Submitted:
03 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
Bovine Respiratory Disease (BRD) remains one of the most consequential health and economic challenges in U.S. beef production, particularly within integrated systems where microbial, environmental, and management factors intersect. This review synthesizes contemporary epidemiological insights, emphasizing BRD’s multifactorial pathogenesis driven by dynamic host-pathogen-environment interactions involving agents such as Mannheimia haemolytica, Pasteurella multocida, and Mycoplasma bovis, alongside stressors from transportation, weaning, and commingling. BRD imposes annual losses exceeding two billion dollars through diminished feed efficiency, reduced carcass yield, increased treatment costs, and mortality. Despite progress in vaccination, biosecurity, and therapeutic interventions, BRD persists due to diagnostic subjectivity and limitations of traditional control measures. The review underscores emerging innovations, including precision livestock technologies, AI-enabled surveillance, and metabolomic biomarkers as transformative tools for early detection and targeted mitigation, while noting barriers related to cost, data harmonization, and scalability. The rising threat of antimicrobial resistance further highlights the need for stewardship frameworks that balance therapeutic effectiveness and public health priorities. Additionally, the paper analyzes policy and economic considerations, arguing for coordinated efforts among producers, veterinarians, researchers, and regulators. BRD is reframed as a systems-level challenge requiring integrated scientific, operational, and regulatory strategies to enhance resilience and sustainability across U.S. beef production.
