Submitted:
27 October 2025
Posted:
29 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
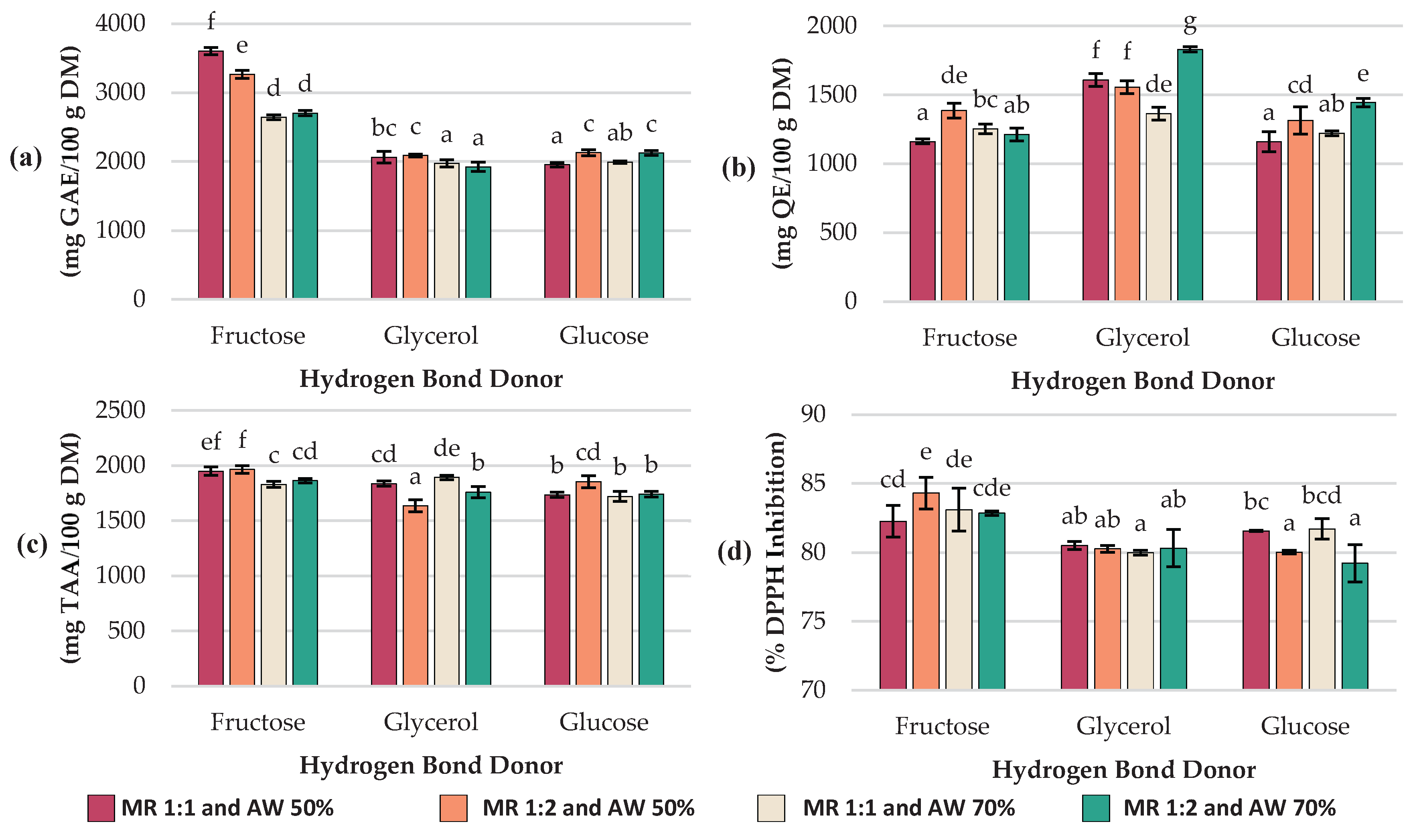
2.1. Evaluation of Total Polyphenols, Flavonoids, Ascorbic Acid, and Antioxidant Capacity in C. aurantium By-Products Extracts
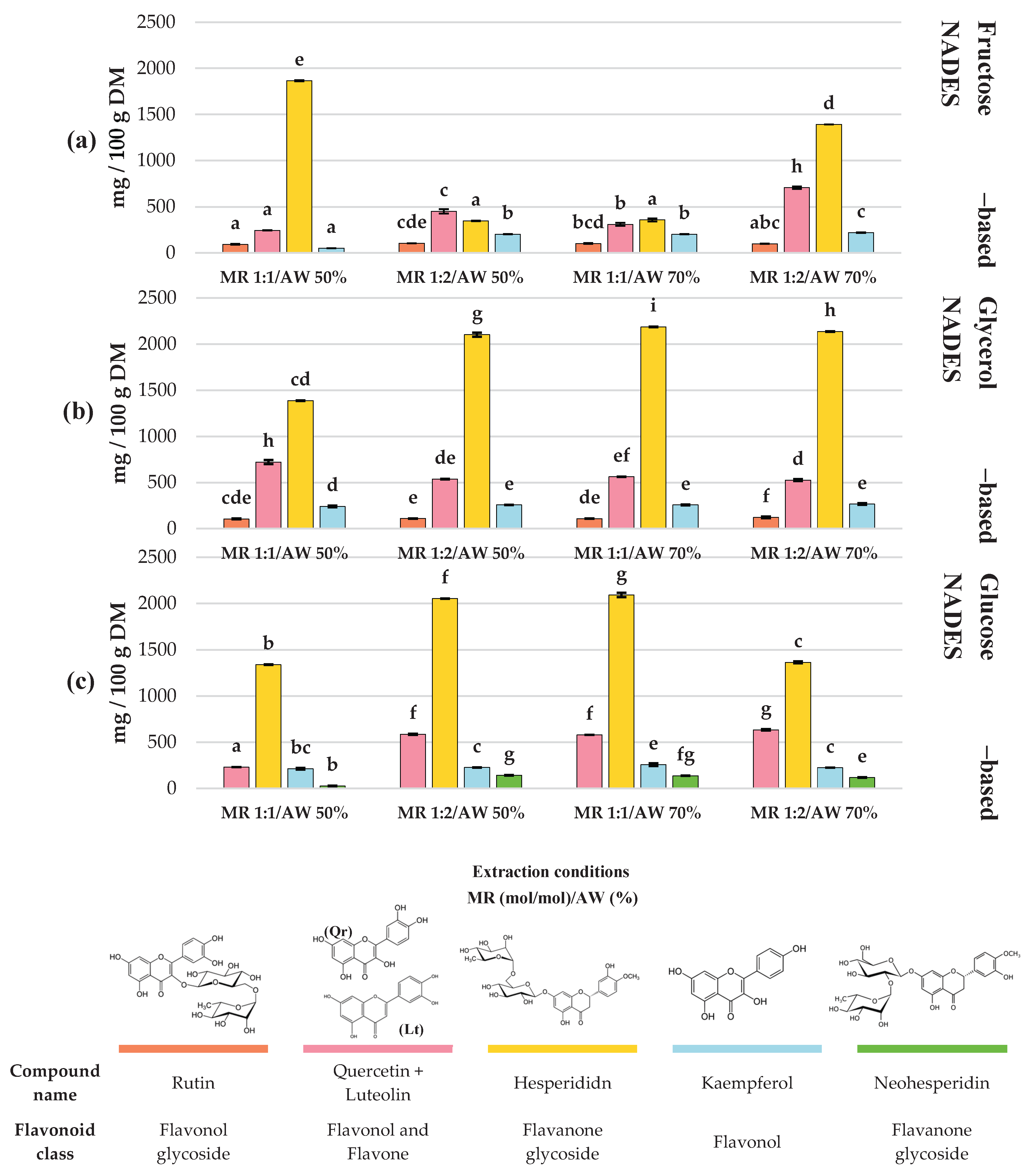
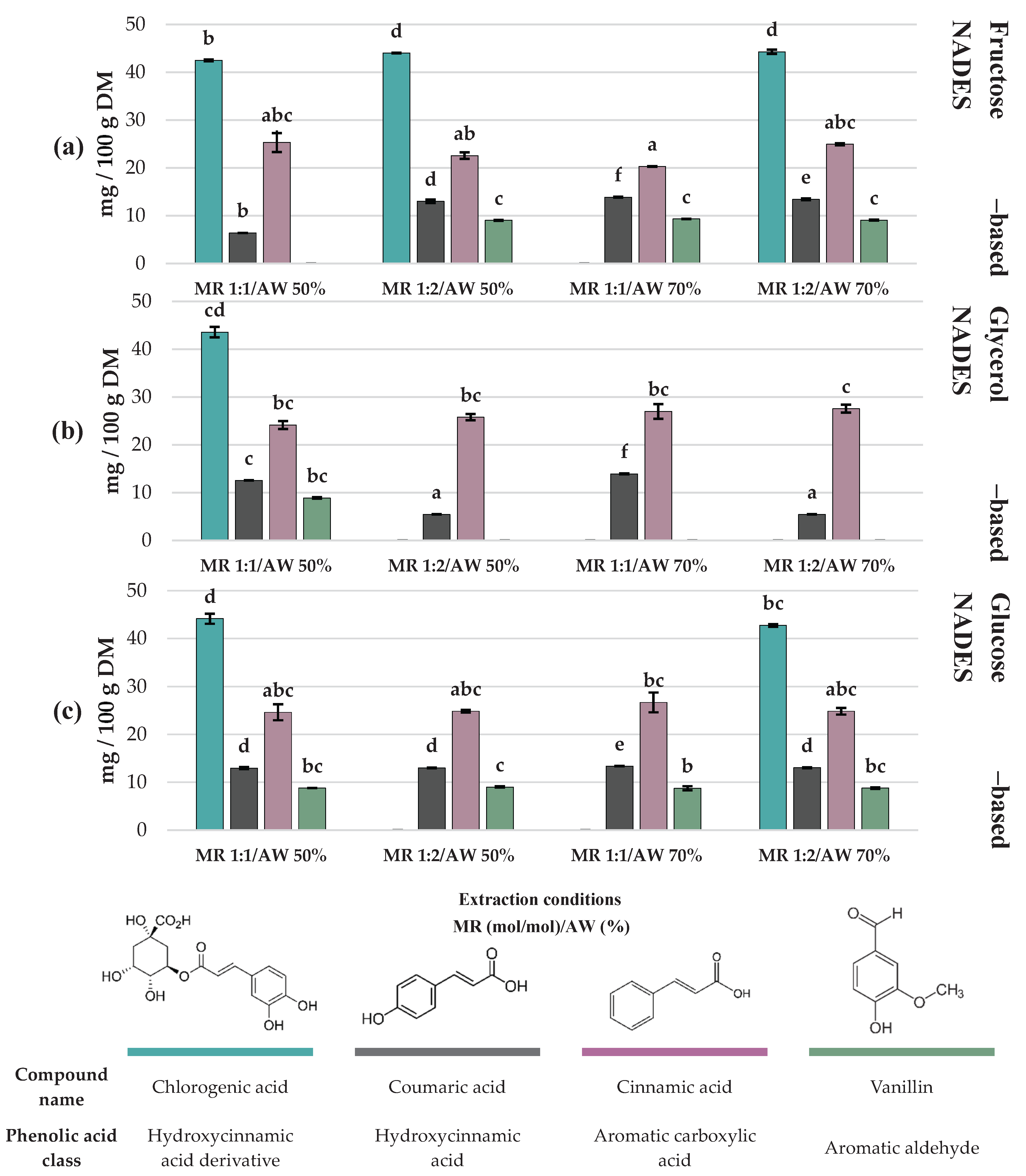
3.2. Evaluation of the Profile of Phenolic Compounds in C. aurantium By-Products Extracts
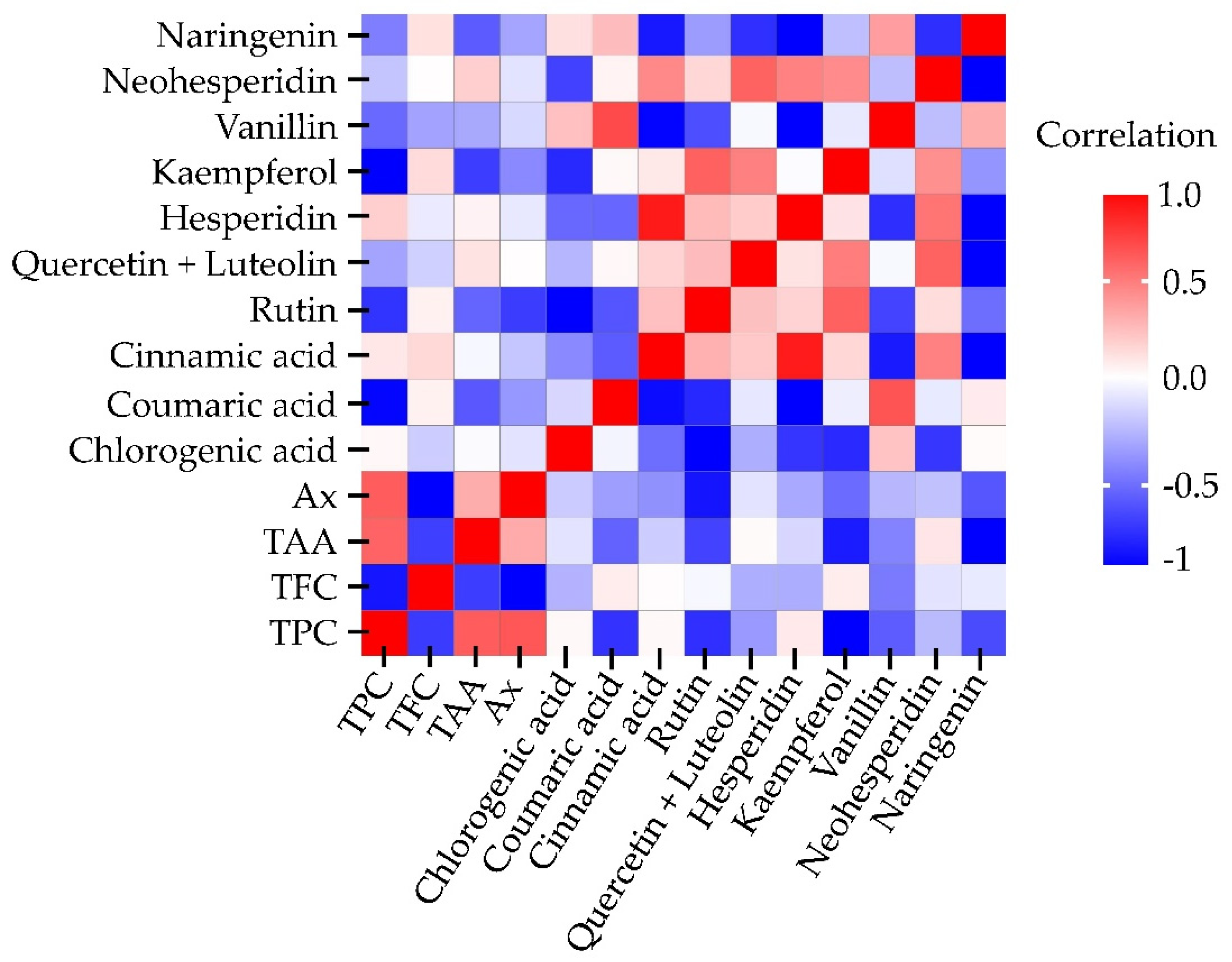
2.3. Pearson Correlation
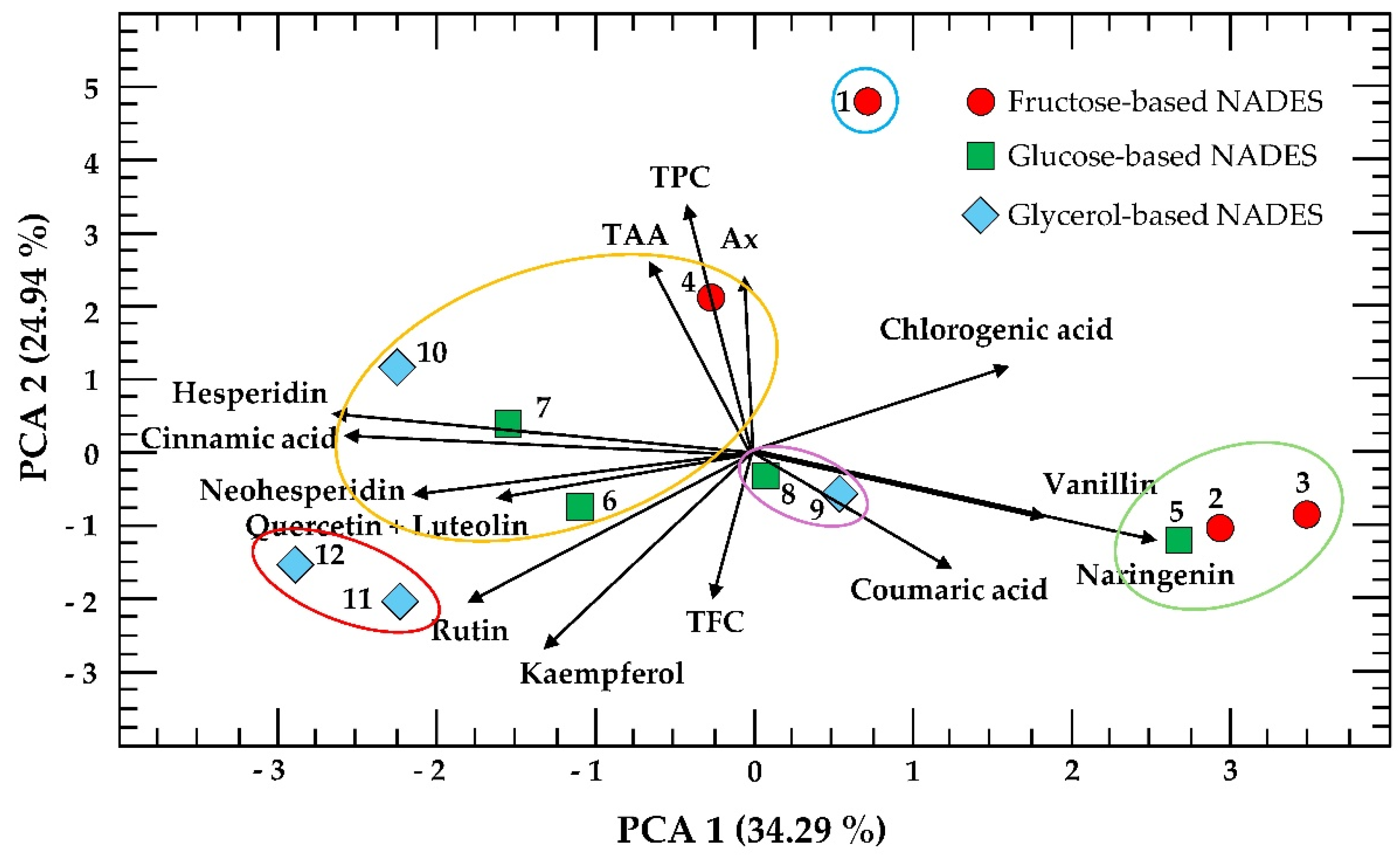
3.4. Principal Component Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Bitter Orange Drying Leaf Process
4.3. Experimental Design
4.4. NADES Preparation
4.5. Ultrasound-Assisted Extraction of Polyphenols Using NADES
4.6. Spectrophotometric Assays
4.6.1. Total Polyphenol Content Determination
4.6.2. Total Flavonoid Content Determination
4.6.3. Total Ascorbic Acid Determination
4.6.4. Antioxidant Capacity Assessment
4.7. Polyphenol Profile
4.8. Statical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rațu, R.N.; Veleșcu, I.D.; Stoica, F.; Usturoi, A.; Arsenoaia, V.N.; Crivei, I.C.; Postolache, A.N.; Lipșa, F.D.; Filipov, F.; Florea, A.M.; Chițea, M.A.; Brumă, I.S. Application of Agri-Food By-Products in the Food Industry. Agriculture 2023, 13(8), 1559. [Google Scholar] [CrossRef]
