Submitted:
28 November 2025
Posted:
03 December 2025
You are already at the latest version
Abstract
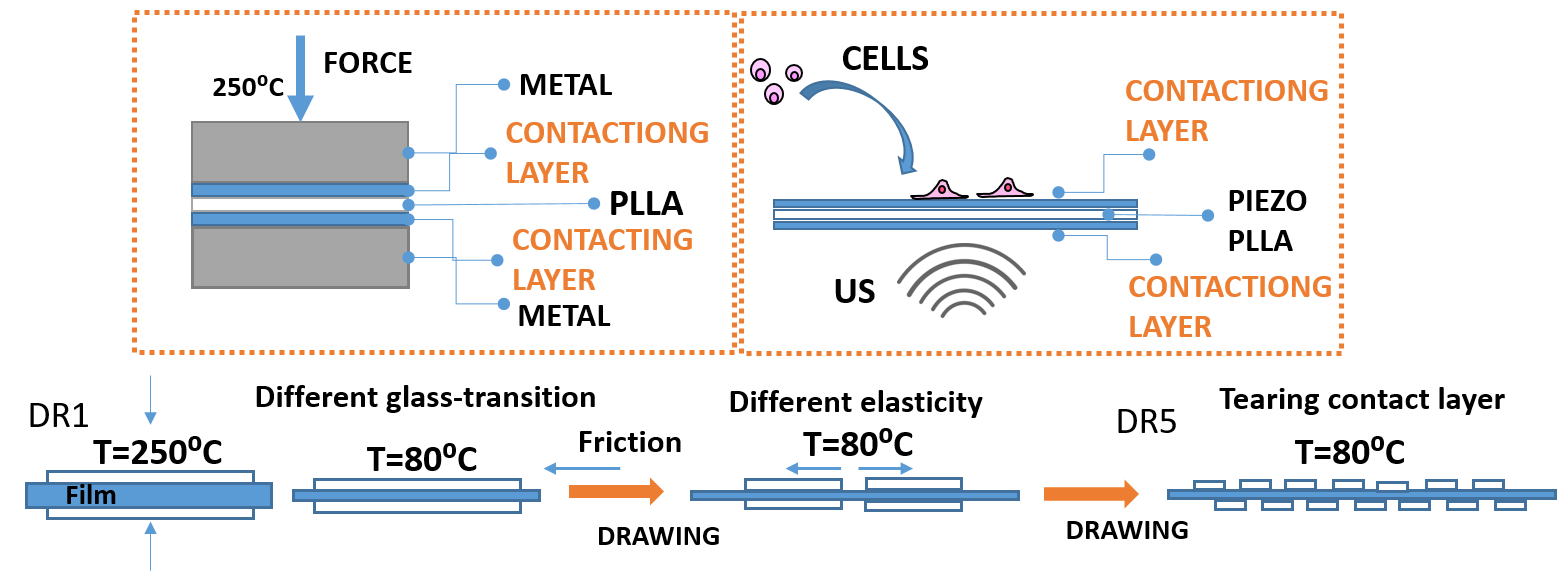
The main limitation of high-temperature drawing approach for tailoring crystallization and molecular orientation of poly-l-lactide (PLLA) toward ultrasound- active piezoelectric structures is set by intrinsic properties of the processed polymer, including low melting / softening elasticity and slow crystallization kinetics. Here we found that application of different contacting layers, including polytetrafluoroethylene (PTFE) (as Teflon and Teflon S), cellulose (paper) or polyimine (Kapton) deposited at the surface of PLLA, significantly affects the drawing process and tailors its oriented crystallization and molecular chain orientation. Consequently the contacting layers contribute to piezoelectric properties of PLLA, affect their activation by ultrasound and generated electro-signal. Human keratinocytes (HaCaT cells) grown stimulated on these surfaces are shown to receive and respond to the transferred stimuli by activation of the cytoskeleton and directional migration. The high-temperature drawing approach with contacting layers is simple, solvent-free and economically continent way for broadening limitations of classical high-temperature drawing which opens new possibilities for further tailoring piezoelectricity of organic piezoelectrics.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
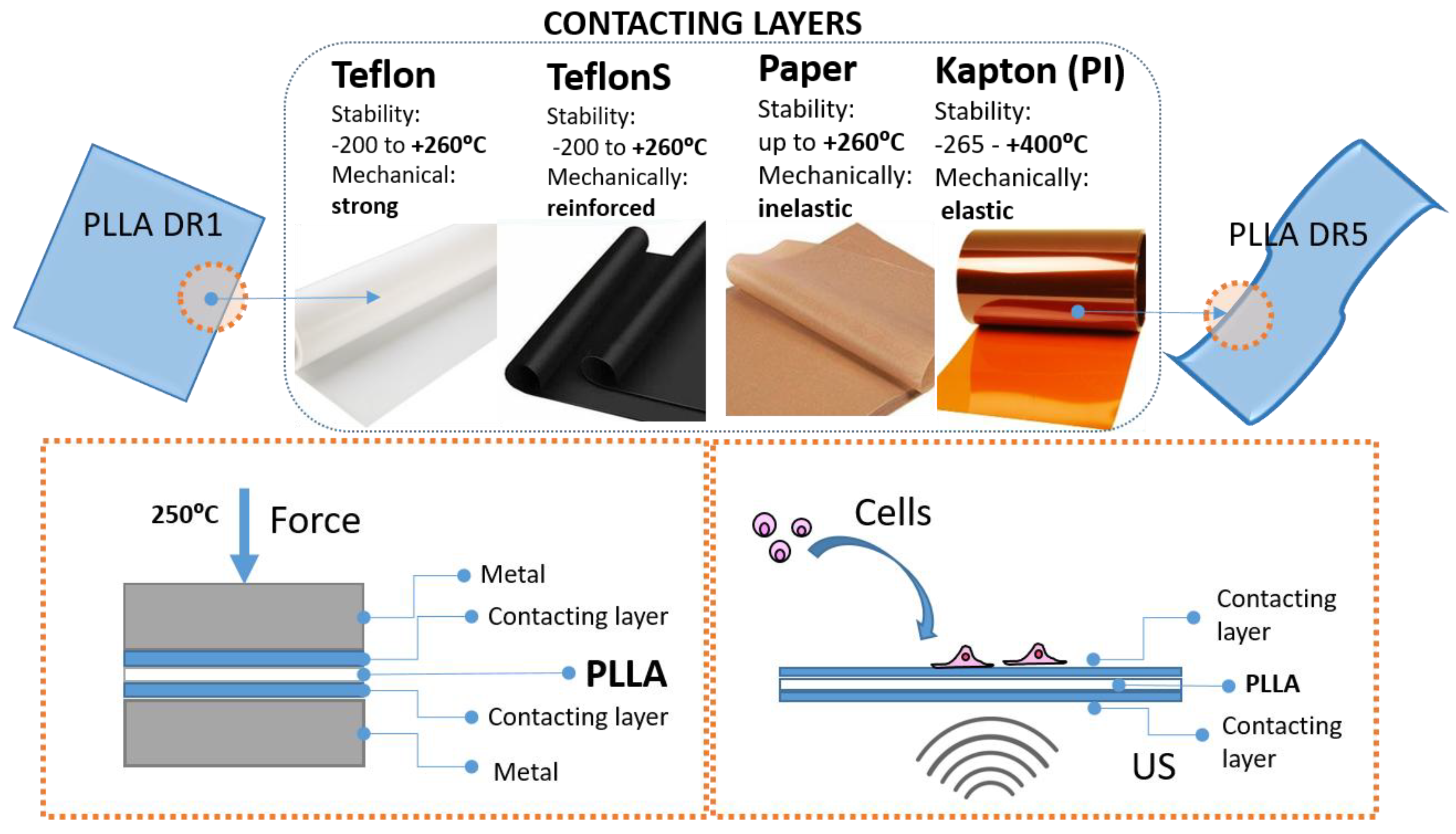
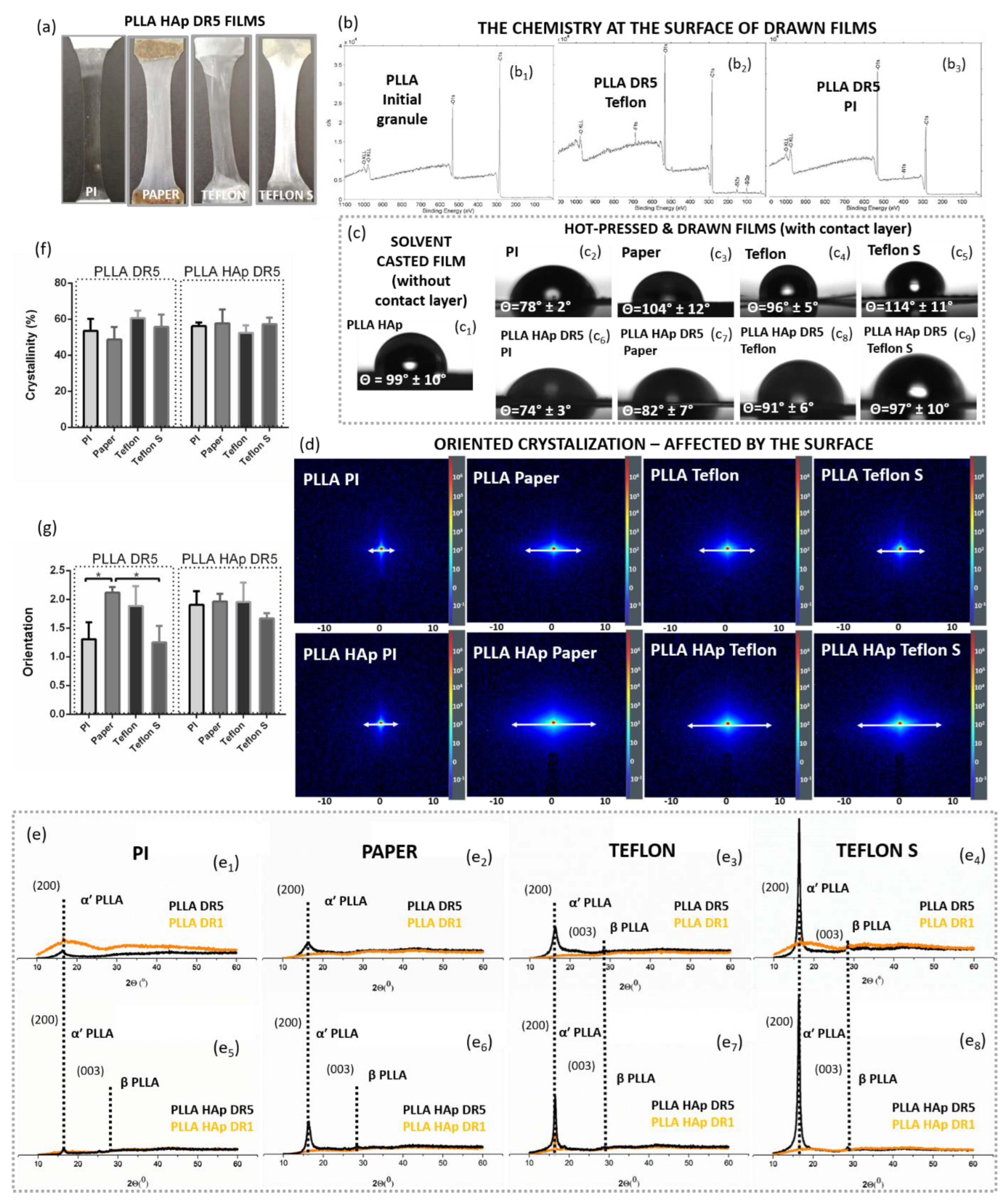
3.1. Surface –Induced Crystallization of PLLA

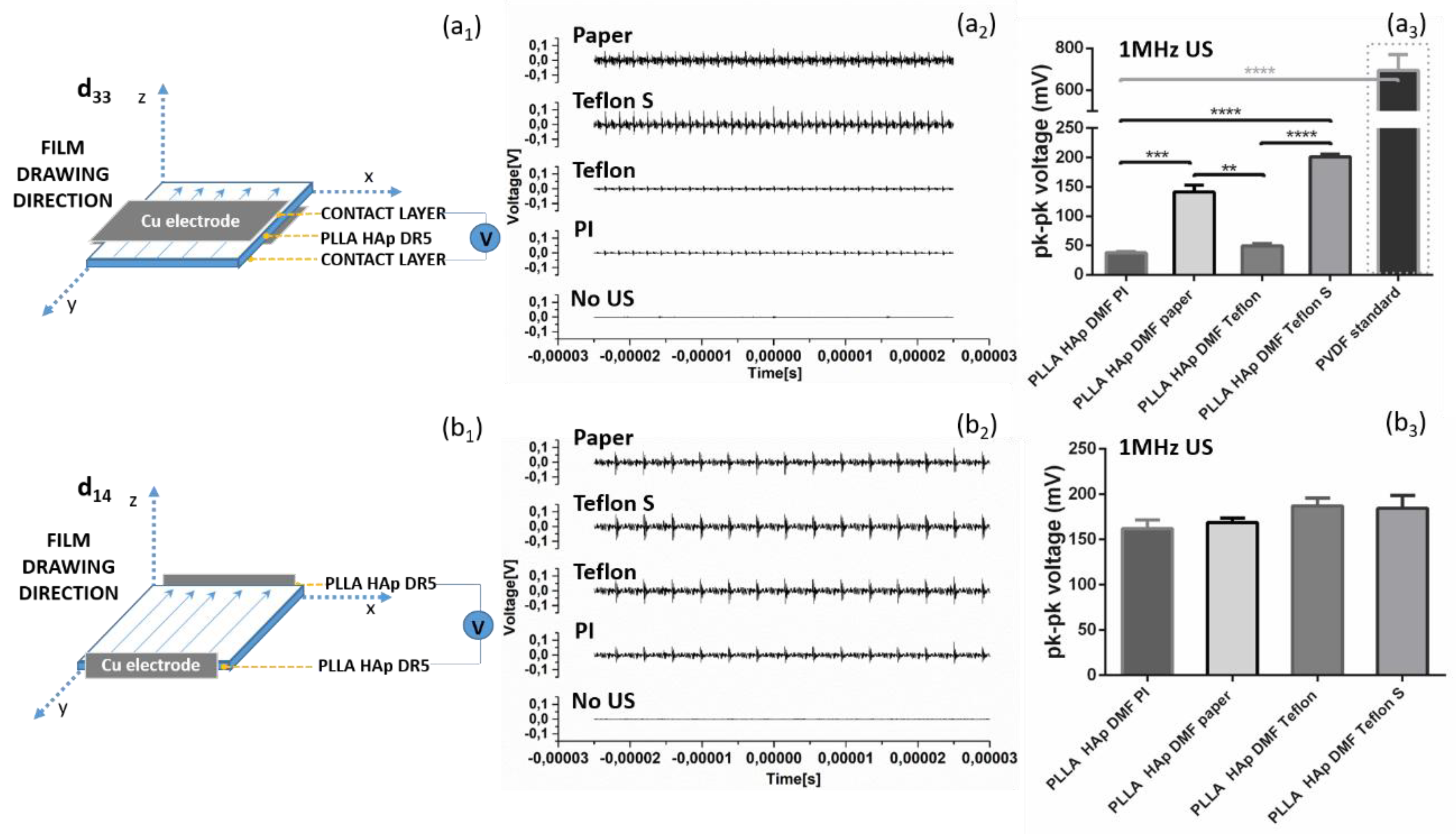
3.2. Ultrasound Activation of PLLA Films with Different Contacting Layers
3.3. Reactive Oxygen Species (ROS) Formation During Biomaterials Ultrasound Activation
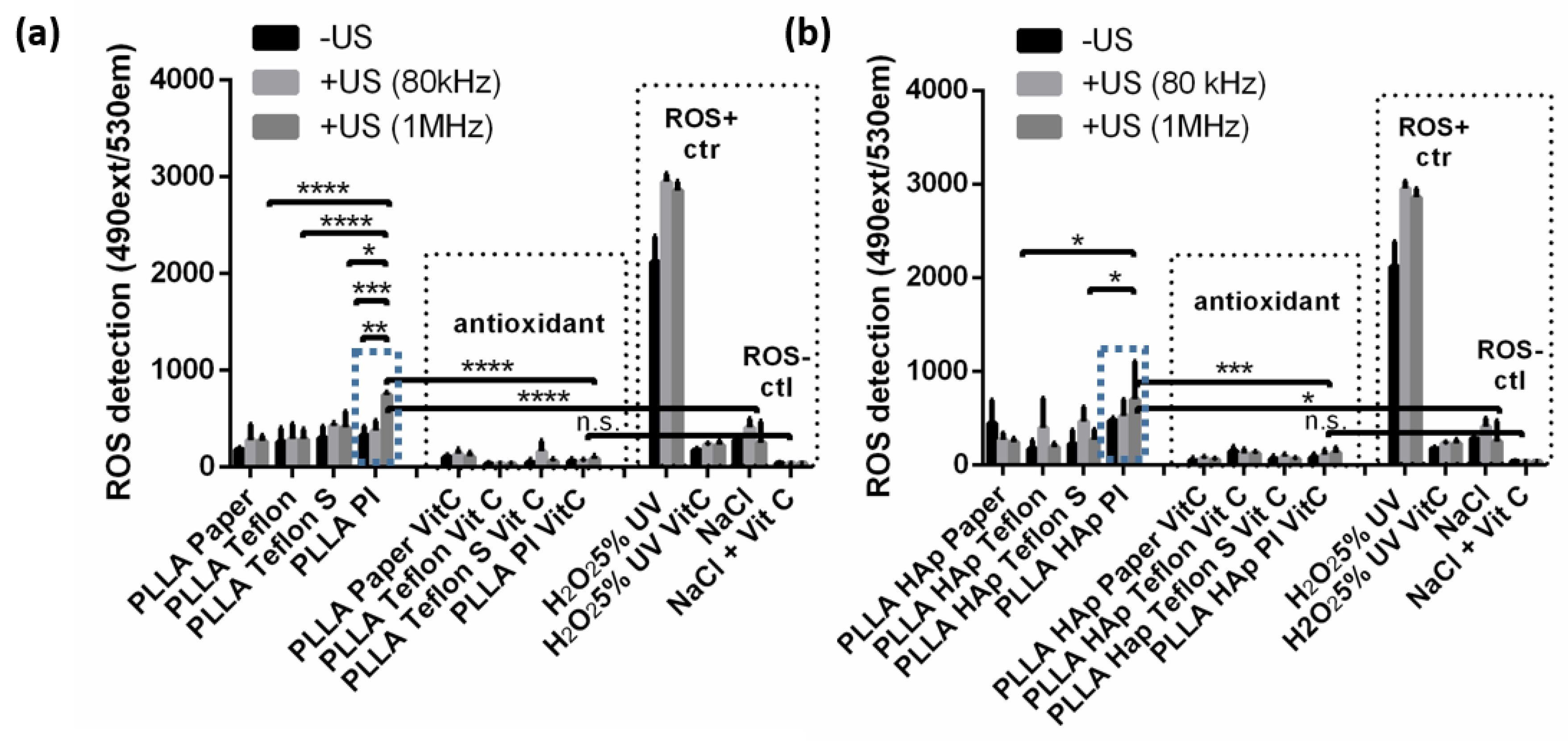
3.4. The Interactions of Human Keratinocyte (HaCaT) Cells with Ultrasound Activated Biomaterials
