1. Introduction: The Missing Link Between Temperature and Immunity
Across virology and immunology, temperature remains one of the most fundamental yet under-utilized determinants of health. Fever-range temperatures are now recognized as an active modulator of immune efficiency rather than a by-product of infection, enhancing leukocyte mobility, cytokine signaling, and antibody binding (Evans, Repasky, & Fisher, 2015.) Viral replication rates and immune performance exhibit opposing dependencies on temperature: viruses generally replicate more efficiently under cooler conditions, whereas the immune system functions best when the body is warm. This asymmetry suggests that even small temperature shifts can determine the outcome of infection.
Most respiratory viruses—rhinovirus, influenza, and many coronaviruses—prefer temperatures around 33–36 °C, typical of the nasal cavity and upper airways. Human immune efficiency, in contrast, peaks near 37–38.5 °C. Within this narrow overlap lies a fragile equilibrium where the infection either escalates or is contained. When body temperature drifts downward within this shared zone, viral kinetics gain an initial advantage; when temperature rises, immune kinetics prevail. Recent immunological reviews confirm that even small shifts in core temperature recalibrate immune cell activation thresholds and influence both innate and adaptive responses (Lee & Amherst, 2021.)
The Body Heating Theory (BHT) explores how deliberate, short bursts of muscular thermogenesis can shift that equilibrium in favor of the host. By increasing core temperature early—often at the first signs of illness—the body can move out of the viral “comfort zone,” reducing replication while activating immune responses. The first onset of shivering represents a natural attempt to achieve this same effect through involuntary muscular contractions. Controlled physical activity simply accelerates the process safely and intentionally.
Because the immune system operates with its own temporal rhythm, typically reaching peak performance after three to four days (≈ 72–96 hours), rapid heating at the beginning of infection may effectively buy time—a physiological grace period—for full immune activation before viral populations reach exponential growth. This time-dependent advantage forms the theoretical foundation for the Body Heating Hypothesis developed later in this paper.
Beyond the theoretical and physiological rationale, one of the key motivations for developing this work is to promote a preventive approach to managing common colds and mild respiratory infections. Most popular over-the-counter remedies are not curative in a strict medical sense; they primarily suppress symptoms to make patients more comfortable while leaving the underlying viral activity largely unaffected. As a result, infections often persist longer, extending discomfort and delaying recovery.
Prolonged colds can also lead to secondary complications such as sinus congestion, bronchial inflammation, disturbed sleep, and weakened immune resilience against subsequent infections. These consequences highlight the need to move beyond symptom management toward active, physiological methods that strengthen the body’s natural defenses and shorten the course of illness—an approach embodied in the Body Heating Theory presented here.
2. Viral Growth Dynamics and Timing of Intervention
The progression of infection can be understood as a temperature-dependent competition between two biological systems with opposing requirements: the virus and the host immune response. Most respiratory viruses, including rhinoviruses and influenza strains, replicate most efficiently at cooler temperatures typical of the upper airways (≈ 33–36 °C), whereas the human immune system performs optimally within a slightly warmer range (≈ 36.5–38.5 °C). Within the narrow overlap of these ranges lies a dynamic equilibrium in which minor temperature shifts can determine whether infection expands or resolves.
When the body cools, viral enzymes gain kinetic advantage; when it warms, immune processes accelerate. This interplay defines the thermal battleground of infection, where even a half-degree change can alter outcomes. Typical respiratory viruses exhibit a time constant of approximately 24 hours for replication doubling, meaning that viral populations can increase substantially within a single day unless checked by immune activation. Thus, temperature regulation during this early phase is critical to infection outcome.
Understanding these reciprocal thermal dependencies clarifies that infection outcome depends not only on immune strength but also on the relative timing of temperature changes within the overlapping comfort zones of the virus and the host. The following section formalizes this relationship into a unified framework—the Body Heating Hypothesis (BHT)—which proposes that deliberate, early thermogenesis can shift this thermal balance decisively in favor of the immune system.
2.1. Viral and Immune Dynamics as a Function of Temperature
Viral population growth follows a sigmoidal pattern with three phases:
Lag (linear) phase – viral replication is slow; immune response just begins.
Exponential phase – viral load doubles rapidly; symptoms intensify.
Plateau/decline phase – immune system regains control.
Intervention through body heating must occur during the lag phase, typically within the first 24–36 hours after infection onset. Once the virus enters exponential growth, thermal manipulation alone cannot halt replication effectively.
Mathematically, if viral replication rate r is inversely proportional to body temperature T, then a 1°C rise can reduce replication rate by 10–15%.
The immune system’s performance, conversely, follows a logistic growth curve that can be advanced in time by early temperature elevation, effectively shifting the immune response curve leftward. This acceleration allows immune activity to peak sooner and overlap more effectively with the early stage of viral replication, increasing the chances of containment before the infection enters its exponential phase. The interplay between these two processes—the declining viral replication rate and the advancing immune activation—is reflected in the modeled curves (
Figure 1 and
Figure 2). This timing alignment, in which a modest thermal increase reshapes both trajectories, is the essence of the proposed strategy.
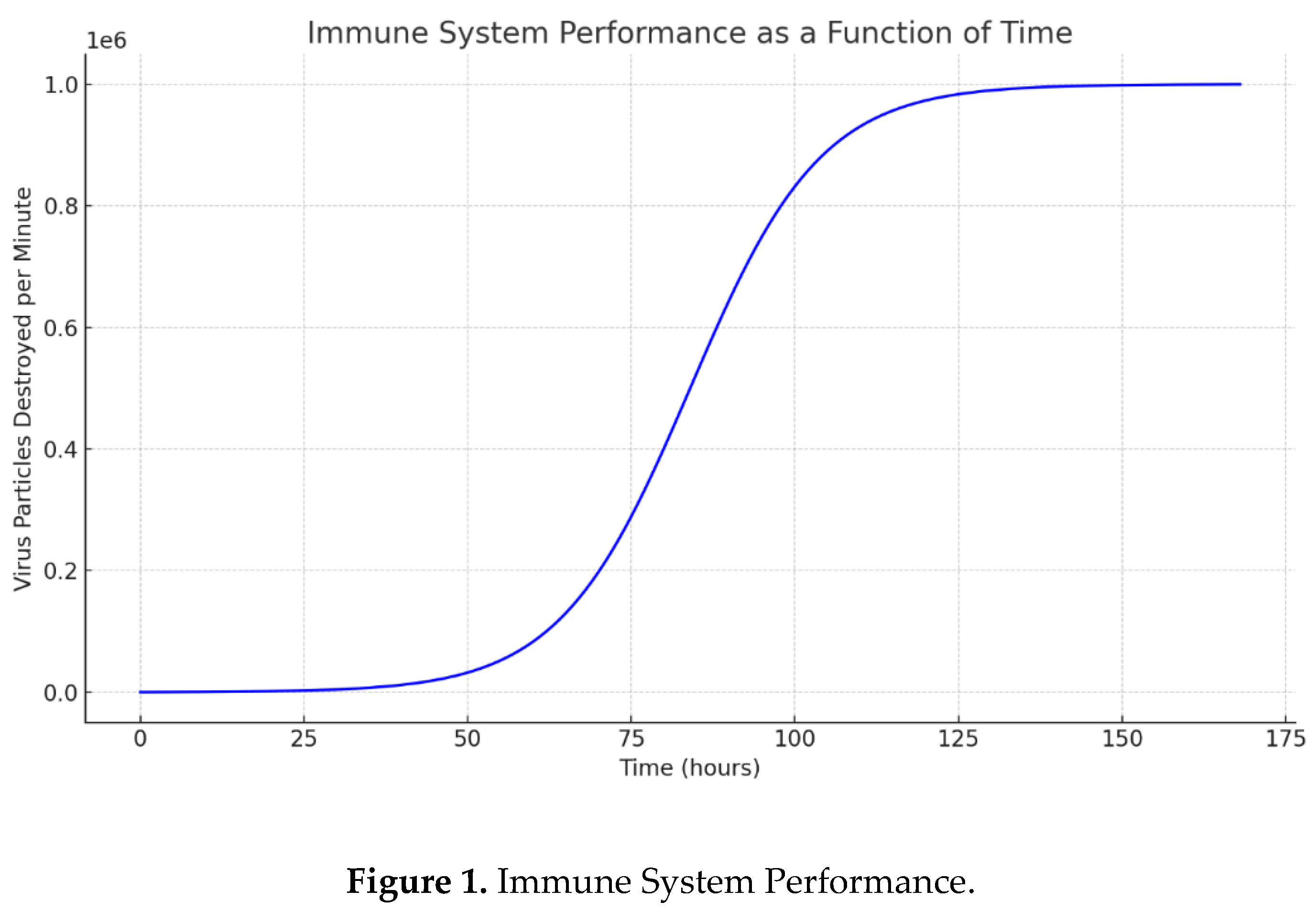
The immune system’s effectiveness can therefore be visualized as a sigmoidal growth curve, rising gradually during the first hours of infection and typically reaching its peak near day 7 (≈168 hours). This curve represents the progressive buildup of immune strength as white blood cells, antibodies, and cytokine signaling increase in coordination.
