Submitted:
16 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
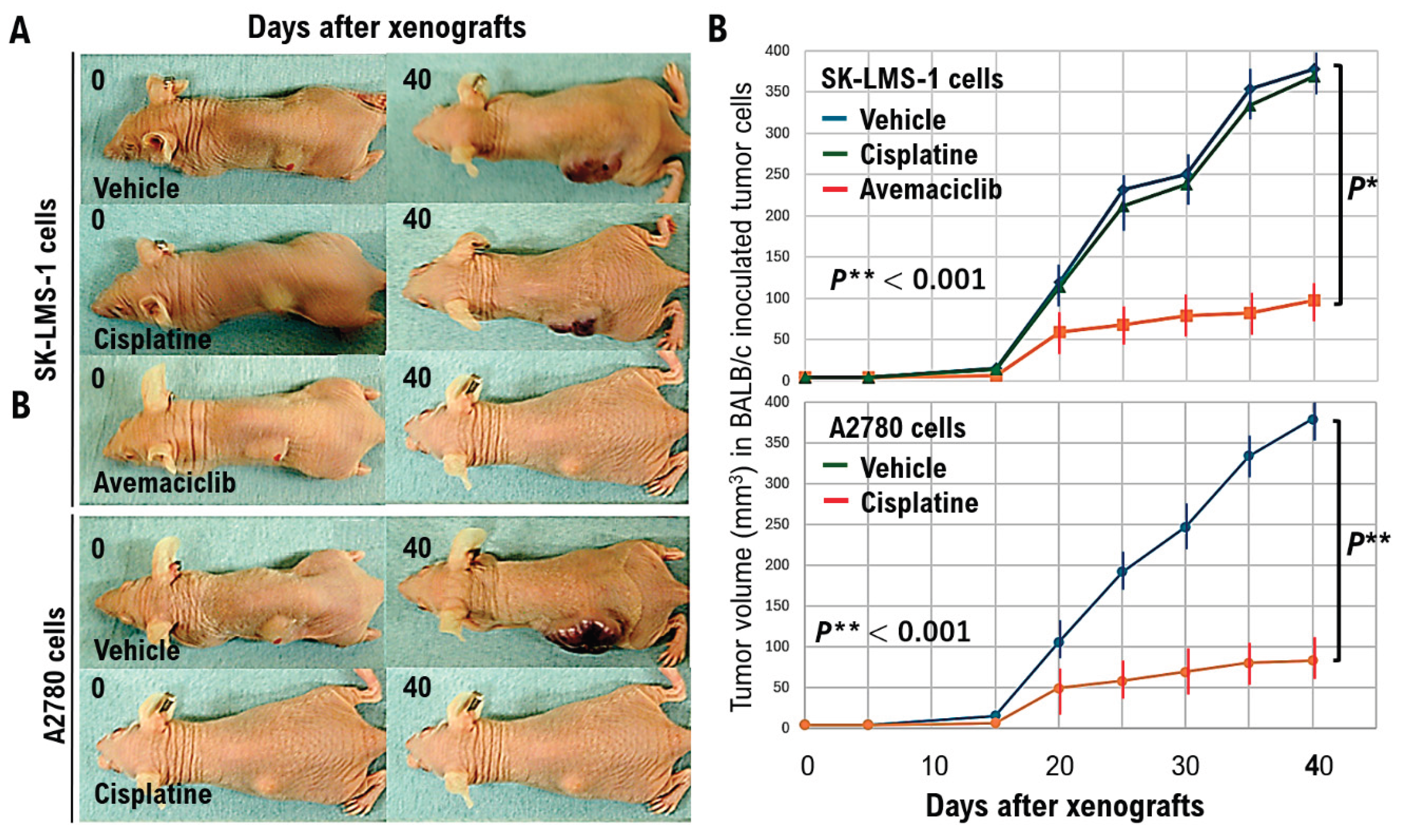
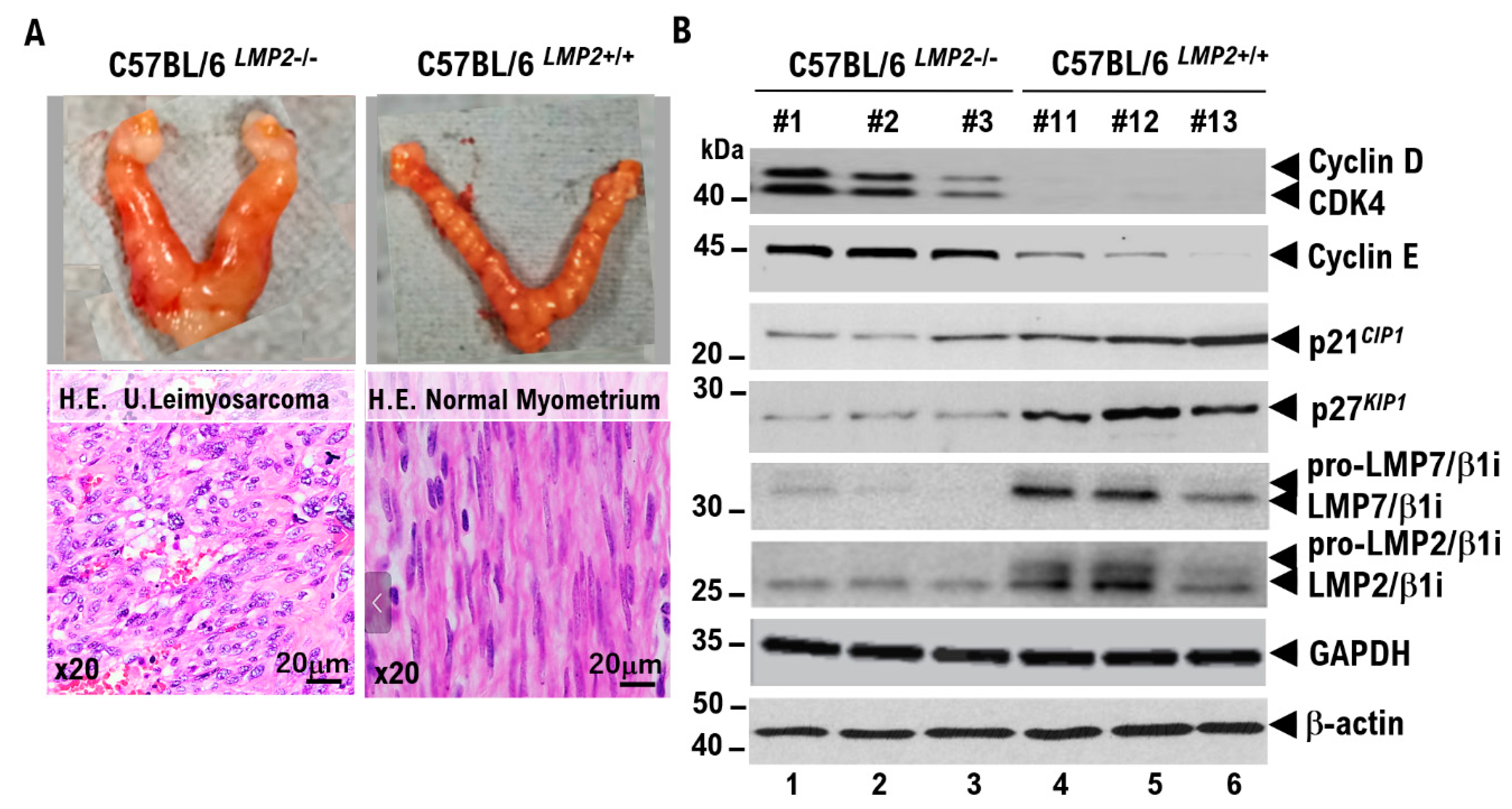
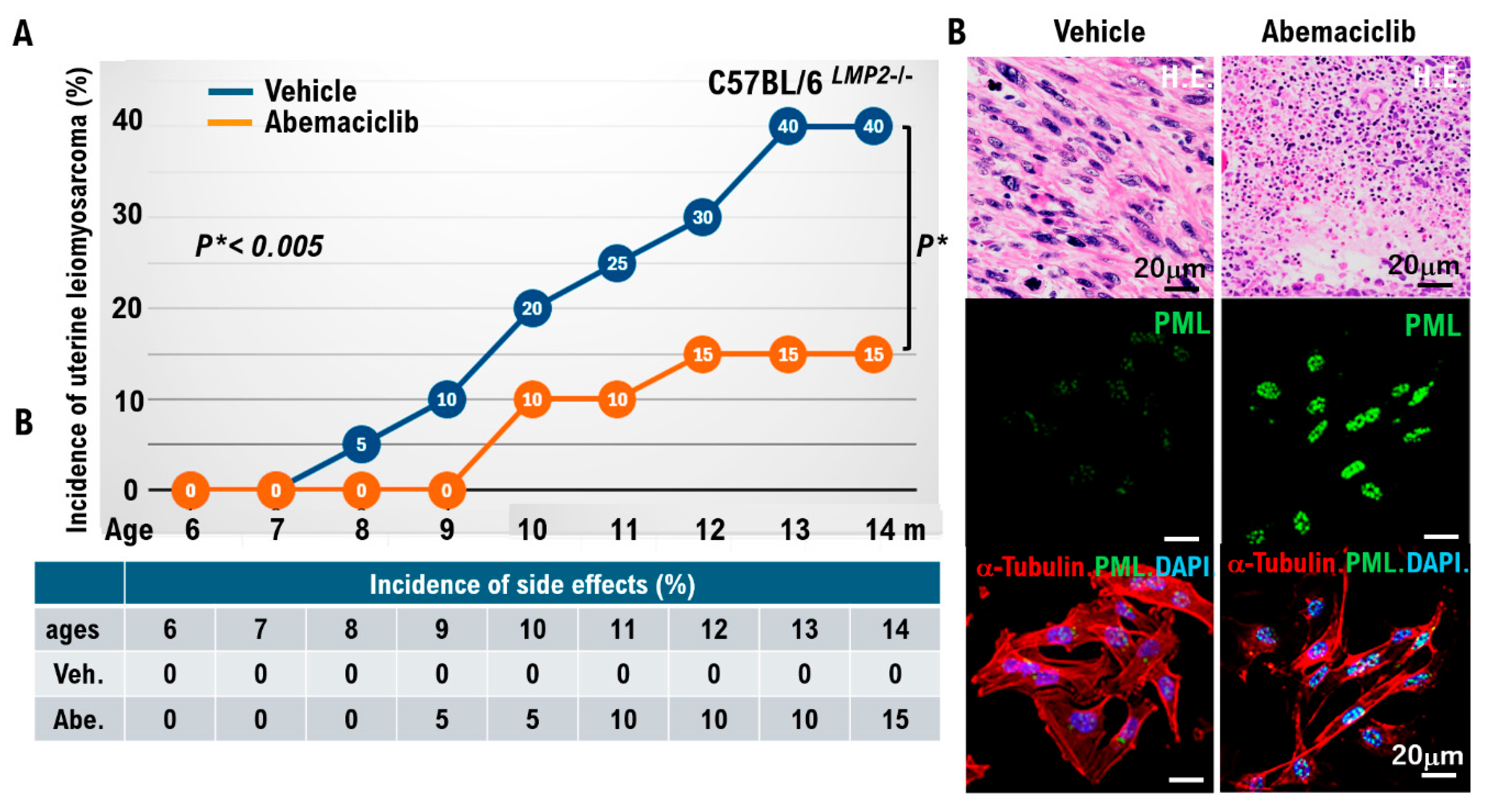
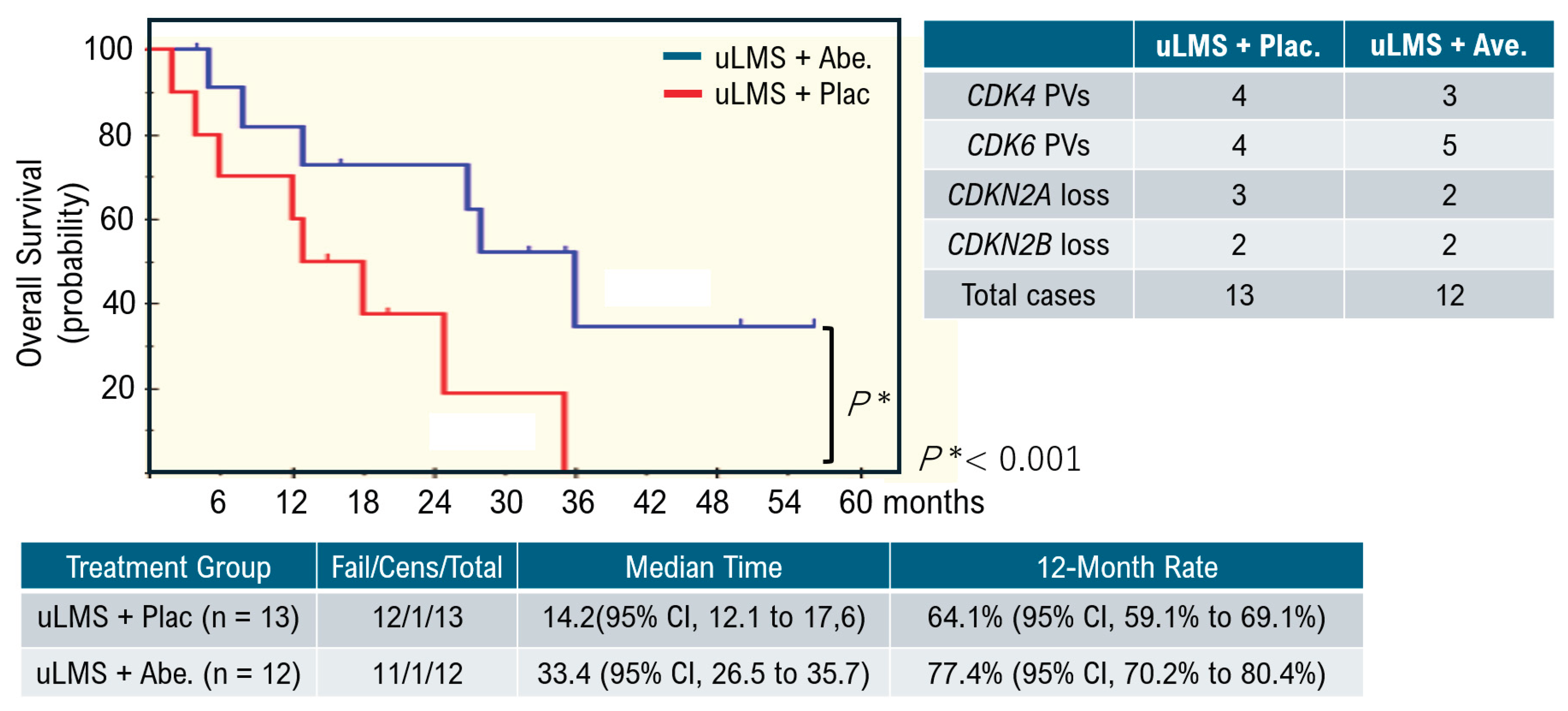
Uterine smooth muscle cells form the uterine framework. The myometrium is the middle layer of the uterine wall, consisting primarily of uterine smooth muscle cells (also called uterine myocytes) but also supports stromal and vascular tissues. Smooth muscle cells differentiate from stem