- Berenguer, C.V.; Andrade, C.; Pereira, J.A.M.; Perestrelo, R.; Câmara, J.S. Current Challenges in the Sustainable Valorization of Agri-Food Wastes: A Review. Processes 2023, 11(1), 20. [Google Scholar] [CrossRef]
- Reguengo, L.M.; Salgaço, M.K.; Sivieri, K.; Júnior, M.R.M. Agro-industrial by-products: Valuable sources of bioactive compounds. Food Research International 2022, 152, 110871. [Google Scholar] [CrossRef]
- Andrade, M.A.; Barbosa, C.H.; Shah, M.A.; Ahmad, N.; Vilarinho, F.; Khwaldia, K.; Silva, A.S.; Ramos, F. Citrus By-Products: Valuable Source of Bioactive Compounds for Food Applications. Antioxidants 2023, 12(1), 38. [Google Scholar] [CrossRef]
- Villar-Luna, H.; Santos-Cervantes, M.E.; Rodríguez-Negrete, E.A.; Méndez-Lozano, J.; Leyva-López, N.E. Economic and Social Impact of Huanglongbing on the Mexico Citrus Industry: A Review and Future Perspectives. Horticulturae 2024, 10(5), 481. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Kato, M. Pesticidal Activity of Citrus Fruits for the Development of Sustainable Fruit-Processing Waste Management and Agricultural Production. Plants 2025, 14(5), 754. [Google Scholar] [CrossRef]
