1. Introduction
Donkeys have lost their traditional role in industrialized communities, leading to a drastic reduction in their numbers, particularly in developed regions such as Europe, over the last two decades. As a result, most European donkey breeds have become endangered or extinct [
1]. Nowadays, donkeys are assuming new roles in society, such as landscape maintenance, flock protection, donkey milk production and participation in social and leisure activities [
2]. Their endangered status and increasing relevance in society makes it crucial to preserve and enhance their genetic pool [
3].
Assisted reproductive technologies (ARTs) play a key role on the conservation of endangered species [
4]. ARTs are often extrapolated from horses to donkeys, despite obtaining poorer fertility outcomes in this species [
5,
6,
7,
8]. Crossbreeding studies have suggested that the lower fertility in donkeys is related to an exacerbated inflammatory response of the endometrium of the jenny [
7,
9], which mechanism remains unclear [
10,
11,
12,
13]. A possible explanation could be the differences on the reproductive physiology and anatomy of jennies and mares. Jennies present proportionally larger internal genitalia, narrower and more tortuous cervix forming a deeper fornix, less endometrial edema in estrus, and more branched uterine glands [
14]. This unique reproductive anatomy of the jennies might contribute to an exacerbated post-breeding endometrial inflammation [
13]. However, to the best of our knowledge, no studies have objectively compared the uterine anatomical differences between jennies and mares.
Histomorphometry has been extensively used to objectively evaluate uterine features of several species including variations among individuals [
15,
16], breeds [
17] and species [
18]. In mares, histomorphometry has been used to evaluate the effect of the estrus cycle [
19]; age and reproductive history [
20]; susceptibility to endometritis [
21]; intrauterine fluid accumulation [
22]; and treatment responses [
22,
23] on the uterine anatomy. Nevertheless, there is limited research on this topic in jennies [
14]. A comparative study could shed light on physiologic anatomical features predisposing to poorer reproductive outcomes in jennies. Thus, the objective of this study was to compare the histomorphometric characteristics of the endometrium of reproductively sound jennies and mares.
2. Materials and Methods
2.1. Animals and Breeding Soundness Evaluation
A total of twelve animals were included in this study: six mares (
Equus caballus), three Purebred Spanish Horse and three Purebred Arabian Horse; and six Andalusian Donkey Breed jennies
(Equus asinus). The age of the animals ranged from 3 to 16 years old. Prior to their inclusion in the study, a reproductive soundness evaluation was conducted. Parity status and reproductive history was recorded. Females were classified as maiden, if they had never been bred; or foaling, if they had foaled the year of the study [
24].
The reproductive soundness evaluation consisted of the assessment of cyclic ovarian activity and the absence of pathological images in the reproductive system evaluated by transrectal ultrasonography (Logiq V2, GE Medical Systems, China), endometrial biopsy, and endometrial cytology. Cytology and biopsy samples were taken in estrus, as explained below. The animals were considered reproductively sound if the percentage of polymorphonuclear leukocytes (PMNs) in endometrial cytology smear was lower than 5% [
25] and the biopsy was Category I or IIa according to Kenney and Doig (1986) endometrial biopsy grading system (KDGS).
2.2. Sample Collection
Ovarian cycle control was performed weekly by seriated ultrasound examinations. Endometrial samples were collected when behavioral estrus signs were displayed, in the presence of a dominant follicle > 30 mm in diameter, uterine edema > 2 (scale 0-3, being 0 absence of edema and 3 maximum edema), and soft, relaxed cervix on vaginal palpation.
After perineal disinfection [
26,
27], guarded endometrial biopsy was collected employing the double sleeve technique [
28,
29] using a biopsy forceps with a cutting surface of 4 x 15 mm (Equivet, Kruuse, Denmark). The tissues were immediately tapped onto a sterile slide for cytology and then transferred into a test tube containing 4% formaldehyde (Qemical, Quality Chemicals S.L., Spain) for fixation.
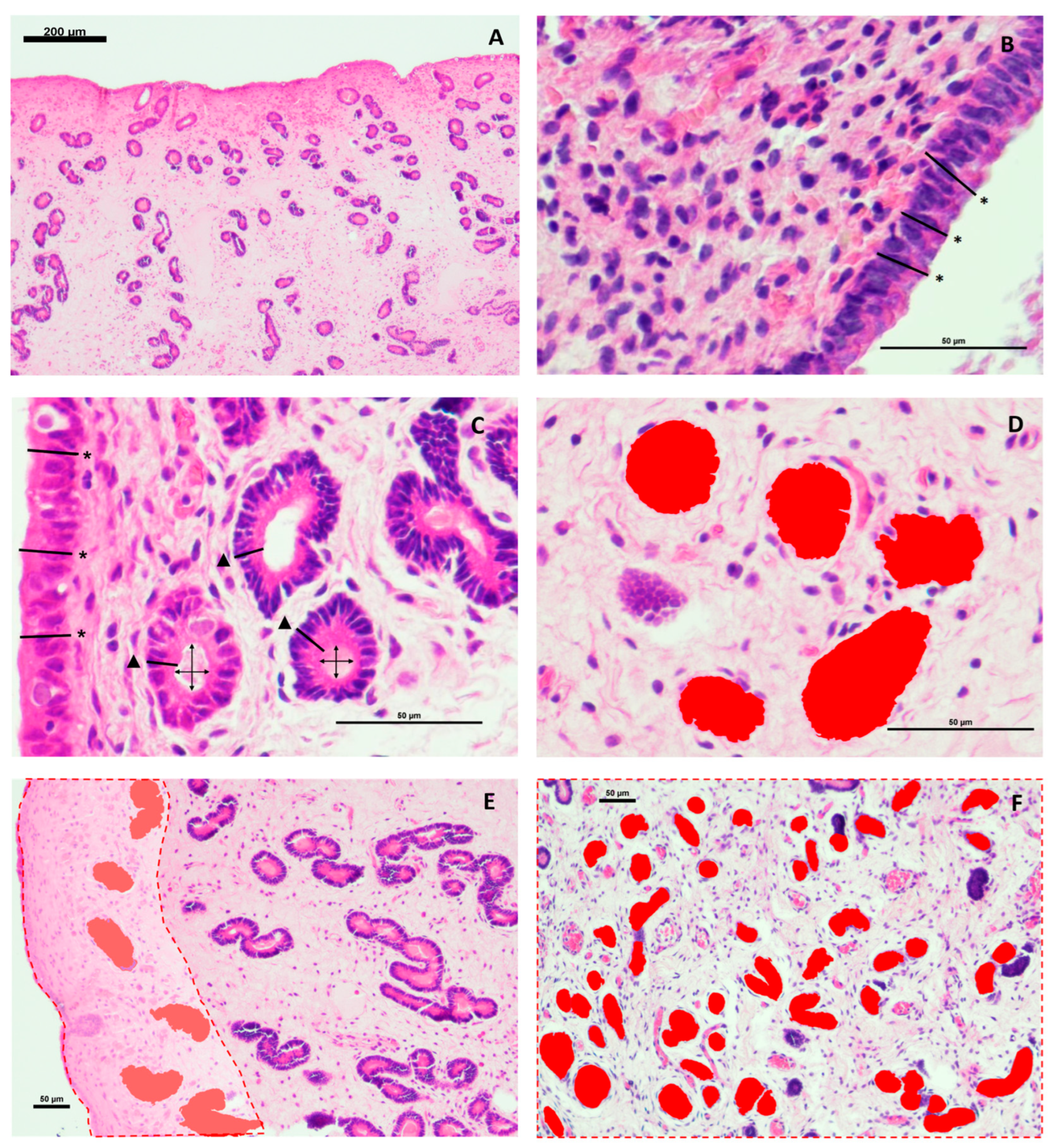
