Submitted:
11 November 2025
Posted:
13 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Faecal Microbiota Transplantation as a Modern Therapeutic Tool
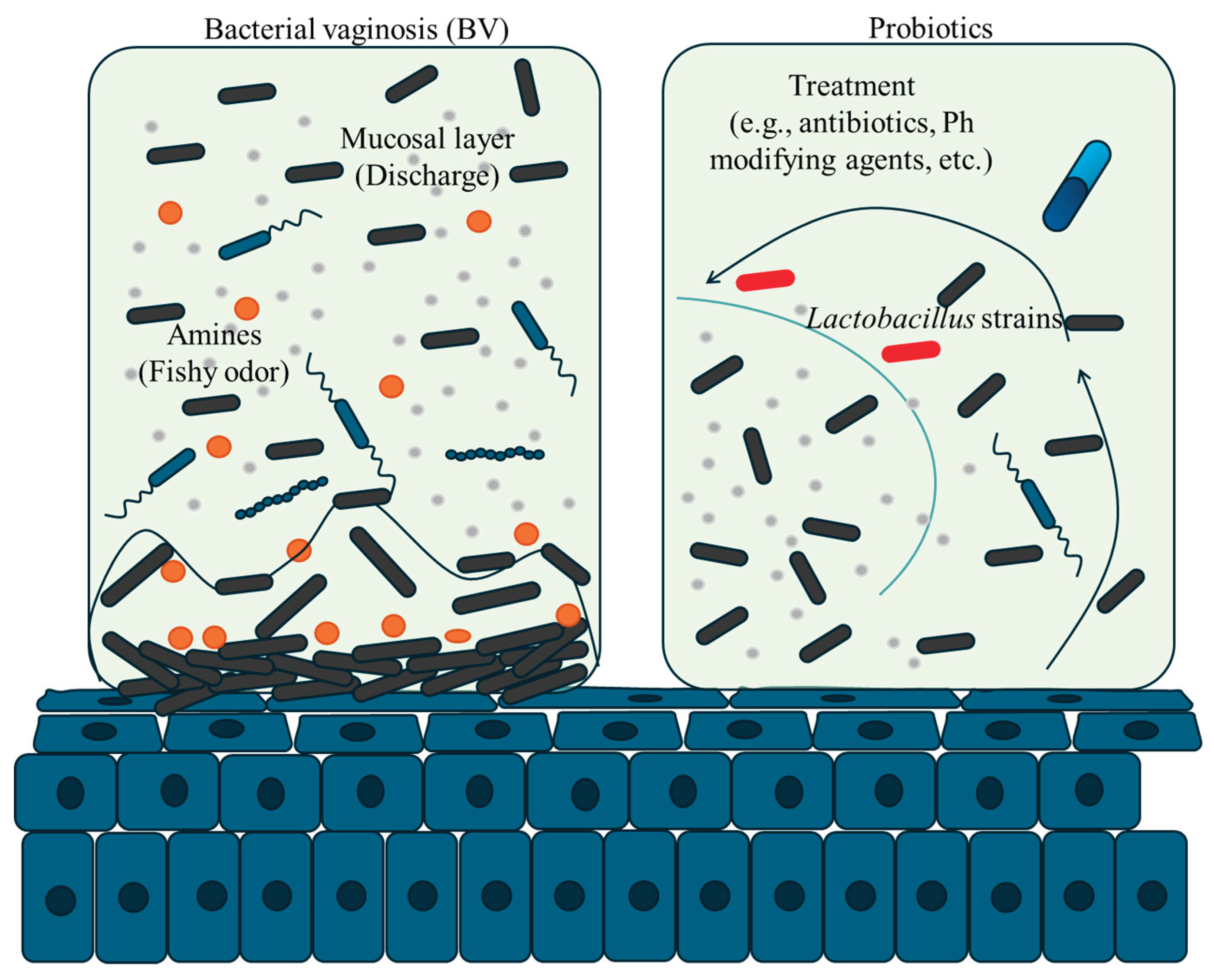
3. Bacterial Vaginosis
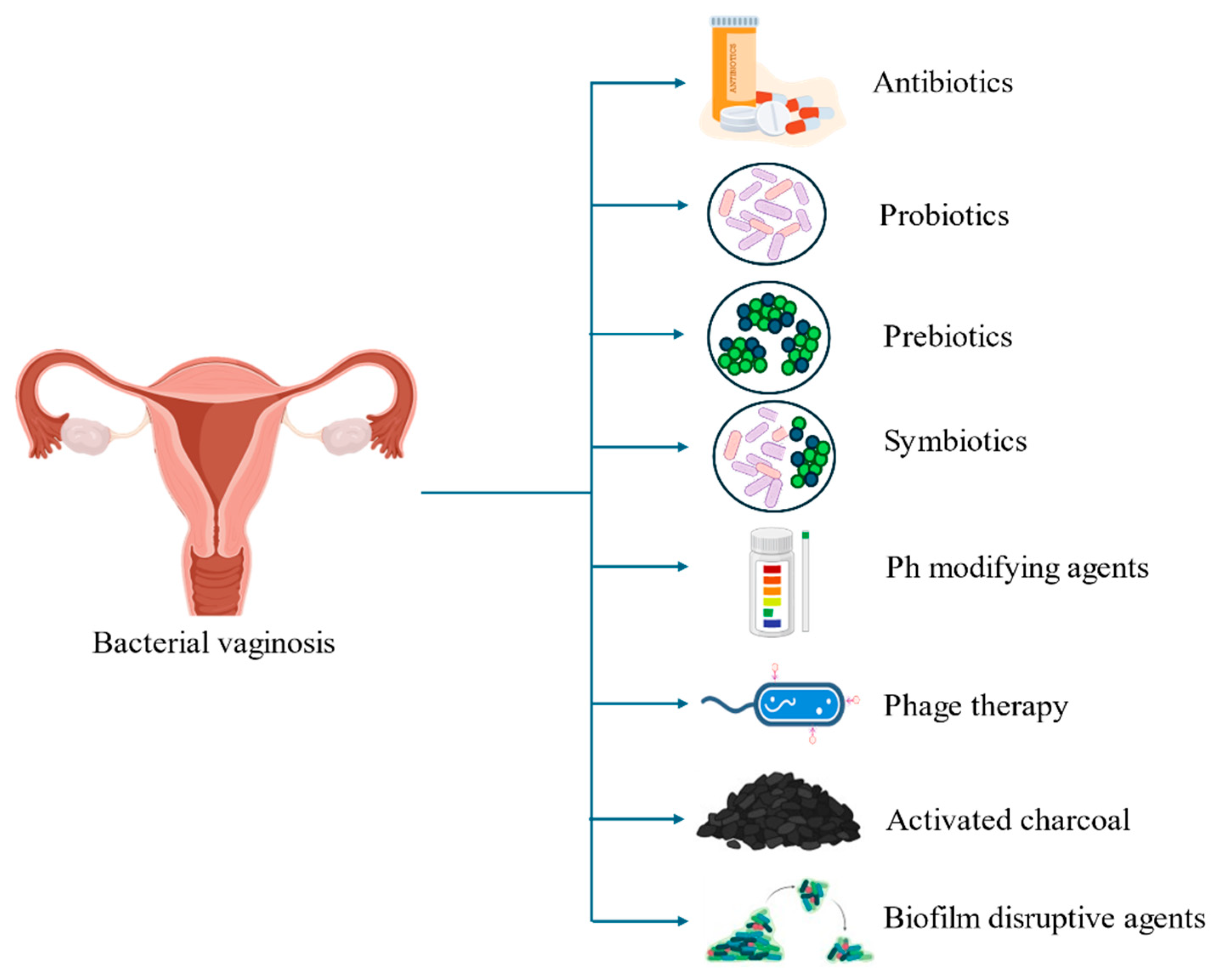
4. Current Treatment Regimens for BV
4.1. Antibiotics
4.2. Probiotics
4.3. Prebiotics
4.4. Symbiotics
4.5. Phage Therapy
4.6. Activated Charcoal
4.7. Biofilm Disruptive Agents
5. A promising Approach Inspired by FMT
6. VMT: As Emerging Concept for BV
6.1. Preconditions Required for VMT
6.1.1. Diagnosis of BV
6.1.2. Inclusion Criteria for Recipients
6.1.3. Exclusion Criteria for Recipients
6.1.4. Inclusion Criteria for Donors
6.1.5. Exclusion Criteria for Donors
6.2. VMT Procedure
6.3. Post-VMT Follow-Up
6.4. High Throughput 16S rRNA Gene Amplicon Sequencing
6.5. Microbial Bioinformatics Analysis
7. Readdressing Challenges in VMT
8. Conclusions
Consent for Publication
Conflicts of Interest
References
- Antonio, M. A. D., Meyn, L. A., Murray, P. J., Busse, B., Hillier, S. L., 2009. Vaginal colonization by probiotic Lactobacillus crispatus CTV-05 is decreased by sexual activity and endogenous. Lactobacilli. J. Infect. Dis. 199, 1506–1513.
