Submitted:
10 November 2025
Posted:
11 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subjects and Experimental Protocol
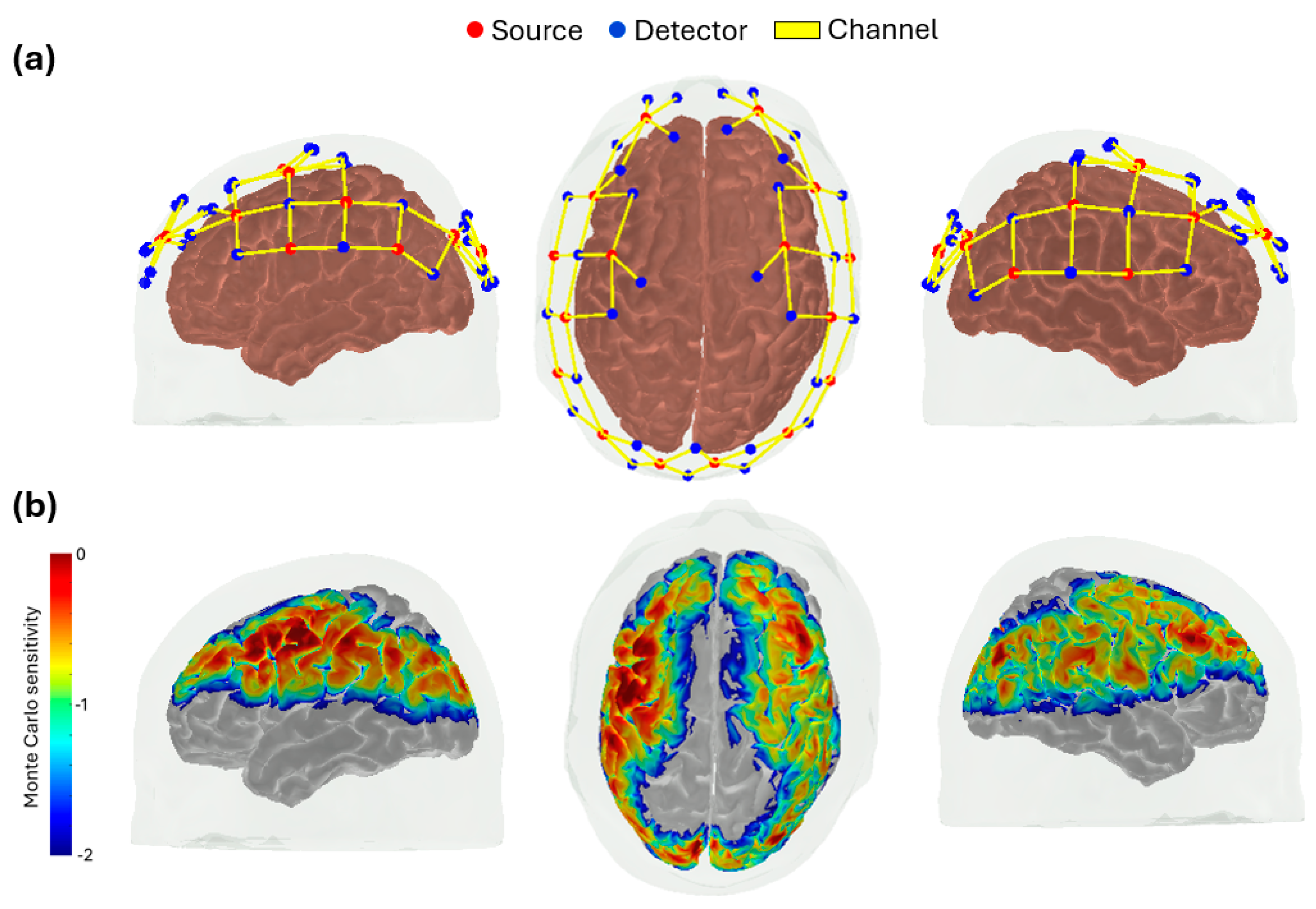
2.2. fNIRS Acquisition and Processing
2.3. Functional Connectivity Analysis
2.4. Statistical Analysis
3. Results
3.1. Power Spectrum of Low-Frequency Oscillations Is Attenuated for Older Adults
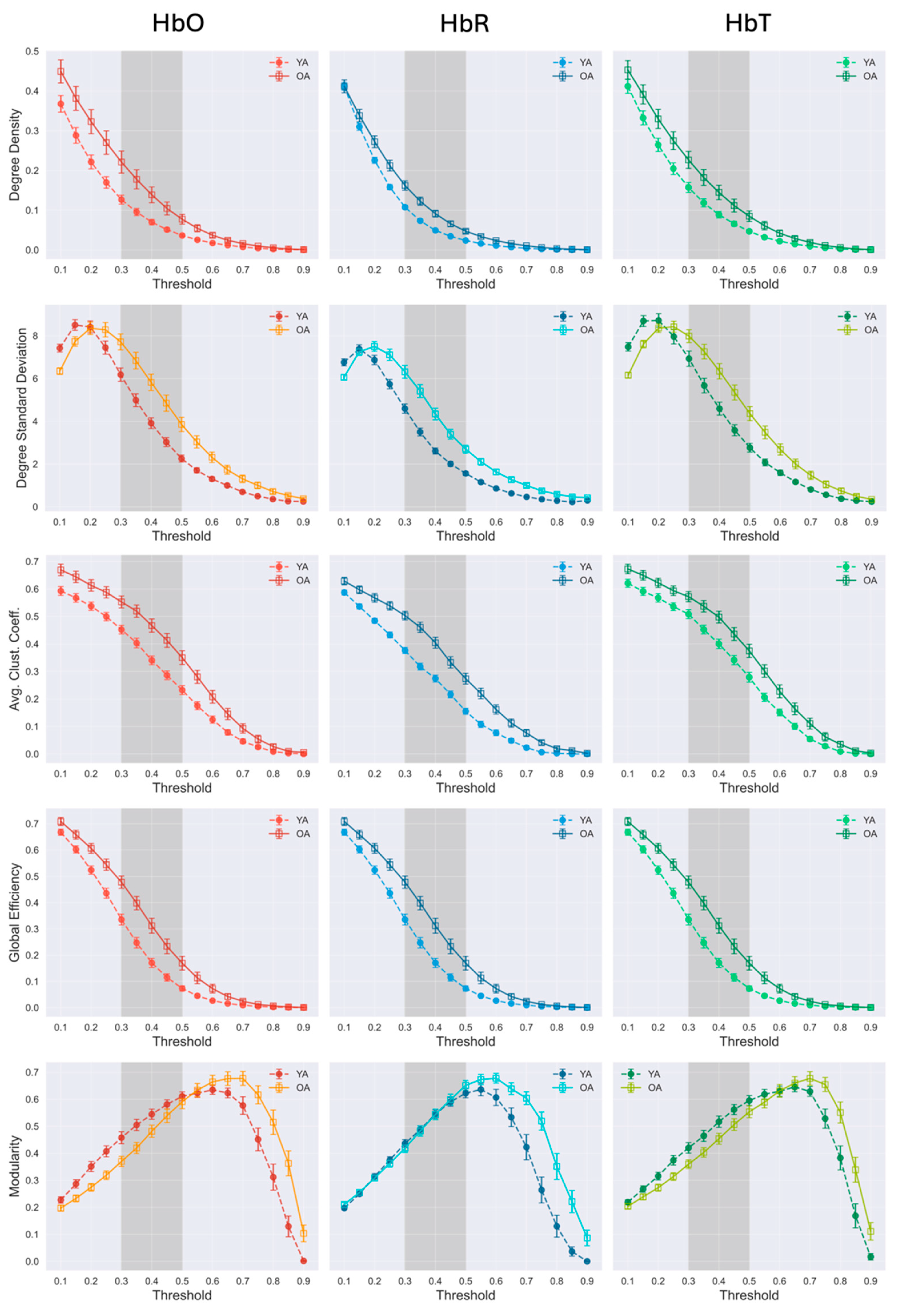
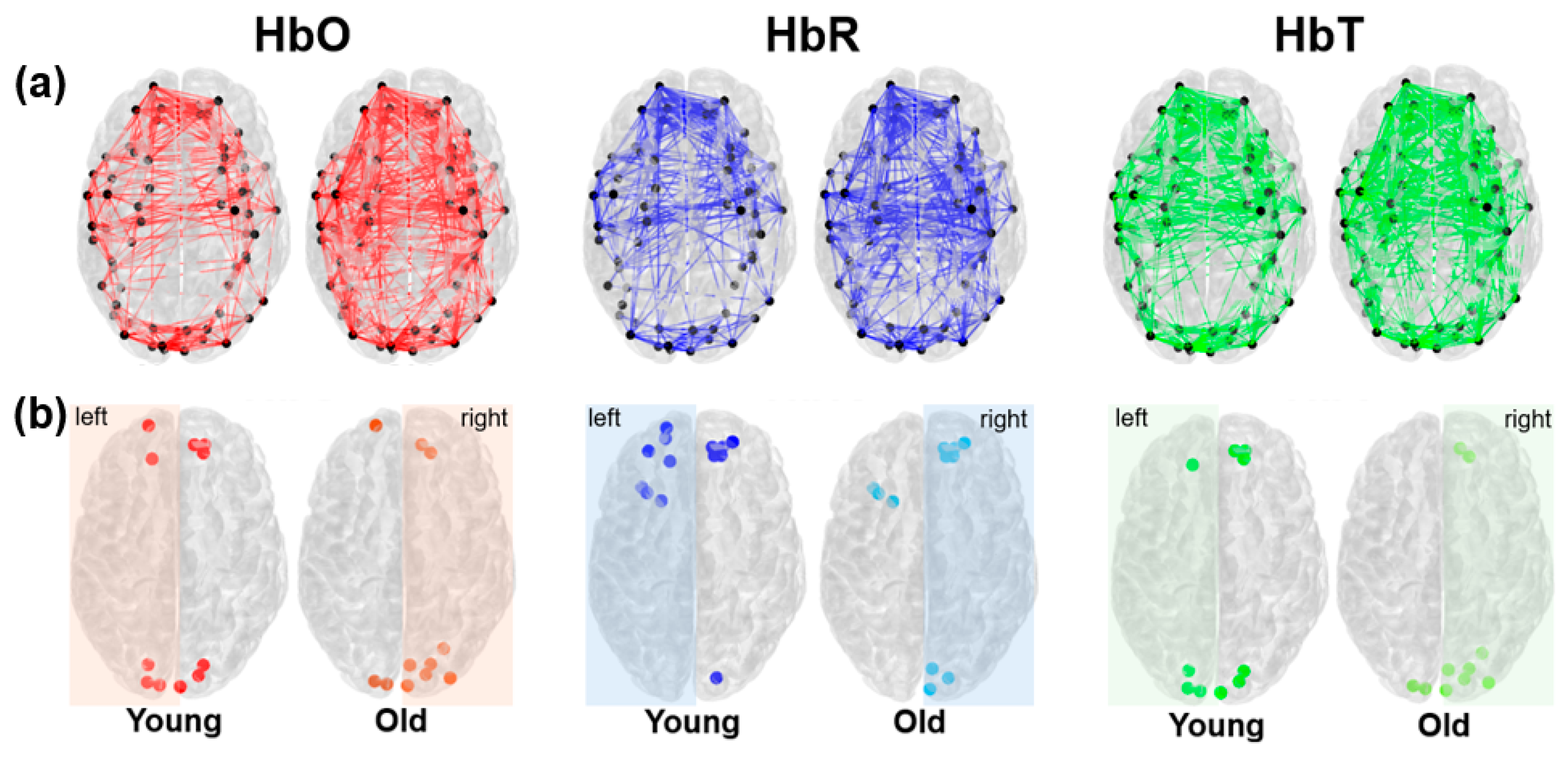
3.2. Resting-State fNIRS Connectivity and Global Network Properties
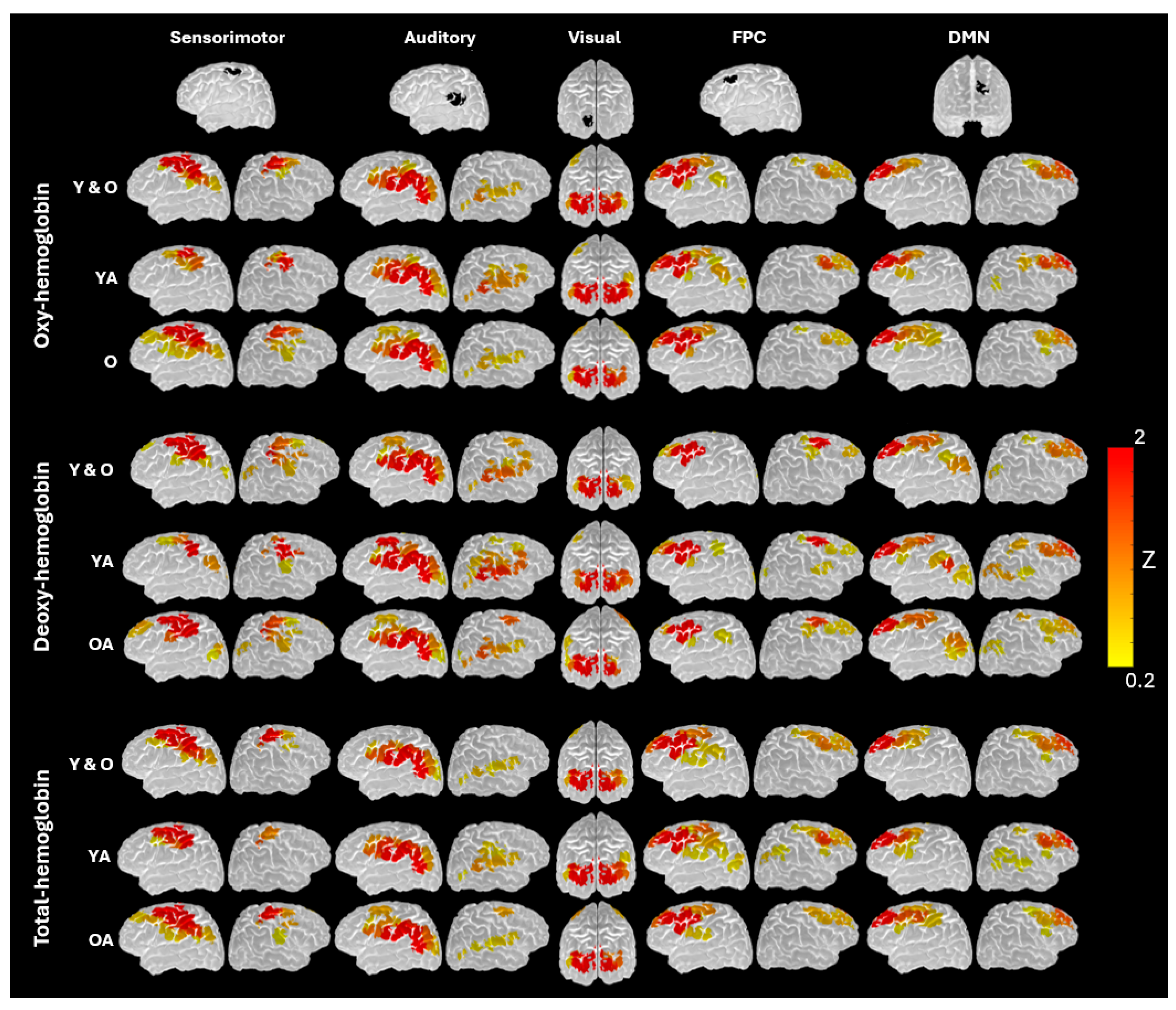
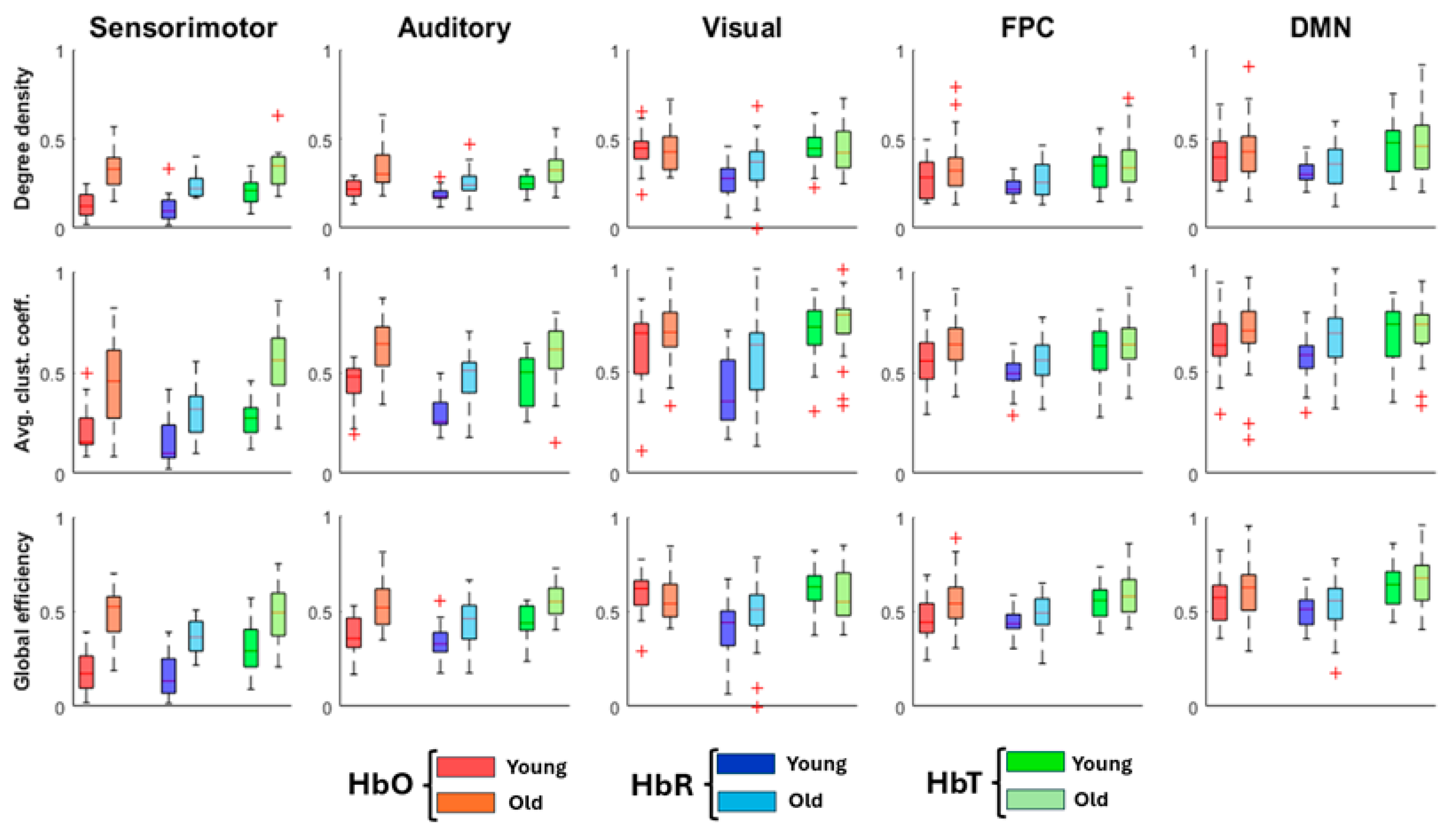
3.2. Network-Specific Topology
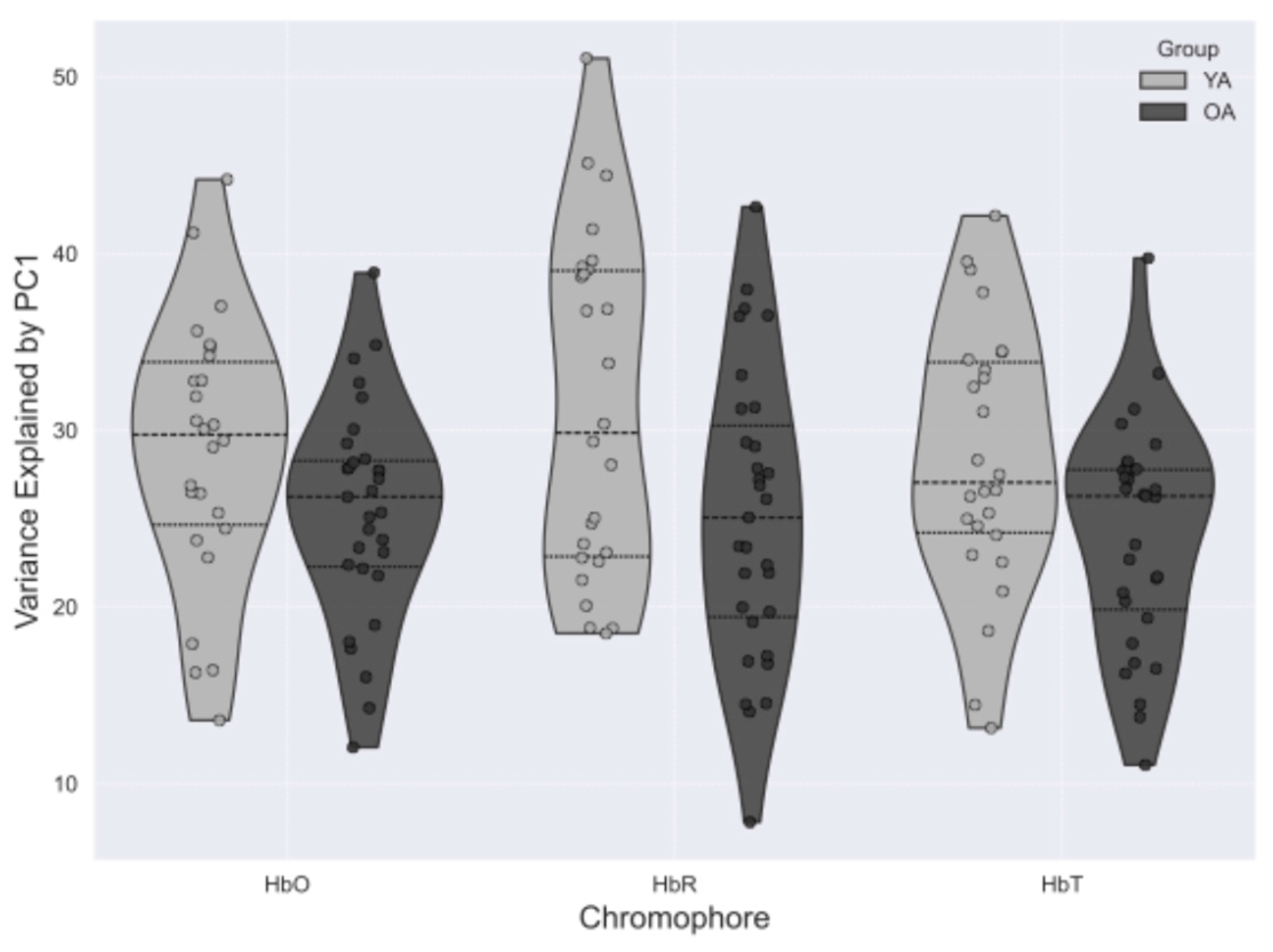
3.3. Influence of Global Signal Regression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| fNIRS | Functional Near-Infrared Spectroscopy |
| rsFC | Resting state functional connectivity |
| HbO | Oxy-hemoglobin |
| HbR | Deoxy-hemoglobin |
| HbT | Total-hemoglobin |
| LFO | Low frequency oscillation |
References
- W. H. Organization. "Ageing and health." World Health Organization. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (accessed October 1, 2025).
- W. Bai et al., "Worldwide prevalence of mild cognitive impairment among community dwellers aged 50 years and older: a meta-analysis and systematic review of epidemiology studies," Age and ageing, vol. 51, no. 8, p. afac173, 2022, doi: 10.1093/ageing/afac173. [CrossRef]
- N. Salari et al., "The global prevalence of mild cognitive impairment in geriatric population with emphasis on influential factors: a systematic review and meta-analysis," BMC geriatrics, vol. 25, no. 1, p. 313, 2025, doi: 10.1186/s12877-025-05967-w. [CrossRef]
