Submitted:
30 October 2025
Posted:
31 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
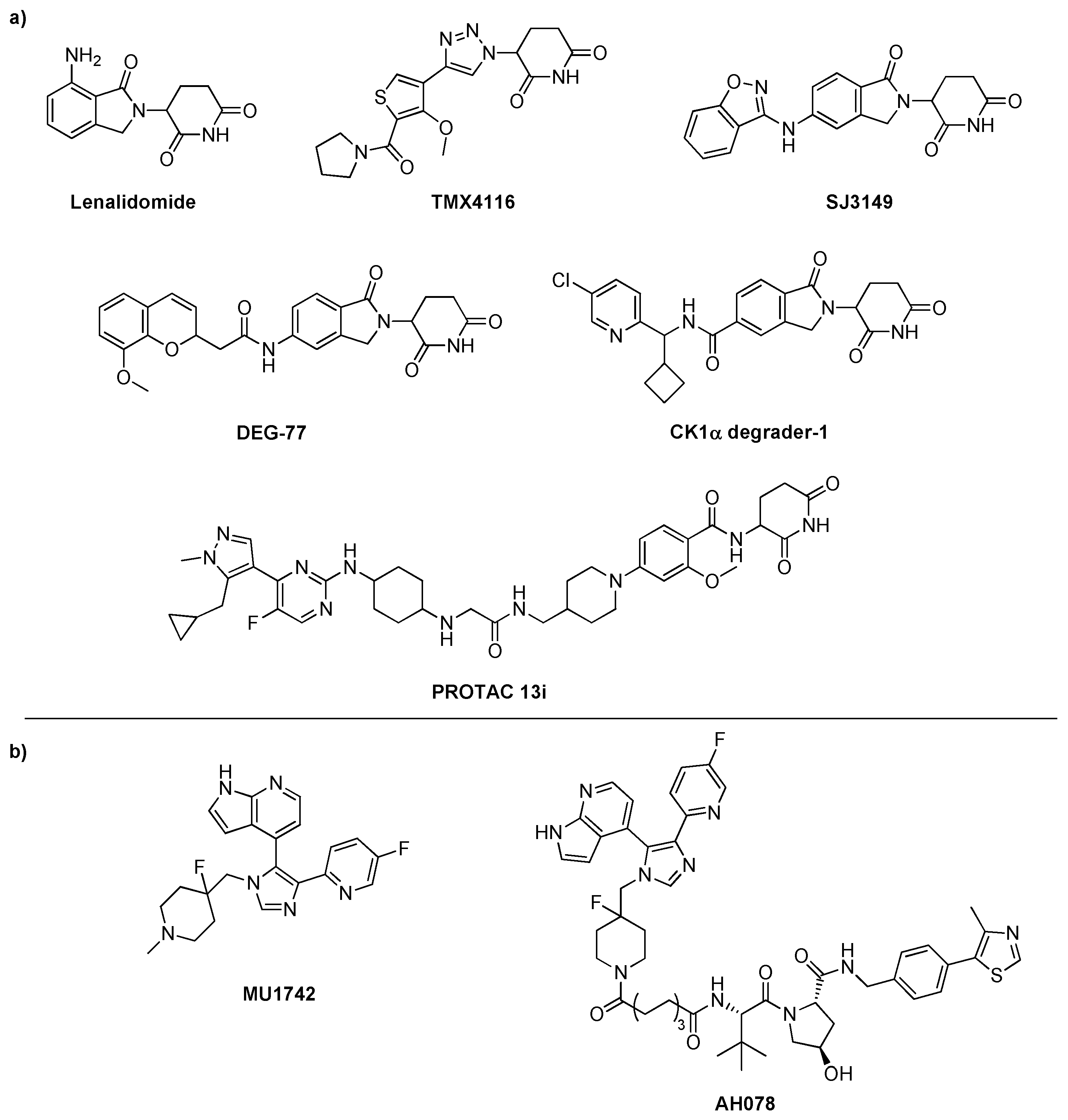
1.1. CK1 isoforms as proteins of interest for PROTAC design
2. Results and Discussion
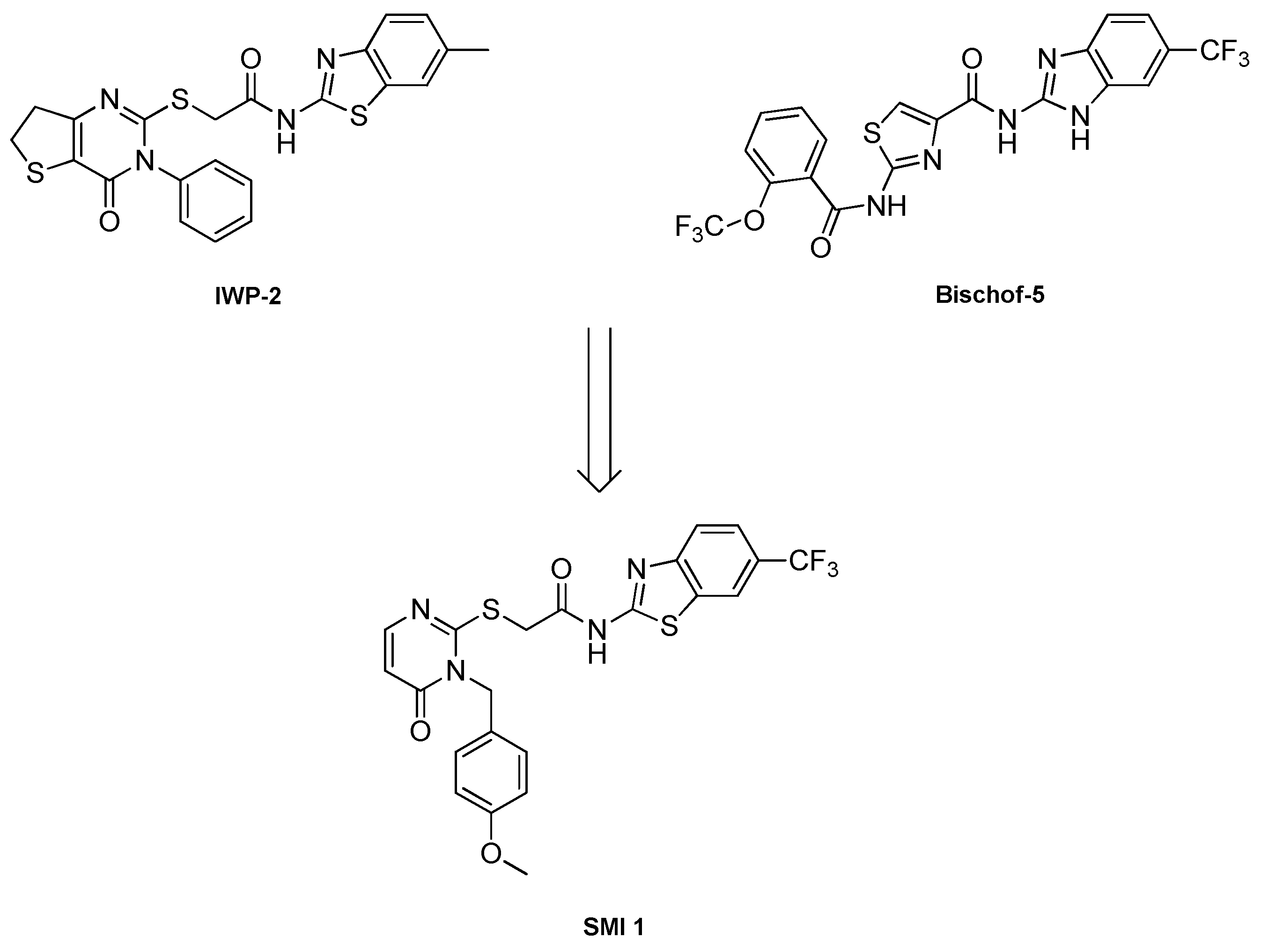
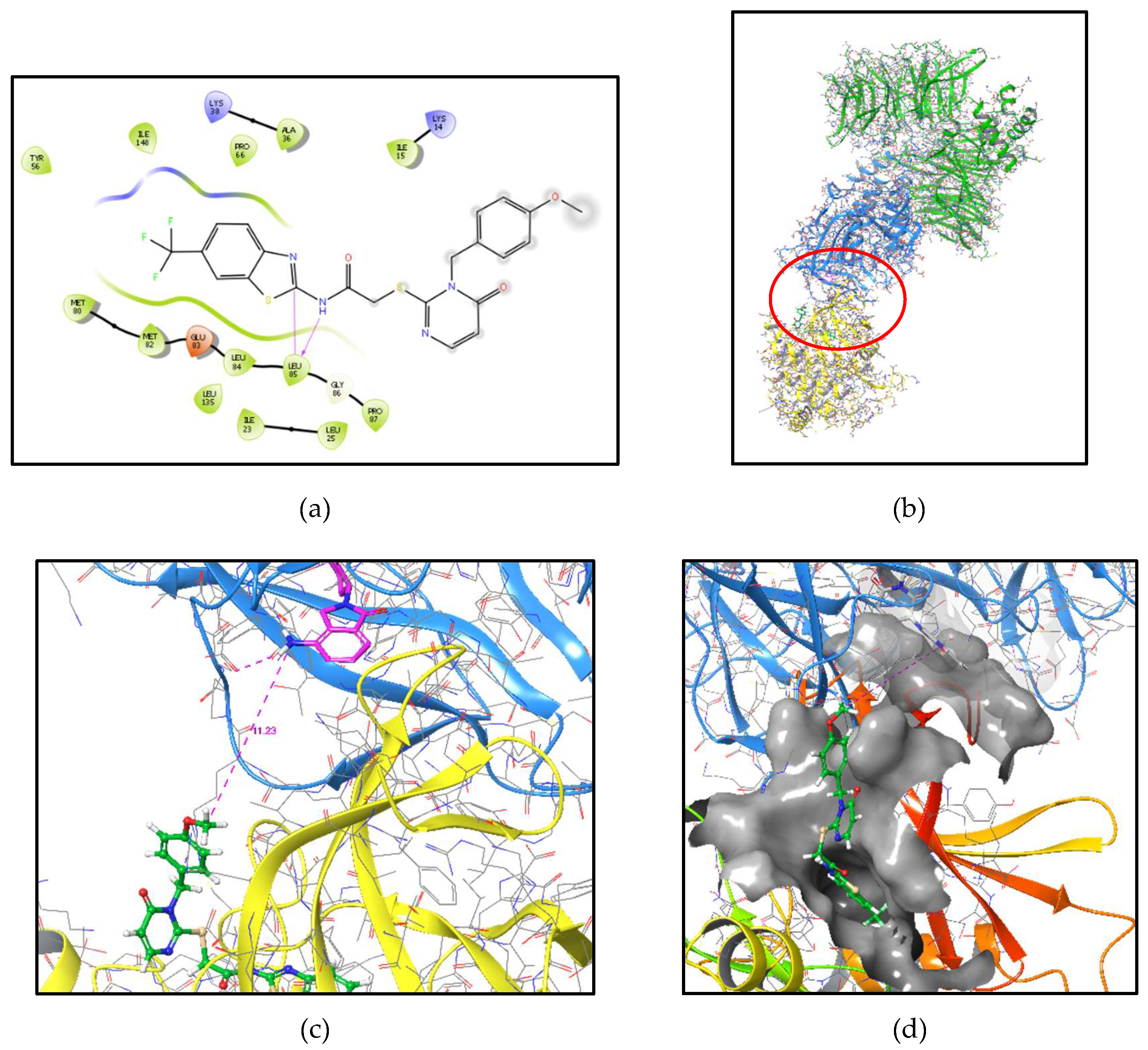
2.1. CK1δ PROTAC design based on SMI 1
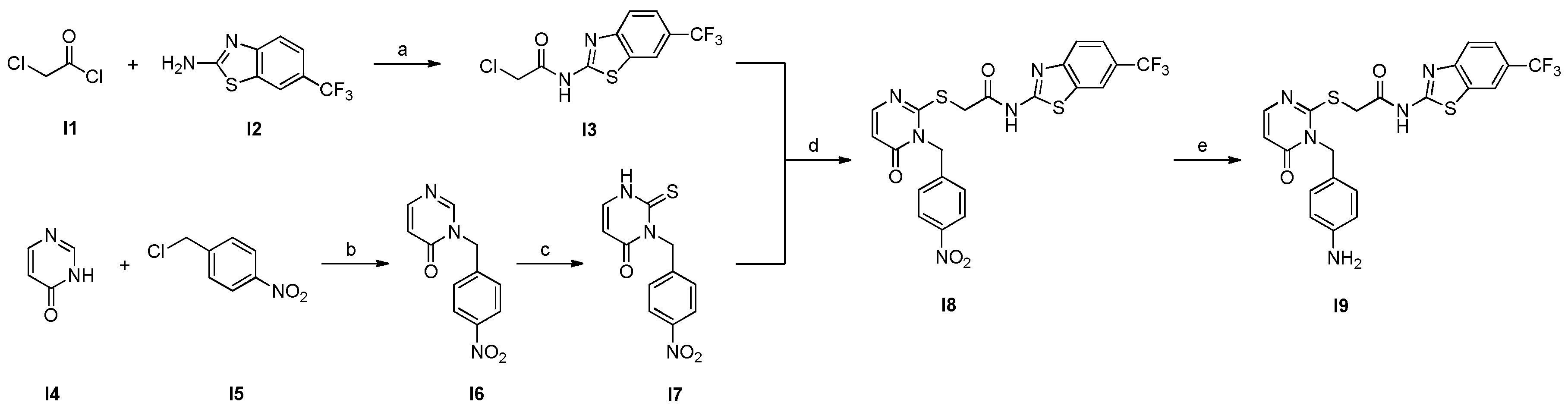
2.2. Synthesis of the key aniline CK1δ POI ligand I9
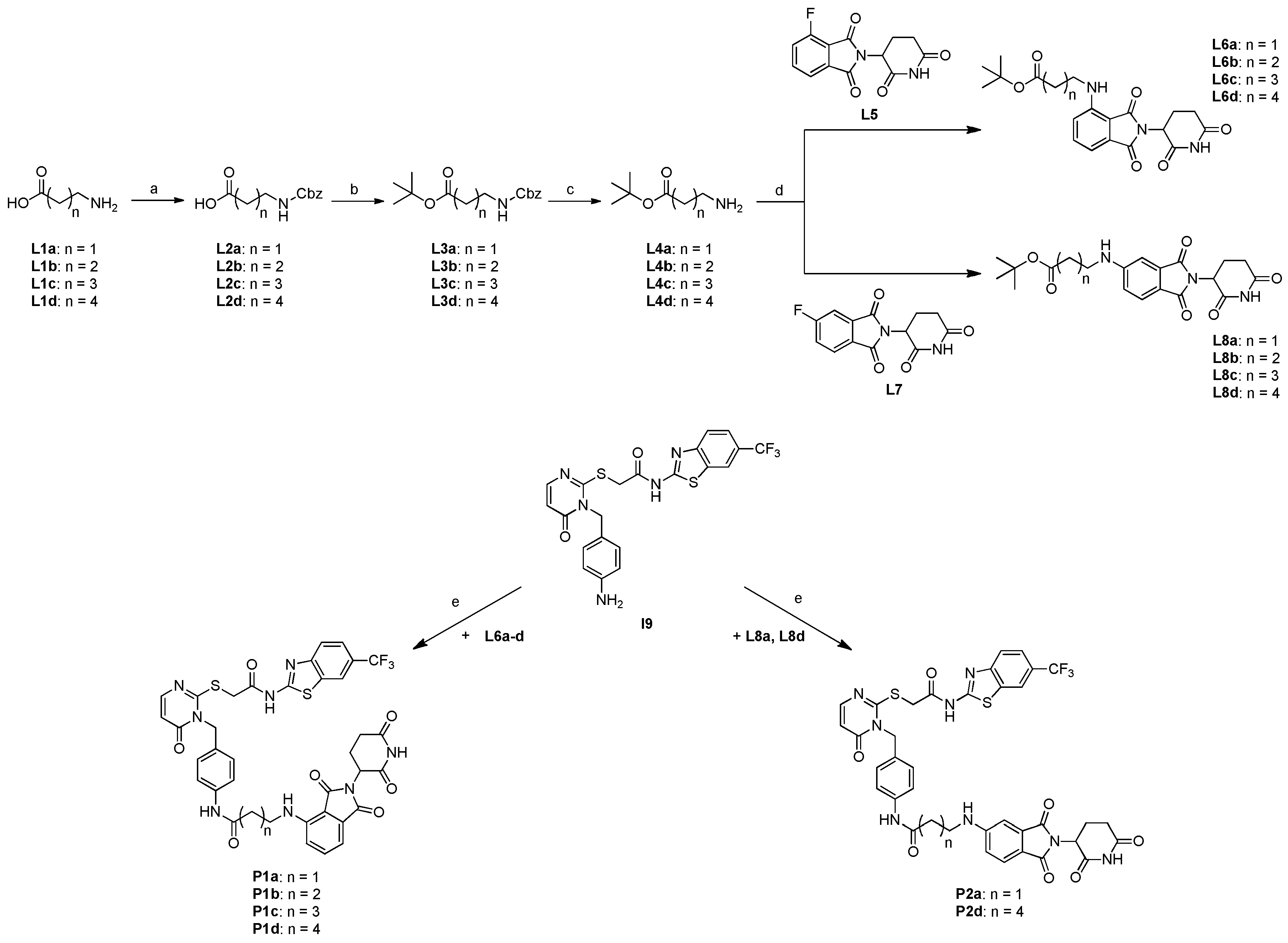
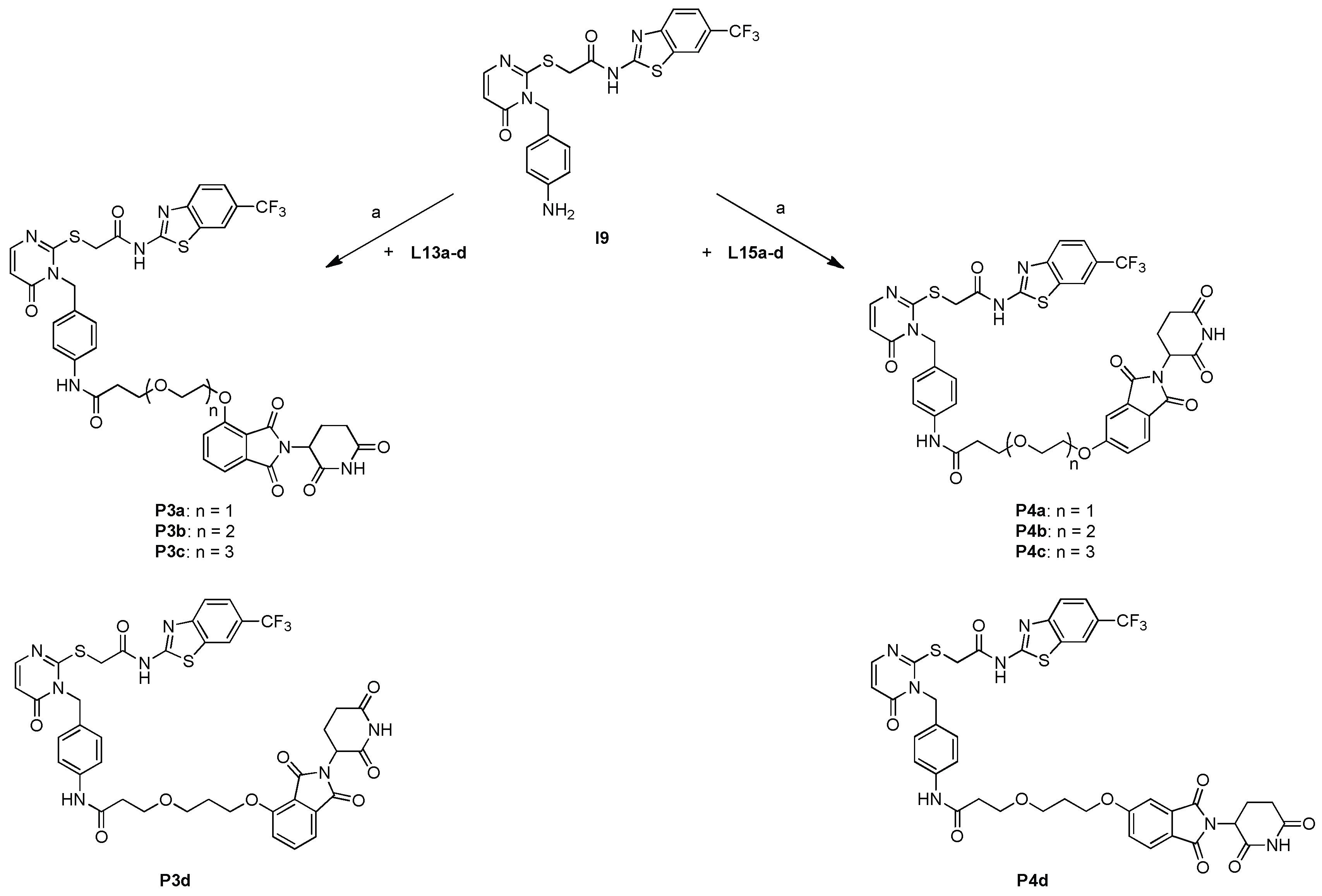
2.3. Synthesis of candidate CK1δ PROTACs containing alkyl linker motifs
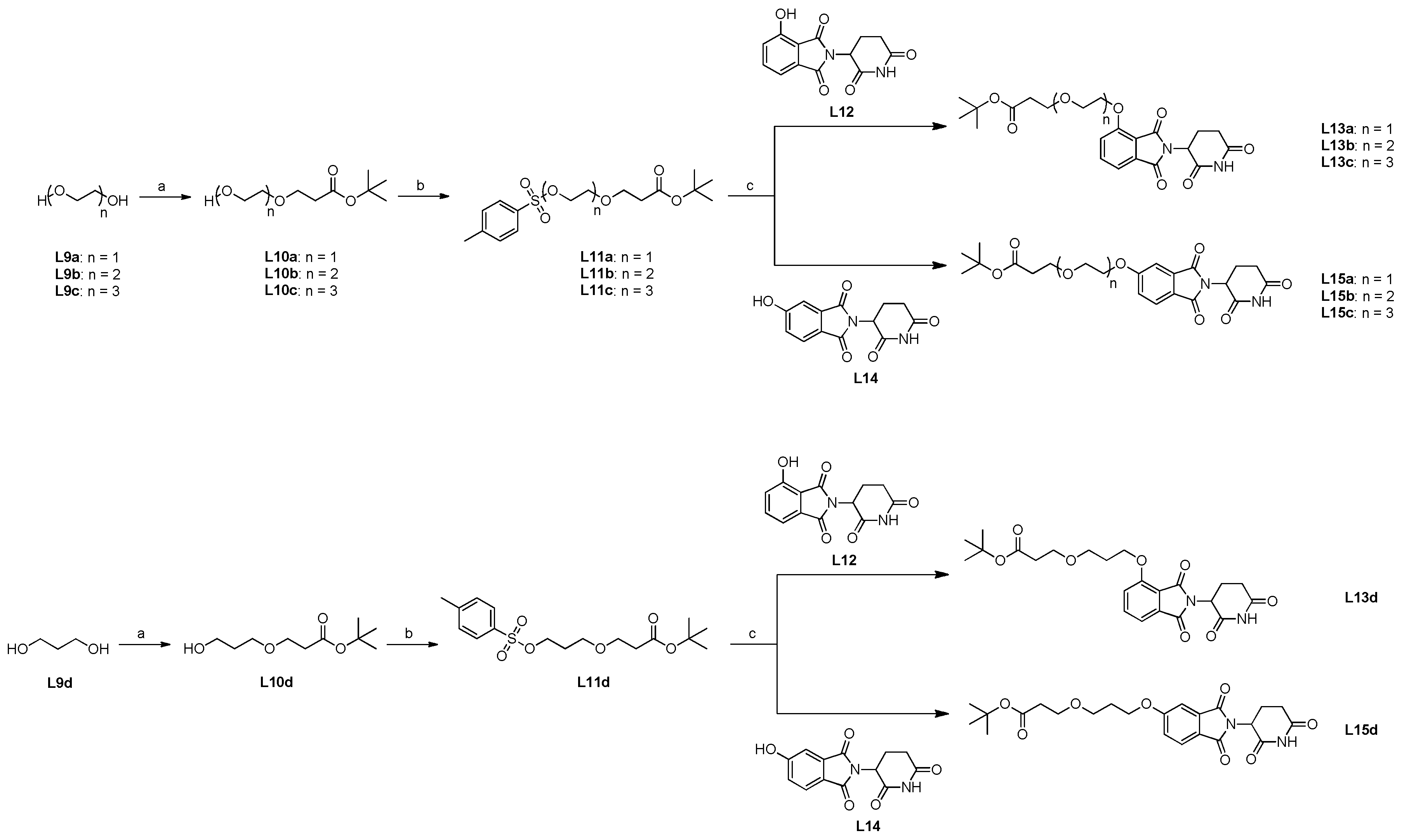
2.4. Synthesis of candidate CK1δ PROTACs containing PEG linker motifs
2.5. Biological Results
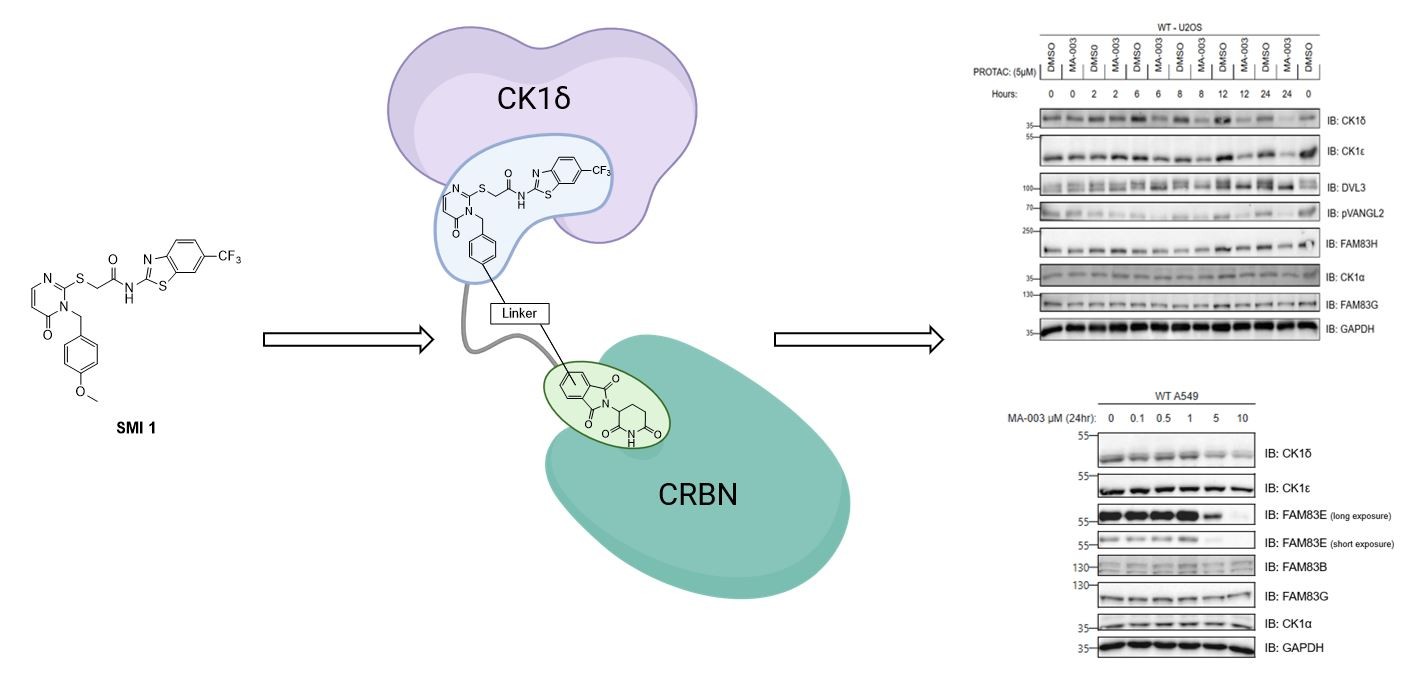
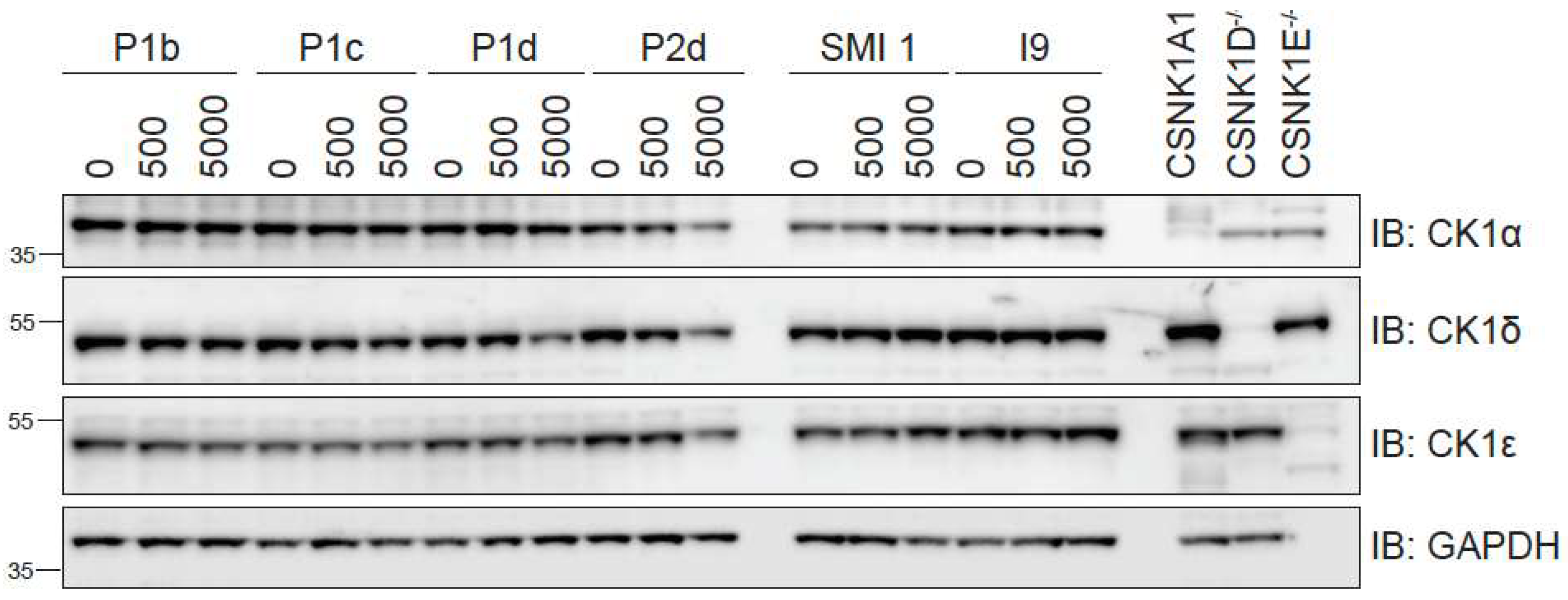
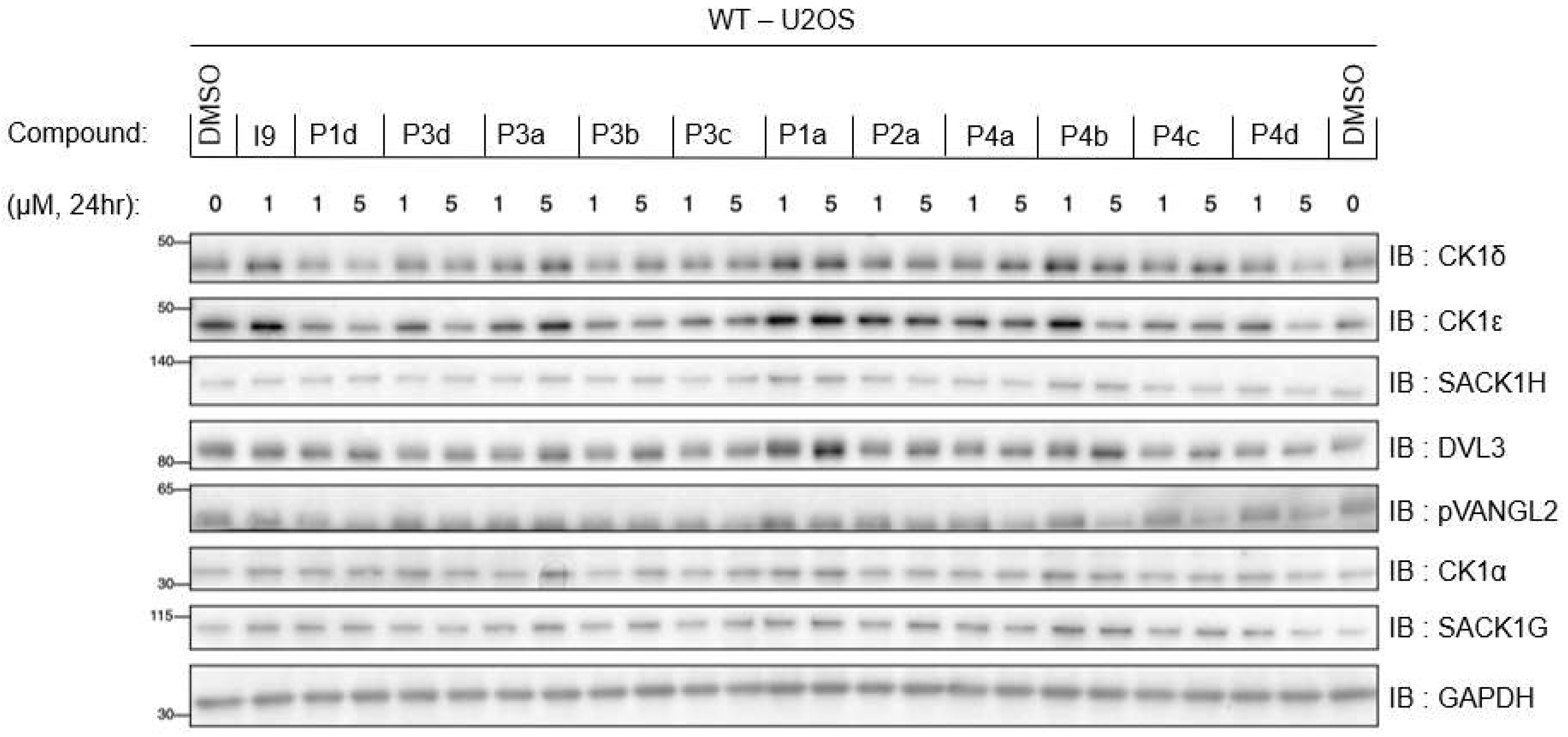
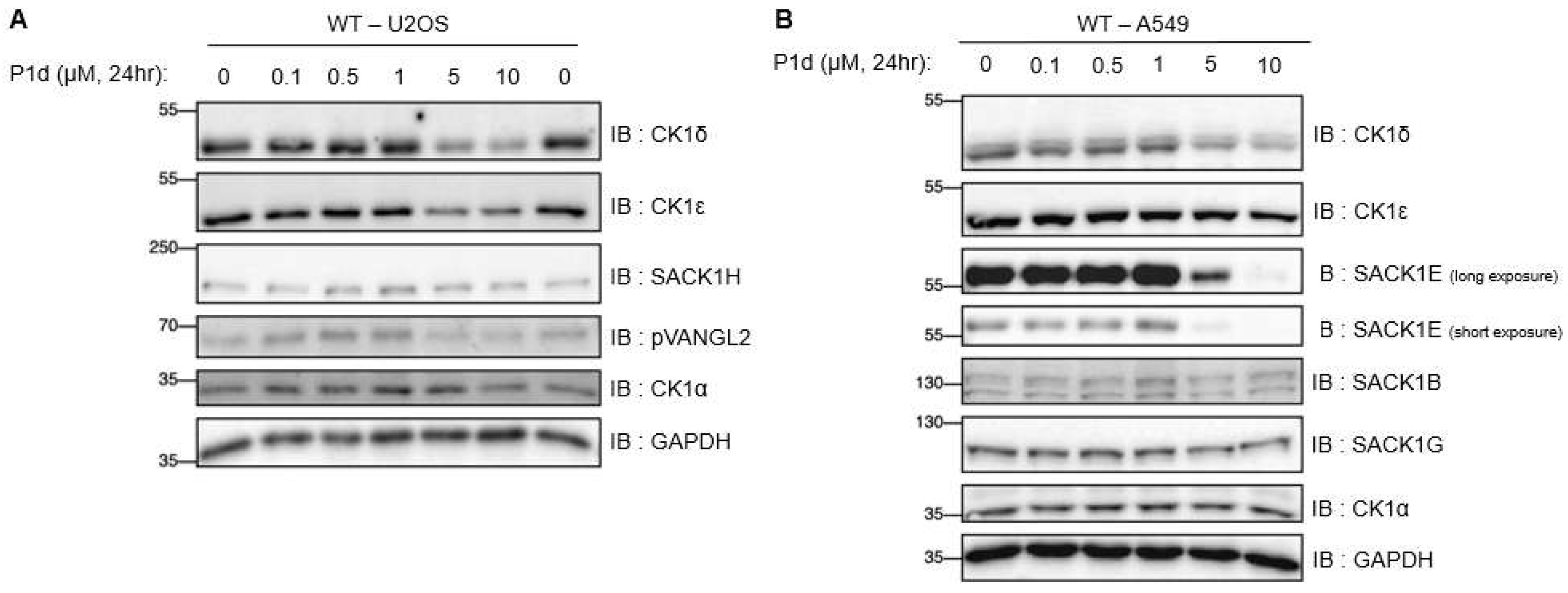
2.5.1. Differential degradation of CK1δ/ε by different PROTACs
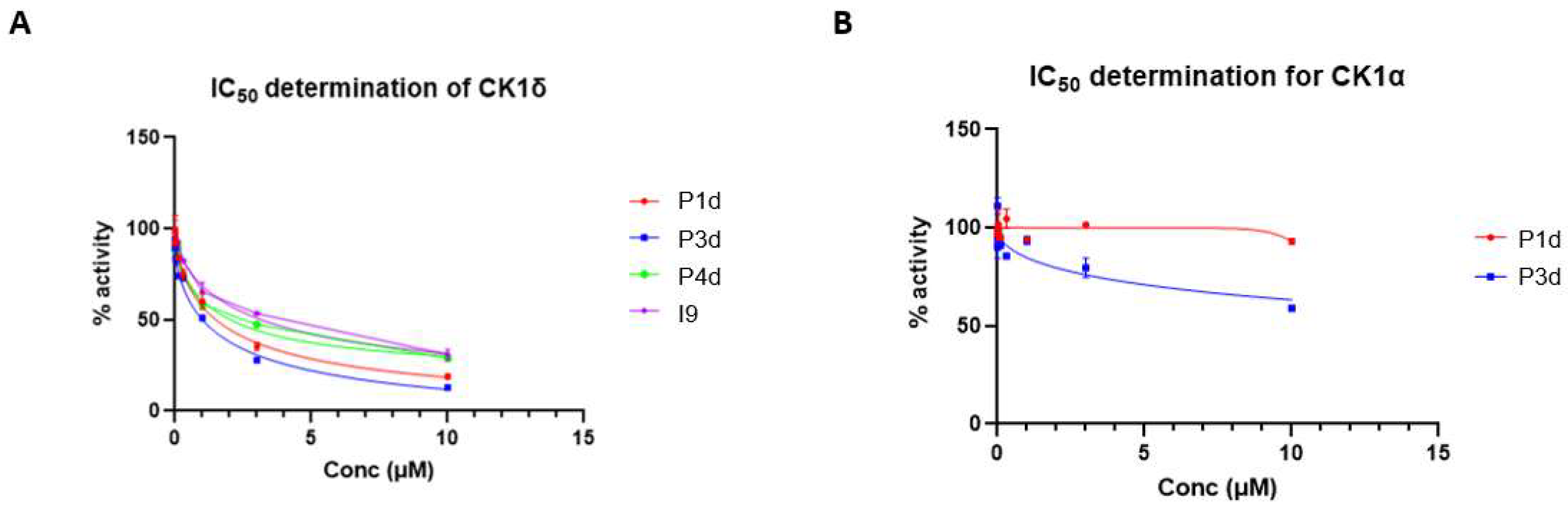
2.5.2. PROTACs as inhibitors of CK1δ/ε
2.5.3. Extended dose-response analysis of CK1δ/ε degradation by the best degrader P1d across multiple cells.
