Submitted:
19 November 2025
Posted:
21 November 2025
You are already at the latest version
Abstract
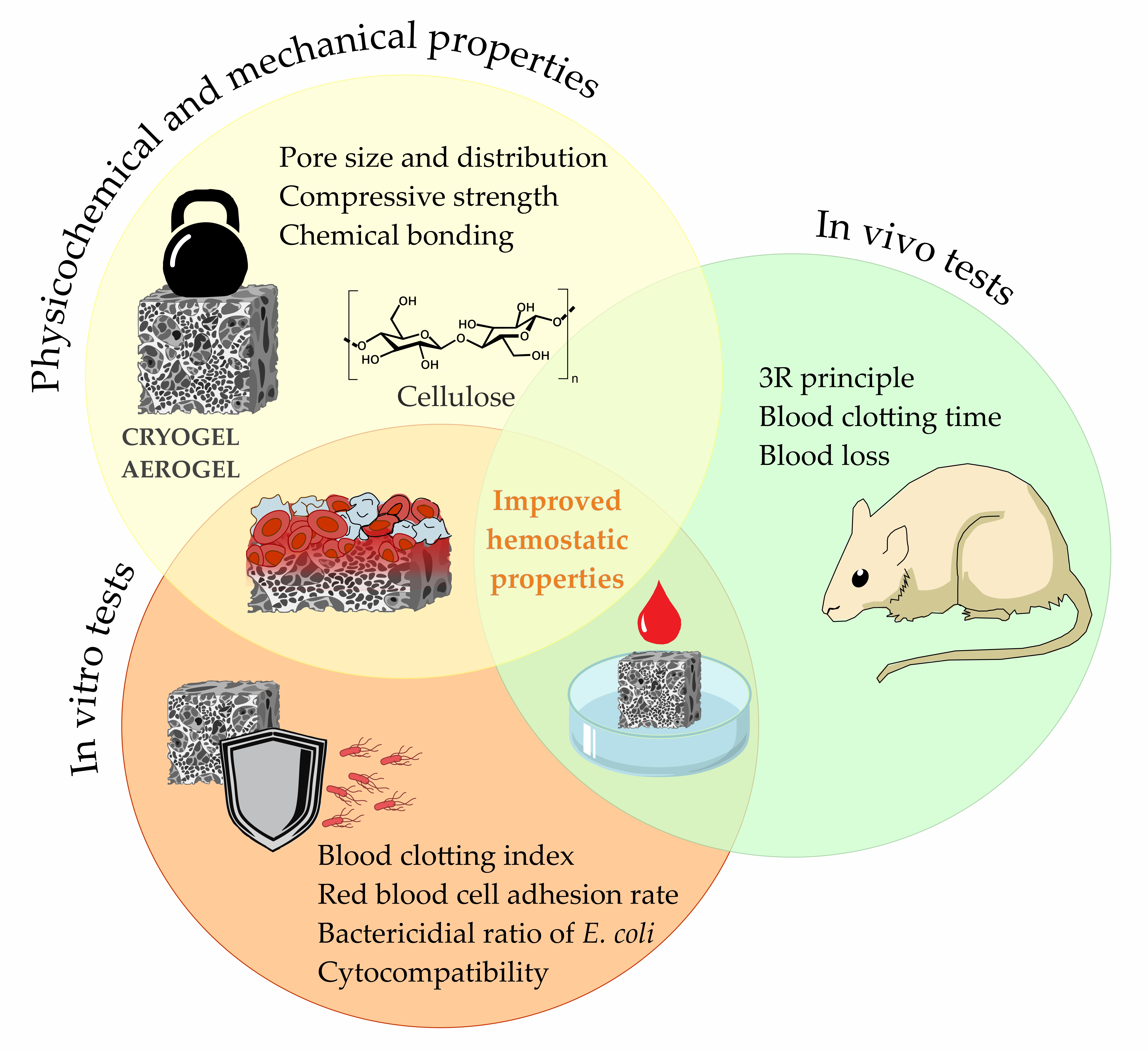
Introduction. Severe bleeding and uncompensated blood loss remain some of the most serious complications associated with trauma and surgical interventions that can threaten life. In recent years, intensive research has been directed toward developing hemostatic materials based on natural and synthetic polymers. Among them, cellulose and its modified derivatives represent one of the most promising sources for creating effective hemostatic systems. The aim of this review was to identify key criteria for the efficiency of cellulose-based gels with hemostatic activity. Methods. Experimental studies aimed at evaluating new hemostatic materials were analyzed based on international sources using the PRISMA methodology. Results. A total of 111 publications were identified. Following the identification and screening stages, 20 articles were selected for the final qualitative synthesis. The analyzed publications include experimental studies focused on the development and analysis of highly porous cellulose-based scaffolds in the form of aerogels and cryogels. The type and origin of cellulose, as well as the influence of additional components and synthesis conditions on gel formation, were investigated. Discussion and Conclusions. Three major groups of key criteria that should be considered when developing new cellulose-based highly porous scaffolds with hemostatic functionality were identified: (I) physicochemical and mechanical properties (pore size distribution, compressive strength, and presence of functional groups); (II) in vitro tests (blood clotting index, red blood cell adhesion rate, hemolysis, cytocompatibility, and antibacterial activity); (III) in vivo hemostatic efficiency (hemostasis time and blood loss) in compliance with the 3Rs policy (replacement, reduction, refinement).
Keywords:
1. Introduction
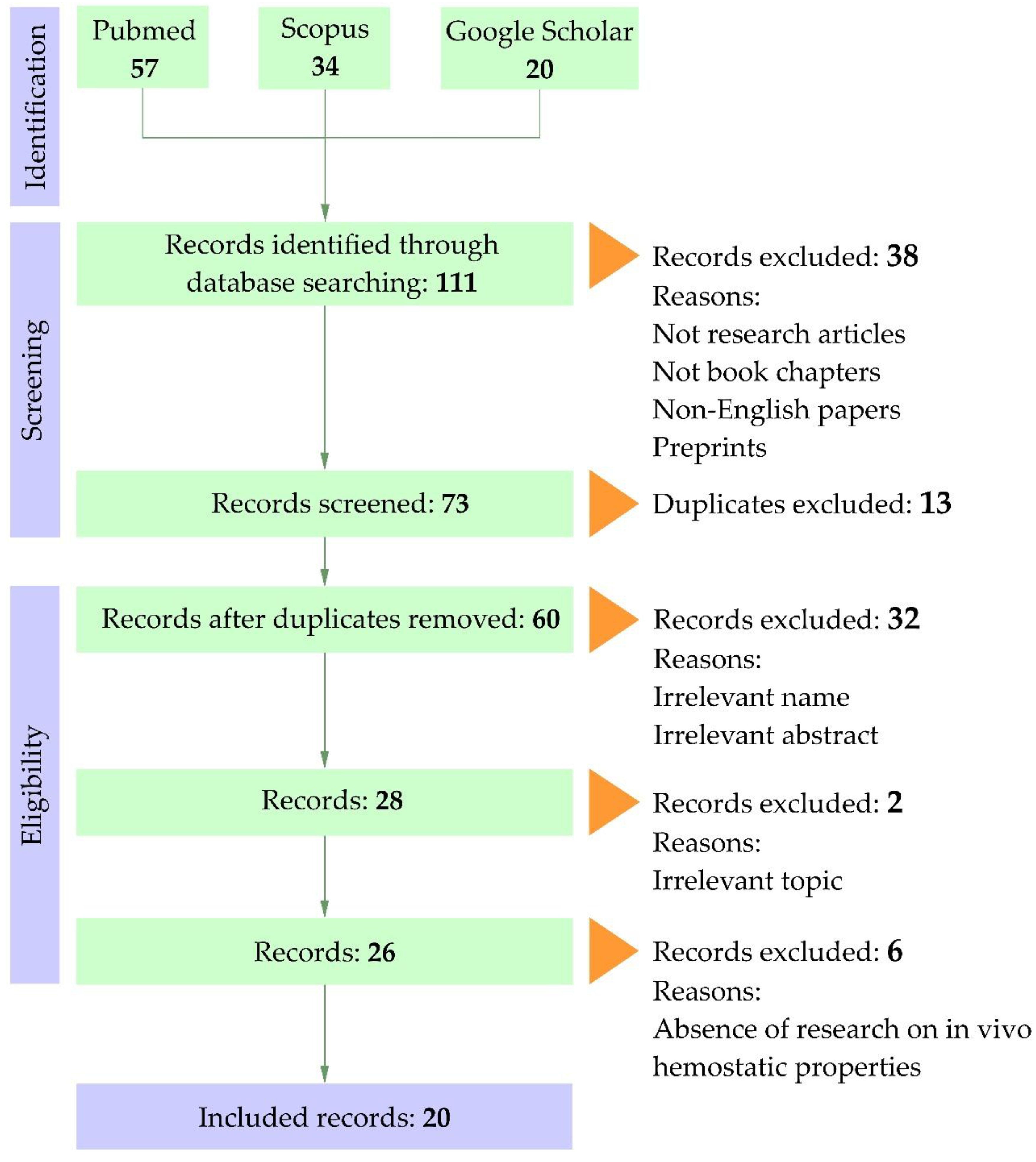
2. Materials and Methods