- United Nations. FAOSTAT: Citrus fruit production. Available online: https://data.un.org/Data.aspx?d=FAO&f=itemCode%3A1804 (accessed on 10 October 2025).
- Shah, M.A.; Tariq, S.; Abuzar, S.M.; Ilyas, K.; Qadees, I.; Alsharif, I.; Anam, K.; Almutairi, R.T.; Al-Regaiey, K.A.; Babalghith, A.O.; Saleem, U.; Malik, Abd.; Almikhlaf, M.A.; Alanazi, Y.F.; Albalawi, M.; Sanches Silva, A. Peel waste of citrus fruits: A valuable and renewable source of polyphenols for the treatment of diabesity. Current Research in Biotechnology 2024, 7, 100204. [Google Scholar] [CrossRef]
- Government of Mexico. National Agricultural Planning (2016-2030). Available online: https://www.gob.mx/cms/uploads/attachment/file/257073/Potencial-C_tricos-parte_uno.pdf (accessed on 10 October 2025).
- Gao, L.; Zhang, H.; Yuan, C.H.; Zeng, L.H.; Xiang, Z.; Song, J.F.; Wang, H.G.; Jiang, J.P. Citrus aurantium ‘Changshan-huyou’—An ethnopharmacological and phytochemical review. Frontiers in Pharmacology 2022, 13, 983470. [Google Scholar] [CrossRef] [PubMed]
