Submitted:
06 November 2025
Posted:
09 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
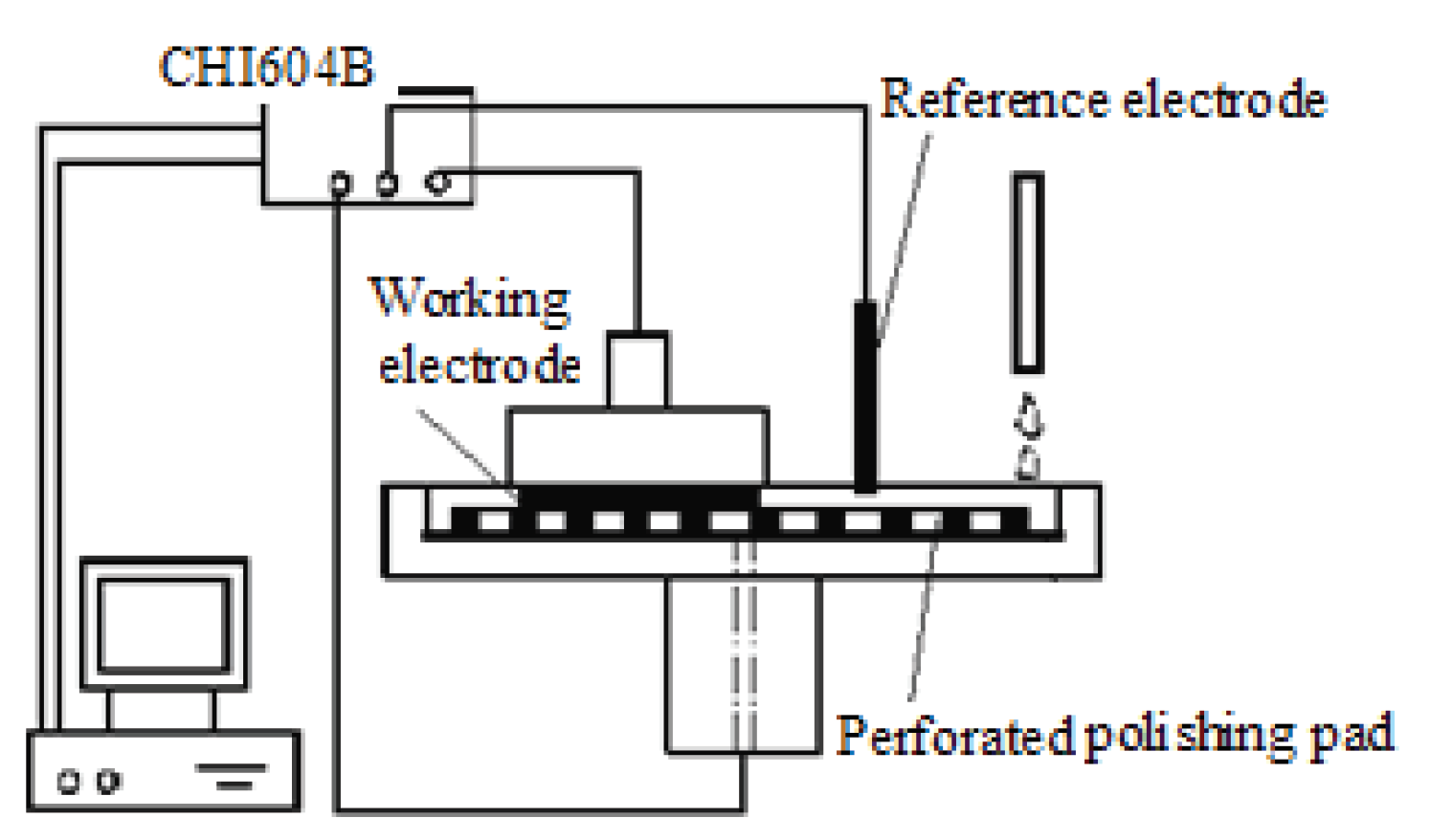
2. Experimental
3. Results and Discussions
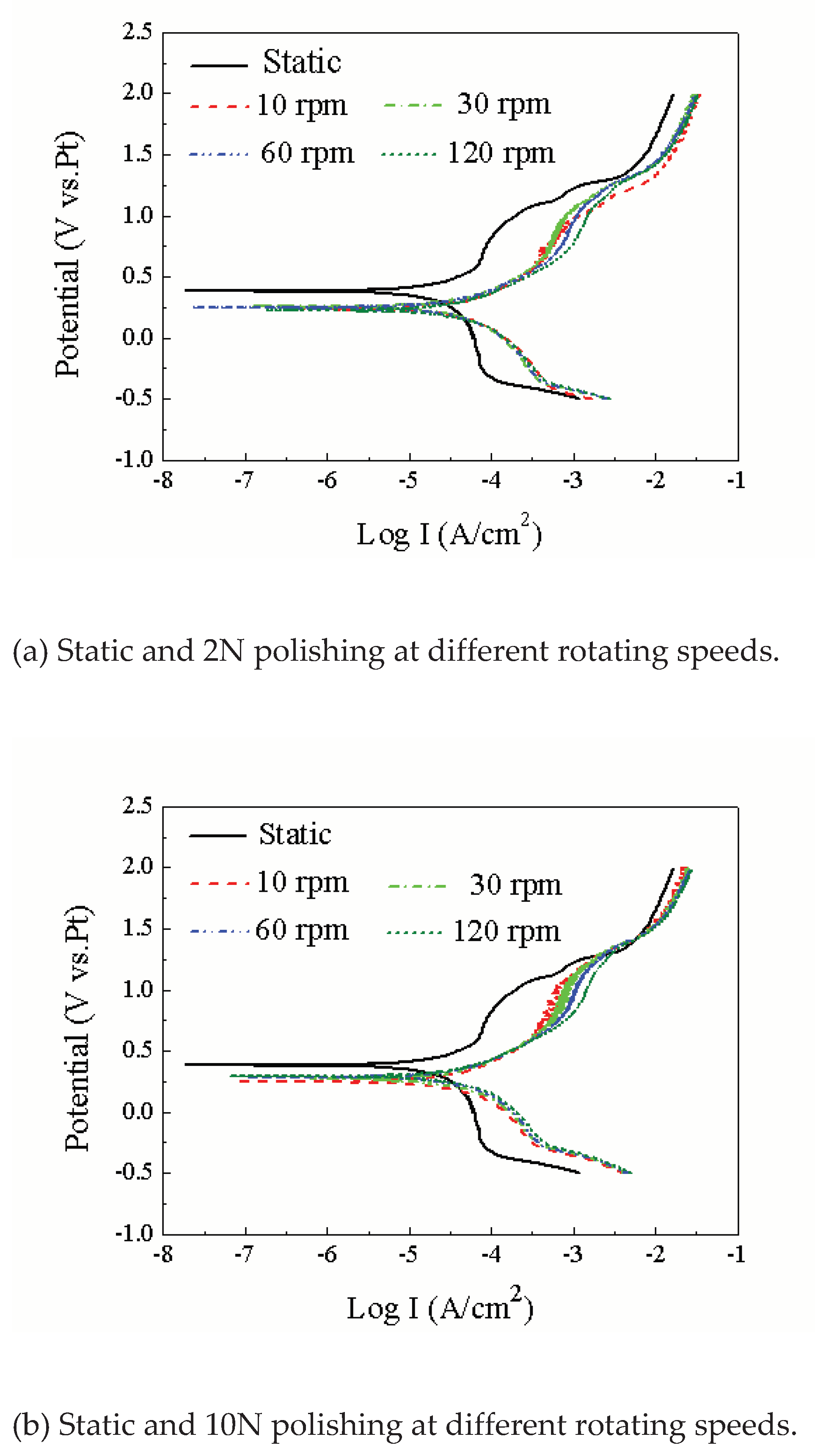
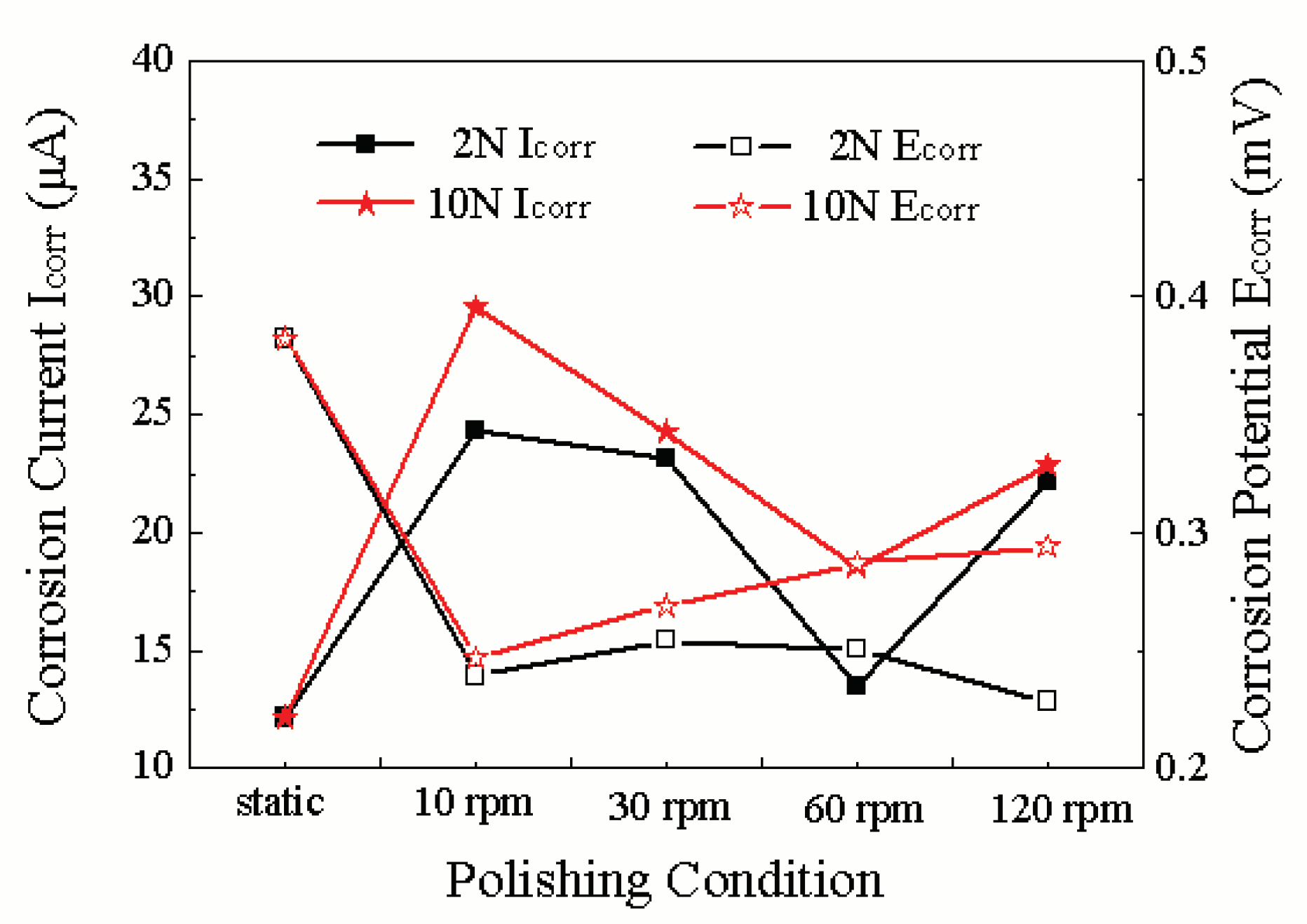
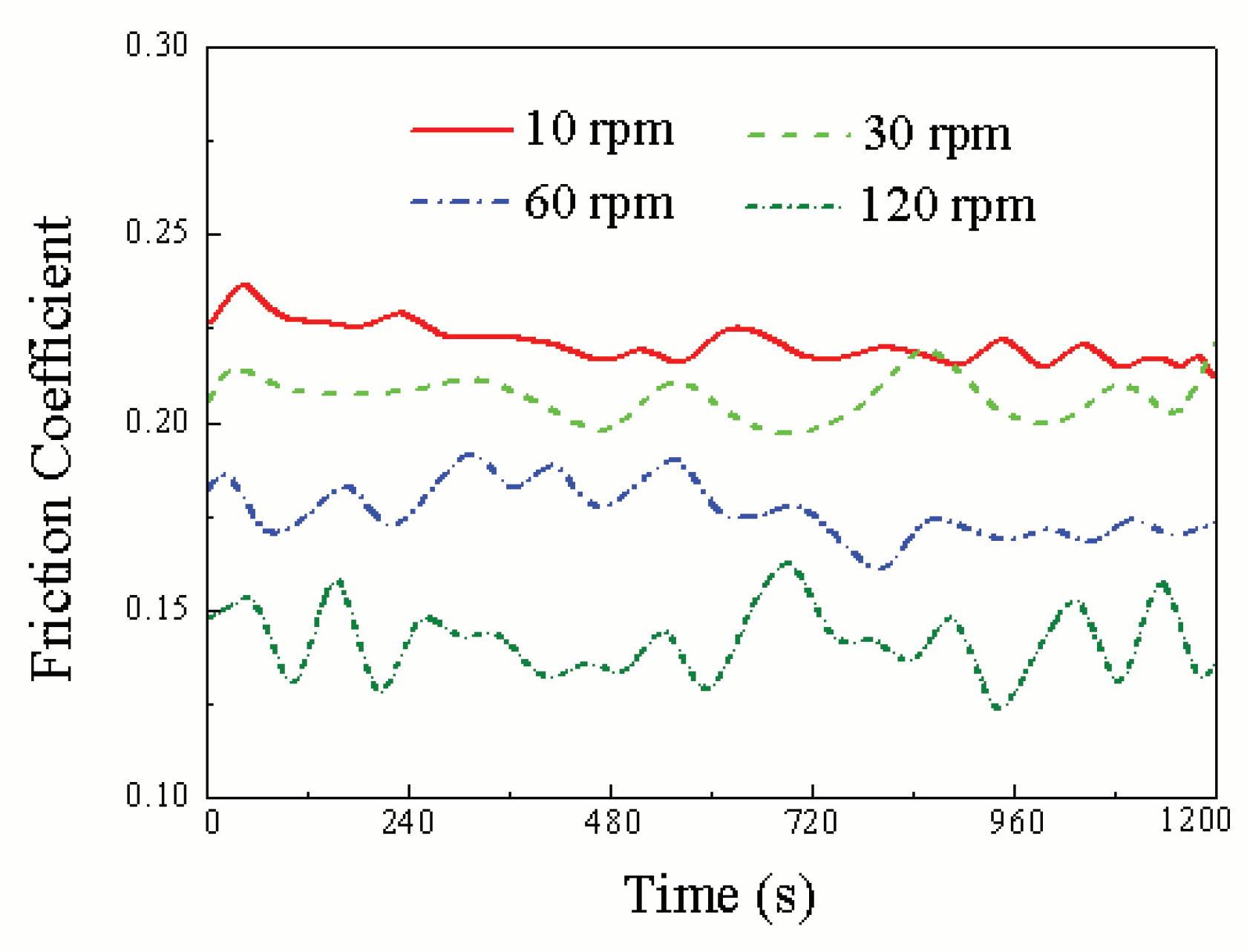
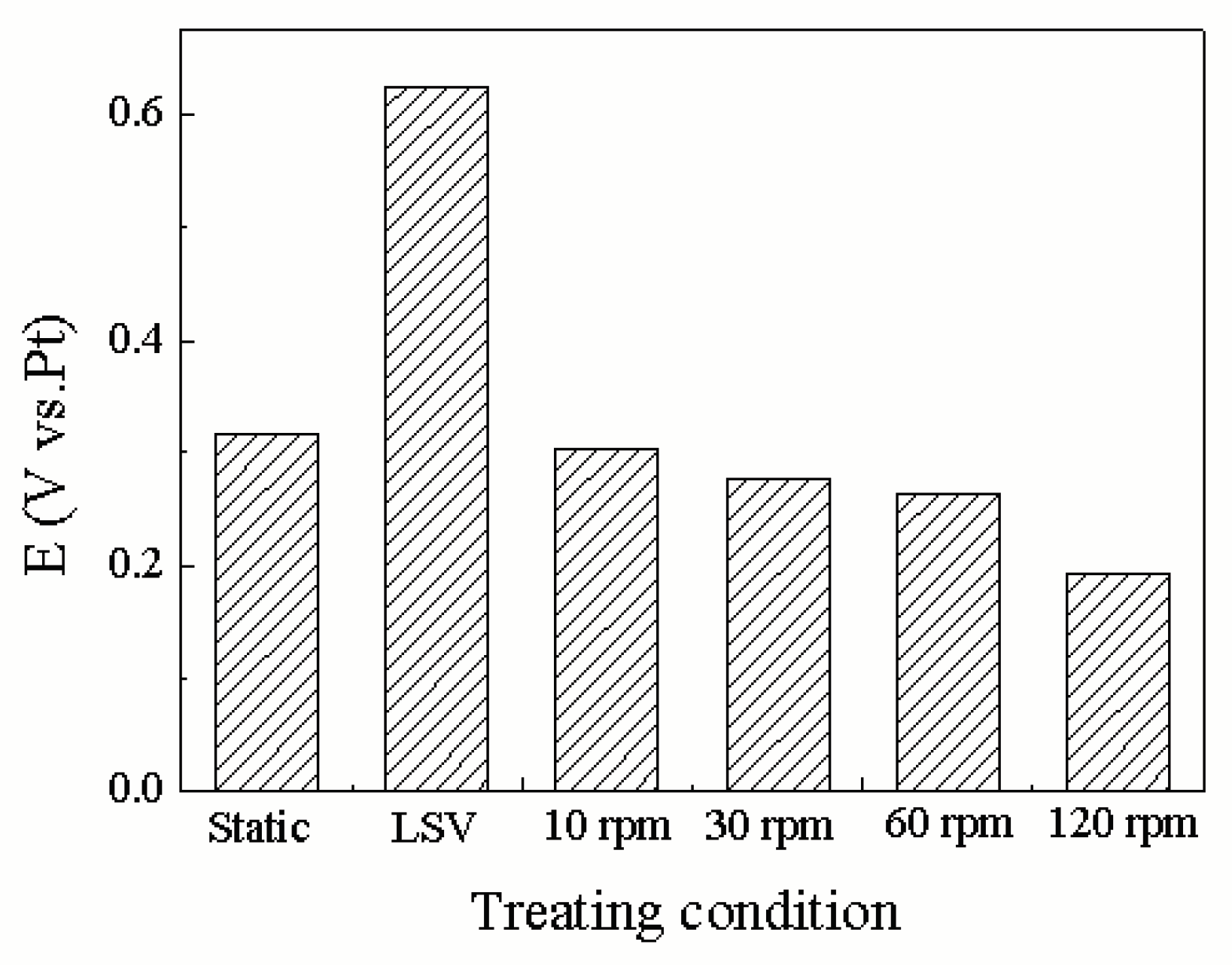
3.1. Influence Mechanism of Force and Linear Velocity on the Triboelectrochemical Performance of Ruthenium ECMP
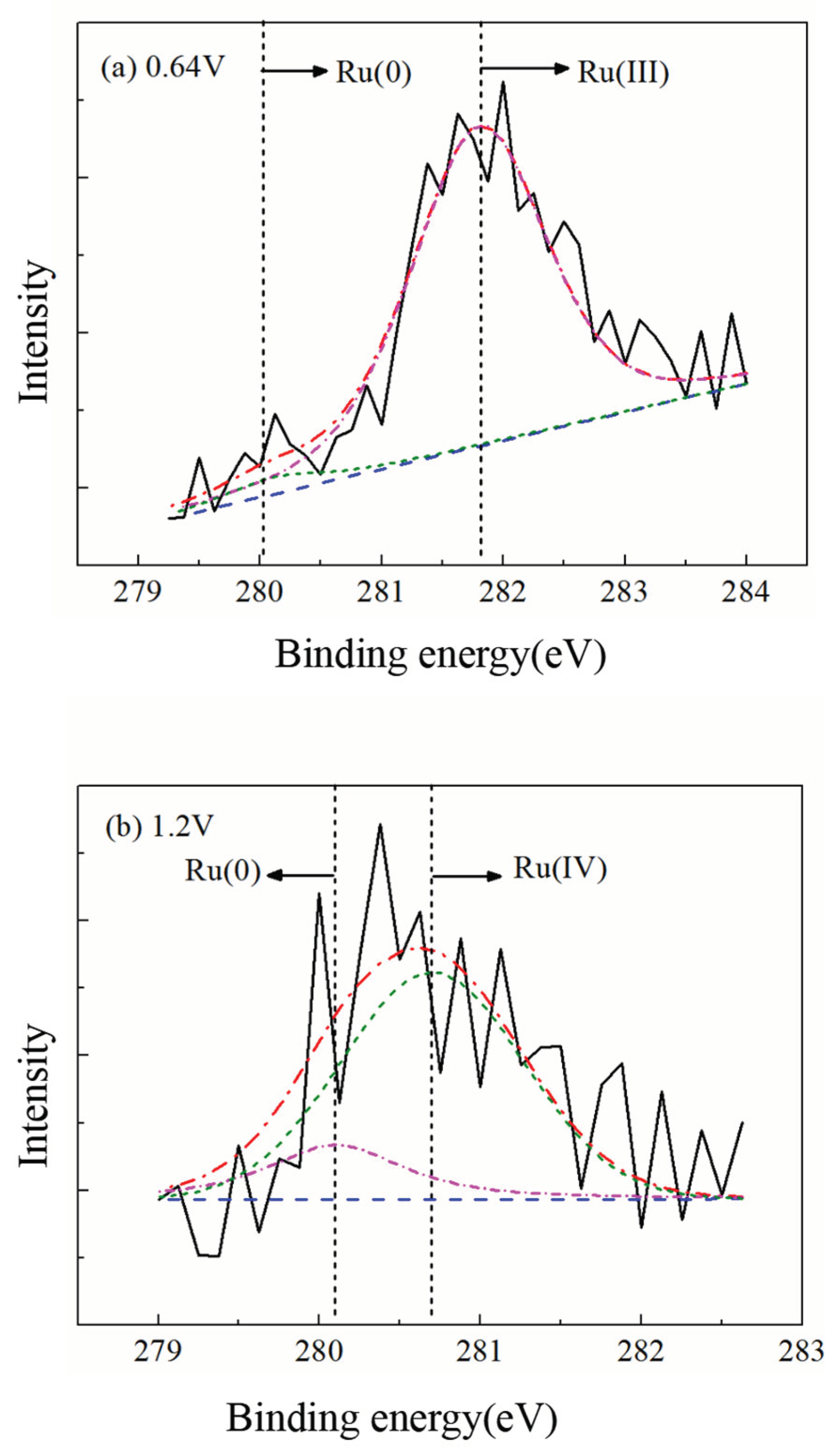
3.2. Effect of Applied Potential on the Surface Characteristics of Ruthenium ECMP
3.3. Comprehensive Evaluation of Influencing Factors in Ruthenium ECMP
4. Conclusions
Acknowledgments
References
- You, Y.; Zhou, J.; Qi, Y.; Luo, C. Research on the Mechanism of Ru Abrasive-free CMP Based on KIO4/FeIII-NTA System. ECS J. Solid State Sci. Technol. 2025, 14, 054007. [Google Scholar] [CrossRef]
- Zhan, N.; He, C.; He, C.; Niu, X.; Zhang, N.-N.; Zhang, N.; Zou, Y.; Liu, J.; Dong, C.; Dong, C.; Li, X.; Li, X.; Zhou, J. Effects of pyrazine and its derivatives as inhibitors on copper film chemical-mechanical polishing properties for ruthenium-based copper interconnect. Colloids Surf. A Physicochem. Eng. Asp. 2024, 688, 133609. [Google Scholar] [CrossRef]
- Dong, C.; Niu, X.; Liu, J.; Zhan, N.; Zou, Y.; He, C.; Li, X.; Luo, F.; Shi, Y.; Cheng, J. Effect of Novel pH Regulators on Copper film Chemical Mechanical Polishing for Ruthenium-Based Copper Interconnect under Weak Alkalinity Conditions. ECS J. Solid State Sci. Technol. 2024, 13, 054001. [Google Scholar] [CrossRef]