2.1. Search Method
2.2. Paper Selection
2.3. Data Extraction
3. Results
4. Discussion`
4.1. Physicochemical and Mechanical Properties of Cellulose-Based Hemostatic Scaffolds
4.2. In Vitro Tests
4.3. In Vivo Hemostatic Efficiency
5. Conclusions
- −
- When preparing gels according to the methodologies described in this review, with minimal modification of synthesis parameters, analysis of the first group of criteria is sufficient.
- −
- When modifying the synthesis process while using similar raw materials, physicochemical and mechanical characterization should be supplemented by in vitro tests to confirm the effect of changes on hemostatic response.
- −
- When developing a fundamentally new gel, it is necessary to analyze all groups of criteria, define the intended hemostatic application, and select an appropriate in vivo bleeding model.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hickman, D. A.; Pawlowski, C. L.; Sekhon, U. D.; Marks, J.; Gupta, A. S. Biomaterials and advanced technologies for hemostatic management of bleeding. Adv. Mater. 2018, 30(4), 1700859. [CrossRef]
- Fu, F.; Zuo, X.; Wang, Y.; Zhao, F.; Li, C.; Zeng, Y.; Wang.; L.; Wang, F. Centrifugal Spinning-Derived Biomimetic Aerogel for Rapid Hemostasis with Minimal Blood Loss. Nano Lett. 2025, 25(15), 6040-6050. [CrossRef]
- Bukatuka, C. F.; Mbituyimana, B.; Xiao, L.; Qaed Ahmed, A. A.; Qi, F.; Adhikari, M.; Shi, Zhijun; Yang, G. Recent Trends in the Application of Cellulose-Based Hemostatic and Wound Healing Dressings. J. Funct. Biomater. 2025, 16(5), 151. [CrossRef]
- Cidreira, A. C. M.; de Castro, K. C.; Hatami, T.; Linan, L. Z.; Mei, L. H. I. Cellulose nanocrystals-based materials as hemostatic agents for wound dressings: a review. Biomed. Microdevices 2021, 23(4), 43. [CrossRef]
- Rezvani Ghomi, E.; Niazi, M.; Ramakrishna, S. The evolution of wound dressings: from traditional to smart dressings. Polym. Advan. Technol. 2023, 34(2), 520-530. [CrossRef]
