Submitted:
03 November 2025
Posted:
05 November 2025
You are already at the latest version
Abstract
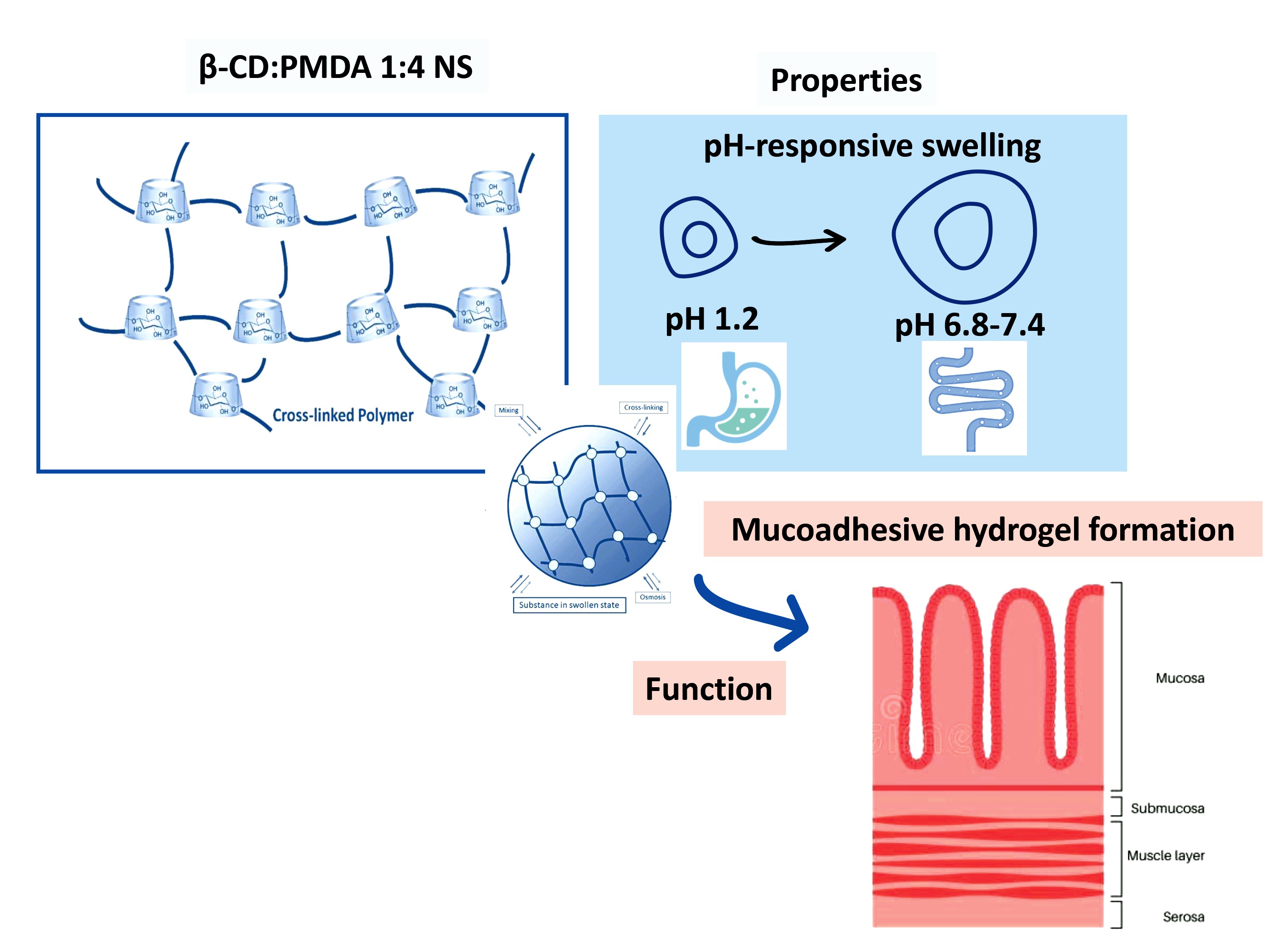
Keywords:
1. Introduction
2. Results and Discussion
2.1. Confirmation of Cross-Linking (FTIR and TGA)
2.2. Particle Size and Surface Charge
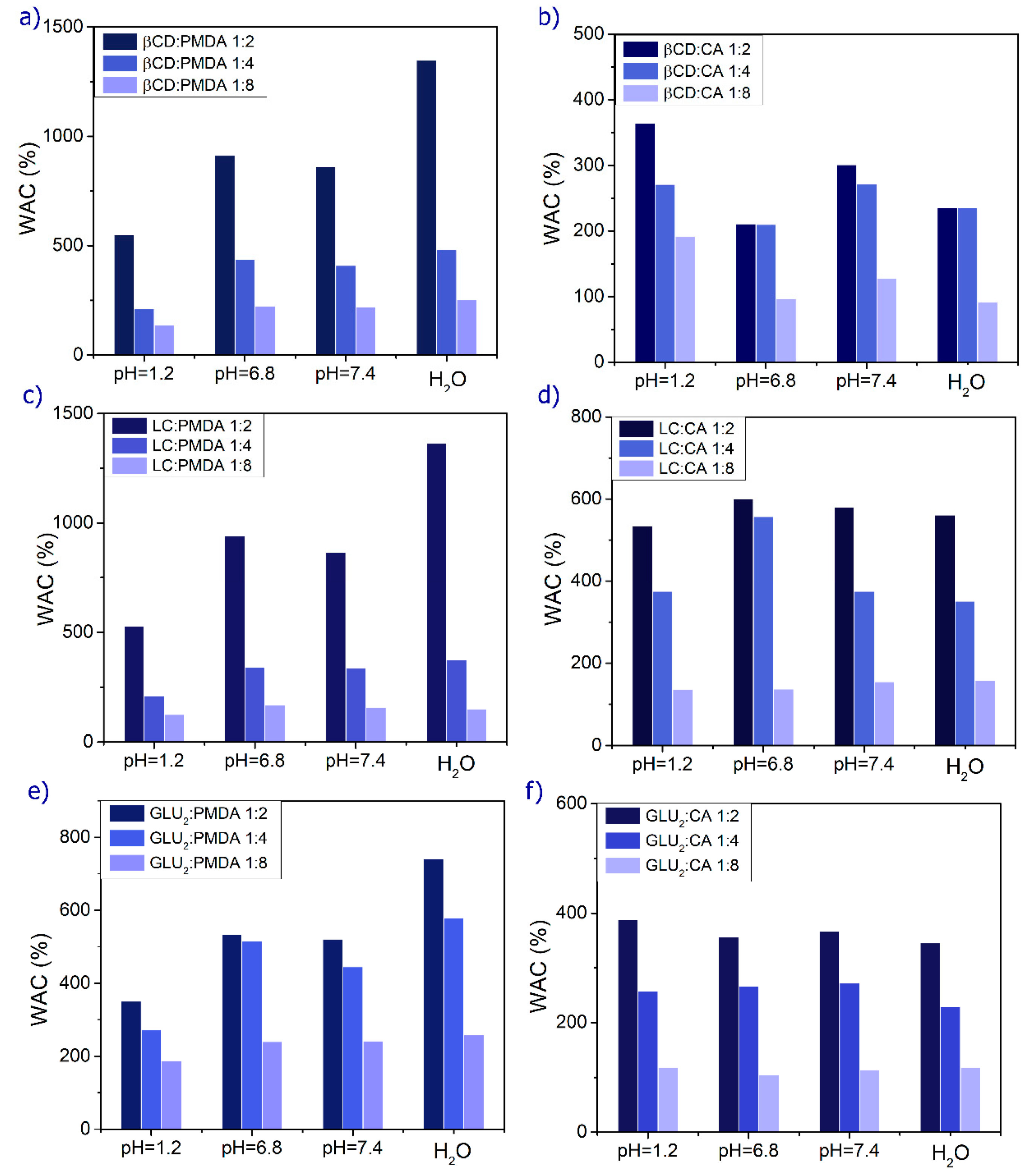
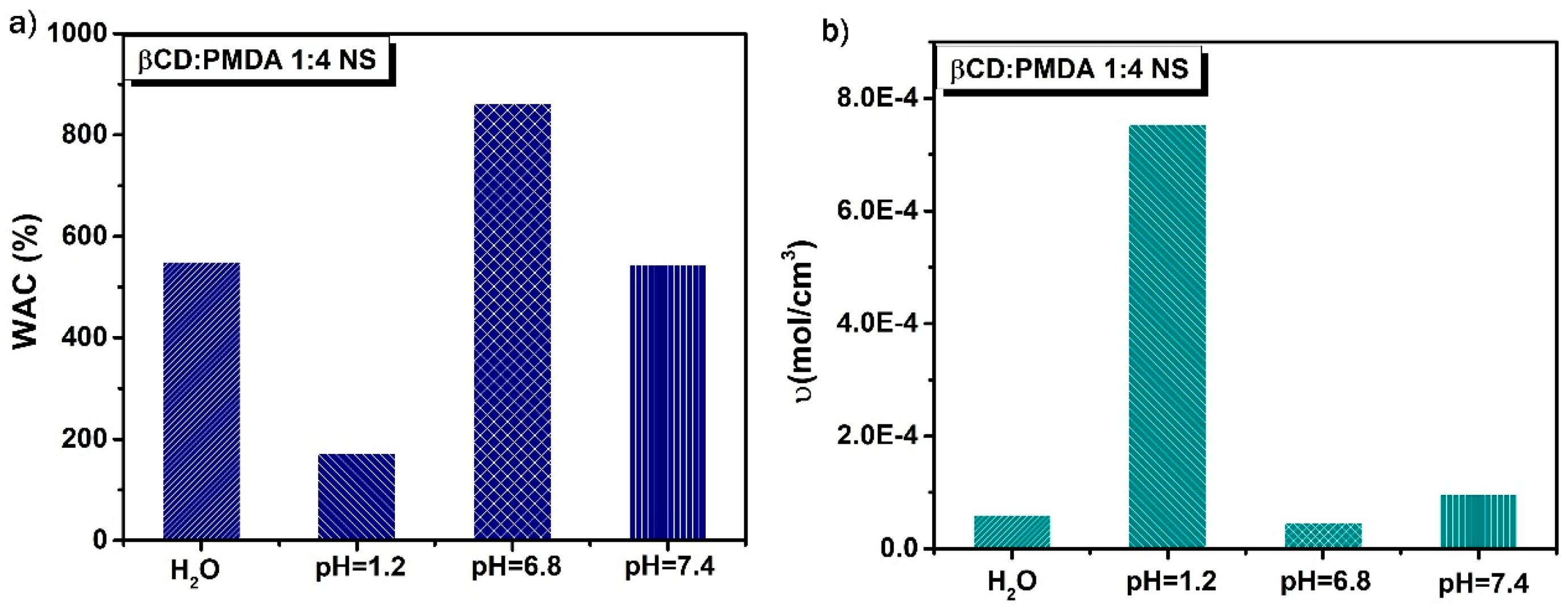
2.3. Swelling Behavior at Different pH
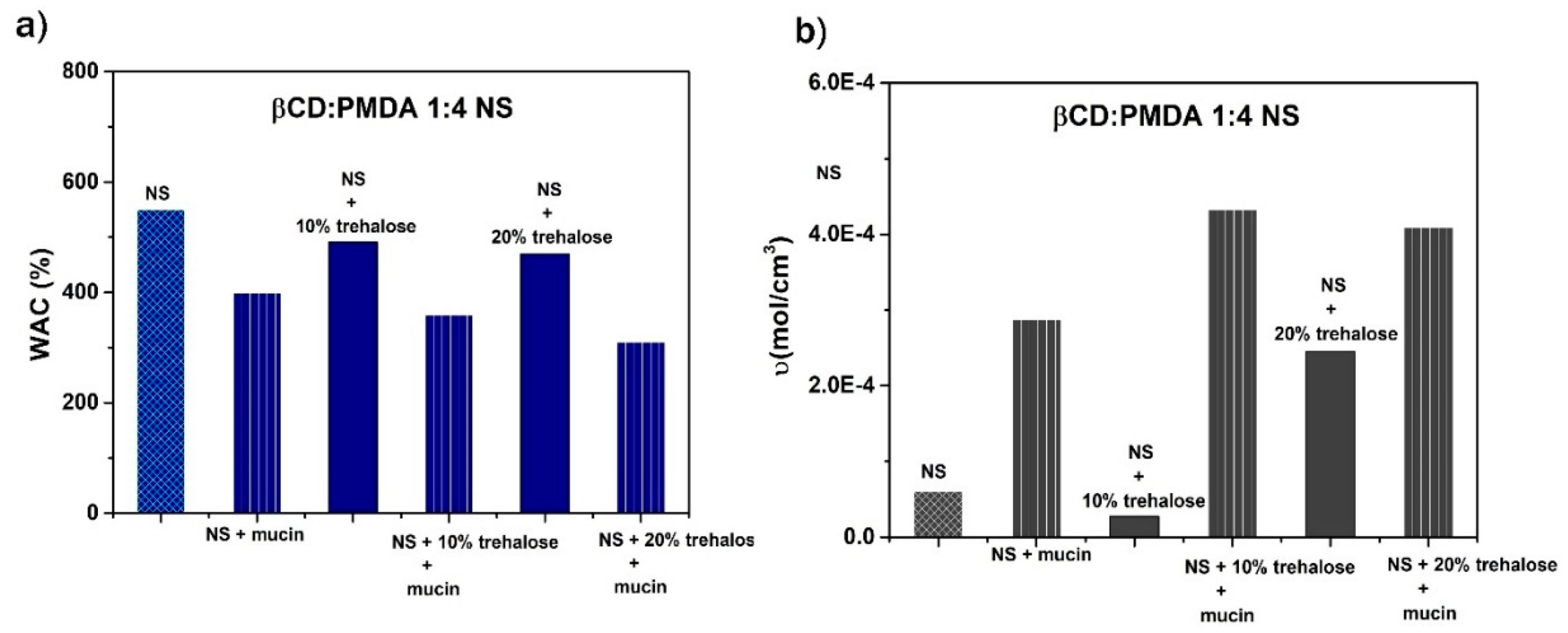
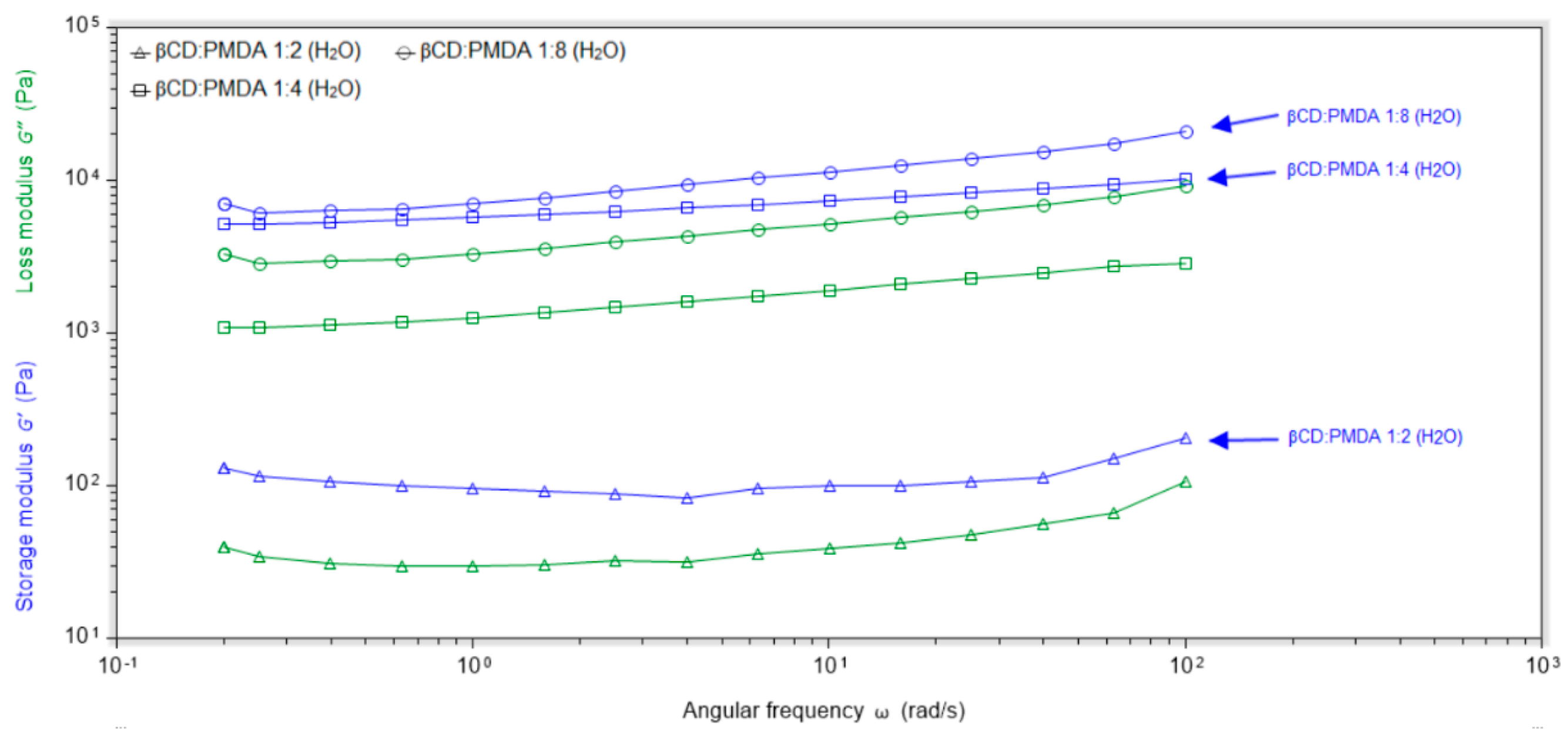
2.4. Cross-Linking Density and Network Properties
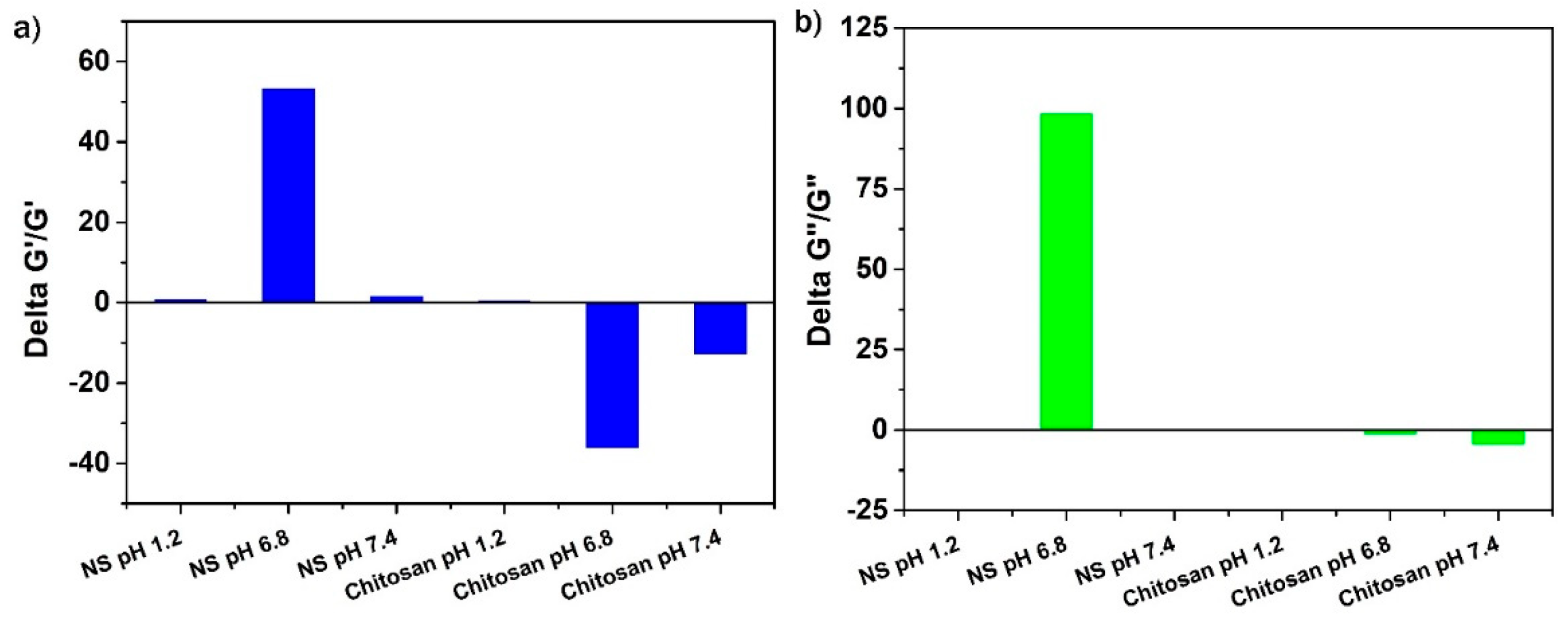
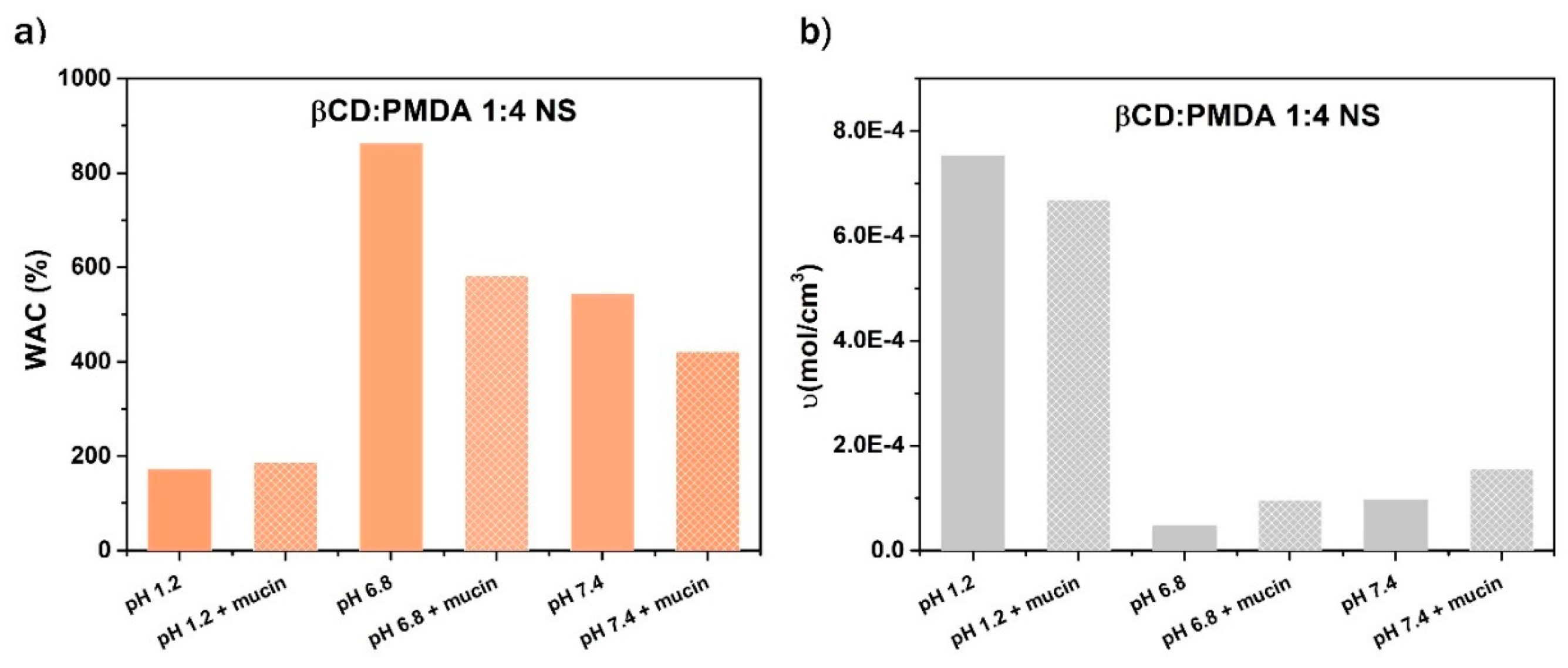
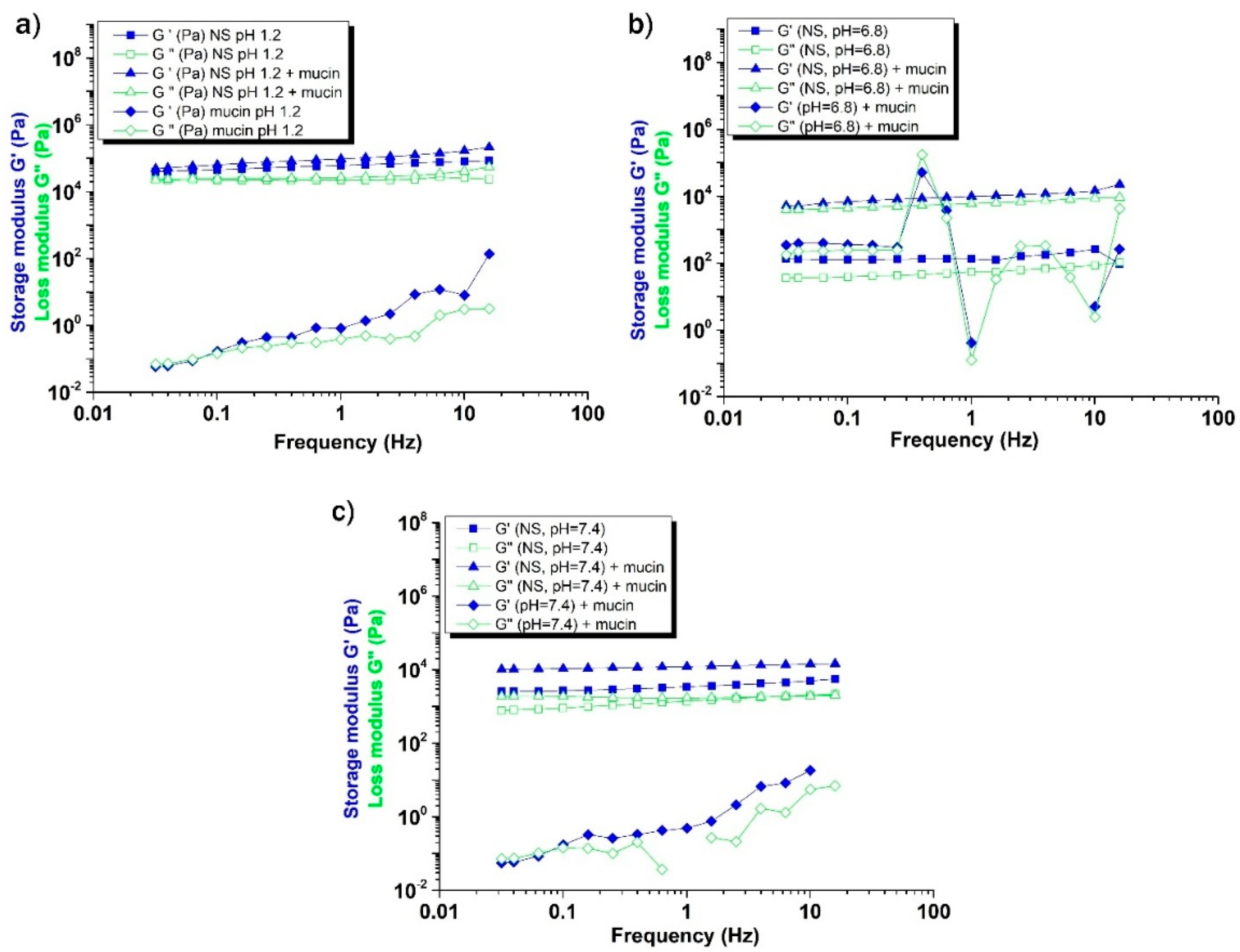
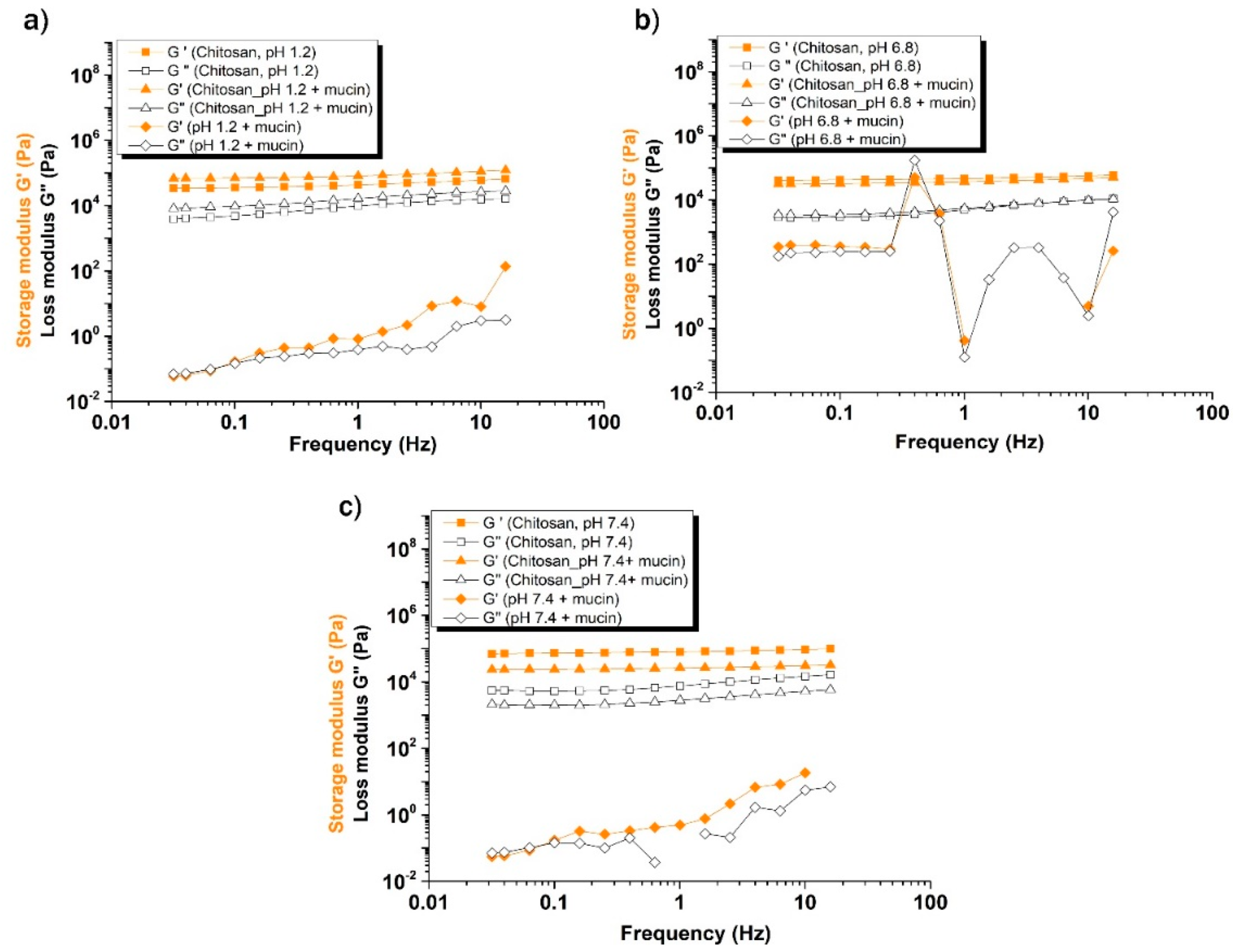