Keywords:
1. Introduction
2. Epidemiology of BRD
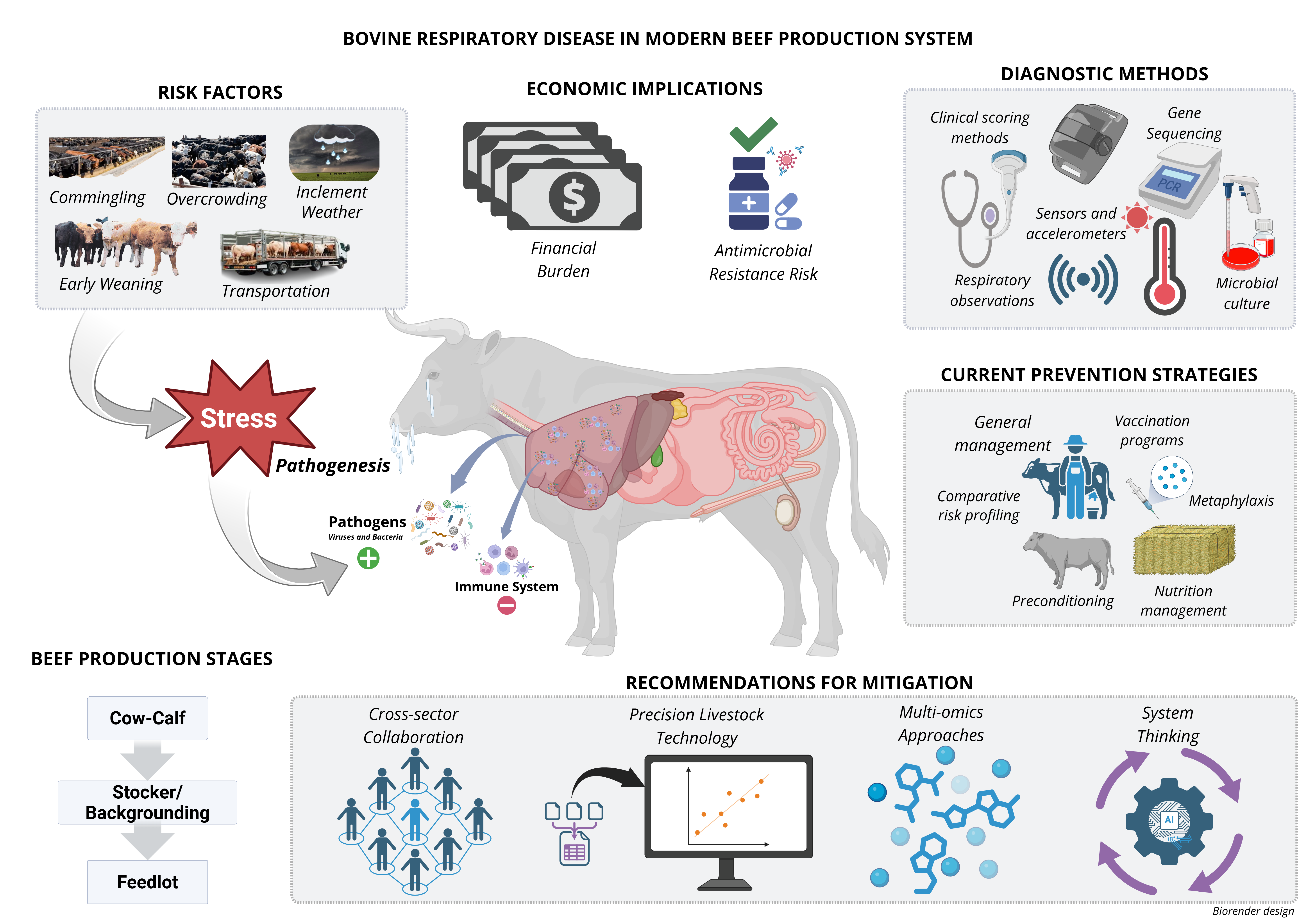
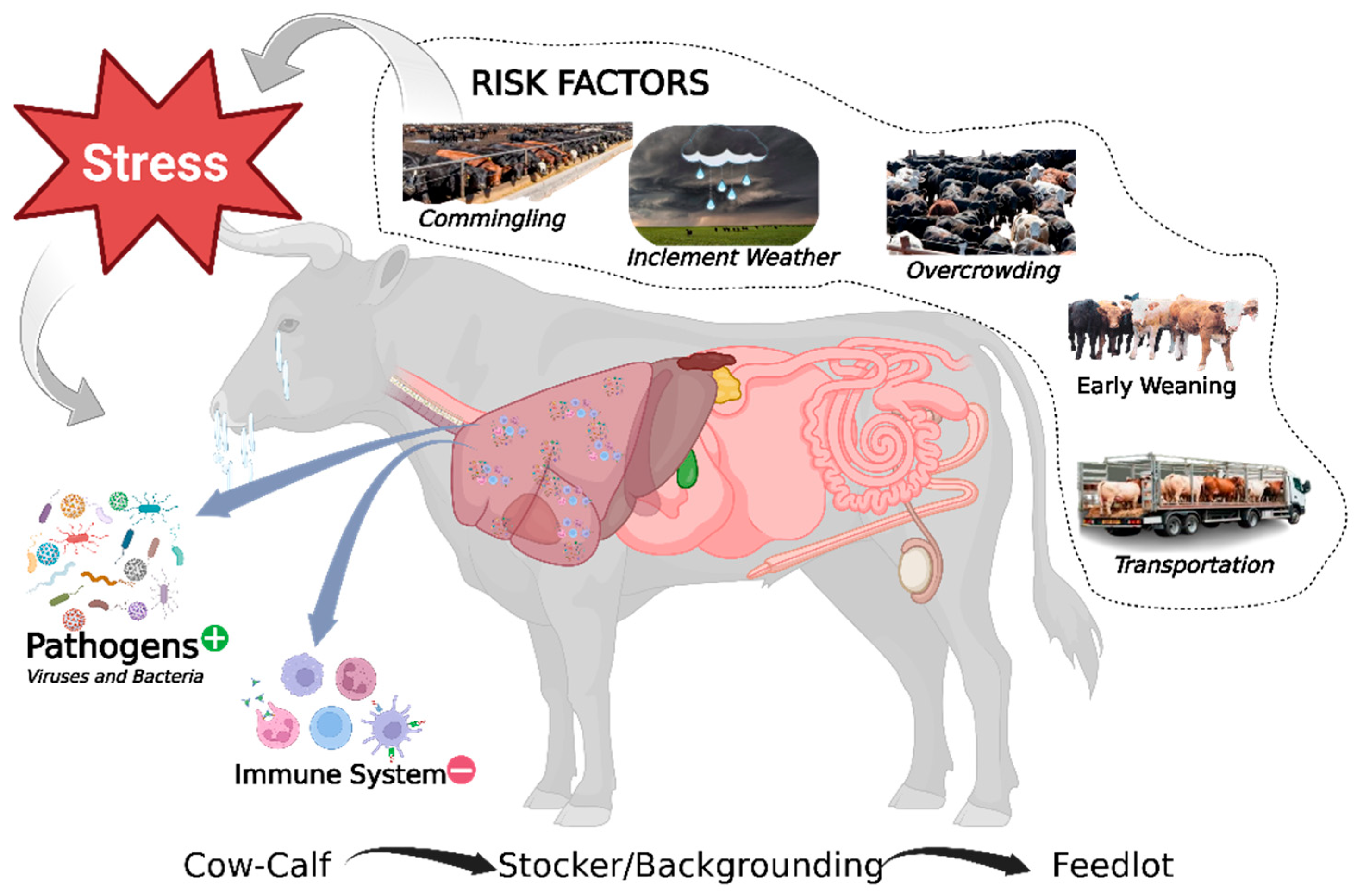
2.1. Etiology, Pathogens, and Risk Factors
2.2. Pathogenesis of BRD
2.3. Clinical Symptoms and Diagnosis
2.4. Herd-Level Dynamics of BRD
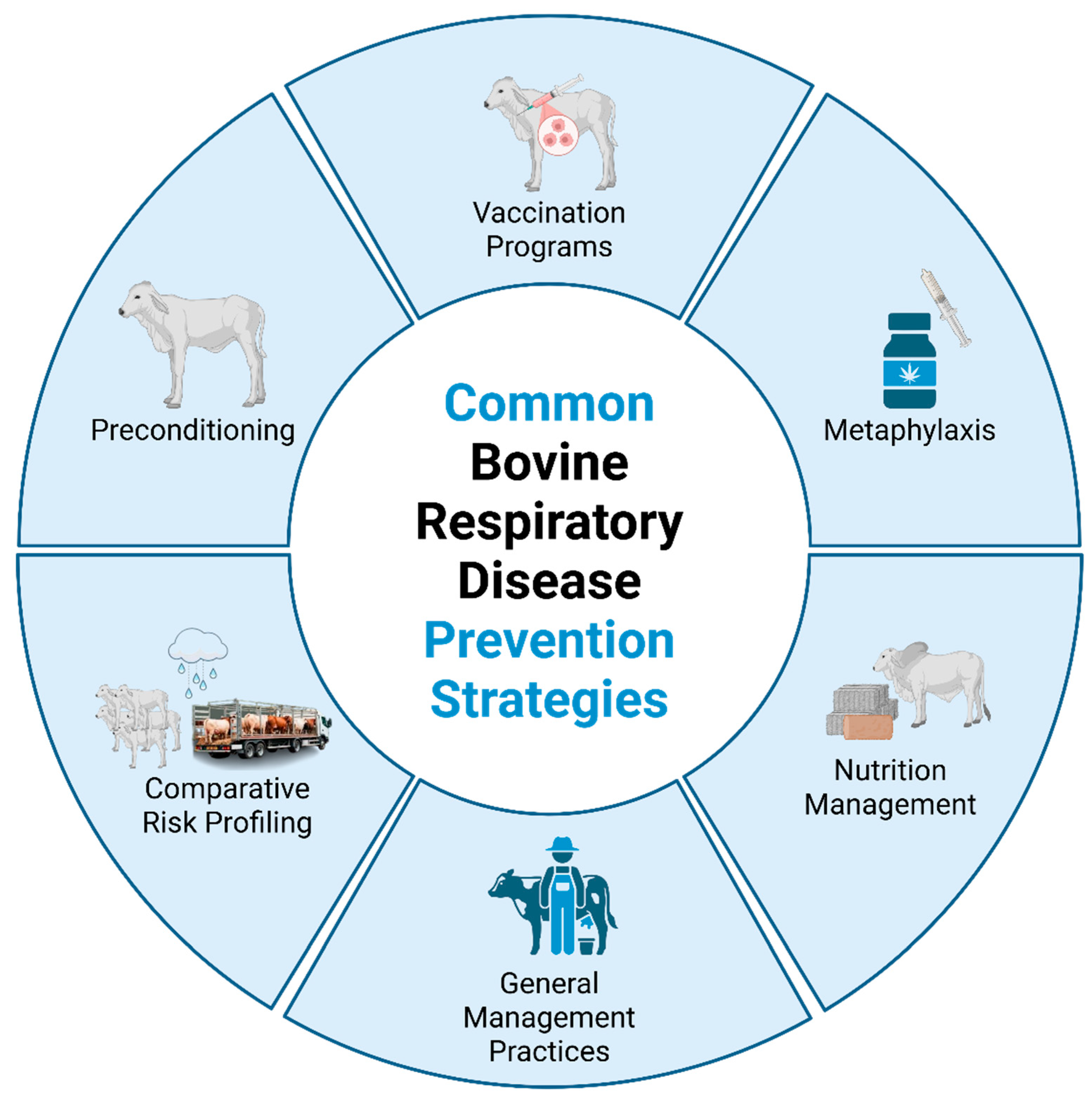
2.5. Current BRD Prevention and Management Strategies
2.5.1. Vaccination Programs
2.5.2. Preconditioning
2.5.3. Metaphylaxis
2.5.4. Comparative Risk Profiling & Segregation of Risk Factors
2.5.5. Nutritional Management
2.5.6. General Management Practices
3. Epidemiological Studies on BRD
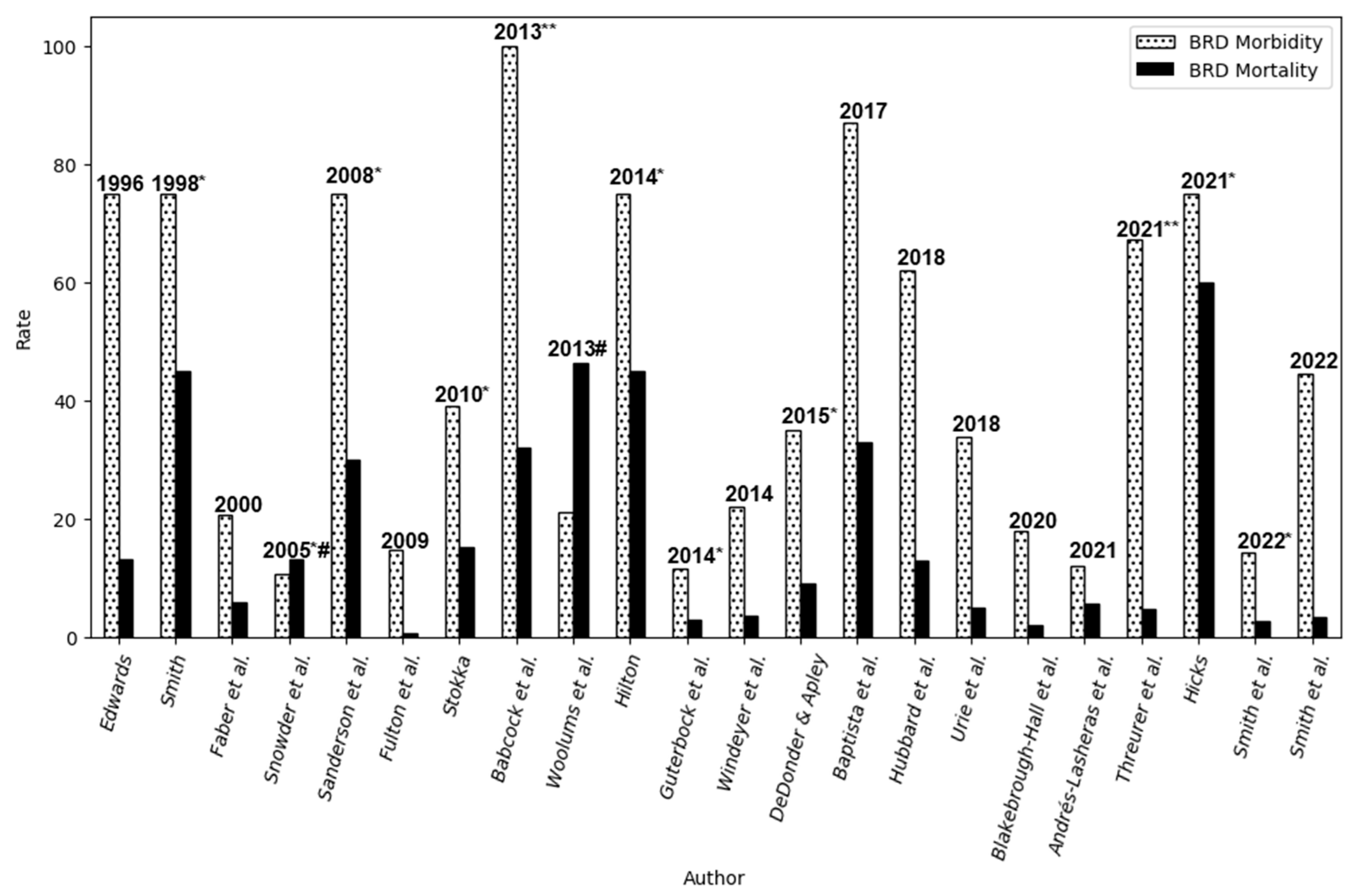
3.1. BRD Incidence, Prevalence, and Epidemiological Patterns
| BRD Morbidity, % | BRD Mortality, % | Year | Author |
|---|---|---|---|
| 75 | 50 | 1996 | [61] |
| 75* | 45* | 1998 | [60] |
| 20.60 | 5.90 | 2000 | [66] |
| 10.52* | 13.1#* | 2005 | [64] |
| 75* | 30* | 2008 | [62] |
| 14.70 | 0.70 | 2009 | [67] |
| 39 | 15.15* | 2010 | [68] |
| 100** | 31.9** | 2013 | [54] |
| 21 | 46.4# | 2013 | [69] |
| 75* | 45* | 2014 | [4] |
| 11.43* | 3 | 2014 | [70] |
| 22 | 3.50 | 2014 | [71] |
| 35* | 9* | 2015 | [72] |
| 87 | 33 | 2017 | [73] |
| 61.90 | 12.90 | 2018 | [13] |
| 33.90 | 5 | 2018 | [74] |
| 18 | 2.10 | 2020 | [21] |
| 12 | 5.60 | 2021 | [75] |
| 67.2** | 4.83** | 2021 | [63] |
| 75 | 60* | 2021 | [65] |
| 14.15* | 2.73* | 2022 | [40] |
| 44.50 | 3.30 | 2022 | [76] |
3.2. Selected Studies and Contributions to the Understanding of BRD Epidemiology
3.2.1. Temporal Disease Patterns and Treatment Outcomes in Feedlot Cattle (Case Study I)