- Salazar, C.; Zizumbo-Villarreal, D.; Colunga-GarcíaMarín, P.; Brush, S. Contemporary Maya Food System in the Lowlands of Northern Yucatan. In Ethnobotany of Mexico. Ethnobiology; Lira, R., Casas, A., Blancas, J., Eds.; Springer: New York, NY, 2016. [Google Scholar] [CrossRef]
- Jorge de Jesús, C.-G.; Margarita, C.T.; Beatriz, C.-T.; Francisco Román, L.-Z.; Gerardo Alberto, M.-P.; Carlos Jesahel, V.-G. Improvements and Evaluation on Bitter Orange Leaves (Citrus aurantium L.) Solar Drying in Humid Climates. Sustainability 2021, 13(16), 9393. [Google Scholar] [CrossRef]
- Grabska-Kobyłecka, I.; Szpakowski, P.; Król, A.; Książek-Winiarek, D.; Kobyłecki, A.; Głąbiński, A.; Nowak, D. Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development. Nutrients 2023, 15(15), 3454. [Google Scholar] [CrossRef]
- Ran, Y.; Li, F.; Xu, Z.; Zeng, K.; Ming, J. Recent advances in dietary polyphenols (DPs): antioxidant activities, nutrient interactions, delivery systems, and potential applications. Food & Function 2024. [Google Scholar] [CrossRef]
- Palos-Hernández, A.; González-Paramás, A.M.; Santos-Buelga, C. Latest Advances in Green Extraction of Polyphenols from Plants, Foods and Food By-Products. Molecules 2025, 30(1), 55. [Google Scholar] [CrossRef]
