Case Report
In clinical practice, uterine leiomyosarcoma is relatively rare but is known to have a poor prognosis. Few reports have described uterine leiomyosarcomas containing ectopic components. Therefore, the malignancy and clinical impact of uterine leiomyosarcomas containing ectopic components remain unknown. Ovarian tumor rupture is relatively common in clinical practice, whereas uterine tumor rupture is rare. In this case, a patient developed abdominal pain after an endoscopic examination of the lower gastrointestinal tract and was diagnosed with ruptured uterine leiomyosarcoma containing ectopic components. Here, we report this case along with a review of the literature.
Patient: 56-year-old female
Height: 156 cm, Weight: 54 kg
Medical history: Total hip replacement for congenital hip dislocation at age 53
Family history: Father with subarachnoid hemorrhage; mother with gastric cancer
Menstrual history: Menarche at age 13; menopause at age 55
Pregnancy/delivery history: None
Chief pregnancy complaint: Abdominal pain
Medications: None
History of Present Illness
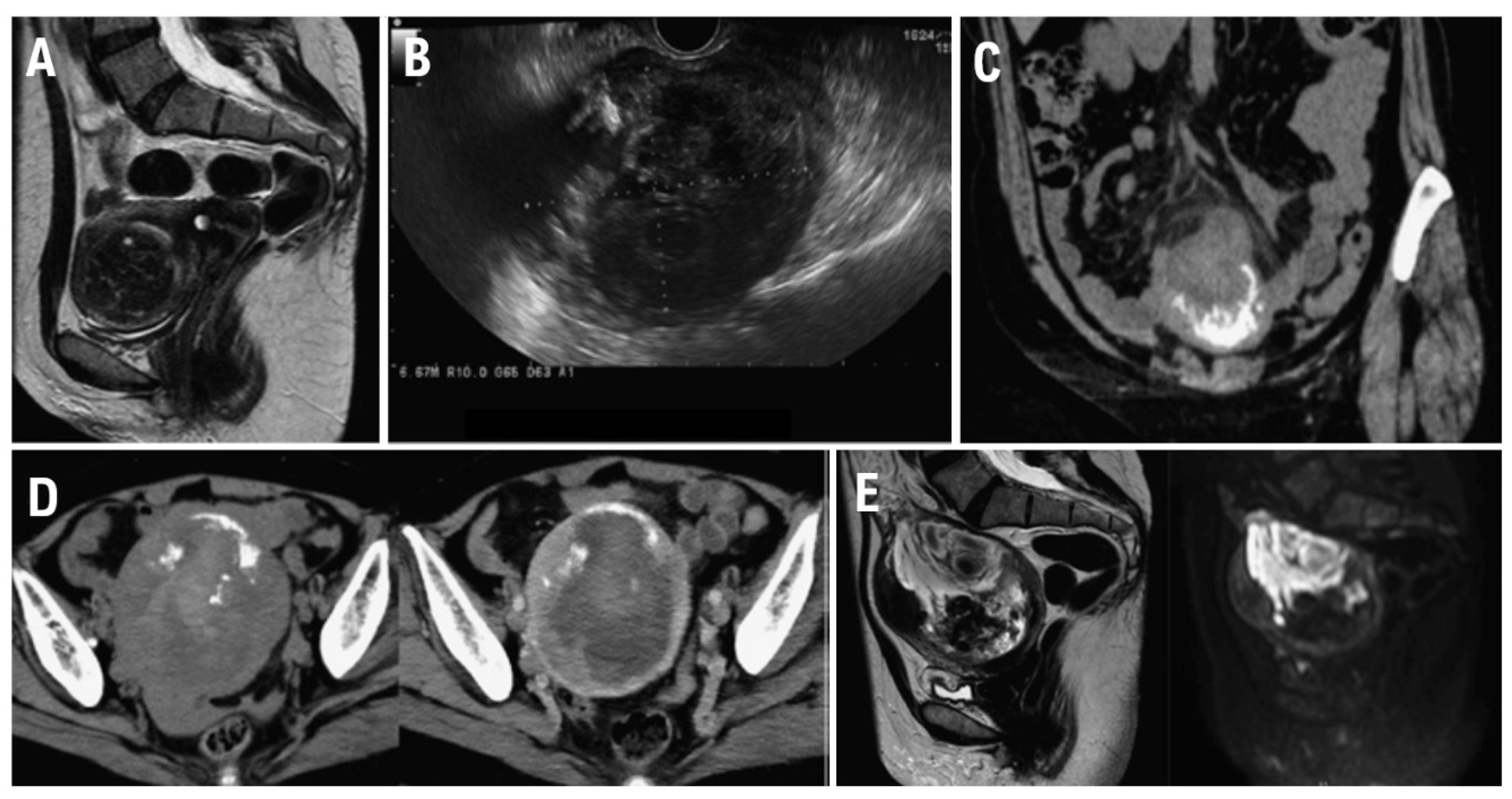
At 48 years of age, the patient was diagnosed with anemia (Hb 8.8 g/dL). She was therefore referred to our gynecological clinic for further evaluation to determine whether a gynecological disease was present. Transvaginal ultrasonography revealed a large 48 mm × 50 mm mass compressing the lining of the anterior uterine wall. Based on contrast-enhanced magnetic resonance imaging (MRI) findings, the mass was diagnosed as a uterine leiomyoma (
Figure 1A). However, because the patient preferred monitoring until menopause, we prescribed iron supplements to treat the anemia and continued tumor follow-up. The tumor did not show any signs of enlargement. At 56 years of age, however, she developed lower abdominal pain after undergoing an endoscopic examination of the lower gastrointestinal tract. Blood test results revealed an elevated C-reactive protein (CRP) level of 8.49 mg/dL. We suspected the development of peritonitis due to delayed perforation and therefore performed contrast-enhanced CT. The imaging showed increased adipose tissue density in the mesenteric lining around the uterus as well as ascites.
Physical examination findings were as follows: blood pressure 135/72 mmHg, pulse 64 beats/min, and mild abdominal tenderness.
