Submitted:
13 November 2025
Posted:
14 November 2025
You are already at the latest version
Abstract
Keywords:
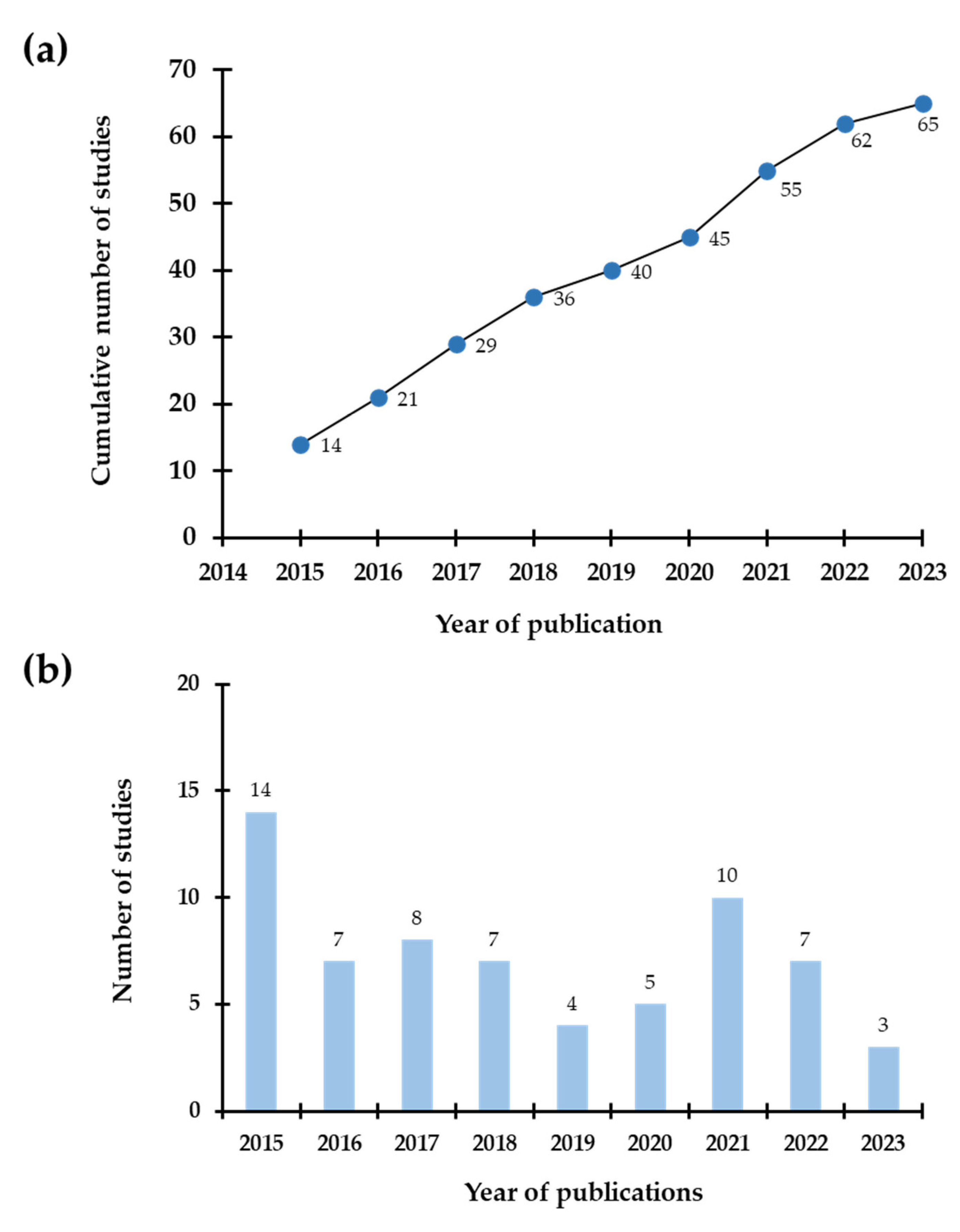
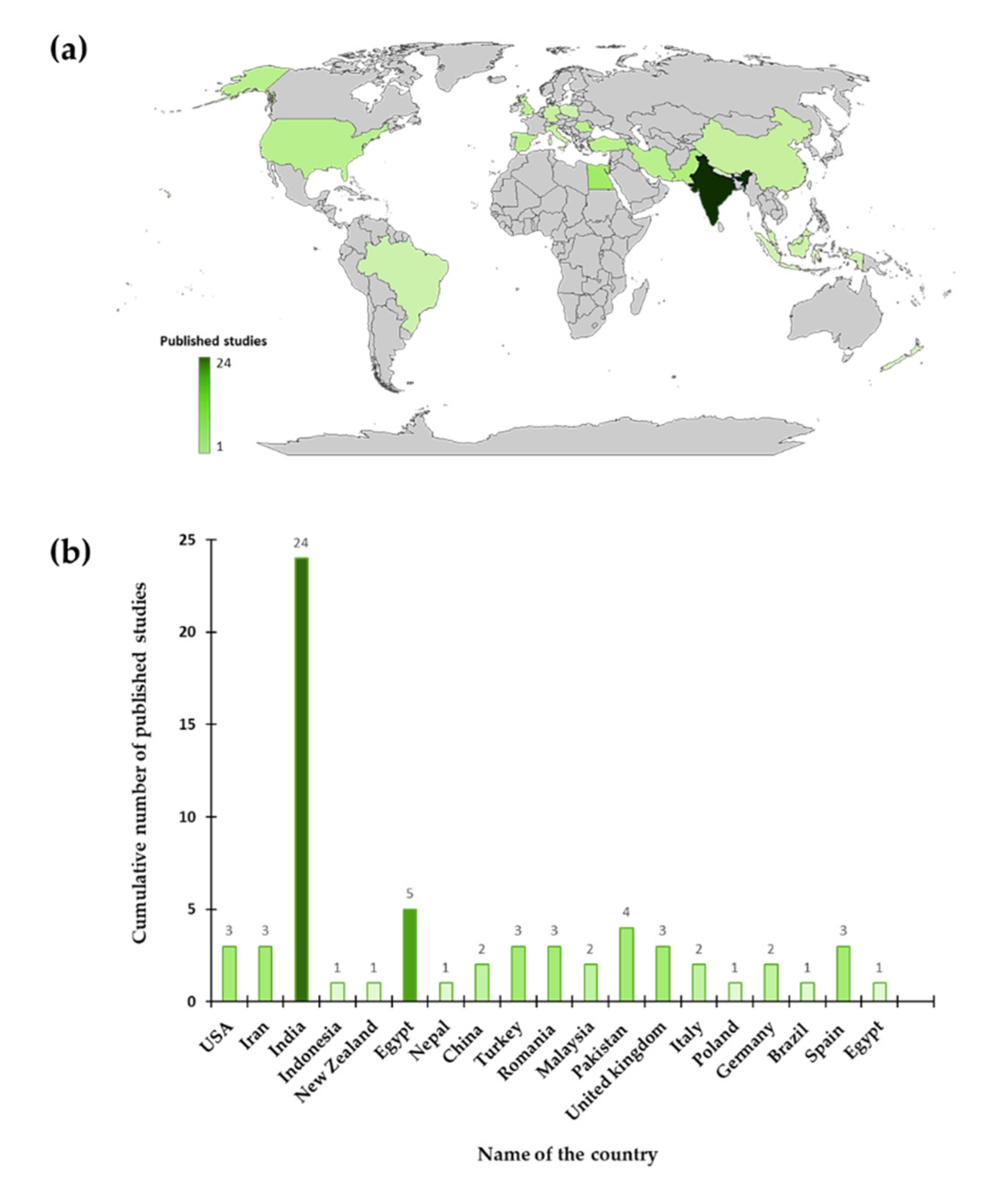
1. Introduction
2. Methodology
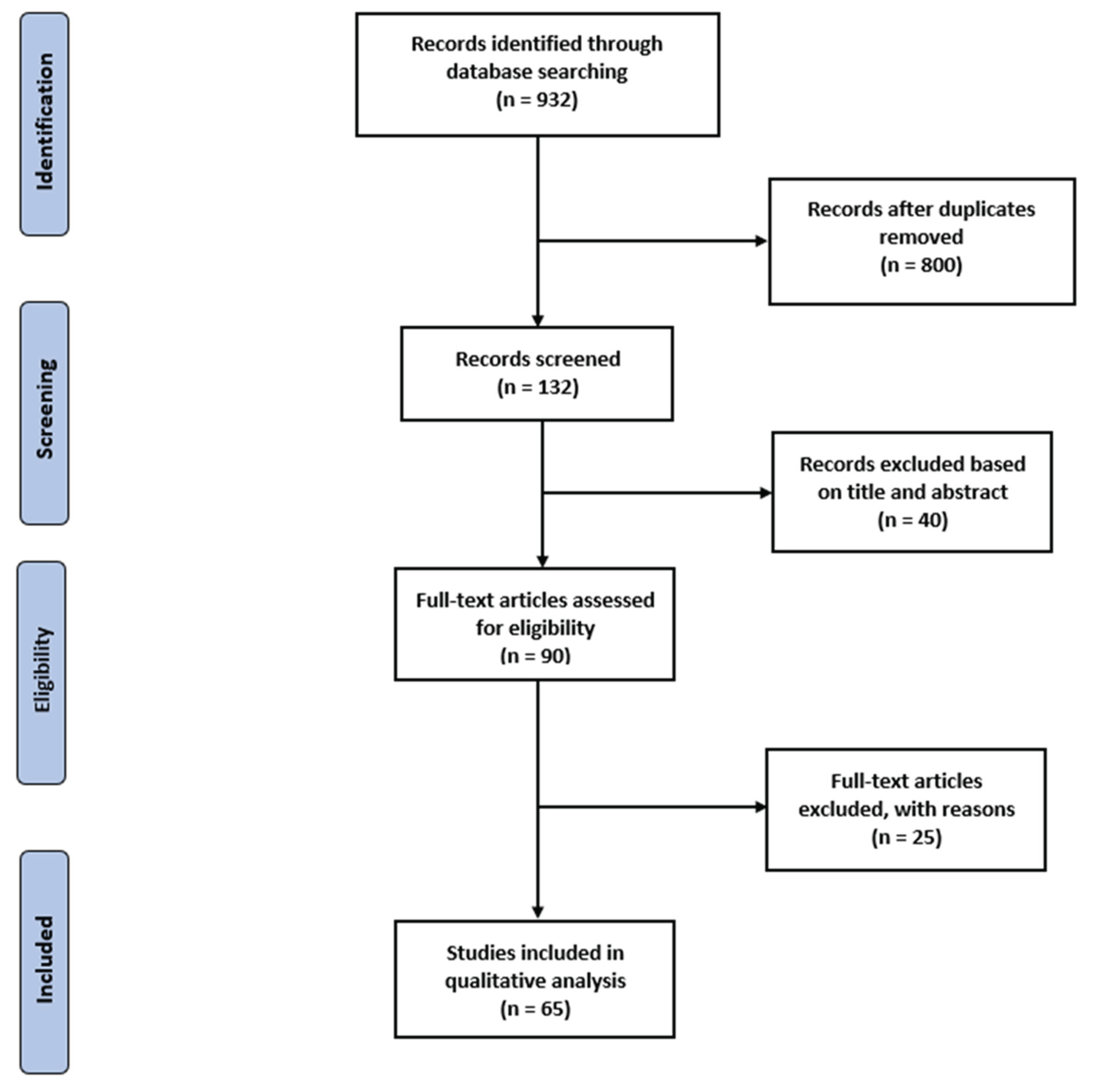
2.1. Search Strategy, Information Sources and Screening Process
2.2. Study Selection
2.3. Data Extraction and Collection
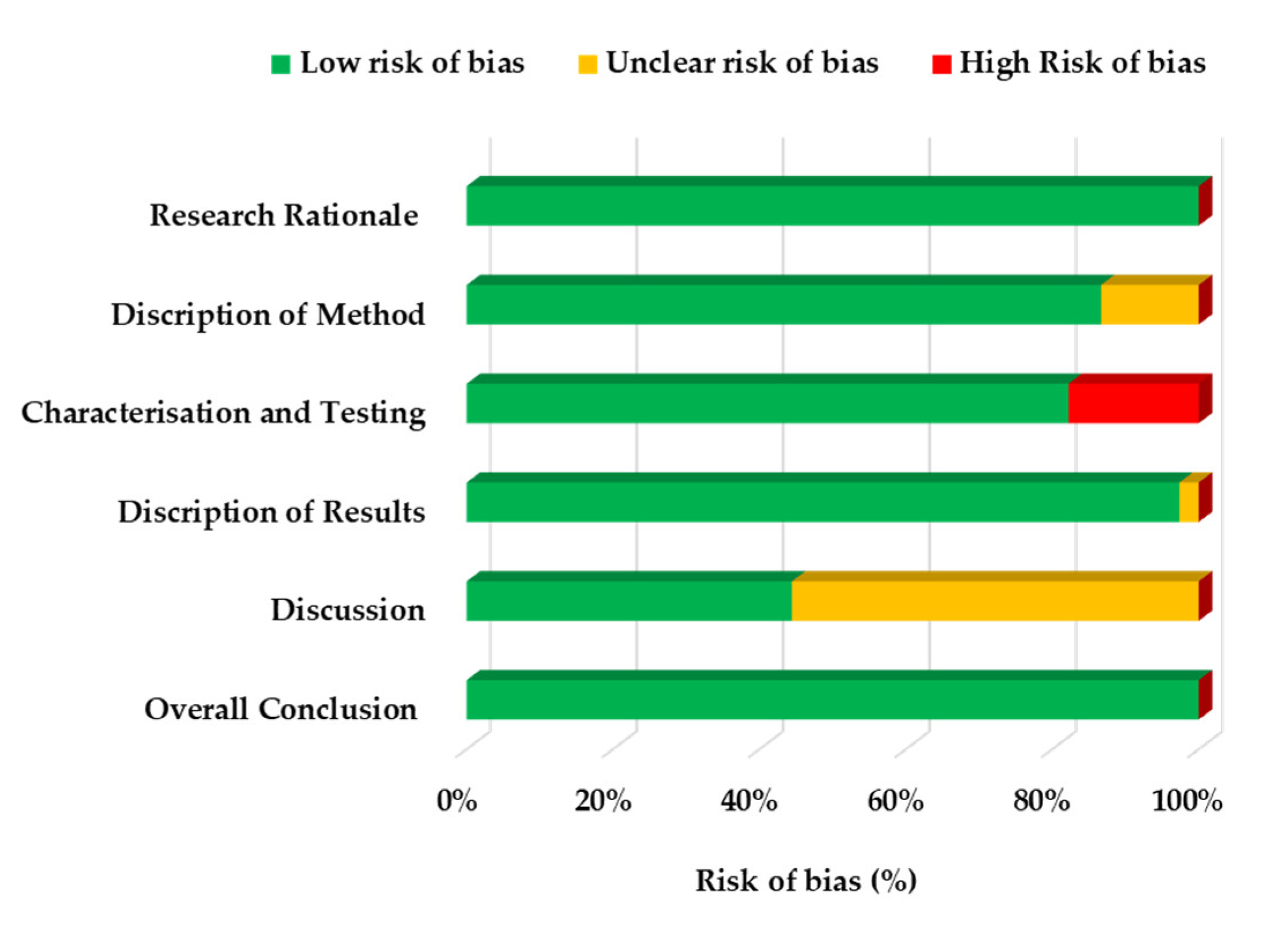
2.4. Risk of Bias Assessment
3. Results and Discussion
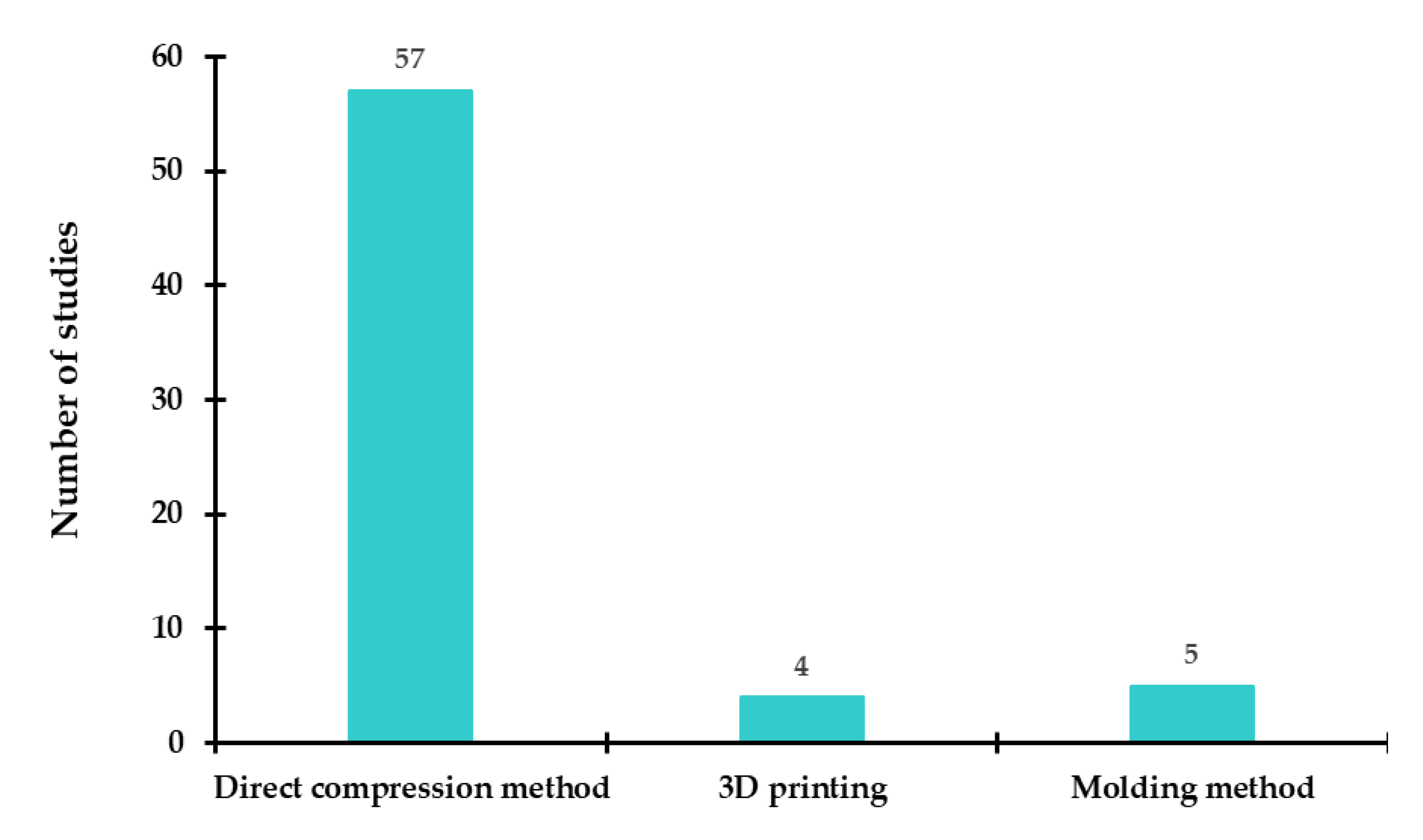
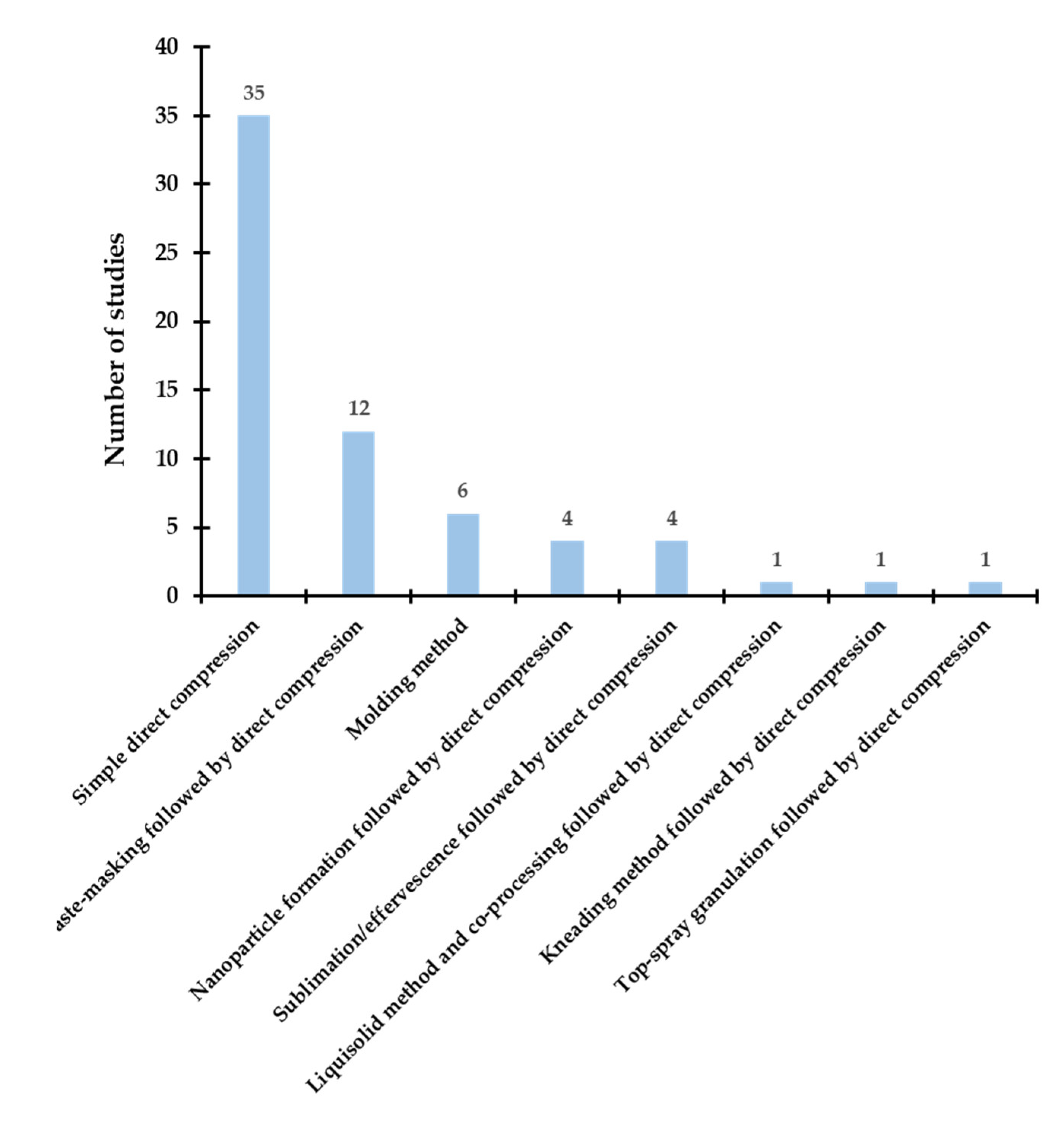
3.1. Techniques and Processes for Developing ODTs
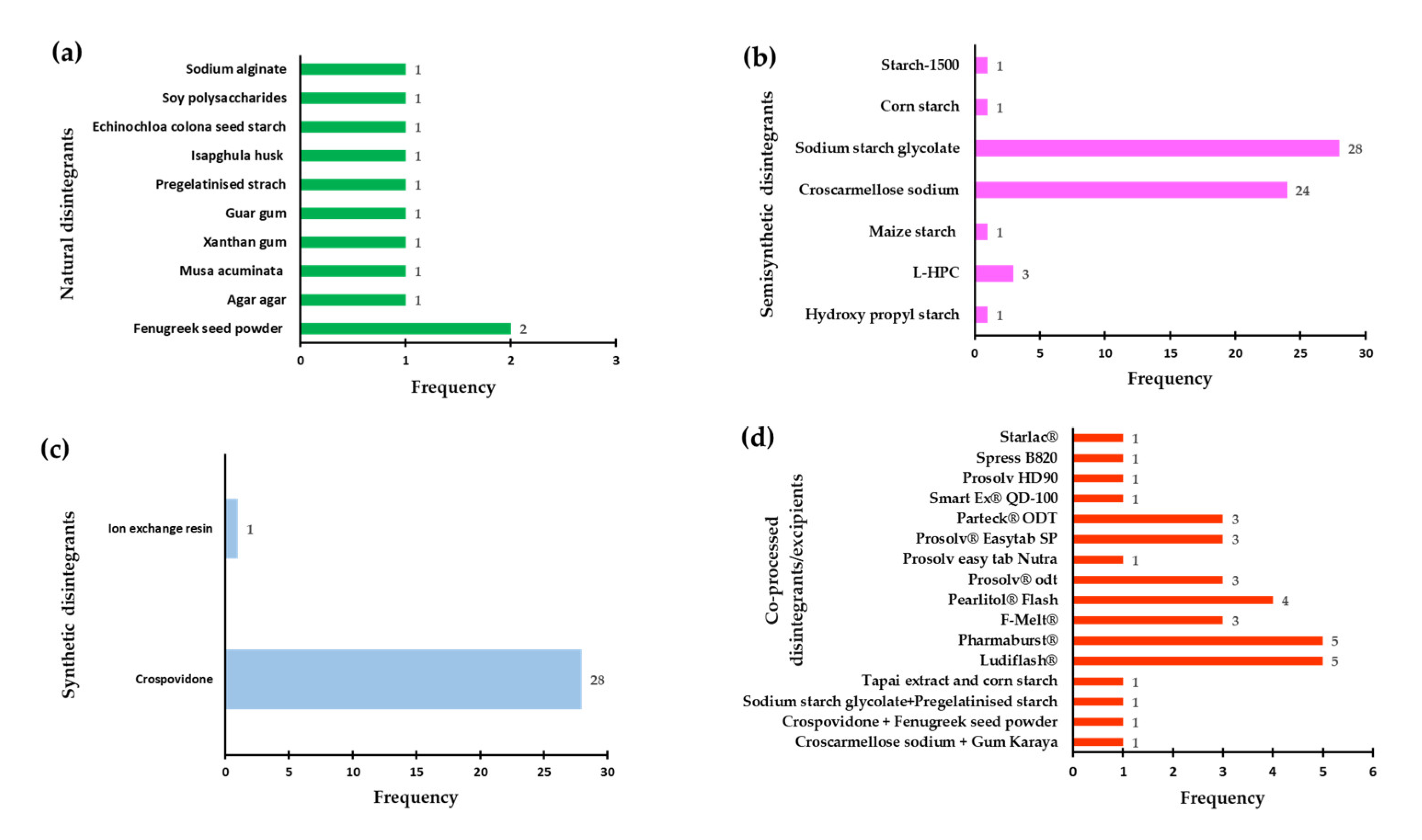
3.2. Use of Disintegrants in ODT Development
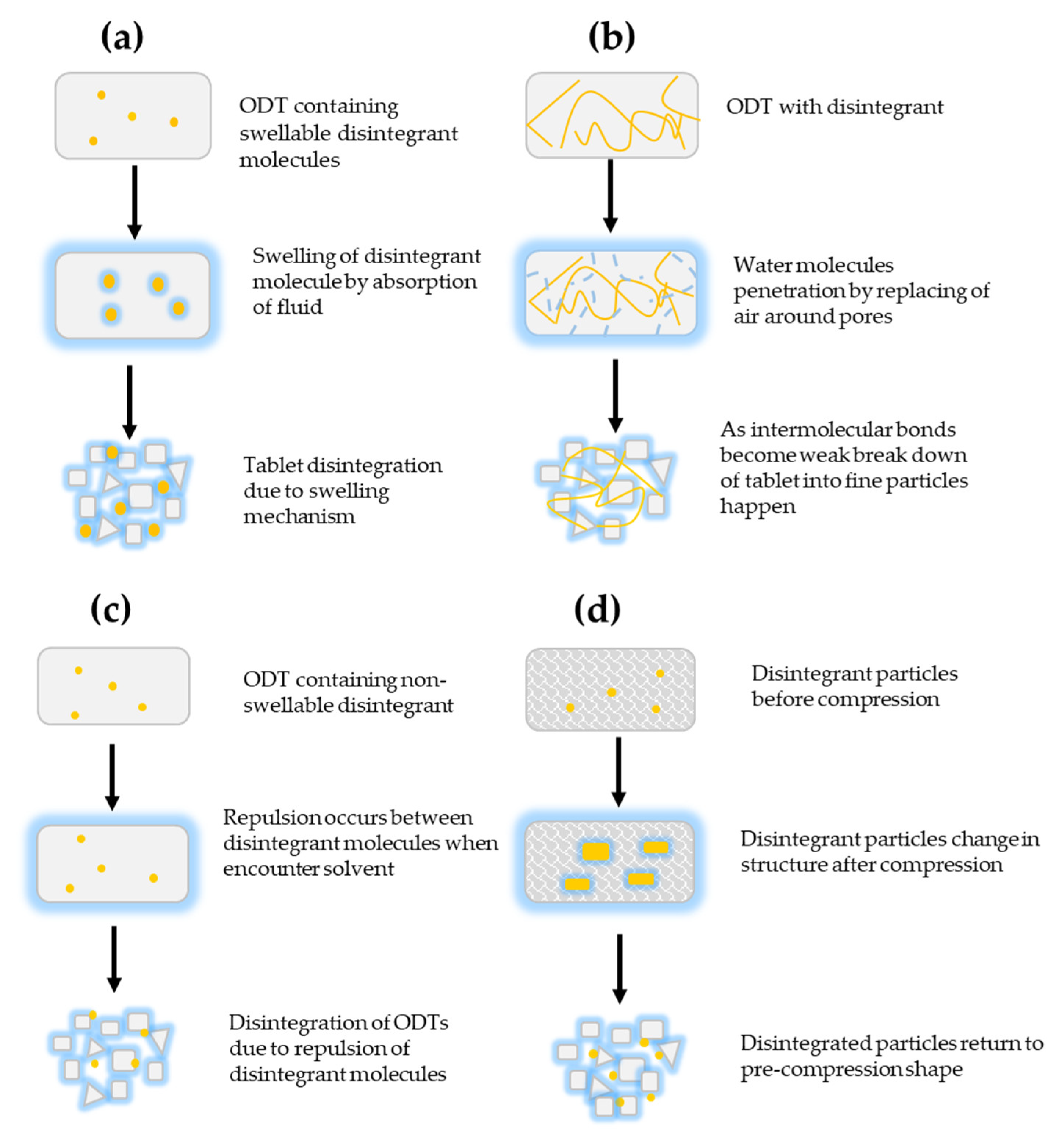
3.2.1. Mechanism of Action of Disintegrants