- Aldunate, M.; Srbinovski, D.; Hearps, A.C.; Latham, C.F.; Ramsland, P.A.; Gugasyan, R.; Cone, R.A.; Tachedjian, G. Antimicrobial and immune modulatory effects of lactic acid and short chain fatty acids produced by vaginal microbiota associated with eubiosis and bacterial vaginosis. Front. Physiol. 2015, 6, 164. [CrossRef]
- Andriessen, E.M.; Wilson, A.M.; Mawambo, G.; Dejda, A.; Miloudi, K.; Sennlaub, F.; Sapieha, P. Gut microbiota influences pathological angiogenesis in obesity-driven choroidal neovascularization. EMBO Mol. Med. 2016, 8, 1366–1379. [CrossRef]
- Amabebe, E.; Anumba, D.O.C. The Vaginal Microenvironment: The Physiologic Role of Lactobacilli. Front. Med. 2018, 5, 181. [CrossRef]
- Anton, L.; Sierra, L.-J.; DeVine, A.; Barila, G.; Heiser, L.; Brown, A.G.; Elovitz, M.A. Common Cervicovaginal Microbial Supernatants Alter Cervical Epithelial Function: Mechanisms by Which Lactobacillus crispatus Contributes to Cervical Health. Front. Microbiol. 2018, 9, 2181. [CrossRef]
- Anton, L.; Ferguson, B.; Friedman, E.S.; Gerson, K.D.; Brown, A.G.; Elovitz, M.A. Gardnerella vaginalis alters cervicovaginal epithelial cell function through microbe-specific immune responses. Microbiome 2022, 10, 1–20. [CrossRef]
- Arroyo-Moreno S., Cummings M., Corcoran D.B., Coffey A., McCarthy R.R.,2022. Identification and characterization of novel endolysins targeting Gardnerella vaginalis biofilms to treat bacterial vaginosis. NPJ Biofilms Microbiomes 19, 8(1), 1-12.