Typical rhinoviruses exhibit a time constant of roughly 24 hours for replication doubling, meaning that even modest delays in early viral growth can translate into markedly reduced symptom severity later.
Figure 1 below illustrates this relationship between immune performance and time, showing how an early thermogenic intervention—achieved through short, controlled bouts of exercise—can shift the response curve leftward. Such a shift enables the immune system to reach peak activity more quickly, thereby suppressing viral proliferation earlier in the infection cycle.
These modeled estimates provide quantitative insight into immune dynamics. Intervention through deliberate body-temperature elevation is expected to help the immune system attain its peak performance in significantly less than 168 hours, reinforcing the premise that timing is critical for successful viral control.
The immune system’s effectiveness can be visualized as a sigmoidal curve rising gradually from the onset of infection and reaching its peak near day 7 (≈168 hours).
These estimates provide some quantitative insight into immune system performance. Intervention by exercise-based body temperature control is expected to help the immune system reach its peak performance earlier — effectively shifting the curve to the left — so that maximum immune activity occurs in less than 168 hours.
To better understand the timing advantage of early heating, let us now consider the complementary view — how the virus population itself evolves over time.
To better understand the timing advantage of early heating, it is useful to examine the complementary process—the evolution of the viral population over time.
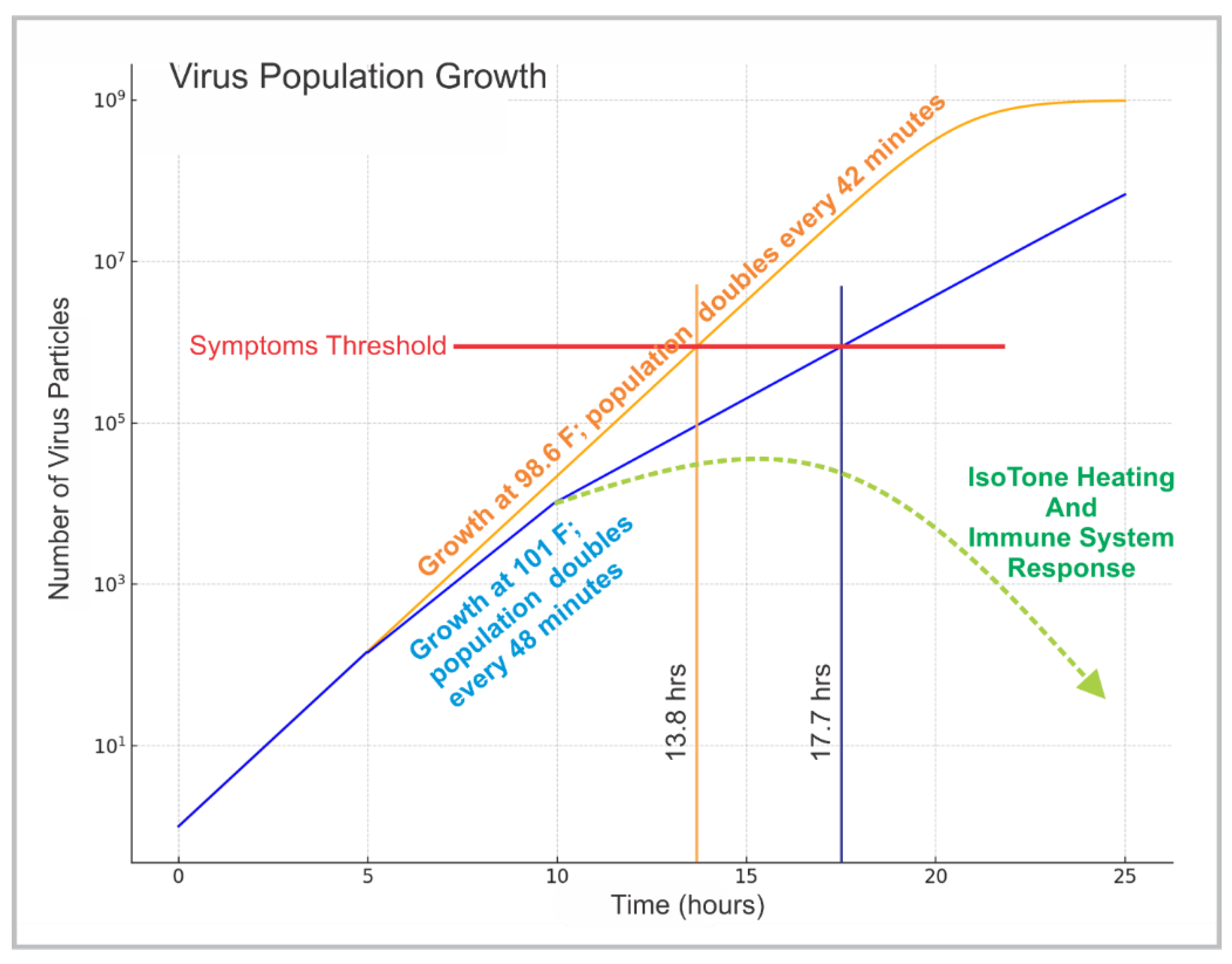
Figure 2 models this dynamic, showing how viral load increases gradually during the lag phase but accelerates sharply once replication mechanisms are fully engaged. This exponential rise represents the critical point where delayed intervention becomes far less effective. The comparison with the immune response curve demonstrates how a small, early increase in body temperature can flatten and delay the viral trajectory while advancing immune dominance, as reflected in the modeled curves (
Figure 1 and
Figure 2).
In a complementary calculation, early exercise-induced thermogenesis was shown to arrest this exponential surge if initiated soon after infection onset. The Symptoms Threshold in the plot marks the viral load at which infection effects reach maximum intensity. Maintaining elevated core temperature during the lag phase prevents the curve from crossing that threshold, keeping viral burden low until immune control is achieved.
Observations derived from the modeled curves indicate that the virus population remains in a slow, linear-growth phase for approximately 36–48 hours before entering exponential expansion. This early interval represents the most critical period for thermogenic intervention—when viral replication is still moderate, and the immune system can respond effectively with minimal physiological stress. Applying controlled heating during this window can dramatically alter infection kinetics by inhibiting viral enzyme activity and stimulating immune signaling before the infection becomes systemically entrenched.
This finding strongly supports the principle of early intervention. Thermogenic sessions—such as brief IsoTone “body-core heat-up” routines or comparable resistance-based movements—should ideally be performed as soon as potential exposure occurs rather than waiting for symptoms to emerge. In practical terms, this might mean initiating a short heating session immediately after returning home from public environments, meetings, or travel. Even a few minutes of deliberate muscular activation can raise core temperature enough to inactivate or destabilize viral particles before replication begins. Such proactive use of controlled thermogenesis functions as a physiological firewall—an immediate, non-pharmaceutical barrier against early infection establishment.
Understanding these reciprocal thermal dependencies clarifies that infection outcome depends not only on immune strength but also on the relative timing of temperature changes within the overlapping comfort zones of the virus and the host. The following section formalizes this relationship into a unified framework—the Body Heating Hypothesis—which proposes that deliberate, early thermogenesis can shift this thermal balance decisively in favor of the immune system.