3.2.2. Predictive Modeling for BRD Outcomes in Commercial Feedyards (Case Study II)
3.2.4. Economic Impacts and Performance Consequences of BRD (Case Study III)
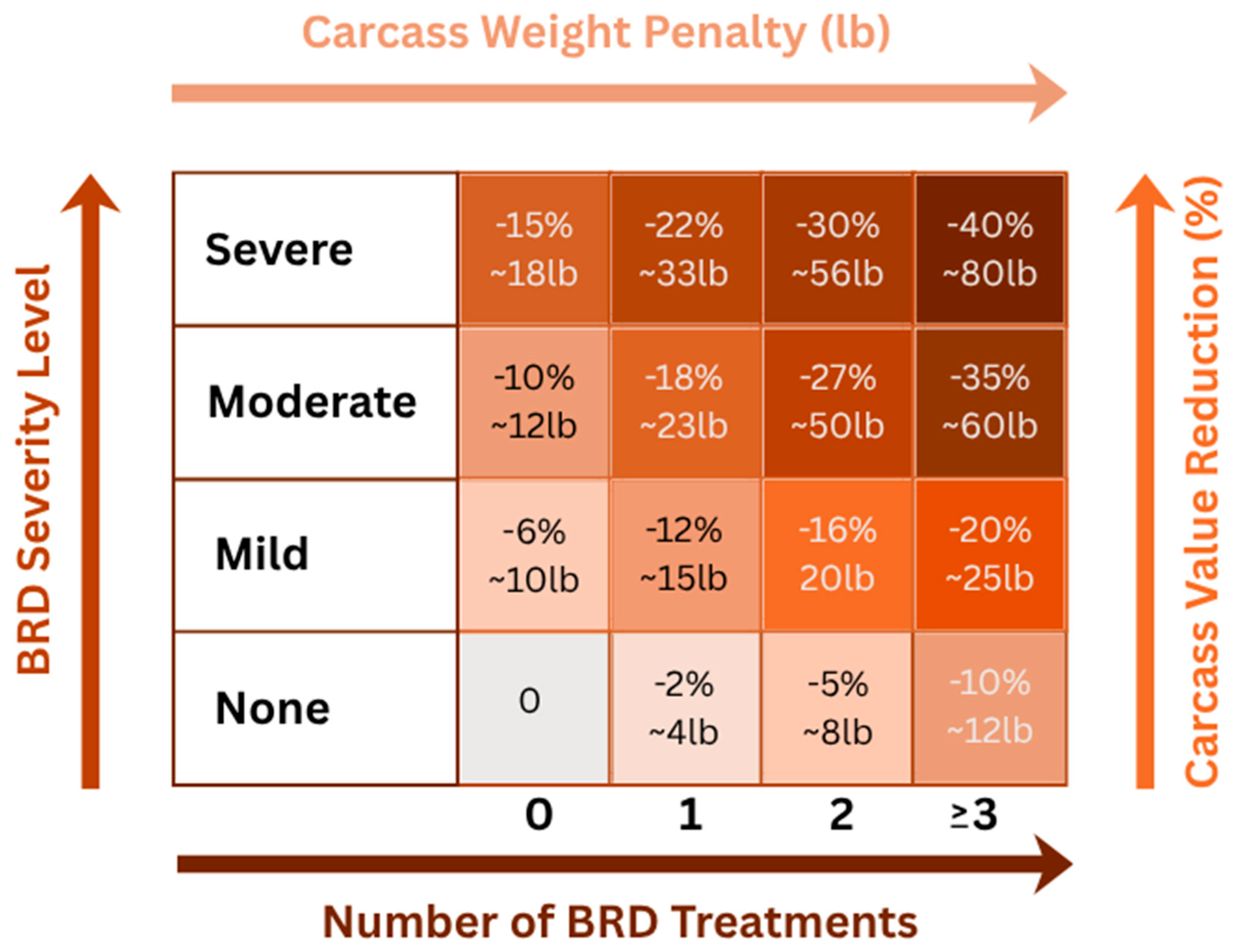
4. Economic Impact of BRD
4.1. Financial Burden and Animal Performance Consequences
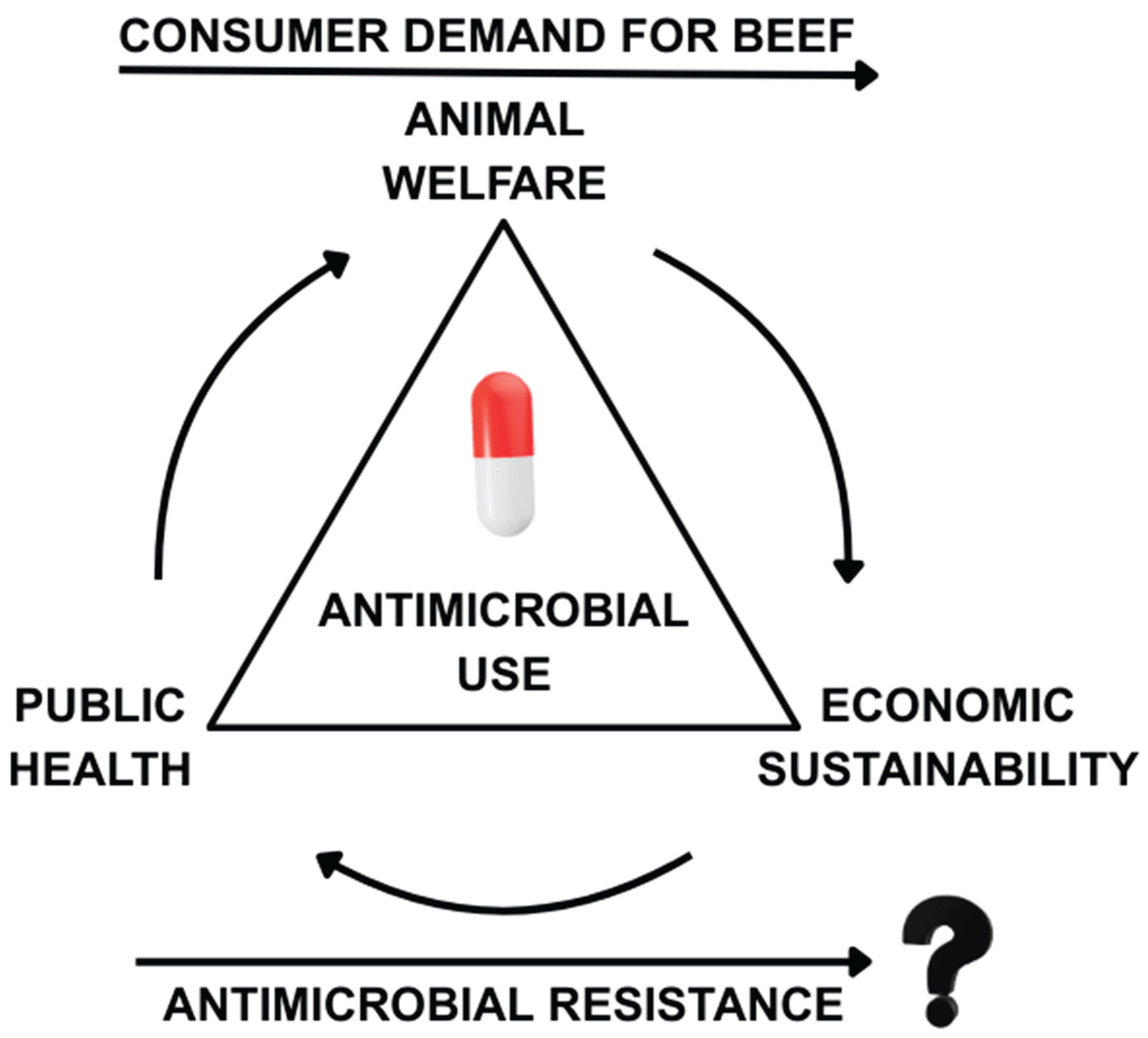
4.2. Economic Ramifications of Antimicrobial Usage and Stewardship in Managing BRD
4.3. Practical Implications for Farmers, Veterinarians, and Policymakers
5. Recommendations
5.1. Areas Requiring Further Exploration and In-Depth Studies
5.2. Suggestions for Refining Preventive Measures and Management Strategies
6. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Baldwin, K.; Williams, B.; Turner, D.; Tsiboe, F.; Raszap Skorbiansky, S.; Sichko, C.; Jones, J.W.; Toossi, S. U.S. Agricultural Policy Review, 2023. 2024 Available online: https://ageconsearch.umn.edu/record/349026 (accessed on Oct 27, 2025).
- Adekunle A.J., Gaines A., Estefano N., Lenin D., Hole P., Khandelwal R., Cooke R., Kaniyamattam K. Mapping Risk Factor Variability and Disease Dynamics of Bovine Respiratory Disease in Beef Production Systems: A Scoping Review. 2025, DOI: In press.
- USDA Feedlot 2011 Part IV Health and health mgt on US feedlot with capacity 1000 above. 2013.
- Hilton, W.M. BRD in 2014: where have we been, where are we now, and where do we want to go? Animal health research reviews 2014, 15, 120–122. [CrossRef]
- Dubrovsky, S.A.; Van Eenennaam, A.L.; Karle, B.M.; Rossitto, P.V.; Lehenbauer, T.W.; Aly, S.S. Epidemiology of bovine respiratory disease (BRD) in preweaned calves on California dairies: The BRD 10K study. Journal of Dairy Science 2019, 102, 7306–7319. [CrossRef]