- J. S. Damoiseaux, "Effects of aging on functional and structural brain connectivity," Neuroimage, vol. 160, pp. 32-40, 2017, doi: 10.1016/j.neuroimage.2017.01.077. [CrossRef]
- M. Fjell and K. B. Walhovd, "Structural brain changes in aging: courses, causes and cognitive consequences," Reviews in the Neurosciences, vol. 21, no. 3, pp. 187-222, 2010, doi: 10.1515/revneuro.2010.21.3.187. [CrossRef]
- L. L. Beason-Held et al., "Changes in brain function occur years before the onset of cognitive impairment," Journal of Neuroscience, vol. 33, no. 46, pp. 18008-18014, 2013, doi: 10.1523/JNEUROSCI.1402-13.2013. [CrossRef]
- J. D. Power, B. L. Schlaggar, and S. E. Petersen, "Studying brain organization via spontaneous fMRI signal," Neuron, vol. 84, no. 4, pp. 681-696, 2014, doi: 10.1016/j.neuron.2014.09.007. [CrossRef]
- M. P. van den Heuvel and H. E. H. Pol, "Exploring the brain network: A review on resting-state fMRI functional connectivity," European Neuropsychopharmacology, vol. 20, no. 8, pp. 519-534, Aug 2010, doi: 10.1016/j.euroneuro.2010.03.008. [CrossRef]
- L. Farras-Permanyer et al., "Age-related changes in resting-state functional connectivity in older adults," Neural regeneration research, vol. 14, no. 9, pp. 1544-1555, 2019, doi: 10.4103/1673-5374.255976. [CrossRef]
- N. Franzmeier et al., "Resting-state global functional connectivity as a biomarker of cognitive reserve in mild cognitive impairment," Brain imaging and behavior, vol. 11, no. 2, pp. 368-382, 2017, doi: 10.1007/s11682-016-9599-1. [CrossRef]