- Boris, S.; Barbés, C. Role played by lactobacilli in controlling the population of vaginal pathogens. Microbes Infect. 2000, 2, 543–546. [CrossRef]
- Boskey, E.; Cone, R.; Whaley, K.; Moench, T. Origins of vaginal acidity: high d/l lactate ratio is consistent with bacteria being the primary source. Hum. Reprod. 2001, 16, 1809–1813. [CrossRef]
- Bradshaw, C.S.; Morton, A.N.; Hocking, J.; Garland, S.M.; Morris, M.B.; Moss, L.M.; Horvath, L.B.; Kuzevska, I.; Fairley, C.K. High Recurrence Rates of Bacterial Vaginosis over the Course of 12 Months after Oral Metronidazole Therapy and Factors Associated with Recurrence. J. Infect. Dis. 2006, 193, 1478–1486. [CrossRef]
- Bragulla, H.H.; Homberger, D.G. Structure and functions of keratin proteins in simple, stratified, keratinized and cornified epithelia. Am. J. Anat. 2009, 214, 516–559. [CrossRef]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and function of the epidermis related to barrier properties. Clin. Dermatol. 2012, 30, 257–262. [CrossRef]
- Bourdin, G.; Navarro, A.; Sarker, S.A.; Pittet, A.; Qadri, F.; Sultana, S.; Cravioto, A.; Talukder, K.A.; Reuteler, G.; Brüssow, H. Coverage of diarrhoea-associated E scherichia coli isolates from different origins with two types of phage cocktails. Microb. Biotechnol. 2014, 7, 165–176. [CrossRef]
- Babu, G.; Singaravelu, B.G.; Srikumar, R.; Reddy, S.V.; Kokan, A. Comparative Study on the Vaginal Flora and Incidence of Asymptomatic Vaginosis among Healthy Women and in Women with Infertility Problems of Reproductive Age. J. Clin. Diagn. Res. 2017, 11, DC18–DC22. [CrossRef]
- Baunwall, S.M.D.; Lee, M.M.; Eriksen, M.K.; Mullish, B.H.; Marchesi, J.R.; Dahlerup, J.F.; Hvas, C.L. Faecal microbiota transplantation for recurrent Clostridioides difficile infection: An updated systematic review and meta-analysis. eClinicalMedicine 2020, 29-30, 100642. [CrossRef]
- Bhujel, R.; Mishra, S.K.; Yadav, S.K.; Bista, K.D.; Parajuli, K. Comparative study of Amsel’s criteria and Nugent scoring for diagnosis of bacterial vaginosis in a tertiary care hospital, Nepal. BMC Infect. Dis. 2021, 21, 1–6. [CrossRef]
- Biazzo, M.; Deidda, G. Fecal Microbiota Transplantation as New Therapeutic Avenue for Human Diseases. J. Clin. Med. 2022, 11, 4119. [CrossRef]
- Breedveld, A.C.; Schuster, H.J.; van Houdt, R.; Painter, R.C.; Mebius, R.E.; van der Veer, C.; Bruisten, S.M.; Savelkoul, P.H.M.; van Egmond, M. Enhanced IgA coating of bacteria in women with Lactobacillus crispatus-dominated vaginal microbiota. Microbiome 2022, 10, 1–11. [CrossRef]
- Coste, I.; Judlin, P.; Lepargneur, J.-P.; Bou-Antoun, S. Safety and Efficacy of an Intravaginal Prebiotic Gel in the Prevention of Recurrent Bacterial Vaginosis: A Randomized Double-Blind Study. Obstet. Gynecol. Int. 2012, 2012, 1–7. [CrossRef]
- Chawla, R.; Bhalla, P.; Chadha, S.; Grover, S.; Garg, S. Comparison of Hay’s Criteria with Nugent’s Scoring System for Diagnosis of Bacterial Vaginosis. BioMed Res. Int. 2013, 2013, 1–5. [CrossRef]
- Coleman, J.S.; A Gaydos, C. Molecular Diagnosis of Bacterial Vaginosis: an Update. J. Clin. Microbiol. 2018, 56. [CrossRef]
- Collins, S.L.; McMillan, A.; Seney, S.; van der Veer, C.; Kort, R.; Sumarah, M.W.; Reid, G. Promising Prebiotic Candidate Established by Evaluation of Lactitol, Lactulose, Raffinose, and Oligofructose for Maintenance of a Lactobacillus-Dominated Vaginal Microbiota. Appl. Environ. Microbiol. 2018, 84, e02200-17. [CrossRef]
- Chee, W.J.Y.; Chew, S.Y.; Than, L.T.L. Vaginal microbiota and the potential of Lactobacillus derivatives in maintaining vaginal health. Microb. Cell Factories 2020, 19, 203. [CrossRef]
- Cheng, L.; Norenhag, J.; Hu, Y.O.O.; Brusselaers, N.; Fransson, E.; Ährlund-Richter, A.; Guðnadóttir, U.; Angelidou, P.; Zha, Y.; Hamsten, M.; et al. Vaginal microbiota and human papillomavirus infection among young Swedish women. npj Biofilms Microbiomes 2020, 6, 1–10. [CrossRef]
- Coudray, M.S.; Madhivanan, P. Bacterial vaginosis—A brief synopsis of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 245, 143–148. [CrossRef]
- Chieng, W.K.; Jalal, M.I.A.; Bedi, J.S.; Zainuddin, A.A.; Mokhtar, M.H.; Abu, M.A.; Chew, K.T.; Azurah, A.G.N. Probiotics, a promising therapy to reduce the recurrence of bacterial vaginosis in women? a systematic review and meta-analysis of randomized controlled trials. Front. Nutr. 2022, 9, 938838. [CrossRef]
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An ecological and evolutionary perspective on human–microbe mutualism and disease. Nature 2007, 449, 811–818. [CrossRef]
- Dareng, E.O.; Ma, B.; Famooto, A.O.; Akarolo-Anthony, S.N.; Offiong, R.A.; Olaniyan, O.; Dakum, P.S.; Wheeler, C.M.; Fadrosh, D.; Yang, H.; et al. Prevalent high-risk HPV infection and vaginal microbiota in Nigerian women. Epidemiology Infect. 2015, 144, 123–137. [CrossRef]
- Donlan, R.M. Preventing biofilms of clinically relevant organisms using bacteriophage. Trends Microbiol. 2009, 17, 66–72. [CrossRef]
- Duar, R.M.; Lin, X.B.; Zheng, J.; Martino, M.E.; Grenier, T.; Pérez-Muñoz, M.E.; Leulier, F.; Gänzle, M.; Walter, J. Lifestyles in transition: evolution and natural history of the genus Lactobacillus. FEMS Microbiol. Rev. 2017, 41, S27–S48. [CrossRef]
- Das Purkayastha, S.; Bhattacharya, M.K.; Prasad, H.K.; Upadhyaya, H.; Das Lala, S.; Pal, K.; Das, M.; Sharma, G.D.; Bhattacharjee, M.J. Contrasting diversity of vaginal lactobacilli among the females of Northeast India. BMC Microbiol. 2019, 19, 1–10. [CrossRef]
- DeLong, K.; Zulfiqar, F.; Hoffmann, D.E.; Tarzian, A.J.; Ensign, L.M. Vaginal Microbiota Transplantation: The Next Frontier. J. Law, Med. Ethic- 2019, 47, 555–567. [CrossRef]
- Delgado-Diaz, D.J.; Jesaveluk, B.; Hayward, J.A.; Tyssen, D.; Alisoltani, A.; Potgieter, M.; Bell, L.; Ross, E.; Iranzadeh, A.; Allali, I.; et al. Lactic acid from vaginal microbiota enhances cervicovaginal epithelial barrier integrity by promoting tight junction protein expression. Microbiome 2022, 10, 1–16. [CrossRef]
- Dequalinium chloride. Health Canada approved product monograph. Revised Sept, 2021. Health Canada. https://pdf.hres.ca/dpd_pm/00063156.PDF (Accessed 25.10. 2022.).