- Coscarella, M.; Nardi, M.; Alipieva, K.; Bonacci, S.; Popova, M.; Procopio, A.; Scarpelli, R.; Simeonov, S. Alternative Assisted Extraction Methods of Phenolic Compounds Using NaDESs. Antioxidants 2024, 13(1), 62. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Suthar, K.J. Advances in the extraction of polyphenols using natural deep eutectic solvents: a comprehensive review. Chem. Pap 2025, 79, 3563–3585. [Google Scholar] [CrossRef]
- Freitas, D.S.; Ribeiro, A.; Cavaco-Paulo, A.; Silva, C. The Versatility of NADES Across Applications. Molecules 2025, 30(19), 3862. [Google Scholar] [CrossRef]
- Molnar, M.; Gašo-Sokač, D.; Komar, M.; Jakovljević Kovač, M.; Bušić, V. Potential of Deep Eutectic Solvents in the Extraction of Organic Compounds from Food Industry By-Products and Agro-Industrial Waste. Separations 2024, 11(1), 35. [Google Scholar] [CrossRef]
- Bautista, F. Clasificación de suelos de la península de Yucatán. In Los territorios kársticos de la península de Yucatán: caracterización, manejo y riesgos.; Bautista F (Coord.); Asociación Mexicana de Estudios sobre el Karst.: Ciudad de México, 2021; pp. 25–38. [Google Scholar]
- Instituto Nacional de Estadística y Geografía (INEGI). Climatología. Available online: https://www.inegi.org.mx/temas/climatologia/ (accessed on 10 October 2025).
