Submitted:
14 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
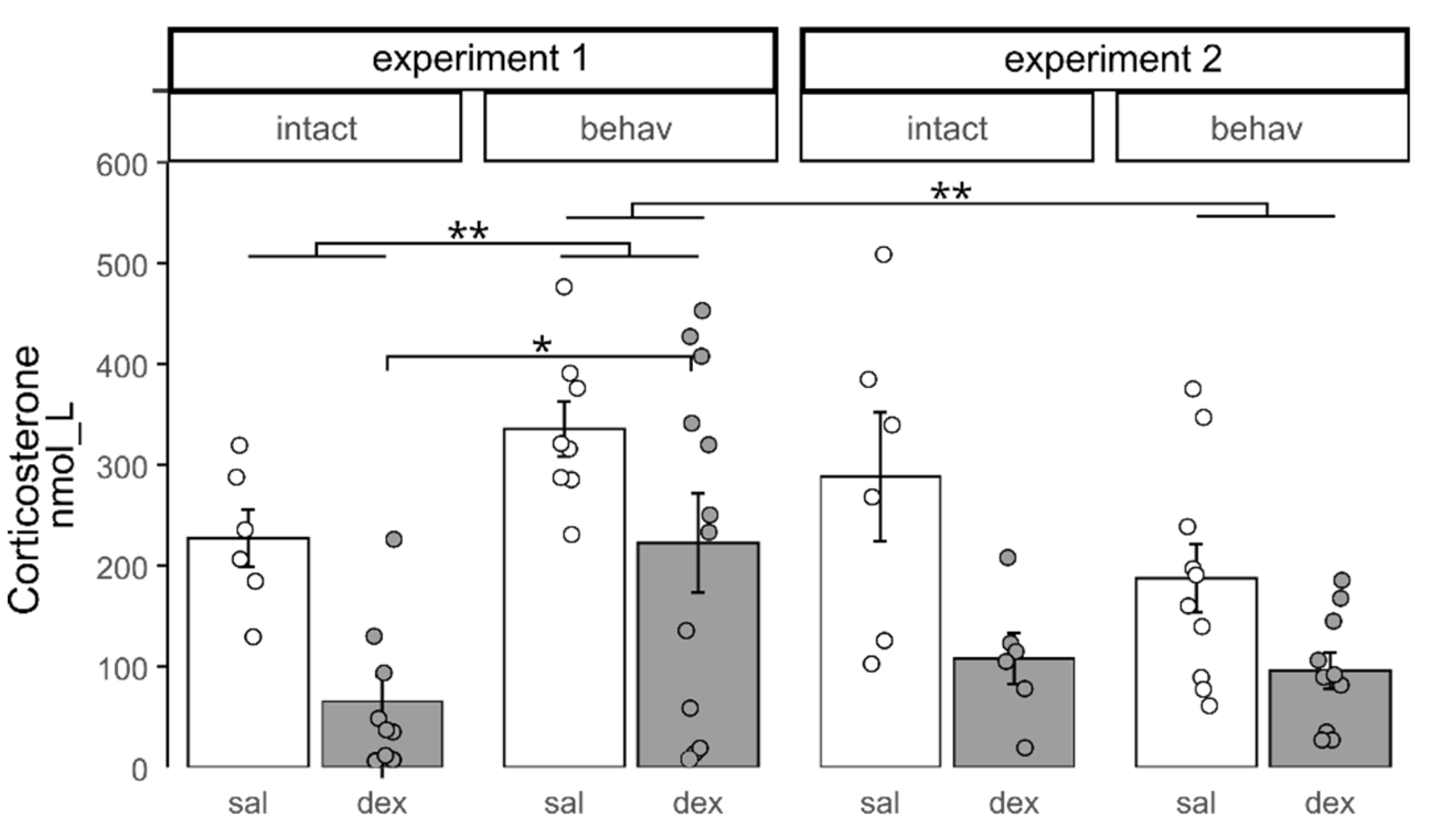
2.1. Effects of Behavioral Testing on Basal Corticosterone Levels in Control
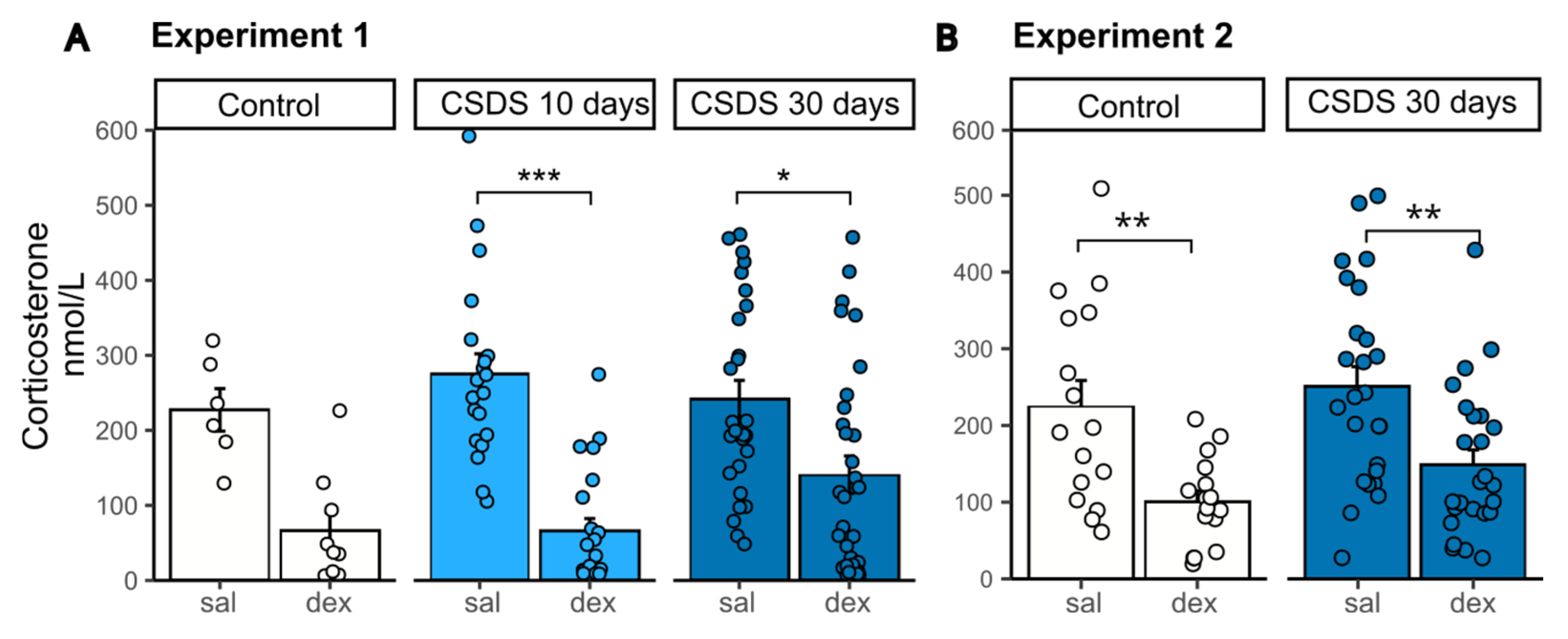
2.2. HPA Axis State Evaluation After 10 and 30 Days of CSDS with DST
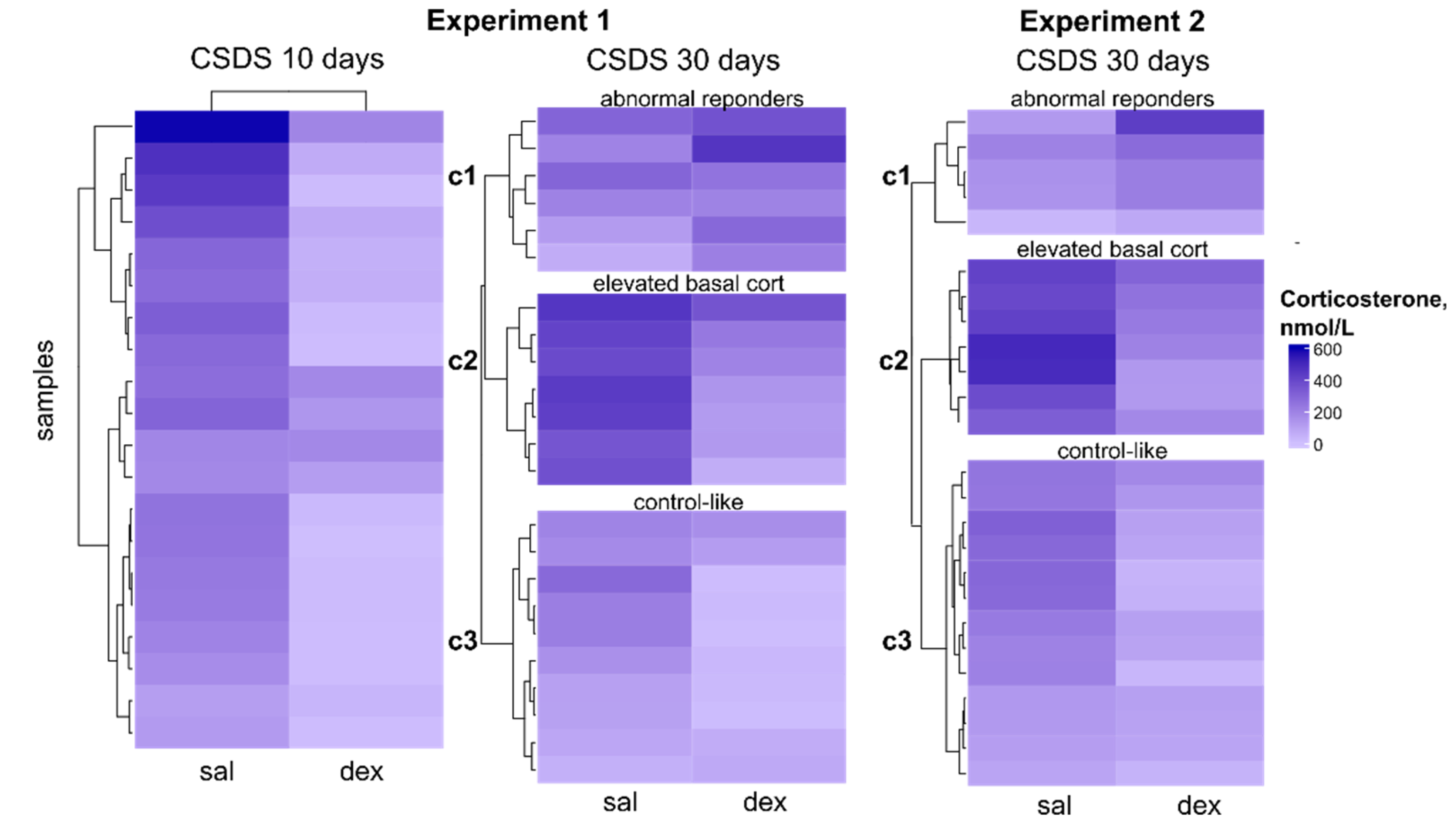
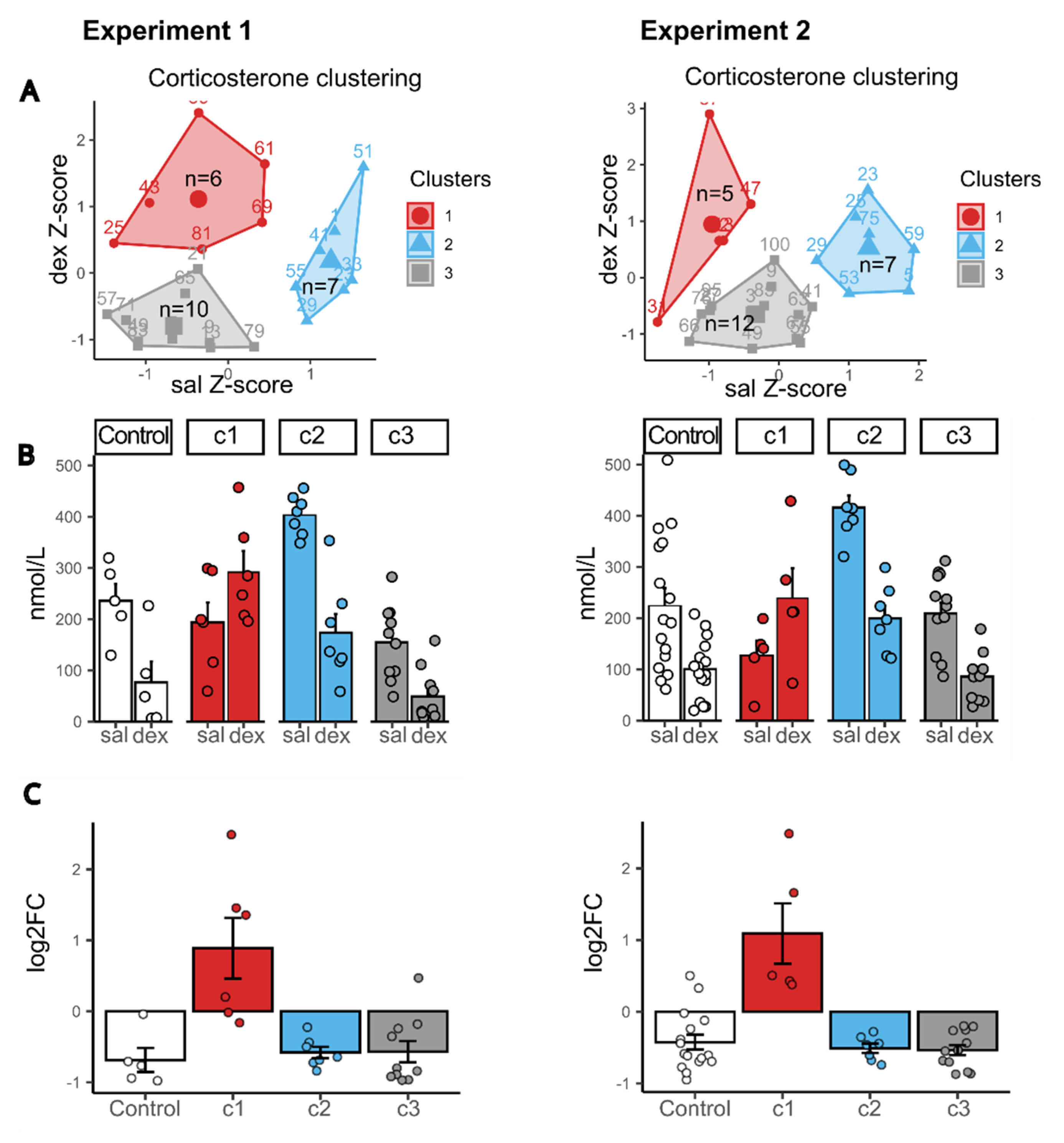
2.3. Clustering by Corticosterone Response in DST
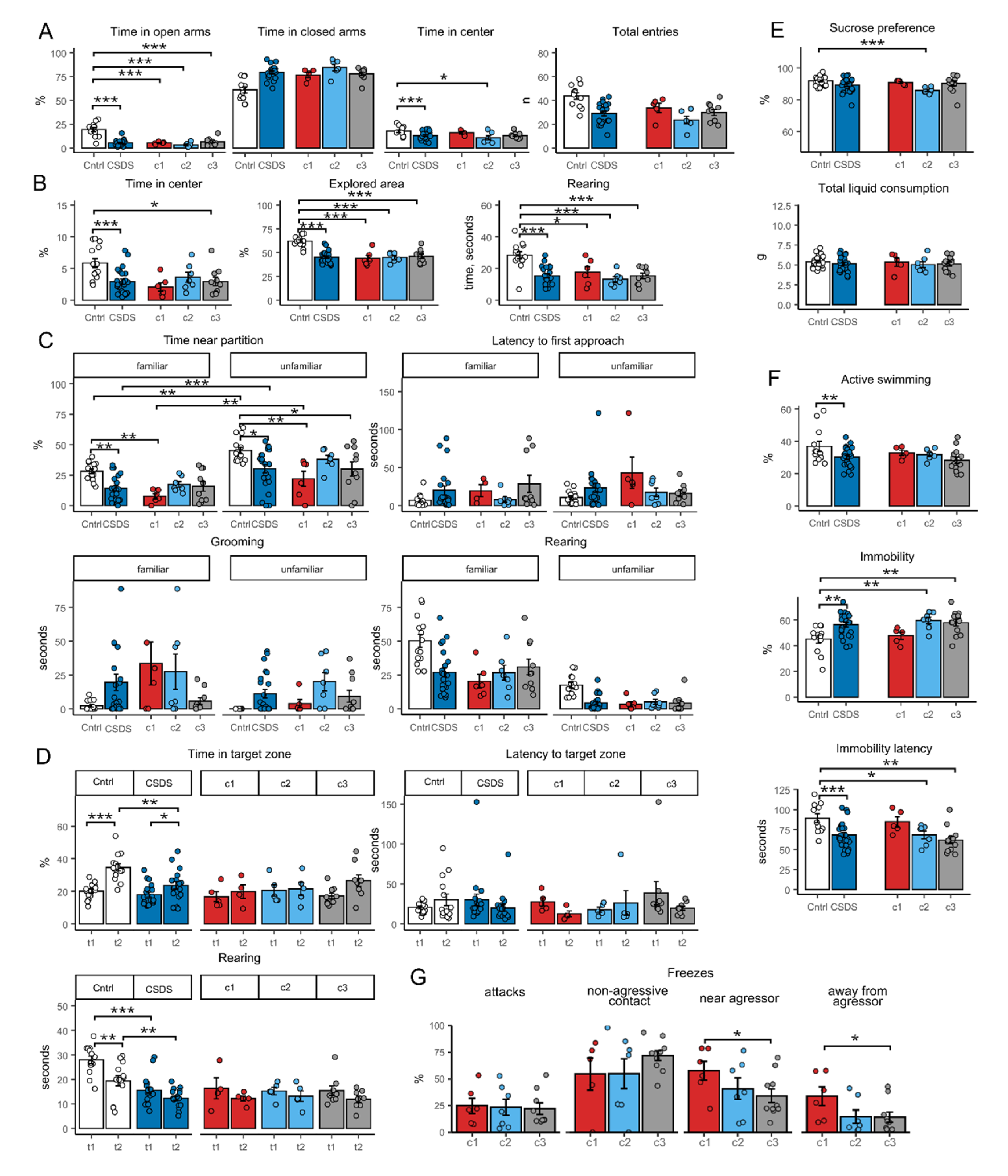
2.4. Behavior Analysis
2.5. Behavioral Characteristics of Animals with Different HPA Axis Response in DST
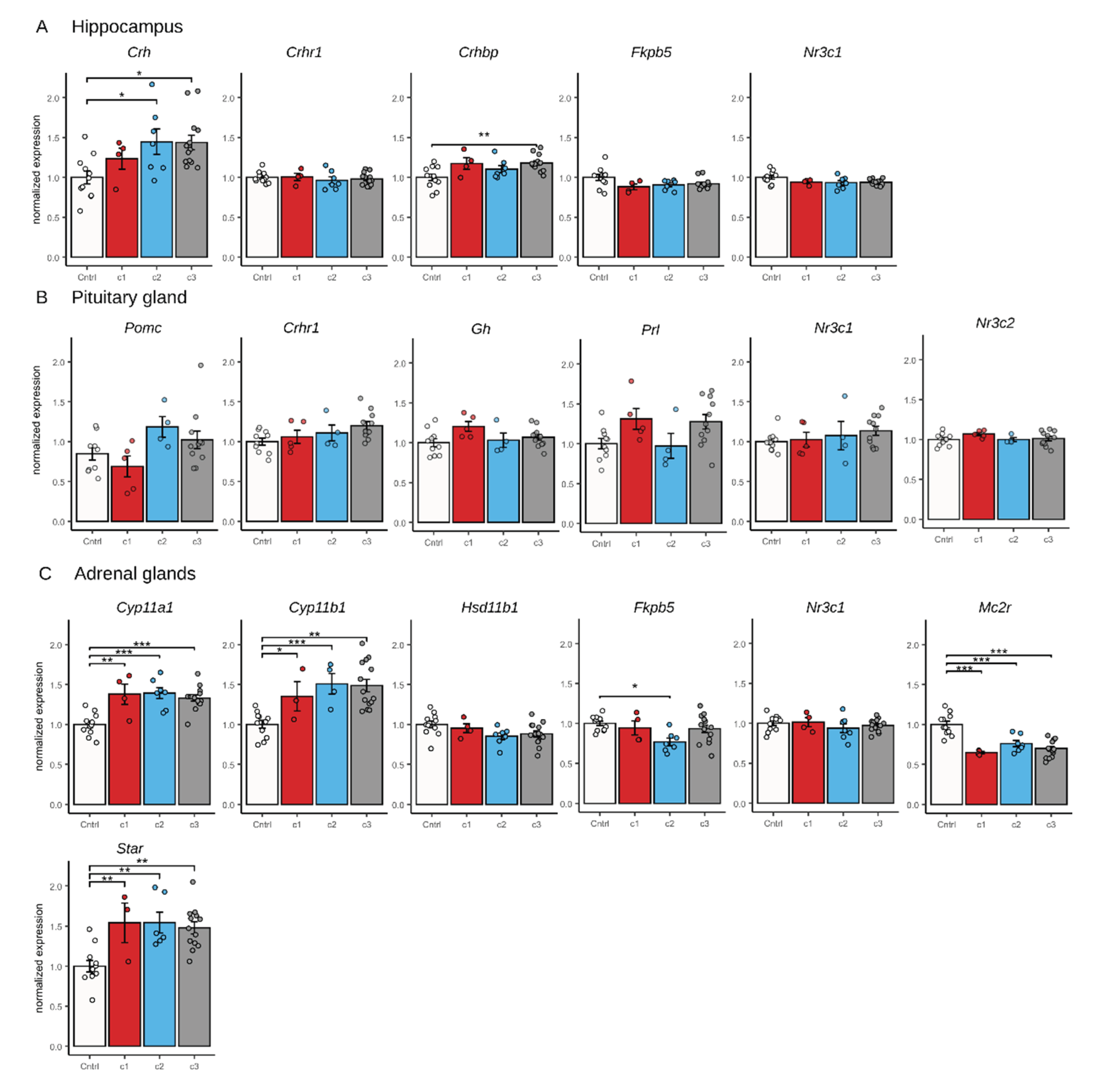
2.6. qPCR Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Chronic Social Defeat Stress
4.3. Experimental Design
4.3.1. Experiment 1
4.3.2. Experiment 2
4.4. Dexamethasone Suppression Test (DST)
4.5. Behavioral Tests
4.5.1. Elevated Plus Maze
4.5.2. Open Field
4.5.3. Social Interaction Test
4.5.4. Partition Test
4.5.5. Social Defeat Session
4.5.6. The Forced Swimming Test
4.5.7. The Sucrose Preference Test
4.6. Enzyme-Linked Immunosorbent Assay
4.7. RNA Extraction
4.9. Statistical Analysis
4.10. Clustering
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Agorastos, A.; Chrousos, G.P. The Neuroendocrinology of Stress: The Stress-Related Continuum of Chronic Disease Development. Mol. Psychiatry 2022, 27, 502–513. [Google Scholar] [CrossRef]
- Avitsur, R.; Powell, N.; Padgett, D.A.; Sheridan, J.F. Social Interactions, Stress, and Immunity. Immunol. Allergy Clin. North Am. 2009, 29, 285–293. [Google Scholar] [CrossRef]
- Holsboer, F. Stress, Hypercortisolism and Corticosteroid Receptors in Depression: Implications for Therapy. J. Affect. Disord. 