- Eckhart, L.; Lippens, S.; Tschachler, E.; Declercq, W. Cell death by cornification. Biochim. et Biophys. Acta (BBA) - Mol. Cell Res. 2013, 1833, 3471–3480. [CrossRef]
- Evans, J.M., Morris, L.S., Marchesi, J.R., 2013. The gut microbiome: the role of a virtual organ in the endocrinology of the host. J Endocrinol. 218 (3), R37-R47.
- Eastment, M.C.; McClelland, R.S. Vaginal microbiota and susceptibility to HIV. AIDS 2018, 32, 687–698. [CrossRef]
- Ferris, D.G.; Litaker, M.S.; Woodward, L.; Mathis, D.; Hendrich, J. Treatment of bacterial vaginosis: a comparison of oral metronidazole, metronidazole vaginal gel, and clindamycin vaginal cream.. 1995, 41, 443–9.
- Farage, M.A.; Miller, K.W.; Sobel, J.D. Dynamics of the Vaginal Ecosystem—Hormonal Influences. Infect. Dis. Res. Treat. 2010, 3. [CrossRef]
- Flint, H.J.; Scott, K.P.; Louis, P.; Duncan, S.H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 2022, 9, 577–589. [CrossRef]
- A Greenberg, S.; Youngster, I.; A Cohen, N.; Livovsky, D.M.; Strahilevitz, J.; Israeli, E.; Melzer, E.; Paz, K.; Fliss-Isakov, N.; Maharshak, N. Five years of fecal microbiota transplantation - an update of the Israeli experience. World J. Gastroenterol. 2018, 24, 5403–5414. [CrossRef]
- Greenbaum, S.; Greenbaum, G.; Moran-Gilad, J.; Weintraub, A.Y. Ecological dynamics of the vaginal microbiome in relation to health and disease. Am. J. Obstet. Gynecol. 2019, 220, 324–335. [CrossRef]
- Gupta, S., Allen-Vercoe, E., Petrof, E.O., 2016. Fecal microbiota transplantation: in perspective. Therap Adv Gastroenterol. 9(2), 229-39.
- Happel, A.-U.; Kullin, B.; Gamieldien, H.; Wentzel, N.; Zauchenberger, C.Z.; Jaspan, H.B.; Dabee, S.; Barnabas, S.L.; Jaumdally, S.Z.; Dietrich, J.; et al. Exploring potential of vaginal Lactobacillus isolates from South African women for enhancing treatment for bacterial vaginosis. PLOS Pathog. 2020, 16, e1008559. [CrossRef]
- Hay, P. Life in the littoral zone: lactobacilli losing the plot. Sex. Transm. Infect. 2005, 81, 100–102. [CrossRef]
- Heinemann, C.; Reid, G. Vaginal microbial diversity among postmenopausal women with and without hormone replacement therapy. Can. J. Microbiol. 2005, 51, 777–781. [CrossRef]
- Hans, V., Rita, V., 2009. Bacterial vaginosis: an update on diagnosis and treatment. Expert Rev. Anti Infect. Ther. 7, 1109–1124.
- Hao, W.; Peng, Y.; Xu, W.; Liao, X.; Li, M.; Luo, S. Diversity of vaginal ecosystem on women in Southern China. 2011 International Conference on Information Science and Technology (ICIST). pp. 1254–1261.
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [CrossRef]
- Hanssen, N.M.; de Vos, W.M.; Nieuwdorp, M. Fecal microbiota transplantation in human metabolic diseases: From a murky past to a bright future?. Cell Metab. 2021, 33, 1098–1110. [CrossRef]
- Hou, K., Wu, Z.X., Chen, X.Y. et al., 2022. Microbiota in health and diseases. Sig Transduct Target Ther. 7, 1-28.
- Macro, O.R.C., International Institute for Population Sciences (IIPS) 2000. National Family Health Survey (NFHS-2), Mumbai, India. 1998, 99, 307-360.
- Jiang, Z.D.; Ajami, N.J.; Petrosino, J.F.; Jun, G.; Hanis, C.L.; Shah, M.; Hochman, L.; Ankoma-Sey, V.; DuPont, A.W.; Wong, M.C.; et al. Randomised clinical trial: faecal microbiota transplantation for recurrent Clostridum difficile infection – fresh, or frozen, or lyophilised microbiota from a small pool of healthy donors delivered by colonoscopy. Aliment. Pharmacol. Ther. 2017, 45, 899–908. [CrossRef]
- Jacob, L., John, M., Kalder, M. & Kostev, K., 2018. Prevalence of vulvovaginal candidiasis in gynecological practices in Germany: a retrospective study of 954,186 patients. Curr. Med. Mycol. 4, 6–11.
