Submitted:
14 November 2025
Posted:
14 November 2025
You are already at the latest version
Abstract
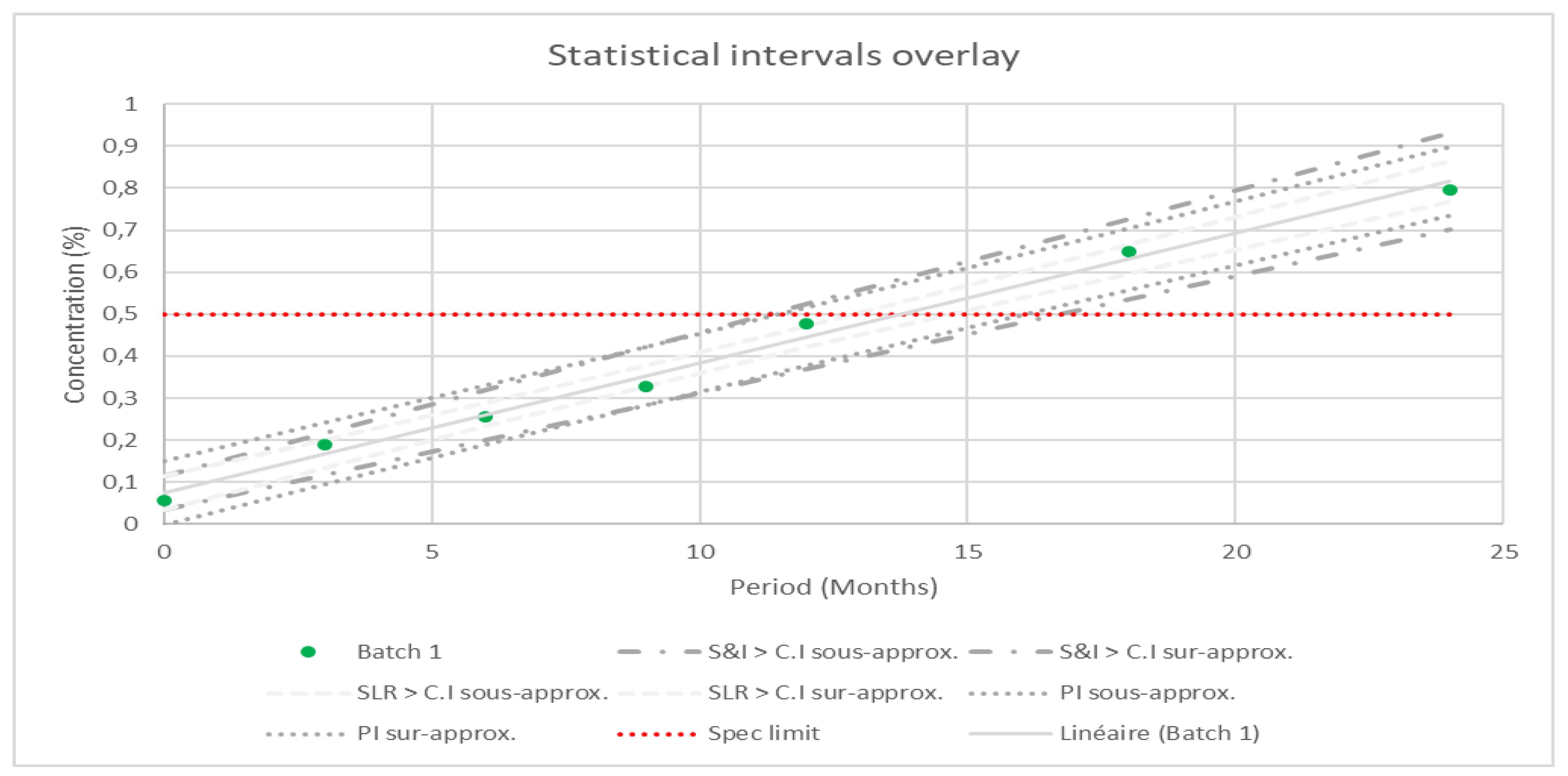
Background/Objectives: The stability of pharmaceutical compounds is a critical quality attribute; it is an essential step in the drug development process. Significant focus is required to understand the variation of quality pharmaceutical compounds under prevailing environmental storage conditions. Simultaneously, many issues arise in understanding updated