- Budtova, T.; Aguilera, D. A.; Beluns, S.; Berglund, L.; Chartier, C.; Espinosa, E.; Gaidukovs, S.; Klimek-Kopyra, A.; Kmita, A.; Lachowicz, D.; Liebner, F.; Platnieks, O.; Rodrigues, A.; Navarro, L.K.T.; Zou, F.; Buwalda, S. J. Biorefinery approach for aerogels. Polymers 2020, 12(12), 2779. [CrossRef]
- Tyshkunova, I. V.; Poshina, D. N.; Skorik, Y. A. Cellulose cryogels as promising materials for biomedical applications. Int. J. Mol. Sci. 2022, 23(4), 2037. [CrossRef]
- Pochinkova, P. A.; Gorbatova, M. A.; Narkevich, A. N.; Grjibovski, A. M. Updated brief recommendations onwriting and presenting systematic reviews: what’s new in PRISMA-2020 guidelines? Marine Medicine 2022, 8(2), 88-101. [CrossRef]
- Long, M.; Yang, X.; Shi, T.; Yang, Y. All-natural aerogel of nanoclay/cellulose nanofibers with hierarchical porous structure for rapid hemostasis. Int. J. Biol. Macromol. 2024, 278, 134592. [CrossRef]
- Li, M.; Dai, Q.; Zhu, S.; Feng, Q.; Qin, Z.; Gao, H.; Cao, X. An ultrafast water absorption composite cryogel containing Iron-doped bioactive glass with rapid hemostatic ability for non-compressible and coagulopathic bleeding. Chem. Eng. J. 2023, 469, 143758. [CrossRef]
- Zhu, Y.; Chen, F.; Wu, M.; Xiang, J.; Yan, F.; Xie, Y.; Tong, Z.; Chen, Y.; Cai, L. Biocompatible and antibacterial Flammulina velutipes-based natural hybrid cryogel to treat noncompressible hemorrhages and skin defects. Front. Bioeng. and Biotech. 2022, 10, 960407. [CrossRef]
- Mahmoodzadeh, A.; Moghaddas, J.; Jarolmasjed, S.; Kalan, A. E.; Edalati, M.; Salehi, R. Biodegradable cellulose-based superabsorbent as potent hemostatic agent. Chem. Eng. J. 2021, 418, 129252. [CrossRef]
- Wan, W.; Feng, Y.; Tan, J.; Zeng, H.; Jalaludeen, R. K.; Zeng, X.; Zheng, B.; Song, J.; Zhang, X.; Chen, S.; Pan, J. Carbonized cellulose aerogel derived from waste pomelo peel for rapid hemostasis of trauma--induced bleeding. Adv. Sci. 2024, 11(19), 2307409. [CrossRef]
- Xu, Z.; Tian, W.; Wen, C.; Ji, X.; Diao, H.; Hou, Y.; Fan, J.; Liu, Z.; Ji, T.; Sun, F.; Wu, D.; Zhang, J. Cellulose-based cryogel microspheres with nanoporous and controllable wrinkled morphologies for rapid hemostasis. Nano Lett. 2022, 22(15), 6350-6358. [CrossRef]