Pelvic examination revealed a small amount of white vaginal discharge, erosions in the uterine vaginal region, a first-sized and mildly tender uterine body, and nonpalpable adnexa.
Transvaginal ultrasonography showed a 90 mm x 69 mm heterogeneous mass with irregular margins in the uterine body (
Figure 1B).
Blood test results revealed no abnormalities except for an elevated platelet count of 500,000/µL. Liver and renal function test results were normal. Serum lactate dehydrogenase (LDH) and total protein (TP) levels were elevated at 401 U/Land 8.2 g/dL, respectively. The fibrinogen level was 604 mg/dL, indicating coagulation abnormalities. CRP levels were 0.37 mg/mL, slightly above the normal range. Tumor marker levels were within normal limits: CEA 1.8 ng/mL, CA19–9 9 U/mL, and CA125 18 U/mL.
Endoscopy of the lower gastrointestinal tract showed no abnormal findings.
Truncal computed tomography (CT) (
Figure 1C) revealed a 10 cm x 11 cm mass with marginal calcification on the anterior uterine wall and cranial protrusions of the uterus. A pale, low-density area suggestive of hemorrhage was observed within the mass, and a small amount of pelvic fluid was present, suggesting the presence of blood. Increased adipose tissue density surrounding the mass suggested possible rupture.
A contrast-enhanced CT performed for preoperative evaluation 8 days after the initial scan showed that the uterine mass had decreased to 9.5 cm × 9.5 cm and the bloody ascites had resolved (
Figure 1D). The fat density surrounding the cranial protrusion of the uterus had decreased, and the wall in that region had thinned. The low-density background area within the mass had become less prominent, suggesting a reduction in bloody fluid. No lymph node enlargement or distant metastasis was observed.
Pelvic contrast-enhanced MRI (
Figure 1E) showed a 10 cm mass located within the muscle layer of the anterior uterine wall. T2W1 demonstrated moderate signal intensity within the mass, and DWI demonstrated high signal intensity. Heterogeneous internal signals suggestive of hemorrhage and necrosis were present. T2W1 also showed signal imaging, which showed a low-signal area on the ventral side of the mass. CT demonstrated areas of calcification, and contrast-enhanced MRI confirmed that both ovaries were normal.
Based on the rapid tumor growth accompanied by bleeding, necrosis, rupture, and elevated LDH levels, rupture of the uterine leiomyosarcoma was suspected. The preoperative stage corresponded to stage IB disease.