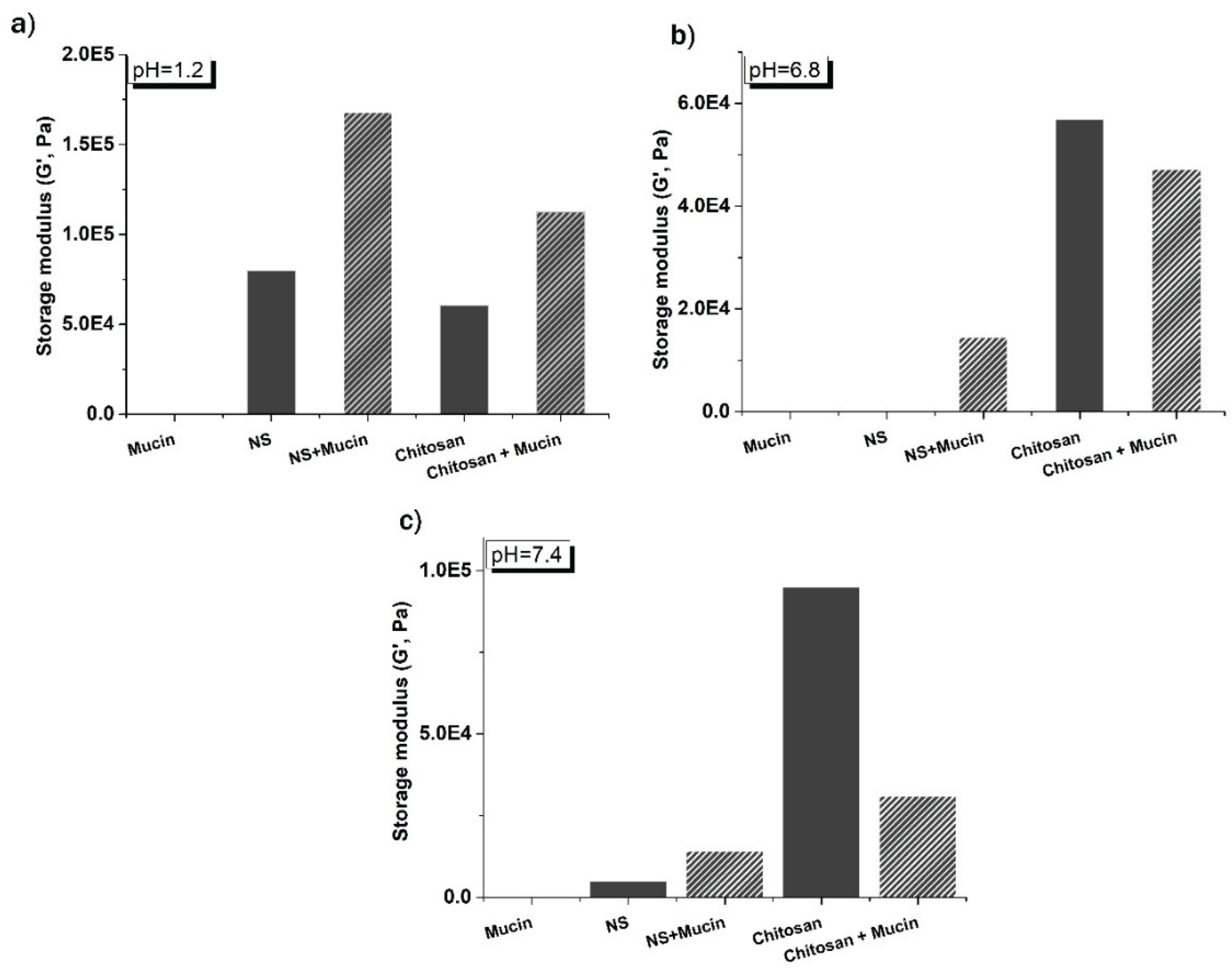
2.5. Mucoadhesive Behavior and Polymer–Mucin Interactions
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Synthesis of Dextrin-Based Polymers
4.2.1. Synthesis of PMDA-Based D-NS
4.2.2. Synthesis of CA-Based D-NS
4.3. Characterization
4.4. Swelling Studies
4.5. Preparation of Mucin Samples
4.6. Cross-Linking Density Determination
4.7. Rheological Analysis
4.8. Mucoadhesion Studies
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| β-CD | β-Cyclodextrin |
| LC | KLEPTOSE® Linecaps |
| GLU2 | GluciDex®2 |
| CA | Citric Acid |
| PMDA | Pyromellitic Dianhydride |
| GI | Gastrointestinal |
| CDs | Cyclodextrins |
| D-NS | Dextrin-based Nanosponges |
| NS | Nanosponge |