regulations, knowledge of data sciences, and appreciation of common practices, presenting a challenge for defining a retest period and in predicting a prolongation of the shelf life of drug products. The purpose of this paper is to conduct a statistical study to assess stability and to forecast a prolongation of drugs shelf-life. Methods: A case study is suggested to identify the most appropriate statistical test for assessing stability. The results of physical and chemical tests are considered to detect changes and variability during different conditions (accelerate, intermediate and real). Results: In the stability study, minimal variability in the content of the substance of interest was obtained using the predictive interval approach over a period of 31 months, and an interval of ±1,2%. Conclusion: The example of the statistical study is given to provide different perspectives on statistical approaches for market approval.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Background
2.2. Basic Statistics
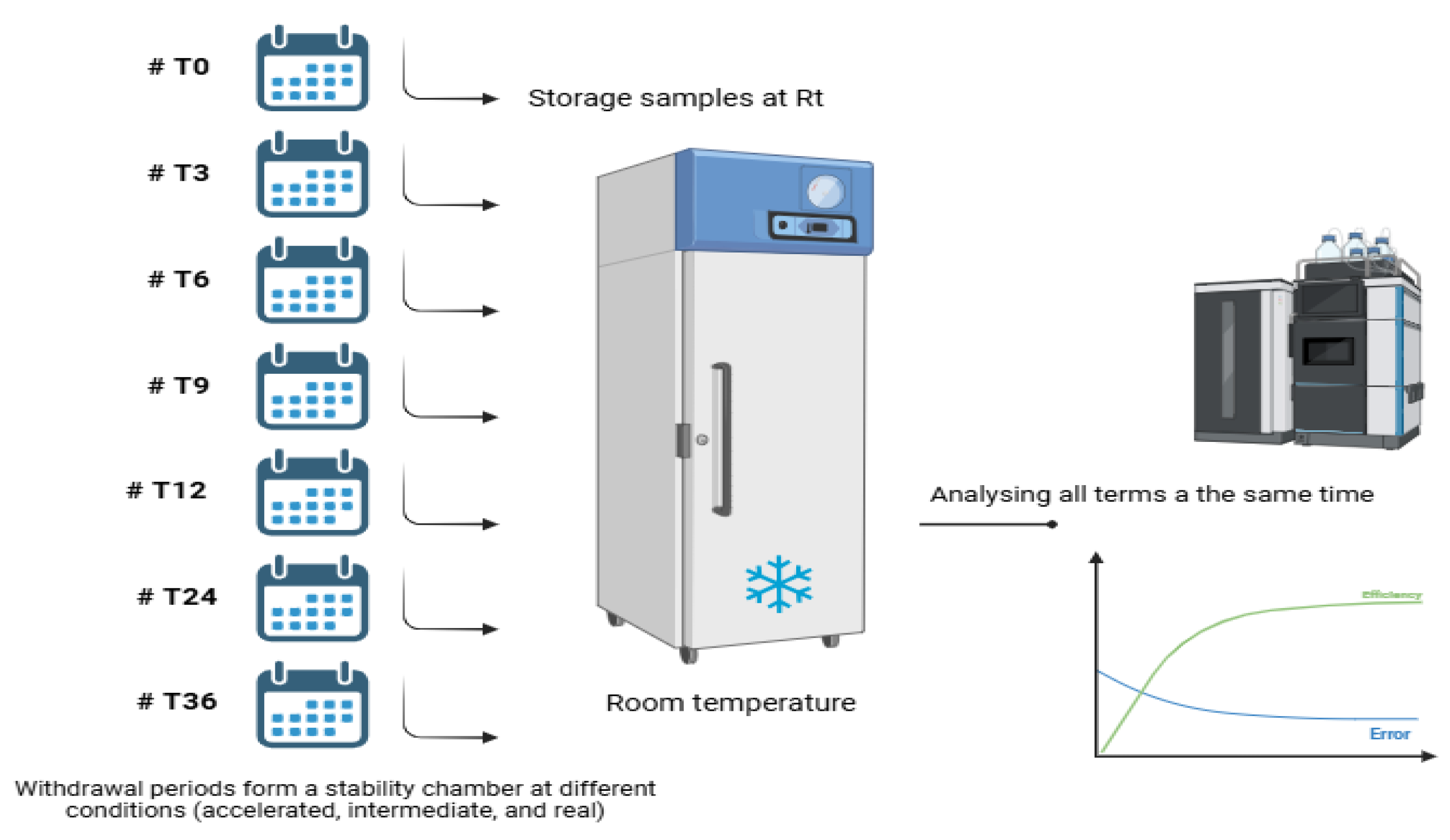
2.3. Stability Evaluation
2.4. Statistical Interval
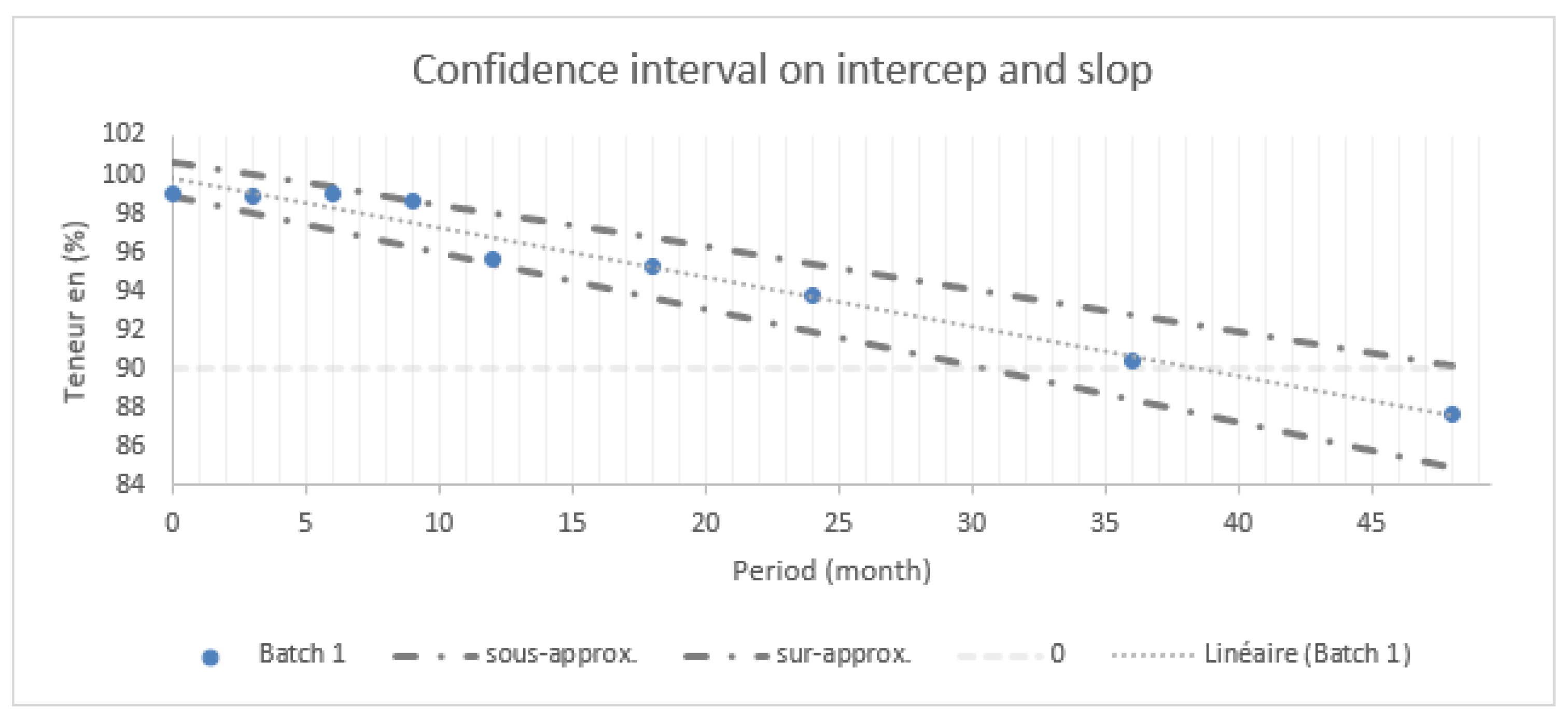
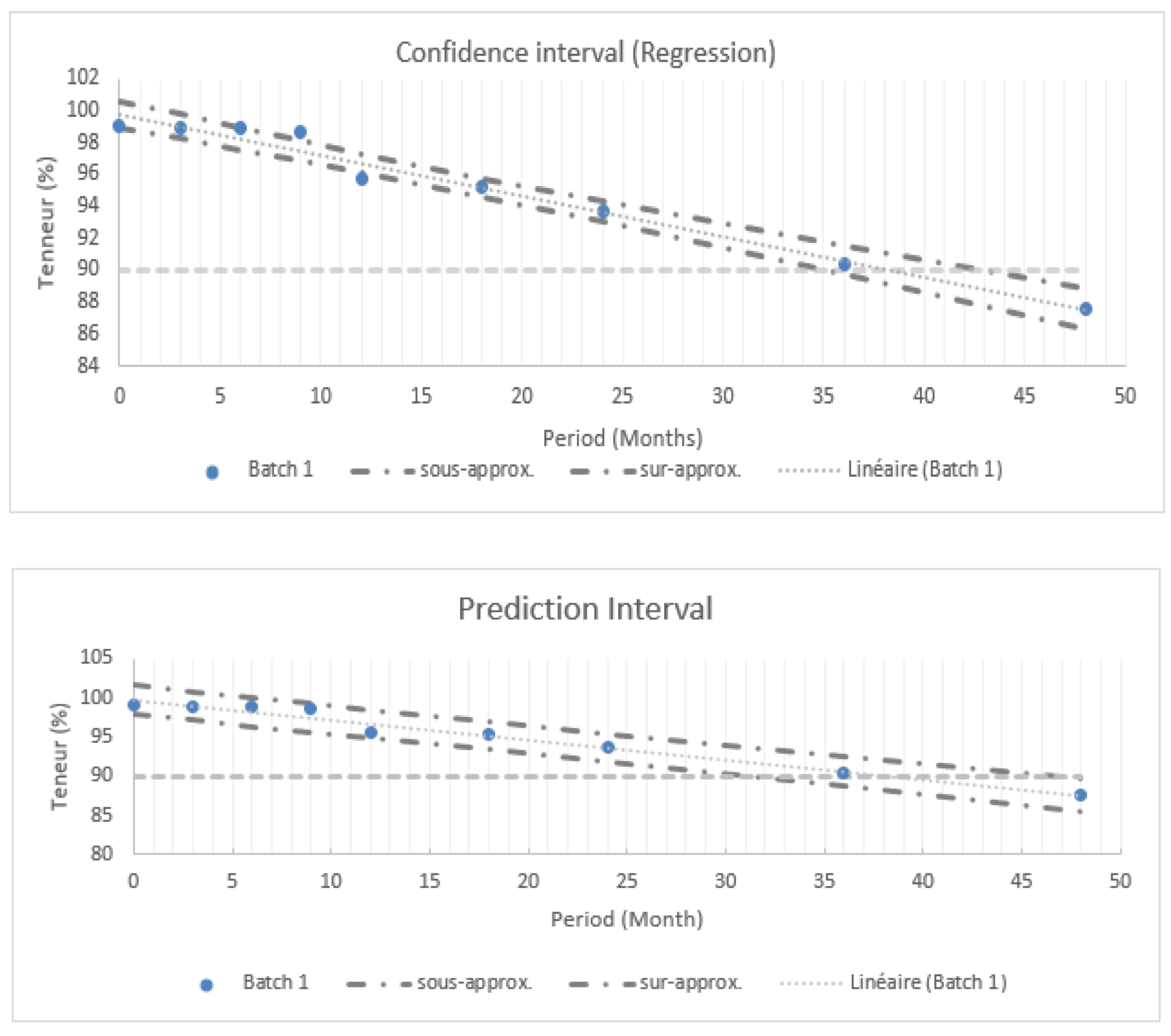
2.4.1. Confidence Interval
2.4.2. Prediction Interval
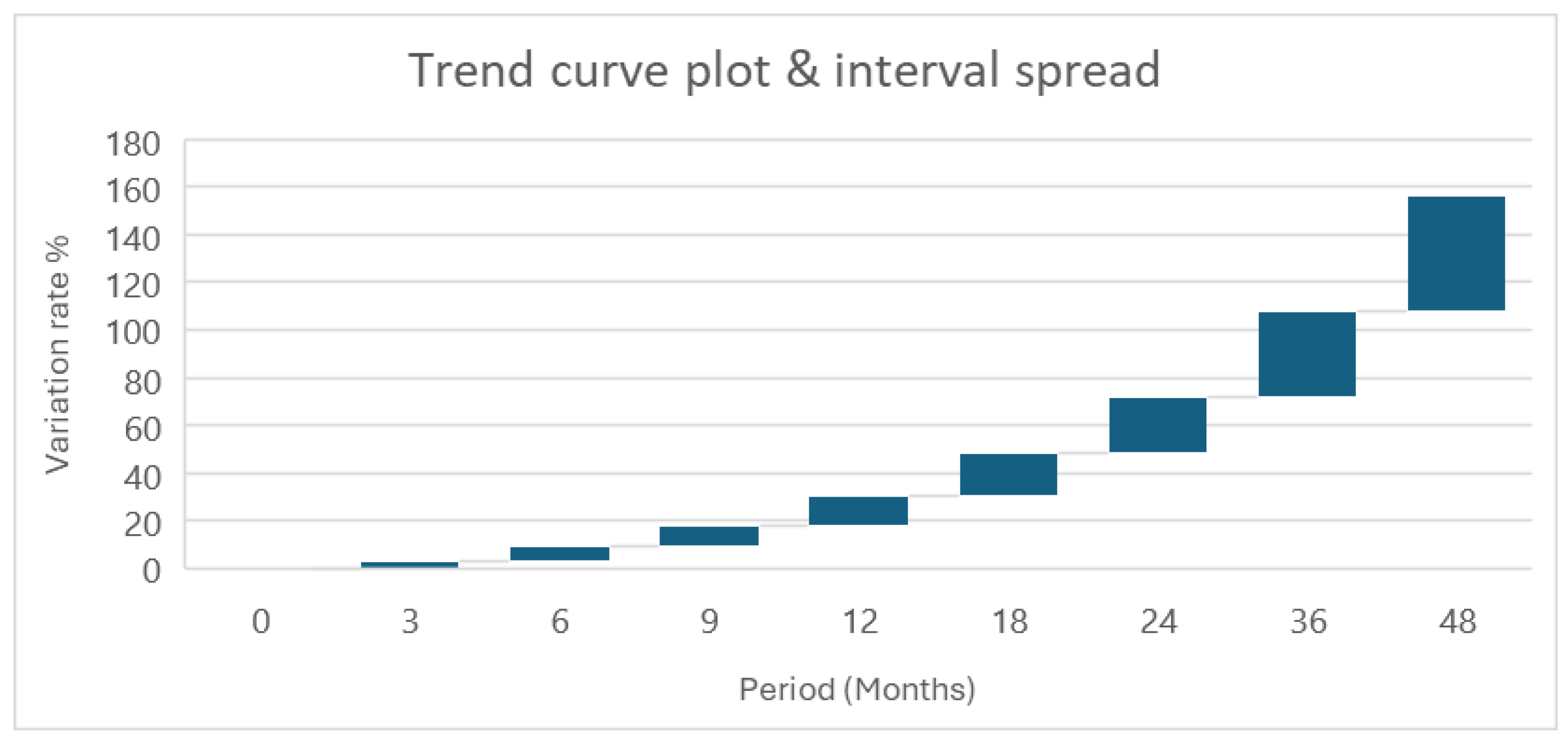
2.5. Stability Uncertainty
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jamrógiewicz, M. & Pieńkowska, K. (2019) TrAC Trends in Analytical Chemistry 111, 118–127. [CrossRef]
- Guideline, I.C.H.H.T. (2003) ICH Q1E. ICH 6, 1–19.
- Guideline, I.H.T. (2003) Q1A (R2), current step 4.
- Khan, H., Ali, M., Ahuja, A., & Ali, J. (2010) Curr Pharm Anal 6, 142–150. [CrossRef]
- Huynh-Ba, K. (2009) Handbook of stability testing in pharmaceutical development: regulations, methodologies, and best practices, Springer. [CrossRef]
- Suresh Kumar, B. V, Kulshrestha, P., & Shiromani, S. (2018) Methods for Stability Testing of Pharmaceuticals 233–260. [CrossRef]
- Yu, B., Zeng, L., Ren, P., & Yang, H. (2018) Stat Biopharm Res 10, 237–243.