A total of 12 endometrial biopsies, 6 from jennies and 6 from mares, were routinely processed and sectioned into 4 μm tissue sections and stained with hematoxylin-eosin [
21,
30], for blind histopathological evaluation and later histomorphometric analyses (
Figure 1A). The parameters luminal (LE) and glandular (GE) epithelium height, glandular lumen diameter (LD), and glandular area (GA) were measured at 400x magnification, whereas the parameters numbver of glands (#G), and glandular tissue percentage (GT) were measured at 100x magnification. Each parameter was recorded separately for SC and SS.
2.3. Endometrial Histomorphometric Evaluation
The endometrial histomorphometry was evaluated using the NIS-Elements Basic Research software version 5.21.03 (Nikon, Tokyo, Japan) on images provided by a camera (DS-Fi3, Nikon, Tokyo, Japan) connected to an inverted microscope (ECLIPSE Ti1-U IVF, Nikon, Tokyo, Japan).
2.3.1. Measurement of Luminal Epithelium Height and Glandular Size Parameters
The height of the luminal epithelium (LE) was measured as the distance from the basement membrane to the apical edge of the epithelial cell [
19], at 400x magnification following the methodology described elsewhere [
19,
20,
22,
31] with modifications (
Figure 1B). The LE was calculated as the mean from 30 measures of each endometrium [
21]. The results were expressed in micrometers.
The height of the glandular epithelium (GE) was also measured as the distance from the basement membrane to the apical border of the epithelial cell, at 400x magnification [
19,
20,
22,
31] (
Figure 1C). For each stratum (glandular epithelium in the stratum compactum, GE-SC; and glandular epithelium in the stratum spongiosum, GE-SS) 30 glands were measured [
21]. The results were expressed in micrometers.
Glandular lumen diameter (LD) was calculated as the mean of two perpendicular diameters in circular section glands randomly selected [
20,
21,
22,
23] from the SC (LD-SC) and from the SS (LD-SS). Measures were taken at 400x magnification. This parameter was measured in all the circular glands found in SC and SS. To standardize the gland selection for the measures, glands were considered circular when the parameter “
circularity” was higher than 0.5.
Circularity was measured by automatically detecting the object area by the NIS-Br software and calculated as: Circularity = 4 x π x
Area /
Perimeter2. The tools
Measure Vertical and
Measure Horizontal were used to ensure exact 90-degree perpendicular measures (
Figure 1C). The results were expressed in micrometers.
Glandular area (GA) was calculated as the mean area of the glands measured separately for each stratum (glandular area in the stratum compactum, GA-SC; and glandular area in the stratum spongiosum, GA-SS), at 400x magnification. GA was individually quantified using the feature
Automated Measurement >
Auto detect, highlighting the selected areas with
Object Colors as shown in
Figure 1D. This tool is based on traditional computer vision algorithms, analyzing pixel intensity levels and variations in adjacent pixels to detect cell or gland boundaries. The results were expressed as µm
2.
2.3.2. Measurement of Glandular Density Parameters
Number of glands (#G) is a glandular density parameter that refers to the number of glands present in a microscopic field. It was measured separately for the SC and the SS (#G-SC, #G-SS), calculated as the mean of the number of glands in 10 fields per stratum at 100x magnification. Glands were quantified as described in previous studies [
17,
19,
31], with some modifications; using the
Object Count tool of NIS-BR software, disregarding the glands that were touching the image borders. Glands were recognized as individual structures when, regardless of their shape, their structure was delimited by glandular epithelium throughout their perimeter, and there was identifiable stromal tissue around them. The results were expressed as number of glands per field.
Glandular tissue percentage (GT) is a glandular density parameter representing the percentage of the endometrial tissue surface covered by glands. In this study, this parameter was evaluated using the methodology described in cattle by Wang et al. [
16] with some modifications. In brief, the area of the glands in the SC and SS was measured separately in 10 microscopic fields, and the corresponding endometrial tissue area was measured using the manual area tool (dashed lines in
Figure 1 E and F). GT was recorded for each microscopic field, stratum, and sample (GT-SC, GT-SS).