2.2. The Body Heating Hypothesis (BHT)
The Body Heating Hypothesis (BHT) extends the thermal and temporal framework outlined above into a coherent model that links infection dynamics, temperature regulation, and muscular thermogenesis. It proposes that deliberate, early, and controlled elevation of core temperature through short bouts of physical activity can favorably alter the timing relationship between viral replication and immune activation.
2.2.1. Thermal Ranges and the “Crossover Zone”
Human physiology functions within a narrow immune comfort zone—approximately 36.5 to 38.5 °C—where cellular and humoral immunity operate most efficiently. Below ≈ 36 °C, leukocyte motility and cytokine signaling slow, while fever-range temperatures trigger heat-shock proteins and enhanced interferon activity.
Respiratory viruses possess their own viral comfort zone, roughly 33–36 °C, corresponding to the cooler mucosal surfaces of the nasal and upper respiratory tract. Above ≈ 37.5 °C, many viral enzymes lose activity; below ≈ 33 °C, replication also falters. The overlap between these zones (≈ 35–37 °C) defines a narrow thermophysiological battlefield in which both systems function, and even a 0.5–1 °C shift can determine which dominates.
Importantly, severe overcooling may drop temperature below the viral comfort range, halting replication entirely—explaining why not everyone exposed to cold develops illness. Yet mild chilling within the overlap region often gives viruses the kinetic edge.
2.2.2. Time Constant and the Grace Period
Typical rhinoviruses and seasonal influenza strains exhibit a replication time constant of ≈ 24 hours during the early exponential phase. Viral load thus doubles roughly once per day until the immune system mounts an effective response, usually after 3–4 time constants (72–96 h). Exercise-induced thermogenesis initiated during the first 24 hours can shorten the effective viral growth window and provide the immune system a grace period—a temporal advantage during which it can reach full activation before viral populations surge.
2.2.3. Exercise as Controlled Thermogenesis
Short, vigorous muscular effort increases core temperature by ≈ 0.5–1.5 °C while enhancing circulation and lymphatic flow. These effects simultaneously suppress viral replication and accelerate immune cell trafficking. Early activation of this process at the first shiver, chill, or throat irritation can therefore pre-empt the virus’s exploitation of its temperature range. Each brief exercise session effectively shifts the body into the immune-dominant zone, buying time equivalent to at least one replication constant and potentially preventing symptom escalation.
2.2.4. The Thermophysiological Crossover Point
The moment when immune efficiency surpasses viral replication rate defines the Thermophysiological
Crossover Point. Achieving this point early determines whether infection plateaus or advances. Controlled thermogenesis can move this crossover leftward in time, shortening illness duration and reducing symptom severity.
2.2.5. Implications for Conventional Treatment
Conventional cold medications generally alleviate symptoms—fever, congestion, or inflammation—without correcting the underlying thermal imbalance. By lowering fever and masking discomfort, they may prolong infection by keeping the body within the viral comfort zone while suppressing the very mechanisms that resolve illness. This paradox underscores the need for a preventive, physiology-based approach that supports, rather than suppresses, the body’s thermal defense.
Developing integrative thermotherapy—combining mild resistance exercise, hydration, and temperature monitoring—could transform early infection management from passive symptom relief to active restoration of immune–thermal equilibrium.
Summary of the BHT Framework
The immune system functions best within ≈ 36.5–38.5 °C; below this, its responsiveness declines.
Most respiratory viruses replicate fastest between ≈ 33–36 °C but are impaired above ≈ 37.5 °C and below ≈ 33 °C.
The overlap of these comfort zones forms the critical battlefield of infection; minor temperature shifts can decide the outcome.
Brief muscular thermogenesis grants a “grace period,” slowing viral kinetics long enough for adaptive immunity to develop.
Conventional medications address symptoms rather than thermal imbalance; active temperature management offers a more direct, curative path.
Collectively, these premises yield a testable prediction: early, controlled body heating through brief exercise can shorten infection duration by shifting immune activation ahead of viral replication by one or more time constants.
3. Shivering and Thermogenesis as Immune Triggers
Shivering represents the body’s intrinsic thermogenic reflex—its built-in version of the same mechanism described in the Body Heating Hypothesis. When peripheral thermoreceptors detect a drop below the core temperature set-point, the hypothalamus initiates rapid, rhythmic contractions of skeletal muscle. These contractions convert chemical energy into heat, raising core temperature typically by 0.5–1 °C within minutes. In the context of viral infection, this involuntary response functions as an early, biologically programmed attempt to restore the immune comfort zone before the virus gains kinetic dominance.
Physiologically, shivering is accompanied by increased cardiac output, elevated oxygen consumption, and activation of sympathetic pathways that accelerate blood and lymph flow. These responses mirror the effects achieved through deliberate muscular exercise but occur automatically—often at the onset of infection when cytokine signaling first resets the hypothalamic thermostat. The resulting temperature elevation stimulates interferon production and enhances leukocyte motility, effectively initiating the immune system’s counter-offensive.
Understanding shivering as a natural, self-regulating form of thermogenesis highlights the continuum between involuntary and voluntary heating responses. While the body’s reflexive mechanisms strive to re-establish thermal balance, intentional activation through controlled movement can amplify and stabilize this process, achieving the same immune benefit without the metabolic stress or discomfort associated with prolonged chills. In this sense, exercise-based heating is not an artificial substitute but a conscious extension of the body’s evolutionary design for thermal defense.
In summary, shivering serves as the body’s instinctive first line of thermal defense—an autonomic counterpart to deliberate muscular thermogenesis. Both rely on the same physiological pathways of energy conversion, circulatory stimulation, and neuroendocrine activation. Yet while shivering is reactive, triggered only after a measurable drop in core temperature, intentional movement can preempt this phase, maintaining the immune system within its optimal functional range.
This distinction underscores a central concept of the Body Heating Theory: the timing of heat generation is as critical as its magnitude. The next section examines how these thermogenic mechanisms influence broader physiological and immunological parameters—linking temperature, circulation, and immune signaling into a unified framework of infection control.
4. Physiological and Immunological Correlates
The effectiveness of body heating as a therapeutic strategy depends on the close integration of thermal, circulatory, and immune functions. Temperature elevation does more than raise metabolic rate—it reorganizes physiological priorities throughout the body. As core temperature rises, vasodilation increases peripheral and mucosal blood flow, improving oxygen delivery and immune-cell access to infected tissues. Elevated circulation enhances lymphatic drainage, facilitating the removal of viral debris and the transport of antigen-presenting cells to lymph nodes where adaptive immunity is initiated.
Within this coordinated response, timing again proves decisive. Early activation of these mechanisms, whether through reflexive shivering or deliberate muscular effort, preserves the immune system’s advantage during the first viral replication cycle. By contrast, delayed or suppressed heating allows viruses to exploit the thermal overlap zone, expanding rapidly before immune dominance is established.
The following subsections examine these interactions in detail—beginning with the temperature-dependent stability of viral structures, then exploring how immune activation and circulatory dynamics respond to controlled thermogenesis.
4.1. Virus Stability and Temperature
Most respiratory viruses display a sharp decline in infectivity above 38°C. Experimental data from airway-cell models demonstrate that common cold viruses replicate far less efficiently at 37–39 °C than at the cooler temperatures typical of nasal passages (Foxman et al., 2015; Evans et al., 2015.) This correlates with the downward bend of the viral curve beyond the ‘Symptoms Threshold’ line in
Figure 2.
For example, rhinoviruses lose structural integrity at 39°C, and SARS-CoV-2 replication is reduced by over 50% at febrile temperatures in vitro. Thus, even mild fever-range heating (38–39°C) can compromise viral replication cycles.
4.2. Immune Activation
Elevated temperature promotes the same early surge of immune efficiency
depicted in Figure 1, confirming that the modeled timing advantage aligns with known physiological mechanisms:
Faster migration of neutrophils and macrophages.
Increased T-cell activation and antibody binding efficiency.
Enhanced production of heat-shock proteins that improve viral antigen presentation.
Exercise-immunology studies refute the idea of post-exercise immune suppression, showing instead that moderate, repeated activity enhances immune surveillance and accelerates recovery (Campbell & Turner, 2018.)
