Submitted:
02 December 2025
Posted:
04 December 2025
You are already at the latest version
Abstract
This review provides an integrated analytical overview of the phenolic constituents of Solenostemma argel, with emphasis on extraction efficiency, structural characterization, and antioxidant-linked bioactivity. Because direct studies on argel phenolics remain limited, a broadened inclusion strategy was adopted. Studies were considered when phenolic-solubilizing solvents were used, when antioxidant-related biological effects (such as antidiabetic, anticancer, or neuroprotective activities) were evaluated, or when chromatographic and spectroscopic techniques applicable to phenolic analysis were employed. Comparative findings indicate that moderately polar solvents—particularly ethanol, methanol, and acetone—produce the highest phenolic yields, especially under ultrasound- or microwave-assisted extraction conditions. Reported variations in total phenolic content (TPC) primarily reflect methodological differences; however, higher TPC values consistently correlate with stronger antioxidant activity across assays. Advanced analytical platforms, including HPLC and NMR, provide the highest accuracy for qualitative and quantitative characterization of major phenolic classes. Overall, this expanded review synthesizes current evidence on phenolic profiling, extraction methodologies, analytical applicability, and antioxidant potential of S. argel, underscoring the plant’s promise as a rich and underexplored source of bioactive phenolic compounds.
Keywords:
1. Introduction
Novelty
2. Methodology
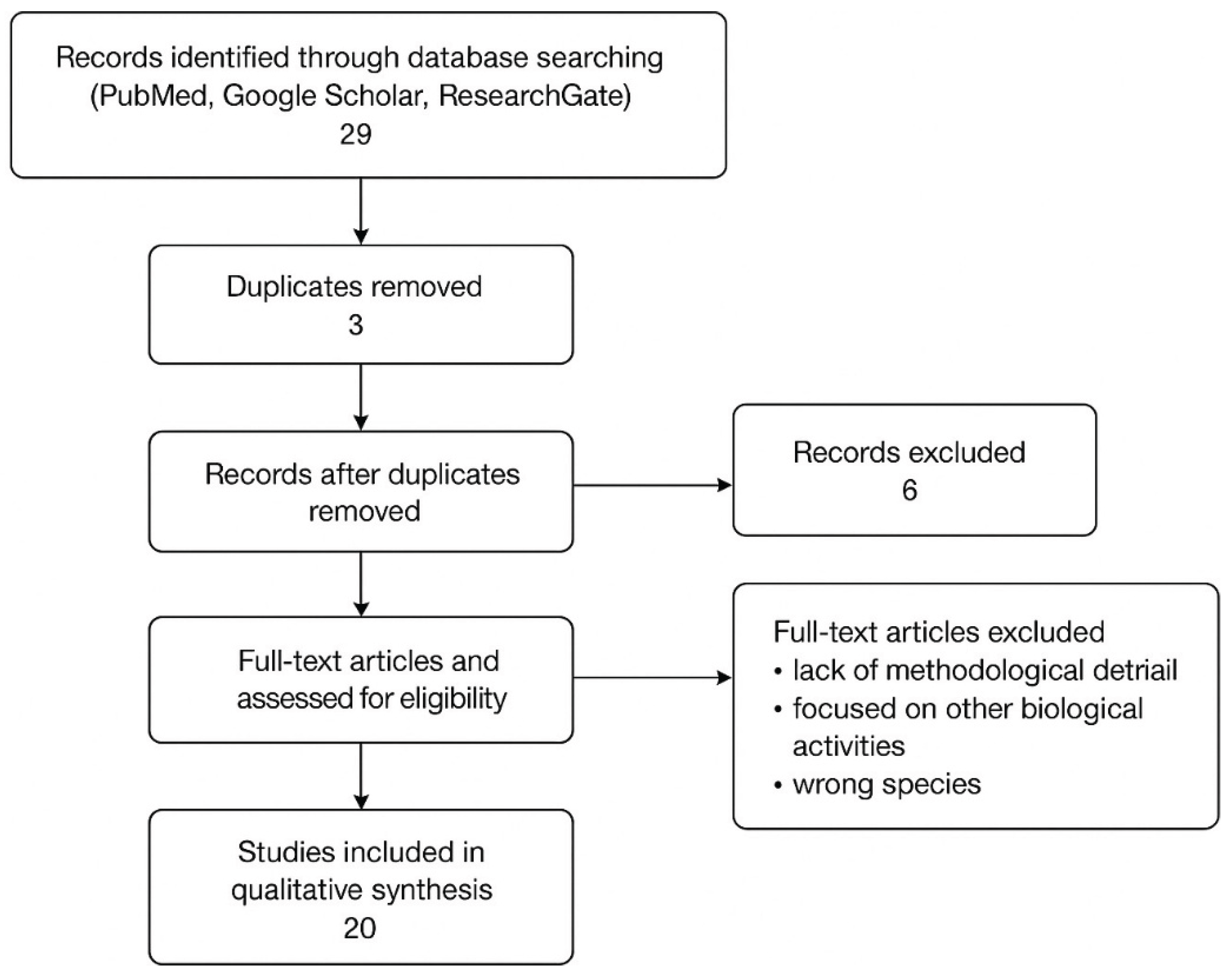
2.1 Study Identification
2.2. Inclusion Criteria
2.3. Comparative Analytical Strategy
3. Phenolic Profile of Solenostemma argel
3.1. Overview of Identified Phenolic Compounds
| Phenolic class | Representative compounds | Notes | References |
|---|---|---|---|
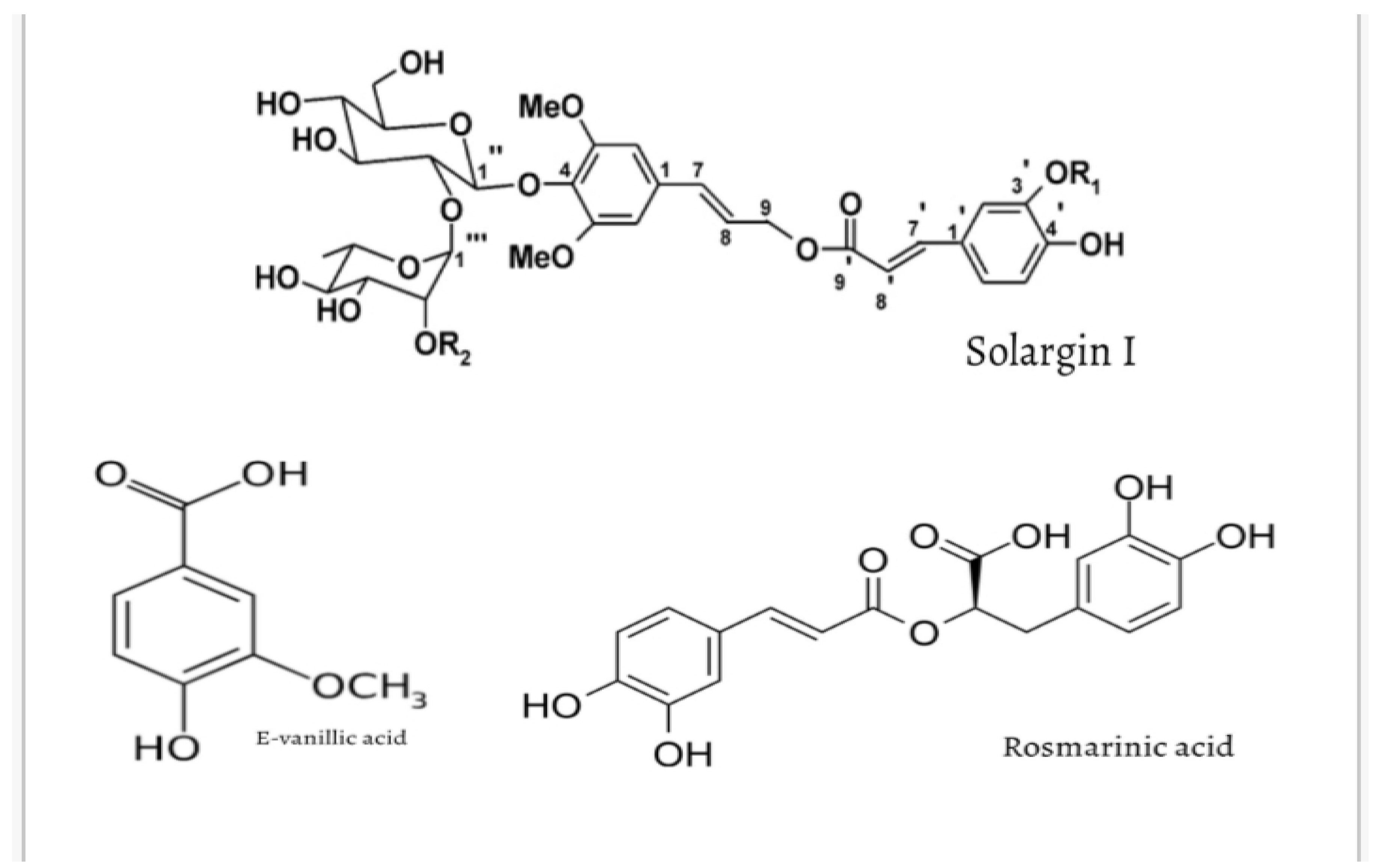
| Phenolic acids | Vanillic acid, E-vanillic acid, Ferulic acid, Chlorogenic acid, Gallic acid, Pyrogallol, Caffeic acid | Highest concentration reported for E-vanillic acid; abundant across leaves and aerial parts | [3,6] |
| Flavonoids | Quercetin, Hesperidin, Narengin, Rutin, Kaempferol, Kaempferol glycosides (3-O-glucoside, 3-O-arabinoside, 7-O-rhamnoside), Apigenin |