- Elshocht, S.V.; Delabie, A.; Dewilde, S.; Meersschaut, J.; Swerts, J.; Tielens, H.; Verdonck, P.; Witters, T.; Vancoille, E. ALD barrier deposition on porous low-k dielectric materials for interconnects. ECS Trans. 2011, 41, 25–32. [Google Scholar] [CrossRef]
- Zhou, W.; Bailey, S.; Sooryakumar, R.; King, S.; Xu, G.; Mays, E.; Ege, C.; Bielefeld, J. Elastic properties of porous low-k dielectric nano-films. J. Appl. Phys. 2011, 110, 043520. [Google Scholar] [CrossRef]
- Xi, K.; He, H.; Xu, D.; Ge, R.J.; Meng, Z.; Jia, X.D.; Yu, X.H. Ultra low dielectric constant polysilsequioxane films using T8(Me4NO)8 as porogen. Thin Solid Films 2010, 518, 4768–4772. [Google Scholar] [CrossRef]
- Chen, F.; Shinosky, M. Soft breakdown characteristics of ultra low-k time-dependent dielectric breakdown for advanced complementary metal-oxide semiconductor technologies. J. Appl. Phys. 2010, 108, 054107. [Google Scholar] [CrossRef]
- Zhao, L.; Tokei, Z.; Croes, K.; Wilson, C.J.; Baklanov, M.; Beyer, G.P.; Claeys, C. Direct observation of the 1/E dependence of time dependent dielectric breakdown in the presence of copper. Appl. Phys. Lett. 2011, 98, 023107. [Google Scholar] [CrossRef]
- Lin, W.C.; Lin, J.; Tsai, T.C.; Hsu, C.M.; Liu, C.C.; Lin, J.F.; Hwang, C.C.; Wu, J.Y. Effects of Cu surface roughness on TDDB for direct polishing ultra-low k dielectric Cu interconnects at 40 nm technology node and beyond. Microelectron. Eng. 2012, 92, 115–118. [Google Scholar] [CrossRef]
- He, M.; Novak, S.; Vanamurthy, L.; Bakhru, H.; Plawsky, J.; Lu, T.M. Cu penetration into low-k dielectric during deposition and bias-temperature stress. Appl. Phys. Lett. 2010, 97, 252901. [Google Scholar] [CrossRef]
- Shin, J.; Kim, H.W.; Hwang, G.S.; Ekerdt, J.G. Chemical routes to ultra thin films for copper barriers and liners. Surf. Coat. Technol. 2007, 201, 9256–9259. [Google Scholar] [CrossRef]