- Wang, B.; Li, X.; Wei, L.; Ma, S.; Wang, J.; Zhu, W.; Wang, H. Engineering high-performance composite cellulose materials for fast hemostasis. ACS Biomater. Sci. Eng. 2024, 10(8), 5313-5326. [CrossRef]
- Wang, M.; Sun, P.; Zhang, J.; Li, D.; Liu, Y.; Xia, Y.; Shao, L.; Jia, M. Intelligent and biocompatible cellulose aerogels featured with high-elastic and fast-hemostatic for epistaxis and wound healing. Int. J. Biol. Macromol. 2024, 277, 134239. [CrossRef]
- Mendes, B. B.; Gómez-Florit, M.; Araújo, A. C.; Prada, J.; Babo, P. S.; Domingues, R. M.; Reis, R.L.; Gomes, M. E. Intrinsically bioactive cryogels based on platelet lysate nanocomposites for hemostasis applications. Biomacromolecules 2020, 21(9), 3678-3692. [CrossRef]
- Chen, L.; Jin, Z.; Kamiya, T.; Liu, X.; Tian, Y.; Xia, R.; Wang, G.; Li, T.; Zhang, Q. Nanoporous zeolite anchored cellulose nanofiber aerogel for safe and efficient hemostasis. Small 2025, 21(21), 2500696. [CrossRef]
- Tripathi, G.; Park, M.; Lim, H.; Lee, B. T. Natural TEMPO oxidized cellulose nano fiber/alginate/dSECM hybrid aerogel with improved wound healing and hemostatic ability. Int. J. Biol. Macromol. 2023, 243, 125226. [CrossRef]
- Wang, Y.; Guo, Y.; Liu, Y.; Zhao, X.; Huang, Y.; Zhang, X.; Hu, X.; Mequanint, K.; Luo, G.; Xing, M. Platelet vesicles synergetic with biosynthetic cellulose aerogels for ultra--fast hemostasis and wound healing. Adv. Healthc. Mater. 2024, 13(17), 2304523. [CrossRef]
- Shakya, K. R.; Nigam, K.; Sharma, A.; Jahan, K.; Tyagi, A. K.; Verma, V. Preparation and assessment of agar/TEMPO-oxidized bacterial cellulose cryogels for hemostatic applications. J. Mater. Chem. B 2024, 12(14), 3453-3468. [CrossRef]
- Cao, S.; Li, Q.; Zhang, S.; Liu, Z.; Lv, X.; Chen, J. Preparation of biodegradable carboxymethyl cellulose/dopamine/Ag NPs cryogel for rapid hemostasis and bacteria-infected wound repair. Int. J. Biol. Macromol. 2022, 222, 272-284. [CrossRef]