Major contributors to antioxidant activity; present in both free and glycosylated forms | [3,6] |
| Phenolic glycosides | Solargins I-IV | Newly identified; structurally unique glycosides | [3,5,7] |
| Other related metabolites | Rosmarinic acid, Coumarins, Cinnamic derivatives | Occur at moderate levels; contribute to total antioxidant capacity | [8] |
3.4. Distribution Across Plant Parts
3.4.1. Leaves
3.4.2. Aerial Parts
3.4.3. Roots and Fruit Peels
3.4.5. Comparative Interpretation
4. Integrated Comparative Analysis
4.1. Extraction Methods
| Extraction Technique | Solvent System | Typical Yield (%) | General Evaluation | References |
|---|---|---|---|---|
| Maceration (RT, days) | Methanol 80%, Ethanol 80% | 10–14% | Simple; long time; moderate yield | [6,15] |
| Soxhlet (60°C) | Methanol 95% | ~12% | Higher yield; risk of thermal degradation | [11] |
| Ultrasound-Assisted Extraction (UAE) | Ethanol 25–50% | — | Best antioxidant potency; good extraction selectivity | [9] |
| Microwave-Assisted Extraction (MAE) | EtOH/H₂O (various ratios) | 15–53% | Highest yields; short time; preserves thermolabile compounds | [9] |
| Hot Water Extraction | Water | Low | Mimics traditional use; weak extraction of phenolics | [10] |
4.2. Total Phenolic Content (TPC)
4.3 Antioxidant Activity
| Extraction Method / Solvent | TPC (mg GAE/g) | Antioxidant Results (DPPH / FRAP / ORAC) | Interpretation | References |
|---|---|---|---|---|
| MAE – EtOH/H₂O | 15–53% (yield) | High ORAC; strong radical scavenging | Best overall extraction efficiency | [9] |
| UAE – EtOH 25–50% | 63–72 | Up to 91% DPPH inhibition | Optimal balance of solvent + sonication | [9] |
| Acetone 80% | 81.45 | IC₅₀ = 48.87 µg/mL (DPPH); highest FRAP | Most effective solvent for phenolics | [10] |
| Ethanol 80% | 62.58 | Moderate DPPH & FRAP | Good extraction but lower bioactivity | [10] |
| Methanol extract | 24.53 | 13.7–44.8% (DPPH) | Lower phenolic levels → weaker activity | [19] |
| Ethyl acetate extract | — | 19–58% inhibition (DPPH) | Extracts moderately polar antioxidant compounds | [16] |
| Hot Water | 46.72 | Lowest activity | Poor extraction of phenolics | [10] |
| Crude extracts (general) | — | Up to 86,263 µmol TE/100g (ORAC) | Rich in mixed flavonoids | [18] |
4.4 Analytical Techniques
| Technique | Purpose in S. argel Studies | Strengths | Limitations | Relevance to Phenolic Analysis | References |