- Chyan, O.; Arunagiri, T.N.; Zhang, Y.; Wallace, R.M.; Kim, M.J.; Hurd, T.Q. Electrodeposition of Copper Thin Film on Ruthenium A Potential Diffusion Barrier for Cu Interconnects. J. Electrochem. Soc. 2003, 150, C347–C350. [Google Scholar] [CrossRef]
- Josell, D.; Wheeler, D.; Witt, C.; Moffat, T.P. Seedless superfill: copper electrodeposition in trenches with ruthenium barriers. Electrochem. Solid-State Lett. 2003, 6, C143–C145. [Google Scholar] [CrossRef]
- Chan, R. Diffusion studies of copper on ruthenium thin film. Electrochem. Solid-State Lett. 2004, 7, G154–G157. [Google Scholar] [CrossRef]
- Mahadevaiyer, K.; Nalaskowski, J.W.; Cook, L.M. Chemical Mechanical Planarization: Slurry Chemistry, Materials, and Mechanisms. Chem. Rev. 2010, 110, 178–204. [Google Scholar]
- Seo, S.C.; Yang, C.C.; Hu, C.K.; Kreber, A.; Fan, S.; Horak, D.; Canaperi, D.; Rao, S.P.; Haran, B.; Doris, B. Thermal Stability of Copper Contact Metallization Using Ru-Containing Liner. Electrochem. Solid-State Lett. 2011, 14, H187–H190. [Google Scholar] [CrossRef]
- Arunagiri, T.N.; Zhang, Y.; Chyan, O.; Bouanani, M.E.; Kim, M.J.; Chen, K.H.; Wu, T.N.; Chen, L.C. 5 nm ruthenium thin film as a directly plateable copper diffusion barrier. Appl. Phys. Lett. 2005, 86, 083104. [Google Scholar] [CrossRef]
- Burke, L.; Naser, N.; Sharna, R. The oxide electrochemistry of ruthenium and its relevance to trench liner applications in damascene copper plating. J. Appl. Electrochem. 2008, 38, 377–384. [Google Scholar] [CrossRef]