2.4. Statistical Analysis
All analyses were performed using the statistical package SPSS v29.0.1.0 (IBM Spain, Madrid, Spain). All histomorphometric parameters were compared between species (jenny vs. mare), parity status (maiden vs. foaling), and strata (SC vs. SS). Data distribution normality was assessed using the Kolmogorov–Smirnov test. The results were compared by one-way ANOVA test followed by post-hoc analysis using the Tukey test. Results were presented as mean ± SEM. The level of significance was set at P < 0.05.
3. Results
3.1. Breeding Soundness Evaluation
The results of the individual breeding soundness evaluation of the animals included in the study are displayed in
Table 1.
3.2. Histological Characteristics of the Endometrial Tissue
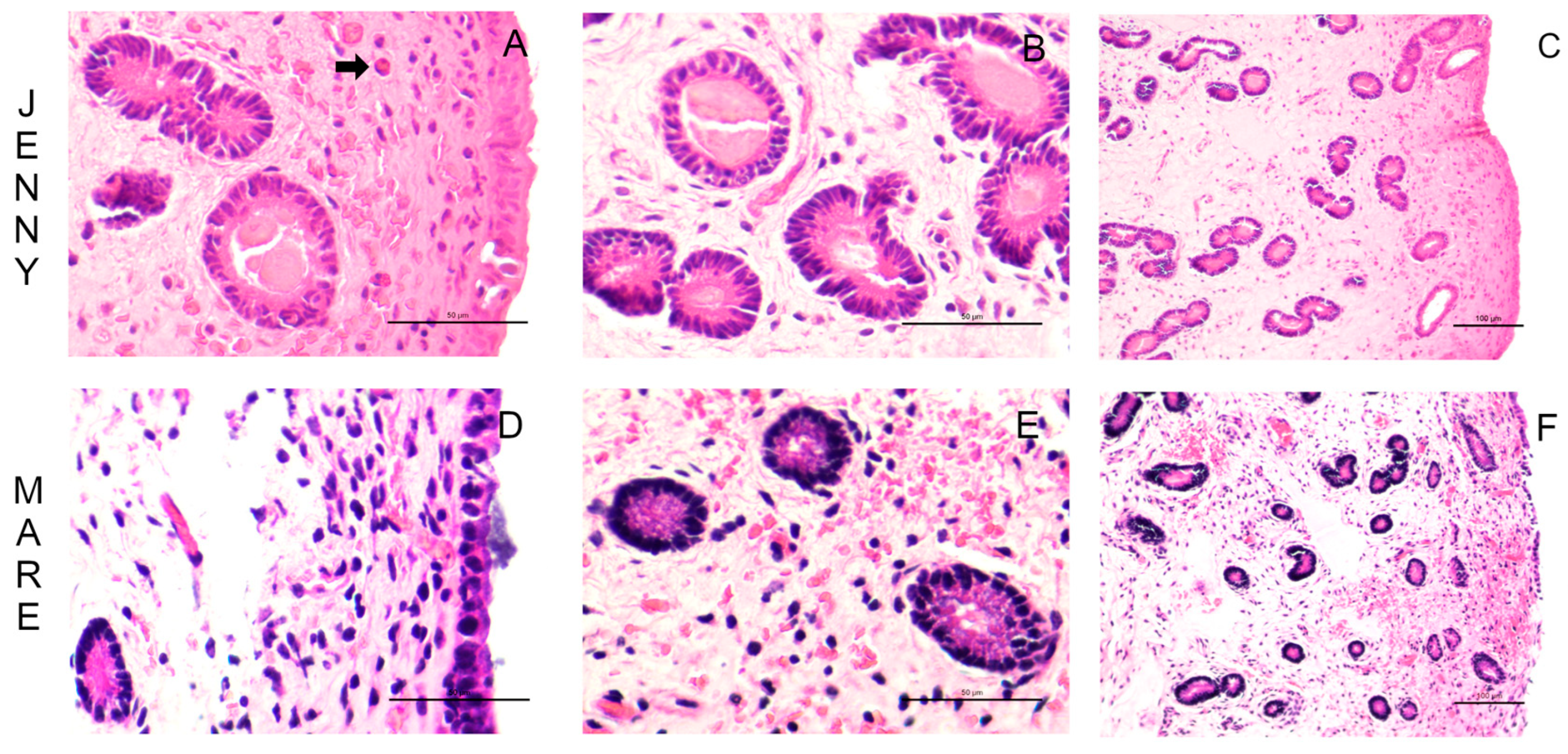
The histological characteristics of the endometrial tissue of mares and jennies are illustrated in
Figure 2. The luminal epithelium was observed under bright field microscopy (
Figure 2A and D), displaying simple cuboidal to columnar epithelium with varying pseudostratification in both species. The lamina propria exhibited estrus-induced stromal edema, and uterine glands showed variable tortuosity (
Figure 2B, C, E, F), apparently higher in jennies. Mild subepithelial inflammatory infiltrate was present, with a more pronounced proportion of eosinophilic infiltrates in jennies. These findings align with previous literature descriptions [
19,
21,
23,
32,
33,
34].