- Q. Lin, M. D. Rosenberg, K. Yoo, T. W. Hsu, T. P. O'Connell, and M. M. Chun, "Resting-state functional connectivity predicts cognitive impairment related to Alzheimer's disease," Frontiers in aging neuroscience, vol. 10, p. 94, 2018, doi: 10.3389/fnagi.2018.00094. [CrossRef]
- P. Ranasinghe and M. S. Mapa, "Functional connectivity and cognitive decline: A review of rs-fMRI, EEG, MEG, and graph theory approaches in aging and dementia," Exploration of Medicine, vol. 5, no. 6, pp. 797-821, 2024, doi: 10.37349/emed.2024.00256. [CrossRef]
- L. K. Ferreira and G. F. Busatto, "Resting-state functional connectivity in normal brain aging," Neuroscience & Biobehavioral Reviews, vol. 37, no. 3, pp. 384-400, 2013, doi: 10.1016/j.neubiorev.2013.01.017. [CrossRef]
- L. Geerligs, R. J. Renken, E. Saliasi, N. M. Maurits, and M. M. Lorist, "A brain-wide study of age-related changes in functional connectivity," Cerebral cortex, vol. 25, no. 7, pp. 1987-1999, 2015, doi: 10.1093/cercor/bhu012. [CrossRef]
- H. Ayaz et al., "Optical imaging and spectroscopy for the study of the human brain: status report," Neurophotonics, vol. 9, Aug 2022, doi: 10.1117/1.NPh.9.S2.S24001. [CrossRef]
- P. Pinti et al., "The present and future use of functional near-infrared spectroscopy (fNIRS) for cognitive neuroscience," Annals of the new York Academy of Sciences, vol. 1464, no. 1, pp. 5-29, 2020, doi: 10.1111/nyas.13948. [CrossRef]
- V. Quaresima and M. Ferrari, "A Mini-Review on Functional Near-Infrared Spectroscopy (fNIRS): Where Do We Stand, and Where Should We Go?," Photonics, vol. 6, no. 3, Sep 2019, Art no. 87, doi: 10.3390/photonics6030087. [CrossRef]