- Brodersen, B.W. Bovine Respiratory Syncytial Virus. The Veterinary Clinics of North America. Food animal practice 2010, 26, 323–333. [CrossRef]
- Chai, J.; Capik, S.F.; Kegley, B.; Richeson, J.T.; Powell, J.G.; Zhao, J. Bovine respiratory microbiota of feedlot cattle and its association with disease. Veterinary research (Paris) 2022, 53, 4. [CrossRef]
- Edwards, T.A. Control Methods for Bovine Respiratory Disease for Feedlot Cattle. The Veterinary Clinics of North America. Food animal practice 2010, 26, 273–284. [CrossRef]
- Griffin, D.; Chengappa, M.M.; Kuszak, J.; McVey, D.S. Bacterial Pathogens of the Bovine Respiratory Disease Complex. The Veterinary Clinics of North America. Food animal practice 2010, 26, 381–394. [CrossRef]
- Ng, T.F.F.; Kondov, N.O.; Deng, X.; Van Eenennaam, A.; Neibergs, H.L.; Delwart, E. A Metagenomics and Case-Control Study To Identify Viruses Associated with Bovine Respiratory Disease. Journal of Virology 2015, 89, 5340–5349. [CrossRef]
- Smith, R.A.; Step, D.L.; Woolums, A.R. Bovine Respiratory Disease. The Veterinary Clinics of North America. Food animal practice 2020, 36, 239–251. [CrossRef]
- Adekunle, A.J.; Kaniyamattam, K.; Cooke, R.F. 388 Epidemiological risk factor dynamics of Bovine Respiratory Disease in U.S. beef production systems. J Anim Sci 2025, 103, 47–48. [CrossRef]
- Hubbard, K.J.; DVM, A.R.; Woolums, DVM; Karisch, B.B.; Blanton, J.R.; Epperson, W.B.; DVM, D.R.; Smith; Smith, D.R. Case report: Analysis of risk factors and production effects following an outbreak of bovine respiratory disease in Stocker cattle. 2018.