- Schneid, S.C., Stärtzel, P.M., Lettner, P., & Gieseler, H. (2011) Pharm Dev Technol 16, 583–590. [CrossRef]
- Capen, R., Christopher, D., Forenzo, P., Huynh-Ba, K., LeBlond, D., Liu, O., O’Neill, J., Patterson, N., Quinlan, M., & Rajagopalan, R. (2018) AAPS PharmSciTech 19, 668–680. [CrossRef]
- Lyon, R.C., Taylor, J.S., Porter, D.A., Prasanna, H.R., & Hussain, A.S. (2006) J Pharm Sci 95, 1549–1560. [CrossRef]
- Mihalovits, M. & Kemény, S. (2020) J Pharm Biomed Anal 188, 113375. [CrossRef]
- Faya, P., Seaman Jr, J.W., & Stamey, J.D. (2018) Stat Med 37, 2599–2615. [CrossRef]
- Buda, V., Baul, B., Andor, M., Man, D.E., Ledeţi, A., Vlase, G., Vlase, T., Danciu, C., Matusz, P., & Peter, F. (2020) Pharmaceutics 12, 86. [CrossRef]
- David, H.A. (1998) Statistical Science 368–377.
- Dodge, Y. (2003) The Oxford dictionary of statistical terms, Oxford University Press, USA.
- Everitt, B.S. & Skrondal, A. (2010).
- Islam, M.A., Al-Shiha, A., Islam, M.A., & Al-Shiha, A. (2018) Foundations of Biostatistics 39–72. [CrossRef]
- Frost, J. (2022) Statistics by Jim. https://statisticsbyjim.com/basics/standard-deviation.
- Frost, J. (2021) Statistics by Jim.
- Wang, E.W., Ghogomu, N., Voelker, C.C.J., Rich, J.T., Paniello, R.C., Nussenbaum, B., Karni, R.J., & Neely, J.G. (2009) Otolaryngology—Head and Neck Surgery 140, 794–799. [CrossRef]
- Meeker, W.Q., Hahn, G.J., & Escobar, L.A. (2017) Statistical intervals: a guide for practitioners and researchers, John Wiley & Sons.
- Huynh-Ba, K. (2009) Handbook of stability testing in pharmaceutical development: regulations, methodologies, and best practices, Springer. [CrossRef]
- Waterman, K.C. (2009) in Handbook of stability testing in pharmaceutical development: regulations, methodologies, and best practices, Springer, pp 115–135.