| AD | Alzheimer’s disease |
| PD | Parkinson’s disease |
| DMSO | Dimethylsulfoxide |
| Et₃N | Triethylamine |
| SHP | Sodium Hypophosphite Monohydrate |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| TGA | Thermogravimetric Analysis |
| DLS | Dynamic Light Scattering |
| N2 | Nitrogen |
| S | Swelling |
| WAC | Water Absorption Capacity |
| G | Shear Modulus |
| G’ | Storage Modulus |
| G” | Loss Modulus |
| ΔG’ | Rheological Synergism |
| ΔG’/G’ | Relative Rheological Synergism |
| -COOH | Carboxylic Acid Groups |
| PDI | Polydispersity Index |
| Z-Ave | Z-average |
| HPH | High-Pressure Homogenization |
References
- J. Lou et al., “Advances in Oral Drug Delivery Systems: Challenges and Opportunities,” Pharmaceutics, vol. 15, no. 484, pp. 1–22, 2023.
- D. Vllasaliu, “Oral administration.pdf,” Front. Drug Deliv., pp. 1–3, 2025.
- J. Reinholz, K. Landfester, and V. Mailänder, “The challenges of oral drug delivery via nanocarriers,” Drug Deliv., vol. 25, no. 1, pp. 1694–1705, 2018.
- S. Bashiardes and C. Christodoulou, “Orally Administered Drugs and Their Complicated Relationship with Our Gastrointestinal Tract,” Microorganisms, vol. 12, no. 242, pp. 1–18, 2024.
- M. Azman, A. H. Sabri, Q. K. Anjani, M. F. Mustaffa, and K. A. Hamid, “Intestinal Absorption Study: Challenges and Absorption Enhancement Strategies in Improving Oral Drug Delivery,” Pharmaceuticals, vol. 15, no. 8, pp. 1–24, 2022.