- Kim, I.K.; Kang, Y.J.; Kwon, T.Y.; Cho, B.G.; Park, J.G.; Park, J.Y.; Park, H.S. Effect of sodium periodate in alumina-based slurry on Ru CMP for metal-insulator-metal capacitor. Electrochem. Solid-State Lett. 2008, 11, H150–H153. [Google Scholar] [CrossRef]
- Lee, W.J.; Park, H.S.; Lee, S.I.; Sohn, H.C. Effects of ceric ammonium nitrate (CAN) additive in HNO3 solution on the electrochemical behaviour of ruthenium for CMP processes. J. Appl. Electrochem. 2004, 34, 119–125. [Google Scholar] [CrossRef]
- Wu, Z.; Dong, C.; Niu, X.; Zhou, J.; He, C.; Li, X.; Hu, B.; Li, J. A novel slurry for Cu films CMP in Ru-based Cu interconnects: Integration of experimental and theoretical calculations. Mater. Sci. Semicond. Process. 2026, 201, 110071. [Google Scholar] [CrossRef]
- Luo, F.; Niu, X.; Yan, H.; Zhang, Y.; Qu, M.; Zhu, Y.; Hou, Z. Synergistic effect of 1,2,4-triazole and phytic acid as inhibitors on copper film CMP for ruthenium-based copper interconnected and the surface action mechanism analysis. Mater. Sci. Semicond. Process. 2023, 156, 107276. [Google Scholar] [CrossRef]
- Gao, F.; Liang, H. Transformable oxidation of tantalum in electrochemical mechanical polishing (ECMP). J. Electron. Mater. 2011, 40, 134–140. [Google Scholar] [CrossRef]
- Lee, S.J.; Chen, Y.H.; Liu, C.P.; Fan, T.J. Electrochemical mechanical polishing of flexible stainless steel substrate for thin-film solar cells. Int. J. Electrochem. Sci. 2013, 8, 6878–6888. [Google Scholar] [CrossRef]
- Cojocaru, P.; Muscolino, F.; Magagnin, L. Effect of organic additives on copper dissolution for e-CMP. Microelectron. Eng. 2010, 87, 2187–2189. [Google Scholar] [CrossRef]
- Wu, Y.; Lei, R.; Jiang, L.; Qian, L. Achieving equivalent removal of Ta and Ru via controlling oxidation for chemical mechanical polishing of advanced barrier layer. Mater. Sci. Semicond. Process. 2024, 180, 108564. [Google Scholar] [CrossRef]