3.3. Endometrial Histomorphometric Evaluation
3.3.1. Results of Luminal Epithelium and Endometrial Glandular Size Parameters
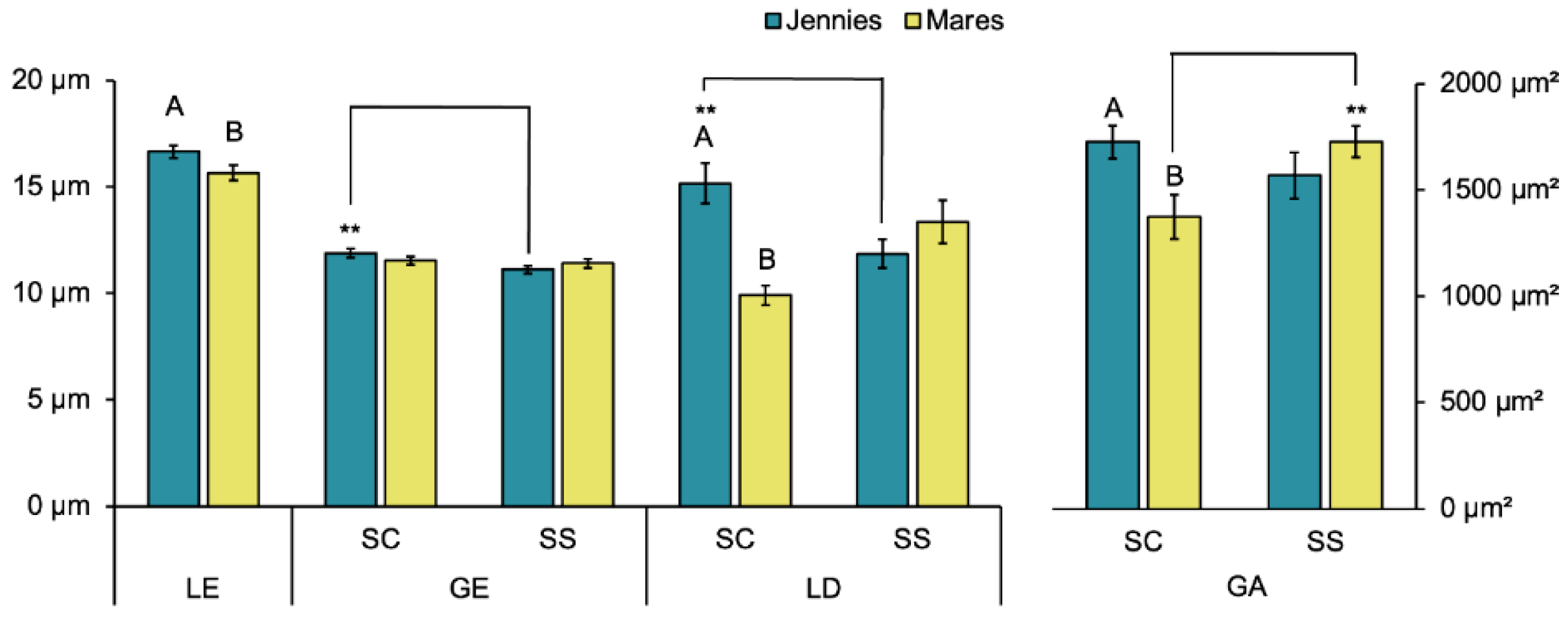
The height of the luminal epithelium (LE), glandular lumen diameter (LD) and glandular area (GA) showed significantly higher values in the stratum compactum (SC) in jennies than in mares (
P < 0.05,
P < 0.001 and
P < 0.05, respectively) (
Figure 3). Significant intraspecies differences (
P < 0.01) between
strata (SC vs. SS) were observed in certain parameters: in jennies, GE-SC and LD-SC were larger than their equivalents in SS; whereas in mares GA-SC was smaller than GA-SS.
The differences among the glandular size parameters grouped according to species and parity status were also compared (
Table 2). In both species, foaling animals presented higher values (
P < 0.001) for LE, GE and GA-SC than maiden. Maiden and foaling jennies exhibited wider (
P < 0.001) LD-SC than mares. There were also significant differences (
P < 0.01) between
strata (SC vs SS) for some parameters: in maiden jennies, GE-SC was taller than GE-SS; in foaling jennies, LD-SC was wider than LD-SS. Foaling mares exhibited narrower LD-SC than LD-SS; maiden mares had smaller GA-SC than GA-SS.
3.3.2. Results of Glandular Density Parameters
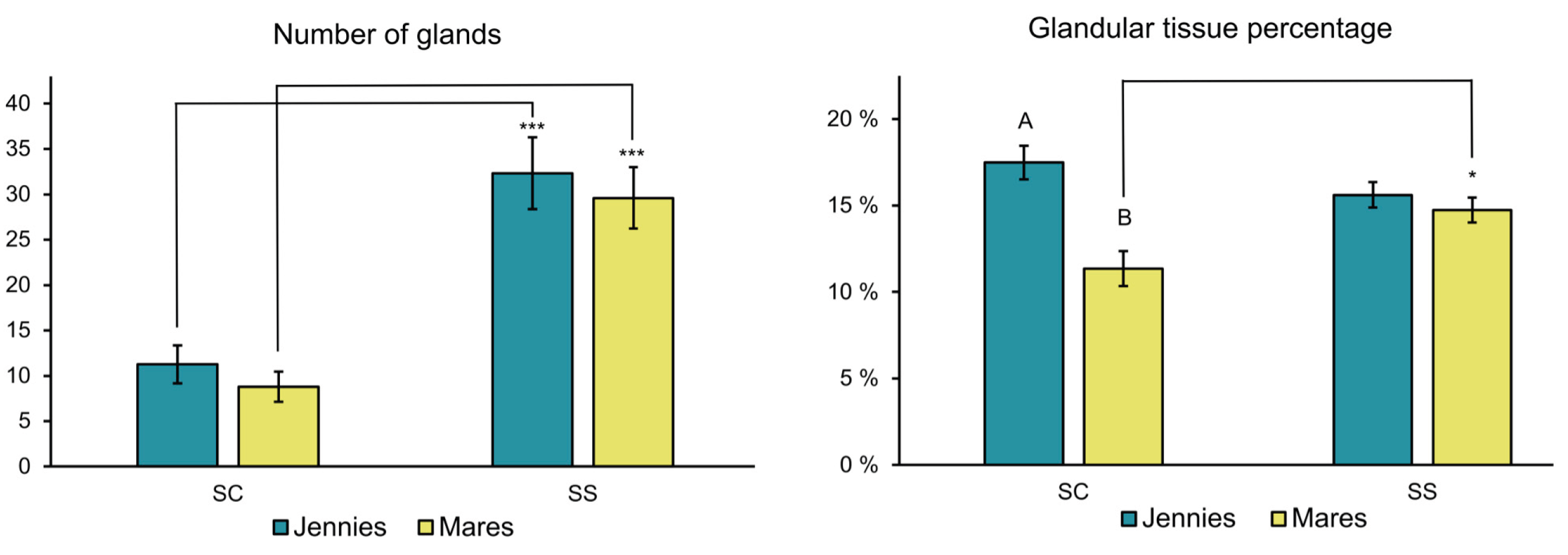
Glandular density parameters revealed further significant differences in glandular tissue distribution between species (
Figure 4). The quantification of glands (#G) was similar in both species (
P > 0.05), while glandular tissue percentage in the SC (GT-SC) was significantly higher in jennies than in mares (
P < 0.01). In mares, GT-SS proportionally covered a greater area (
P < 0.05) than GT-SC.