2001, 62, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Jochems, J.; Teegarden, S.L.; Chen, Y.; Boulden, J.; Challis, C.; Ben-Dor, G.A.; Kim, S.F.; Berton, O. Enhancement of Stress Resilience Through Histone Deacetylase 6–Mediated Regulation of Glucocorticoid Receptor Chaperone Dynamics. Biol. Psychiatry 2015, 77, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Quan, N.; Avitsur, R.; Stark, J.L.; He, L.; Shah, M.; Caligiuri, M.; Padgett, D.A.; Marucha, P.T.; Sheridan, J.F. Social Stress Increases the Susceptibility to Endotoxic Shock. J. Neuroimmunol. 2001, 115, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Baumeister, D.; Lightman, S.L.; Pariante, C.M. The HPA Axis in the Pathogenesis and Treatment of Depressive Disorders: Integrating Clinical and Molecular Findings. Psychopathol. Rev. 2016, a3, 64–76. [Google Scholar] [CrossRef]
- Bondar, N.; Bryzgalov, L.; Ershov, N.; Gusev, F.; Reshetnikov, V.; Avgustinovich, D.; Tenditnik, M.; Rogaev, E.; Merkulova, T. Molecular Adaptations to Social Defeat Stress and Induced Depression in Mice. Mol. Neurobiol. 2018, 55, 3394–3407. [Google Scholar] [CrossRef]
- Ceruso, A.; Martínez-Cengotitabengoa, M.; Peters-Corbett, A.; Diaz-Gutierrez, M.J.; Martínez-Cengotitabengoa, M. Alterations of the HPA Axis Observed in Patients with Major Depressive Disorder and Their Relation to Early Life Stress: A Systematic Review. Neuropsychobiology 2020, 79, 417–427. [Google Scholar] [CrossRef]
- Carnevali, L.; Montano, N.; Tobaldini, E.; Thayer, J.F.; Sgoifo, A. The Contagion of Social Defeat Stress: Insights from Rodent Studies. Neurosci. Biobehav. Rev. 2020, 111, 12–18. [Google Scholar] [CrossRef]
- Schmidt, M.V.; Sterlemann, V.; Ganea, K.; Liebl, C.; Alam, S.; Harbich, D.; Greetfeld, M.; Uhr, M.; Holsboer, F.; Müller, M.B. Persistent Neuroendocrine and Behavioral Effects of a Novel, Etiologically Relevant Mouse Paradigm for Chronic Social Stress during Adolescence. Psychoneuroendocrinology 2007, 32, 417–429. [Google Scholar] [CrossRef]