- Rojas, H.A.; White, B.J.; Amrine, D.E.; Larson, R.L. Predicting Bovine Respiratory Disease Risk in Feedlot Cattle in the First 45 Days Post Arrival. Pathogens (Basel) 2022, 11, 442. [CrossRef]
- Ellis, J.A. Update on viral pathogenesis in BRD. Animal Health Research Reviews 2009, 10, 149–153. [CrossRef]
- Muggli-Cockett, N.E.(.S.U., Logan); Cundiff, L.V.; Gregory, K.E. Genetic analysis of bovine respiratory disease in beef calves during the first year of life. Journal of animal science 1992, 70, 2013–2019. [CrossRef]
- Panciera, R.J.; Confer, A.W. Pathogenesis and Pathology of Bovine Pneumonia. The Veterinary Clinics of North America. Food Animal Practice 2010, 26, 191–214. [CrossRef]
- Ferraro, S.; Fecteau, G.; Dubuc, J.; Francoz, D.; Rousseau, M.; Roy, J.; Buczinski, S. Scoping review on clinical definition of bovine respiratory disease complex and related clinical signs in dairy cows. Journal of Dairy Science 2021, 104, 7095–7108. [CrossRef]
- Griffin, D. The monster we don't see: subclinical BRD in beef cattle. Animal health research reviews 2014, 15, 138–141. [CrossRef]
- Buczinski, S.; Pardon, B. Bovine Respiratory Disease Diagnosis. The Veterinary Clinics of North America. Food animal practice 2020, 36, 399–423. [CrossRef]
- Blakebrough-Hall, C.; Dona, A.; D’occhio, M.J.; McMeniman, J.; González, L.A. Diagnosis of Bovine Respiratory Disease in feedlot cattle using blood 1H NMR metabolomics. Scientific Reports 2020, 10, 115. [CrossRef]
- Berman, J.; Francoz, D.; Abdallah, A.; Dufour, S.; Buczinski, S. Evaluation of inter-rater agreement of the clinical signs used to diagnose bovine respiratory disease in individually housed veal calves. Journal of Dairy Science 2021, 104, 12053–12065. [CrossRef]
- Flöck, M. Diagnostic ultrasonography in cattle with thoracic disease. The Veterinary Journal 2004, 167, 272–280. [CrossRef]
- Fowler, J.; Stieger-Vanegas, S.M.; Vanegas, J.A.; Bobe, G.; Poulsen, K.P. Comparison of Thoracic Radiography and Computed Tomography in Calves with Naturally Occurring Respiratory Disease. Front Vet Sci 2017, 4. [CrossRef]
- Galland, J.C.; House, J.K.; Hyatt, D.R.; Hawkins, L.L.; Anderson, N.V.; Irwin, C.K.; Smith, B.P. Prevalence of Salmonella in beef feeder steers as determined by bacterial culture and ELISA serology. Veterinary Microbiology 2000, 76, 143–151. [CrossRef]
- Goto, Y.; Fukunari, K.; Suzuki, T. Multiplex RT-qPCR Application in Early Detection of Bovine Respiratory Disease in Healthy Calves. Viruses 2023, 15, 669. [CrossRef]
- Conrad, C.C.; Daher, R.K.; Stanford, K.; Amoako, K.K.; Boissinot, M.; Bergeron, M.G.; Alexander, T.; Cook, S.; Ralston, B.; Zaheer, R.; Niu, Y.D.; McAllister, T. A Sensitive and Accurate Recombinase Polymerase Amplification Assay for Detection of the Primary Bacterial Pathogens Causing Bovine Respiratory Disease. Front Vet Sci 2020, 7. [CrossRef]
- Jiminez, J.; Timsit, E.; Orsel, K.; van der Meer, F.; Guan, L.L.; Plastow, G. Whole-Blood Transcriptome Analysis of Feedlot Cattle With and Without Bovine Respiratory Disease. Front Genet 2021, 12. [CrossRef]
- Fox, J.T.; Spire, M.F. Near infrared spectroscopy as a potential method to detect bovine respiratory disease. Kansas Agricultural Experiment Station Research Reports 2004, 97–99. [CrossRef]
- Schaefer, A.L.; Cook, N.J.; Church, J.S.; Basarab, J.; Perry, B.; Miller, C.; Tong, A.K.W. The use of infrared thermography as an early indicator of bovine respiratory disease complex in calves. Research in Veterinary Science 2007, 83, 376–384. [CrossRef]
- Cantor, M.C.; Casella, E.; Silvestri, S.; Renaud, D.L.; Costa, J.H.C. Using Machine Learning and Behavioral Patterns Observed by Automated Feeders and Accelerometers for the Early Indication of Clinical Bovine Respiratory Disease Status in Preweaned Dairy Calves. Front Anim Sci 2022, 3. [CrossRef]
- Carpentier, L.; Berckmans, D.; Youssef, A.; Berckmans, D.; van Waterschoot, T.; Johnston, D.; Ferguson, N.; Earley, B.; Fontana, I.; Tullo, E.; Guarino, M.; Vranken, E.; Norton, T. Automatic cough detection for bovine respiratory disease in a calf house. Biosystems Engineering 2018, 173, 45–56. [CrossRef]
- Haddadi, S.; Koziel, J.A.; Engelken, T.J. Analytical approaches for detection of breath VOC biomarkers of cattle diseases -A review. Analytica Chimica Acta 2022, 1206, 339565. [CrossRef]
- Gao, Y.; Liu, M. Application of machine learning based genome sequence analysis in pathogen identification. Front Microbiol 2024, 15. [CrossRef]
- Adekunle, A.J.; Rahimifar, A.; Kaniyamattam, K. 48. An agent-based modeling framework for bovine respiratory disease management in integrated beef supply chains. Animal - Science proceedings 2025, 16, 579–581. [CrossRef]
- Kniffen, E.; Adekunle, A.; Agnoor, P.; Akpenyi, O.; Bose, S. Stochastic Modeling of Bovine Respiratory Disease Using Agent-Based Modeling, AMS: 2026/01/06.
- Sanguinetti, V. Disease control practices used to prevent morbidity and mortality in preweaned beef calves. UCalgary 2025 Available online: https://chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://ucalgary.scholaris.ca/server/api/core/bitstreams/5be2208e-1b17-4c09-88da-21521c95a017/content.
- Antonopoulos, A.; Ciria, N.; Regan, Á; Tubay, J.; Ciaravino, G.; Hayes, B.; Lambert, S.; Vergne, T.; Velkers, F.; Biebaut, E.; Viltrop, A.; Dewulf, J.; Charlier, J.; Fischer, E.; Palau, A.A. PARAMETRA: A transmission modelling database for livestock diseases. Preventive Veterinary Medicine 2025, 245, 106668. [CrossRef]
- Computational and analytical approaches towards epidemic spread containment of temporal animal trade networks. (Accessed on Oct 27, 2025).
- Smith, K.J.; Amrine, D.E.; Larson, R.L.; Theurer, M.E.; White, B.J. Determining relevant risk factors associated with mid- and late-feeding-stage bovine respiratory disease morbidity in cohorts of beef feedlot cattle. Applied Animal Science 2022, 38, 373–379. [CrossRef]
- Biesheuvel, M.M.; Ward, C.; Penterman, P.; van Engelen, E.; van Schaik, G.; Deardon, R.; Barkema, H.W. Within-herd transmission of Mycoplasma bovis infections after initial detection in dairy cows. J Dairy Sci 2024, 107, 516–529. [CrossRef]
- Isoda, N.; Sekiguchi, S.; Ryu, C.; Notsu, K.; Kobayashi, M.; Hamaguchi, K.; Hiono, T.; Ushitani, Y.; Sakoda, Y. Serosurvey of Bovine Viral Diarrhea Virus in Cattle in Southern Japan and Estimation of Its Transmissibility by Transient Infection in Nonvaccinated Cattle. Viruses 2025, 17, 61. [CrossRef]
- Rossi, G.; Smith, R.L.; Pongolini, S.; Bolzoni, L. Modelling farm-to-farm disease transmission through personnel movements: from visits to contacts, and back. Sci Rep 2017, 7, 2375. [CrossRef]
- Hill, E.M.; Prosser, N.S.; Ferguson, E.; Kaler, J.; Green, M.J.; Keeling, M.J.; Tildesley, M.J. Modelling livestock infectious disease control policy under differing social perspectives on vaccination behaviour. PLOS Computational Biology 2022, 18, e1010235. [CrossRef]
- Chumachenko, D. A theoretical framework for agent-based modelling of infectious disease dynamics under misinformation and vaccine hesitancy. Radioelectronic and Computer Systems 2025, 2025, 6–28. [CrossRef]
- Kamel, M.S.; Davidson, J.L.; Verma, M.S. Strategies for Bovine Respiratory Disease (BRD) Diagnosis and Prognosis: A Comprehensive Overview. Animals 2024, 14, 627. [CrossRef]
- Gorden, P.J.; Plummer, P. Control, Management, and Prevention of Bovine Respiratory Disease in Dairy Calves and Cows. The Veterinary Clinics of North America. Food Animal Practice 2010, 26, 243–259. [CrossRef]
- Lalman, D.; Ward, C.E. Effects of Preconditioning on Health, Performance and Prices of Weaned Calves. American Association of Bovine Practitioners. Conference. Proceedings of the ... Annual Conference 2005, 44–50. [CrossRef]
- Taylor, J.D.; Fulton, R.W.; Lehenbauer, T.W.; Step, D.L.; Confer, A.W. The epidemiology of bovine respiratory disease: what is the evidence for preventive measures? Canadian Veterinary Journal 2010, 51, 1351–1359. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21358927.