Surgical findings:
Rupture of the tumor mass on the anterior uterine wall was observed, with hematoma and tumor contents exposed to the abdominal cavity. Adhesion between the tumor and ileum was observed near the rupture site, raising suspicion of tumor infiltration. Macroscopically, both adnexa were normal. No lesions suggestive of disseminated metastasis were identified in the abdominal cavity; therefore, a simple total hysterectomy, bilateral salpingo-oophorectomy, partial omentectomy, and partial ileectomy were performed.
Histopathological findings:
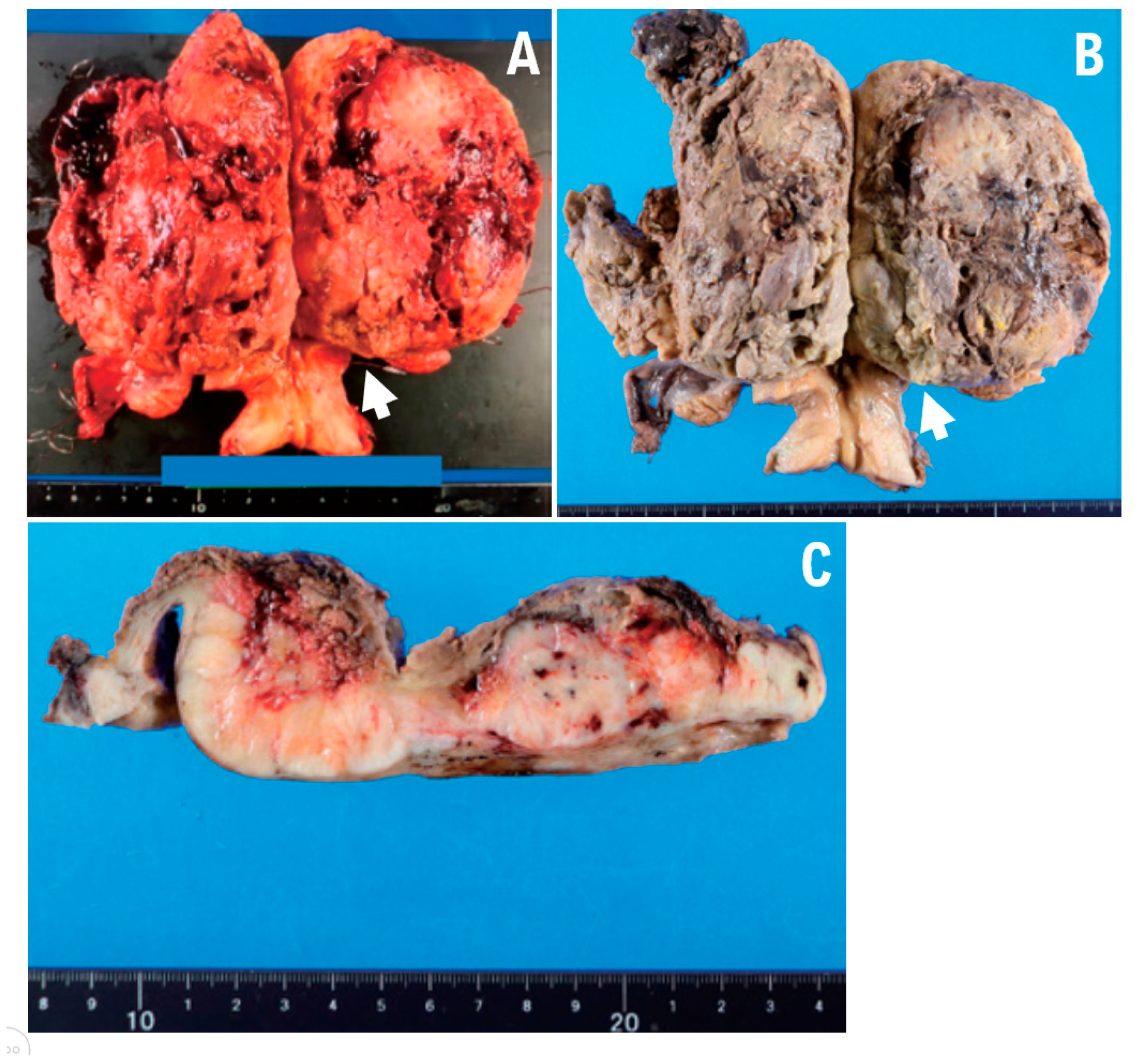
A 12 cm hemorrhagic lesion was identified in the uterine body (
Figure 2A–C).