- R. Kumar, T. Islam, and M. Nurunnabi, “Mucoadhesive carriers for oral drug delivery,” J Control Release, vol. 351, pp. 504–559, 2022.
- S. Alaei and H. Omidian, “Mucoadhesion and Mechanical Assessment of Oral Films,” Eur. J. Pharm. Sci., vol. 159, pp. 1–17, 2021.
- J. Bagan et al., “Mucoadhesive Polymers for Oral Transmucosal Drug Delivery: A Review,” Curr. Pharm. Des., vol. 18, no. 34, pp. 5497–5514, 2012.
- R. Sharma et al., “Recent advances in biopolymer-based mucoadhesive drug delivery systems for oral application,” J. Drug Deliv. Sci. Technol., vol. 91, pp. 1–18, 2024.
- J. D. Smart, “The basics and underlying mechanisms of mucoadhesion,” Adv. Drug Deliv. Rev., vol. 57, no. 11, pp. 1556–1568, 2005.
- B. Chatterjee, N. Amalina, P. Sengupta, and U. K. Mandal, “Mucoadhesive Polymers and Their Mode of Action: A Recent Update,” J. Appl. Pharm. Sci., vol. 7, no. 5, pp. 195–203, 2017.
- Z. Jawadi, C. Yang, Z. S. Haidar, P. L. Santa Maria, and S. Massa, “Bio-Inspired Muco-Adhesive Polymers for Drug Delivery Applications,” Polymers (Basel)., vol. 14, pp. 1–28, 2022.
- B. M. Boddupalli, Z. N. K. Mohammed, R. Nath A., and D. Banji, “Mucoadhesive drug delivery system: An overview,” J. Adv. Pharm. Technol. Res., vol. 1, no. 4, pp. 381–387, 2010.
- L. Serra, J. Doménech, and N. Peppas, “Engineering Design and Molecular Dynamics of Mucoadhesive Drug Delivery Systems as Targeting Agents,” Eur J Pharm Biopharm, vol. 71, no. 3, pp. 1–7, 2009.