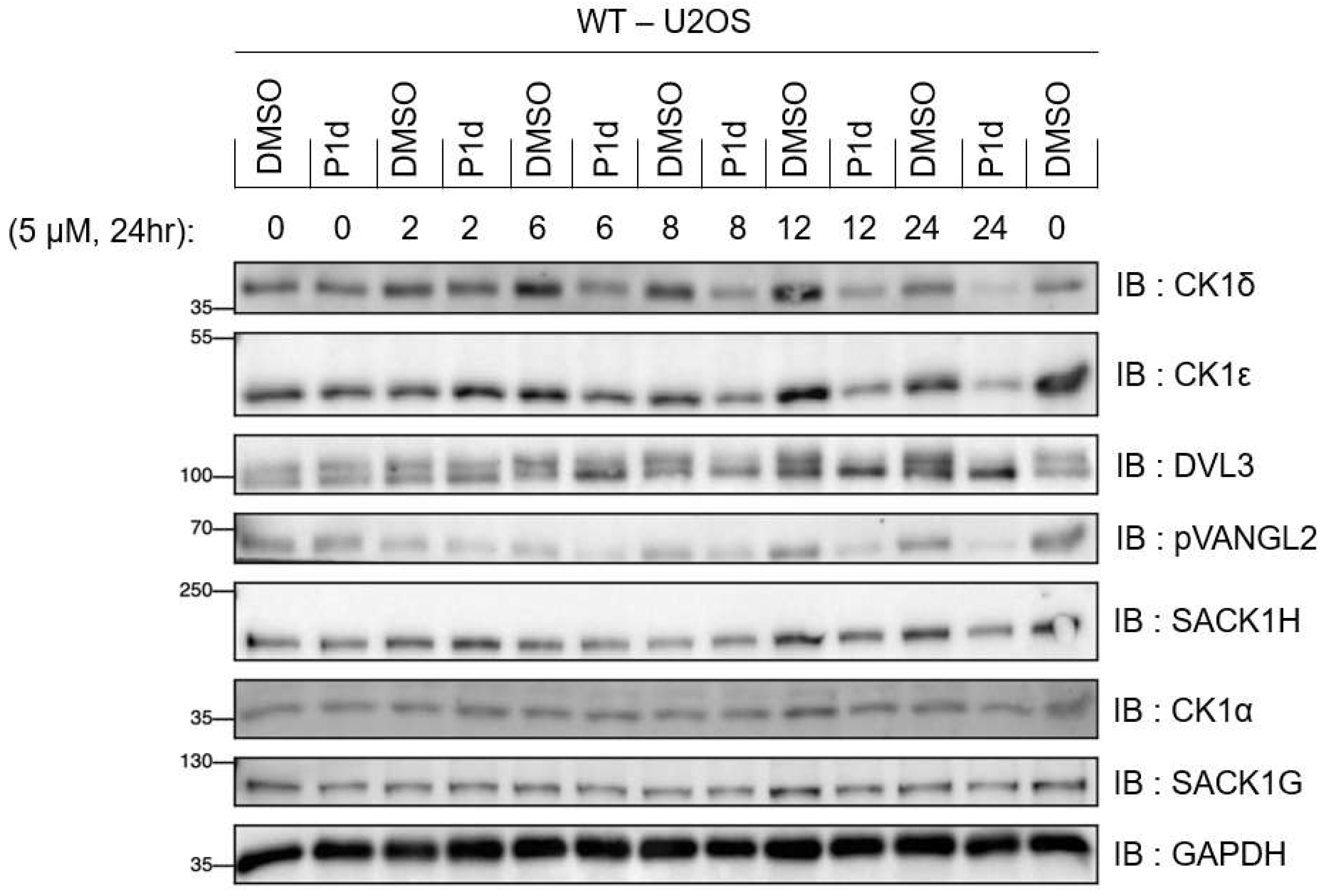
2.5.4. Kinetic analysis of CK1δ/ε degradation by P1d
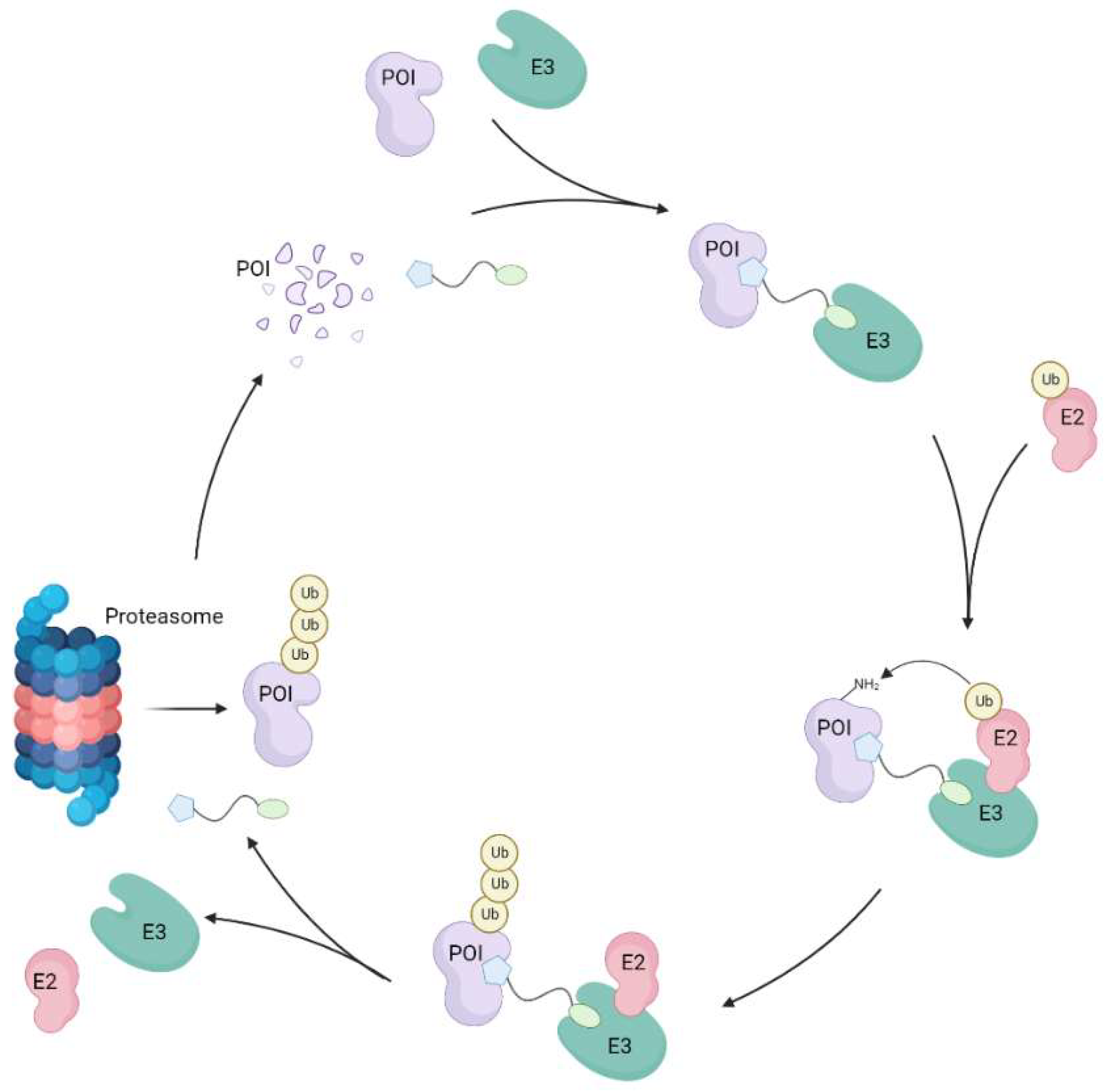
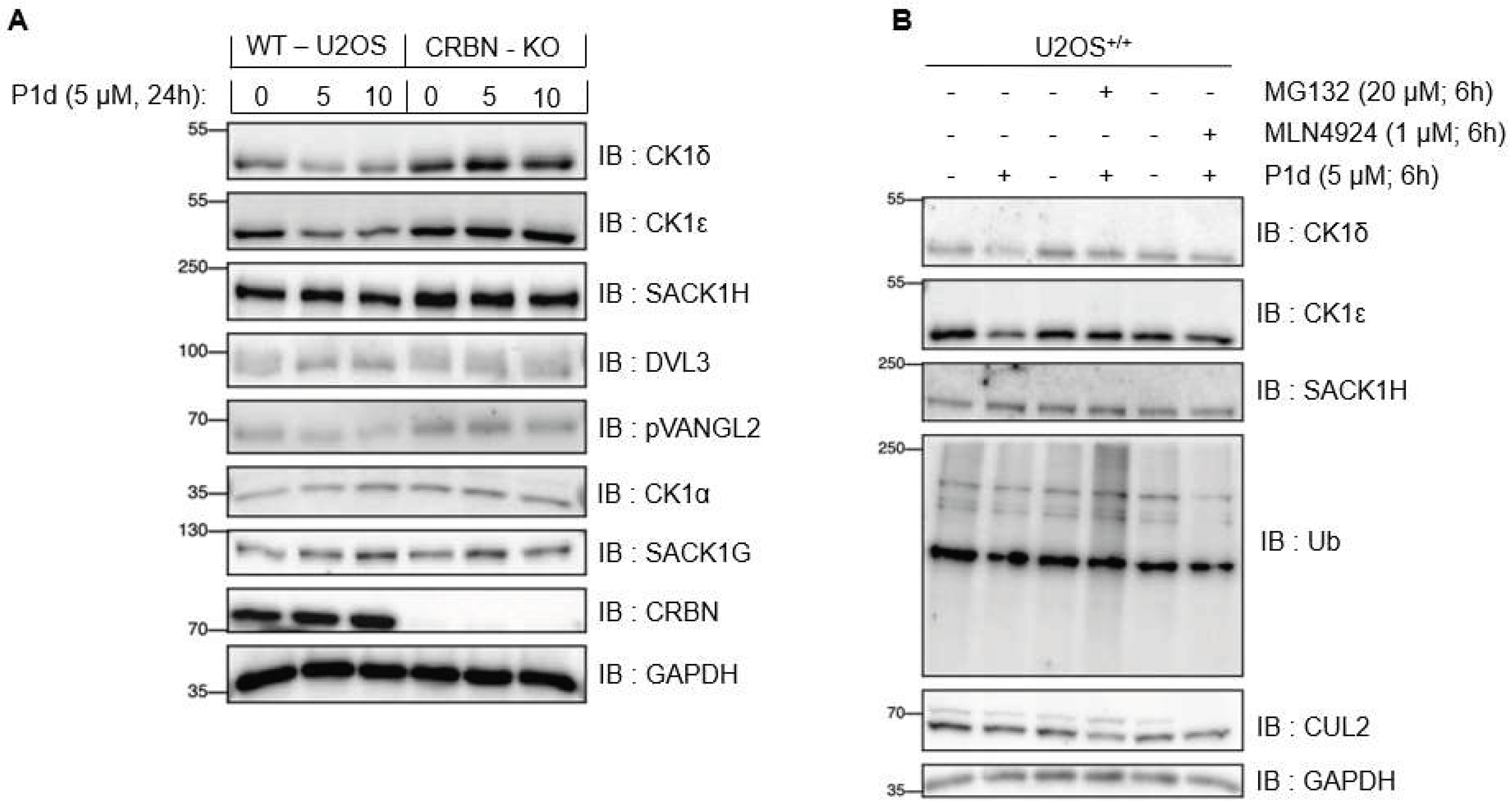
2.5.5. Establishing mode of action for P1d
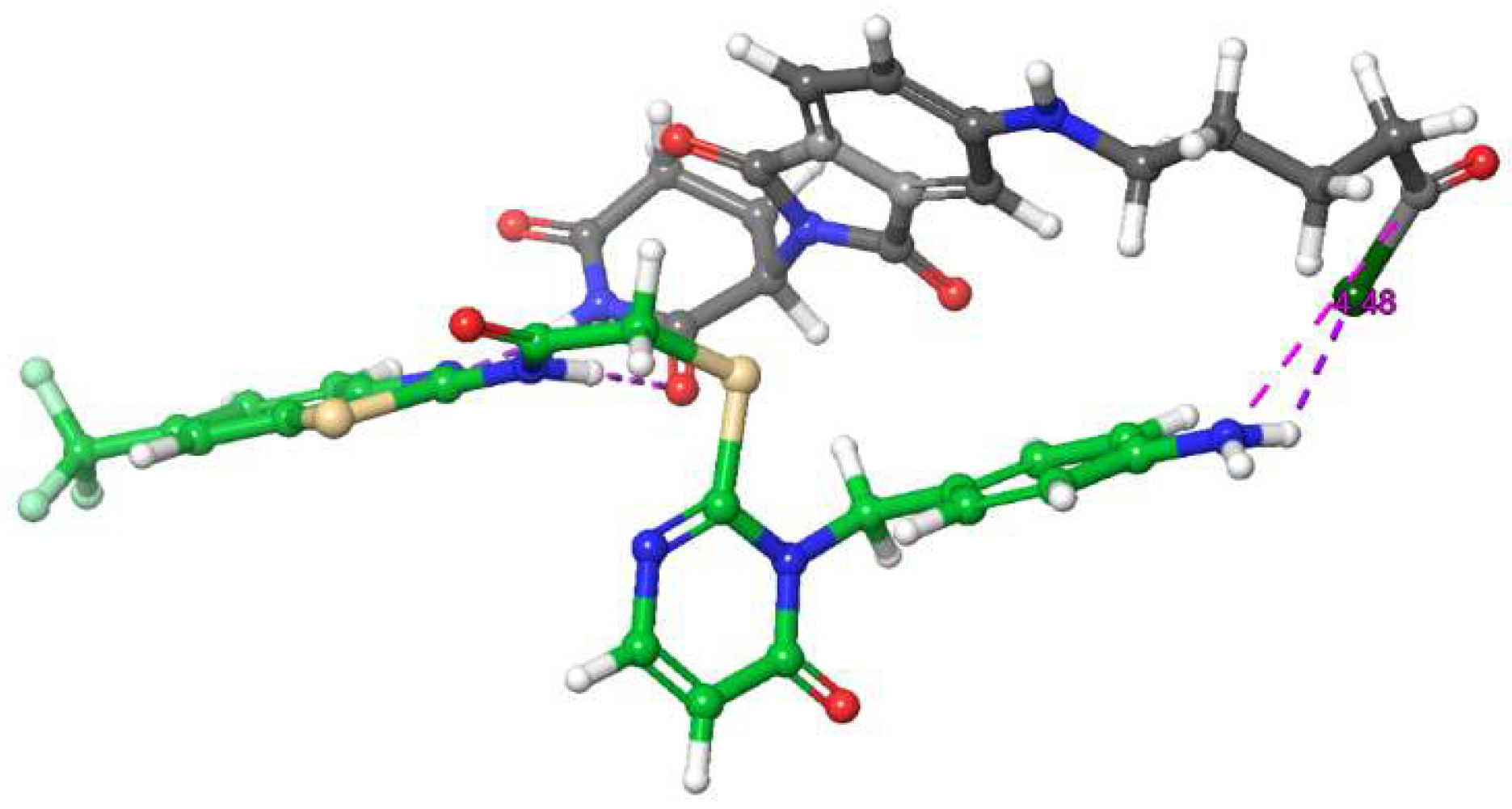
2.5.6. Correlation between Computed and Experimental Linker Dimensions
3. Materials and Methods
3.1. Molecular Modeling
3.2. Chemistry
3.2.1. General
3.2.2. Synthesis of Compound I3
3.2.3 Synthesis of Compound I6
3.2.4. Synthesis of Compound I7
3.2.5. Synthesis of Compound I8
3.2.6. Synthesis of Compound I9
3.2.7. General Procedure for the Synthesis of Linker Compounds L2a-d
3.2.8. General Procedure for the Synthesis of Linker Compounds L3a-d
3.2.9. General Procedure for the Synthesis of Linker Compounds L4a-d
3.2.10. General Procedure for the Synthesis of Compounds L6a-d
3.2.11. General Procedure for the Synthesis of Compounds L8a-d
3.2.12. General Procedure for the Synthesis of Linker Compounds L10a-d
3.2.13. General Procedure for the Synthesis of Linker Compounds L11a-d