- Li, Y.; Chu, C.; Chen, C.; Sun, B.; Wu, J.; Wang, S.; Ding, W.; Sun, D. Quaternized chitosan/oxidized bacterial cellulose cryogels with shape recovery for noncompressible hemorrhage and wound healing. Carbohyd. Polym. 2024, 327, 121679. [CrossRef]
- Pan, S.; Li, Y.; Tong, X.; Chen, L.; Wang, L.; Li, T.; Zhang, Q. Strongly-adhesive easily-detachable carboxymethyl cellulose aerogel for noncompressible hemorrhage control. Carbohyd. Polym. 2023, 301, 120324. [CrossRef]
- Chen, L.; Pan, S.; Wang, L.; Liu, X.; Jin, Z.; Xia, R.; Zhang, Q. Superhydrophobic cellulose nanofiber aerogels for efficient hemostasis with minimal blood loss. ACS Appl. Mater. Inter. 2024, 16(36), 47294-47302. [CrossRef]
- Liu, L.; Liu, L.; Chen, L.; Chen, G.; Wei, Y.; Hong, F. F. Synthesis of hemostatic aerogel of TEMPO-oxidized cellulose nanofibers/collagen/chitosan and in vivo/vitro evaluation. Mater. Today Bio 2024, 28, 101204. [CrossRef]
- Wang, L.; Zhang, C.; Zhao, W.; Li, W.; Wang, G.; Zhou, X.; Zhang, Q. Water-swellable cellulose nanofiber aerogel for control of hemorrhage from penetrating wounds. ACS Appl. Bio Mater. 2022, 5(10), 4886-4895. [CrossRef]
- Jin, Z.; Chen, L.; Liu, X.; Xia, R.; Li, W.; Wang, G.; Zhang, Q. Zeolite firmly anchored regenerated cellulose aerogel for efficient and biosafe hemostasis. Int. J. Biol. Macromol. 2025, 304, 140743. [CrossRef]
- Lawrence, M., Jiang, Y. (2017). Porosity, pore size distribution, micro-structure. In Bio-aggregates Based Building Materials. RILEM State-of-the-Art Reports; Amziane, S.; Collet, F. Еds.; vol 23. Dordrecht: Springer, Netherlands, 2017; pp. 39-71. [CrossRef]
- Schimper, C. B.; Pachschwöll, P.; Maitz, M. F.; Werner, C.; Rosenau, T.; Liebner, F. Hemocompatibility of cellulose phosphate aerogel membranes with potential use in bone tissue engineering. Front. Bioeng. and Biotech. 2023, 11, 1152577. [CrossRef]
- Bouras, H.; Zou, F.; Tillier, Y.; Buwalda, S.; Budtova, T. Starch aerogels, cryogels and xerogels for biomedical applications. In Advanced Functional Polymers for Medicine; Utrecht, Netherlands, 2024.