- Jain, J.P.; Bristow, C.C.; Pines, H.A.; Harvey-Vera, A.; Rangel, G.; Staines, H.; Patterson, T.L.; Strathdee, S.A. Factors in the HIV risk environment associated with bacterial vaginosis among HIV-negative female sex workers who inject drugs in the Mexico-United States border region. BMC Public Heal. 2018, 18, 1032. [CrossRef]
- Javed, A.; Parvaiz, F.; Manzoor, S. Bacterial vaginosis: An insight into the prevalence, alternative treatments regimen and it's associated resistance patterns. Microb. Pathog. 2019, 127, 21–30. [CrossRef]
- Jakobsen, R.R.; Haahr, T.; Humaidan, P.; Jensen, J.S.; Kot, W.P.; Castro-Mejia, J.L.; Deng, L.; Leser, T.D.; Nielsen, D.S. Characterization of the Vaginal DNA Virome in Health and Dysbiosis. Viruses 2020, 12, 1143. [CrossRef]
- Junca, H.; Pieper, D.H.; Medina, E. The emerging potential of microbiome transplantation on human health interventions. Comput. Struct. Biotechnol. J. 2022, 20, 615–627. [CrossRef]
- Koumans, E.H.; Markowitz, L.E.; Hogan, V.; the CDC BV Working Group Indications for Therapy and Treatment Recommendations for Bacterial Vaginosis in Nonpregnant and Pregnant Women: A Synthesis of Data. Clin. Infect. Dis. 2002, 35, S152–S172. [CrossRef]
- Kelly, C.R.; Kahn, S.; Kashyap, P.; Laine, L.; Rubin, D.; Atreja, A.; Moore, T.; Wu, G. Update on Fecal Microbiota Transplantation 2015: Indications, Methodologies, Mechanisms, and Outlook. Gastroenterology 2015, 149, 223–237. Erratum in 2015, 149, 223–237. [CrossRef]
- Kaushic, C., Nguyen, P.V., 2016. Immunology of the female reproductive mucosa. Encyclopedia Immunobiology. 63–72.
- Kim, J.-M.; Park, Y.J. Probiotics in the Prevention and Treatment of Postmenopausal Vaginal Infections: Review Article. J. Menopausal Med. 2017, 23, 139–145. [CrossRef]
- Kolaček, S., et al., 2017. Commercial probiotic products: a call for improved quality control. A position paper by the ESPGHAN working group for probiotics and prebiotics. J. Pediatr. Gastroenterol. Nutr. 65 (1), 117–124.
- Khangwal, I.; Shukla, P. Prospecting prebiotics, innovative evaluation methods, and their health applications: a review. 3 Biotech 2019, 9, 1–7. [CrossRef]
- Kalia, N.; Singh, J.; Kaur, M. Microbiota in vaginal health and pathogenesis of recurrent vulvovaginal infections: a critical review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 1–19. [CrossRef]
- Larsson, P.-G.; Brandsborg, E.; Forsum, U.; Pendharkar, S.; Andersen, K.K.; Nasic, S.; Hammarström, L.; Marcotte, H. Extended antimicrobial treatment of bacterial vaginosis combined with human lactobacilli to find the best treatment and minimize the risk of relapses. BMC Infect. Dis. 2011, 11, 223–223. [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [CrossRef]
- Lev-Sagie, A.; Goldman-Wohl, D.; Cohen, Y.; Dori-Bachash, M.; Leshem, A.; Mor, U.; Strahilevitz, J.; Moses, A.E.; Shapiro, H.; Yagel, S.; et al. Vaginal microbiome transplantation in women with intractable bacterial vaginosis. Nat. Med. 2019, 25, 1500–1504. [CrossRef]
- Landlinger, C.; Tisakova, L.; Oberbauer, V.; Schwebs, T.; Muhammad, A.; Latka, A.; Van Simaey, L.; Vaneechoutte, M.; Guschin, A.; Resch, G.; et al. Engineered Phage Endolysin Eliminates Gardnerella Biofilm without Damaging Beneficial Bacteria in Bacterial Vaginosis Ex Vivo. Pathogens 2021, 10, 54. [CrossRef]
- Liu, H.-F.; Yi, N. A systematic review and meta-analysis on the efficacy of probiotics for bacterial vaginosis. 2022, 26, 90–98. [CrossRef]
- Mikamo, H., Sato., Hayasaki, Y. et al., 2000. Vaginal microflora in healthy women with Gardnerella vaginalis. J Infect Chemother. 6(3), 173–177.
- Martinez, R.C.R.; Franceschini, S.A.; Patta, M.C.; Quintana, S.M.; Nunes, A.C.; Moreira, J.L.S.; Anukam, K.C.; Reid, G.; De Martinis, E.C.P. Analysis of Vaginal Lactobacilli from Healthy and Infected Brazilian Women. Appl. Environ. Microbiol. 2008, 74, 4539–4542. [CrossRef]
- Menard, J.-P. Antibacterial treatment of bacterial vaginosis: current and emerging therapies. Int. J. Women's Heal. 2011, 3, 295–305. [CrossRef]
- Ma, B.; Forney, L.J.; Ravel, J. Vaginal Microbiome: Rethinking Health and Disease. Annu. Rev. Microbiol. 2012, 66, 371–389. [CrossRef]
- Maynard, C.L.; Elson, C.O.; Hatton, R.D.; Weaver, C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nature 2012, 489, 231–241. [CrossRef]
- Menon, G.K.; Cleary, G.W.; Lane, M.E. The structure and function of the stratum corneum. Int. J. Pharm. 2012, 435, 3–9. [CrossRef]
- Mastromarino, P.; Hemalatha, R.; Barbonetti, A.; Cinque, B.; Cifone, M.; Tammaro, F.; Francavilla, F. Biological control of vaginosis to improve reproductive health. 2014, 140, S91–S97.
- Mendling, W., 2016. Vaginal Microbiota. Advances in experimental medicine and biology. 902, 83–93.