- Z. Davoudi, G. Kali, D. Braun, M. H. Azizi, and A. Bernkop-Schnürch, “Highly thiolated corn starch for enhanced mucoadhesion and permeation,” Int. J. Pharm., vol. 680, pp. 1–12, 2025.
- K. Ahmad et al., “Enhancing mucoadhesion: Exploring rheological parameters and texture profile in starch solutions, with emphasis on micro-nanofiber influence,” Int. J. Biol. Macromol., vol. 275, no. 1299, pp. 1–19, 2024.
- D. R. Lu, C. M. Xiao, and S. J. Xu, “Starch-based completely biodegradable polymer materials,” Express Polym. Lett., vol. 3, no. 6, pp. 366–375, 2009.
- <i>18. </i>B. D. Ulery, L. S. B. D. Ulery, L. S. Nair, and C. T. Laurencin, “Biomedical Applications of Biodegradable Polymers Bret,” J Polym Sci B Polym Phys, vol. 49, no. 12, pp. 832–864, 2011.
- T. Loftsson and M. E. Brewster, “Pharmaceutical Applications of Cyclodextrins. 1. Drug Solubilization and Stabilization,” J. Pharm. Sci., vol. 85, no. 10, pp. 1017–1025, 1996.
- G. Kali, S. Haddadzadegan, and A. Bernkop-Schnürch, “Cyclodextrins and derivatives in drug delivery: New developments, relevant clinical trials, and advanced products,” Carbohydr. Polym., vol. 324, pp. 1–23, 2024.
- Z. Liu, L. Ye, J. Xi, J. Wang, and Z. Feng, “Cyclodextrin polymers: Structure, synthesis, and use as drug carriers,” Prog. Polym. Sci., vol. 118, pp. 1–24, 2021.
- M. Agnes, E. Pancani, M. Malanga, E. Fenyvesi, and I. Manet, “Implementation of Water-Soluble Cyclodextrin-Based Polymers in Biomedical Applications: How Far Are We?,” Macromol. Biosci., vol. 22, no. 8, pp. 1–26, 2022.
- P. Sherje, B. R. Dravyakar, D. Kadam, and M. Jadhav, “Cyclodextrin-based nanosponges: A critical review,” Carbohydr. Polym., vol. 173, no. 1, pp. 37–49, 2017.
- Krabicová et al., “History of cyclodextrin nanosponges,” Polymers (Basel)., vol. 12, no. 5, pp. 1–23, 2020.
- F. Trotta, M. Zanetti, and R. Cavalli, “Cyclodextrin-based nanosponges as drug carriers,” Beilstein J. Org. Chem., vol. 8, pp. 2091–2099, 2012.
- G. Hoti et al., “Effect of the Cross-linking Density on the Swelling and Rheological Behavior of Ester-Bridged β-Cyclodextrin Nanosponges,” Materials (Basel)., vol. 14, no. 3, pp. 1–20, 2021.
- Y.-J. Wang and L. Wang, “Structures and Properties of Commercial Maltodextrins from Corn, Potato, and Rice Starches,” Starch/Staerke, vol. 52, no. 7–8, pp. 296–304, 2000.
- M. Münster et al., “Comparative in vitro and in vivo taste assessment of liquid praziquantel formulations,” Int. J. Pharm., vol. 529, no. 1–2, pp. 310–318, 2017.
- D. G. Gadhave et al., “Neurodegenerative disorders: Mechanisms of degeneration and therapeutic approaches with their clinical relevance,” Ageing Res. Rev., vol. 99, 2024.
- G. Hoti et al., “A Comparison between the Molecularly Imprinted and Non- Molecularly Imprinted Cyclodextrin- Based Nanosponges for the Transdermal Delivery of Melatonin,” Polymers (Basel)., vol. 15, no. 1543, pp. 1–27, 2023.
- F. Madsen, K. Eberth, and J. D. Smart, “A rheological examination of the mucoadhesive/mucus interaction: the effect of mucoadhesive type and concentration,” J. Control. Release, vol. 50, no. 1–3, pp. 167–178, 1998.
- P. Sriamornsak and N. Wattanakorn, “Rheological synergy in aqueous mixtures of pectin and mucin,” Carbohydr. Polym., vol. 74, no. 3, pp. 474–481, 2008.
- W. Wang, J. Wang, Y. Kang, and A. Wang, “Synthesis, swelling and responsive properties of a new composite hydrogel based on hydroxyethyl cellulose and medicinal stone,” Compos. Part B, vol. 42, no. 4, pp. 809–818, 2011.
- T. M. FitzSimons, E. V. Anslyn, and A. M. Rosales, “Effect of pH on the Properties of Hydrogels Cross - Linked via Dynamic Thia-Michael Addition Bonds,” ACS Polym. Au, vol. 2, no. 2, pp. 129–136, 2022.
- M. A. Güler, M. K. Gök, A. K. Figen, and S. Özgümüş, “Swelling, mechanical and mucoadhesion properties of Mt/starch-g-PMAA nanocomposite hydrogels,” Appl. Clay Sci., vol. 112–113, pp. 44–52, 2015.
- H. M. Yildiz, L. H. M. Yildiz, L. Speciner, C. Ozdemir, D. E. Cohen, and R. L. Carrier, “Food-associated Stimuli Enhance Barrier Properties of Gastrointestinal Mucus,” Biomaterials, vol. 54, pp. 1–8, 2015.
- E. Y. Chen, D. Daley, Y.-C. Wang, M. Garnica, C.-S. Chen, and W.-C. Chin, “Functionalized carboxyl nanoparticles enhance mucus dispersion and hydration,” Sci. Rep., vol. 2, 2012.
- M. Collado-González, Y. G. Espinosa, and F. M. Goycoolea, “Interaction Between Chitosan and Mucin: Fundamentals and Applications,” Biomimetics, vol. 4, no. 32, pp. 1–20, 2019.
- M. Ahmad, C. Ritzoulis, W. Pan, and J. Chen, “Biologically-relevant interactions, phase separations and thermodynamics of chitosan–mucin binary systems,” Process Biochem., vol. 94, no. December 2019, pp. 152–163, 2020.
- S. Rossi, F. Ferrari, M. C. Bonferoni, and C. Caramella, “Characterization of chitosan hydrochloride-mucin rheological interaction: influence of polymer concentration and polymer:mucin weight ratio,” Eur. J. Pharm. Sci., vol. 12, no. 4, pp. 479–485, 2001.
- T. M. M. Ways, W. M. Lau, and V. V. Khutoryanskiy, Chitosan and its derivatives for application in mucoadhesive drug delivery systems, vol. 10. 2018.