- C. Udina et al., "Functional near-infrared spectroscopy to study cerebral hemodynamics in older adults during cognitive and motor tasks: a review," Frontiers in aging neuroscience, vol. 11, p. 367, 2020, doi: 10.3389/fnagi.2019.00367. [CrossRef]
- L. Li et al., "Whole-cortical graphical networks at wakeful rest in young and older adults revealed by functional near-infrared spectroscopy," Neurophotonics, vol. 5, no. 3, pp. 035004-035004, 2018, doi: 10.1117/1.NPh.5.3.035004. [CrossRef]
- S. Jiang et al., "Functional connectivity and characteristics of cortical brain networks of elderly individuals under different motor cognitive tasks based on functional near-infrared spectroscopy," Frontiers in Human Neuroscience, vol. 19, p. 1563338, 2025, doi: 10.3389/fnhum.2025.1563338. [CrossRef]
- L. Blum et al., "Effects of aging on functional connectivity in a neurodegenerative risk cohort: Resting state versus task measurement using near-infrared spectroscopy," Scientific reports, vol. 12, no. 1, p. 11262, 2022, doi: 10.1038/s41598-022-13326-7. [CrossRef]
- T. J. Huppert, S. G. Diamond, M. A. Franceschini, and D. A. Boas, "HomER: a review of time-series analysis methods for near-infrared spectroscopy of the brain," Applied Optics, vol. 48, no. 10, pp. D280-D298, Apr 2009, doi: 10.1364/ao.48.00d280. [CrossRef]
- L. Pollonini, C. Olds, H. Abaya, H. Bortfeld, M. S. Beauchamp, and J. S. Oghalai, "Auditory cortex activation to natural speech and simulated cochlear implant speech measured with functional near-infrared spectroscopy," Hearing research, vol. 309, pp. 84-93, 2014, doi: 10.1016/j.heares.2013.11.007. [CrossRef]