- Miller, E.A.; Beasley, D.E.; Dunn, R.R.; Archie, E.A. Lactobacilli Dominance and Vaginal pH: Why Is the Human Vaginal Microbiome Unique?. Front. Microbiol. 2016, 7, 1936. [CrossRef]
- Myles, I. A., Earland, N. J., Anderson, E. D., Moore, I. N. et al., 2019. First-in-human topical microbiome transplantation with Roseomonas mucosa for atopic dermatitis. JCI insight, 3(9), e120608. [CrossRef]
- Ma D, Chen Y, Chen T. Vaginal microbiota transplantation for the treatment of bacterial vaginosis: a conceptual analysis. FEMS Microbiol Lett. 366(4), 1-13. [CrossRef]
- Marrazzo, J.M.; Dombrowski, J.C.; Wierzbicki, M.R.; Perlowski, C.; Pontius, A.; Dithmer, D.; Schwebke, J. Safety and Efficacy of a Novel Vaginal Anti-infective, TOL-463, in the Treatment of Bacterial Vaginosis and Vulvovaginal Candidiasis: A Randomized, Single-blind, Phase 2, Controlled Trial. Clin. Infect. Dis. 2019, 68, 803–809. [CrossRef]
- Muzny, C.A.; Łaniewski, P.; Schwebke, J.R.; Herbst-Kralovetz, M.M. Host–vaginal microbiota interactions in the pathogenesis of bacterial vaginosis. Curr. Opin. Infect. Dis. 2020, 33, 59–65. [CrossRef]
- Muzny, C.A.M.; Kardas, P. A Narrative Review of Current Challenges in the Diagnosis and Management of Bacterial Vaginosis. Sex. Transm. Dis. 2020, 47, 441–446. [CrossRef]
- Nurainiwati, S.A.; Ma'ROef, M.; Pravitasari, D.N.; Putra, P.Y.P. Effectivity and efficacy probiotics for Bacterial Vaginosis treatments: Meta-analysis. Infect. Dis. Model. 2022, 7, 597–604. [CrossRef]
- Ochiel, D.O.; Fahey, J.V.; Ghosh, M.; Haddad, S.N.; Wira, C.R. Innate Immunity in the Female Reproductive Tract: Role of Sex Hormones in Regulating Uterine Epithelial Cell Protection Against Pathogens. Curr. Women s Heal. Rev. 2008, 4, 102–117. [CrossRef]
- Oduyebo, O.; I Anorlu, R.; Ogunsola, F.T. The effects of antimicrobial therapy on bacterial vaginosis in non-pregnant women. Cochrane Database Syst. Rev. 2009, CD006055. [CrossRef]
- Oyinlola, O. O., Rose, I. A., Folasade, T. O., 2009. The effects of antimicrobial therapy on bacterial vaginosis in non-pregnant women. Cochrane Database Syst. Rev. 8, CD006055.
- E O'Hanlon, D.; Moench, T.R.; A Cone, R. In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect. Dis. 2011, 11, 200–200. [CrossRef]
- O’hanlon, D.E.; Moench, T.R.; Cone, R.A. Vaginal pH and Microbicidal Lactic Acid When Lactobacilli Dominate the Microbiota. PLOS ONE 2011, 8, e80074. [CrossRef]
- Onderdonk, A.B.; Delaney, M.L.; Fichorova, R.N. The Human Microbiome during Bacterial Vaginosis. Clin. Microbiol. Rev. 2016, 29, 223–238. [CrossRef]
- Otsuki, K.; Imai, N. Effects of lactoferrin in 6 patients with refractory bacterial vaginosis. Biochem. Cell Biol. 2017, 95, 31–33. [CrossRef]
- Oliveira, H.; São-José, C.; Azeredo, J. Phage-Derived Peptidoglycan Degrading Enzymes: Challenges and Future Prospects for In Vivo Therapy. Viruses 2018, 10, 292. [CrossRef]
- Petrova, M.I., Marianne, V.D.B., Jan, B. et al., 2013. Vaginal microbiota and its role in HIV transmission and infection. Fems Microbiol Rev, 37:762–92.
- Pilla, R.; Gaschen, F.P.; Barr, J.W.; Olson, E.; Honneffer, J.; Guard, B.C.; Blake, A.B.; Villanueva, D.; Khattab, M.R.; AlShawaqfeh, M.K.; et al. Effects of metronidazole on the fecal microbiome and metabolome in healthy dogs. J. Veter- Intern. Med. 2020, 34, 1853–1866. [CrossRef]
- Plummer, E.L.; Bradshaw, C.S.; Doyle, M.; Fairley, C.K.; Murray, G.L.; Bateson, D.; Masson, L.; Slifirski, J.; Tachedjian, G.; Vodstrcil, L.A. Lactic acid-containing products for bacterial vaginosis and their impact on the vaginal microbiota: A systematic review. PLOS ONE 2021, 16, e0246953. [CrossRef]
- Bascuñana, R.P.; Veses, V.; Sheth, C. Effectiveness of fecal microbiota transplant for the treatment of Clostridioides difficile diarrhea: a systematic review and meta-analysis. Lett. Appl. Microbiol. 2021, 73, 149–158. [CrossRef]
- Quraishi, M.N.; Widlak, M.; Bhala, N.; Moore, D.; Price, M.; Sharma, N.; Iqbal, T.H. Systematic review with meta-analysis: the efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection. Aliment. Pharmacol. Ther. 2017, 46, 479–493. [CrossRef]
- Reid, G.; Charbonneau, D.; Erb, J.; Kochanowski, B.; Beuerman, D.; Poehner, R.; Bruce, A.W. Oral use ofLactobacillus rhamnosusGR-1 andL. fermentumRC-14 significantly alters vaginal flora: randomized, placebo-controlled trial in 64 healthy women. FEMS Immunol. Med Microbiol. 2003, 35, 131–134. [CrossRef]
- Rousseau, V.; Lepargneur, J.; Roques, C.; Remaud-Simeon, M.; Paul, F. Prebiotic effects of oligosaccharides on selected vaginal lactobacilli and pathogenic microorganisms. Anaerobe 2005, 11, 145–153. [CrossRef]
- Rönnqvist, P.D.J.; Forsgren-Brusk, U.B.; Grahn-Håkansson, E.E. Lactobacilli in the female genital tract in relation to other genital microbes and vaginal pH. Acta Obstet. et Gynecol. Scand. 2006, 85, 726–735. [CrossRef]
- Reichman, O.; Akins, R.; Sobel, J.D. Boric Acid Addition to Suppressive Antimicrobial Therapy for Recurrent Bacterial Vaginosis. Sex. Transm. Dis. 2009, 36, 732–734. [CrossRef]
- Robinson, C.J.; Bohannan, B.J.M.; Young, V.B. From Structure to Function: the Ecology of Host-Associated Microbial Communities. Microbiol. Mol. Biol. Rev. 2010, 74, 453–476. [CrossRef]
- Raybardhan, S.; Ng, W.; Tomassi, J.; Katz, K. Self-stool banking as a source for fecal microbiota transplantation: A pilot study. Off. J. Assoc. Med Microbiol. Infect. Dis. Can. 2017, 2, 9–14. [CrossRef]
- Redelinghuys, M.J.; Ehlers, M.M.; Dreyer, A.W.; Kock, M.M. Normal flora and bacterial vaginosis in pregnancy: an overview. Crit. Rev. Microbiol. 2016, 42, 1–12. [CrossRef]
