Submitted:
26 November 2025
Posted:
26 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
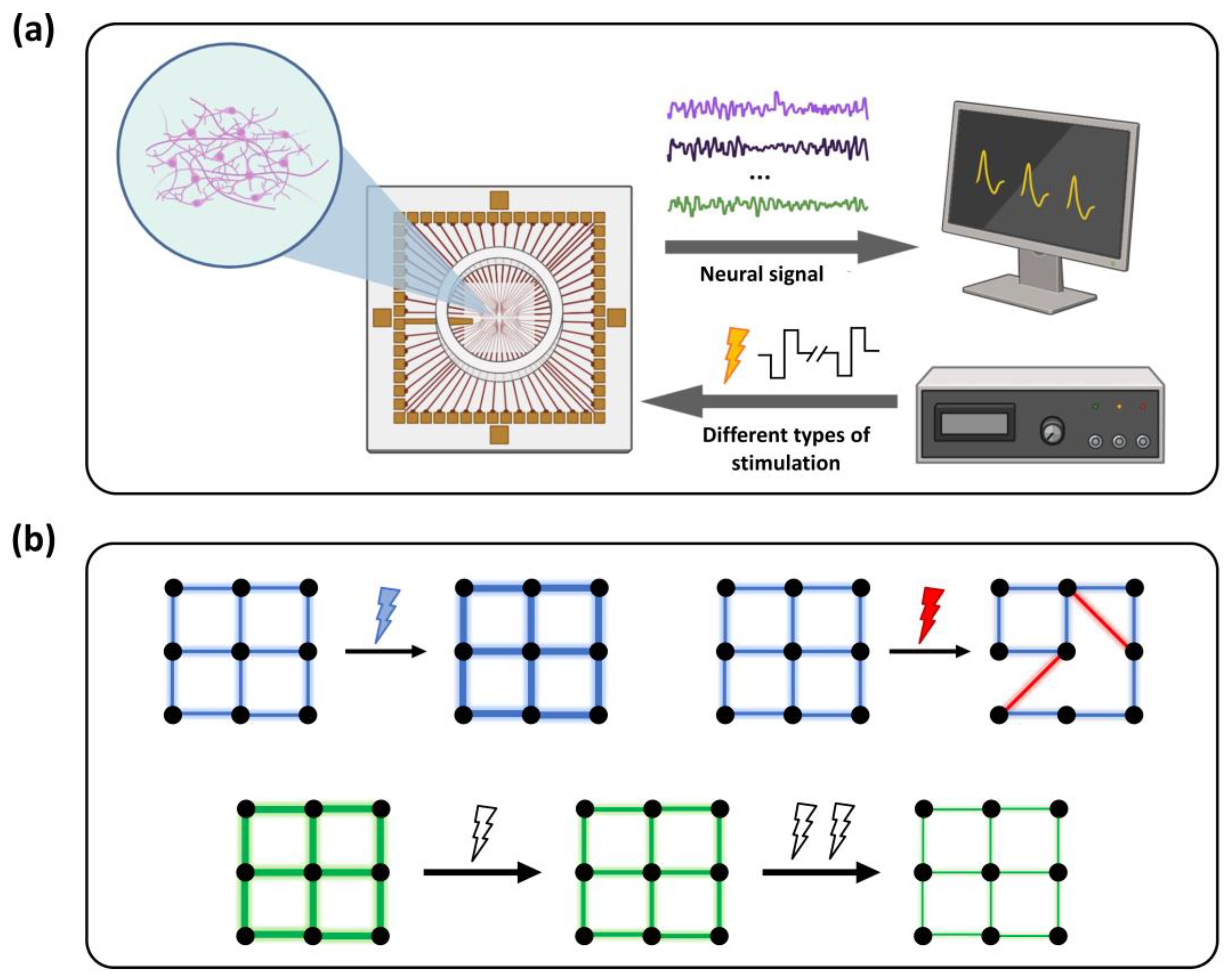
2. Materials and Methods
2.1. Reagents and Apparatus
2.2. Fabrication of In Vitro Microelectrode Arrays and Neuron Cultivation
2.3. Modulation of Network Connectivity Induced by Electrical Stimulation at Different Frequencies
2.4. Modulation of Neuronal Firing Patterns by Rhythmic Stimulation
2.5. Electrophysiological Analysis
3. Results
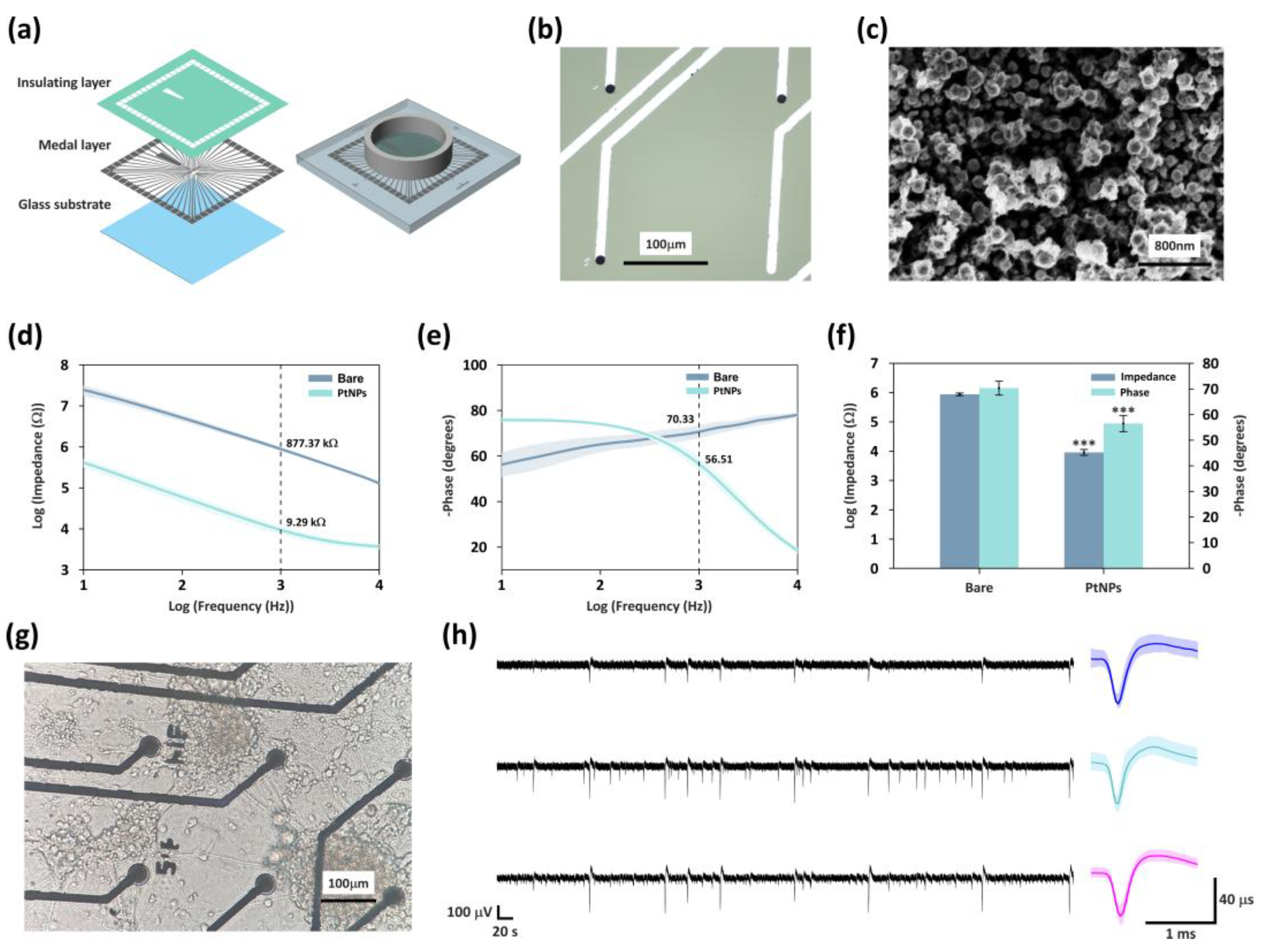
3.1. Enhanced Electrical Performance of MEA via Modification
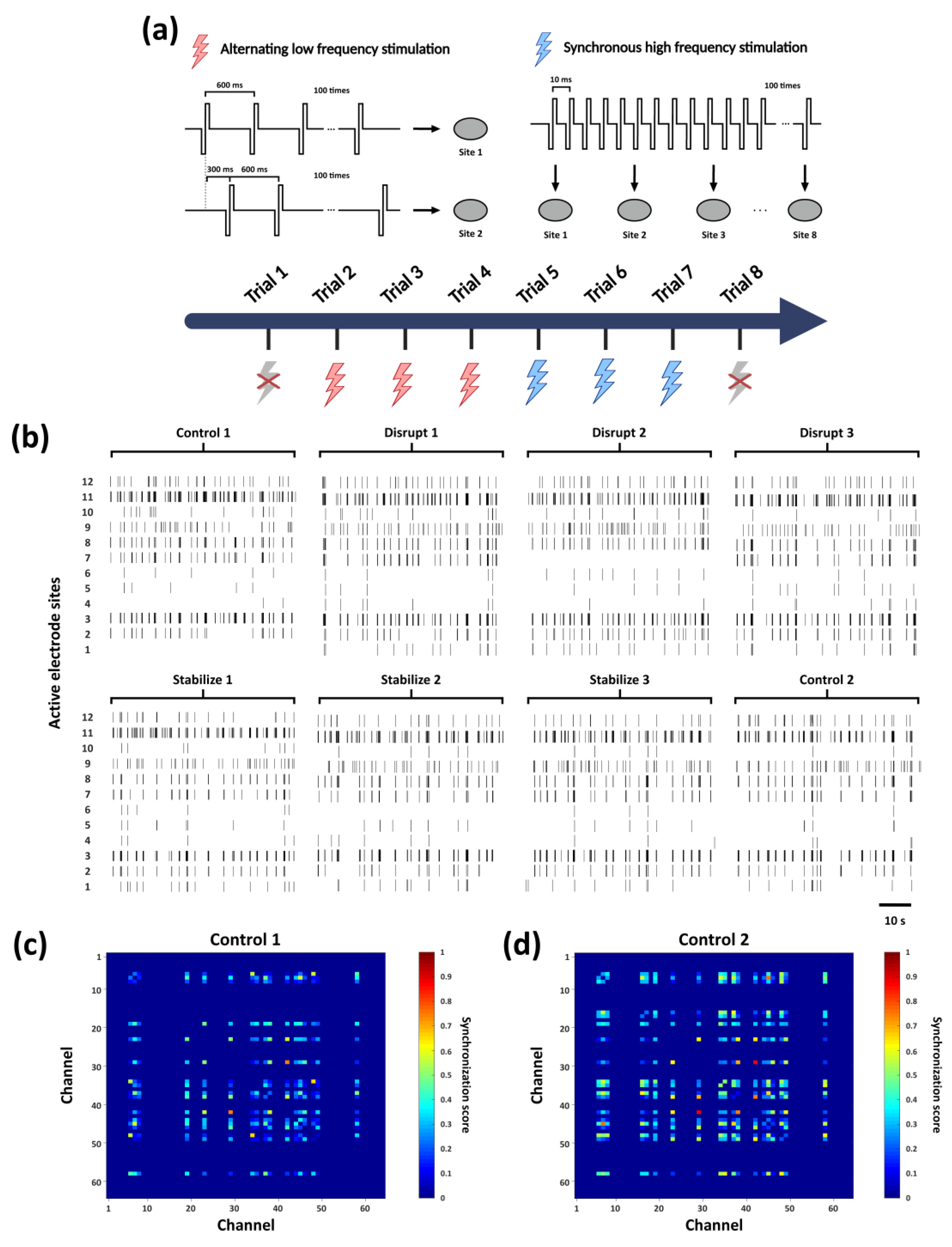
3.2. Modulation of Network Firing Synchrony Following Electrical Stimulation Trials
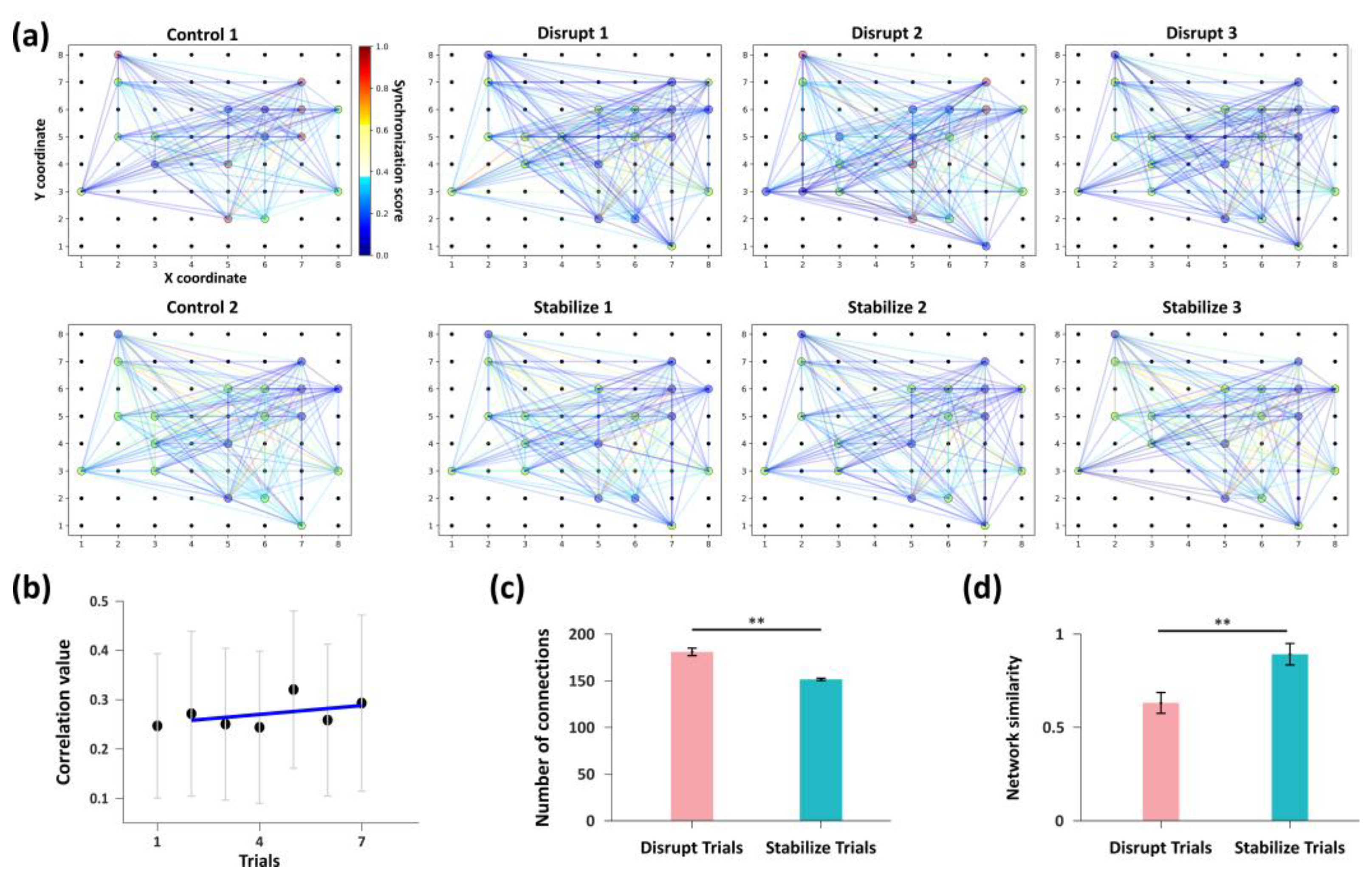