3.2.14. General Procedure for the Synthesis of Compounds L13a-d
3.2.15. General Procedure for the Synthesis of Compounds L15a-d
3.2.16. General Procedure for the Synthesis of PROTAC Compounds P1a-d, P2a, P2d, P3a-d and P4a-d
3.3. Biology
3.3.1. Cell culture, drug treatment, and lysis
3.3.3. In vitro kinase assays for IC50 determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CK1 | Casein Kinase 1 |
| PROTAC | Proteolysis Targeting Chimera |
| PEG | Polyethylene Glycol |
| MOA | Mode of Action |
| SMI | Small Molecule Inhibitor |
| POI | Protein of Interest |
| UPS | Ubiquitin Proteasome System |
| APC | Adenomatous Polyposis Coli |
| AXIN | Axis Inhibition Protein |
| GSK3 protein | Glycogen Synthase Kinase 3 Protein |
| HRII | Hydrophobic Region II |
| ATP | Adenosine triphosphate |
| TPD | Targeted Protein Degradation |
| IWP | Inhibitor of Wnt Production |
| CRBN | Cereblon |
| p-MeO | Para-Methoxy |
| SAR | Structure Activity Relationship |
| TEA | Triethylamine |
| DMSO | Dimethylsulfoxide |
| DMF | Dimethylformamide |
| equiv. | Equivalent |
| TFA | Trifluororacetic acid |