|---|---|---|---|---|---|
| HPLC | Quantification of phenolic acids and flavonoids; profiling of major compounds | High-resolution separation; reproducible quantification; widely validated for phenolics | Limited ability to identify unknowns without MS coupling | Strongly suitable for routine phenolic profiling and quantification | [3,5,6,20,21,22] |
| UPLC–MS | Identification of phenolic acids, flavonoids, and novel glycosides (e.g., Solargins I–IV) | High sensitivity; structural elucidation; detection of minor and complex metabolites | Requires advanced instrumentation and expertise | Essential for comprehensive phenolic profiling and discovering new phenolics | [3,5,6,20,21,22] |
| GC–MS | Mainly used for volatile oils and non-phenolic constituents | Excellent for volatile and semi-volatile compounds; rich spectral libraries | Phenolics generally non-volatile; requires derivatization | Indirect relevance—useful for evaluating analytical feasibility and method transferability | [3,9,22] |
| NMR | Structural confirmation of newly identified compounds (e.g., Solargins) | Definitive structural clarification; essential for new compound validation | Low sensitivity; requires high-purity isolates; time-consuming | Crucial for full structural characterization of isolated phenolic glycosides | [5,6] |
| UV–Vis Spectrophotometry | Used for general phenolic assays (TPC) | Simple; rapid; cost-effective | Non-specific; subject to interference | Useful for total phenolic estimation but not compound-level analysis | [7,9,10,19] |
5. Discussion
6. Limitations
7. Conclusions
Recommendations
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. Journal of Natural Products 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Ogbodo, J.O.; Agbo, C.P.; Echezona, A.C.; Ezike, T.C.; Emencheta, S.C.; Onyia, O.C.; Iguh, T.C.; Ihim, S.A. Therapeutic role of phenolic antioxidants in herbal medicine. In Upaganlawar, A.B.; Dhote, V.V., Raja, M.K.M. Health Benefits of Phenolic Antioxidants, Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2022; pp. 149–166. [Google Scholar]
- Abdel-Sattar, E.; El-Shiekh, R.A. A comprehensive review on Solenostemma argel (Del. ) Hayne: An Egyptian medicinal plant. Bulletin of the Faculty of Pharmacy, Cairo University 2024, 62, Article 3. [Google Scholar] [CrossRef]