- Pérez, M.; Dominguez-López, I.; Lamuela-Raventós, R.M. The chemistry behind the folin–ciocalteu method for the estimation of (poly) phenol content in food: Total phenolic intake in a mediterranean dietary pattern. Journal of agricultural and food chemistry 2023, 71(46), 17543–17553. [Google Scholar] [CrossRef]
- Edrisi, S.; Bakhshi, H. Separation of polyphenolic compounds from Citrus aurantium L. peel by deep eutectic solvents and their recovery using a new DES-based aqueous two-phase system. Journal of Molecular Liquids 2024, 402, 124790. [Google Scholar] [CrossRef]
- Shrivastav, G.; Prava Jyoti, T.; Chandel, S.; Singh, R. Eco-Friendly Extraction: Innovations, Principles, and Comparison with Traditional Methods. Separation & Purification Reviews 2024, 54, 241–257. [Google Scholar] [CrossRef]
- Abdallah, H.B.; Abbassi, A.; Trabelsi, A.; Krichen, Y.; Chekir-Ghedira, L.; Ghedira, K. Optimization of ultrasound-assisted extraction of polyphenols and flavonoids from Citrus aurantium L. var. amara Engl. fruit peel using response surface methodology. Biomass Conv. Bioref 2024, 14, 14139–14151. [Google Scholar] [CrossRef]
- Amador-Luna, V.M.; Herrero, M.; de la Parra, M.J.; Arribas, Á.G.; Ibáñez, E.; Montero, L. Maximizing the neuroprotection from Citrus aurantium leaves: Optimization of a blended extract from a sequential extraction process with compressed fluids and NADES. Advances in Sample Preparation 2025, 13, 100149. [Google Scholar] [CrossRef]
- Barp, L.; Višnjevec, A.M.; Moret, S. Pressurized liquid extraction: A powerful tool to implement extraction and purification of food contaminants. Foods 2023, 12, 2017. [Google Scholar] [CrossRef]
- Alizadeh, B.B.; Falah, F.; Vasiee, A.; TABATABAEI, Y.F.; Mortazavi, S.A. Antimicrobial effect of Citrus aurantium essential oil on some foodborne pathogens and determination of its chemical compounds, total phenolcontent, total flavonoids content and antioxidant potential. 2019; ISSN 2588-4824. [Google Scholar]
- Estrada-Sierra, N.A.; Rincon-Enriquez, G.; Urías-Silvas, J.E.; Bravo, S.D.; Villanueva-Rodríguez, S.J. Impact of ripening, harvest season, and the nature of solvents on antioxidant capacity, flavonoid, and p-synephrine concentrations in Citrus aurantium extracts from residue. Future Foods 2022, 6, 100153. [Google Scholar] [CrossRef]
- Bessarabov, V.; Lisovyi, V.; Lyzhniuk, V.; Derkach, T.; Kuzmina, G.; Gureyeva, S. Development and Characterisation of Polymeric Solid Dispersed Systems of Hesperidin, Obtained by Centrifugal Fibre Formation. Heliyon 2025, 11(4), e42702. [Google Scholar] [CrossRef]