| HATU | Hexafluorophosphate Azabenzotriazole Tetramethyl Uronium |
| DIPEA | N, N-Diisopropylethylamine |
| EDC | 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide |
| HOBt | Hydroxybenzotriazole |
| DMAP | 4-Dimethylaminopyridine |
| MeCN | Acetonitrile |
| RP-HPLC | Reversed Phase High Performance Liquid Chromatography |
| MS | Mass Spectrometry |
| HR-MS | High resolution Mass Spectrometry |
| NMR | Nuclear Magnetic Resonance |
| EA | Ethyl acetate |
| MeOH | Methanol |
References
- Békés, M.; Langley, D. R.; Crews, C. M. Nature reviews. Drug discovery 2022, 3, 181. [CrossRef]
- Li, K.; Crews, C. M. Chemical Society reviews 2022, 12, 5214. [CrossRef]
- Nalawansha, D. A.; Crews, C. M. Cell Chemical Biology 2020, 8, 998. [CrossRef]
- Hershko, A.; Ciechanover, A. Annual review of biochemistry 1998, 425. [CrossRef]
- Kleiger, G.; Mayor, T. Trends in cell biology 2014, 6, 352. [CrossRef]
- Nandi, D.; Tahiliani, P.; Kumar, A.; Chandu, D. Journal of biosciences 2006, 1, 137. [CrossRef]
- Paiva, S.-L.; Da Silva, S. R.; Araujo, E. D. de; Gunning, P. T. Journal of medicinal chemistry 2018, 2, 405. [CrossRef]