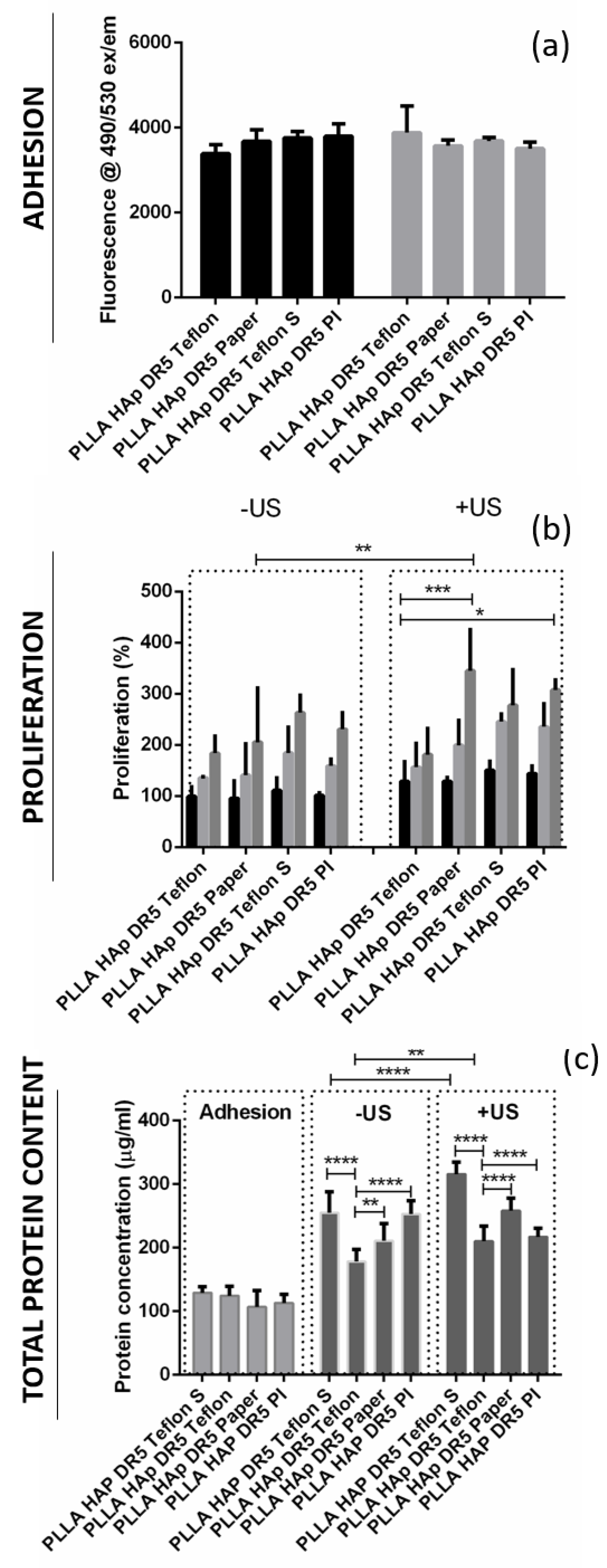
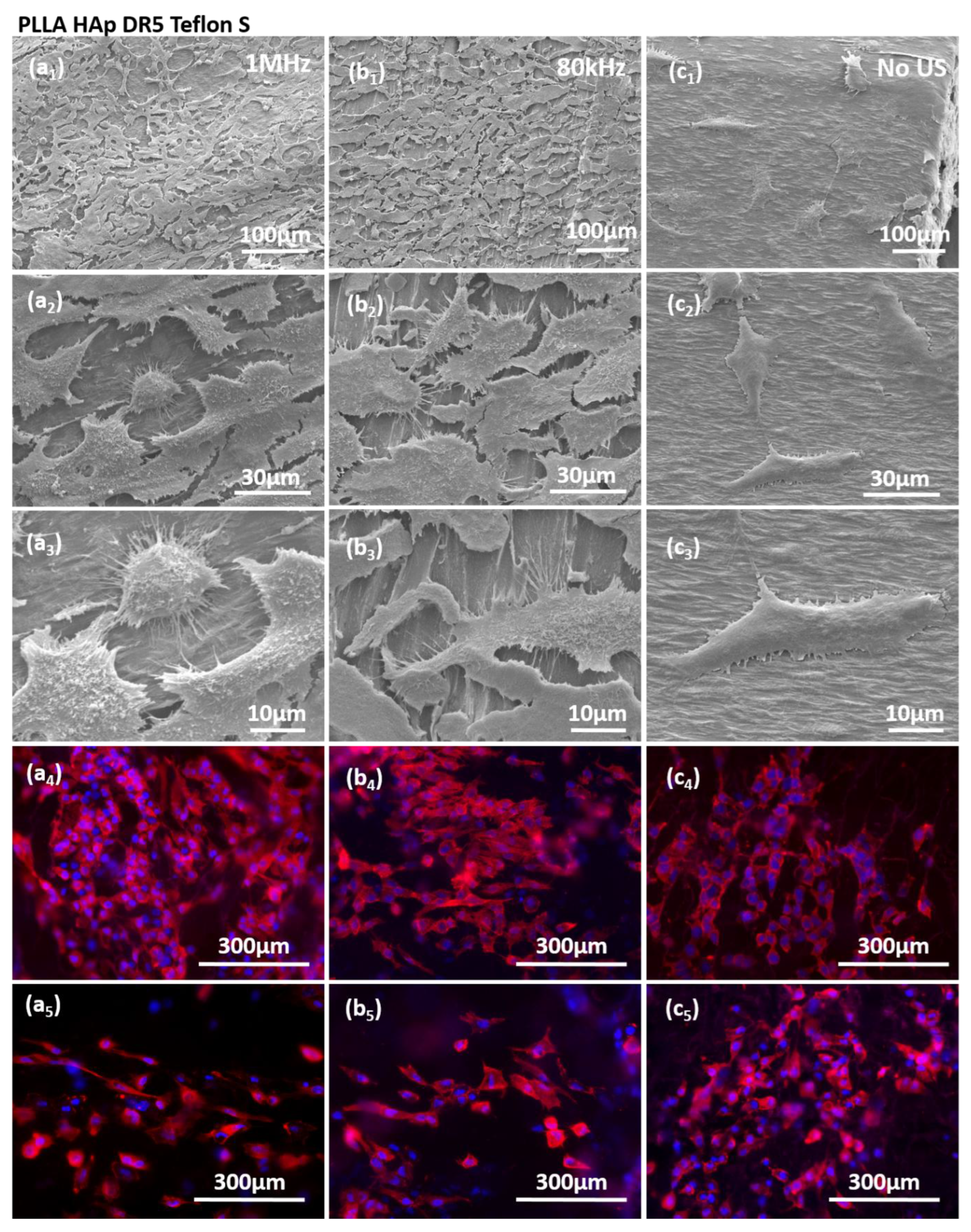
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PLLA | Poly-l-lactide |
| PI | Polyimide |
| PTFE | Polytetrafluoroethylene |
| ROS | Reactive oxygen species |
| HAp | Hydroxyapatite |
| DHR | Dihydrorhodamine 123 |
| DMF | N,N-Dimethylformamide |
| DR | Drawing ratio |
| DMEM | Dulbecco's Modified Eagle Medium |
| BCA | Bicinchoninic acid assay |
References
- Zhang, S.; Zhang, H.; Sun, J.; Javanmardi, N.; Li, T.; Jin, F.; He, Y.; Zhu, G.; Wang, Y.; Wang, T.; Feng, Z.Q. A review of recent advances of piezoelectric poly-L-lactic acid for biomedical applications. Int. J. Biol. Macromol. 2024, 276, 133748. [Google Scholar] [CrossRef]
- Khan, A.; Joshi, R.; Sharma, M.K.; Huang, C.J.; Yu, J.H.; Wang, Y.L.; Lin, Z.H. The potential of organic piezoelectric materials for next-generation implantable biomedical devices. Nano Trends 2024, 6, 100032. [Google Scholar] [CrossRef]