cells. Broadly, two types of tumor cells derived from uterine smooth muscle cells can arise in the uterine smooth muscle layer. Benign uterine leiomyomas and malignant uterine leiomyosarcomas arise from the uterine smooth muscle layer. Because uterine leiomyoma cells display diverse morphologies, differential diagnosis from other uterine smooth muscle tumors can be challenging. Uterine leiomyosarcoma also has a poor prognosis due to frequent metastasis and recurrence. Overseas clinical trials have shown limited efficacy and noted adverse effects of existing antitumor agents. Therefore, we analyzed molecular pathological characteristics of uterine leiomyosarcoma in a spontaneous mouse model (C57BLACK/6LMP2-/-) that we previously reported in collaboration with Professor Susumu Tonegawa of the MIT–Picower Institute. We identified activation of cell-cycle G0/G1 inducers. Intraperitoneal abemaciclib, which targets cell cycle G0/G1 inducers (CDK4/cyclin D, CDK2/cyclin E), in C57BLACK/6LMP2-/- mice reduced uterine leiomyosarcoma incidence in C57BLACK/6LMP2-/- mice. We then enrolled 25 patients with advanced metastatic uterine leiomyosarcoma who had activating pathogenic variants in G0/G1-inducing factors identified by cancer gene panel testing. They were assigned to the abemaciclib or placebo groups, and median treatment time and 12-month treatment rate were compared. Results demonstrated efficacy in patients with advanced metastatic uterine leiomyosarcoma with activating cell-cycle variants. Because this trial involved a small cohort, larger studies are warranted.