3.3. Frequency-Dependent Bidirectional Modulation of Network Connectivity by Distinct Stimulation Patterns
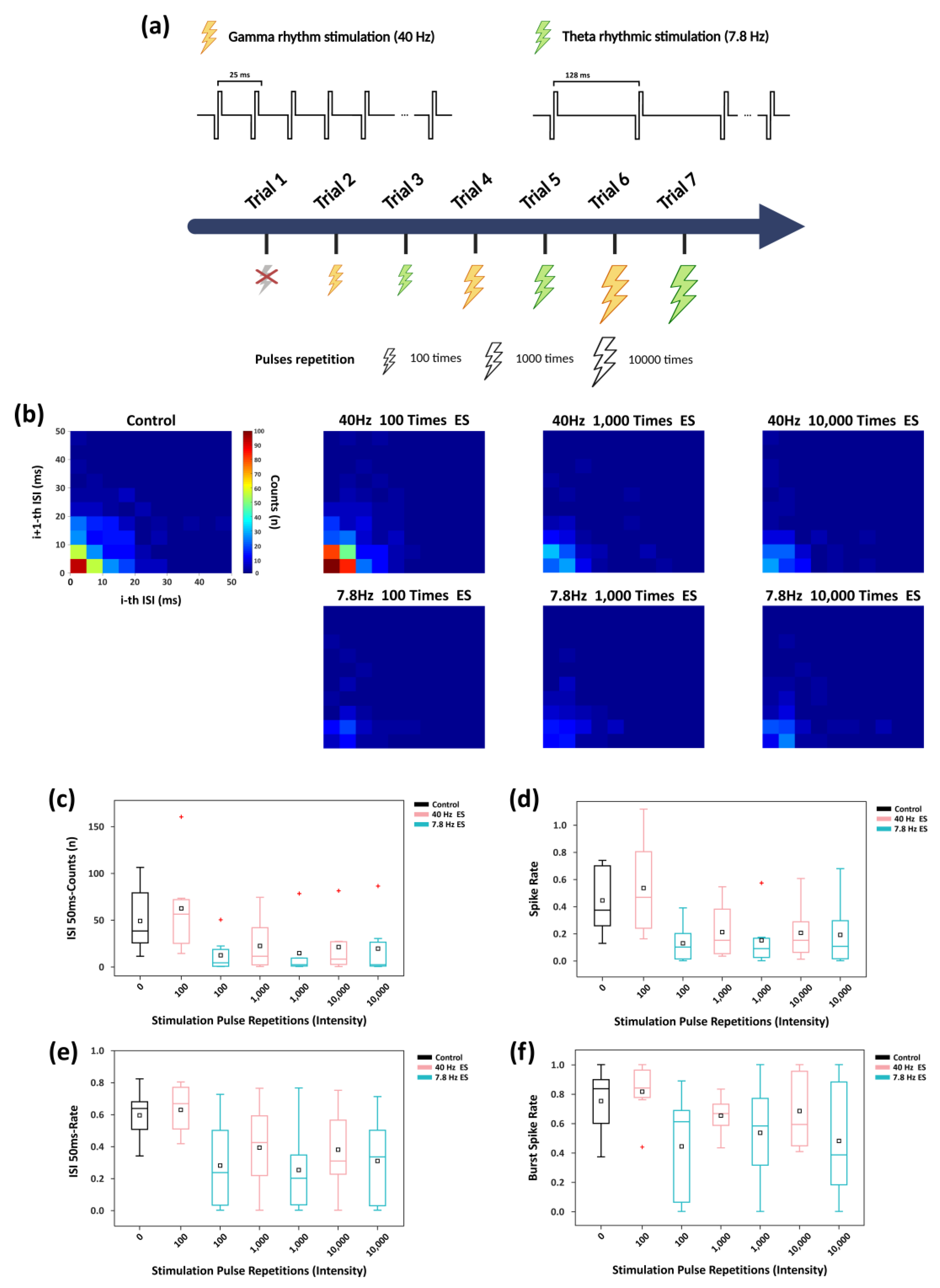
3.4. Rhythmic Stimulation with Varying Intensities Modulates ISI Distribution and Firing Rate and of Network
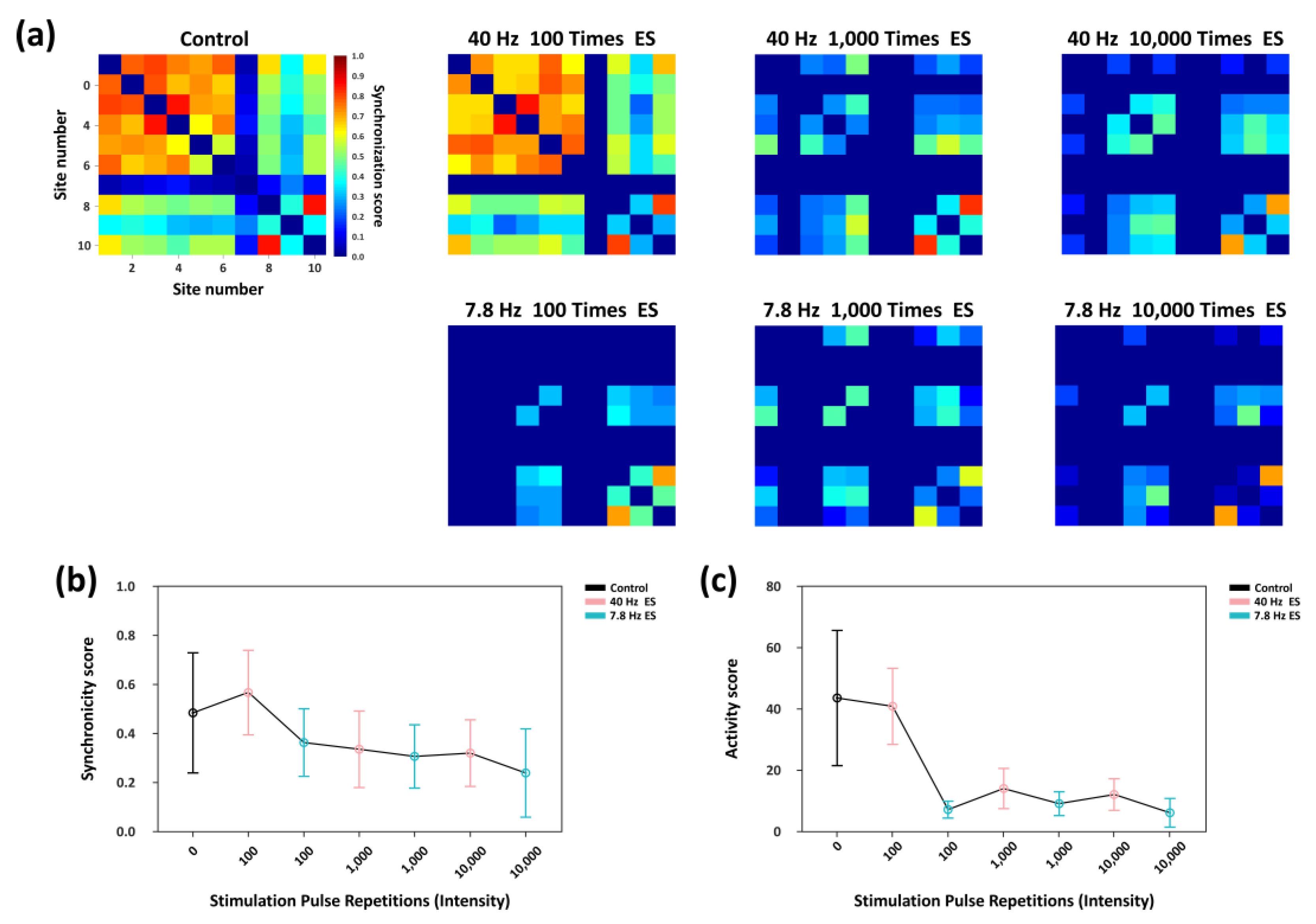
3.5. Adaptive Suppression of Network Synchrony Activity Under Prolonged Rhythmic Stimulation
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
References
- Abbott, L.F.; Nelson, S.B. Synaptic plasticity: Taming the beast. Nature neuroscience 2000, 3, 1178–1183. [Google Scholar] [CrossRef]
- Chen, Z.; Liang, Q.; Wei, Z.; et al. An overview of in vitro biological neural networks for robot intelligence. Cyborg and Bionic Systems 2023, 4, 0001. [Google Scholar] [CrossRef]