- Xu, Y.; Ma, T.; Liu, Y.; Tan, B.; Zhang, S.; Wang, Y.; Song, G. Effect of ethylenediamine on CMP performance of ruthenium in H2O2-based slurries. RSC Adv. 2021, 12, 228–240. [Google Scholar] [CrossRef]
- Wang, C.; Zhou, J.; Luo, C.; Wang, C.; Zhang, X. Synergist effect of potassium periodate and potassium persulfate on improving removal rate of Ruthenium during chemical mechanical polishing. Mater. Sci. Eng. B 2020, 262, 114764. [Google Scholar] [CrossRef]
- Bian, Y.-F.; Zhai, W.-J.; Cheng, Y.-Y. Electrolyte composition and galvanic corrosion for ruthenium/copper electrochemical mechanical polishing. Rare Met. 2020, 39, 1–7. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, T.; Wang, J.; Liu, Y.; Lu, X. Effects of KIO4 concentration and pH values of the solution relevant for chemical mechanical polishing of ruthenium. Microelectron. Eng. 2016, 151, 30–37. [Google Scholar] [CrossRef]
- Ma, H.; Zhou, J.; Wang, C.; Zhang, Y.; Li, Z.; Zhang, X.; Liu, G. Role of Diethylene Triaminepentaacetic Acid Pentapotassium Salt on Ruthenium CMP in H2O2-Based Slurries. ECS J. Solid State Sci. Technol. 2022, 11, 124002. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, T.; Mei, H.; Zhou, W.; Lu, X. Synergetic effect of potassium molybdate and benzotriazole on the CMP of ruthenium and copper in KIO4-based slurry. Appl. Surf. Sci. 2014, 320, 531–537. [Google Scholar] [CrossRef]
- Wang, C.; Wang, C.; Li, H.; Zhou, J.; Zhang, X.; Tian, Y.; Xu, C. Effect of UV Radiation on Oxidation for Ru CMP. ECS J. Solid State Sci. Technol. 2021, 10, 034007. [Google Scholar] [CrossRef]