- Schunicht, O.C. Preconditioning beef calves is an economic no-brainer. American Association of Bovine Practitioners. Conference. Proceedings of the ... Annual Conference 2017, 59–62. [CrossRef]
- Hodder, A., Impact on feeding behavior of beef calves preconditioning on ranch and commingling. Masters, University of Calgary, 2022.
- Nickell, J.S.; White, B.J. Metaphylactic Antimicrobial Therapy for Bovine Respiratory Disease in Stocker and Feedlot Cattle. The Veterinary Clinics of North America. Food animal practice 2010, 26, 285–301. [CrossRef]
- Maier, G.U.; Love, W.J.; Karle, B.M.; Dubrovsky, S.A.; Williams, D.R.; Champagne, J.D.; Anderson, R.J.; Rowe, J.D.; Lehenbauer, T.W.; Van Eenennaam, A.L.; Aly, S.S. A novel risk assessment tool for bovine respiratory disease in preweaned dairy calves. Journal of Dairy Science 2020, 103, 9301–9317. [CrossRef]
- Babcock, A.H.; Cernicchiaro, N.; White, B.J.; Dubnicka, S.R.; Thomson, D.U.; Ives, S.E.; Scott, H.M.; Milliken, G.A.; Renter, D.G. A multivariable assessment quantifying effects of cohort-level factors associated with combined mortality and culling risk in cohorts of U.S. commercial feedlot cattle. Preventive veterinary medicine 2013, 108, 38–46. [CrossRef]
- Lehtoranta, L.; Latvala, S.; Lehtinen, M.J. Role of Probiotics in Stimulating the Immune System in Viral Respiratory Tract Infections: A Narrative Review. Nutrients 2020, 12, 3163. [CrossRef]
- Palomares, R.A. Trace Minerals Supplementation with Great Impact on Beef Cattle Immunity and Health. Animals 2022, 12, 2839. [CrossRef]
- Markey, J., Evaluation of NutraGen®, an Immunomodulatory Feed Additive, in Calves Subjected to a Combination Viral and Bacterial Bovine Respiratory Disease Challenge - ProQuest. PhD, Oklahoma State University, 2024.
- Voland, L.; Ortiz-Chura, A.; Tournayre, J.; Martin, B.; Bouchon, M.; Nicolao, A.; Pomiès, D.; Morgavi, D.P.; Popova, M. Duration of dam contact had a long effect on calf rumen microbiota without affecting growth. Front Vet Sci 2025, 12. [CrossRef]
- Sweiger, S.H.; Nichols, M.D. Control Methods for Bovine Respiratory Disease in Stocker Cattle. The Veterinary Clinics of North America. Food animal practice 2010, 26, 261–271. [CrossRef]
- Smith, R.A. Impact of disease on feedlot performance: a review. Journal of Animal Science 1998, 76, 272. [CrossRef]
- Edwards, A. Respiratory diseases of feedlot cattle in central USA. The Bovine Practitioner 1996, 5–7. [CrossRef]
- Sanderson, M.W.; Wagner, B.A.; Dargatz, D.A. Risk factors for initial respiratory disease in the United States' feedlots based on producer-collected daily morbidity counts. 2008.
- Theurer, M.E.; Johnson, M.D.; Fox, T.; McCarty, T.M.; McCollum, R.M.; Jones, T.M.; Alkire, D.O. Bovine respiratory disease during the mid-portion of the feeding period: Observations of frequency, timing, and population from the field. Applied Animal Science 2021, 37, 52–58. [CrossRef]
- Snowder, G.; Van Vleck, L. D.,; Cundiff, L. V.,; Bennett, G. L. Bovine respiratory disease in feedlot cattle environmental, genetic, and economic factors. American Society of Animal Science 2005, 84, 1999–2008. [CrossRef]
- Hicks, B. BEEF CATTLE RESEARCH UPDATE. 2021.
- Faber, R.; Hartwig, N.; Busby, D.; BreDahl, R. The Costs and Predictive Factors of Bovine Respiratory Disease in Standardized Steer Tests. Iowa State University Animal Industry Report 2000, 1. Available online: https://www.iastatedigitalpress.com/air/article/id/7342/ (accessed on Oct 27, 2025).
- Taylor, J.D.; Fulton, R.W.; Lehenbauer, T.W.; Step, D.L.; Confer, A.W. The epidemiology of bovine respiratory disease: What is the evidence for predisposing factors? Public Health 2010, 51.
- Stokka, G.L. Prevention of Respiratory Disease in Cow/Calf Operations. The Veterinary Clinics of North America. Food Animal Practice 2010, 26, 229–241. [CrossRef]
- Woolums, A.R.; Berghaus, R.D.; Smith, D.R.; White, B.J.; Engelken, T.J.; Irsik, M.B.; Matlick, D.K.; Jones, A.L.; Ellis, R.W.; Smith, I.J.; Mason, G.L.; Waggoner, E.R. Producer survey of herd-level risk factors for nursing beef calf respiratory disease. J Am Vet Med Assoc 2013, 243, 538–547. [CrossRef]
- Guterbock, W.M. The impact of BRD: the current dairy experience. Anim Health Res Rev 2014, 15, 130–134. [CrossRef]
- Windeyer, M.C.; Leslie, K.E.; Godden, S.M.; Hodgins, D.C.; Lissemore, K.D.; LeBlanc, S.J. Factors associated with morbidity, mortality, and growth of dairy heifer calves up to 3 months of age. Preventive Veterinary Medicine 2014, 113, 231–240. [CrossRef]
- DeDonder, K.D.; Apley, M.D. A review of the expected effects of antimicrobials in bovine respiratory disease treatment and control using outcomes from published randomized clinical trials with negative controls. Vet Clin North Am Food Anim Pract 2015, 31, 97–111, vi. [CrossRef]
- Baptista, A.L.; Rezende, A.L.; Fonseca, P.d.A.; Massi, R.P.; Nogueira, G.M.; Magalhães, L.Q.; Headley, S.A.; Menezes, G.L.; Alfieri, A.A.; Saut, J.P.E. Bovine respiratory disease complex associated mortality and morbidity rates in feedlot cattle from southeastern Brazil. J Infect Dev Ctries 2017, 11, 791–799. [CrossRef]
- Urie, N.J.; Lombard, J.E.; Shivley, C.B.; Kopral, C.A.; Adams, A.E.; Earleywine, T.J.; Olson, J.D.; Garry, F.B. Preweaned heifer management on US dairy operations: Part V. Factors associated with morbidity and mortality in preweaned dairy heifer calves. Journal of Dairy Science 2018, 101, 9229–9244. [CrossRef]
- Andrés-Lasheras, S.; Ha, R.; Zaheer, R.; Lee, C.; Booker, C.W.; Dorin, C.; Van Donkersgoed, J.; Deardon, R.; Gow, S.; Hannon, S.J.; Hendrick, S.; Anholt, M.; McAllister, T.A. Prevalence and risk factors associated with antimicrobial resistance in bacteria related to bovine respiratory disease—a broad cross-sectional study of beef cattle at entry into Canadian feedlots. Frontiers in Veterinary Science 2021, 8, 692646. [CrossRef]
- Smith, K.J.; Amrine, D.E.; Larson, R.L.; Theurer, M.E.; Szaz, J.I.; Bryant, T.; White, B.J. Risk factors for mid- and late-feeding-stage bovine respiratory morbidity and mortality based on individual animal treatments of beef feedlot cattle. Applied Animal Science 2022, 38, 360–372. [CrossRef]