- L. Pollonini, H. Bortfeld, and J. S. Oghalai, "PHOEBE: a method for real time mapping of optodes-scalp coupling in functional near-infrared spectroscopy," Biomedical optics express, vol. 7, no. 12, pp. 5104-5119, 2016, doi: 10.1364/BOE.7.005104. [CrossRef]
- S. L. Novi et al., "Functional near-infrared spectroscopy for speech protocols: characterization of motion artifacts and guidelines for improving data analysis," Neurophotonics, vol. 7, 1, p. 015001, 2020, doi: 10.1117/1.NPh.7.1.015001. [CrossRef]
- R. C. Mesquita, M. A. Franceschini, and D. A. Boas, "Resting state functional connectivity of the whole head with near-infrared spectroscopy," Biomedical Optics Express, vol. 1, no. 1, pp. 324-336, Aug 2010, doi: 10.1364/boe.1.000324. [CrossRef]
- S. L. Novi, R. Rodrigues, and R. C. Mesquita, "Resting state connectivity patterns with near-infrared spectroscopy data of the whole head," Biomedical Optics Express, vol. 7, no. 7, pp. 2524-2537, Jul 2016, doi: 10.1364/boe.7.002524. [CrossRef]
- B. R. White et al., "Resting-state functional connectivity in the human brain revealed with diffuse optical tomography," Neuroimage, vol. 47, no. 1, pp. 148-156, Aug 2009, doi: 10.1016/j.neuroimage.2009.03.058. [CrossRef]
- Abdalmalak et al., "Effects of Systemic Physiology on Mapping Resting-State Networks Using Functional Near-Infrared Spectroscopy," Frontiers in Neuroscience, vol. 16, Mar 2022, Art no. 803297, doi: 10.3389/fnins.2022.803297. [CrossRef]
- X. Quan et al., "Frequency-specific changes of amplitude of low-frequency fluctuations in patients with acute basal ganglia ischemic stroke," Neural plasticity, vol. 2022, no. 1, p. 4106131, 2022, doi: 10.1155/2022/4106131. [CrossRef]