- Di, H.; Zhou, P.; Lu, Y.; Yang, K.; Guo, D. Role of alumina particles in chemical-mechanical synergies in ruthenium polishing. J. Mater. Process. Technol. 2024, 330, 118477. [Google Scholar] [CrossRef]
- Shao, S.; Wu, B.; Wang, P.; He, P.; Qu, X.-P. Investigation on inhibition of ruthenium corrosion by glycine in alkaline sodium hypochlorite based solution. Appl. Surf. Sci. 2020, 506, 144976. [Google Scholar] [CrossRef]
- Zhan, N.; Niu, X.; Liu, J.; Zuo, Y.; Yan, H.; Luo, F.; Qu, M.; Xian, W.; Zhou, J. Surface Interaction Effect and Mechanism of Methionine Derivatives as Novel Inhibitors for Alkaline Copper CMP: Insights from Molecular Simulation and Experimental Analysis. ECS J. Solid State Sci. Technol. 2023, 12, 094001. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, J.; Niu, X.; Zhang, K.; Wang, Z.; Cui, Y.; Wang, R. Chemical Interactions and Mechanisms of Different pH Regulators on Copper and Cobalt Removal Rate of Copper Film CMP for GLSI. ECS J. Solid State Sci. Technol. 2019, 8, P99–P105. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, J.; Wang, C.; Niu, X.; Tian, Q.; Xiao, Y.; Wang, R. Controlling the Removal Rate Selectivity of Ruthenium to Copper during CMP by Using Guanidine Carbonate and 1,2,4-Triazole. ECS J. Solid State Sci. Technol. 2018, 7, P567–P574. [Google Scholar] [CrossRef]
- Kristin, G.S. An Electrochemical Investigation of the Removal and Planarization of Copper and Ruthenium. Ph.D. Thesis, Columbia University, New York, NY, USA, 2010. [Google Scholar]