- Smith, D.R.; Wills, R.W.; Woodruff, K.A. Epidemiology’s Adoption of System Dynamics is a Natural Extension of Population Thinking. Veterinary Clinics of North America: Food Animal Practice 2022, 38, 245–259. [CrossRef]
- Windeyer, M.C.; Timsit, E.; Barkema, H. Bovine respiratory disease in pre-weaned dairy calves: Are current preventative strategies good enough? Vet J 2017, 224, 16–17. [CrossRef]
- Woolums, A.R.; Karisch, B.B.; Frye, J.G.; Epperson, W.; Smith, D.R.; Blanton, J.; Austin, F.; Kaplan, R.; Hiott, L.; Woodley, T.; Gupta, S.K.; Jackson, C.R.; McClelland, M. Multidrug-resistant Mannheimia haemolytica isolated from high-risk beef stocker cattle after antimicrobial metaphylaxis and treatment for bovine respiratory disease. Veterinary Microbiology 2018, 221, 143–152. [CrossRef]
- Smith, K.J.; White, B.J.; Amrine, D.E.; Larson, R.L.; Theurer, M.E.; Szasz, J.I.; Bryant, T.C.; Waggoner, J.W. Evaluation of First Treatment Timing, Fatal Disease Onset, and Days from First Treatment to Death Associated with Bovine Respiratory Disease in Feedlot Cattle. Veterinary Sciences 2023, 10. [CrossRef]
- Heinen, L.; White, B.J.; Amrine, D.E.; Larson, R.L. Evaluation of predictive models to determine final outcome for feedlot cattle based on information available at first treatment for bovine respiratory disease. American journal of veterinary research 2023, 84, 1–8. [CrossRef]
- Blakebrough-Hall; McMeniman, J.P.; González, L.A. An evaluation of the economic effects of bovine respiratory disease on animal performance, carcass traits, and economic outcomes in feedlot cattle defined using four BRD diagnosis methods. Journal of animal science 2020, 98, 1–11. [CrossRef]
- White, B.J.; Larson, B.L. Impact of bovine respiratory disease in U.S. beef cattle. Animal health research reviews 2020, 21, 132–134. [CrossRef]
- Watts, J.L.; Sweeney, M.T. Antimicrobial Resistance in Bovine Respiratory Disease Pathogens: Measures, Trends, and Impact on Efficacy. The Veterinary Clinics of North America. Food animal practice 2010, 26, 79–88. [CrossRef]
- Dubrovsky, S.A.; Van Eenennaam, A.L.; Aly, S.S.; Karle, B.M.; Rossitto, P.V.; Overton, M.W.; Lehenbauer, T.W.; Fadel, J.G. Preweaning cost of bovine respiratory disease (BRD) and cost-benefit of implementation of preventative measures in calves on California dairies: The BRD 10K study. Journal of Dairy Science 2020, 103, 1583–1597. [CrossRef]
- Overton, M.W. Economics of respiratory disease in dairy replacement heifers. Animal Health Research Reviews 2020, 21, 143–148. [CrossRef]
- Schneider, M.J.; Tait, R.G.; Busby, W.D.; Reecy, J.M. An evaluation of bovine respiratory disease complex in feedlot cattle: Impact on performance and carcass traits using treatment records and lung lesion scores. J Anim Sci 2009, 87, 1821–1827. [CrossRef]
- Brooks, K.R.; Raper, K.C.; Ward, C.E.; Holland, B.P.; Krehbiel, C.R.; Step, D.L. Economic effects of bovine respiratory disease on feedlot cattle during backgrounding and finishing phases1. The Professional Animal Scientist 2011, 27, 195–203. [CrossRef]
- Wilson, B.K.; Step, D.L.; Maxwell, C.L.; Gifford, C.A.; Richards, C.J.; Krehbiel, C.R. Effect of bovine respiratory disease during the receiving period on steer finishing performance, efficiency, carcass characteristics, and lung scores. Prof Anim Sci 2017, 33, 24–36. [CrossRef]
- Roeber, D.L.; Speer, N.C.; Gentry, J.G.; Tatum, J.D.; Smith, C.D.; Whittier, J.C.; Jones, G.F.; Belk, K.E.; Smith, G.C. Feeder Cattle Health Management: Effects on Morbidity Rates, Feedlot Performance, Carcass Characteristics, and Beef Palatability12. The Professional Animal Scientist 2001, 17, 39–44. [CrossRef]
- Heinen, L.; White, B.J.; Larson, R.L.; Kopp, D.; Pendell, D.L. Economic impact of mortality prediction by predictive model at first and second treatment for bovine respiratory disease. American Journal of Veterinary Research 2024, 85. [CrossRef]
- De Koster, J.; Tena, J.; Stegemann, M.R. Treatment of bovine respiratory disease with a single administration of tulathromycin and ketoprofen. Veterinary record 2022, 190, no. [CrossRef]
- Magouras, I.; Dürr, S.; Martínez-López, B.; Brookes, V.J. Editorial: Insights in veterinary epidemiology and economics: 2023. Front Vet Sci 2025, 12. [CrossRef]
- Soares Filipe, J.F.; Ciliberti, M.G.; Attard, G.; Ratti, G.; Rocchi, M.S.L. Editorial: Biosecurity of infectious diseases in veterinary medicine. Front Vet Sci 2025, 12, 1665683. [CrossRef]
- Cazer, C.L.; Basran, P.; Ivanek-Miojevic, R. From bark to bytes: artificial intelligence transforming veterinary medicine. Am J Vet Res 2025, 86, S4–S5. [CrossRef]
- Lhermie, G.; Sauvage, P.; Tauer, L.W.; Chiu, L.V.; Kanyiamattam, K.; Ferchiou, A.; Raboisson, D.; Scott, H.M.; Smith, D.R.; Grohn, Y.T. Economic effects of policy options restricting antimicrobial use for high-risk cattle placed in U.S. feedlots. PloS one 2020, 15, e0239135. [CrossRef]
- Kaniyamattam, K.; Tauer, L.W.; Gröhn, Y.T. System Economic Costs of Antibiotic Use Elimination in the US Beef Supply Chain. Frontiers in Veterinary Science 2021, 8, 606810. [CrossRef]
- Patel, S.J.; Wellington, M.; Shah, R.M.; Ferreira, M.J. Antibiotic Stewardship in Food-producing Animals: Challenges, Progress, and Opportunities. Clinical therapeutics 2020, 42, 1649–1658. [CrossRef]
- Adekunle, A.J.; Kaniyamattam, K. Feedlot economic visualization decision support tool for sustainable beef production. J Anim Sci 2025, 103, 246–247. [CrossRef]
- Abell, K.M.; Theurer, M.E.; Larson, R.L.; White, B.J.; Apley, M. A mixed treatment comparison meta-analysis of metaphylaxis treatments for bovine respiratory disease in beef cattle. J Anim Sci 2017, 95, 626–635. [CrossRef]
- Centeno-Martinez, R.E.; Klopp, R.N.; Koziol, J.; Boerman, J.P.; Johnson, T.A. Dynamics of the nasopharyngeal microbiome of apparently healthy calves and those with clinical symptoms of bovine respiratory disease from disease diagnosis to recovery. Front Vet Sci 2023, 10. [CrossRef]
- Munir, S.; Ashfaq, U.A.; Qasim, M.; Fatima, T.; Aslam, S.; Sarfraz, M.H.; Kober, A.K.M.H.; Khurshid, M. Chapter 4 - Molecular omics: a promising systems biology approach to unravel host-pathogen interactions. In Systems Biology Approaches for Host-Pathogen Interaction Analysis; Ashraf, M.T.; Khan, A.A.; Aldakheel, F.M., Eds.; Academic Press: 2024; pp. 81–102.