- Zarnadze, S.; Bäuerle, P.; Santos-Torres, J.; et al. Cell-specific synaptic plasticity induced by network oscillations. Elife 2016, 5, e14912. [Google Scholar] [CrossRef]
- Ter Wal, M.; Linde-Domingo, J.; Lifanov, J.; et al. Theta rhythmicity governs human behavior and hippocampal signals during memory-dependent tasks. Nature communications 2021, 12, 7048. [Google Scholar] [CrossRef]
- Abubaker, M.; Al Qasem, W.; Pilátová, K.; et al. Theta-gamma-coupling as predictor of working memory performance in young and elderly healthy people. Molecular Brain 2024, 17, 74. [Google Scholar] [CrossRef]
- Bocchio, M.; Nabavi, S.; Capogna, M. Synaptic plasticity, engrams, and network oscillations in amygdala circuits for storage and retrieval of emotional memories. Neuron 2017, 94, 731–743. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T. Neural oscillations and information flow associated with synaptic plasticity. Sheng li xue bao:[Acta physiologica Sinica] 2011, 63, 412–422. [Google Scholar] [PubMed]
- Chao, Z.C.; Bakkum, D.J.; Wagenaar, D.A.; et al. Effects of random external background stimulation on network synaptic stability after tetanization: A modeling study. Neuroinformatics 2005, 3, 263–280. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Sun, T.; Shimoda, S.; et al. Bio-inspired engineering of a perfusion culture platform for guided three-dimensional nerve cell growth and differentiation. Lab on a Chip 2022, 22, 1006–1017. [Google Scholar] [CrossRef]