The comparison of density parameters grouped within species and parity status was also evaluated (
Table 3). The number of glands (#G) did not significantly differ (
P > 0.05) among species or parity status. In foaling jennies, GT-SC covered a larger (
P < 0.001) area compared to mares (both foaling and maiden). Additionally, GT-SC in maiden jennies was significantly larger (
P < 0.001) than in their mare counterparts. Regarding differences observed between
strata, in both species, there were fewer #G (
P < 0.001) in the SC. No differences between
strata were observed for GT (
P > 0.05).
Although #G did not show significant differences (P > 0.05) among species or parity status, GT differed between jennies and mares in the SC. Accordingly to glandular size parameters, the percentage of glandular tissue covered a greater area (P < 0.01) of the SC of the endometrium in jennies than in mares when comparing species within the same parity status.
4. Discussion
This study presents the first histomorphometric comparison between the endometrium of jennies and mares, providing objective differences among the endometrial features of these species. This novel characterization of the endometrial tissue of jennies provides valuable insights into their unique physiological traits that could play a role in the different response after artificial insemination in these species [
7,
9].
Our study revealed significant differences between jennies and mares glandular size parameters. During estrus, the luminal epithelium (LE) was significantly taller in jennies than in mares. The glandular epithelium (GE) of the glands in the stratum compactum (SC) was also taller in maiden jennies than maiden mares. The observed increase in epithelial height (luminal and glandular) in jennies compared to mares suggests a heightened level of glandular activity [
32,
35]. This fact was supported by the finding that LE and GE were also taller in animals that had foaled on that year than in maiden animals in both species. Glandular lumen diameter and area (LD-SC and GA-SC) were also larger in jennies compared to mares, reinforcing the idea of increased glandular activity, with the possible subsequent secretion accumulation in this species. Exuberant secretion may enlarge glands and impair their clearance, leading to glandular secretion accumulation. This build-up fluid could trap inflammatory debris, which would promote further mucus production and bacterial growth, creating a self-sustaining cycle of inflammation [
36].
The presence of larger glands in jennies and foaling animals, as indicated by increased LD-SC and GA-SC, may result in an environment rich in uterine secretion and inflammatory mediators, which could facilitate bacterial colonization and increase the risk of uterine contamination [
37]. Previous studies have shown that pathogenic microorganisms causing endometritis in mares are able to reside in the epithelia cells and within the endometrial glands [
38,
39,
40,
41]. Their presence in these locations has been proposed to reflect distinct survival mechanism: dormant bacteria inside the cells may evade detection until activated by specific triggers [
38]; and within the glandular lumen, phagocytic binding may be impaired [
41] while accessing the nutrient-rich glandular fluid [
39]. Previous studies have shown that larger glandular structures are more prevalent in older mares with poor reproductive outcomes [
20], and larger glands are also related to greater susceptibility to a more exacerbated post-breeding inflammation [
21,
22]. These findings agree with the results found in other species like the dog; bitches with cystic endometrial hyperplasia show glandular enlargement and mucus retention, creating favorable conditions for bacterial growth and predisposing these animals to suffer from pyometra [
42]. According to these findings, wider glands (as those found in foaling females and jennies in particular) could be more predisposed to heightened inflammatory responses. This hypothesis would explain some reproductive differences found in jennies, such as higher basal polymorphonuclear leukocytes (PMNs) counts in endometrial samples [
13,
43,
44], a more exacerbated post-breeding inflammatory response [
12,
45], and lower pregnancy rates after AI [
7,
9,
46].
In addition to the size of luminal epithelium and glandular structures, glandular density across the stromal layers shows a wider picture of the endometrium. In our study, glandular density was assessed by counting the number of glands per field (#G) and calculating the percentage of tissue covered by glands (GT). The latter incorporated the parameter gland area using a novel methodology for mares, adapted from what has been applied in other species [
16]. This approach was chosen for its accuracy and ease of implementation in comparison to previous analogue approaches [
17,
19,
20,
21,
31]. Our findings revealed that, in jennies, GT covered a larger area within the SC, while the #G did not differ significantly between species. The fact that GT covered a wider area in jennies and #G was similar for both species, stands by the finding that jennies display wider glands which accumulate more secretion in the glandular lumen, which may contribute to bacteria colonization and heightened inflammatory response, as previously discussed [
36,
38,
41]. Previous studies conducted in mares found that an increased number of glands in mares is associated with a higher susceptibility to endometritis [
21], while higher glandular density has been linked to intrauterine fluid accumulation [
22]. These findings reaffirm that the higher amount of glandular tissue found in jennies might be linked to a more exacerbated post-breeding inflammation. In contrast, a study evaluating the histomorphometric features of older mares with poor reproductive outcomes reported a lower number of glands, concluding that insufficient functional tissue could lead to early embryo death [
20]. However, this study did not take into consideration the percentage of glandular tissue these glands represented. These findings emphasize the need for future studies to further investigate glandular density and its potential implications in subfertile mares and jennies.
Remarkably, our results showed that significant differences in glandular size and density between jennies and mares were almost exclusively limited to the stratum compactum (SC), with minimal variations found in the deeper stratum spongiosum (SS). The SC seems to be more sensitive to environmental and physiological changes [
47,
48], and probably more noteworthy from a diagnostic point of view. These results highlight the importance of differentiating between
strata when performing histomorphometric analyses, as significant differences may be overlooked without this separation.