- Bhatnagar, S.; Vining, C. Facilitation of Hypothalamic–Pituitary–Adrenal Responses to Novel Stress Following Repeated Social Stress Using the Resident/Intruder Paradigm. Horm. Behav. 2003, 43, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Ruis, M.A.W.; Te Brake, J.H.A.; Buwalda, B.; De Boer, S.F.; Meerlo, P.; Korte, S.M.; Blokhuis, H.J.; Koolhaas, J.M. Housing Familiar Male Wildtype Rats Together Reduces the Long-Term Adverse Behavioural and Physiological Effects of Social Defeat. Psychoneuroendocrinology 1999, 24, 285–300. [Google Scholar] [CrossRef] [PubMed]
- Bartolomucci, A.; Pederzani, T.; Sacerdote, P.; Panerai, A.E.; Parmigiani, S.; Palanza, P. Behavioral and Physiological Characterization of Male Mice under Chronic Psychosocial Stress. Psychoneuroendocrinology 2004, 29, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.C.; Davis, J.M. DST Studies in Psychotic Depression: A Meta-Analysis. Am. J. Psychiatry 1997, 154, 1497–1503. [Google Scholar] [CrossRef]
- Nuller, J.L.; Ostroumova, M.N. Resistance to Inhibiting Effect of Dexamethasone in Patients with Endogenous Depression. Acta Psychiatr. Scand. 1980, 61, 169–177. [Google Scholar] [CrossRef]
- Stetler, C.; Miller, G.E. Depression and Hypothalamic-Pituitary-Adrenal Activation: A Quantitative Summary of Four Decades of Research. Psychosom. Med. 2011, 73, 114–126. [Google Scholar] [CrossRef]
- Young, E.A.; Akana, S.; Dallman, M.F. Decreased Sensitivity to Glucocorticoid Fast Feedback in Chronically Stressed Rats. Neuroendocrinology 1990, 51, 536–542. [Google Scholar] [CrossRef]
- Skupio, U.; Tertil, M.; Sikora, M.; Golda, S.; Wawrzczak-Bargiela, A.; Przewlocki, R. Behavioral and Molecular Alterations in Mice Resulting from Chronic Treatment with Dexamethasone: Relevance to Depression. Neuroscience 2015, 286, 141–150. [Google Scholar] [CrossRef]