- Liu, M.; Yu, X.; Qin, C.; Zhao, Q.; Wang, S.; Wang, H. Green and efficient extraction of Aurantii Fructus Immaturus flavonoids by natural deep eutectic solvents: Component identification, extraction mechanism and biological activity. Journal of Molecular Liquids 2025, 128543. [Google Scholar] [CrossRef]
- Fanali, C.; Della Posta, S.; Dugo, L.; Gentili, A.; Mondello, L.; De Gara, L. Choline-chloride and betaine-based deep eutectic solvents for green extraction of nutraceutical compounds from spent coffee ground. Journal of Pharmaceutical and Biomedical Analysis 2020, 189, 113421. [Google Scholar] [CrossRef]
- Shraim, A.M.; Ahmed, T.A.; Rahman, M.M.; Hijji, Y.M. Determination of total flavonoid content by aluminum chloride assay: A critical evaluation. Lwt 2021, 150, 111932. [Google Scholar] [CrossRef]
- Mansour, R. Determination of nutritional composition in citrus fruits (C. aurantium) during maturity. Nutrition & Food Science 2019, 49(2), 299–317. [Google Scholar] [CrossRef]
- Sir Elkhatim, K.A.; Elagib, R.A.A.; Hassan, A.B. Content of phenolic compounds and vitamin C and antioxidant activity in wasted parts of Sudanese citrus fruits. Foods 2018, 6, 1214–1219. [Google Scholar] [CrossRef]
- Tarikuzzaman, M.; Sagar, V.; Wong, M.J.; Lynam, J.G. Temperature Effects on Physiochemical Characteristics of Sugar--Based Natural Deep Eutectic Solvents. Advances in Materials Science and Engineering 2024, 6641317. [Google Scholar] [CrossRef]
- Ahmad, I.; Hussain-Shaikh, R.; Khurshid, A.; Khurshid, A.; Anwar-Baqai, Z. Photolysis of ascorbic acid in aqueous solution: A kinetic study. Baqai Journal of Health Sciences 2019, 22. [Google Scholar]
- Wołosiak, R.; Drużyńska, B.; Derewiaka, D.; Piecyk, M.; Majewska, E.; Ciecierska, M.; Worobiej, E.; Pakosz, P. Verification of the Conditions for Determination of Antioxidant Activity by ABTS and DPPH Assays—A Practical Approach. Molecules 2022, 27, 50. [Google Scholar] [CrossRef]
- Ramírez-Sucre, M.O.; Avilés-Betanzos, K.A.; López-Martínez, A.; Rodríguez-Buenfil, I.M. Evaluation of polyphenol profile from citrus peel obtained by natural deep eutectic solvent/ultrasound extraction. Processes 2024, 12(10), 2072. [Google Scholar] [CrossRef]
- Gulcin, İ.; Alwasel, S.H. DPPH radical scavenging assay. Processes 2023, 11(8), 2248. [Google Scholar] [CrossRef]
- Qader, I.B.; Laguerre, M.; Lavaud, A.; Tenon, M.; Prasad, K.; Abbott, A.P. Selective Extraction of antioxidants by formation of a deep eutectic mixture through mechanical mixing. ACS Sustainable Chemistry & Engineering 2023, 11, 4168–4176. [Google Scholar] [CrossRef]