- M. K. Amin and J. S. Boateng, “Enhancing Stability and Mucoadhesive Properties of Chitosan Nanoparticles by Surface Modification with Sodium Alginate and Polyethylene Glycol for Potential Oral Mucosa Vaccine Delivery,” Mar. Drugs, vol. 20, pp. 1–22, 2022.
- V. M. de Oliveira Cardoso, M. P. D. Gremião, and B. S. F. Cury, “Mucin-polysaccharide interactions: A rheological approach to evaluate the effect of pH on the mucoadhesive properties,” Int. J. Biol. Macromol., vol. 149, pp. 234–245, 2020.
- R. G. Riley et al., “An investigation of mucus / polymer rheological synergism using synthesised and characterised poly (acrylic acid)s,” Int. J. Pharm., vol. 217, pp. 87–100, 2001.

| Samples | ZP (mV) |
| GLU2:CA 1:2 | 0.96 |
| GLU2:CA 1:4 | 0.25 |
| GLU2:CA 1:8 | 1.55 |
| LC: CA 1:2 | 1.56 |
| LC: CA 1:4 | 2.08 |
| LC: CA 1:8 | 1.80 |
| β-CD: CA 1:2 | 0.80 |
| β-CD: CA 1:4 | 0.23 |
| β-CD: CA 1:8 | 1.49 |
| β-CD: PMDA 1:2 | 9.49 |
| β-CD: PMDA 1:4 | 0.32 |
| β-CD: PMDA 1:8 | 1.14 |
| LC: PMDA 1:2 | 1.25 |
| LC:PMDA 1:4 | 0.80 |
| LC:PMDA 1:8 | 0.98 |
| GLU2:PMDA 1:2 | 0.65 |
| GLU2:PMDA 1:4 | 0.69 |
| GLU2:PMDA 1:8 | 0.15 |
| Samples |
Z-Ave (d.nm) |
PdI | Pk 1 Mean Int (d.nm) | Pk 2 Mean Int (d.nm) |
| GLU2:CA 1:2 | 3341.0153.4 | 0.382 0.006 | 1848.0 172.9 | 5444 200.3 |
| GLU2:CA 1:4 | 2512.0237.4 | 0.464 0.037 | 2120.0 804.0 | 5167 45.54 |
| GLU2:CA 1:8 | 1644.073.2 | 0.820 0.241 | 1061.0 42.2 | 1851 2868 |
| LC: CA 1:2 | 666.310.8 | 0.485 0.01 | 1463.0 401.5 | 262.7 149.3 |
| LC: CA 1:4 | 2094.099.5 | 0.428 0.055 | 1594.0 386 | 3529 2903 |
| LC: CA 1:8 | 965.654.7 | 0.639 0.107 | 1959.0 874.7 | 1694 2177 |
| β-CD: CA 1:2 | 2145.087.1 | 0.514 0.040 | 2469.0 1137.0 | 3634 2241 |
| β-CD: CA 1:4 | 2507.055.4 | 0.322 0.039 | 1910 83.7 | 5503 52.37 |
| β-CD: CA 1:8 | 2097.0221.1 | 0.481 0.074 | 1987 552.7 | 182.5 21.34 |
| β-CD: PMDA 1:2 | 742.5103.2 | 0.757 0.156 | 667 63.26 | 136.4 2.689 |
| β-CD: PMDA 1:4 | 790.67.1 | 0.496 0.025 | 1511 441.3 | 243 134.2 |
| β-CD: PMDA 1:8 | 1141.025.4 | 0.468 | 2017 478.9 | 344.5 145.4 |
| LC:PMDA 1:2 | 581.99.8 | 0.422 0.009 | 837.3 91.92 | 1664 2550 |
| LC:PMDA 1:4 | 762.413.9 | 0.358 0.058 | 1247 206.6 | 212.7 35.88 |
| LC:PMDA 1:8 | 126743.8 | 0.544 0.042 | 1716 1265 | 3159 2385 |
| GLU2:PMDA 1:2 | 1212.035.0 | 0.498 0.031 | 2035 683.7 | 278.2 172.2 |
| GLU2:PMDA 1:4 | 804.048.0 | 0.571 0.010 | 2557 795 | 381.6 330.7 |
| GLU2:PMDA 1:8 | 964.570.3 | 0.872 0.012 | 1036 120.8 | 247.8 81.34 |
| Samples |
Z-Ave±SD (nm) |
PDI | Zeta Potential ±SD (mV) |
| GLU2:CA 1:4 | 186.60 ± 2.54 | 0.25 ± 0.02 | -34.31 ± 0.78 |
| LC: CA 1:4 | 298.90 ± 9.08 | 0.21 ± 0.03 | -28.58 ± 0.52 |
| β-CD: CA 1:4 | 441.70 ± 9.87 | 0.26 ± 0.02 | -22.75 ± 0.43 |
| β-CD: PMDA 1:4 | 286.60 ± 3.94 | 0.20 ± 0.01 | -30.21 ± 0.75 |
| LC:PMDA 1:4 | 382.76 ± 13.96 | 0.30 ± 0.04 | -30.16 ± 1.28 |
| GLU2:PMDA 1:4 | 177.80 ± 2.66 | 0.11 ± 0.01 | -22.50 ± 0.76 |
| Sample (βCD: PMDA 1:4 NS) | WAC (%) | υ(mol/cm3) |
| Deionized H2O | 550 | 6.04E-5 |
| Gastric fluid (pH=1.2) | 172 | 7.53E-4 |
| Intestinal fluid (pH=6.8) | 863 | 4.64E-5 |
| Intestinal fluid (pH=7.4) | 544 | 9.65E-5 |
| Samples | G’ (Pa) (pH=1.2) | G’ (Pa) (pH=6.8) | G’ (Pa) (pH=7.4) | Delta G’/G’ (pH=1.2) | Delta G’/G’ (pH=6.8) | Delta G’/G’ (pH=7.4) |
| Mucin | 8.05 | 4.95 | 18.22 | |||
| NS | 80009.00 | 260.29 | 4940.29 | |||
| NS + Mucin | 167957.00 | 14444.50 | 14132.70 | 1.09 | 53.45 | 1.85 |
| Chitosan | 60420.80 | 56871.10 | 94889.30 | |||
| Chitosan + Mucin | 112273.00 | 47256.30 | 30918.20 | 0.64 | -36.26 | -12.90 |
| Nanosponges | Molar ratio (Dextrin: PMDA) | Glucose unit: PMDA | m (PMDA), g |
| β-CD/LC/GLU2: PMDA | 1:2 | 1:0.29 | 1.87 |
| 1:4 | 1:0.57 | 3.75 | |
| 1:8 | 1:1.14 | 7.51 | |
| β-CD/LC/GLU2: CA | 1:2 | 1:0.29 | 1.49 |
| 1:4 | 1:0.57 | 2.98 | |
| 1:8 | 1:1.14 | 5.96 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