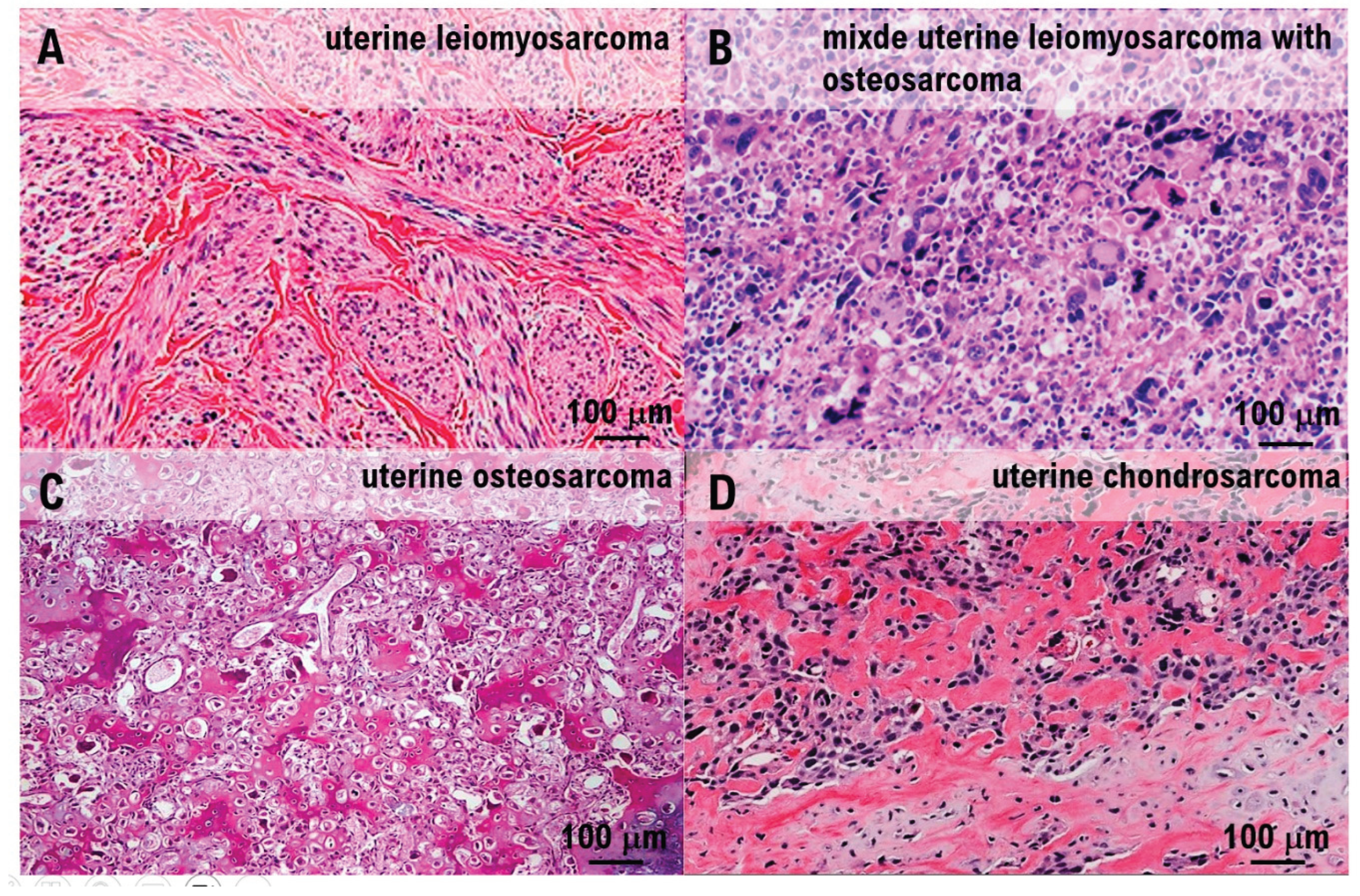
Histologically, irregular fascicular proliferation of spindle cells with marked nuclear atypia was observed. Approximately 12 mitotic figures per 10 HPF and areas of coagulation necrosis were present, supporting the diagnosis of uterine leiomyosarcoma (
Figure 3A–B). Multinucleated giant cells were scattered throughout the tumor tissue (
Figure 3C), and ectopic osteosarcomatous and chondrosarcomatous elements were identified (
Figure 3D). Ectopic bone and cartilage elements were observed extensively in 29 of the 33 pathological sections containing uterine leiomyosarcoma. A 12 cm mass lesion accompanied by hemorrhage was present in the uterine body (
Figure 2A–C).