In order to identify the physiological interspecific differences in the endometrium, this study only included reproductively healthy jennies and mares, meeting the criteria of i) no endometrial inflammation (PMNs < 5%) and ii) low degree of fibrotic endometrial degeneration with scores I or IIa according to the KDGS [
33]. Future studies including samples showing signs of inflammation or fibrotic endometrial degeneration (KDGS IIb or III) could deepen our understanding on the reproductive adaptations of these closely related species and how endometrial status might affect reproductive outcomes.
5. Conclusions
In summary, the comparison of histomorphometry characteristics between reproductively sound jennies and mares revealed significant endometrial differences, with jennies displaying larger luminal epithelium, glandular size, and density compared to mares. These anatomical differences might play a role in the more exacerbated uterine response after AI in jennies. Further research is needed to understand the physiological mechanism underlying the uterine response in jennies.
Author Contributions
Conceptualization, P. Vallejo-Soto and I. Ortiz; methodology, P. Vallejo-Soto and I. Ortiz; formal analysis, P. Vallejo-Soto and I. Ortiz; investigation, P. Vallejo-Soto, I. Ortiz, R. Herrera-García, C. Álvarez-Delgado, J. Gómez-Laguna, A. de Santiago, M. Manrique, A. Gonzalez Ariza, and J.M. Leon Jurado; resources, J. Gómez-Laguna, A. de Santiago, M. Manrique, A. Gonzalez Ariza, J.M. Leon Jurado, M. Hidalgo, and J. Dorado; data curation, P. Vallejo-Soto and R. Herrera-García; writing—original draft preparation, P. Vallejo-Soto and I. Ortiz; writing—review and editing, P. Vallejo-Soto, I. Ortiz, C. Álvarez-Delgado, J. Gómez-Laguna, M. Hidalgo, and J. Dorado; visualization, P. Vallejo-Soto, I. Ortiz, M. Hidalgo, and J. Dorado; supervision, I. Ortiz, M. Hidalgo, and J. Dorado; project administration, M. Hidalgo and J. Dorado; funding acquisition, M. Hidalgo and J. Dorado. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by project grant UCO-13811332-R from FEDER Andalucía 2014-2020 and project grant PID2020-116090RB-100 (Ministerio de Ciencia e Innovación, Spain), which also supported P. Vallejo-Soto and R. Herrera-García.
Institutional Review Board Statement
The animal study protocol was approved by the Ethical Committee for Animal Experimentation of the Universidad de Córdoba (No. 2024PI/18) and are in accordance with Spanish (RD 53/2013) and European (Directive 2010/63/EU) legislation on the protection of animals used for scientific purposes.
Data Availability Statement
Acknowledgments
The authors would like to thank the Military Horse Breeding Centre of Ecija of the Spanish Army and the Provincial Agropecuary Centre of Córdoba for allowing access to their herds. Preliminary results of this study were presented at the 17th International Conference of the AERA (Asociación Española de Reproducción Animal), Madrid, Spain, 17-19 October 2024.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| #G |
Number of glands |
| AI |
Artificial Insemination |
| ARTs |
Assisted reproductive technologies |
| GA |
Glandular area |
| GE |
Glandular epithelium |
| GT |
Glandular tissue percentage |
| KDGS |
Kenney and Doig biopsy grading system |
| LD |
Lumen diameter |
| LE |
Luminal epithelium |
| SC |
Stratum compactum |
| SS |
Stratum spongiosum |
References
- Food and Agriculture Organization (FAO). Available online: Domestic Animal Diversity Information System (DAD-IS) https://www.fao.org/dad-is/en/ (accessed 13th March 2025).
- Camillo, F.; Rota, A.; Biagini, L.; Tesi, M.; Fanelli, D.; Panzani, D. The Current Situation and Trend of Donkey Industry in Europe. J Equine Vet Sci 2018, 65, 44–9. [Google Scholar] [CrossRef]
- Hedrick, P.W.; Garcia-Dorado, A. Understanding Inbreeding Depression, Purging, and Genetic Rescue. Trends Ecol Evol 2016, 31, 940–52. [Google Scholar] [CrossRef]
- Hildebrandt, T.B.; Hermes, R.; Goeritz, F.; Appeltant, R.; Colleoni, S.; de Mori, B.; Diecke, S.; Drukker, M.; Galli, C.; Hayashi, K.; Lazzari, G.; Loi, P.; Payne, J.; Renfree, M.; Seet, S.; Stejskal, J.; Swegen, A.; Williams, S.A.; Zainuddin, Z.Z.; Holtze, S. The ART of bringing extinction to a freeze – History and future of species conservation, exemplified by rhinos. Theriogenology 2021, 169, 76–88. [Google Scholar] [CrossRef]
- De Oliveira, J.V.; Oliveira, P.V.L.F.; e Oña, C.M.M.; Guasti, P.N.; Monteiro, G.A.; da Silva, Y.F.R.S.; de Mello Papa, P.; Alvarenga, M.A.; Dell’Aqua Junior, J.A.; Ozanam Papa, F. Strategies to improve the fertility of fresh and frozen donkey semen. Theriogenology 2016, 85, 1267–1273. [Google Scholar] [CrossRef]
- Panzani, D.; Fanelli, D.; Camillo, F.; Rota, A. Embryo technologies in donkeys (Equus Asinus). Theriogenology 2020, 156, 130–7. [Google Scholar] [CrossRef]