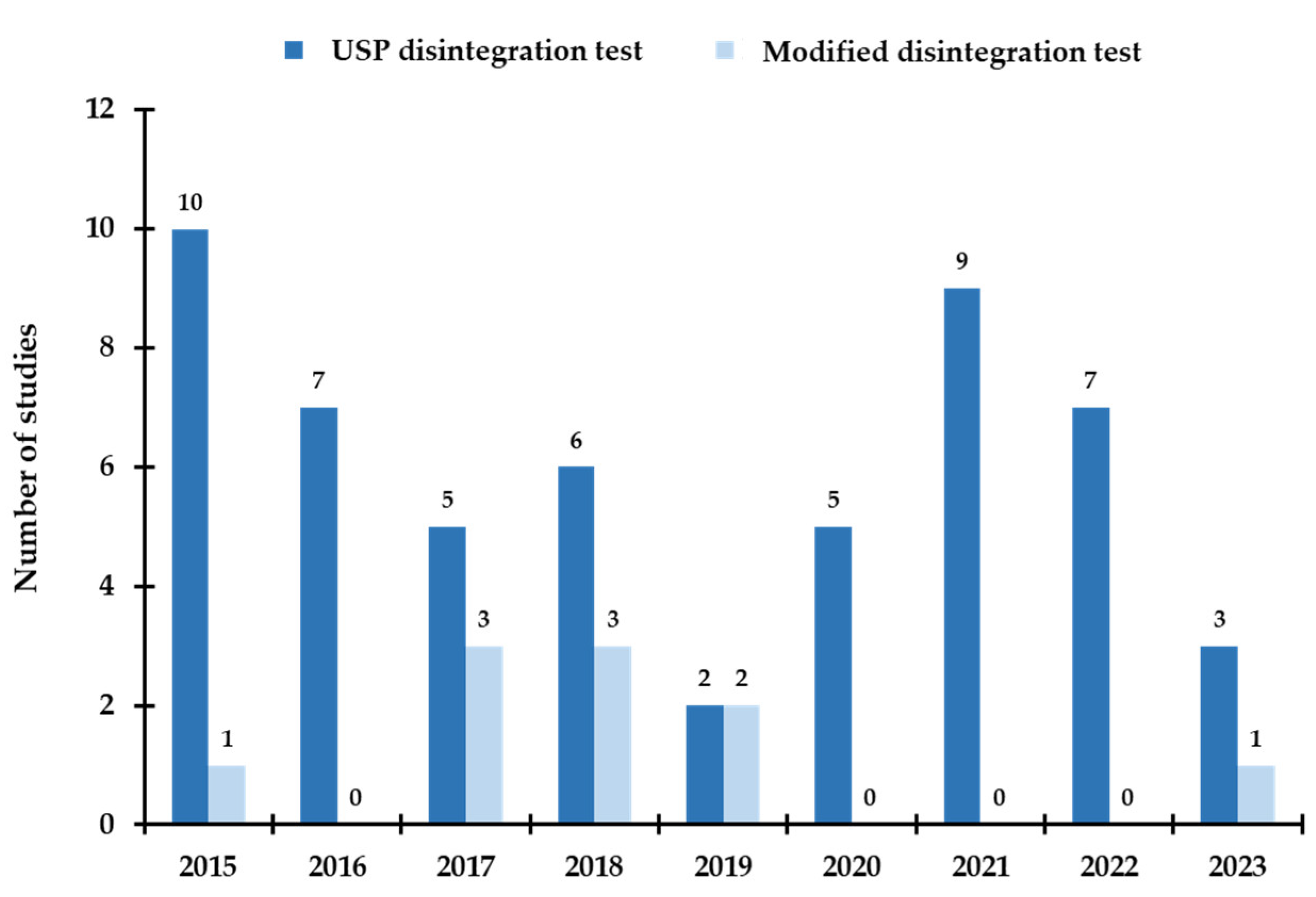
3.3. Disintegration Test and Practical Approaches for Determining Disintegration Time
4. Future Directions and Outlook
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prateek, S.; Ramdayal, G.; Kumar, S.U.; Ashwani, C.; Ashwini, G.; Mansi, S. Fast dissolving tablets: a new venture in drug delivery. Am. j. PharmTech res. 2012, 2, 252–279. [Google Scholar]
- Ivanovska, V.; Rademaker, C.M.; van Dijk, L.; Mantel-Teeuwisse, A.K.J.P. Pediatric drug formulations: a review of challenges and progress. Pediatrics. 2014, 134, 361–372. [Google Scholar] [CrossRef]
- Kozarewicz, P. Regulatory perspectives on acceptability testing of dosage forms in children. Int. J. Pharm. 