- Popovic, D.; Vucic, D.; Dikic, I. Nature medicine 2014, 11, 1242. [CrossRef]
- Jiang, Y.; Beaudet, A. L. Current opinion in pediatrics 2004, 4, 419. [CrossRef]
- Paiva, S.-L.; Crews, C. M. Current Opinion in Chemical Biology 2019, 111. [CrossRef]
- An, S.; Fu, L. EBioMedicine 2018, 553. [CrossRef]
- Berndsen, C. E.; Wolberger, C. Nature Structural & Molecular Biology 2014, 4, 301. [CrossRef]
- Schreiber, A.; Peter, M. Biochimica et biophysica acta 2014, 1, 163. [CrossRef]
- Alabi, S. B.; Crews, C. M. The Journal of biological chemistry 2021, 100647. [CrossRef]
- Bondeson, D. P.; Mares, A.; Smith, I. E. D.; Ko, E.; Campos, S.; Miah, A. H.; Mulholland, K. E.; Routly, N.; Buckley, D. L.; Gustafson, J. L.; Zinn, N.; Grandi, P.; Shimamura, S.; Bergamini, G.; Faelth-Savitski, M.; Bantscheff, M.; Cox, C.; Gordon, D. A.; Willard, R. R.; Flanagan, J. J.; Casillas, L. N.; Votta, B. J.; Besten, W. den; Famm, K.; Kruidenier, L.; Carter, P. S.; Harling, J. D.; Churcher, I.; Crews, C. M. Nature chemical biology 2015, 8, 611. [CrossRef]
- Zagidullin, A.; Milyukov, V.; Rizvanov, A.; Bulatov, E. Exploration of targeted anti-tumor therapy 2020, 5, 381. [CrossRef]