- X.-N. Zuo et al., "The oscillating brain: complex and reliable," Neuroimage, vol. 49, no. 2, pp. 1432-1445, 2010, doi: 10.1016/j.neuroimage.2009.09.037. [CrossRef]
- J. W. Barker, A. Aarabi, and T. J. Huppert, "Autoregressive model based algorithm for correcting motion and serially correlated errors in fNIRS," Biomedical Optics Express, vol. 4, no. 8, pp. 1366-1379, Aug 2013, doi: 10.1364/boe.4.001366. [CrossRef]
- C. M. Aasted et al., "Anatomical guidance for functional near-infrared spectroscopy: AtlasViewer tutorial," Neurophotonics, vol. 2, no. 2, Apr-Jun 2015, Art no. 020801, doi: 10.1117/1.NPh.2.2.020801. [CrossRef]
- M. Rubinov and O. Sporns, "Complex network measures of brain connectivity: Uses and interpretations," NEUROIMAGE, vol. 52, no. 3, pp. 1059-1069, 2010-09-01 2010, doi: 10.1016/j.neuroimage.2009.10.003. [CrossRef]
- S. Achard and E. Bullmore, "Efficiency and cost of economical brain functional networks," PLoS computational biology, vol. 3, no. 2, p. e17, 2007, doi: 10.1371/journal.pcbi.0030017. [CrossRef]
- D. S. Bassett and O. Sporns, "Network neuroscience," Nature neuroscience, vol. 20, no. 3, pp. 353-364, 2017, doi: 10.1038/nn.4502. [CrossRef]
- E. Bullmore and O. Sporns, "Complex brain networks: graph theoretical analysis of structural and functional systems," Nature reviews neuroscience, vol. 10, no. 3, pp. 186-198, 2009, doi: 10.1038/nrn2575. [CrossRef]
- Fornito, Andrew Zalesky, and E. Bullmore, Fundamentals of brain network analysis. Academic press, 2016.