Keywords:
Introduction
Diagnostic Methods
Prognosis and Treatment
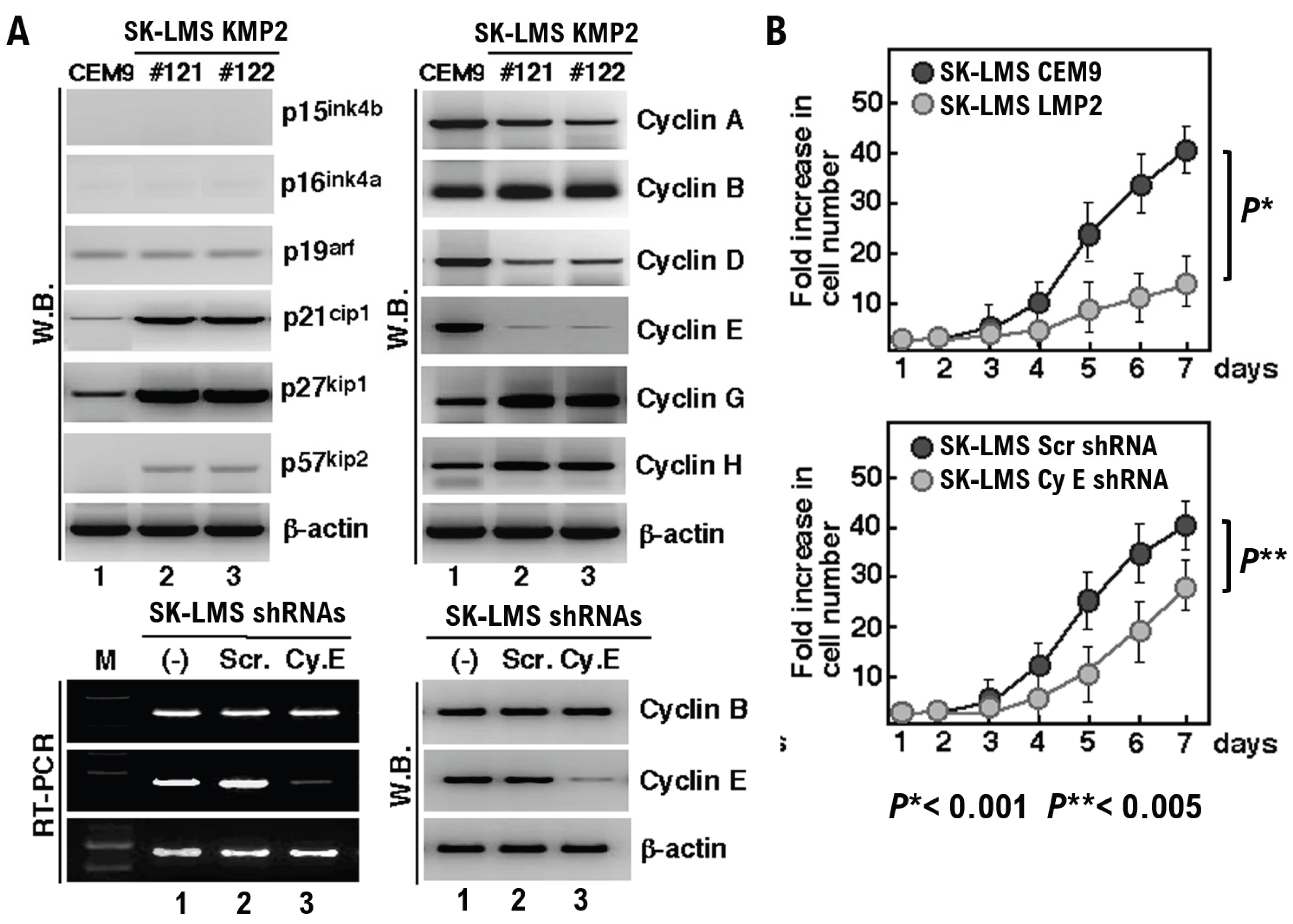
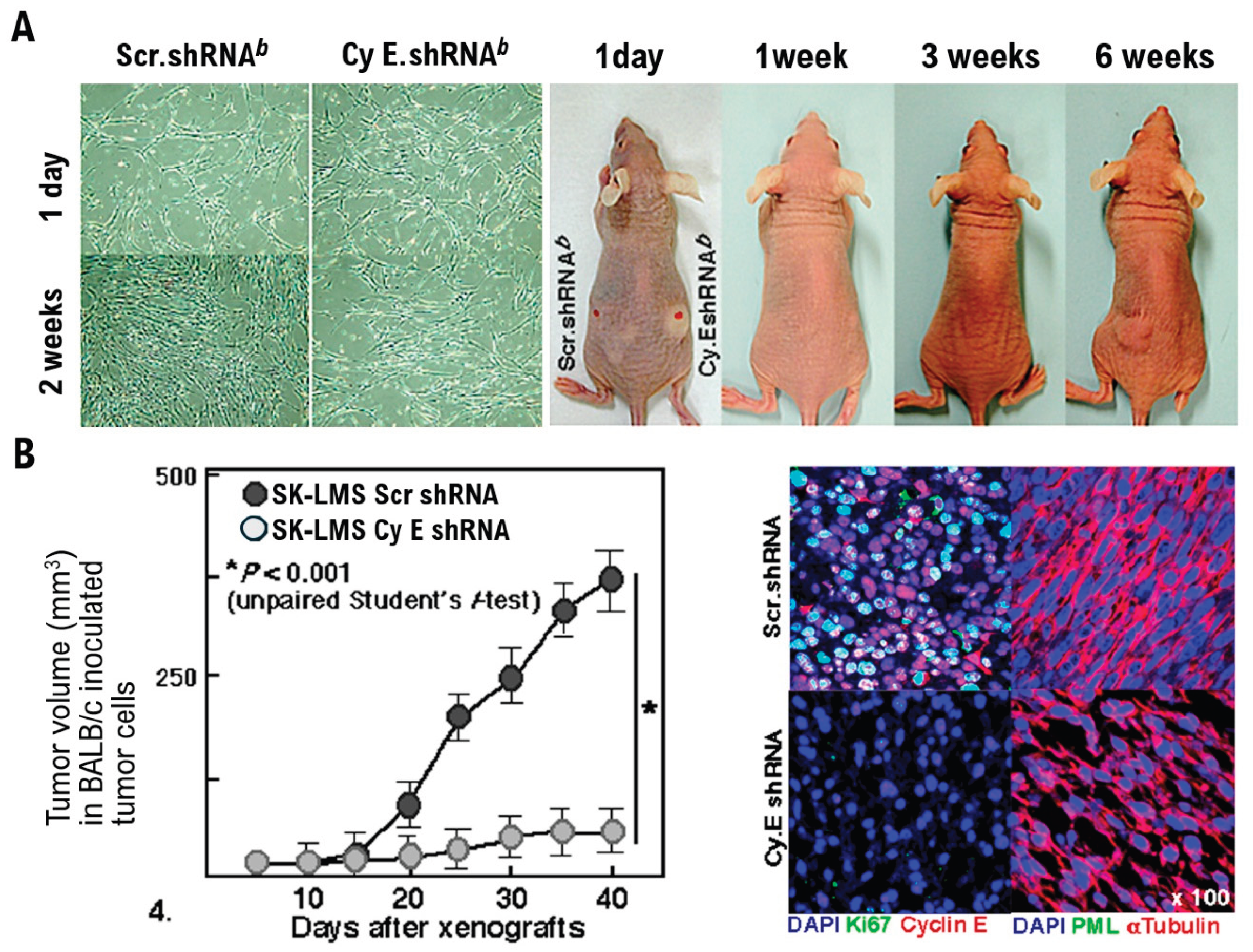
Results
Discussion
Material and Methods
- 1.
- DNA Transfection and Isolation of Flat Revertants
- 2.
- Reverse Transcription-polymerase Chain Reaction Analysis (RT-PCR)
- 3.
- Flow Cytometry
- 4.
- Xenograft Studies
- 6.
- IHC
- 7.
- b-Galactosidase staining
- 8.
- Ethics approval and consent to participate
- 9.
- Ethical compliance with human study
ARRIVE Checklist Documentation
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aguilar HN, Mitchell BF. Physiological pathways and molecular mechanisms regulating uterine contractility. Hum Reprod Update. 2010 Nov-Dec;16(6):725-44. [CrossRef]
- NCI Dictionary of Cancer Terms”. National Cancer Institute. Retrieved 2017-12-27.