No neoplastic changes are observed in the endometrium. Benign leiomyomas were not identified. The Ki67 labeling index was 50%. No evidence of tumor invasion was found in the ileum, which was attached to and resected with the tumor. No malignant findings were present in the cervix, bilateral adnexa, or greater omentum. Based on these histological and pathological findings, the patient was diagnosed with ruptured uterine leiomyosarcoma containing ectopic components (pT1bN0M0).
Postoperative course:
In this case, rupture of the uterine leiomyosarcoma was observed, and our medical staff determined that the risk of recurrence and metastasis was high. Therefore, we administered six cycles of doxorubicin as postoperative adjuvant chemotherapy. Contrast-enhanced CT performed after completion of adjuvant therapy revealed no evidence of recurrence. However, a contrast-enhanced CT scan performed one year after surgery revealed a 9 mm mass in the right upper lobe of the lung. Thoracoscopic partial resection was performed, and pathological examination confirmed that the mass was a metastatic uterine leiomyosarcoma with osteosarcoma and chondrosarcoma.
Discussion
Leiomyosarcomas with ectopic components are extremely rare, even when an extrauterine origin is considered, and uterine leiomyosarcomas with ectopic components arising from the uterus are particularly uncommon. 1)–-3) Examples of sarcomatous lesions with ectopic components include osteosarcoma, angiosarcoma, rhabdomyosarcoma, and liposarcoma. 4) The uterine leiomyosarcoma in this case contained osteosarcoma and chondrosarcoma components. Among previously reported cases of sarcomas with ectopic components, there are few cases of uterine leiomyosarcomas with osteosarcomatous and chondrosarcomatous elements. To the best of our knowledge, only seven such cases have been reported to date.
Because uterine leiomyosarcoma containing ectopic components has been reported to originate from other organs, medical staff should perform a full-body assessment at the time of diagnosis to determine whether lesions are present in other organs. 3) In this case, imaging studies revealed no extrauterine lesions, and the tumor was considered primary to the uterus. Based on the imaging and pathological findings, the patient was diagnosed with uterine leiomyosarcoma containing rare osteosarcomatous and chondrosarcomatous components.
This case was also accompanied by rupture of the uterine leiomyosarcoma. Rupture of uterine tumors is rare, and only four cases of uterine leiomyosarcoma rupture have been reported. 9)–12) In reviews of uterine fibroid rupture, causes such as traffic injuries and post-defecation events have been described. 13)
However, uterine leiomyosarcoma rupture may occur due to tumor growth 9) or as a result of abdominal pressure, such as during defecation. 10) In the present case, the tumor was discovered when the patient developed abdominal pain immediately after endoscopic evaluation of the lower gastrointestinal tract; therefore, rupture due to external stimuli was suspected. However, at the previous routine examination, no significant increase in tumor size had been noted, suggesting rapid interval growth. Exogenous uterine rupture generally does not occur without substantial external force, such as trauma.
Although there have been reports of iatrogenic rupture during intrauterine balloon insertion for postpartum hemorrhage 14), there are no reports describing its use in non-pregnant women. It is highly likely that the primary cause of uterine leiomyosarcoma rupture is rapid tumor growth. The high Ki67 labeling index of the tumor also suggests rapid proliferation.