- Rodríguez-Dorado, R.; López-Iglesias, C.; García-González, C. A.; Auriemma, G.; Aquino, R. P.; Del Gaudio, P. Design of aerogels, cryogels and xerogels of alginate: Effect of molecular weight, gelation conditions and drying method on particles’ micromeritics. Molecules 2019, 24(6), 1049. [CrossRef]
- Croll, T. I.; Gentz, S.; Mueller, K.; Davidson, M.; O’Connor, A. J.; Stevens, G. W.; Cooper-White, J. J. Modelling oxygen diffusion and cell growth in a porous, vascularising scaffold for soft tissue engineering applications. Chem. Eng. Sci. 2005, 60(17), 4924-4934. [CrossRef]
- Buchtová, N.; Budtova, T. Cellulose aero-, cryo-and xerogels: towards understanding of morphology control. Cellulose 2016, 23(4), 2585-2595. [CrossRef]
- Yeo, J. H.; Lam, Y. W.; Fraser, S. T. Cellular dynamics of mammalian red blood cell production in the erythroblastic island niche. Biophys. Rev. 2019, 11(6), 873-894. [CrossRef]
- Zhao, X.; Guo, B.; Wu, H.; Liang, Y.; Ma, P. X. Injectable antibacterial conductive nanocomposite cryogels with rapid shape recovery for noncompressible hemorrhage and wound healing. Nat. Comm. 2018, 9(1), 2784. [CrossRef]
- Darya, G.; Mohammadi, H.; Dehghan, Z.; Nakhaei, A.; Derakhshanfar, A. Animal models of hemorrhage, parameters, and development of hemostatic methods. Lab. Anim. Res. 2025, 41(1), 5. [CrossRef]
| Form of the hemostatic agent |
Type and origin of cellulose |
Additional component | Conditions for obtaining highly porous scaffolds |
Ref |
|---|---|---|---|---|
| Aerogel | TEMPO-oxidized cellulose nanofibers (ScienceK Co., Ltd.) |
Halloysite | Concentration 1% Dissolution for 2 h Freezing temperature -60 °C for 24 h Vacuum freeze dryer |
[9] |
| Cryogel | Hydroxyethyl cellulose (Aladdin Chemistry) |
Quaternized chitosan and Iron-doped bioactive glass | Concentration 0.1%, 0.2%, and 0.4% Freezing temperature -20 °C for 36-48 h Freeze-dryer |
[10] |
| Cryogel | Hydroxyethyl cellulose (Shandong Head Reagent Co., Ltd.) |
Flammulina velutipes extract | Concentration 2% Freezing temperature -20 °C for 12 h Washing in deionized water, 24 h Freeze dryer -50 °C, 24 h |
[11] |
| Aerogel | Carboxymethyl cellulose and hydroxyl ethyl cellulose (Sigma-Aldrich) |
Tranexamic acid | Substitution of the solvent (water) with isopropyl alcohol and hexane 1 method of drying: in Petri dishes in the oven for 15 h 1 method of drying: freezing temperature -20 °C for 24 h Freeze dryer |
[12] |
| Aerogel | Carbonized cellulose from pomelo peel waste (Jiangyong Xiangyou, Yongzhou) | - | Substitution of the solvent (water) with ethanol Freeze dryer |
[13] |
| Cryogel microspheres |
Microcrystalline cellulose (Innochem Beijing) |
Polydopamine | Substitution of the solvent (water) with butanol 50% Freeze dryer |