- Yadav, K.; Manivannan, R.; Victoria, S.N. Electrochemical characterization of ruthenium dissolution and chemical mechanical polishing in hydrogen peroxide based slurries. Mater. Today Proc. 2019, 18, 1220–1228. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, T.; Jiang, L.; Lu, X. Surface characteristics of ruthenium in periodate-based slurry during chemical mechanical polishing. Appl. Surf. Sci. 2015, 351, 401–409. [Google Scholar] [CrossRef]
- Jiang, L.; He, Y.; Li, Y.; Luo, J. Effect of ionic strength on ruthenium CMP in H2O2-based slurries. Appl. Surf. Sci. 2014, 317, 332–337. [Google Scholar] [CrossRef]
- Tamboli, D.; Osso, J.O.; McEvoy, T.; Vega, L.F.; Rao, M.; Banerjee, G. Investigating the compatibility of ruthenium liners with copper interconnects. ECS Trans. 2010, 33, 181–187. [Google Scholar] [CrossRef]
- Yeung, H.; Chan, H.; Zou, S.Z.; Weaver, M.J. Mechanistic differences between electrochemical and gas-phase thermal oxidation of platinum-group transition metals as discerned by surface-enhanced raman spectroscopy. J. Phys. Chem. B 1999, 103, 11141–11151. [Google Scholar]
- Liu, J.; Lei, J.; Magtoto, N.; Garza, M.; Kelber, J.A. The effects of an iodine surface layer on Ru reactivity in air and during Cu electrodeposition. J. Electrochem. Soc. 2005, 152, G115–G121. [Google Scholar] [CrossRef]
- Lee, W.J.; Park, H.S. Development of novel process for Ru CMP using ceric ammonium nitrate (CAN)-containing nitric acid. Appl. Surf. Sci. 2004, 228, 410–417. [Google Scholar] [CrossRef]
- Ahadi, M.M.; Attar, M.M. OCP measurement: A method to determine CPVC. Sci. Iran. 2007, 14, 369–372. [Google Scholar]
- Kim, I.K.; Cho, B.G.; Park, J.G.; Park, J.Y.; Park, H.S. Effect of pH in Ru Slurry with Sodium Periodate on Ru CMP. J. Electrochem. Soc. 2009, 156, H188–H192. [Google Scholar] [CrossRef]