2014, 469, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.K.W.; Xu, Y.; Worsley, A.; Wong, I.C.K. Oral transmucosal drug delivery for pediatric use. Adv. Drug Delivery Rev. 2014, 73, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Wagner-Hattler, L.; Kiene, K.; Bielicki, J.; Pfister, M.; Puchkov, M.; Huwyler, J. High Acceptability of an Orally Dispersible Tablet Formulation by Children. Children 2021, 8, 194. [Google Scholar] [CrossRef]
- Zajicek, A.; Fossler, M.J.; Barrett, J.S.; Worthington, J.H.; Ternik, R.; Charkoftaki, G.; Lum, S.; Breitkreutz, J.; Baltezor, M.; Macheras, P. A report from the pediatric formulations task force: perspectives on the state of child-friendly oral dosage forms. AAPS J 2013, 15, 1072–1081. [Google Scholar] [CrossRef]
- Ernest, T.B.; Craig, J.; Nunn, A.; Salunke, S.; Tuleu, C.; Breitkreutz, J.; Alex, R.; Hempenstall, J. Preparation of medicines for children – A hierarchy of classification. Int. J. Pharm. 2012, 435, 124–130. [Google Scholar] [CrossRef]
- Nahata, M.C.; Allen, L.V. Extemporaneous drug formulations. Clin. Ther. 2008, 30, 2112–2119. [Google Scholar] [CrossRef]
- Best, B.M.; Capparelli, E.V.; Diep, H.; Rossi, S.S.; Farrell, M.J.; Williams, E.; Lee, G.; van den Anker, J.N.; Rakhmanina, N. Pharmacokinetics of lopinavir/ritonavir crushed versus whole tablets in children. J. Acquired Immune Defic. Syndr. 2011, 58, 385–391. [Google Scholar] [CrossRef]
- UNICEF and WHO. Sources and prices of selected medicines for children, 2009 Available online:. Available online: https://www.unicef.org/supply/index_47129.html (accessed on 03).