- M. E. Newman and M. Girvan, "Finding and evaluating community structure in networks," Physical review E, vol. 69, no. 2, p. 026113, 2004, doi: 10.1103/PhysRevE.69.026113. [CrossRef]
- R. Albert and A.-L. Barabási, "Statistical mechanics of complex networks," Reviews of modern physics, vol. 74, no. 1, p. 47, 2002, doi: 10.1103/RevModPhys.74.47. [CrossRef]
- C. Zhang, C. C. Dougherty, S. A. Baum, T. White, and A. M. Michael, "Functional connectivity predicts gender: Evidence for gender differences in resting brain connectivity," Human brain mapping, vol. 39, no. 4, pp. 1765-1776, 2018, doi: 10.1002/hbm.23950. [CrossRef]
- M. L. Schroeter, O. Schmiedel, and D. Y. von Cramon, "Spontaneous low-frequency oscillations decline in the aging brain," Journal of Cerebral Blood Flow & Metabolism, vol. 24, no. 10, pp. 1183-1191, 2004, doi: 10.1097/01.WCB.0000135231.90164.40. [CrossRef]
- S. Song et al., "Low-frequency oscillations in cerebrovascular and cardiovascular hemodynamics: Their interrelationships and the effect of age," Microvascular research, vol. 102, pp. 46-53, 2015, doi: 10.1016/j.mvr.2015.08.004. [CrossRef]
- M. D'Esposito, L. Y. Deouell, and A. Gazzaley, "Alterations in the BOLD fMRI signal with ageing and disease: a challenge for neuroimaging," Nature Reviews Neuroscience, vol. 4, no. 11, pp. 863-872, 2003, doi: 10.1038/nrn1246. [CrossRef]
- D. Kumral et al., "BOLD and EEG signal variability at rest differently relate to aging in the human brain," NeuroImage, vol. 207, p. 116373, 2020, doi: 10.1016/j.neuroimage.2019.116373. [CrossRef]
- S. L. Novi, A. C. Carvalho, R. M. Forti, F. Cendes, C. L. Yasuda, and R. C. Mesquita, "Revealing the spatiotemporal requirements for accurate subject identification with resting-state functional connectivity: a simultaneous fNIRS-fMRI study," Neurophotonics, vol. 10, no. 1, Jan 2023, Art no. 013510, doi: 10.1117/1.NPh.10.1.013510. [CrossRef]
- H. Li, X. Yang, and L. Gong, "Functional near-infrared spectroscopy for identifying mild cognitive impairment and Alzheimer's disease: a systematic review," Frontiers in Neurology, vol. 16, p. 1578375, 2025, doi: 10.3389/fneur.2025.1578375. [CrossRef]
- T. K. K. Ho et al., "Deep learning-based multilevel classification of Alzheimer’s disease using non-invasive functional near-infrared spectroscopy," Frontiers in aging neuroscience, vol. 14, p. 810125, 2022, doi: 10.3389/fnagi.2022.810125. [CrossRef]
- H. Santosa, X. Zhai, F. Fishburn, P. J. Sparto, and T. J. Huppert, "Quantitative comparison of correction techniques for removing systemic physiological signal in functional near-infrared spectroscopy studies," Neurophotonics, vol. 7, no. 3, pp. 035009-035009, 2020, doi: 10.1117/1.NPh.7.3.035009. [CrossRef]