- Burke, L.D.; Whelan, D.P. The behaviour of ruthenium anodes in base. J. Electroanal. Chem. 1979, 103, 179–187. [Google Scholar] [CrossRef]
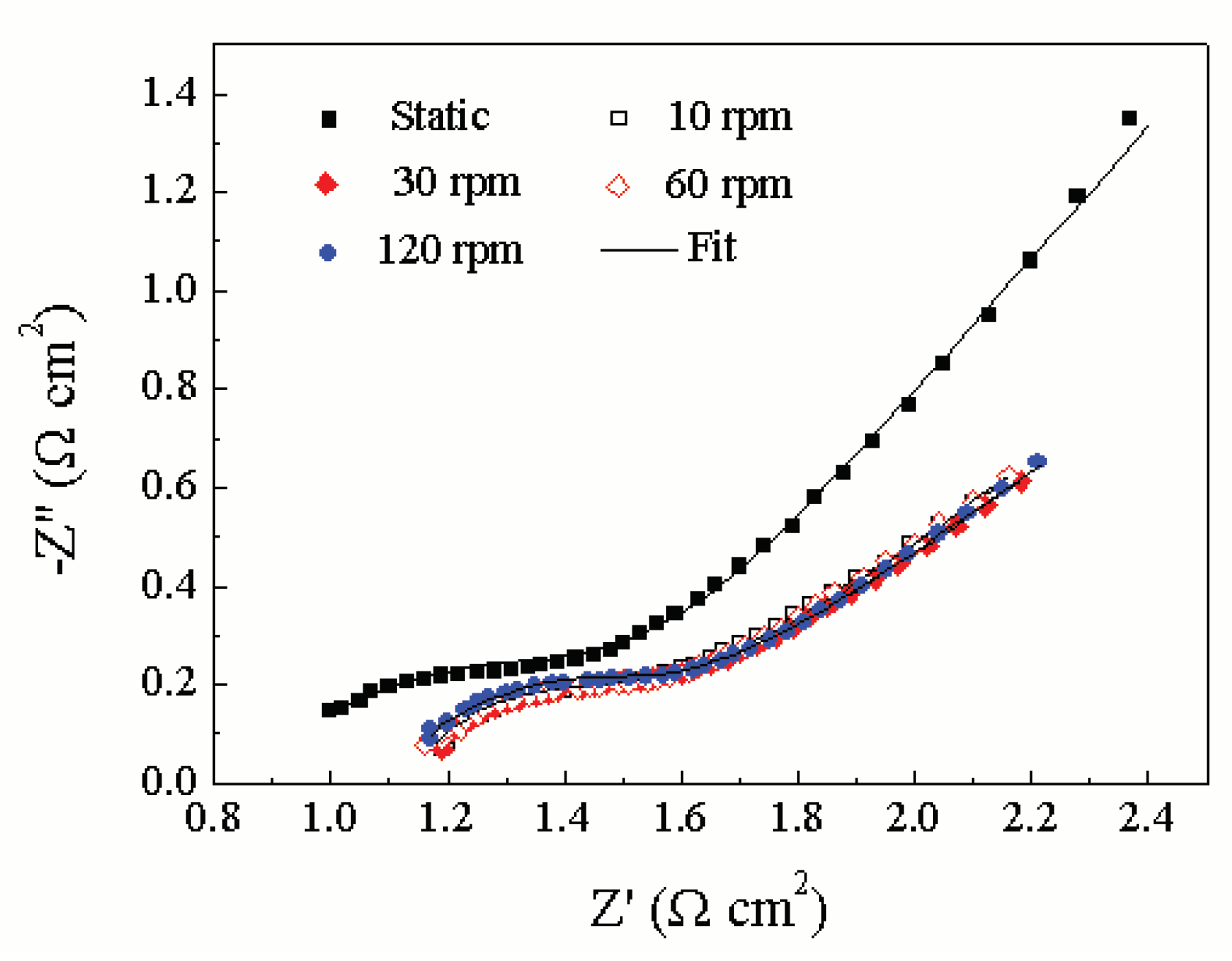
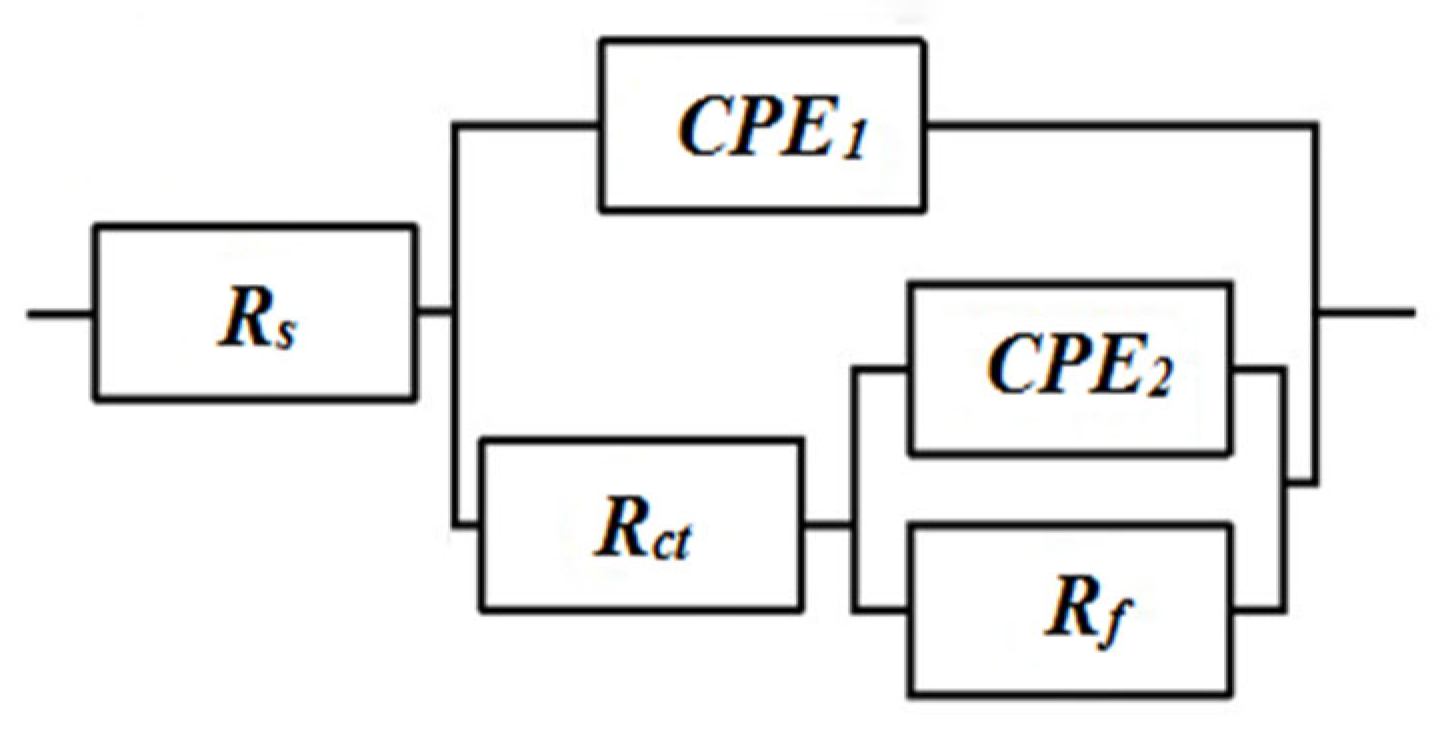

| CPE1 /(F·cm-2) | Rct /(Ω·cm2) | |
|---|---|---|
| Static | 0.0007916 (62.27) | 0.3541 (9.867) |
| 10 rpm | 0.0007312 (26.95) | 0.3604 (9.277) |
| 30 rpm | 0.0005719 (48.93) | 0.3777 (15.88) |
| 60 rpm | 0.0005682 (33.22) | 0.3814 (10.18) |
| 120 rpm | 0.0005098 (33.67) | 0.4272 (9.905) |
| Potential/V | C | N | O | Ru |
|---|---|---|---|---|
| No bias | – | – | 0.64 | 99.36 |
| 0.64 | 11.44 | 1.59 | 1.98 | 84.99 |
| 1.0 | 11.84 | 2.67 | 2.79 | 82.70 |
| 2.0 | 5.48 | 10.27 | 20.61 | 63.64 |
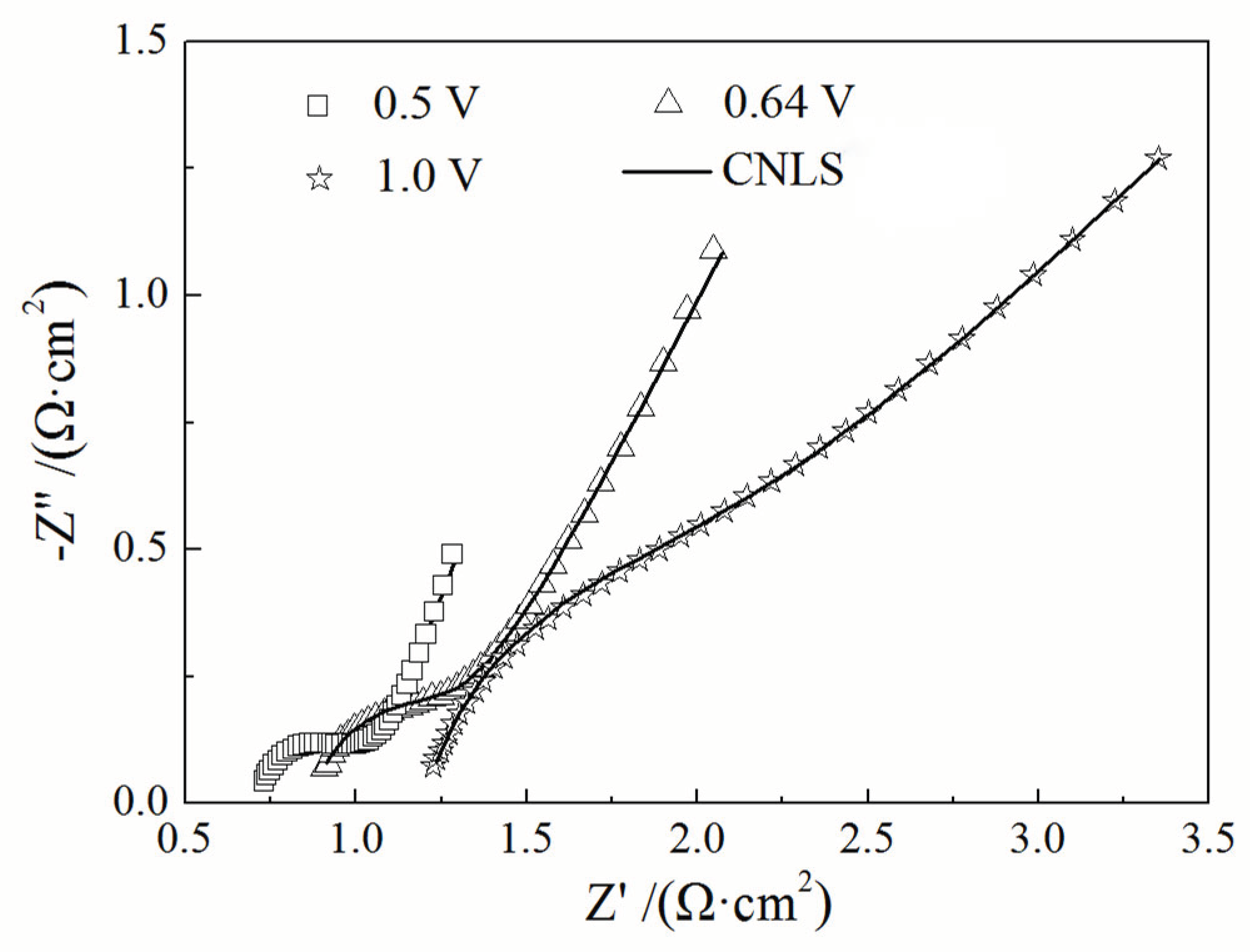
| Potential / (V vs. Pt) | CPE1 /(F·cm-2) | Rct /(Ω·cm2) |
|---|---|---|
| 0.5 | 0.002563 | 0.3746 |
| 0.64 | 0.001355 | 0.4915 |
| 1.0 | 0.0007592 | 0.6590 |
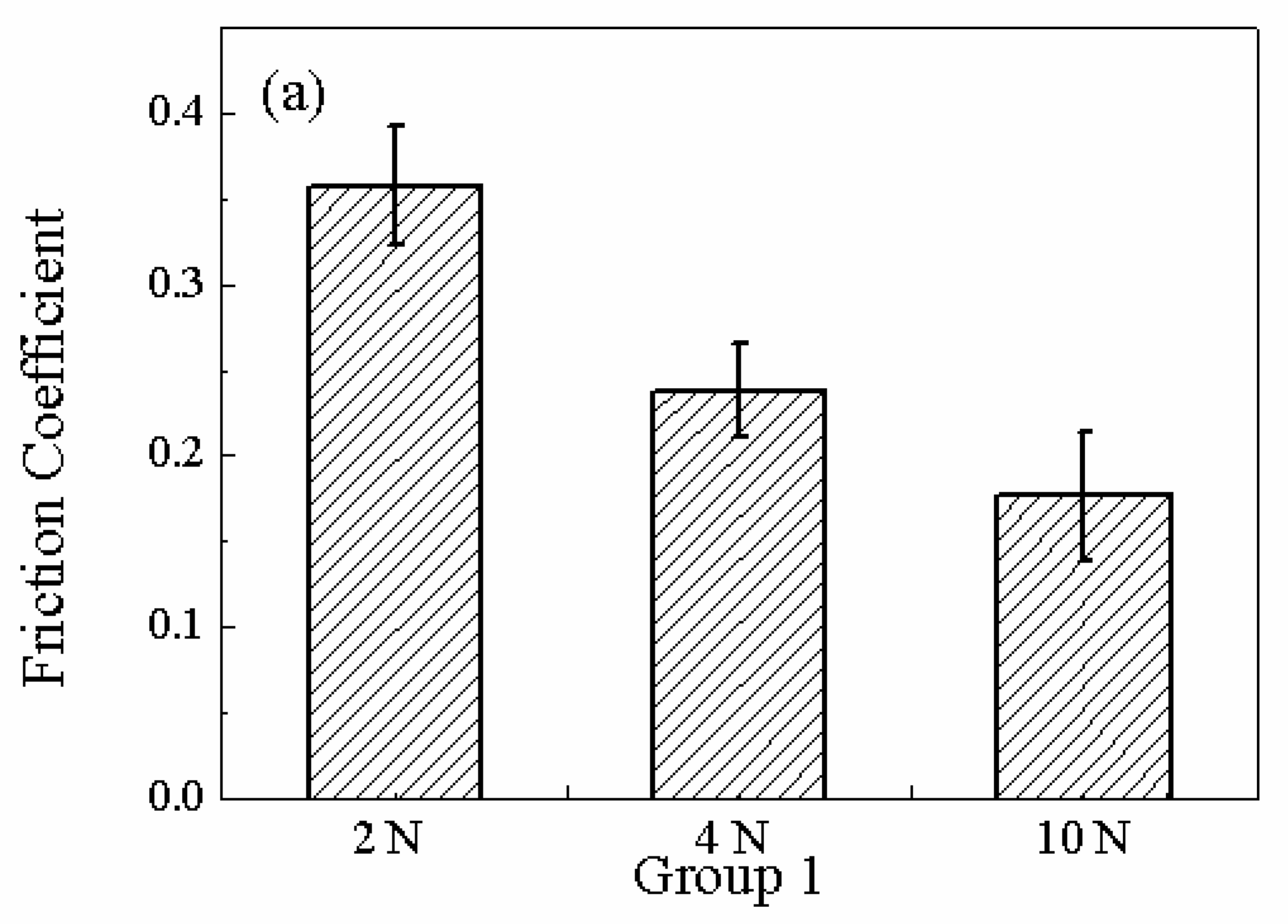
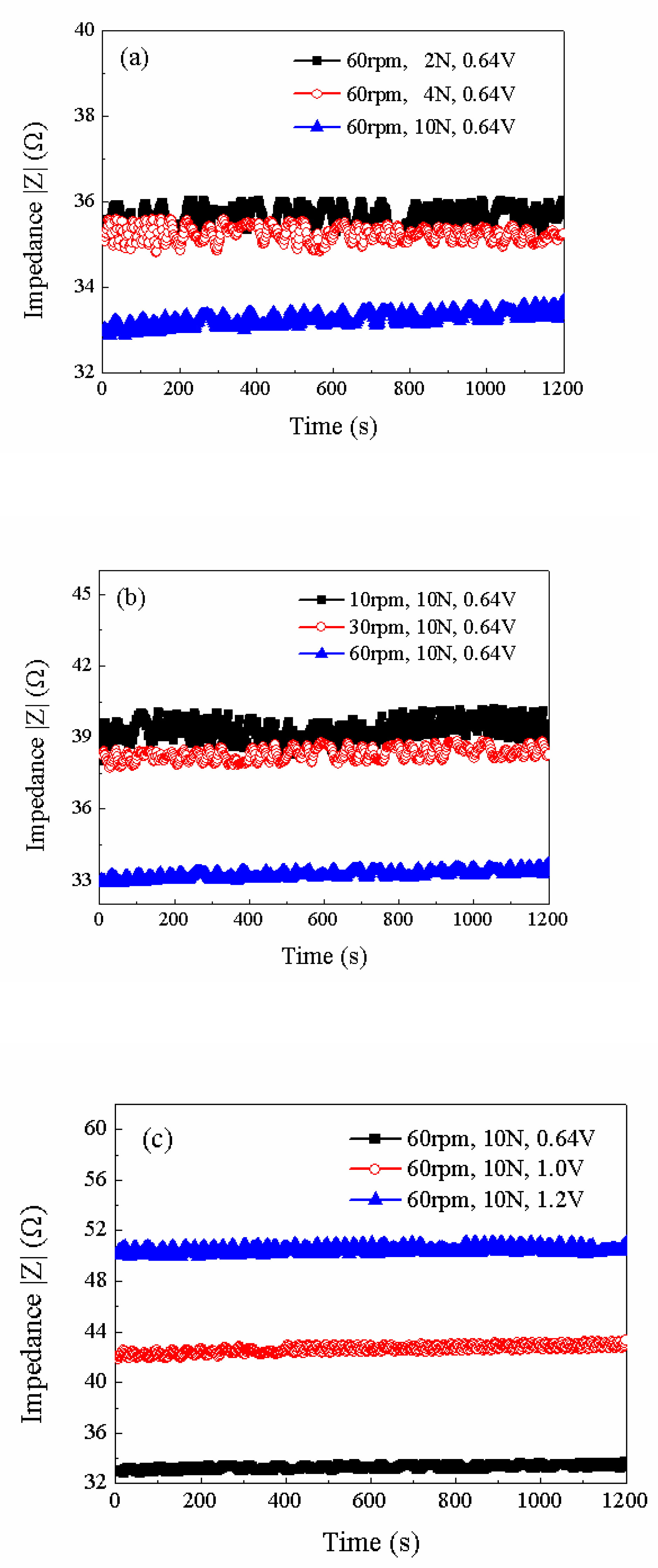
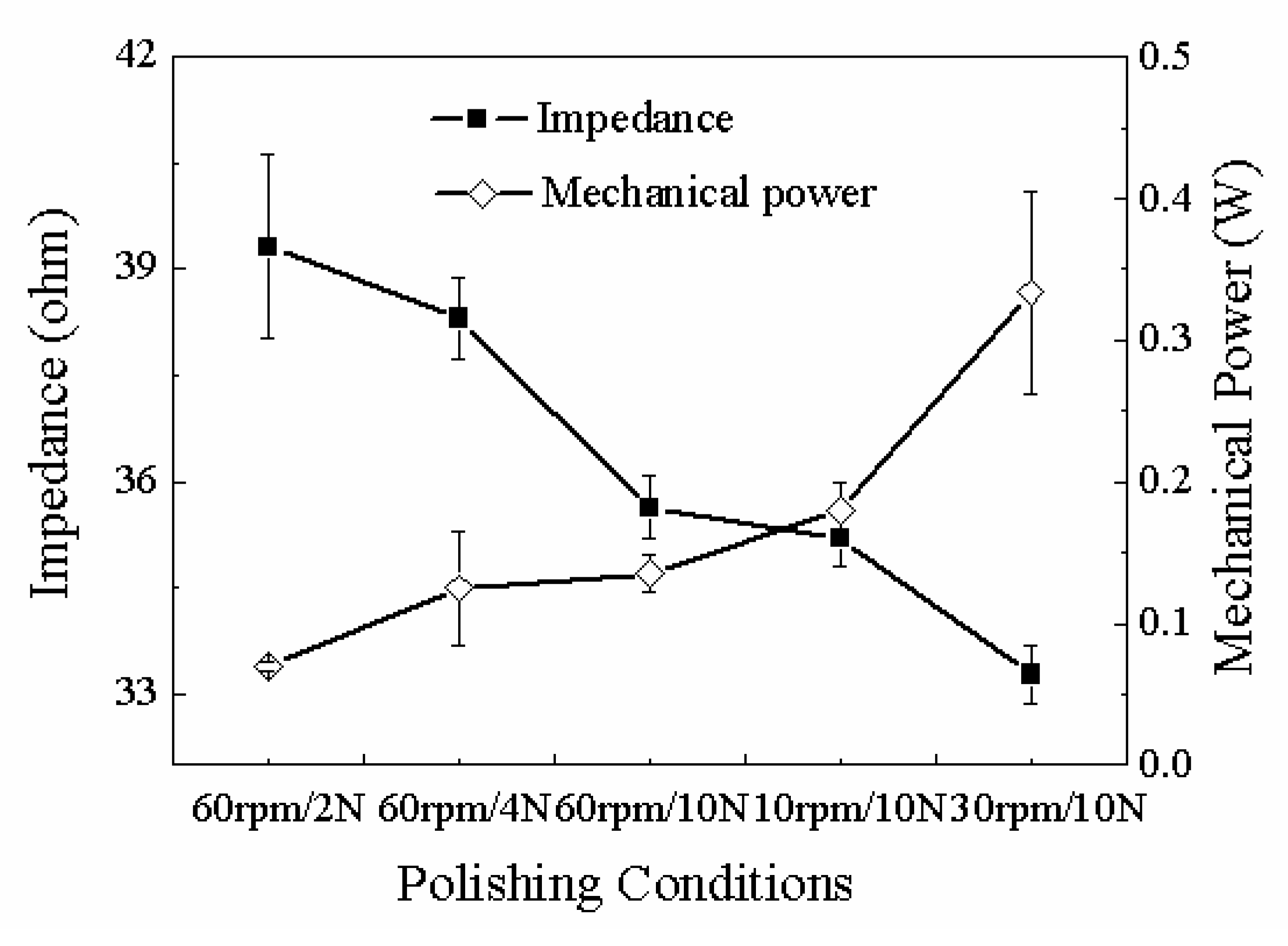
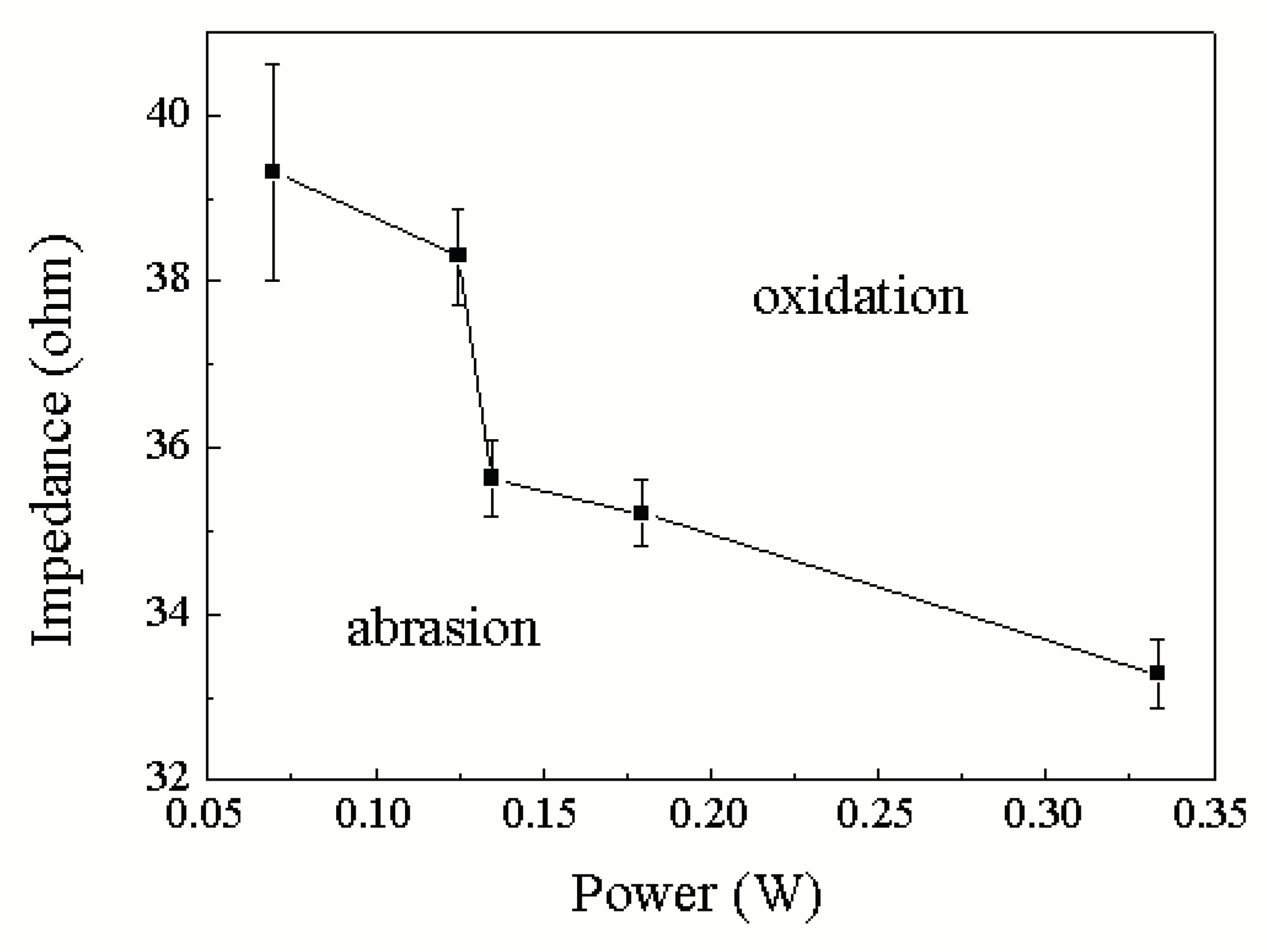
| Group | Fixed parameters | Variables | Average impedance(Ω) | Power(W) |
|---|---|---|---|---|
| 1 | 60rpm/0.64V | 2N | 35.6366±0.4466 | 0.1350±0.0132 |
| 4N | 35.2091±0.4 | 0.1794±0.0202 | ||
| 10N | 33.2810±0.4190 | 0.3333±0.0711 | ||
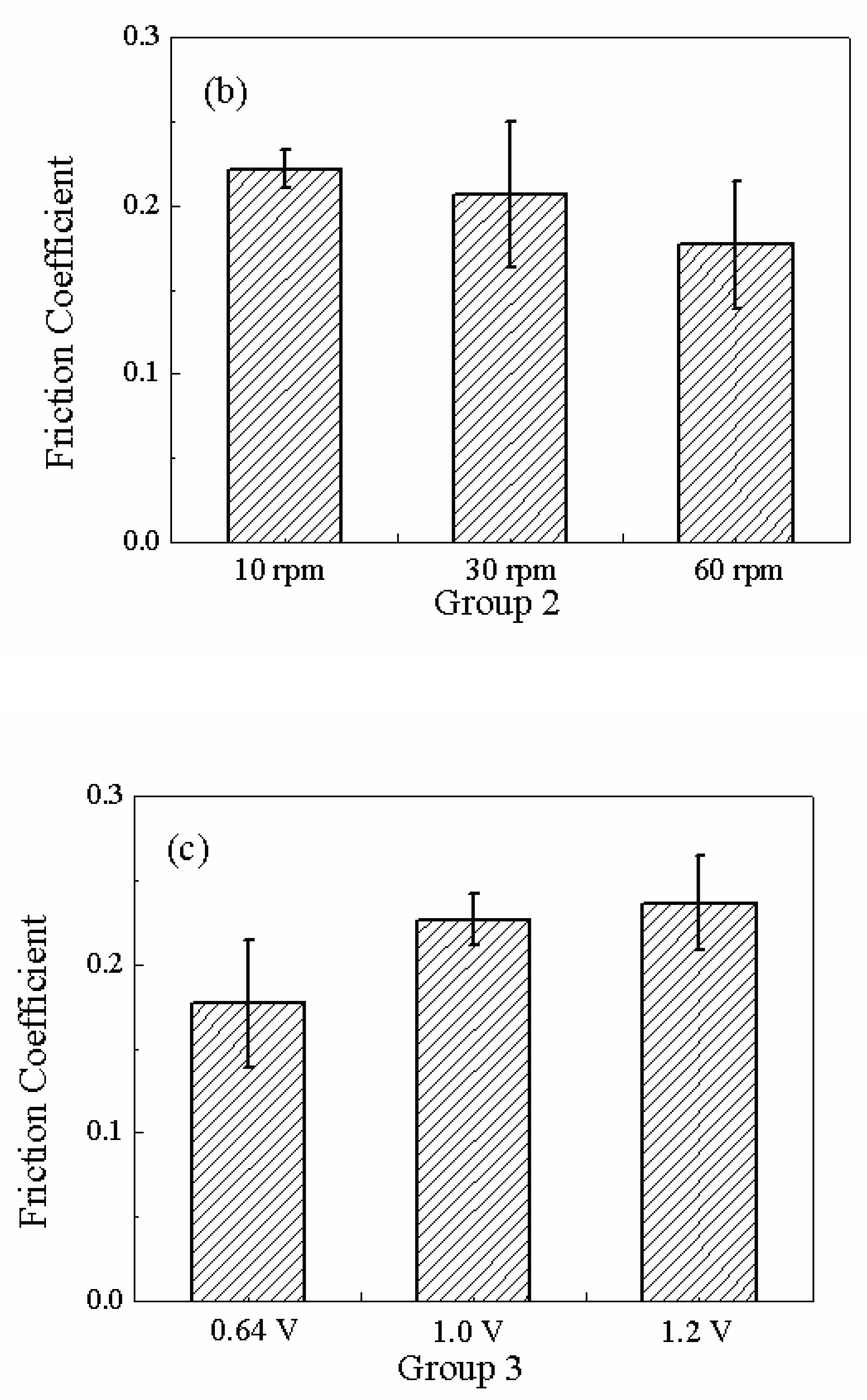
| 2 | 10N/0.64V | 10rpm | 39.3130±1.293 | 0.0697±0.0035 |
| 30rpm | 38.3030±0.5830 | 0.1249±0.0405 | ||
| 60rpm | 33.2810±0.4190 | 0.3333±0.0711 | ||
| 3 | 60rpm/10N | 0.64V | 33.2810±0.4190 | 0.3333±0.0711 |
| 1.0V | 42.6213±0.7113 | 0.4275±0.0283 | ||
| 1.2V | 50.5326±0.5626 | 0.4464±0.0539 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