Uterine leiomyosarcoma has a poor prognosis, with a 5-year survival rate of 40–70% for stage I–II tumors. 1) In this case, because the tumor had ruptured, the medical staff believed the risk of recurrence was higher than that for typical stage I tumors and therefore administered postoperative chemotherapy. However, despite completing chemotherapy, the patient developed recurrence and metastasis. Although the impact of rupture on the prognosis of uterine leiomyosarcoma remains unclear, rupture has been reported as an independent poor prognostic factor in gastrointestinal stromal tumors (GISTs), which are malignant stromal tumors originating from the gastrointestinal tract. Thus, ruptured uterine leiomyosarcomas may also have a poor prognosis. In addition, some reports indicate that leiomyosarcomas containing ectopic components originating from other sites have a poor prognosis 16), and that osteosarcoma and chondrosarcoma arising in the uterus also show poor outcomes 17). However, it is still uncertain whether uterine leiomyosarcomas with ectopic components have worse prognoses than ordinary uterine leiomyosarcomas. In this case, no recurrence was observed on contrast-enhanced CT at the completion of adjuvant chemotherapy, but lung metastases were detected one year after surgery, suggesting high tumor malignancy and limited benefit from postoperative adjuvant chemotherapy.
Unlike uterine leiomyomas, uterine leiomyosarcoma can develop after menopause, even in women as old as 80 years. The 5-year survival rate for uterine leiomyosarcoma is less than 20% 18). Therefore, early diagnosis and surgical treatment are essential for long-term survival.
Ethical considerations
Institutional Review Board (IRB) Approval and Consent to Participate: This research on human cancer genome information derived from results of cancer genome profiling was conducted by Cancer Genomic Medicine Members at the Kyoto University Hospital, its affiliated hospitals, and the National Hospital Organization Kyoto Medical Center in accordance with institutional guidelines (IRB approval no. 50-201504, NHOKMC-2023-2, and H31-cancer-2). All patients received an explanation of the clinical trial and provided informed consent to participate in this study. Clinical research conducted by us complies with the Declaration of Helsinki.
Ethics committee name: IRB of the National Hospital Organization Headquarter (Meguro, Tokyo, Japan, approval code: H31-cancer-2; approval date: November 09, 2019, and August 14, 2025).
Ethics committee name: IRB of Kyoto University Hospital (Kyoto, Kyoto, Japan, approval code: R34005; approval date: August 01, 2025).
Ethical compliance with human study
This study involves human subjects and has been approved by the ethics committee and IRB (Institutional Review Board) of the institution to which it is affiliated. This paper contains personally identifiable personal and/or medical information, as well as case reports/medical histories. Therefore, in accordance with our anonymization policy, it has been sufficiently anonymized to prevent the identification of individual patients. The authors obtained consent directly from the patients.
The authors attended research ethics education through the Education for Research Ethics and Integrity (APRIN e-learning program (eAPRIN)) agency. The completion numbers for the authors are AP0000151756, AP0000151757, AP0000151769, and AP000351128.
This study involves research with human materials and was approved by the institutional ethics committee(s) and IRBs (Ethics Committee for research with animals in National Hospital Organization Headquarter; Meguro, Tokyo, Japan).
Ethics committee name: IRB of the National Hospital Organization Headquarters (approval code: H31-1-2; approval date: November 09, 2019, and June 17, 2023, approved code: R07-1-2).
Author Contributions
TH and YA reviewed the content of the case reports and selected the images obtained from the contrast-enhanced MRI examinations. TH, YA, and IK compiled the patient’s medical history and chief complaints. TH, YA, and IK were the attending physicians and reviewed the case report and blood test results. IK reviewed the draft case report.
Funding
The costs of conducting our clinical research are covered by the following research grants: Japan Society for Promoting Science for TH (no. 19K09840), START-program Japan Science and Technology Agency (JST) for TH (no. STSC20001), National Hospital Organization Multicenter Clinical Study for TH (grant no. 2019-Cancer in general-02), and Japan Agency for Medical Research and Development (AMED) (no. 22ym0126802j0001), Tokyo, Japan. These research funds were provided by Japanese government research institutes, and therefore the funders were not involved in the establishment of the clinical research program, the collection and analysis of data, the public dissemination of research findings, or the preparation of the manuscript.
Informed Consent Statement
This research includes clinical/human materials, therefore Informed consent is required.
Data Availability Statement
The data are available and publicly available on various websites. Details are provided in the first paragraph of the Results section. Information from this clinical study and the associated transparency statement in the medical journal article are available online.
https://kyoto.hosp.go.jp/html/guide/medicalinfo/ clinical research/expand/gan.html (accessed on 15 March 2025).
Acknowledgments
The authors want to thank Dr. Kohji Moriyoshi at The pathology division, National Hospital Organization, Kyoto Medical Center. The authors also want to acknowledge all clinical staff for clinical research at Kyoto University Hospital and the National Hospital Organization Kyoto Medical Center.