- Troup, R. I.; Fallan, C.; Baud, M. G. J. Exploration of targeted anti-tumor therapy 2020, 5, 273. [CrossRef]
- Poongavanam, V.; Peintner, S.; Abeje, Y.; Kölling, F.; Meibom, D.; Erdelyi, M.; Kihlberg, J. ACS medicinal chemistry letters 2025. [CrossRef]
- Cyrus, K.; Wehenkel, M.; Choi, E.-Y.; Han, H.-J.; Lee, H.; Swanson, H.; Kim, K.-B. Molecular bioSystems 2011, 2, 359. [CrossRef]
- Knippschild, U.; Krüger, M.; Richter, J.; Xu, P.; García-Reyes, B.; Peifer, C.; Halekotte, J.; Bakulev, V.; Bischof, J. Frontiers in oncology 2014, 96. [CrossRef]
- Narasimamurthy, R.; Hunt, S. R.; Lu, Y.; Fustin, J.-M.; Okamura, H.; Partch, C. L.; Forger, D. B.; Kim, J. K.; Virshup, D. M. Proceedings of the National Academy of Sciences 2018, 23, 5986. [CrossRef]
- Sinnberg, T.; Wang, J.; Sauer, B.; Schittek, B. BMC cancer 2016, 594. [CrossRef]
- Fulcher, L. J.; Sapkota, G. P. The Biochemical journal 2020, 23, 4603. [CrossRef]
- Gybeľ, T.; Čada, Š.; Klementová, D.; Schwalm, M. P.; Berger, B.-T.; Šebesta, M.; Knapp, S.; Bryja, V. The Journal of biological chemistry 2024, 7, 107407. [CrossRef]