- Wang, R.; Sui, J.; Wang, X. Natural Piezoelectric Biomaterials: A Biocompatible and Sustainable Building Block for Biomedical Devices. ACS Nano 2022, 11, 17708–17728. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Z.; Li, X.; Zheng, Y.; Yang, Z. Design and Manufacturing of Piezoelectric Biomaterials for Bioelectronics and Biomedical Applications. Chem. Rev. 2025, 125, 9875–9929. [Google Scholar] [CrossRef] [PubMed]
- Simamora, P. & Chern, W. Poly-L-lactic acid: an overview. Journal of drugs in dermatology : JDD 2006, 5, 5–436. [Google Scholar]
- Elmowafy, E.M.; Tiboni, M.; Soliman, M.E. Biocompatibility, biodegradation and biomedical applications of poly(lactic acid)/poly(lactic-co-glycolic acid) micro and nanoparticles. J. Pharm. Investig. 2019, 49, 347–380. [Google Scholar] [CrossRef]
- Yoon, S.D.; Kwon, Y.S.; Lee, K.S. Biodegradation and biocompatibility of poly L-lactic acid implantable mesh. Int. Neurourol. J. 2017, 21 (Suppl 1), S48–54. [Google Scholar] [CrossRef]
- Su, D.; Yang, W.; He, T.; Wu, J.; Zou, M.; Liu, X.; Li, R.; Wang, S.; Lai, C.; Wang, J. Clinical applications of a novel poly-L-lactic acid microsphere and hyaluronic acid suspension for facial depression filling and rejuvenation. J. Cosmet. Dermatol. 2024, 23, 3508–3516. [Google Scholar] [CrossRef]
- Wei, Y.; Li, Z.; Yu, T.; Chen, Y.; Yang, Q.; Wen, K.; Liao, J.; Li, L. Ultrasound-activated piezoelectric biomaterials for cartilage regeneration. Ultrason. Sonochem. 2025, 117, 107353. [Google Scholar] [CrossRef] [PubMed]
- Vinikoor, T.; et al. Injectable and biodegradable piezoelectric hydrogel for osteoarthritis treatment. Nat. Commun. 2023. [Google Scholar] [CrossRef]
- Ochiai, T.; Fukada Eiichi. Electromechanical Properties of Poly-L-Lactic Acid. Jpn. J. Appl. Phys. 1998, 37, 3374–3376. [Google Scholar] [CrossRef]