- Lee, C.-W.; Fang, Y.-P.; Chu, M.-C.; Chung, Y.-J.; Chi, H.; Tang, C.-W.; So, E.C.; Lin, H.-C.; Lin, H.-C. Differential Mechanisms of Synaptic Plasticity for Susceptibility and Resilience to Chronic Social Defeat Stress in Male Mice. Biochem. Biophys. Res. Commun. 2021, 562, 112–118. [Google Scholar] [CrossRef]
- Wang, W.; Liu, L.; Yang, X.; Gao, H.; Tang, Q.-K.; Yin, L.-Y.; Yin, X.-Y.; Hao, J.-R.; Geng, D.-Q.; Gao, C. Ketamine Improved Depressive-like Behaviors via Hippocampal Glucocorticoid Receptor in Chronic Stress Induced- Susceptible Mice. Behav. Brain Res. 2019, 364, 75–84. [Google Scholar] [CrossRef]
- Wang, W.; Liu, W.; Duan, D.; Bai, H.; Wang, Z.; Xing, Y. Chronic Social Defeat Stress Mouse Model: Current View on Its Behavioral Deficits and Modifications. Behav. Neurosci. 2021, 135, 326–335. [Google Scholar] [CrossRef]
- Han, Q.-Q.; Yang, L.; Huang, H.-J.; Wang, Y.-L.; Yu, R.; Wang, J.; Pilot, A.; Wu, G.-C.; Liu, Q.; Yu, J. Differential GR Expression and Translocation in the Hippocampus Mediates Susceptibility vs. Resilience to Chronic Social Defeat Stress. Front. Neurosci. 2017, 11, 287. [Google Scholar] [CrossRef]
- Kuehner, J.N.; Walia, N.R.; Seong, R.; Li, Y.; Martinez-Feduchi, P.; Yao, B. Social Defeat Stress Induces Genome-Wide 5mC and 5hmC Alterations in the Mouse Brain. G3 Genes Genomes Genet. 2023, 13, jkad114. [Google Scholar] [CrossRef]
- O’Toole, N.; Zhang, T.-Y.; Wen, X.; Diorio, J.; Silveira, P.P.; Labonté, B.; Nestler, E.J.; Meaney, M.J. Epigenetic Signatures of Chronic Social Stress in Stress-Susceptible Animals 2019, bioRxiv, 690826. [CrossRef]
- Wollman, R. Robustness, Accuracy, and Cell State Heterogeneity in Biological Systems. Curr. Opin. Syst. Biol. 2018, 8, 46–50. [Google Scholar] [CrossRef]
- Hebda-Bauer, E.K.; Simmons, T.A.; Sugg, A.; Ural, E.; Stewart, J.A.; Beals, J.L.; Wei, Q.; Watson, S.J.; Akil, H. 3xTg-AD Mice Exhibit an Activated Central Stress Axis During Early-Stage Pathology. J. Alzheimers Dis. 2012, 33, 407–422. [Google Scholar] [CrossRef]