- Wood, J.; Butler, C.; Hood, K.; Kelly, M.; Verheij, T.; Little, P.; Torres, A.; Blasi, F.; Schaberg, T.; Goossens, H. Antibiotic prescribing for adults with acute cough/LRTI: congruence with guidelines. Eur. Respir. J. 2011, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Li, L.; Cui, M.; Han, Y.; Karahan, H.E.; Chow, V.T.K.; Xu, C. Cold Chain-Free Storable Hydrogel for Infant-Friendly Oral Delivery of Amoxicillin for the Treatment of Pneumococcal Pneumonia. ACS Appl. Mater. Interfaces. 2017, 9, 18440–18449. [Google Scholar] [CrossRef]
- Thomson, S.A.; Tuleu, C.; Wong, I.C.; Keady, S.; Pitt, K.G.; Sutcliffe, A.G. Minitablets: new modality to deliver medicines to preschool-aged children. Pediatrics. 2009, 123, e235–e238. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.; Bickmann, D.; Breitkreutz, J.; Chariot-Goulet, M. Delivery devices for the administration of paediatric formulations: Overview of current practice, challenges and recent developments. Int. J. Pharm. 2011, 415, 221–231. [Google Scholar] [CrossRef]
- Salunke, S.; Giacoia, G.; Tuleu, C. The STEP (Safety and Toxicity of Excipients for Paediatrics) database. Part 1—A need assessment study. Int. J. Pharm. 2012, 435, 101–111. [Google Scholar] [CrossRef]
- Batchelor, H.K.; Marriott, J.F. Formulations for children: problems and solutions. Br. J. Clin. Pharmacol. 2015, 79, 405–418. [Google Scholar] [CrossRef]
- Ali, A.A.; Charoo, N.A.; Abdallah, D.B. Pediatric drug development: formulation considerations. Drug Dev. Ind. Pharm. 2014, 40, 1283–1299. [Google Scholar] [CrossRef]
- van Riet-Nales, D.A.; de Neef, B.J.; Schobben, A.F.; Ferreira, J.A.; Egberts, T.C.; Rademaker, C.M. Acceptability of different oral formulations in infants and preschool children. Arch. Dis. Child. 2013, 98, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, H.G. WHO guideline development of paediatric medicines: Points to consider in pharmaceutical development. Int. J. Pharm. 2012, 435, 134–135. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Reflection paper: formulations of choice for the paediatric population (EMEA/CHMP/PEG/194810/2005). Available from: https://www.ema.europa.eu/en/formulations-choicepaediatric- population (Accessed on December 24, 2024). 24 December 2024.
- Almajidi, Y.; Maraie, N. An Overview on Oroslippery Technique as A Promising Alternative for Tablets used in Dysphagia. Research J. Pharm. and Tech. 2019, 12, 4545–4549. [Google Scholar] [CrossRef]
- Lopez, F.L.; Ernest, T.B.; Tuleu, C.; Gul, M.O. Formulation approaches to pediatric oral drug delivery: benefits and limitations of current platforms. Expert Opin. Drug Delivery. 2015, 12, 1727–1740. [Google Scholar] [CrossRef]
- Chinwala, M. Recent Formulation Advances and Therapeutic Usefulness of Orally Disintegrating Tablets (ODTs). Pharmacy. 2020, 8, 186. [Google Scholar] [CrossRef] [PubMed]
- Alderborn, G. Tablets and compaction. In Aulton’s Pharmaceutics - The design and manufacture of medicines. 3rd ed.; Churchill Livingstone, Edinburgh: 2007.
- Belayneh, A.; Molla, F.; Kahsay, G. Formulation and Optimization of Monolithic Fixed-Dose Combination of Metformin HCl and Glibenclamide Orodispersible Tablets. Adv. Pharmacol. Pharm. Sci. 2020, 2020, 3546597–3546597. [Google Scholar] [CrossRef]
- Alyami, H.; Koner, J.; Huynh, C.; Terry, D.; Mohammed, A.R. Current opinions and recommendations of paediatric healthcare professionals - The importance of tablets: Emerging orally disintegrating versus traditional tablets. PLoS One. 2018, 13, e0193292. [Google Scholar] [CrossRef]
- Alyami, H.; Koner, J.; Dahmash, E.Z.; Bowen, J.; Terry, D.; Mohammed, A.R. Microparticle surface layering through dry coating: impact of moisture content and process parameters on the properties of orally disintegrating tablets. J. Pharm. Pharmacol. 2017, 69, 807–822. [Google Scholar] [CrossRef] [PubMed]
- A AlHusban, F.; M El-Shaer, A.; J Jones, R.; R Mohammed, A. Recent patents and trends in orally disintegrating tablets. Recent Pat. Drug Delivery Formulation. 2010, 4, 178–197. [Google Scholar] [CrossRef]
- Slavkova, M.; Breitkreutz, J. Orodispersible drug formulations for children and elderly. Eur. J. Pharm. Sci. 2015, 75, 2–9. [Google Scholar] [CrossRef]
- Karavasili, C.; Gkaragkounis, A.; Fatouros, D.G. Patent landscape of pediatric-friendly oral dosage forms and administration devices. Expert Opin. Ther. Pat. 2021, 1–23. [Google Scholar] [CrossRef]
- Mfoafo, K.A.; Omidian, M.; Bertol, C.D.; Omidi, Y.; Omidian, H. Neonatal and pediatric oral drug delivery: Hopes and hurdles. Int. J. Pharm. 2021, 120296. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009, 151, 264–269. [Google Scholar]
- Yousaf, M.; Nirwan, J.S.; Smith, A.M.; Timmins, P.; Conway, B.R.; Ghori, M.U. Raft-forming polysaccharides for the treatment of gastroesophageal reflux disease (GORD): Systematic review. J. Appl. Polym. Sci. 2019, 136, 48012. [Google Scholar] [CrossRef]
- Hasan, S.S.; Zaidi, S.T.R.; Nirwan, J.S.; Ghori, M.U.; Javid, F.; Ahmadi, K.; Babar, Z.-U.-D. Use of central nervous system (CNS) medicines in aged care homes: a systematic review and meta-analysis. J. Clin. Med. 2019, 8, 1292. [Google Scholar] [CrossRef]
- Nirwan, J.S.; Hasan, S.S.; Babar, Z.-U.-D.; Conway, B.R.; Ghori, M.U. Global Prevalence and Risk Factors of Gastro-oesophageal Reflux Disease (GORD): Systematic Review with Meta-analysis. Sci. Rep. 2020, 10, 1–4. [Google Scholar] [CrossRef]
- Nirwan, J.S.; Hasan, S.S.; Conway, B.R.; Ghori, M.U. Investigating the association between education level and gastroesophageal reflux disease (GERD): A systematic review and meta-analysis. Turk. J. Gastroenterol. 2019, 30 (Suppl. 3), S892–S893. [Google Scholar]
- Nirwan, J.S.; Hasan, S.S.; Conway, B.R.; Ghori, M.U. Investigating the association between diet and gastroesophageal reflux disease (GERD): A systematic review and meta-analysis. Turk. J. Gastroenterol. 2019, 30 (Suppl. 3), S616–S618. [Google Scholar]
- Nirwan, J.S.; Hasan, S.S.; Conway, B.R.; Ghori, M.U. Investigating the association between body mass index and gastroesophageal reflux disease: A systematic review and meta-analysis. Turk. J. Pharm. Sci. 2019, 30, S592–S593. [Google Scholar]
- Electronic Medicines Compendium, Database on the Internet. Datapharm Communications Ltd. [Internet]. 2021 [cited 20 May 2021]. Available from: http://emc.medicines.org.uk.