- Kaniyamattam, K.; Adekunle, A.; Vettil, V.K.; Rejimon, S.P.; Pi, Y.; Tao, J.; Mendes, E.D.M.; Tedeschi, L.O. 400 Design and development of artificial intelligence-driven decision support systems for sustainable livestock systems in the United States. J Anim Sci 2025, 103, 109–110. [CrossRef]
- Puig, A.; Ruiz, M.; Bassols, M.; Fraile, L.; Armengol, R. Technological Tools for the Early Detection of Bovine Respiratory Disease in Farms. Animals (Basel) 2022, 12, 2623. [CrossRef]
- Theurer, M.E.; White, B.J.; Renter, D.G. Optimizing Feedlot Diagnostic Testing Strategies Using Test Characteristics, Disease Prevalence, and Relative Costs of Misdiagnosis. Veterinary Clinics of North America: Food Animal Practice 2015, 31, 483–493. [CrossRef]
- Vysyaraju, U.S.R.; Moon, S.; Mendes, E.D.M.; Adekunle, A.J.; Kaniyamattam, K.; Pi, Y. 47. Activity recognition in beef cattle Calan gate using CLIP and YOLO v11. Animal - Science proceedings 2025, 16, 578–579. [CrossRef]
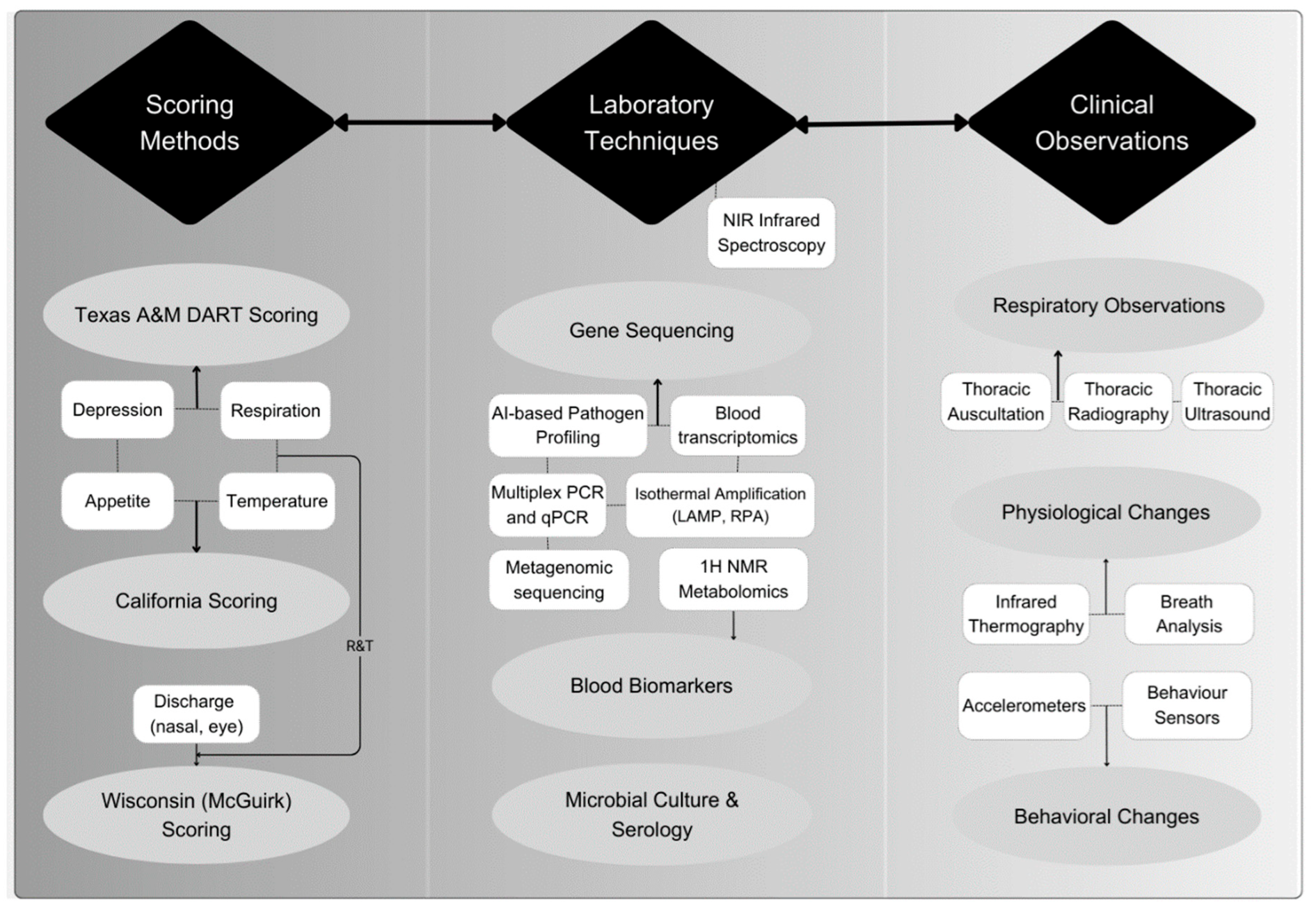
| Method | Principle/Target | Advantages | Limitations | Key References |
| DART Scoring | Subjective clinical signs: depression, appetite, respiration, temperature | Low-cost; easy to implement quickly in the field | Low sensitivity/specificity (<70%); high inter-observer variability. | [19,20,22] |
| Thoracic Auscultation | Detects abnormal lung sounds via a stethoscope | Simple, immediate insights | Requires expertise; limited sensitivity, especially for lower lung lesions | [20] |
| Thoracic Ultrasound (TUS) | Visualizes lung consolidations and pleural lesions | High sensitivity (~85%); early lesion detection | Equipment cost, operator dependency, and animal handling are needed | [23] |
| Thoracic Radiography | X-ray imaging of lung structures | Can detect deep parenchymal lesions | Superimposition issues require anesthesia or restraint | [24] |
| Bacterial Culture & Serology | Identification of pathogen presence and immune response | Gold standard for pathogen ID; supports antimicrobial sensitivity testing | Takes days; may miss fastidious pathogens; serology may lack specificity | [25] |
| Multiplex PCR and qPCR | Detects DNA/RNA of multiple pathogens (viral & bacterial) | High sensitivity/specificity; can detect co-infections; quantitative | Infrastructure and cost barriers; positives may reflect colonization, not disease | [26] |
| Isothermal Amplification (LAMP, RPA) | Rapid on-site detection of specific pathogens | Field-friendly; colorimetric readout; ~1 hr results | Requires primer design; possible false positives; early-stage tool | [27] |
| ¹H NMR Metabolomics | Identifies systemic metabolic biomarkers in blood | High accuracy (~85%); reveals host response patterns | High cost; requires lab and ML models; less field-ready | [21] |
| Blood Transcriptomics (DEGs) | Uses gene-expression profiling to detect immune response | Early detection potential; highly specific insights | Requires RNA sequencing; high cost; technical demands | [28] |
| Near-Infrared Spectroscopy (NIRS) | Measures biochemical fingerprint in fluids/tissues | Non-invasive; potential rapid testing without a blood draw | Early research stage; needs standardization | [29] |
| Infrared Thermography | Measures surface temperature (eye/orbit) | Non-contact; early fever detection | Variable with ambient conditions; needs calibration | [30] |
| Accelerometers and Behavior Sensors | Detect changes in activity and rumination | Can detect illness ~3 days before clinical signs | Initial cost, data noise; requires algorithms | [31] |
| Acoustic Monitoring (Cough sensors) | Detects cough frequency in pens | Automated, non-invasive; early outbreak detection | Background noise interference | [32] |
| Breath Analysis (e.g., NO, VOCs) | Measures volatile biomarkers linked to inflammation | Non-invasive; rapid results | Technical complexity; needs calibration | [33] |
| Metagenomic Sequencing & AI-based Pathogen Profiling | Deep sequencing with ML models for pathogen signature | Potential for high-throughput, sensitive detection | High cost; bioinformatics demands; emerging technology | [34] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