- Zhou, J.-J.; Gao, Y.; Zhang, X.; Kosten, T.A.; Li, D.-P. Enhanced Hypothalamic NMDA Receptor Activity Contributes to Hyperactivity of HPA Axis in Chronic Stress in Male Rats. Endocrinology 2018, 159, 1537–1546. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Singh, R.; Arora, G.; Dangol, A.; Goyal, S. Biomarkers of Major Depressive Disorder: Knowing Is Half the Battle. Clin. Psychopharmacol. Neurosci. 2021, 19, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Kudryavtseva, N.N.; Bakshtanovskaya, I.V.; Koryakina, L.A. Social Model of Depression in Mice of C57BL/6J Strain. Pharmacol. Biochem. Behav. 1991, 38, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Avgustinovich D.F., Kovalenko I.L., Bondar N.P. Choice of “control” in experimental researches of animal social interactions in mice. Ross Fiziol Zh Im I M Sechenova. 2005, 91(12): 1454-1468. [PubMed]
- Walf, A.A.; Frye, C.A. The Use of the Elevated plus Maze as an Assay of Anxiety-Related Behavior in Rodents. Nat. Protoc. 2007, 2, 322–328. [Google Scholar] [CrossRef]
- File, S.E.; Seth, P. A Review of 25 Years of the Social Interaction Test. Eur. J. Pharmacol. 2003, 463, 35–53. [Google Scholar] [CrossRef]
- Kudryavtseva, N.N. Use of the “Partition” Test in Behavioral and Pharmacological Experiments. Neurosci. Behav. Physiol. 2003, 33, 461–471. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Le Pichon, M.; Jalfre, M. Depression: A New Animal Model Sensitive to Antidepressant Treatments. Nature 1977, 266, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Willner, P.; Towell, A.; Sampson, D.; Sophokleous, S.; Muscat, R. Reduction of Sucrose Preference by Chronic Unpredictable Mild Stress, and Its Restoration by a Tricyclic Antidepressant. Psychopharmacology (Berl.) 1987, 93. [CrossRef]