- Gu W, Hong X, Le Bras A, Nowak WN, Issa Bhaloo S, Deng J, Xie Y, Hu Y, Ruan XZ, Xu Q. Smooth muscle cells differentiated from mesenchymal stem cells are regulated by microRNAs and suitable for vascular tissue grafts. J Biol Chem. 2018 May 25;293(21):8089-8102. [CrossRef]
- Swartz CD, Afshari CA, Yu L, Hall KE, Dixon D. Estrogen-induced changes in IGF-I, Myb family and MAP kinase pathway genes in human uterine leiomyoma and normal uterine smooth muscle cell lines. Mol Hum Reprod. 2005 Jun;11(6):441-50. [CrossRef]
- Preoperative medical therapy before surgery for uterine fibroids. Puscasiu L, Vollenhoven B, Nagels HE, Melinte IM, Showell MG, Lethaby A. Cochrane Database Syst Rev. 2025 Apr 4;4(4):CD000547. [CrossRef]
- Leitao MM, Soslow RA, Nonaka D, Olshen AB, Aghajanian C, Sabbatini P, Dupont J, Hensley M, Sonoda Y, Barakat RR, Anderson S. Tissue microarray immunohistochemical expression of estrogen, progesterone, and androgen receptors in uterine leiomyomata and leiomyosarcoma. Cancer. 2004 Sep 15;101(6):1455-62. [CrossRef]
- De Bruyn C, Ceusters J, Vanden Brande K, Timmerman S, Froyman W, Timmerman D, Van Rompuy AS, Coosemans A, Van den Bosch T. Ultrasound features using MUSA terms and definitions in uterine sarcoma and leiomyoma: cohort study. Ultrasound Obstet Gynecol. 2024 May;63(5):683-690. [CrossRef]
- Callegaro D, Fiore M, Gronchi A. Personalizing surgical margins in retroperitoneal sarcomas. Expert Rev Anticancer Ther. 2015 May;15(5):553-67. [CrossRef]
- leiomyosarcoma symptoms Sarcoma; information for leiomyosarcoma families. https://www.leiomyosarcoma.org/symptoms/.
- Adams CL, Dimitrova I, Post MD, Gibson L, Spillman MA, Behbakht K, Bradford AP. Identification of a novel diagnostic gene expression signature to discriminate uterine leiomyoma from leiomyosarcoma. Exp Mol Pathol. 2019 Oct;110:104284. [CrossRef]
- Matsumoto Y, Hayashi T, Amano Y, Abiko K, Konishi I. DEVELOPMENT OF ENDOSALPINGIOSIS IN PATIENTS WITH A HISTORY OF BREAST CANCER. Georgian Med News. 2024 Oct;(355):72-76.
- Xu X, Zhao S, Xue L. Leiomyosarcomas of the inferior vena cava: diagnostic features on contrast-enhanced CT, ultrasonography and MRI. Front Oncol. 2025 Feb 4;15:1442674. eCollection 2025. [CrossRef]
- Klein CR, Koob S, Tischler V, Heine A, Brossart P, Feldmann G, Mayer K. Chronification of metastatic leiomyosarcoma in 9 lines of therapy by precision oncology: a case report and review of the literature. Front Oncol. 2025 Sep 2;15:1626478. eCollection 2025. [CrossRef]
- unç M, Akıllı H, Kuşçu E, Ayhan A. Timing and survival benefits of cytoreduction in patients with recurrent leiomyosarcoma. T Arch Gynecol Obstet. 2025 Oct 17. [CrossRef]
- Gadducci A, Guerrieri ME. Pharmacological treatment for uterine leiomyosarcomas. Expert Opin Pharmacother. 2015 Feb;16(3):335-46. [CrossRef]
- Hayashi T, Faustman DL. Development of spontaneous uterine tumors in low molecular mass polypeptide-2 knockout mice. Cancer Res. 2002 Jan 1;62(1):24-7.
- Hayashi T, Horiuchi A, Sano K, Hiraoka N, Kanai Y, Shiozawa T, Tonegawa S, Konishi I. Mice-lacking LMP2, immuno-proteasome subunit, as an animal model of spontaneous uterine leiomyosarcoma. Protein Cell. 2010 Aug;1(8):711-7. [CrossRef]
- Abemaciclib; https://en.wikipedia.org/wiki/Abemaciclib.
- Hayashi T, Horiuchi A, Sano K, Hiraoka N, Kasai M, Ichimura T, Sudo T, Tagawa Y, Nishimura R, Ishiko O, Kanai Y, Yaegashi N, Aburatani H, Shiozawa T, Konishi I. Potential role of LMP2 as tumor-suppressor defines new targets for uterine leiomyosarcoma therapy. Sci Rep. 2011;1:180. [CrossRef]
- Geng, Y., Yu, Q., Sicinska, E., Das, M., Schneider, J. E., Bhattacharya, S., Rideout, W. M., III, Bronson, R. T., Gardner, H., Sicinski, P. Cyclin E ablation in the mouse. Cell 114: 431-443, 2003.