[14] |
| Aerogel | Microcrystalline cellulose (Anhui Shanhe Pharmaceutical Co., Ltd.) |
Gelatin, Diatomite | Substitution of the solvent (water) with ethanol Freezing temperature -20 °C for 12 h Freeze dryer for 18 h |
[15] |
| Aerogel | Carboxymethyl cellulose and TEMPO-oxide cellulose nanofibers |
- | Concentration 0.1-0.5% Substitution of the solvent (water) with butyldehydrodiketone ethylene glycol for 24 h Freezing temperature -75 °C, 12 h Freeze dryer for 48 h |
[16] |
| Cryogel | Microcrystalline cellulose powder (Sigma-Aldrich) |
Platelet lysate | Concentration 1.2-2.4% Freezing temperature -80 °C Freeze dryer |
[17] |
| Aerogel | Carboxymethyl cellulose nanofibers, dry bleached wood pulp powders |
Zeolite powder | Concentration 1% Freezing temperature -20 °C for 12 h Freeze dryer -50 °C. |
[18] |
| Aerogel | TEMPO oxidized cellulose nano fiber (TOCNF) |
Alginate and decellularized pig skin fragments |
Concentration 1% alginate, 1% TOCNF Freeze dryer |
[19] |
| Aerogel | Oxidized bacterial cellulose G. xylinum | Platelet extracellular vesicles | Concentration 0.2-1.2%; Freezing temperature -20 °C (3 freeze-thaw cycles of 15 h each) Substitution of the solvent (water) with tert-butanol for 12 h Freeze dryer |
[20] |
| Cryogel | TEMPO oxidized bacterial cellulose (BC) K. xylinus |
Agar | Concentration 1% agar and 20, 30, 40% OBC w/w of agar Substitution of the solvent (water) with methanol Freezing temperature -80 °C; Freeze dryer for 24 h |
[21] |
| Cryogel | Carboxymethyl cellulose (Macklin Co., Ltd.) |
Dopamine, silver nanoparticles | Concentration 2% Freezing temperature -20 °C for 36 h Freeze dryer |
[22] |
| Cryogel | Oxidized bacterial cellulose A. xylinum | Quaternized chitosan | Concentration 5% Freezing temperature − 80 °C; Freeze dryer |
[23] |
| Aerogel | Carboxymethyl cellulose (Ever Bright Enterprise Development Co., Ltd.) |
N-hydroxysuccinimide ether | Concentration 2% Freezing temperature -80 °C; Freeze dryer Heating for cross-linking − 80 °C for 1 h |
[24] |
| Aerogel | Bacterial cellulose (Hainan Yeguo Foods Co., Ltd.) | Polydophamine and modified fluoroalkyl chains | Concentration 1% Freezing Freeze dryer -50 °C |
[25] |
| Aerogel | TEMPO-oxidized cellulose nanofibers | Сollagen/chitosan | Concentration 1% Freezing temperature -80 °C, 12 h Freeze dryer, 48 h |
[26] |
| Aerogel | Carboxymethyl cellulose nanofibers, bleached wood pulp |
Сitric acid | Concentration 1% Freezing temperature 4 °C for 2 h and -80 °C for 6 h Freeze dryer Heating for cross-linking − 80 °C for 1 h |
[27] |
| Aerogel | Carboxymethyl cellulose (Shandong Senxin Environmental Protection Technology Co., Ltd.) |