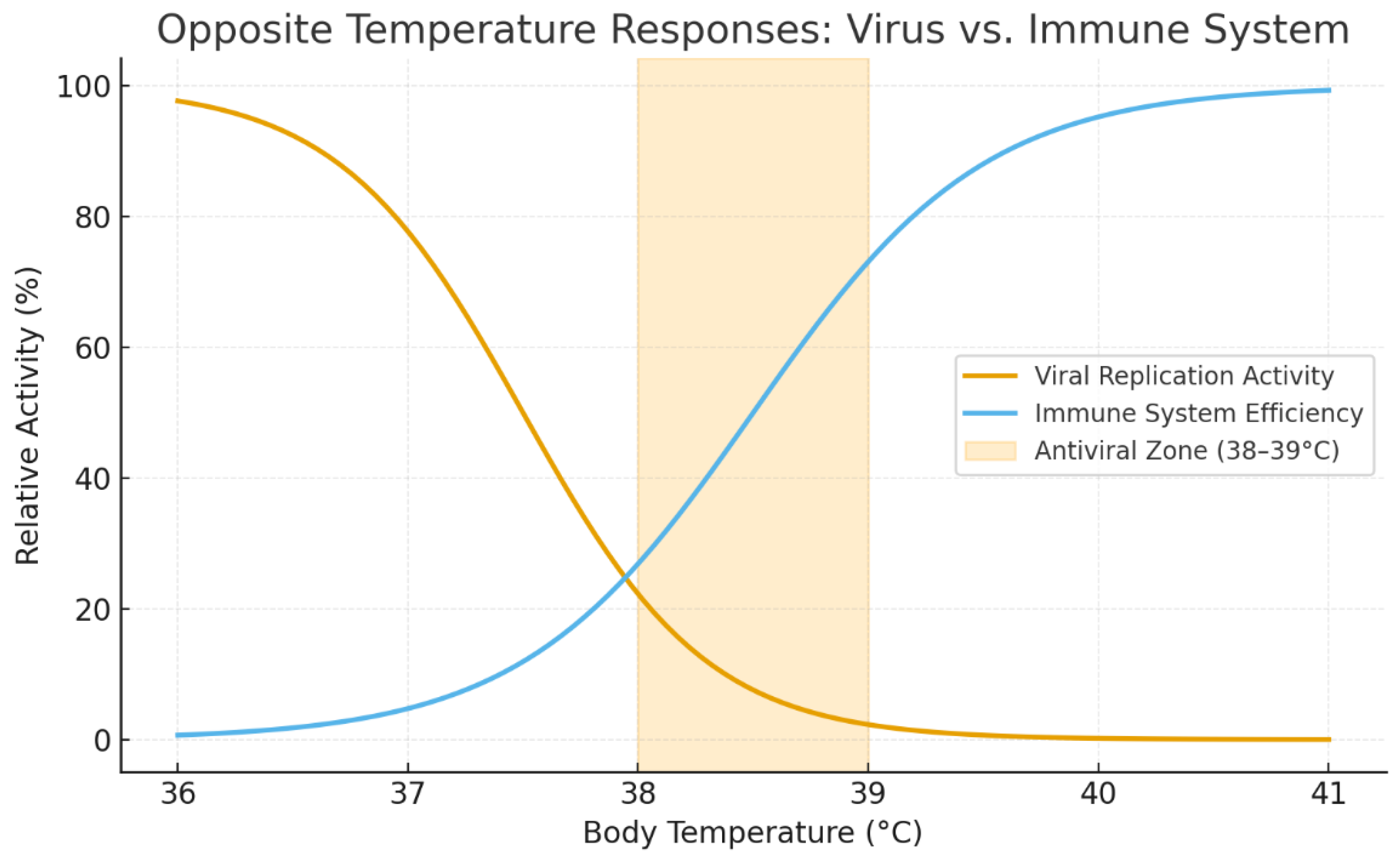
The opposing dependencies of viral replication and immune efficiency on temperature are illustrated in
Figure 3. Viral activity declines steeply above ≈ 37.5 °C, while immune effectiveness rises within the same range, reaching its functional peak near 38.5–39 °C. The intersection between the two curves defines the
antiviral zone—a narrow thermal window where even a 0.5–1 °C shift can reverse infection trajectory. This relationship provides the quantitative rationale for controlled body heating: deliberate muscular thermogenesis moves the body into this immune-dominant range long enough to inhibit viral replication and accelerate recovery.
This relationship provides the quantitative rationale for controlled body heating: deliberate muscular thermogenesis moves the body into this immune-dominant range long enough to inhibit viral replication and accelerate recovery.
4.3. Blood and Lymphatic Circulation
IsoTone’s resistance-based motion accelerates lymph flow, critical because the lymphatic system lacks its own pump and relies on muscular contraction. Adequate hydration further supports this process by maintaining lymph fluid volume and facilitating the transport and removal of viral debris through the same circulation that exercise activates. This supports antigen clearance and faster immune response mobilization. The lymphatic system’s role as both a transport and immune-signaling network underscores how muscular contraction directly amplifies immune communication and clearance (Olive & Bastien, 2023.) Comprehensive analyses of lymphatic transport reveal that rhythmic muscular motion is the primary driver of lymph propulsion and antigen delivery to lymph nodes (Hoeh et al., 2018.)
Taken together, these thermophysiological interactions reveal that temperature control during infection is not a passive byproduct of illness but an active, adaptive process. The body’s capacity to generate internal heat through muscular effort creates a dynamic system of feedback between temperature, circulation, and immune signaling—one that can be intentionally harnessed to accelerate recovery. Yet not all forms of heating are equivalent in effect or safety. External methods such as saunas, hot baths, or electric warming raise surface temperature but have limited influence on core thermal and immune dynamics, while internal or active heating through muscular work produces heat directly within deep tissues, where viruses replicate and immune cells operate. The next section compares these two modes of thermogenesis, examining their physiological efficiency, safety profiles, and therapeutic implications.
5. Comparative Analysis: Passive vs. Active Heating
Although both passive and active heating can elevate body temperature, their physiological pathways and clinical implications differ profoundly. Passive methods—such as warm baths, saunas, electric blankets, or external heat pads—transfer energy from the environment inward, producing comfort and superficial warmth but only modest increases in core temperature.
By contrast,
active thermogenesis, generated by voluntary muscular contraction, produces heat internally at the very sites where immune reactions occur and viruses replicate. This distinction defines not only the depth of heating but also its biological consequences. Passive heating primarily dilates surface vessels and relaxes musculature, while active heating simultaneously enhances cardiovascular output, stimulates lymphatic flow, and activates neuromuscular feedback that supports immune signaling. The effectiveness of any heating strategy therefore depends less on the external temperature applied than on the
origin and distribution of the heat itself.
| Method |
Mechanism |
Depth of Heating |
Safety |
Portability |
| Blanket / clothing |
Surface insulation |
Low (skin only) |
High |
High |
| Sauna / hot bath |
External conduction |
Moderate |
Moderate (risk of fainting) |
Low |
| IsoTone exercise |
Internal metabolic generation |
High (core) |
Adjustable / Safe |
Very high |
Active thermogenesis via IsoTone therefore offers superior physiological efficiency with minimal risk and immediate availability. As shown in the comparison above, passive methods provide surface comfort and temporary relief, while active approaches generate measurable physiological change at the core level where immune responses and viral activity intersect.
The comparison highlights that only active, muscle-driven thermogenesis can achieve deep, sustained heating without external equipment or risk of overheating. This insight sets the foundation for integrating IsoTone-based exercise not merely as an emergency intervention, but as a preventive and complementary practice for year-round immune resilience discussed in the following section.
In summary, passive heating methods provide transient surface warmth and subjective comfort but do little to alter the internal thermodynamic or immunological environment where infection develops. Active, muscle-driven thermogenesis, by contrast, produces targeted heating at the cellular level, improves oxygen and nutrient delivery, and triggers systemic immune activation without reliance on external heat sources. This internal origin of heat makes active methods inherently self-regulating: as core temperature rises, muscular effort naturally declines, preventing overheating while sustaining optimal immune conditions.
Recognizing this difference transforms body heating from a comfort measure into a functional preventive tool. The following section expands this perspective, exploring how controlled thermogenesis can be integrated into daily routines to maintain immune readiness and reduce susceptibility to infection.