- Fanelli, D.; Moroni, R.; Bocci, C.; Camillo, F.; Rota, A.; Panzani, D. Interspecific and Intraspecific Artificial Insemination in Domestic Equids. Animals 2023, 13:582. [CrossRef]
- Hidalgo, M. Sperm vitrification in horses and donkeys. J Equine Vet Sci 2025, 145, 105340. [Google Scholar] [CrossRef]
- Vidament, M.; Vincent, P.; Martin, F.X.; Magistrini, M.; Blesbois, E. Differences in ability of jennies and mares to conceive with cooled and frozen semen containing glycerol or not. Anim Reprod Sci 2009, 112, 22–35. [Google Scholar] [CrossRef]
- Rota, A.; Panzani, D.; Sabatini, C.; Camillo, F. Donkey jack (Equus asinus) semen cryopreservation: Studies of seminal parameters, post breeding inflammatory response, and fertility in donkey jennies. Theriogenology 2012, 78, 1846–1854. [Google Scholar] [CrossRef]
- Vilés, K.; Rabanal, R.; Rodríguez-Prado, M.; Miró, J. Effect of ketoprofen treatment on the uterine inflammatory response after AI of jennies with frozen semen. Theriogenology 2013, 79, 1019–1026. [Google Scholar] [CrossRef]
- Miró, J.; Papas, M. Post–Artificial Insemination Endometrial Inflammation and Its Control in Donkeys. J Equine Vet Sci 2018, 65, 38–43. [Google Scholar] [CrossRef]
- Canisso, I.F.; Panzani, D.; Miró, J.; Ellerbrock, R.E. Key Aspects of Donkey and Mule Reproduction. Veterinary Clinics of North America - Equine Practice 2019, 35, 607–642. [Google Scholar] [CrossRef]
- Abdelnaeim, M. Morphological Characteristics of the Donkey (Equus asinus) Uterus during Estrus: Light, Scanning and Transmission Electron Microscopic Study. Journal of Agricultural and Veterinary Sciences, Qassim University 2008, 2, 45–57. [Google Scholar]
- Alfradique, V.A.P.; Netto, D.L.S.; Alves, S.V.P.; Machado, A.F.; Novae,s C.M.; Penitente-Filho, J.M.; Machado-Neves, M.; Lopes, M.S.; Guimaraes, S.E.F The impact of FSH stimulation and age on the ovarian and uterine traits and histomorphometry of prepubertal gilts. Domest Anim Endocrinol 2023, 83, 106786. [CrossRef]
- Wang, C.K.; Robinson, R.S.; Flint, A.P.F.; Mann, G.E. Quantitative analysis of changes in endometrial gland morphology during the bovine oestrous cycle and their association with progesterone levels. Reproduction 2007, 134, 365–371. [Google Scholar] [CrossRef]
- Lefranc, A.C.; Allen, W.R. ; Influence of breed and oestrous cycle on endometrial gland surface density in the mare. Equine Vet J 2007, 39, 506–510. [Google Scholar] [CrossRef]
- Abdoon, A.S.S.; Soliman, S.S.; Nagy, A.M. Uterotubal junction of the bovine (Bos taurus) versus the dromedary camel (Camelus dromedarius): Histology and histomorphometry. Reproduction in Domestic Animals 2024, 59, e14665. [Google Scholar] [CrossRef]
- Mansour, G.D.; Ferreira, A.M.R.; Fernandes, F.T.; Henry, M. Histomorphometry of epithelial structures of the mare’s endometrium. Revista Brasileira de Ciência Veterinária 2004, 11, 44–48. [Google Scholar] [CrossRef]
- Dubrovskaya, A.B.; Lebedeva, L.F.; Schukis, K.A. Comparative histomorphological characteristics of the endometrium of young and aged mares in estrus and diestrus. IOP Conf Ser Earth Environ Sci 2019, 341, 012067. [Google Scholar] [CrossRef]
- Herrera, M.; Herrera, J.M.; Cantatore, S.; Aguilar, J.; Felipe, A.; Fumuso, E. Comparative histomorphological study of endometrium in mares. Journal of Veterinary Medicine Series C: Anatomia Histologia Embryologia 2018, 47, 153–158. [Google Scholar] [CrossRef]
- Rasch, K.; Schoon, H.A.; Sieme, H.; Klug, E. Histomorphological endometrial status and influence of oxytocin on the uterine drainage and pregnancy rate in mares. Equine Vet J 1996, 28, 455–460. [Google Scholar] [CrossRef]
- Herrera, M.F.; Otermin, M.; Herrera, J.M.; Simoy, M.V.; Bianchi, C.P.; Aguilar, J.J.; Fumuso, E.A. Effect of Mycobacterium cell wall fraction on endometrial histomorphometry of mares resistant and susceptible to persistent breeding-induced endometritis. Theriogenology 2020, 156, 2–10. [Google Scholar] [CrossRef]
- Love, C.C. Evaluation of reproductive efficiency. In Equine breeding management and artificial insemination, 2nd ed.; Samper J.C. Publisher: Saunders: Philadelphia, USA, 2009; pp. 289–94. [Google Scholar] [CrossRef]
- Card, C. Post-breeding inflammation and endometrial cytology in mares. Theriogenology 2005, 64, 580–588. [Google Scholar] [CrossRef]
- Hinrichs, K.; Cummings, M.R.; Sertich, P.L.; Kenney, R.M. Clinical significance of aerobic bacterial flora of the uterus, vagina, vestibule, and clitoral fossa of clinically normal mares. J Am Vet Med Assoc 1988, 193, 72–75. [Google Scholar] [CrossRef]
- Dascanio, J.J. Uterine Lavage. In Equine Reproductive Procedures, 2nd ed.; Dascanio, J.J., McCue, P., Eds.; Publisher: John Wiley & Sons: Hoboken, USA, 2021; pp. 127–130. [Google Scholar] [CrossRef]
- Aguilar, J.; Hanks, M.; Shaw, D.J.; Else, R.; Watson, E. Importance of using guarded techniques for the preparation of endometrial cytology smears in mares. Theriogenology 2006, 66, 423–430. [Google Scholar] [CrossRef]