- El-shiekh, R.A.; Al-Mahdy, D.A.; Mouneir, S.M.; Hifnawy, M.S.; Abdel-Sattar, E.A. Anti-obesity effect of argel (Solenostemma argel) on obese rats fed a high fat diet. Journal of Ethnopharmacology 2019, 238, 111893. [Google Scholar] [CrossRef] [PubMed]
- Kamel, M.S. Acylated phenolic glycosides from Solenostemma argel. Phytochemistry 2003, 62, 1247–1250. [Google Scholar] [CrossRef] [PubMed]
- Demmak, R.G.; Bordage, S.; Bensegueni, A.; Boutaghane, N.; Hennebelle, T.; Mokrani, E.H.; Sahpaz, S. Chemical constituents from Solenostemma argel and their cholinesterase inhibitory activity. Natural Product Sciences 2019, 25, 115–121. [Google Scholar] [CrossRef]
- Hassabelrasoul, H.; Moriguchi, M.; Kang, B.; Siribel, A.A.; Kuse, M. Isolation and identification of metabolites from ethyl acetate leaf extract of Solenostemma argel. Agriculture and Natural Resources 2021, 55, 757–763. [Google Scholar] [CrossRef]
- Azer, D.D.; Kahlil, A.F.; Hafez, A.A.; El-Hadidy, E.M. Hepato effect of argel herb (Solenostemma argel) against carbon tetrachloride-induced liver damage in albino rats. International Journal of Family Studies, Food Science and Nutrition Health 2021, 4, 142–161. [Google Scholar] [CrossRef]
- Ahmed, I.A.M.; et al. Optimization of ultrasound-assisted extraction of phenolic compounds and antioxidant activity from argel (Solenostemma argel Hayne) leaves using response surface methodology (RSM). Journal of Food Science and Technology 2020, 57, 3071–3080. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.A.; Gaafar, A.A.; Salama, Z.A.; El Baz, F.K. Anti-inflammatory and antioxidant activity of Solenostemma argel extract. International Journal of Pharmacognosy and Phytochemical Research 2015, 7, 635–641. [Google Scholar]
- Abouzaid, O.A.R.; Mansour, S.Z.; Sabbah, F. Evaluation of the antitumor activity of Solenostemma argel in the treatment of lung carcinoma induced in rats. Benha Veterinary Medical Journal 2018, 35, 178–189. [Google Scholar] [CrossRef]
- Hamadnalla, H.M.Y.; El-Jack, M.M. Phytochemical screening and antibacterial activity of Solenostemma argel: A medicinal plant. Acta Scientific Agriculture 2019, 3, 2–4. [Google Scholar]
- Farah, A.A.; Ahmed, E.H. Beneficial antibacterial, antifungal and anti-insecticidal effects of ethanolic extract of Solenostemma argel leaves. Mediterranean Journal of Biosciences 2016, 1, 184–191. [Google Scholar]