6. Complementary and Preventive Aspects
The same mechanisms that make controlled body heating effective during infection can also serve as a preventive measure when practiced regularly. Mild, periodic thermogenesis enhances vascular tone, optimizes microcirculation, and maintains immune readiness even in the absence of illness. By repeatedly shifting the body into its immune comfort zone, these brief temperature elevations reinforce adaptive thermoregulation and improve resistance to both viral and inflammatory stressors. Integrating short thermogenic sessions—whether through structured resistance exercise or spontaneous daily movement—thus provides a simple yet potent form of physiological conditioning.
When combined with hydration, balanced nutrition, and adequate rest, this approach functions as a complementary health practice, strengthening overall resilience and reducing the likelihood that routine viral exposures progress into symptomatic infection. Preventive thermogenesis also supports recovery from fatigue and improves metabolic efficiency, offering additional benefits across cardiovascular, respiratory, and musculoskeletal systems. In this sense, maintaining warmth through controlled muscular activation becomes not merely an adjunct to fitness but a continuous strategy for immune optimization.
Vaccination and nutrition (vitamins C, D, zinc) remain essential; heating complements, not replaces, immune protection.
Chronic cold exposure (e.g., refrigerated workplaces) correlates with higher viral incidence—an observation consistent with the model. Extended exposure effectively keeps the infection curve in the steep growth zone of Figure 2, delaying the crossover to immune control shown in Figure 1.
IsoTone preventive micro-sessions—five 4-minute bursts daily—can maintain circulatory vigor and thermal resilience during winter months.
In this broader preventive framework, IsoTone becomes more than a reactionary tool—it evolves into a daily instrument of physiological maintenance, harmonizing circulation, temperature balance, and neuromuscular activation. Regular use fosters an internal environment that is less favorable to viral persistence and more supportive of overall systemic health.
These same thermoregulatory and circulatory mechanisms also explain IsoTone’s demonstrated benefits in seemingly unrelated conditions, from muscular cramps to hypothermic exposure, forming the scientific bridge to the broader applications reviewed in the next section.
Viewed collectively, these preventive practices demonstrate that controlled thermogenesis can be more than a temporary defense against infection—it can serve as an ongoing mechanism for maintaining systemic balance and health. Regular activation of internal heat generation enhances circulation, stabilizes metabolic rate, and promotes efficient immune surveillance, particularly in environments where sedentary habits, artificial cooling, or stress suppress natural thermoregulation.
As a result, preventive body heating not only reduces the likelihood of respiratory infections but may also mitigate broader risks linked to chronic low-grade inflammation, vascular stiffness, and impaired recovery. The next section extends this perspective, exploring how the same thermophysiological principles apply across multiple health domains and clinical contexts.
A detailed regimen for applying these preventive principles is outlined in
Section 11.
7. Broader Applications and Evidence Parallels
The physiological mechanisms that underpin controlled body heating extend beyond viral defense and general prevention; they form a foundation for multiple therapeutic and performance-enhancing applications. The same increase in core temperature, circulation, and neuromuscular activation that suppresses viral activity also promotes tissue repair, pain reduction, and recovery from fatigue.
Clinical and empirical observations support this broader view: thermogenic stimulation has shown measurable benefits in managing muscular cramps, joint stiffness, and circulation-related disorders, while improving functional outcomes in both rehabilitation and athletic training. These parallels confirm that the principles governing temperature, perfusion, and immune balance represent a unifying biological process rather than an isolated response to infection.
Observations across diverse applications—ranging from cramp prevention and therapeutic massage to cold-weather survival and extended immobility during travel—reveal a consistent pattern: controlled internal heating enhances cellular resilience, restores circulation, and supports the body’s natural repair systems. This section summarizes these broader findings to illustrate the scientific coherence of the IsoTone method across multiple health domains.
The same thermogenic mechanism underlies IsoTone’s documented roles in:
Cramp prevention: localized heating and massage reset hyperactive neuromuscular loops.
Therapeutic massage: heat-enhanced blood flow accelerates recovery and tissue repair.
Cold survival scenarios: maintaining body temperature under hypothermic stress.
Airplane and sedentary environments: circulation stimulation to prevent thrombosis.
These cross-domain examples reinforce the central claim: controlled heat generation through muscular resistance is a versatile survival and health mechanism.
Taken together, these examples demonstrate that the benefits of controlled thermogenesis are not confined to viral defense but represent a universal physiological principle: warmth generated from within sustains vitality, mobility, and recovery across nearly all body systems. IsoTone serves as a practical embodiment of this principle—transforming muscular effort into a therapeutic source of heat, circulation, and neural activation.
The cumulative evidence points toward a unified model of internal heating as a cornerstone of human health, setting the stage for a broader discussion of its evolutionary logic and implications presented in the following section.
Taken together, these findings suggest that thermogenesis functions as a universal physiological integrator—linking metabolic activity, circulation, and immune performance into a single adaptive framework. Whether applied to viral suppression, muscular recovery, or chronic pain management, the mechanism remains fundamentally the same: internally generated heat restores balance where localized cooling, reduced blood flow, or cellular stress disrupt homeostasis.
This consistency across systems implies that the capacity for heat generation evolved not only for survival in cold environments but as a core element of self-repair and defense. The next section examines this concept from an evolutionary perspective, exploring how the ability to generate and regulate internal warmth shaped human adaptation, resilience, and the fundamental interplay between movement and health.
8. Discussion: Evolutionary Logic
The ability to generate and regulate internal heat represents one of the most defining evolutionary achievements of the human organism. Long before it became a conscious health strategy, thermogenesis served as a fundamental survival mechanism—allowing early humans to maintain metabolic stability, remain active in cold environments, and sustain immune function despite environmental stress. Unlike externally dependent species, humans evolved a powerful internal heating system that couples movement with immunity: muscular activity not only produced mechanical work but simultaneously preserved the temperature range in which immune and enzymatic processes operate most effectively. In this sense, every act of motion carried a dual purpose—supporting both survival and resistance to disease.
Over time, as lifestyles grew more sedentary and external sources of comfort replaced physical effort, this evolutionary synergy weakened. Modern heating, clothing, and pharmacological aids insulated humans from environmental variation but also reduced the frequency of natural thermogenic episodes that once sustained vascular tone and immune vigilance. The Body Heating Theory seeks to reconnect this ancient biological link between movement, warmth, and health, translating an evolutionary advantage into a modern preventive practice grounded in measurable physiology.
In this context, IsoTone represents a technological return to a deeply rooted biological strategy: reclaiming the body’s intrinsic capacity to heal through motion-induced warmth. The following discussion examines this principle as an adaptive legacy of evolution, connecting ancient physiology with modern self-regulation.
The immune system’s design already incorporates temperature elevation (fever) as a fundamental defense strategy. Fever is not a malfunction or collateral symptom—it is a tightly regulated, evolutionarily conserved process triggered by cytokines such as interleukin-1, interleukin-6, and tumor necrosis factor-α, which act on the hypothalamus to raise the body’s thermal set point. This controlled rise in core temperature inhibits replication of many pathogens while simultaneously enhancing immune cell performance. The body, in effect, uses heat as a selective biological weapon. Fever’s evolutionary persistence suggests that natural selection favored organisms capable of leveraging internal heat as a defense strategy rather than suppressing it pharmaceutically (Rand & White, 2020.)
The proposed IsoTone-based method does not replace this natural fever mechanism; rather, it accelerates and amplifies it by initiating thermogenesis before the body fully commits to fever production. Instead of waiting for the immune system to react chemically, deliberate muscular activation produces heat mechanically, achieving the same end through different physiological channels. Muscle contraction is one of the most efficient heat-producing processes in the body, responsible for nearly 80% of thermogenesis during physical effort. By harnessing this process intentionally and early, IsoTone transforms a passive biological reflex into an active, self-regulated defense response—an “artificial fever” that remains entirely physiologically compatible.