- Khizer, Z.; Sadia, A.; Sharma, R.; Farhaj, S.; Nirwan, J.S.; Kakadia, P.G.; Hussain, T.; Yousaf, A.M.; Shahzad, Y.; Conway, B.R. Drug Delivery Approaches for Managing Overactive Bladder (OAB): A Systematic Review. Pharmaceuticals. 2021, 14, 409. [Google Scholar] [CrossRef]
- Cantor, S.L.; Khan, M.A.; Gupta, A. Development and optimization of taste-masked orally disintegrating tablets (ODTs) of clindamycin hydrochloride. Drug Dev. Ind. Pharm. 2015, 41, 1156–1164. [Google Scholar] [CrossRef]
- Usmani, M.T.; Shoaib, M.H.; Nasiri, M.I.; Yousuf, R.I.; Zaheer, K.; Ahmed, K. Development and evaluation of orally disintegrating tablets of Montelukast sodium by direct compression method. Trop. J. Pharm. Res. 2015, 14, 1–6. [Google Scholar] [CrossRef]
- Pimparade, M.B.; Morott, J.T.; Park, J.-B.; Kulkarni, V.I.; Majumdar, S.; Murthy, S.; Lian, Z.; Pinto, E.; Bi, V.; Durig, T. Murthy, R. Development of taste masked caffeine citrate formulations utilizing hot melt extrusion technology and in vitro–in vivo evaluations. Int. J. Pharm. 2015, 487, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Shahtalebi, M.A.; Tabbakhian, M.; Koosha, S. Formulation and evaluation of orally disintegrating tablet of ondansetron using natural superdisintegrant. J. HerbMed Pharmacol. 2015, 4, 102–109. [Google Scholar]
- Kumari, P.K.; Rao, Y.S.; Devi, A.L.; Mallikarjun, P. Formulation and Evaluation of Orally Disintegrating Tablets of Amlodipine Besylate Using Novel Co-Processed Superdisintegrants. Int. J. Pharm. Sci. Rev. Res. 2015, 34, 17–23. [Google Scholar]
- Rao, P.; Rao, G.; Mallikarjun, V. Formulation and in- vitro evaluation of fast-disintegrating tablets of flupirtine. Int. J. Pharm. Technol. 2015, 7, 8289–8301. [Google Scholar]
- Samran, K. Methochlopramide orally disintegrating tablet formulation using co-processed excipient of solid tapai extract and corn starch. Int. J. PharmTech Res. 2015, 8, 91–98. [Google Scholar]
- Elwerfalli, A.M.; Al-Kinani, A.; Alany, R.G.; ElShaer, A. Nano-engineering chitosan particles to sustain the release of promethazine from orodispersables. Carbohydr. Polym. 2015, 131, 447–461. [Google Scholar] [CrossRef] [PubMed]
- Labib, G.S. Novel levocetirizine HCl tablets with enhanced palatability: synergistic effect of combining taste modifiers and effervescence technique. Drug Des., Dev. Ther. 2015, 9, 5135–5146. [Google Scholar] [CrossRef] [PubMed]
- El Sisi, A.M.; Kharshoum, R.M.; Ali, A.A.; Hosny, K.M.; Abd-Elbary, A. Preparation and Evaluation of Risperidone Oro-dispersible Tablets. Int. J. Pharm. Sci. Rev. Res. 2015, 30, 1–6. [Google Scholar]
- Subedi, R.; Poudel, K.; Budhathoki, U.; Thapa, P. Taste Masking and Formulation of Ondansetron Hydrochloride Mouth Dissolving Tablets. Int. J. Pharm Sci. Res. 2015, 6. [Google Scholar]
- Chen, Y.-D.; Liang, Z.-Y.; Cen, Y.-Y.; Zhang, H.; Han, M.-G.; Tian, Y.-Q.; Zhang, J.; Li, S.-J.; Yang, D.-S. Development of oral dispersible tablets containing prednisolone nanoparticles for the management of pediatric asthma. Drug Des., Dev. Ther. 2015, 9, 5815. [Google Scholar]
- Sharma, D.; Singh, G.; Kumar, D.; Singh, M. Formulation development and evaluation of fast disintegrating tablets of salbutamol sulphate, cetirizine hydrochloride in combined pharmaceutical dosage form: a new era in novel drug delivery for pediatrics and geriatrics. J. Drug Delivery. 2015, 2015. [Google Scholar] [CrossRef]
- Khan, W.U. Fabrication and evaluation of fast dissolving dosage form of domperidone. Pharm. Lett. 2015, 7, 221–236. [Google Scholar]
- Aslani, A.; Beigi, M. Design, formulation, and physicochemical evaluation of montelukast orally disintegrating tablet. Int. J. Prev. Med. 2016, 7. [Google Scholar] [CrossRef]
- Shukla, S.; Mishra, D.K.; Jain, D.K. Designing of Fast Disintegrating Tablets for Antihypertensive Agent Using Superdisintegrants. Res. J. Pharm. Technol. 2016, 9, 527–532. [Google Scholar] [CrossRef]
- Sahoo, C.K.; Sahoo, N.K.; Sahu, M.; Sarangi, D.K. Formulation and Evaluation of Orodispersible Tablets of Granisetron Hydrochloride Using Agar as Natural Super disintegrants. Pharm. Methods. 2016, 7, 17–22. [Google Scholar] [CrossRef]
- Yıldız, S.; Aytekin, E.; Yavuz, B.; Bozdağ Pehlivan, S.; Ünlü, N. Formulation studies for mirtazapine orally disintegrating tablets. Drug Dev. Ind. Pharm. 2016, 42, 1008–1017. [Google Scholar] [CrossRef]
- Shazly, G.A.; Ibrahim, M.A. Losartan potassium taste-masked oral disintegrating tablets for hypertensive patients. Lat. Am. J. Pharm. 2016, 35, 110–117. [Google Scholar]
- Pandey, P.; K Marwaha, R.; Nanda, A.; Dureja, H. Spray-dried nanoparticles-in-microparticles system (NiMS) of acetazolamide using central composite design. Nanosci. Nanotechnol.-Asia. 2016, 6, 146–156. [Google Scholar] [CrossRef]
- Vishwasrao, S.S.; Jadhav, A. Studies on formulation and evaluation of orally disintegrating tablets using Musa acuminata as a natural disintegrant for paediatric use. Indian Drugs. 2016, 53, 55–58. [Google Scholar] [CrossRef]
- Sipos, E.; Oltean, A.R.; Szabó, Z.-I.; RÉDAI, E.; Nagy, G.D. Application of the SeDeM expert systems in the preformulation studies of pediatric ibuprofen ODT tablets. Acta Pharm. 2017, 67, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Moqbel, H.A.; ElMeshad, A.N.; El-Nabarawi, M.A. Comparative study of different approaches for preparation of chlorzoxazone orodispersible tablets. Drug Dev. Ind. Pharm. 2017, 43, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Lal, M.; Lai, M.; Estrada, M.; Zhu, C. Developing a Flexible Pediatric Dosage Form for Antiretroviral Therapy: A Fast-Dissolving Tablet. J. Pharm. Sci. 2017, 106, 2173–2177. [Google Scholar] [CrossRef]
- Ranganathan, V.; Yoong, J. Development and evaluation of mouth dissolving tablets using natural super disintegrants. J. Young Pharm. 2017, 9, 332–335. [Google Scholar] [CrossRef]
- Khan, H.; Sarfraz, R.M.; Maheen, S.; Mahmood, A.; Shamim, A.; Sher, M. Development and evaluation of novel antihypertensive orodispersible tablets. Pak. J. Pharm. Sci. 2017, 30, 1557–1565. [Google Scholar]
- Tarke, S.; Shanmugasundaram, P. Formulation and evaluation of fast Dissolving tablets of Antihypertensive Drug. Res. J. Pharm. Tech. 2017, 10, 155–160. [Google Scholar] [CrossRef]
- Chandrasekaran, P.; Kandasamy, R. Development of oral flexible tablet (OFT) formulation for pediatric and geriatric patients: a novel age-appropriate formulation platform. AAPS PharmSciTech. 2017, 18, 1972–1986. [Google Scholar] [CrossRef] [PubMed]
- Orubu, S.E.; Hobson, N.J.; Basit, A.W.; Tuleu, C. The Milky Way: paediatric milk-based dispersible tablets prepared by direct compression–a proof-of-concept study. J. Pharm. Pharmacol. 2017, 69, 417–431. [Google Scholar] [CrossRef]
- Liu, Y.; Li, P.; Qian, R.; Sun, T.; Fang, F.; Wang, Z.; Ke, X.; Xu, B. A novel and discriminative method of in vitro disintegration time for preparation and optimization of taste-masked orally disintegrating tablets of carbinoxamine maleate. Drug Dev. Ind. Pharm. 2018, 44, 1317–1327. [Google Scholar] [CrossRef]
- Yıldız, S.; Aytekin, E.; Yavuz, B.; Bozdağ Pehlivan, S.; Vural, İ.; Ünlü, N. Development and evaluation of orally disintegrating tablets comprising taste-masked mirtazapine granules. Pharm. Dev. Technol. 2018, 23, 488–495. [Google Scholar] [CrossRef]
- Mennini, N.; Orlandini, S.; Furlanetto, S.; Pasquini, B.; Mura, P. Development and Optimization by Quality by Design Strategies of Frovatriptan Orally Disintegrating Tablets for Migraine Management. Curr. Drug Delivery. 2018, 15, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Siemiradzka, W.; Szulc-Musiol, B.; Bulas, L.; Jankowski, A. Effect of selected excipients and the preparation method on the parameters of orally disintegrating tablets containing ondansetron. Acta Pol. Pharm. 2018, 75, 733–740. [Google Scholar]
- G, S.; Pg, M.; K, C.; M, L.; S, A. Formulation and evaluation of fast dissolving tablet of ketorolac tromethamine. Asian J. Pharm. Clin. Res. 2018, 11, 163–169. [Google Scholar] [CrossRef]
- Türkmen, Ö.; Şenyiğit, Z.A.; Baloğlu, E. Formulation and evaluation of fexofenadine hydrochloride orally disintegrating tablets for pediatric use. J. Drug Deliv Sci.Technol. 2018, 43, 201–210. [Google Scholar] [CrossRef]
- Husseiny, R.A.; Abu Lila, A.S.; Abdallah, M.H.; El-ghamry, H.A. Fast disintegrating tablet of Valsartan for the treatment of pediatric hypertension: In vitro and in vivo evaluation. J. Drug Deliv Sci.Technol. 2018, 43, 194–200. [Google Scholar] [CrossRef]
- Gupta, M.M.; Gupta, I.K.; Baboota, S.; Gupta, M. Formulation Development and Evaluation of Orally Disintegrating Tablet of Taste Masked Azithromycin. Lat. Am. J. Pharm. 2019, 38, 1478–1484. [Google Scholar]