- Chau, J., Altan, S., Burggraeve, A., Coppenolle, H., Kifle, Y.W., Prokopcova, H., Van Daele, T., & Sterckx, H. (2023) AAPS PharmSciTech 24, 250. [CrossRef]
- Xu, Z., Ding, Z., Zhang, Y., Liu, X., Wang, Q., Shao, S., & Liu, Q. (2023) Virus Res 323, 198997. [CrossRef]
- Evers, A., Clénet, D., & Pfeiffer-Marek, S. (2022) Pharmaceutics 14, 375. [CrossRef]
- Singh, S., Junwal, M., Modhe, G., Tiwari, H., Kurmi, M., Parashar, N., & Sidduri, P. (2013) TrAC Trends in Analytical Chemistry 49, 71–88. [CrossRef]
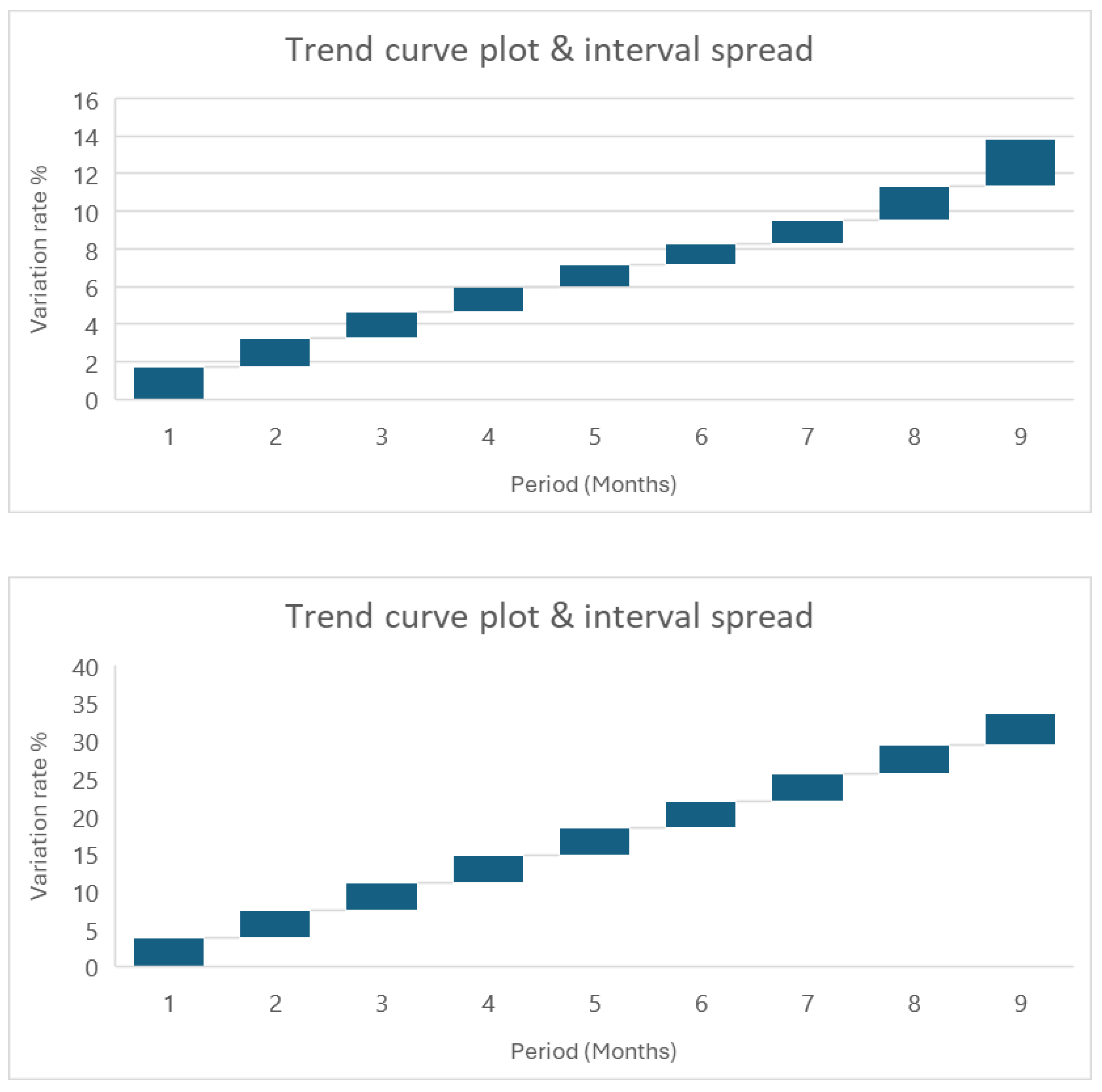
| Times (month) | 35 °C | 45 °C | 55 °C | 65 °C |
| 1 | 99,2 | 98,7 | 97 | 94,3 |
| 2 | 98,7 | 97 | 93,4 | 87,9 |
| 3 | 98,5 | 96,4 | 90,5 | 79,1 |
| Parameters | 35 °C | 45 °C | 55 °C | 65 °C |
|---|---|---|---|---|
| Variance | 0,1 | 1,4 | 10,6 | 58,2 |
| Standard deviation | 0,36 | 1,19 | 3,26 | 7,63 |
| Absolute change | 0,7 | 2,3 | 6,5 | 15,2 |
| Relative change | 0,007 | 0,023 | 0,067 | 0,161 |
| Percentage change | 0,7 | 2,3 | 6,7 | 16,1 |
| Confidence Interval | 35 °C | 45 °C | 55 °C | 65 °C |
|---|---|---|---|---|
| Upper limits interval | 99,7 | 100,3 | 101,7 | 106,1 |
| Lower limits interval | 97,9 | 94,4 | 85,5 | 68,1 |
| Temperature °C | Calculated statistic | Critical value |
| 35 | 4,04 | 12,70 |
| 45 | 3,62 | |
| 55 | 16,08 | |
| 65 | 10,96 |
| 0 | 3 | 6 | 9 | 12 | 18 | 24 | 36 | 48 | |
| Batch 1 | 99,01 | 98,9 | 98,97 | 98,64 | 95,7 | 95,28 | 93,74 | 90,35 | 87,61 |
| Batch 2 | 101,13 | 99,45 | 97,71 | 98,53 | 97,22 | 95,87 | 93,41 | 90,34 | 88,04 |
| Batch 3 | 99,63 | 99,39 | 99,29 | 97,45 | 96,23 | 94,38 | 93,53 | 90,47 | 87,06 |
| Approach | Time period (Month) | Uncertainty (%) |
|---|---|---|
| SLR-CI | 35 months | ± 1,40 |
| S&I-CI | 30 months | ± 1,24 |
| PI | 31 months | ± 1,20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