From an evolutionary perspective, this approach mirrors one of the oldest survival strategies in nature. Early humans and their mammalian ancestors did not survive environmental cooling through passive insulation alone, but through movement-induced heat production. Shivering, running, and the instinctive urge to stay active in cold weather were not mere behaviors—they were essential life-preserving responses shaped by selective pressure. Individuals capable of rapid internal heating gained a decisive advantage: they resisted hypothermia, maintained immune vigilance, and could recover from infections that crippled less adaptable members of the population.
Over evolutionary time, this link between motion and immunity became hardwired into our physiology. The neuromuscular system, vascular system, and immune system co-evolved as a coordinated network, where muscle activity supported not only mobility but also circulatory and immunological resilience. Every contraction promotes blood and lymph movement, oxygenation, and heat distribution—processes essential to survival in a world where both predators and pathogens were constant threats.
In this light, the modern tendency toward physical inactivity can be viewed as an evolutionary mismatch. Our ancestors relied on constant muscular engagement to maintain warmth, metabolic balance, and immune strength, whereas contemporary lifestyles often confine people to sedentary environments with external climate control. The result is an under-stimulated thermoregulatory system and a less responsive immune baseline. The IsoTone device re-creates the missing physiological signal by allowing individuals to restore natural heat-movement cycles within minutes, even in static or confined conditions such as offices, airplanes, or hospital recovery rooms.
Moreover, IsoTone’s method of controlled resistance carries an important evolutionary symmetry: it converts effort directly into heat and circulation, just as hunting, climbing, or shelter-building once did—but without requiring space, risk, or extreme endurance. This makes it not merely an exercise device but a modern re-expression of a primal survival mechanism. Through IsoTone, humans can once again leverage the same thermogenic processes that sustained them for millennia, now refined and directed with precision.
In summary, fever and movement are two expressions of the same evolutionary principle—internal heat as protection. Fever arises involuntarily from infection; IsoTone thermogenesis invokes it voluntarily through motion. Both share the same goal: to create an internal environment hostile to pathogens and supportive of immune and metabolic harmony. IsoTone thus bridges biology and technology, offering a compact and controllable tool that revives an ancient adaptation for modern health and longevity.
Understanding thermogenesis as an evolutionary adaptation reframes body heating not as an artificial intervention, but as a restoration of an ancient equilibrium between physiology and environment. Modern humans possess the same intrinsic mechanisms that once enabled survival through cold, infection, and stress—yet these systems now lie underused in an age defined by comfort and inactivity.
By consciously reactivating this capacity through brief, controlled physical effort, it becomes possible to reclaim a natural form of resilience that evolution refined but modern living subdued. The next section translates these concepts into practical application, outlining how deliberate thermogenic exercise can be structured, measured, and studied to validate the Body Heating Theory in clinical and real-world settings.
9. Practical Implementation and Research Outlook
Translating the Body Heating Theory into practice requires structured yet adaptable methods that enable individuals and researchers to apply its principles safely and effectively. The objective is twofold: first, to make the body’s natural heat-defense mechanisms readily accessible through deliberate, controlled movement; and second, to design research pathways that quantify the resulting physiological and immunological benefits.
Controlled muscular thermogenesis can be achieved with simple, portable resistance tools or minimal-equipment exercises capable of generating rapid internal heating without external aids. When guided by measured feedback—such as temperature change, heart rate, or perceived exertion—these sessions can reproduce the beneficial effects of fever or shivering while maintaining user comfort and control.
The following subsections outline practical recommendations for implementation, preventive conditioning, and experimental validation, establishing a foundation for both individual use and future clinical research.
9.1. Controlled Heating Principles and Instrumentation
Implementing controlled thermogenesis begins with short, well-defined exercise sessions designed to elevate core temperature safely and predictably. The aim is not maximal exertion but precision in thermal response—achieving a measurable rise of approximately 0.5–1.5 °C within minutes through rhythmic muscular engagement. By integrating brief thermogenic bouts into daily routines, users can actively maintain immune readiness while minimizing fatigue or overexertion. The following guidelines outline practical methods and parameters for applying this approach across diverse age and fitness levels.
A portable adaptive-resistance device fulfills this function by automatically matching the user’s force in both compression and expansion phases, creating continuous 360-degree resistance. Every movement engages large muscle groups, producing proportional energy conversion into metabolic heat. Within three to five minutes of moderate-to-vigorous use, most individuals experience a core-temperature rise of 0.5–1.5 °C, sufficient to push internal conditions beyond the viral comfort range and enhance immune kinetics.
The principal advantages of such an adaptive-resistance mechanism include:
Precision and safety: self-adjusting load prevents overheating or overexertion.
Accessibility: compact design allows use in nearly any environment—at home, at work, in hospitals, or during travel.
Dual functionality: resistance exercise and therapeutic self-massage, supporting both fitness and recovery.
Thermal efficiency: metal and composite elements retain and radiate warmth, reinforcing post-exercise heating effects.
This approach merges physiology with practicality: it translates muscular effort directly into internal energy, turning motion into medicine. The device or comparable method serves only as a facilitator—the essential principle is controlled muscular thermogenesis, achievable wherever deliberate movement is possible.
With this foundation, the following section presents the Early-Response Routine designed to employ these principles during the crucial first hours of infection onset.
9.2. Early-Response Routine (“Anti-Cold Protocol”)
The same thermogenic principles that accelerate recovery during infection can be applied proactively to prevent illness and support chronic-care rehabilitation. Regular activation of internal heat generation sustains vascular elasticity, improves circulation, and helps the immune system remain responsive under environmental or metabolic stress. In clinical contexts, these methods may complement existing therapies by enhancing local perfusion and metabolic turnover, thereby accelerating healing and recovery. The recommendations that follow describe timing, frequency, and safe integration of thermogenic activity within both preventive and therapeutic programs.
The Early-Response Routine, sometimes referred to as the Anti-Cold Protocol, is designed to generate rapid internal heat by engaging multiple large muscle groups through brief, rhythmic resistance movements. Each motion should be performed with deliberate intensity, emphasizing controlled breathing and full muscular engagement. The entire sequence can be completed in just a few minutes, even in confined spaces such as offices, homes, or recovery settings.
Expanded daily scheduling guidelines appear in
Section 11, Practical Regimen for Applying BHT.
Recommended sequence
Elbow Squeeze: perform rapid push-pull motions at chest level to activate arm, shoulder, and chest muscles, stimulating circulation through the upper body.
Front Press: execute forward horizontal presses to raise temperature in the pectoral and upper-spinal regions.
High Press: extend the arms overhead with firm resistance to accelerate cardiovascular and respiratory response.
Iso-Squat Front/Back: incorporate a full-body drive by alternating short squats and upright extensions, engaging the legs, core, and back to maximize heat generation.
Each round should include 40–50 repetitions per exercise at near-maximum but sustainable effort. Three rounds are sufficient to elevate core temperature by approximately 0.5–1.5 °C in most individuals. Rest briefly between rounds to maintain hydration and prevent overexertion.
Supportive measures
Drink electrolyte-balanced water before and after the session to offset fluid loss from heating.
Avoid heavy clothing; allow heat to disperse evenly across the body.
Continue 2–3 short sessions daily until symptoms subside.
This rapid-response approach enables individuals to harness their natural thermoregulatory mechanisms at the earliest indication of infection. By initiating deep muscular thermogenesis before the virus enters exponential growth, the body gains valuable hours—often enough to suppress replication and prevent progression to severe symptoms.
When applied consistently, the Anti-Cold Protocol demonstrates how motion itself can become an immediate form of preventive medicine: a safe, drug-free, and evolutionarily grounded strategy for reinforcing immune readiness whenever the first signs of illness appear.
This rapid-response approach demonstrates the body’s capacity to counter infection through self-generated thermogenesis; when practiced regularly, the same mechanism that defends against early viral invasion can also serve as a foundation for sustained immune conditioning—an idea explored in the following section.
9.3. Early Intervention and Timing of Response
The effectiveness of thermogenic intervention depends critically on timing. Infection modeling and empirical observation indicate that viral populations remain in a slow, linear phase for roughly 36–48 hours following exposure before entering exponential growth. This period defines a narrow but powerful therapeutic opportunity—when the viral load is still manageable and immune mechanisms can easily regain control.