- Liu, J.; Xiao, Q.; Xiao, J.; Niu, C.; Li, Y.; Zhang, X.; Zhou, Z.; Shu, G.; Yin, G. Signal transduction and targeted therapy 2022, 1, 3. [CrossRef]
- MacDonald, B. T.; Tamai, K.; He, X. Developmental cell 2009, 1, 9. [CrossRef]
- Bryja, V.; Bernatík, O. 2014, 207. [CrossRef]
- Xu, P.; Ianes, C.; Gärtner, F.; Liu, C.; Burster, T.; Bakulev, V.; Rachidi, N.; Knippschild, U.; Bischof, J. Gene 2019, 144005. [CrossRef]
- Mazzoldi, E. L.; Pastò, A.; Ceppelli, E.; Pilotto, G.; Barbieri, V.; Amadori, A.; Pavan, S. Frontiers in oncology 2019, 1211. [CrossRef]
- Cheong, J. K.; Virshup, D. M. The international journal of biochemistry & cell biology 2011, 4, 465. [CrossRef]
- Nishiguchi, G.; Caine, E. A.; McGowan, K.; Shi, Z.; Aggarwal, A.; Mayasundari, A.; Price, J.; Yang, L.; Li, Y.; Fu, X.; Mascibroda, L. G.; Das, S.; Daniels, D. L.; Urh, M.; Klco, J. M.; Riching, K. M.; Rankovic, Z. ACS medicinal chemistry letters 2024. [CrossRef]
- MedchemExpress.com. Available online: https://www.medchemexpress.com/ck1%CE%B1-degrader-1.html (accessed on 6 March 2025).