- Venzala, E.; García-García, A.L.; Elizalde, N.; Delagrange, P.; Tordera, R.M. Chronic Social Defeat Stress Model: Behavioral Features, Antidepressant Action, and Interaction with Biological Risk Factors. Psychopharmacology (Berl.) 2012, 224, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.P.; Brivio, E.; Santambrogio, A.; De Donno, C.; Kos, A.; Peters, M.; Rost, N.; Czamara, D.; Brückl, T.M.; Roeh, S.; et al. Single-Cell Molecular Profiling of All Three Components of the HPA Axis Reveals Adrenal ABCB1 as a Regulator of Stress Adaptation. Sci. Adv. 2021, 7, eabe4497. [Google Scholar] [CrossRef]
- Hassamal, S. Chronic Stress, Neuroinflammation, and Depression: An Overview of Pathophysiological Mechanisms and Emerging Anti-Inflammatories. Front. Psychiatry 2023, 14, 1130989. [Google Scholar] [CrossRef]
- Nandam, L.S.; Brazel, M.; Zhou, M.; Jhaveri, D.J. Cortisol and Major Depressive Disorder—Translating Findings From Humans to Animal Models and Back. Front. Psychiatry 2020, 10, 974. [Google Scholar] [CrossRef]
- Buwalda; De Boer; Schmidt; Felszeghy; Nyakas; Sgoifo; Van Der Vegt; Tilders; Bohus; Koolhaas Long--Lasting Deficient Dexamethasone Suppression of Hypothalamic--Pituitary--Adrenocortical Activation Following Peripheral CRF Challenge in Socially Defeated Rats. J. Neuroendocrinol. 1999, 11, 513–520. [CrossRef]
- Hennings, J.M.; Ising, M.; Uhr, M.; Holsboer, F.; Lucae, S. Effects of Weariness of Life, Suicide Ideations and Suicide Attempt on HPA Axis Regulation in Depression. Psychoneuroendocrinology 2021, 131, 105286. [Google Scholar] [CrossRef]
- Jokinen, J.; Nordström, A.-L.; Nordström, P. ROC Analysis of Dexamethasone Suppression Test Threshold in Suicide Prediction after Attempted Suicide. J. Affect. Disord. 2008, 106, 145–152. [Google Scholar] [CrossRef]
- Juruena, M.F.; Bocharova, M.; Agustini, B.; Young, A.H. Atypical Depression and Non-Atypical Depression: Is HPA Axis Function a Biomarker? A Systematic Review. J. Affect. Disord. 2018, 233, 45–67. [Google Scholar] [CrossRef]