Conflicts of Interest
Authors have nothing to disclose. .
References
- Parikh, P.; Maheshwari, A.; Rekhi, B. Two uncommon cases of uterine leiomyosarcomas displaying heterol- ogous osteosarcomatous de-differentiation. J. Cancer Res. Ther. 2015, 11, 654. [Google Scholar] [CrossRef] [PubMed]
- Rawish, K.R.; Fadare, O. Dedifferentiated Leiomyosar- coma of the Uterus with Heterologous Elements : A Potential Diagnostic Pitfall. Case Rep. Obstet. Gynecol. 2012, 2012, 534634. [Google Scholar] [PubMed]
- Chen, E.; O’cOnnell, F.; Fletcher, C.D.M. Dedifferentiated leiomyosarcoma : Clinicopathological analysis of 18 cases. Histopathology 2011, 59, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Anh, T.T.; Holloway, R.W. Metastatic leiomyosarcoma of the uterus with heterologous differentiation to Malignant Mesenchymoma. Int. J. Gynecol. Pathol. 2012, 31, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Damjanov, I.; Fan, F. Chondrosarcomatous differentiation of a metastatic leiomyosarcoma of the uterus. Virchows Arch. 2006, 449, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Kew, C.C.; Putti, T.C.; Razvi, K. Malignant mesenchy- moma arising from a uterine leiomyoma in the meno- pause. Gynecol. Oncol. 2004, 95, 712–715. [Google Scholar] [CrossRef] [PubMed]
- den Bakker, M.A.; Hegt, V.N.; Sleddens, H.B.F.M.; Nuijten, A.S.M.; Dinjens, W.N.M. Malignant mesenchymoma of the uterus, arising in a leiomyoma. Histopathology 2002, 40, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Vakiani, M.; Mawad, J.; Talerman, A. Heterologous sarcomas of the uterus. Int. J. Gynecol. Pathol. 1982, 1, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Özcan, J.; Dülger, Ö.; Küpelioğlu, L.; Gönenç, A.I.; Erşahin, A. Uterine sarcoma in a 14 year-old girl presenting with uterine rupture. Gynecol. Oncol. Rep. 2014, 10, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Barua, R.; Olesnicky, G. Uterine leiomyosarcoma pre- senting with haemoperitoneum. Med. J. Aust. 1988, 145, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Farhi, J.; Dicker, D.; Ben-Rafael, Z. Leiomyosarcoma presenting as spontaneous haemoperitoneum. Int. J. Gynaecol. Obstet. 1993, 40, 249–250. [Google Scholar] [CrossRef] [PubMed]
- Oda, H.; Hirakawa, H. Spontaneous rupture of uterine smooth muscle tumour presenting acute abdominal pain and haemoperitoneum. BMJ Case Rep. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.H.; Cohen, S.C.; Lamaro, V.P. Intra-abdominal haemorrhage from uterine fibroids : A systematic review of the literature. BMC Surg. 2020, 20, 70. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.T.; Lozada, M.J.; Gorrindo, P.; Peralta, F.M. Massive Hemorrhage From Suspected Iatrogenic Uterine Rupture. Obstet. Gynecol. 2018, 132, 1494–1497. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, Q. Prognostic Indicators for Gastroin- testinal Stromal Tumors : A Review. Transl. Oncol. 2020, 13, 100812. [Google Scholar] [CrossRef] [PubMed]
- Gaeta, R.; Matera, D.; Muratori, F.; Roselli, G.; Baldi, G.; Campanacci, D.A.; Franchi, A. Dedifferentiated soft tissue leiomyosarcoma with heterologous osteosarcoma component: Case report and review of the literature. Clin. Sarcoma Res. 2020, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Fadare, O. Heterologous and rare homologous sarco- mas of the uterine corpus : A clinicopathologic review. Adv. Anat. Pathol. 2011, 18, 60–74. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Kawano, M.; Sano, K.; Ichimura, T.; Gur, G.; Yaish, P.; Zharhary, D.; Kanai, Y.; Tonegawa, S.; Shiozawa, T.; et al. A novel diagnostic biomarker for human uterine leiomyosarcoma: PSMB9/β1i. Chin. Clin. Oncol. 2017, 6, 22. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).