- Russo, R.; Karadja, E.; De Seta, F. Evidence-based mixture containing Lactobacillus strains and lactoferrin to prevent recurrent bacterial vaginosis: a double blind, placebo controlled, randomised clinical trial. Benef. Microbes 2019, 10, 19–26. [CrossRef]
- Sobel, J.D.; Schmitt, C.; Meriwether, C. Long-Term Follow-Up of Patients with Bacterial Vaginosis Treated with Oral Metronidazole and Topical Clindamycin. J. Infect. Dis. 1993, 167, 783–784. [CrossRef]
- Stappenbeck, T.S.; Hooper, L.V.; Gordon, J.I. Developmental regulation of intestinal angiogenesis by indigenous microbes via Paneth cells. Proc. Natl. Acad. Sci. 2002, 99, 15451–15455. [CrossRef]
- Stoyancheva, G.; Marzotto, M.; Dellaglio, F.; Torriani, S. Bacteriocin production and gene sequencing analysis from vaginal Lactobacillus strains. Arch. Microbiol. 2014, 196, 645–653. [CrossRef]
- Srinivasan, S.; Morgan, M.T.; Fiedler, T.L.; Djukovic, D.; Hoffman, N.G.; Raftery, D.; Marrazzo, J.M.; Fredricks, D.N. Metabolic Signatures of Bacterial Vaginosis. mBio 2015, 6, e00204-15. [CrossRef]
- Smith, S.B.; Ravel, J. The vaginal microbiota, host defence and reproductive physiology. J. Physiol. 2016, 595, 451–463. [CrossRef]
- Serrano, M.G.; Parikh, H.I.; Brooks, J.P.; Edwards, D.J.; Arodz, T.J.; Edupuganti, L.; Huang, B.; Girerd, P.H.; Bokhari, Y.A.; Bradley, S.P.; et al. Racioethnic diversity in the dynamics of the vaginal microbiome during pregnancy. Nat. Med. 2019, 25, 1001–1011. [CrossRef]
- Shukla, A.; Sobel, J.D. Vulvovaginitis Caused by Candida Species Following Antibiotic Exposure. Curr. Infect. Dis. Rep. 2019, 21, 44. [CrossRef]
- Secnidasole oral granules. US Food and Drug Administration (FDA) approved product information. Revised September 2017. US National Library of Medicine. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/209363s015lbl.pdf (Accessed on June 17.06. 2021).
- Surapaneni, S., Akins, R., & Sobel, J. D., 2021. Recurrent Bacterial Vaginosis: An Unmet Therapeutic Challenge. Experience With a Combination Pharmacotherapy Long-Term Suppressive Regimen. Sexually transmitted diseases. 48(10), 761–765.
- Tibaldi, C.; Cappello, N.; Latino, M.; Masuelli, G.; Marini, S.; Benedetto, C. Vaginal and endocervical microorganisms in symptomatic and asymptomatic non-pregnant females: risk factors and rates of occurrence. Clin. Microbiol. Infect. 2009, 15, 670–679. [CrossRef]
- Tominaga, K.; Sato, S.; Hayashi, M. Activated charcoal as an effective treatment for bacterial vaginosis. Pers. Med. Universe 2012, 1, 54–57. [CrossRef]
- Tachedjian, G.; O’hAnlon, D.E.; Ravel, J. The implausible “in vivo” role of hydrogen peroxide as an antimicrobial factor produced by vaginal microbiota. Microbiome 2018, 6, 1–5. [CrossRef]
- Tinidazole tablet . US Food and Drug Administration (FDA) approved product information. Revised Dec, 2019. US National Library of Medicine. Available at: https://dailymed.nlm.nih.gov/dailymed/ (Accessed on 30.04.2021).
- Vallor, A.C.; Antonio, M.A.D.; Hawes, S.E.; Hillier, S.L. Factors Associated with Acquisition of, or Persistent Colonization by, Vaginal Lactobacilli: Role of Hydrogen Peroxide Production. J. Infect. Dis. 2001, 184, 1431–1436. [CrossRef]
- Vodstrcil, L.A.; Muzny, C.A.; Plummer, E.L.; Sobel, J.D.; Bradshaw, C.S. Bacterial vaginosis: drivers of recurrence and challenges and opportunities in partner treatment. BMC Med. 2021, 19, 1–12. [CrossRef]
- Vieira-Baptista, P.; De Seta, F.; Verstraelen, H.; Ventolini, G.M.; Lonnee-Hoffmann, R.; Lev-Sagie, A. The Vaginal Microbiome: V. Therapeutic Modalities of Vaginal Microbiome Engineering and Research Challenges. J. Low. Genit. Tract Dis. 2022, 26, 99–104. [CrossRef]
- Vitali, B.; Abruzzo, A.; Parolin, C.; Palomino, R.A.Ñ.; Dalena, F.; Bigucci, F.; Cerchiara, T.; Luppi, B. Association of Lactobacillus crispatus with fructo-oligosaccharides and ascorbic acid in hydroxypropyl methylcellulose vaginal insert. Carbohydr. Polym. 2016, 136, 1161–1169. [CrossRef]
- Vijay, A.; Valdes, A.M. RETRACTED ARTICLE: Role of the gut microbiome in chronic diseases: a narrative review. Eur. J. Clin. Nutr. 2021, 76, 489–501. [CrossRef]
- Wira, C.R.; Fahey, J.V.; Sentman, C.L.; Pioli, P.A.; Shen, L. Innate and adaptive immunity in female genital tract: cellular responses and interactions. Immunol. Rev. 2005, 206, 306–335. [CrossRef]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and function of the epidermis related to barrier properties. Clin. Dermatol. 2012, 30, 257–262. [CrossRef]
- Wang, Z.; He, Y.; Zheng, Y. Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 3859. [CrossRef]
- Wu, S.; Hugerth, L.W.; Schuppe-Koistinen, I.; Du, J. The right bug in the right place: opportunities for bacterial vaginosis treatment. npj Biofilms Microbiomes 2022, 8, 1–11. [CrossRef]
- Xiao, Y.; Angulo, M.T.; Lao, S.; Weiss, S.T.; Liu, Y.-Y. An ecological framework to understand the efficacy of fecal microbiota transplantation. Nat. Commun. 2020, 11, 1–17. [CrossRef]
- Yazdanbakhsh, M.; Kremsner, P.G.; van Ree, R. Allergy, Parasites, and the Hygiene Hypothesis. Science 2002, 296, 490–494. [CrossRef]
- Youngster, I.; Mahabamunuge, J.; Systrom, H.K.; Sauk, J.; Khalili, H.; Levin, J.; Kaplan, J.L.; Hohmann, E.L. Oral, frozen fecal microbiota transplant (FMT) capsules for recurrent Clostridium difficile infection. BMC Med. 2016, 14, 1–4. [CrossRef]
- Yadav, D.; Khanna, S. Safety of fecal microbiota transplantation for Clostridioides difficile infection focusing on pathobionts and SARS-CoV-2. Ther. Adv. Gastroenterol. 2021, 14. [CrossRef]
- Zoetendal, E.G.; Cheng, B.; Koike, S.; I Mackie, R. Molecular microbial ecology of the gastrointestinal tract: from phylogeny to function.. 2004, 5, 31–47.