- Diermeier-Daucher S, Brockhoff G. Dynamic proliferation assessment in flow cytometry. Curr Protoc Cell Biol. 2010 Sep;Chapter 8:Unit 8.6.1-23. [CrossRef]
- Wang ZG, Ruggero D, Ronchetti S, Zhong S, Gaboli M, Rivi R, Pandolfi PP. PML is essential for multiple apoptotic pathways. Nat Genet. 1998 Nov;20(3):266-72. [CrossRef]
- Bernardi R, Papa A, Pandolfi PP. Regulation of apoptosis by PML and the PML-NBs. Oncogene. 2008 Oct 20;27(48):6299-312. [CrossRef]
- Cai Y, Zhou H, Zhu Y, Sun Q, Ji Y, Xue A, Wang Y, Chen W, Yu X, Wang L, Chen H, Li C, Luo T, Deng H. Elimination of senescent cells by beta-galactosidase-targeted prodrug attenuates inflammation and restores physical function in aged mice. Cell Res. 2020 Jul;30(7):574-589. [CrossRef]
- Valieva Y, Ivanova E, Fayzullin A, Kurkov A, Igrunkova A. Senescence-Associated beta-Galactosidase Detection in Pathology. Diagnostics (Basel). 2022 Sep 25;12(10):2309. [CrossRef]
- Van Kaer L, Ashton-Rickardt PG, Eichelberger M, Gaczynska M, Nagashima K, Rock KL, Goldberg AL, Doherty PC, Tonegawa S. Altered peptidase and viral-specific T cell response in LMP2 mutant mice. Immunity. 1994 Oct;1(7):533-41. [CrossRef]
- 276. https://www.foundationmedicine.com/test/foundationone-cdx.
- 287. poly (ADP-ribose) polymerase (PARP) inhibitors; https://www.cancer.gov/publications/dictionaries/cancer-terms/def/poly-adp-ribose-polymerase-inhibitor.
- Han Y, Wei L. Novel clinical potential of poly (ADP-ribose) polymerase inhibitors in triple-negative breast cancer: Mechanistic insights and clinical applications (Review). Oncol Lett. 2025 Mar 4;29(5):215. [CrossRef]
- Pautier P, Italiano A, Piperno-Neumann S, Chevreau C, Penel N, Firmin N, Boudou-Rouquette P, Bertucci F, Lebrun-Ly V, Ray-Coquard I, Kalbacher E, Bompas E, Collard O, Isambert N, Guillemet C, Rios M, Le Cesne A, Balleyguier C, Archambaud B, Duffaud F; French Sarcoma Group. Doxorubicin-Trabectedin with Trabectedin Maintenance in Leiomyosarcoma. N Engl J Med. 2024 Sep 5;391(9):789-799. [CrossRef]
- 31. Pautier P, Italiano A, Piperno-Neumann S, Chevreau C, Penel N, Firmin N, Boudou-Rouquette P, Bertucci F, Balleyguier C, Lebrun-Ly V, Ray-Coquard I, Kalbacher E, Bardet A, Bompas E, Collard O, Isambert N, Guillemet C, Rios M, Archambaud B, Duffaud F; French Sarcoma Group. Doxorubicin alone versus doxorubicin with trabectedin followed by trabectedin alone as first-line therapy for metastatic or unresectable leiomyosarcoma (LMS-04): a randomised, multicentre, open-label phase 3 trial. Lancet Oncol. 2022 Aug;23(8):1044-1054. [CrossRef]
- Pollack SM, Redman MW, Baker KK, Wagner MJ, Schroeder BA, Loggers ET, Trieselmann K, Copeland VC, Zhang S, Black G, McDonnell S, Gregory J, Johnson R, Moore R, Jones RL, Cranmer LD. Assessment of Doxorubicin and Pembrolizumab in Patients With Advanced Anthracycline-Naive Sarcoma: A Phase 1/2 Nonrandomized Clinical Trial. JAMA Oncol. 2020 Nov 1;6(11):1778-1782. [CrossRef]
- Hensley ML, Maki R, Venkatraman E, Geller G, Lovegren M, Aghajanian C, Sabbatini P, Tong W, Barakat R, Spriggs DR. Gemcitabine and docetaxel in patients with unresectable leiomyosarcoma: results of a phase II trial. J Clin Oncol. 2002 Jun 15;20(12):2824-31. [CrossRef]
- Hensley ML, Miller A, O’Malley DM, Mannel RS, Behbakht K, Bakkum-Gamez JN, Michael H. Randomized phase III trial of gemcitabine plus docetaxel plus bevacizumab or placebo as first-line treatment for metastatic uterine leiomyosarcoma: an NRG Oncology/Gynecologic Oncology Group study.J Clin Oncol. 2015 Apr 1;33(10):1180-5. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