- Marom, S.; Shahaf, G. Development, learning and memory in large random networks of cortical neurons: Lessons beyond anatomy. Quarterly reviews of biophysics 2002, 35, 63–87. [Google Scholar] [CrossRef]
- Jimbo, Y.; Kasai, N.; Torimitsu, K.; et al. A system for MEA-based multisite stimulation. IEEE transactions on biomedical engineering 2003, 50, 241–248. [Google Scholar] [CrossRef]
- Martinoia, S.; Bonzano, L.; Chiappalone, M.; et al. In vitro cortical neuronal networks as a new high-sensitive system for biosensing applications. Biosensors and Bioelectronics 2005, 20, 2071–2078. [Google Scholar] [CrossRef] [PubMed]
- Caporale, N.; Dan, Y. Spike timing–dependent plasticity: A Hebbian learning rule. Annu. Rev. Neurosci. 2008, 31, 25–46. [Google Scholar] [CrossRef] [PubMed]
- Bliss, T.V.P.; Lømo, T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. The Journal of physiology 1973, 232, 331–356. [Google Scholar] [CrossRef] [PubMed]
- Linden, D.J. Long-term synaptic depression in the mammalian brain. Neuron 1994, 12, 457–472. [Google Scholar] [CrossRef]
- Dura-Bernal, S.; Zhou, X.; Neymotin, S.A.; et al. Cortical spiking network interfaced with virtual musculoskeletal arm and robotic arm. Frontiers in neurorobotics 2015, 9, 13. [Google Scholar] [CrossRef]
- Azimirad, V.; Ramezanlou, M.T.; Shahabi, P. Learning of 2 dof robotic arm using integrated architecture of neural network and spike timing dependent plasticity[C]//2018 6th RSI International Conference on Robotics and Mechatronics (IcRoM). IEEE, 2018: 126-129.
- Chao, Z.C.; Bakkum, D.J.; Potter, S.M. Shaping embodied neural networks for adaptive goal-directed behavior. PLoS computational biology 2008, 4, e1000042. [Google Scholar] [CrossRef]
- Kagan, B.J.; Kitchen, A.C.; Tran, N.T.; et al. In vitro neurons learn and exhibit sentience when embodied in a simulated game-world. Neuron 2022, 110, 3952–3969. [Google Scholar] [CrossRef]
- Shahaf, G.; Marom, S. Learning in networks of cortical neurons. Journal of Neuroscience 2001, 21, 8782–8788. [Google Scholar] [CrossRef]
- Masumori, A.; Maruyama, N.; Sinapayen, L.; et al. Emergence of sense-making behavior by the stimulus avoidance principle: Experiments on a robot behavior controlled by cultured neuronal cells[C]//Artificial Life Conference Proceedings. One Rogers Street, Cambridge, MA 02142-1209, USA journals-info@ mit. edu: MIT Press, 2015: 373-380.