Implementing controlled heating during this phase can halt or substantially delay infection progression. A practical recommendation is to perform one or two brief thermogenic sessions shortly after potential exposure—such as after social gatherings, air travel, or time spent in enclosed public spaces—and repeat periodically during the first 24 hours. These “early-response” sessions act as preventive micro-interventions, promoting circulation, boosting innate immunity, and elevating tissue temperature above the viral comfort range before replication can accelerate.
Such early action requires minimal effort yet yields disproportionate benefits, often preventing symptoms entirely or reducing illness duration to a fraction of its normal course. In a broader health context, this approach represents a behavioral evolution in infection management—from reactive symptom relief to proactive thermal immunity.
9.4. Preventive and Clinical Applications
To advance the Body Heating Theory from conceptual model to clinical standard, systematic research is required to quantify its effects on immune performance, viral kinetics, and recovery outcomes. Controlled comparative studies—contrasting early thermogenic intervention with conventional rest or medication-based protocols—can determine the magnitude and timing of measurable benefits.
Complementary physiological metrics such as temperature profiles, heart-rate variability, cytokine levels, and symptom duration should be tracked to evaluate efficacy. The subsections below propose initial study designs and validation strategies for future experimental work.
In practical terms, prevention and early response rely on the same physiological mechanism—timely muscular activation that elevates core temperature and mobilizes the immune system before viral replication accelerates. For both clinical and personal use, the following guidelines summarize the recommended practice:
At first symptom: initiate a focused heating session lasting 2–5 minutes to counteract the earliest phase of infection when viral populations remain low and most vulnerable.
Frequency: repeat the session every 3–4 hours during waking periods for the first 24 hours to sustain elevated immune readiness and inhibit viral growth.
Hydration and rest: maintain adequate fluid balance and avoid overexertion, as dehydration and fatigue may offset thermogenic benefits.
Preventive conditioning: incorporate brief, moderate sessions into daily wellness routines as thermal conditioning—a simple, self-applied method for improving circulation and maintaining immune responsiveness during colder months or sedentary work conditions.
For clinical adaptation, these principles could be integrated into rehabilitation programs, geriatric care, or post-infection recovery protocols, where maintaining circulation and moderate core temperature can improve healing and reduce complications.
The consistent application of these preventive and clinical strategies not only strengthens individual immunity but also provides an emerging framework for quantitative study—linking measurable physiological effects, such as temperature and circulation changes, to documented improvements in immune performance, as discussed in the next section.
9.5. Integration into Public Health and Preventive Medicine
The long-term success of controlled thermogenesis depends not only on individual practice and research validation but also on its inclusion within broader preventive-health frameworks. Education about the physiological role of temperature in immunity should become part of public-health messaging, emphasizing that mild, regular physical activation can reduce both infection incidence and duration. Workplace wellness programs, senior-care facilities, and post-rehabilitation settings could adopt short thermogenic routines as daily preventive measures, much like existing recommendations for hydration or stretching.
Integration at this level requires interdisciplinary collaboration among clinicians, physiologists, and policymakers to define safe guidelines, monitoring standards, and inclusion criteria for at-risk populations. Community-based studies could provide real-world data on adherence, outcomes, and quality-of-life improvements, helping to translate laboratory findings into scalable health-promotion strategies. In this way, the Body Heating Theory extends beyond a scientific model to a practical public-health tool—linking physiology, behavior, and education in a single, accessible framework for disease prevention.
Public-health programs could incorporate timing-based preventive strategies derived from the Body Heating Theory, emphasizing the importance of early thermogenic activation within the first 48 hours of potential viral exposure. Simple public messaging—encouraging short “heat-up” sessions after travel, meetings, or crowded interactions—could significantly reduce transmission rates and symptom severity. When practiced consistently, such brief interventions would serve as a population-wide immune conditioning tool, complementing vaccination and hygiene campaigns by targeting the physiological conditions that favor viral establishment.
Together, these practical and research initiatives translate the Body Heating Theory from conceptual framework into a measurable, testable approach to immune enhancement and viral suppression. By combining self-applied thermogenic exercise with structured clinical evaluation, it becomes possible to quantify the physiological thresholds and time constants that define optimal immune activation. Such work will not only confirm the mechanistic basis of controlled thermogenesis but also establish safe, replicable standards for everyday use in both preventive health and rehabilitation. With a clear experimental roadmap and growing physiological evidence, the transition from theory to application is within reach—setting the stage for the broader implications summarized in the Conclusion that follows.
10. Conclusion and Future Directions
The Body Heating Theory proposes that deliberate, early, and controlled elevation of core body temperature through brief, vigorous muscular activity can suppress viral replication, accelerate immune activation, and shorten illness duration. The theoretical framework identifies a measurable overlap between two thermal comfort zones—one favoring viral replication (≈ 33–36 °C) and one favoring immune efficiency (≈ 36.5–38.5 °C). Within this crossover range, even minor temperature shifts can reverse the competitive balance between host and pathogen.
Exercise-induced thermogenesis not only modifies this thermal environment but also introduces a grace period during which the immune system can reach its peak effectiveness before viral growth accelerates. By raising core temperature quickly—often by 0.5–1.5 °C—controlled physical activity moves the body out of the viral comfort zone and into the immune-dominant range. The average time constant of infection development for typical cold viruses is about 24 hours; interrupting or shortening even one of these early replication cycles can markedly reduce symptom severity and overall illness duration.
Equally important, the theory underscores that conventional over-the-counter cold remedies primarily treat symptoms rather than causes. By lowering fevers and suppressing natural inflammatory responses, they may inadvertently extend infection by keeping the body within the viral comfort zone while blunting its thermoregulatory defense. This paradox highlights the need to rethink current therapeutic strategies. Instead of masking discomfort, future interventions should aim to restore physiological balance by supporting the body’s natural temperature-based immune mechanisms.
The approach proposed here—controlled, exercise-based body heating—offers a scientifically grounded, testable means of achieving that goal. It aligns with evolutionary logic, reinforces innate defenses, and provides a foundation for developing integrative thermotherapy protocols that combine moderate physical activation, hydration, and monitored temperature feedback. Through such methods, early-stage infections could be managed proactively, reducing reliance on symptom-suppressing drugs and promoting faster, natural recovery.
The following section summarizes a practical daily schedule translating these findings into safe, repeatable thermogenic sessions for cold prevention.
Looking ahead, research should focus on quantifying the physiological limits, timing thresholds, and biochemical markers associated with thermogenic immune activation. Comparative trials can clarify the magnitude of benefit relative to conventional treatments and identify optimal session durations and temperature targets for various populations. Equally promising are community-level initiatives that incorporate controlled thermogenesis into public-health programs, using simple, measurable exercises to enhance immune readiness year-round. Digital health tools, wearable sensors, and adaptive-resistance devices could facilitate personalized feedback, ensuring both safety and efficacy.
Ultimately, the Body Heating Theory reframes movement and temperature as complementary elements of immune health. By viewing exercise not merely as conditioning but as a direct, measurable antiviral intervention, this framework points toward a new paradigm in preventive medicine—one that replaces passive symptom management with active, physiological self-defense. Continued interdisciplinary research will determine its full clinical and societal potential, but the principle is clear: the heat we generate may be our most ancient and most underused medicine.
11. Practical Regimen for Applying the Body Heating Theory
Translating the Body Heating Theory into a usable preventive and therapeutic method requires a structured yet flexible regimen that reproduces the benefits of mild fever through self-regulated muscular thermogenesis. The objective is to elevate core temperature into the antiviral zone (≈ 38.3–39.0 °C) for controlled periods while avoiding fatigue or dehydration.
11.1. Physiological Rationale
Short, rhythmic bouts of resistance-based exercise create internal heat at the precise depth where immune and viral processes interact. Each thermogenic pulse temporarily suppresses viral replication and accelerates immune signaling. Repetition of these pulses through the day mimics the natural wave-like pattern of fever, giving the immune system multiple activation windows without the risks associated with continuous exertion.
A comprehensive review of physical activity and immunity confirms that moderate thermogenic exercise consistently elevates circulating immune markers and reduces respiratory infection risk (Nieman & Wentz, 2019.)