- Guerin, S.; Tofail, S.A.M.; Thompson, D. Organic piezoelectric materials: milestones and potential. NPG Asia Materials 2019. [Google Scholar] [CrossRef]
- Ali, M.; Bathaei, M.J.; Istif, E.; Karimi, S.N.H. & Beker, L. Biodegradable Piezoelectric Polymers: Recent Advancements in Materials and Applications. Adv. Healthc. Mater. 2023, 12, 1–32. [Google Scholar]
- Schönlein, R.; et al. Piezoelectric polylactic acid-based biomaterials: Fundamentals, challenges and opportunities in medical device design. Biomaterials 2026, 324. [Google Scholar] [CrossRef]
- Merhi, Y.; et al. Advancing green electronics: tunable piezoelectric enhancement in biodegradable poly(l-lactic acid) PLLA films through thermal-strain engineering. Nanoscale Horizons 2025, 10, 1414–1427. [Google Scholar] [CrossRef]
- Vukomanović, M.; et al. Filler-Enhanced Piezoelectricity of Poly-L-Lactide and Its Use as a Functional Ultrasound-Activated Biomaterial. Small 2023. [Google Scholar] [CrossRef]
- Udovc, L.; Spreitzer, M.; Vukomanovic, M. Towards hydrophilic piezoelectric poly-L-lactide films: optimal processing, post-heat treatment and alkaline etching. Polym. J. 2020, 52, 299–311. [Google Scholar] [CrossRef]
- Saeidlou, S.; Huneault, M.A.; Li, H.; Park, C.B. Poly(lactic acid) crystallization. Progress in Polymer Science 2012. [Google Scholar] [CrossRef]
- Huang, A.; et al. Mechanical properties, crystallization characteristics, and foaming behavior of polytetrafluoroethylene-reinforced poly(lactic acid) composites. Polym. Eng. Sci. 2017. [Google Scholar] [CrossRef]
- Ren, Q.; et al. Cellulose nanofiber reinforced poly (lactic acid) with enhanced rheology, crystallization and foaming ability. Carbohydr. Polym. 2022. [Google Scholar] [CrossRef]
- Chen, S.; et al. The effect of polytetrafluoroethylene particle size on the properties of biodegradable poly(butylene succinate)-based composites. Sci. Rep. 2021. [Google Scholar] [CrossRef]
- Jevtić, M.; et al. Crystal structure of hydroxyapatite nanorods synthesized by sonochemical homogeneous precipitation. Cryst. Growth Des. 2008. [Google Scholar] [CrossRef]
- Jevtić, M. & Uskokovic, D.P. Influence of Urea as Homogeneous Precipitation Agent on Sonochemical Hydroxyapatite Synthesis. Mater. Sci. Forum 2007. [Google Scholar] [CrossRef]
- Aničić, N.; Vukomanović, M.; Koklič, T.; Suvorov, D. Fewer Defects in the Surface Slows the Hydrolysis Rate, Decreases the ROS Generation Potential, and Improves the Non-ROS Antimicrobial Activity of MgO. Small 2018. [Google Scholar] [CrossRef]
- Piwowarczyk, J.; et al. XPS and FTIR studies of polytetrafluoroethylene thin films obtained by physical methods. Polymers (Basel). 2019. [Google Scholar] [CrossRef]
- Shi, J.; Gong, C.; Tian, X.; Yang, S.; Chu, P.K. Optical properties and chemical structures of Kapton-H film after proton irradiation by immersion in a hydrogen plasma. Appl. Surf. Sci. 2012. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