- Geracioti, T.D.; Loosen, P.T.; Gold, P.W.; Kling, M.A. Cortisol, Thyroid Hormone, and Mood in Atypical Depression: A Longitudinal Case Study. Biol. Psychiatry 1992, 31, 515–519. [Google Scholar] [CrossRef]
- Hamilton, J.L.; Alloy, L.B. Atypical Reactivity of Heart Rate Variability to Stress and Depression across Development: Systematic Review of the Literature and Directions for Future Research. Clin. Psychol. Rev. 2016, 50, 67–79. [Google Scholar] [CrossRef]
- Brailean, A.; Curtis, J.; Davis, K.; Dregan, A.; Hotopf, M. Characteristics, Comorbidities, and Correlates of Atypical Depression: Evidence from the UK Biobank Mental Health Survey. Psychol. Med. 2020, 50, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Lyndon, B.; Parker, G.; Morris, G.; Das, P.; Outhred, T.; Hamilton, A.; Bassett, D.; Baune, B.T.; Berk, M.; Boyce, P.; et al. Is Atypical Depression Simply a Typical Depression with Unusual Symptoms? Aust. N. Z. J. Psychiatry 2017, 51, 868–871. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Tejada, J.; Arregi, A.; Gómez-Lázaro, E.; Vegas, O.; Azpiroz, A.; Garmendia, L. Coping with Chronic Social Stress in Mice: Hypothalamic-Pituitary-Adrenal/ Sympathetic-Adrenal-Medullary Axis Activity, Behavioral Changes and Effects of Antalarmin Treatment: Implications for the Study of Stress-Related Psychopathologies. Neuroendocrinology 2013, 98, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Ahuir, A.; Fita-Torró, J.; Proft, M. Capturing and Understanding the Dynamics and Heterogeneity of Gene Expression in the Living Cell. Int. J. Mol. Sci. 2020, 21, 8278. [Google Scholar] [CrossRef]
- You, S.-T.; Jhou, Y.-T.; Kao, C.-F.; Leu, J.-Y. Experimental Evolution Reveals a General Role for the Methyltransferase Hmt1 in Noise Buffering. PLOS Biol. 2019, 17, e3000433. [Google Scholar] [CrossRef]