- Kabir, Q.A.; Wasti, A.N.; Nazir, S.U.; Adnan, M.; Azhar, R.F.; Bukhari, F.; Amer, M. Formulation Development and In Vitro Evaluation of Mouth Dissolving Tablets of Domperidone. Lat. Am. J. Pharm. 2019, 38, 1775–1783. [Google Scholar]
- Jaya, S.; Amala, V. Formulation and in vitro evaluation of oral disintegrating tablets of amlodipine besylate. Int. J. Appl. Pharm. 2019, 11, 49–54. [Google Scholar] [CrossRef]
- Lopalco, A.; Curci, A.; Lopedota, A.; Cutrignelli, A.; Laquintana, V.; Franco, M.; Denora, N. Pharmaceutical preformulation studies and paediatric oral formulations of sodium dichloroacetate. Eur. J. Pharm. Sci. 2019, 127, 339–350. [Google Scholar] [CrossRef]
- El Aita, I.; Rahman, J.; Breitkreutz, J.; Quodbach, J. 3D-Printing with precise layer-wise dose adjustments for paediatric use via pressure-assisted microsyringe printing. Eur. J. Pharm. Biopharm. 2020, 157, 59–65. [Google Scholar] [CrossRef]
- de Freitas Neto, J.L.; do Nascimento Gomes Barbosa, I.; de Melo, C.G.; Ângelos, M.A.; dos Santos Mendes, L.M.; Ferreira, M.R.A.; Rolim, L.A.; Soares, L.A.; da Silva, R.M.F.; Neto, P.J.R. Development of Pediatric Orodispersible Tablets Based on Efavirenz as a New Therapeutic Alternative. Curr. HIV Res. 2020, 18, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Ghogari, I.S.; Jain, P.S. Development of orally disintegrating tablets of memantine hydrochloride—A remedy for Alzheimer’s disease. Int. J. Appl. Pharm. 2020, 12, 147–152. [Google Scholar] [CrossRef]
- Dennison, T.J.; Smith, J.C.; Badhan, R.K.; Mohammed, A.R. Formulation and Bioequivalence Testing of Fixed-Dose Combination Orally Disintegrating Tablets for the Treatment of Tuberculosis in the Paediatric Population. J. Pharm. Sci. 2020, 109, 3105–3113. [Google Scholar] [CrossRef]
- Swamivelmanickam, M.; Venkatesan, P.; Sangeetha, G. Preparation and Evaluation of Mouth Dissolving Tablets of Loratadine by Direct Compression Method. Res. J. Pharm. Technol. 2020, 13, 2629–2633. [Google Scholar] [CrossRef]
- Panhale, D.P.; Bachhav, R.S.; Gondkar, S.B. Formulation and evaluation of orodispersible tablets of apremilast by inclusion complexation using beta-cyclodextrin. Ind. J. Pharm. Edu. Res 2021, 55, S112–S121. [Google Scholar] [CrossRef]
- Kumar, R.; Verma, R.; Kaushik, R.; Kaushik, P.; Pandey, P.; Purohit, D.; Mittal, V.; Kaushik, D. Optimization and In vitro Evaluation of Famotidine Loaded Effervescent Orally Disintegrating Tablets Using Central Composite Design. Curr. Drug Ther. 2021, 16, 336–344. [Google Scholar] [CrossRef]
- Alami-Milani, M.; Salatin, S.; Nasiri, E.; Jelvehgari, M. Preparation and optimization of fast disintegrating tablets of isosorbide dinitrate using lyophilization method for oral drug delivery. Ther. Delivery 2021, 12, 523–538. [Google Scholar] [CrossRef]
- Shamling, S.S.; Chavan, S.; Chandrakant, G.P.; Kharat, R.T.; Kulkarni, A.S. Isolation, Characterization, Chemical Modification and Application of Echinochloa Colona Starch as Tablet Disintegrant. Int. J. Pharm. Sci. Nanotechnol. 2021, 14, 5529–5537. [Google Scholar] [CrossRef]
- Mahesh, P.; Raman, S. Formulation and evaluation of fast dissolving tablet of prasugrel. Res. J. Pharm. Technol. 2021, 14, 4077–4079. [Google Scholar] [CrossRef]
- Eduardo, D.-T.; Ana, S.-E. A micro-extrusion 3D printing platform for fabrication of orodispersible printlets for pediatric use. Int. J. Pharm. 2021, 605, 120854. [Google Scholar] [CrossRef]
- Darwesh, A.Y.; El-Dahhan, M.S.; Meshali, M.M. A new dual function orodissolvable/dispersible meclizine HCL tablet to challenge patient inconvenience: in vitro evaluation and in vivo assessment in human volunteers. Drug Delivery Transl. Res. 2021, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kokott, M.; Lura, A.; Breitkreutz, J.; Wiedey, R. Evaluation of two novel co-processed excipients for direct compression of orodispersible tablets and mini-tablets. Eur. J. Pharm. Biopharm. 2021, 168, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Singhal, P.; Chamola, P.; Kumar, V.J.; Kumar, S.; Rout, A. Optimized fast disintegrating tablets, boosted oseltamivir phosphate orally fast disintegrating tablets. J. Med. Pharm. Allied Sci. 2021, 10, 3781–3788. [Google Scholar] [CrossRef]
- Suárez-González, J.; Magariños-Triviño, M.; Díaz-Torres, E.; Cáceres-Pérez, A.R.; Santoveña-Estévez, A.; Fariña, J.B. Individualized orodispersible pediatric dosage forms obtained by molding and semi-solid extrusion by 3D printing: A comparative study for hydrochlorothiazide. J. Drug Delivery Sci. Technol. 2021, 66, 102884. [Google Scholar] [CrossRef]
- Azmi, N.H.S.; Ming, L.C.; Uddin, A.H.; Sarker, Z.I.; Bin, L.K. Investigation of the Effects of Excipients in the Compounding of Amlodipine Besylate Orally Disintegrating Tablets. Int. J. Pharm. Compd. 2022, 26, 80–87. [Google Scholar]
- Thakker, P.; Shah, J.; Mehta, T.; Shetty, V.; Aware, R.; Kuchekar, A. Development and evaluation of taste masked orally disintegrating tablets of pioglitazone hydrochloride. J. Res. Pharm. 2022, 26. [Google Scholar] [CrossRef]
- Adsare, V.; Prakash, L.R.; Gholap, P.; Kanthale, S.B.; Choudante, S. To enhance the solubility of ivermectin with physical mixing method for the preparation of orodispersible tablets. J. Pharm. Negat. Results 2022, 964–971. [Google Scholar]
- Iurian, S.; Bogdan, C.; Suciu, Ș.; Muntean, D.-M.; Rus, L.; Berindeie, M.; Bodi, S.; Ambrus, R.; Tomuță, I. Milk Oral Lyophilizates with Loratadine: Screening for New Excipients for Pediatric Use. Pharmaceutics 2022, 14, 1342. [Google Scholar] [CrossRef] [PubMed]
- Vlad, R.-A.; Antonoaea, P.; Todoran, N.; Rédai, E.-M.; Bîrsan, M.; Muntean, D.-L.; Imre, S.; Hancu, G.; Farczádi, L.; Ciurba, A. Development and Evaluation of Cannabidiol Orodispersible Tablets Using a 23-Factorial Design. Pharmaceutics 2022, 14, 1467. [Google Scholar] [CrossRef] [PubMed]
- Mhetre, R.L.; Kadam, P.S.; Gadhire, P.H.; Lajurkar, G.; Kagde, A.D.; Dhole, S.N. Formulation and Evaluation of Naproxen Orodispersible Tablets. Int. J. Pharm. Sci. Nanotechnol 2022, 15, 6055–6060. [Google Scholar] [CrossRef]
- Canadell-Heredia, R.; Suñé-Pou, M.; Nardi-Ricart, A.; Pérez-Lozano, P.; Suñé-Negre, J.; García-Montoya, E. Formulation and development of paediatric orally disintegrating carbamazepine tablets. Saudi Pharm. J. 2022, 30, 1612–1622. [Google Scholar] [CrossRef]
- Irshad, A.; Yousuf, R.I.; Shoaib, M.H.; Qazi, F.; Saleem, M.T.; Siddiqui, F.; Ahmed, F.R.; Jabeen, S.; Farooqi, S.; Khan, M.Z. Effect of starch, cellulose and povidone based superdisintegrants in a QbD-based approach for the development and optimization of Nitazoxanide orodispersible tablets: Physicochemical characterization, compaction behavior and in-silico PBPK modeling of its active metabolite Tizoxanide. J. Drug Delivery Sci. Technol. 2023, 79, 104079. [Google Scholar]
- Teaima, M.H.; El-Messiry, H.M.A.; Osman, T.D.; El-Nabarawi, M.A.; Helal, D.A. The effect of co-processed excipients during formulation and evaluation of pediatric levetiracetam orodispersible tablets in rats. Int. J. Appl. Pharm. 2023, 15, 318–323. [Google Scholar] [CrossRef]
- Hu, J.; Fitaihi, R.; Abukhamees, S.; Abdelhakim, H.E. Formulation and Characterisation of Carbamazepine Orodispersible 3D-Printed Mini-Tablets for Paediatric Use. Pharmaceutics 2023, 15, 250. [Google Scholar] [CrossRef] [PubMed]
- Ghori, M.U.; Conway, B.R. Powder compaction: Compression properties of cellulose ethers. Br. J. Pharm. 2016, 1, 19–29. [Google Scholar] [CrossRef]
- Ganesh, N.; Deshpande, K. Orodispersible tablets: an overview of formulation and technology. Int. J. Pharma Bio Sci. 2011, 2, 726–734. [Google Scholar]
- Okwuosa, T.C.; Soares, C.; Gollwitzer, V.; Habashy, R.; Timmins, P.; Alhnan, M.A. On demand manufacturing of patient-specific liquid capsules via co-ordinated 3D printing and liquid dispensing. Eur. J. Pharm. Sci. 2018, 118, 134–143. [Google Scholar] [CrossRef]
- Fina, F.; Madla, C.M.; Goyanes, A.; Zhang, J.; Gaisford, S.; Basit, A.W. Fabricating 3D printed orally disintegrating printlets using selective laser sintering. Int. J. Pharm. 2018, 541, 101–107. [Google Scholar] [CrossRef]
- Vaz, V.M.; Kumar, L. 3D Printing as a Promising Tool in Personalized Medicine. AAPS PharmSciTech 2021, 22, 1–20. [Google Scholar] [CrossRef]
- Desai, P.M.; Liew, C.V.; Heng, P.W.S. Review of Disintegrants and the Disintegration Phenomena. J. Pharm. Sci. 2016, 105, 2545–2555. [Google Scholar] [CrossRef]
- Markl, D.; Zeitler, J.A. A review of disintegration mechanisms and measurement techniques. Pharm. Res. 2017, 34, 890–917. [Google Scholar] [CrossRef]
- Aulton, M.E.; Summers, M. Tablets and compaction. Pharmaceutics The Science of Dosage Form Design, 4th. Philadelphia: Churchill Livingstone 2013, pp. 397-439.