11.2. Session Structure
To apply the Body Heating Theory effectively, each session must follow a reproducible structure that balances heating efficiency, comfort, and safety. The goal is not endurance training but precisely timed thermogenesis that moves the body into the immune-dominant temperature range without inducing strain. The parameters below summarize the optimal framework for a single IsoTone session—its duration, intensity, recovery period, and temperature targets—allowing the user to regulate effort while achieving measurable physiological impact.
| Parameter |
Recommended Range |
Purpose |
| Duration |
20–25 minutes |
Achieve a 0.8–1.2 °C core rise; light perspiration indicates target reached |
| Intensity |
Moderate to vigorous rhythmic IsoTone motion engaging large muscle groups |
Maximize uniform internal heating |
| Recovery |
10–15 minutes gentle movement and hydration |
Prevent over-heating and maintain circulation |
| Temperature goal |
38.3–39.0 °C core, 36–37 °C nasal mucosa |
Enter antiviral zone without distress |
Each session should include several continuous squeeze–expand cycles (≈ 40–50 reps) emphasizing full-body coordination and controlled breathing.
11.3. Recommended IsoTone Thermal–Immune Intervention Schedule
While a single session can generate a transient antiviral effect, the true benefit of controlled thermogenesis emerges from rhythmic repetition throughout the day. The following schedule translates the Body Heating Theory into a practical daily pattern that parallels the body’s natural circadian and immune rhythms. By spacing brief heating sessions at key times—morning activation, midday peak, afternoon maintenance, and evening recovery—the regimen sustains immune alertness while avoiding fatigue. Each period contributes to the cumulative “warm time” required for consistent antiviral protection.
| Time of Day |
Duration |
Physiological Goal |
Typical Activities |
| Morning (after waking) |
10–15 min mild warm-up |
Initiate circulation, pre-heat nasal mucosa |
Light IsoTone motions or brisk walk |
| Midday / Early afternoon |
20–25 min moderate session |
Raise core to 38.5 °C; primary antiviral cycle |
Full IsoTone resistance routine |
| Late afternoon |
10 min maintenance |
Sustain immune activation, improve lymph flow |
Short rhythmic micro-session |
| Evening |
15–20 min gentle session |
Second immune wave, relaxation |
Lower-intensity IsoTone or stretching |
| Cumulative “warm time” |
1.5–2 hours / day |
Maintain repeated antiviral heating cycles |
— |
This schedule ensures two to three thermogenic peaks daily, approximating the amplitude and timing of physiological fever oscillations.
11.4. Timing in Relation to Exposure or Early Symptoms
Timing remains the decisive factor in determining whether thermogenic intervention merely supports recovery or completely prevents illness. Because viral replication accelerates exponentially after the first 24–36 hours of exposure, the earliest application of heat can interrupt the infection cycle before symptoms fully develop. The guidelines below specify how to align IsoTone sessions with this biological clock—initiating them immediately after potential exposure and repeating them during the critical early phase when viruses are still vulnerable to temperature-driven suppression.
Begin a session within 24 hours after potential viral exposure (travel, meetings, crowded events).
During the first 36–48 hours of infection, repeat 10–15 minute sessions every 3–4 hours while awake to inhibit viral multiplication during its slow-growth phase.
Continue two daily sessions until symptoms subside or full recovery occurs.
11.5. Supportive Measures
Thermogenesis is most effective when supported by complementary physiological practices that maintain fluid balance, airway warmth, and gradual cooling. Hydration, in particular, plays a dual role: it prevents dehydration from heat production and enhances immune efficiency by sustaining plasma and lymphatic flow. This synergy between fluid movement and muscle-driven circulation enables the lymphatic system to dilute and flush viral debris, reinforcing the antiviral benefits of each session. The following measures ensure that heating remains both effective and restorative.
Hydration: drink warm, electrolyte-balanced water before and after each session. Hydration does more than replace fluid loss — it enhances immune efficiency by maintaining plasma volume and stimulating lymphatic circulation already energized by vigorous muscular activity. The resulting fluid flow through the lymphatic system helps dilute and wash out viral particles and metabolic waste, accelerating immune clearance from tissues. Controlled laboratory trials show that combined heat stress and hydration during exercise modulate leukocyte activity and cytokine production, confirming the thermal–immune link in vivo (Xue et al., 2016.)
Breathing: use nasal inhalation to warm and humidify upper airways.
Complementary local heating: 10 minutes of moist steam (≈ 42–44 °C) after the main session to raise nasal/throat temperature.
Cooling phase: gradual return to baseline; avoid immediate cold exposure.
11.6. Integration into Daily Life
The regimen can be implemented in offices, homes, or travel settings using the portable IsoTone device. Even brief micro-sessions (5–10 min) between longer bouts maintain circulatory vigor and immune alertness. Regular use throughout colder months functions as thermal conditioning, reducing the likelihood that routine exposures progress into illness.
11.7. Summary of Therapeutic Logic
The regimen described above translates the theoretical framework of the Body Heating Theory into an actionable sequence of physiological events. Each stage—from temperature elevation to recovery—serves a specific immune purpose and follows a logical progression rooted in thermodynamics and virology. The key relationships summarized below capture the essence of this method: how temperature, timing, repetition, and self-regulation converge to create a safe, repeatable, and scientifically grounded strategy for viral prevention and recovery.
Meta-analytic findings in older adults indicate that both acute and chronic exercise elevate Th1/Th2 balance and enhance immune responsiveness without harmful stress effects (Barron et al., 2020.)
Temperature elevation: moves the body out of the viral comfort zone (≈ 33–36 °C) into the immune-dominant zone (≈ 38–39 °C).
Timing: each pulse adds an immune “grace period” of several hours, equivalent to slowing one replication cycle.
Repetition: two or more pulses per day sustain antiviral pressure without exhaustion.
Safety: self-regulated intensity prevents overheating; hydration and short rest restore balance.
Closing Perspective
The arguments presented here are not merely theoretical — they are a call to reconsider how we interpret illness, recovery, and prevention in everyday life. Humanity’s capacity for self-heating, once a tool for survival, remains an untapped resource for maintaining health in a world where physical effort has largely been replaced by comfort. If we learn to re-engage this capacity deliberately—through brief, purposeful movement rather than dependence on symptomatic relief—we may rediscover a fundamental principle of biological balance.
The science of body heating is, at its core, the science of cooperation between motion, energy, and life itself. By restoring that cooperation—and learning to act in the first moments after exposure—we can reclaim a measure of the resilience that evolution designed, and modern living has allowed to fade.
References
- Evans, S. S., Repasky, E. A., & Fisher, D. T. (2015). Fever and the thermal regulation of immunity: The immune system feels the heat. Nature Reviews Immunology, 15(6), 335-349. [CrossRef]
- Lee, S., & Amherst, E. (2021). Deciphering the relationship between temperature and immunity. Discovery Immunology, 3(1), kyae001.
- Rand, D. & White, M. (2020). Let fever do its job: The meaning of fever in the pandemic era. Evolution, Medicine, and Public Health, 9(1), 26.
- Foxman, E. F., et al. (2015). Temperature-dependent innate defense against the common cold virus limits viral replication at warm temperatures. Proceedings of the National Academy of Sciences (PNAS), 112(3), 827-832. [CrossRef]
- Campbell, J. P., & Turner, J. E. (2018). Debunking the myth of exercise-induced immune suppression. Frontiers in Immunology, 9, 648. [CrossRef]
- Nieman, D. C., & Wentz, L. M. (2019). The compelling link between physical activity and the body’s defense system. Journal of Sport and Health Science, 8(3), 201-217. [CrossRef]
- Olive, M. T., & Bastien, K. M. (2023). Regulation of Immune Function by the Lymphatic System. Frontiers in Immunology, 10.3389/fimmu.2019.00470. [CrossRef]
- Xue, Y., et al. (2016). Effect of exercise, heat stress, and hydration on immune cell number and function during exercise. International Journal of Sports Medicine, 27(9), 705-712.
- Hoeh, R., et al. (2018). Transport and immune functions of the lymphatic system. Annual Review of Physiology, 82, 1-25.
- Barron, M., et al. (2020). Effect of acute and chronic exercise on immune markers of Th1/Th2 cells in older adults: A systematic review. Frontiers in Physiology, (2025 forthcoming). [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).