- Taha, L.E.; Bakhit, S.M.A.; Al-Sa’aidi, J.A.A.; Uro, A.B. The anti-hyperglycemic effect of Solenostemma argel compared with Glibenclamide. Al-Qadisiya Journal of Veterinary Medical Sciences 2014, 13, 113–117. [Google Scholar]
- Taj Al-Deen, A.; Al-Naqeb, G. Hypoglycemic effect and in vitro antioxidant activity of methanolic extract from argel (Solenostemma argel) plant. International Journal of Herbal Medicine 2014, 2, 128–131. [Google Scholar]
- Abdel-Motaal, F.F.; Maher, Z.M.; Ibrahim, S.F.; El-Mleeh, A.; Behery, M.; Metwally, A.A. Comparative studies on the antioxidant, antifungal, and wound healing activities of Solenostemma argel ethyl acetate and methanolic extracts. Applied Sciences 2022, 12, 4121. [Google Scholar] [CrossRef]
- El-Zayat, M.M.; Eraqi, M.M.; Alfaiz, F.A.; Elshaer, M.M. Antibacterial and antioxidant potential of some Egyptian medicinal plants used in traditional medicine. Journal of King Saud University–Science 2021, 33, 101466. [Google Scholar] [CrossRef]
- Ounaissi, K.; Pertuit, D.; Mitaine-Offer, A.-C.; Miyamoto, T.; Tanaka, C.; Delemasure, S.; Dutartre, P.; Smati, D.; Lacaille-Dubois, M.-A. New pregnane and phenolic glycosides from Solenostemma argel. Fitoterapia 2016, 114, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Maad, A.H.; Al-Gamli, A.H.; Shamarekh, K.Sh.; Refat, M.; Shayoub, M.E. Antiproliferative and apoptotic effects of Solenostemma argel leaf extracts on colon cancer cell line HCT-116. Biomedical & Pharmacology Journal 2024, 17, 1987–1996. [Google Scholar] [CrossRef]
- Abd Alhady, M.R.; Hegazi, G.A.; Abo El-Fadl, R.E.; Desoukey, S.Y. Biosynthetical capacity of kaempferol from in vitro produced argel (Solenostemma argel) callus. Research Journal of Applied Biotechnology 2016, Special Issue.
- El-Beltagi, H.S.; Abdel-Mobdy, Y.E.; Abdel-Rahim, E. Toxicological influences of cyfluthrin attenuated by Solenostemma argel extracts on carbohydrate metabolism of male albino rats. Fresenius Environmental Bulletin 2017, 26, 1673–1681. [Google Scholar]
- Soliman, M.S.M.; Abdella, A.; Khidr, Y.A.; Osman, H.G.O.; Aladadh, M.A.; Elsanhoty, R.M. Pharmacological activities and characterization of phenolic and flavonoid compounds in Solenostemma argel extract. Research Square 2022. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