Zeolite | Concentration 7%; Dissolution in ethanol and acid for 24 h; Washing with water Freeze dryer |
[28] |
| Pore size | Porosity, % | Compression stress, MPa | Ref | |
|---|---|---|---|---|
| Macropores, µm | Mesopores, nm | |||
| 7.0-19.0 | ∼13.00 | 0.070 | [9] | |
| 0.070 | [10] | |||
| 100–200 | 0.020 | [11] | ||
| 151.6 ± 8.6 | 4.53–16.87 | 70.0 | 0.082 | [12] |
| 80.0 | [13] | |||
| 16.00–55.00 | 96.9 | [14] | ||
| 94.9 | [15] | |||
| 15.0–25.0 | 50-300 | 0.018 | [16] | |
| 88.9 ± 1.5 | [17] | |||
| 110 | 21.37 ± 1.81 | [18] | ||
| 0.002 | [19] | |||
| 30.0 | 97.4 ± 0.4 | [20] | ||
| 6.3 ± 0.3 | 1.50–2.50 | 0.700 | [21] | |
| > 80.0 | [22] | |||
| ∼100-200 | 30.08 | 0.004 | [23] | |
| [24] | ||||
| 50-200 | 0.013 | [25] | ||
| 94.8 | 0.097 | [26] | ||
| 30-100 | 0.065 | [27] | ||
| ∼100 | 5.10 | [28] | ||
| BCI, % | Red blood cell adhesion rate, % |
Hemolysis, % | Cytocompatibility, % | Bactericidal ratio of E. coli, % |
Ref |
|---|---|---|---|---|---|
| 59.47 ± 4.92 | [9] | ||||
| 11.50 ± 0.87 | 52.9 | < 5.00 | 80.12 | 95.0 | [10] |
| < 5.00 | 100 | 100 | [11] | ||
| 0.80 | 80.0 | 0.66 ± 0.05 | 100 | [12] | |
| 10.0 | 55.0 | < 5.0 | 100 | [13] | |
| 3.80 | 3.00 | >99.3 | 34.6 | [14] | |
| 12.3 | < 5.00 | >94 | [15] | ||
| < 5.00 | >90 | + | [16] | ||
| 2.30 | 91.2 ± 8.2 | [17] | |||
| 8.2 | [18] | ||||
| 4.0 | 2.68 ± 2.03 | > 90 | [19] | ||
| < 0.05 | < 5.00 | 100 | [20] | ||
| 89.76 ± 0.49 | 3.20 ± 0.43 | [21] | |||
| < 1.0 | < 5.00 | 100 | [22] | ||
| < 5.00 | 70.0 | [23] | |||
| 0.025 | < 5.00 | + | [24] | ||
| 7.4 ± 2.5 | 7.50 ± 4.00 | > 95 | 88.2 | [25] | |
| < 4.00 | > 80 | 100 | [26] | ||
| < 1.00 | > 90 | 95.0 | [27] | ||
| 10.0 | 1.37 | > 90 | > 95.0 | [28] |
| Anesthetic | Blood clotting time, seconds | Blood loss, g | Ref | ||
|---|---|---|---|---|---|
| liver | tail | femoral artery | |||
| Pentobarbital | 98.0 ± 24.0 | 2.100 ± 0.870 | [9] | ||
| Pentobarbital | 42.9 ± 2.2 | 0.300 ± 0.070 | [10] | ||
| Isoflurane | ∼70 | 0.150 | [11] | ||
| Ketamine and xylazine |
179 | 0.800 | [12] | ||
| Pentobarbital | 91.6 ± 5.5 | 0.293 ± 0.032 | [13] | ||
| + | 43.5 ± 9.8 | 0.740 ± 0.250 | [14] | ||
| Tribromoethanol | 37.4 ± 5.3 | 0.330 ± 0.006 | [15] | ||
| [16] | |||||
| Medetomidine | [17] | ||||
| + | 131.8 ± 10.2 | 0.210 ± 0.490 | [18] | ||
| Isoflurane | 25.0 | 10.0 | [19] | ||
| 98.6 ± 17.3 | 0.760 ± 0.090 | [20] | |||
| 38.0 | 0.15 | [21] | |||
| 47.00 ± 4.2 | 0.065 ± 0.018 | [22] | |||
| Isoflurane | 20.0 | 0.08 | [23] | ||
| + | 0.025 ± 0.010 | [24] | |||
| 106.2 ± 37.3 | 0.013 ± 0.010 | [25] | |||
| Chloral hydrate solution |
41.0 | 0.039 | [26] | ||
| Tribromoethanol | 349.8 | 0.380 | [27] | ||
| Avertin | 162.0 ± 6.0 | 0.286 ± 0.014 | [28] | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