- Rasmussen, C.D.; Petersen, M.R.; Bojesen, A.M.; Pedersen, H.G.; Lehn-Jensen, H.; Christoffersen, M. Equine Infectious Endometritis—Clinical and Subclinical Cases. J Equine Vet Sci 2015, 35, 95–104. [Google Scholar] [CrossRef]
- Álvarez-Delgado, C.; Ruedas-Torres, I.; Sánchez-Carvajal, J.M.; Priego-Capote, F.; Castillo-Peinado, L.; Galán-Relaño, Á.; Moreno, P.J.; Díaz-Bueno, E.; Lozano-Buenestado, B.; Rodríguez-Gómez, I.M.; Carrasco, L.; Pallarés, F.J.; Gómez-Laguna, J. Impact of supplementation with dihydroxylated vitamin D3 on performance parameters and gut health in weaned Iberian piglets under indoor/outdoor conditions. Porcine Health Manag 2023, 9, 15. [Google Scholar] [CrossRef]
- Leishman, D.; Miller, R.B.; Doig, P.A. A Quantitative Study of the Histological Morphology of the Endometrium of Normal and Barren Mares. Can J Comp Med 1982, 46, 17–20. [Google Scholar]
- Kenney, R.M. Cyclic and pathologic changes of the mare endometrium as detected by biopsy, with a note on early embryonic death. J Am Vet Med Assoc 1978, 172, 242–262. [Google Scholar] [CrossRef]
- Kenney, R.M.; Doig, P.A. Equine endometrial biopsy. Current Therapy in Theriogenology 1986, 2, 723–729. [Google Scholar]
- Vilés, K.; Rabanal, R.; Rodríguez-Prado, M.; Miró, J. Influence of seminal plasma on leucocyte migration and amount of COX-2 protein in the jenny endometrium after insemination with frozen-thawed semen. Anim Reprod Sci 2013, 143, 57–63. [Google Scholar] [CrossRef]
- Van Camp, S.D. Endometrial biopsy of the mare. A review and update. Vet Clin North Am Equine Pract 1988, 4, 229–245. [Google Scholar] [CrossRef]
- Causey, R.C. Mucus and the mare: How little we know. Theriogenology 2007, 68, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Troedsson, M.H.T.; Liu, I.K.M.; Thurmond, M. Function of uterine and blood-derived polymorphonuclear neutrophils in mares susceptible and resistant to chronic uterine infection: phagocytosis and chemotaxis. Biol Reprod 1993, 49, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.R.; Skive, B.; Christoffersen, M.; Lu, K.; Nielsen, J.M.; Troedsson, M.H.T.; Bojesen, A.M. Activation of persistent Streptococcus equi subspecies zooepidemicus in mares with subclinical endometritis. Vet Microbiol 2015, 179, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Ferris, R.A.; Mccue, P.M.; Borlee, G.I.; Glapa, K.E.; Martin, K.H.; Mangalea, M.R.; Hennet, M.L.; Wolfe, M.L.; Broeckling, C.D.; Borlee, B.R. Model of Chronic Equine Endometritis Involving a Pseudomonas aeruginosa Biofilm. Infection and Inmunity 2017, 85, e00332–17. [Google Scholar] [CrossRef]
- Skive, B.; Rohde, M.; Molinari, G.; Braunstein, T.H.; Bojesen, A.M. Streptococcus equi subsp. zooepidemicus invades and survives in epithelial cells. Front Cell Infect Microbiol 2017, 7, 465. [Google Scholar] [CrossRef]
- Köhne, M.; Hüsch, R.; Tönissen, A.; Schmidt, M.; Müsken, M.; Böttcher, D.; Hirnet, J.; Plötz, M.; Kittler, S.; Sieme, H. Isolation and characterization of bacteriophages specific to Streptococcus equi subspecies zooepidemicus and evaluation of efficacy ex vivo. Front Microbiol 2024, 15, 1448958. [Google Scholar] [CrossRef]
- Veiga, G.A.L.; Miziara, R.H.; Angrimani, D.S.R.; Papa, P.C.; Cogliati, B.; Vannucchi, C.I. Cystic endometrial hyperplasia-pyometra syndrome in bitches: Identification of hemodynamic, inflammatory, and cell proliferation changes. Biol Reprod 2017, 96, 58–69. [Google Scholar] [CrossRef]
- Radar-Chafirovitch, A.; Catarino, J.; Lourenço, L.; Payan-Carreira, R.; Ferreira-Dias, G.; Miró, J.; Quaresma, M.; Pires, M.A. Evaluation of Inflammatory Infiltrated in the Endometrium of the Jenny (Equus asinus). J Comp Pathol 2022, 191, 46. [Google Scholar] [CrossRef]
- Wu, R.; Yu, F.; Holyoak, G.R.; Gao, Y.; Zhu, Y.; Li, J. Use of the endometrial histopathology to improve diagnosis of donkeys with endometritis. Equine Vet Educ 2023, 36, 43–50. [Google Scholar] [CrossRef]
- Lopes Costa, L.M.; Sancler-Silva, Y.F.R.; Albino, M.V.; Fontes, C.S.; Texeira, C.S.; Freitas, M.S.; Kladt, L.V.; Schultz, E.B. Does the uterine inflammatory response differ between mares and donkey jennies inseminated with frozen donkey semen? J Equine Vet Sci 2023, 125, 104703. [Google Scholar] [CrossRef]
- Diaz-Jimenez, M.; Rota, A.; Dorado, J.; Consuegra, C.; Pereira, B.; Camillo, F.; Panzani, D.; Fanelli, D.; Tesi, M.; Monaco, D.; Hidalgo, M. First pregnancies in jennies with vitrified donkey semen using a new warming method. Animal 2021, 15, 100097. [Google Scholar] [CrossRef]
- Nielsen, J.M. Endometritis in the mare: A diagnostic study comparing cultures from swab and biopsy. Theriogenology 2005, 64, 510–518. [Google Scholar] [CrossRef]
- Rudolph, N.; Schoon, H.A.; Schöniger, S. Immunohistochemical characterisation of immune cells in fixed equine endometrial tissue: A diagnostic relevant method. Pferdeheilkunde 2017, 33, 524–537. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).