- Kumar, R.S.; Kumari, A. Superdisintegrant: crucial elements for mouth dissolving tablets. J. drug delivery ther. 2019, 9, 461–468. [Google Scholar] [CrossRef]
- Alam, M.T.; Parvez, N.; Sharma, P.K. FDA-Approved Natural Polymers for Fast Dissolving Tablets. J. Pharm. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Stoltenberg, I.; Breitkreutz, J. Orally disintegrating mini-tablets (ODMTs) – A novel solid oral dosage form for paediatric use. Eur. J. Pharm. Biopharm. 2011, 78, 462–469. [Google Scholar] [CrossRef]
- Türkmen, Ö.; Ay Şenyiğit, Z.; Baloğlu, E. Formulation and evaluation of fexofenadine hydrochloride orally disintegrating tablets for pediatric use. J. Drug Delivery Sci. Technol. 2018, 43, 201–210. [Google Scholar] [CrossRef]
- Pahwa, R.; Gupta, N. Superdisintegrants in the development of orally disintegrating tablets: a review. Int. J. Pharm. Sci. Res. 2011, 2, 2767–2780. [Google Scholar]
- Shihora, H.; Panda, S. Superdisintegrants, utility in dosage forms: a quick review. J Pharm Sci Biosci Res. 2011, 1, 148–153. [Google Scholar]
- Berardi, A.; Bisharat, L.; Blaibleh, A.; Pavoni, L.; Cespi, M. A simple and inexpensive image analysis technique to study the effect of disintegrants concentration and diluents type on disintegration. J. Pharm. Sci. 2018, 107, 2643–2652. [Google Scholar] [CrossRef] [PubMed]
- Md, N.A.; Sharma, S.K.; Jaimini, M.; Mohan, S.; Chatterjee, A. Fast dissolving dosage forms: Boon to emergency conditions. Int. J. Ther. Appl. 2014, 16, 1–7. [Google Scholar]
- Kumar, S.; Garg, S. Fast dissolving tablets (FDTs): Current status, new market opportunities, recent advances in manufacturing technologies and future prospects. Int. J. Pharm. Pharm. Sci. 2014, 6, 22–35. [Google Scholar]
- Desai, P.M.; Er, P.X.H.; Liew, C.V.; Heng, P.W.S. Functionality of disintegrants and their mixtures in enabling fast disintegration of tablets by a quality by design approach. AAPS PharmSciTech. 2014, 15, 1093–1104. [Google Scholar] [CrossRef]
- Rewar, S.; Singh, C.; Bansal, B.; Pareek, R.; Sharma, A. Oral dispersible tablets: An overview; development, technologies and evaluation. Int. J. Res. Dev. Pharm. Life. Sci. 2014, 3, 1245–1257. [Google Scholar]
- Sharma, N.; Pahuja, S.; Sharma, N. Immediate release tablets: a review. Int. J. Pharm. Sci. Res. 2019, 10, 3607–3618. [Google Scholar]
- Kaur, V.; Mehara, N. A Review on: Importance of Superdisintegrants on Immediate Release Tablets. International Journal of Research and Scientific Innovation 2016, 3, 39–43. [Google Scholar]
- Chotaliya, M.K.B.; Chakraborty, S. Overview of oral dispersible tablets. Int. J. PharmTech Res. 2012, 4, 1712–1720. [Google Scholar]
- Rowe, R.C.; Sheskey, P.; Quinn, M. Handbook of pharmaceutical excipients; Libros Digitales-Pharmaceutical Press: 2009.
- Wasilewska, K.; Winnicka, K. How to assess orodispersible film quality? A review of applied methods and their modifications. Acta Pharm. 2019, 69, 155–176. [Google Scholar] [CrossRef]
- Desai, N.; Redfearn, A.; MacLeod, G.; Tuleu, C.; Hanson, B.; Orlu, M. How Do Orodispersible Tablets Behave in an In Vitro Oral Cavity Model: A Pilot Study. Pharmaceutics. 2020, 12, 651. [Google Scholar] [CrossRef]
- Koner, J.S.; Rajabi-Siahboomi, A.R.; Missaghi, S.; Kirby, D.; Perrie, Y.; Ahmed, J.; Mohammed, A.R. Conceptualisation, Development, Fabrication and In Vivo Validation of a Novel Disintegration Tester for Orally Disintegrating Tablets. Sci. Rep. 2019, 9, 12467. [Google Scholar] [CrossRef] [PubMed]
- Hooper, P.; Lasher, J.; Alexander, K.S.; Baki, G. A new modified wetting test and an alternative disintegration test for orally disintegrating tablets. J. Pharm. Biomed. Anal. 2016, 120, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Jeong, S.H.; Callihan, J.; Kim, J.; Park, K. Preparation of fast-dissolving tablets based on mannose. Am. Chem. Soc. 2006, 340–351. [Google Scholar]
- Khan, S.; Kataria, P.; Nakhat, P.; Yeole, P. Taste masking of ondansetron hydrochloride by polymer carrier system and formulation of rapid-disintegrating tablets. AAPS PharmSciTech. 2007, 8, E127–E133. [Google Scholar] [CrossRef]
| Excipient | Ingredients |
|---|---|
| Ludiflash® | Mannitol |
| Crospovidone | |
| Polyvinyl acetate dispersion | |
| Pharmaburst® | Crospovidone |
| Mannitol | |
| Sorbitol | |
| Precipitated silicon dioxide | |
| F-Melt® | Crospovidone |
| Xylitol | |
| D-Mannitol | |
| Microcrystalline cellulose | |
| Magnesium aluminometasilicate | |
| Pearlitol® Flash | Corn Starch |
| Mannitol | |
| Parteck® ODT | Mannitol |
| Croscarmellose sodium | |
| Prosolv® Easytab SP | Sodium starch Glycolate |
| Microcrystalline cellulose | |
| Colloidal Silicon Dioxide | |
| Sodium Stearyl Fumarate | |
| Prosolv® ODT | Crospovidone |
| Microcrystalline cellulose | |
| Colloidal Silicon Dioxide | |
| Mannitol | |
| Fructose |
| Disintegrant | Source/Nature | Properties | Mechanism |
|---|---|---|---|
| Natural disintegrant | |||
| Karaya gum | Obtained from Sterculia urens trees | Soluble in hot and cold water | Swelling |
| Fenugreek seed powder | From mucilage of Fenugreek seed | Soluble in hot water | Swelling |
| Agar | Mucilage derived from Gelidium amansii Lamouroux and other red algae species | Soluble in boiling water. Practically insoluble in cold water | Swelling |
| Musa acuminate | Obtained from mango peel | - | Wicking |
| Guar gum | Obtained from endosperm seeds of Cyamopsis tetragonolobus | In cold water or hot water disperses and swells immediately | Swelling |
| Ispagol husk | Extracted from the seeds of Plantago ovata | Soluble in water | Swelling |
| Xanthan gum | Obtained from Xanthomonas campestris | Soluble in cold or warm water | Swelling |
| Chitosan | Derived from shells of crustaceans (like shrimp and crabs) and from fungi cell wall | Sparingly soluble in water | Swelling |
| Sodium alginate | Sodium salt of alginic acid | Soluble slowly in water, hygroscopic nature | Swelling |
| Semisynthetic disintegrant | |||
| Croscarmellose sodium | Cross-lined sodium CMC form | 200 mesh particle size | Swelling |
| Sodium starch glycolate | Low substituted crosslinked carboxymethyl ether of poly-glucopyranose | 140mesh particle size Insoluble in water |
Uptake of water followed by rapid swelling |
| L-HPC | Low hydroxy propyl cellulose | Insoluble, but swells in water | Swelling and wicking |
| Hydroxy propyl starch | Obtained from natural starch | Insoluble in water | Swelling |
| Maize starch | Extracted from plant sources through a series of processing steps | Practically insoluble in cold water. In water with temperature 37°C starch swells | Swelling |
| Synthetic disintegrants | |||
| Crospovidone | Crosslinked polymer of N-vinyl-2-pyrrolidone | Practically insoluble in water | Swelling and wicking |
| Ion exchange resins | Crosslinked polyacrylic | Insoluble in water | swelling |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