- Tuttle, A.H.; Philip, V.M.; Chesler, E.J.; Mogil, J.S. Comparing Phenotypic Variation between Inbred and Outbred Mice. Nat. Methods 2018, 15, 994–996. [Google Scholar] [CrossRef]
- Sabbagh, J.J.; O’Leary, J.C.; Blair, L.J.; Klengel, T.; Nordhues, B.A.; Fontaine, S.N.; Binder, E.B.; Dickey, C.A. Age-Associated Epigenetic Upregulation of the FKBP5 Gene Selectively Impairs Stress Resiliency. PLoS ONE 2014, 9, e107241. [Google Scholar] [CrossRef]
- Silva, R.C.; Dattilo, V.; Perusi, G.; Mazzelli, M.; Maffioletti, E.; Bazzanella, R.; Bortolomasi, M.; Cattaneo, A.; Gennarelli, M.; Minelli, A. Transcriptional Modulation of Stress-Related Genes in Association with Early Life Stress Exposure and Trauma-Focused Psychotherapy in Treatment-Resistant Depression Patients. J. EMDR Pract. Res. 2023, 17, 119–138. [Google Scholar] [CrossRef]
- Amasi-Hartoonian, N.; Sforzini, L.; Cattaneo, A.; Pariante, C.M. Cause or Consequence? Understanding the Role of Cortisol in the Increased Inflammation Observed in Depression. Curr. Opin. Endocr. Metab. Res. 2022, 24, 100356. [Google Scholar] [CrossRef]
- Daley--Yates, P.T. Inhaled Corticosteroids: Potency, Dose Equivalence and Therapeutic Index. Br. J. Clin. Pharmacol. 2015, 80, 372–380. [Google Scholar] [CrossRef]
- Karssen, A.M.; Meijer, O.C.; Berry, A.; Sanjuan Piñol, R.; De Kloet, E.R. Low Doses of Dexamethasone Can Produce a Hypocorticosteroid State in the Brain. Endocrinology 2005, 146, 5587–5595. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS : A Free, Versatile Open--source Event--logging Software for Video/Audio Coding and Live Observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- Khotskin, N.V.; Fursenko, D.V.; Bazovkina, D.V.; Kulikov, V.A.; Kulikov, A.V. Automatic Measurement of Spatial Learning Characteristics in Mice in an Underlit Morris Water Maze. Neurosci. Behav. Physiol. 2015, 45, 771–775. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