- Wang, K.; Jiang, M.; Liu, H.; Meng, C.; Li, M.; Lu, H. Bioorganic chemistry 2024, 107319. [CrossRef]
- Haag, A.; Němec, V.; Janovská, P.; Bartošíková, J.; Adhikari, B.; Müller, J.; Schwalm, M. P.; Čada, Š.; Ohmayer, U.; Daub, H.; Kim, Y.; Born, F.; Wolf, E.; Bryja, V.; Knapp, S. Journal of medicinal chemistry 2025, 1, 506. [CrossRef]
- García-Reyes, B.; Witt, L.; Jansen, B.; Karasu, E.; Gehring, T.; Leban, J.; Henne-Bruns, D.; Pichlo, C.; Brunstein, E.; Baumann, U.; Wesseler, F.; Rathmer, B.; Schade, D.; Peifer, C.; Knippschild, U. Journal of medicinal chemistry 2018, 9, 4087. [CrossRef]
- Liu, C.; Witt, L.; Ianes, C.; Bischof, J.; Bammert, M.-T.; Baier, J.; Kirschner, S.; Henne-Bruns, D.; Xu, P.; Kornmann, M.; Peifer, C.; Knippschild, U. International journal of molecular sciences 2019, 24. [CrossRef]
- Schade, D.; Plowright, A. T. Journal of medicinal chemistry 2015, 24, 9451. [CrossRef]
- Chen, B.; Dodge, M. E.; Tang, W.; Lu, J.; Ma, Z.; Fan, C.-W.; Wei, S.; Hao, W.; Kilgore, J.; Williams, N. S.; Roth, M. G.; Amatruda, J. F.; Chen, C.; Lum, L. Nature chemical biology 2009, 2, 100. [CrossRef]
- Bischof, J.; Leban, J.; Zaja, M.; Grothey, A.; Radunsky, B.; Othersen, O.; Strobl, S.; Vitt, D.; Knippschild, U. Amino acids 2012, 4, 1577. [CrossRef]
- Petzold, G.; Fischer, E. S.; Thomä, N. H. Nature 2016, 7597, 127. [CrossRef]
- Hernández-Núñez, E.; Tlahuext, H.; Moo-Puc, R.; Torres-Gómez, H.; Reyes-Martínez, R.; Cedillo-Rivera, R.; Nava-Zuazo, C.; Navarrete-Vazquez, G. European journal of medicinal chemistry 2009, 7, 2975. [CrossRef]
- Roopan, S. M.; Khan, F. R. N.; Mandal, B. K. Tetrahedron letters 2010, 17, 2309. [CrossRef]
- Xu, J.; Yadan, J.-C. Synlett 1995, 03, 239. [CrossRef]
- Schmuck, C.; Rehm, T.; Geiger, L.; Schäfer, M. The Journal of organic chemistry 2007, 16, 6162. [CrossRef]
- Kim, K.; Lee, D. H.; Park, S.; Jo, S.-H.; Ku, B.; Park, S. G.; Park, B. C.; Jeon, Y. U.; Ahn, S.; Kang, C. H.; Hwang, D.; Chae, S.; Du Ha, J.; Kim, S.; Hwang, J. Y.; Kim, J.-H. Scientific reports 2019, 1, 19654. [CrossRef]
- Ghosh, A. K.; Shahabi, D. Tetrahedron letters 2021.
- Abeje, Y. E.; Wieske, L. H. E.; Poongavanam, V.; Maassen, S.; Atilaw, Y.; Cromm, P.; Lehmann, L.; Erdelyi, M.; Meibom, D.; Kihlberg, J. Journal of medicinal chemistry 2025, 1, 638. [CrossRef]
- Cromm, P. M.; Samarasinghe, K. T. G.; Hines, J.; Crews, C. M. Journal of the American Chemical Society 2018, 49, 17019. [CrossRef]
- Kaucká, M.; Petersen, J.; Janovská, P.; Radaszkiewicz, T.; Smyčková, L.; Daulat, A. M.; Borg, J.-P.; Schulte, G.; Bryja, V. Cell communication and signalling : CCS 2015, 2. [CrossRef]
- Yang, W.; Garrett, L.; Di Feng; Elliott, G.; Liu, X.; Wang, N.; Wong, Y. M.; Choi, N. T.; Yang, Y.; Gao, B. Cell research 2017, 12, 1466. [CrossRef]
- Fulcher, L. J.; Bozatzi, P.; Tachie-Menson, T.; Wu, K. Z. L.; Cummins, T. D.; Bufton, J. C.; Pinkas, D. M.; Dunbar, K.; Shrestha, S.; Wood, N. T.; Weidlich, S.; Macartney, T. J.; Varghese, J.; Gourlay, R.; Campbell, D. G.; Dingwell, K. S.; Smith, J. C.; Bullock, A. N.; Sapkota, G. P. Science signalling 2018, 531. [CrossRef]
- Glennie, L.; Curnutt, N.; Cartwright, T.; Dunbar, K.; Le Chatelier, B.; Wood, N. T.; Macartney, T. J.; Woo, C. M.; Sapkota, G. P. bioRxiv : the preprint server for biology 2025. [CrossRef]
- Dunbar, K.; Jones, R. A.; Dingwell, K.; Macartney, T. J.; Smith, J. C.; Sapkota, G. P. Life science alliance 2021, 2. [CrossRef]
| I9 | L8b-c | Solvent | Base (5 equiv.) |
Coupling Reagents | Temperature [° C] | Reaction Time | Additive |
|---|---|---|---|---|---|---|---|
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | DMSO | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | DMF | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | THF | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 25 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | Potassium carbonate | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | TEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | DBU | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | Mole sieve (3 Å) |
| 2 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 2 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | - |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | Glutarimide (5 equiv.) |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | EDC, HOBT, DMAP | 40 °C | 48 h | Glutarimide (10 equiv.) |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | HATU | 40 °C | 5 h | - |
| 1 equiv. | 1 equiv. | MeCN | Potassium carbonate | HATU | 40 °C | 5 h | - |
| 1 equiv. | 1 equiv. | MeCN | TEA | HATU | 40 °C | 5 h | - |
| 1 equiv. | 1 equiv. | MeCN | DBU | HATU | 40 °C | 5 h | - |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | HATU | 40 °C | 5 h | Mole sieve (3 Å) |
| 1 equiv. | 1 equiv. | MeCN | DIPEA | HATU | 70 °C | 5 h | - |
| PROTAC | Linker length (Å)1 |
|---|---|
| P1a | 4.179 |
| P1b | 4.991 |
| P1c | 6.428 |
| P1d | 7.413 |
| P2a | 4.179 |
| P2d | 7.413 |
| P3a | 7.103 |
| P3b | 10.712 |
| P3c | 14.062 |
| P3d | 8.492 |
| P4a | 7.103 |
| P4b | 10.712 |
| P4c | 14.062 |
| P4d | 8.492 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