- Ibarra-Lecue, I.; Haegens, S.; Harris, A.Z. Breaking down a rhythm: Dissecting the mechanisms underlying task-related neural oscillations. Frontiers in Neural Circuits 2022, 16, 846905. [Google Scholar] [CrossRef]
- Massobrio, P.; Tessadori, J.; Chiappalone, M.; et al. In vitro studies of neuronal networks and synaptic plasticity in invertebrates and in mammals using multielectrode arrays. Neural plasticity 2015, 2015, 196195. [Google Scholar] [CrossRef]
- Nyhus, E.; Curran, T. Functional role of gamma and theta oscillations in episodic memory. Neuroscience & Biobehavioral Reviews 2010, 34, 1023–1035. [Google Scholar] [CrossRef]
- Tamura, M.; Spellman, T.J.; Rosen, A.M.; et al. Hippocampal-prefrontal theta-gamma coupling during performance of a spatial working memory task. Nature communications 2017, 8, 2182. [Google Scholar] [CrossRef]
- He, E.; Zhou, Y.; Luo, J.; et al. Sensitive detection of electrophysiology and dopamine vesicular exocytosis of hESC-derived dopaminergic neurons using multifunctional microelectrode array. Biosensors and Bioelectronics 2022, 209, 114263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Deng, Y.; Liu, Y.; et al. Investigating Communication Dynamics in Neuronal Network using 3D Gold Microelectrode Arrays. ACS Nano 2024. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Xu, S.; Song, Y.; et al. In situ detection of neurotransmitters and epileptiform electrophysiology activity in awake mice brains using a nanocomposites modified microelectrode array. Sensors and Actuators B: Chemical 2019, 288, 601–610. [Google Scholar] [CrossRef]
- Ray, J.; Peterson, D.A.; Schinstine, M.; et al. Proliferation, differentiation, and long-term culture of primary hippocampal neurons. Proceedings of the National Academy of Sciences 1993, 90, 3602–3606. [Google Scholar] [CrossRef]
- Cano-Jaimez, M.; Tagliatti, E.; Mendonca, P.R.F.; et al. Preparation of dissociated mouse primary neuronal cultures from long-term cryopreserved brain tissue. Journal of Neuroscience Methods 2020, 330, 108452. [Google Scholar] [CrossRef]
- Wagenaar, D.A.; Pine, J.; Potter, S.M. Searching for plasticity in dissociated cortical cultures on multi-electrode arrays. Journal of negative results in biomedicine 2006, 5, 16. [Google Scholar] [CrossRef]
- Blondel, V.D.; Guillaume, J.L.; Lambiotte, R.; et al. Fast unfolding of communities in large networks. Journal of statistical mechanics: Theory and experiment 2008, 2008, P10008. [Google Scholar] [CrossRef]
- Lam, D.; Enright, H.A.; Cadena, J.; et al. Tissue-specific extracellular matrix accelerates the formation of neural networks and communities in a neuron-glia co-culture on a multi-electrode array. Scientific reports 2019, 9, 4159. [Google Scholar] [CrossRef]
- Berger, B.; Waterman, M.S.; Yu, Y.W. Levenshtein distance, sequence comparison and biological database search. IEEE transactions on information theory 2020, 67, 3287–3294. [Google Scholar] [CrossRef]
- Li, S.; Liu, Y.; Hua, S.; et al. Advanced Brain-on-a-Chip for Wetware Computing: A Review. Advanced Science 2025, 12, e08120. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Liu, Y.; Wang, Y.; et al. Different patterns of electrical activity lead to long-term potentiation by activating different intracellular pathways. Journal of Neuroscience 2015, 35, 621–633. [Google Scholar] [CrossRef] [PubMed]
- Pariz, A.; Esfahani, Z.G.; Parsi, S.S.; et al. High frequency neurons determine effective connectivity in neuronal networks. Neuroimage 2018, 166, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Li, K.T.; He, X.; Zhou, G.; et al. Rational designing of oscillatory rhythmicity for memory rescue in plasticity-impaired learning networks. Cell Reports 2022, 39. [Google Scholar] [CrossRef]
- Watrous, A.J.; Lee, D.J.; Izadi, A.; et al. A comparative study of human and rat hippocampal low-frequency oscillations during spatial navigation. Hippocampus 2013, 23, 656–661. [Google Scholar] [CrossRef]
- Jacobs, J. Hippocampal theta oscillations are slower in humans than in rodents: Implications for models of spatial navigation and memory. Philosophical Transactions of the Royal Society B: Biological Sciences 2014, 369, 20130304. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