- P. Lanka, H. Bortfeld, and T. J. Huppert, "Correction of global physiology in resting-state functional near-infrared spectroscopy," Neurophotonics, vol. 9, no. 3, pp. 035003-035003, 2022, doi: 10.1117/1.NPh.9.3.035003. [CrossRef]
- E. Butters et al., "Brain Network Analysis in Alzheimer’s Disease and Mild Cognitive Impairment using High-Density Diffuse Optical Tomography," bioRxiv, p. 2025.04. 28.651132, 2025, doi: 10.1101/2025.04.28.651132. [CrossRef]
- S. Weis, K. R. Patil, F. Hoffstaedter, A. Nostro, B. Yeo, and S. B. Eickhoff, "Sex classification by resting state brain connectivity," Cerebral cortex, vol. 30, no. 2, pp. 824-835, 2020, doi: 10.1093/cercor/bhz129. [CrossRef]
- D. Tomasi and N. D. Volkow, "Gender differences in brain functional connectivity density," Human brain mapping, vol. 33, no. 4, pp. 849-860, 2012, doi: 10.1002/hbm.21252. [CrossRef]
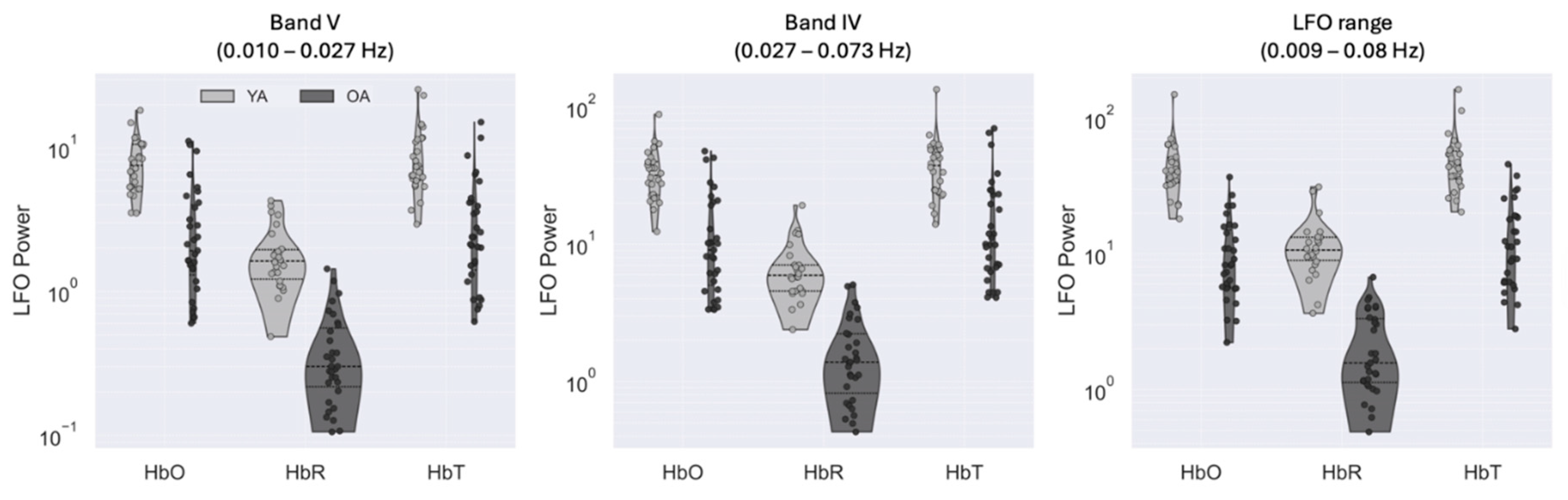
| Resting state | Band V | Band IV | |
| HbO | |||
| YA | 40.7 [33.6 – 51.5] | 7.6 [5.4–10.6] | 31.8 [22.9–40.1] |
| OA | 8.3 [5.6 – 15.1] | 1.9 [1.3–4.0] | 9.1 [5.0–19.7] |
| HbR | |||
| YA | 10.7 [8.9 – 13.3] | 1.6 [1.1 – 3.3] | 5.9 [4.6 – 7.1] |
| OA | 1.6 [ 1.1 – 3.3] | 0.30 [0.22 – 0.56] | 1.4 [ 0.82 – 2.2] |
| HbT | |||
| YA | 45.4 [35.9 – 57.3] | 7.5 [ 5.9 – 11.6] | 37.6 [24.4 – 49.3] |
| OA | 11.3 [ 6.2 – 18.8] | 2.4 [1.4 – 4.2] | 10.0 [6.7 – 23.1] |
| Degree density | Avg. CC | Global Efficiency | |
| HbO | |||
| YA | 5.4 [5.3 – 5.5] | 1.97 [1.96 – 1.99] | 2.94 [2.92 – 2.95] |
| OA | 3.4 [3.3 – 3.5] | 1.72 [1.70 – 1.73] | 2.29 [2.27 – 2.30] |
| HbR | |||
| YA | 7.2 [7.0 – 7.4] | 2.26 [2.24 – 2.29] | 3.10 [3.09 – 3.12] |
| OA | 5.3 [5.0 – 5.6] | 1.84 [1.83 – 1.85] | 2.59 [2.57 – 2.60] |
| HbT | |||
| YA | 5.1 [4.9 – 5.3] | 1.85 [1.84 –1.86] | 2.63 [2.62 – 2.64] |
| OA | 3.3 [3.2 – 3.4] | 1.67 [1.65 – 1.68] | 2.22 [2.21 – 2.23] |
| Graph Properties | ||||
| Degree density | Avg. CC | Global Efficiency | Modularity | |
| HbO | ||||
| YA | 0.07 [0.05 – 0.10] | 0.39 [0.31 – 0.43] | 0.20 [0.14 – 0.26] | 0.55 [0.51 – 0.58] |
| OA | 0.13 [0.09 – 0.18] | 0.49 [0.42 – 0.56] | 0.34 [0.27 – 0.41] | 0.48 [0.42 – 0.54] |
| HbR | ||||
| YA | 0.06 [0.05 – 0.08] | 0.30 [0.25 – 0.34] | 0.16 [0.13 – 0.22] | 0.55 [0.49 – 0.59] |
| OA | 0.09 [0.07 – 0.12] | 0.43 [0.37 – 0.51] | 0.28 [0.21 – 0.36] | 0.54 [0.48 – 0.60] |
| HbT | ||||
| YA | 0.09 [0.07 – 0.12] | 0.43 [0.36 – 0.48] | 0.29 [0.18 – 0.34] | 0.52 [0.46 – 0.56] |
| OA | 0.16 [0.10 – 0.18] | 0.50 [0.46 – 0.57] | 0.38 [0.28 – 0.45] | 0.45 [0.40 – 0.51] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