- Zeng, Z.-M.; Liao, Q.-P.; Yao, C.; Geng, L.; Feng, L.-H.; Shi, H.-R.; Xin, X.-Y.; Li, P.; Wang, H.-L.; Pang, Y.-C.; et al. Directed shift of vaginal flora after topical application of sucrose gel in a phase III clinical trial: a novel treatment for bacterial vaginosis. Chinese medical journal 2010, 123, 2051–7.
- Terrier, M.-C.Z.; Frossard, J.-L.; Simonet, M.L. [Recurrent Clostridium difficile infections: the importance of the intestinal microbiota]. World journal of gastroenterology 2014, 9, 1898, 1900–4.
- Zimmermann, P.; Curtis, N. The effect of antibiotics on the composition of the intestinal microbiota - a systematic review. J. Infect. 2019, 79, 471–489. [CrossRef]
- Zmora, N.; Soffer, E.; Elinav, E. Transforming medicine with the microbiome. Sci. Transl. Med. 2019, 11, eaaw1815. [CrossRef]
- Zhang, Q.; Zhang, L.; Ross, P.; Zhao, J.; Zhang, H.; Chen, W. Comparative Genomics of Lactobacillus crispatus from the Gut and Vagina Reveals Genetic Diversity and Lifestyle Adaptation. Genes 2020, 11, 360. [CrossRef]
| S. No | Therapies | Dose | Duration | Adverse effects | References |
| 1. | Metronidazole | 400 mg orally for every 12 hours or 0.75% gel 5 g once daily | 5-7 days 5 days |
Metallic taste, nausea, and transient neutropenia | Muzny and Kardas, 2020 |
| 2. | Clindamycin | 300 mg orally every 12 hours or 100 mg intravaginally once daily |
7 days 3 days |
Vulvovaginal candidiasis and gastrointestinal side effects. |
https://dailymed.nlm. nih.gov/dailymed/ (Accessed 28, 06.2021). |
| 3. | Tinidazole | 2 g orally once daily 1 g orally once daily |
2 days 5 days |
Metallic/bitter taste, nausea, and weakness or fatigue |
https://dailymed.nlm. nih.gov/dailymed/ (Accessed 30.04.2021 |
| 4. | Secnidazole | 2 g packet (orally) once | Vulvovaginal candidiasis. Headache, nausea, diarrhoea and abdominal pain |
https://www.accessdata.fda.gov/ drugsatfda_docs/label/2021/ 209363s015lbl.pdf (Accessed 17.06. 2021 |
|
| 5. | Dequalinium chloride | 10 mg tablet intra vaginally | 6 nights | Product use is limited |
https://pdf.hres.ca/dpd _pm/00063156.PDF (Accessed 25.08. 2022). |
| S. No. | Prebiotics | Beneficial effects | Adverse effect | References |
| 1. | Oligosaccharides | Selectively help in promoting the enrichment of lactobacilli Increase lactic acid production Prevent the growth of anaerobic bacteria by inhibiting adhesion and replication through the secretion of antibacterial substances. |
Not observed | Rousseau et al, 2005 |
| 2. | Vaginal sucrose gel | Therapeutic cure rate was 61% after 21–35 days treatment At 5-7 days, the Nugent score showed significantly higher levels of lactobacilli in the sucrose gel group treatment as versus the metronidazole group. |
Promote the development of candidiasis | Zeng et al, 2010 |
| 3. | Disaccharide lactulose | Promote the enrichment of vaginal lactobacilli including L. crispatus and non-stimulating of C. albicans and other harmful bacteria spotted in BV. | Not observed | Collins et al, 2018 |
| 4 | Trifolium pratense (red clover) extract and Galacto-oligosaccharides | Nugent score ≤ 3 | Not observed | Coste et al, 2012 |
| 5. | Lactoferrin | Improve the BV by gradual increasing of lactobacilli | Not observed | Otsuki and Imai, 2017 |
| CitationsClinicalTrials.gov Identifier | Population | Study Focus | Primary cure rate |
| Kelly et al, 2015 | N=46 | FMT vs Antibiotic |
91% |
| Youngster et al, 2016 | N=180 | FMT Delivery Method |
82% |
| Jiang e al, 2017 | N=72 | FMT Material Processing | 100% |
| Myles et al, 2018 | N=15 | Topical Microbiome Transplant | 75% |
| Lev-Sagie et al, 2019 | N=5 | VMT for Recurrent Bacterial Vaginosis | 75% |
| NCT04046900 | N=134 | VMT for Recurrent Bacterial Vaginosis | Primary completion on December 2024 |
| NCT04517487 | N=100 | Biological: Vaginal Microbiome Transplantation | Primary completion on December 2024 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