- Mansinhos, I.; Gonçalves, S.; Rodríguez-Solana, R.; Ordóñez-Díaz, J.L.; Moreno-Rojas, J.M.; Romano, A. Ultrasonic-assisted extraction and natural deep eutectic solvents combination: A green strategy to improve the recovery of phenolic compounds from Lavandula pedunculata subsp. lusitanica (chaytor) franco. Antioxidants 2021, 10, 582. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods in enzymology 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Hagos, M.; Redi-Abshiro, M.; Chandravanshi, B.S.; Yaya, E.E. New analytical methods for the determination of ascorbic acid content in aqueous extracts of flesh, peel and seeds of pumpkin (Cucurbita maxima). Bulletin of the Chemical Society of Ethiopia 2022, 36(2), 277–290. [Google Scholar] [CrossRef]
- Chel-Guerrero, L.D.; Oney-Montalvo, J.E.; Rodríguez-Buenfil, I.M. Phytochemical characterization of by-products of habanero pepper grown in two different types of soils from Yucatán, Mexico. Plants 2021, 10, 779. [Google Scholar] [CrossRef]
- Avilés-Betanzos, K.A.; Cauich-Rodríguez, J.V.; González-Ávila, M.; Scampicchio, M.; Morozova, K.; Ramírez-Sucre, M.O.; Rodríguez-Buenfil, I.M. Natural Deep Eutectic Solvent Optimization to Obtain an Extract Rich in Polyphenols from Capsicum chinense Leaves Using an Ultrasonic Probe. Processes 2023, 11, 1729. [Google Scholar] [CrossRef]
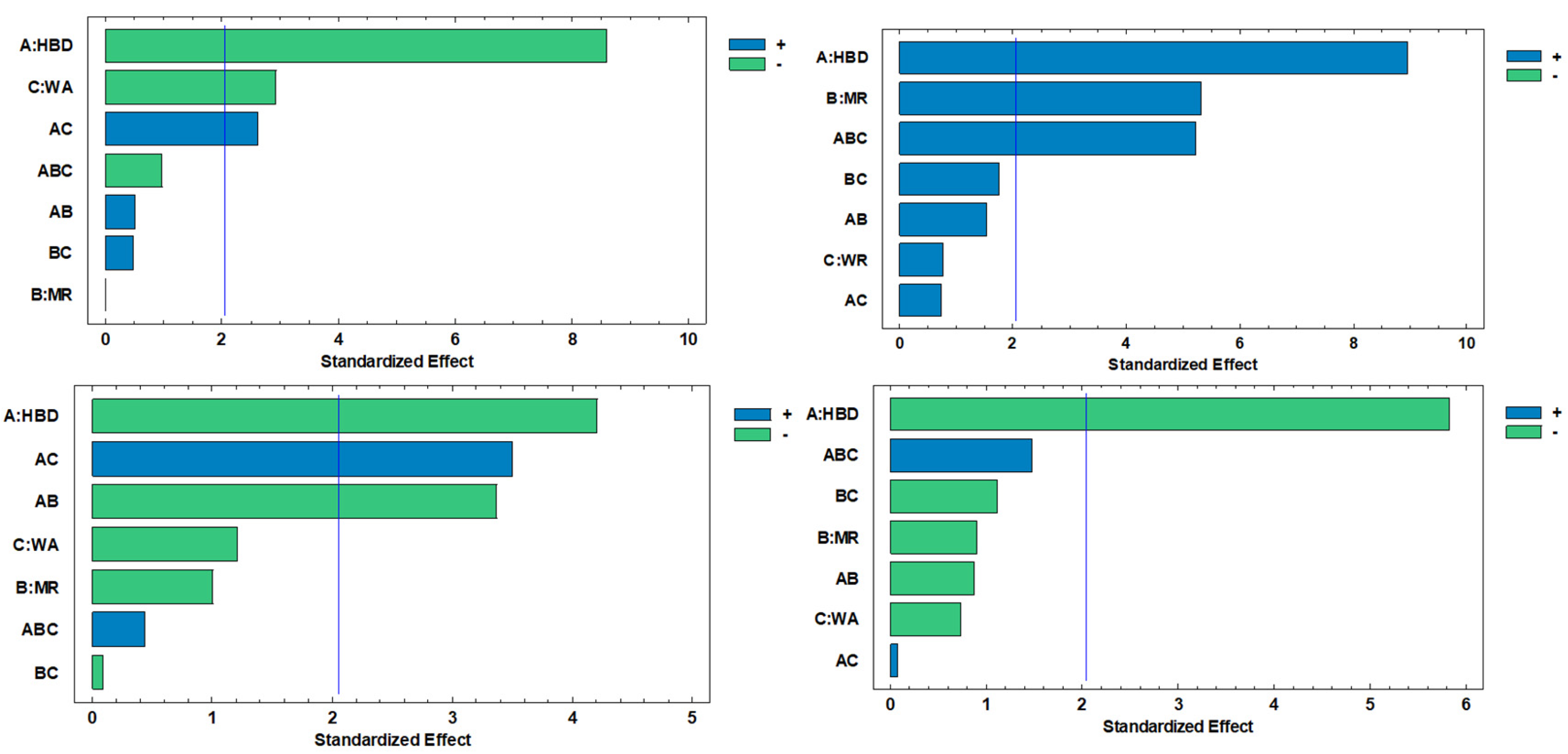
| Individual polyphenol | Main factors and interactions | |||||||
|---|---|---|---|---|---|---|---|---|
| A | B | C | AB | AC | BC | ABC | ||
| Protocatechuic acid | 0.0037 | 0.0137 | 0.0137 | 0.0037 | 0.0037 | 0.0137 | 0.0037 | |
| Catechin | 0.0057 | 0.0192 | 0.0192 | 0.0057 | 0.0057 | 0.0192 | 0.0057 | |
| Chlorogenic acid | 0.0001 | 0.9715 | 0.0009 | 0.0001 | 0.9388 | <0.0001 | 0.9629 | |
| Coumaric acid | 0.0238 | 0.0476 | 0.0368 | <0.0001 | 0.1007 | 0.0709 | 0.1149 | |
| Cinnamic acid | 0.0009 | 0.4800 | 0.2571 | 0.9053 | 0.0198 | 0.2370 | 0.0084 | |
| Rutin | 0.0001 | 0.0052 | 0.1023 | 0.4432 | 0.2213 | 0.1135 | 0.0179 | |
| Quercetin + Luteolin | 0.0025 | 0.0021 | 0.0229 | 0.0003 | 0.0138 | 0.8641 | 0.8109 | |
| Hesperidin | 0.0001 | 0.8547 | 0.6346 | 0.1349 | 0.0946 | 0.7030 | 0.0003 | |
| Kaempferol | <0.0001 | 0.0060 | 0.0005 | 0.0058 | 0.0057 | 0.0037 | 0.0120 | |
| Vanillin | 0.0018 | 0.9833 | 0.9733 | 0.0026 | 0.0020 | 0.9282 | 0.0021 | |
| Neohesperidin | 0.0271 | 0.0106 | 0.0014 | 0.0845 | 0.6498 | 0.0941 | 0.0079 | |
| Naringenin | 0.0124 | 0.2887 | 0.2423 | 0.9439 | 0.9439 | 0.2690 | 0.0124 | |
| #EXP | Encoded values | Real values | Response variables* | ||||
|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | HBD | MR (mol/mol) |
AW (%) |
||
| 1 | -1 | -1 | -1 | Fructose | 1 | 50 | Y1 |
| 2 | 0 | -1 | -1 | Glycerol | 1 | 50 | Y2 |
| 3 | 1 | -1 | -1 | Glucose | 1 | 50 | Y3 |
| 4 | -1 | 1 | -1 | Fructose | 2 | 50 | Y4 |
| 5 | 0 | 1 | -1 | Glycerol | 2 | 50 | Y5 |
| 6 | 1 | 1 | -1 | Glucose | 2 | 50 | Y6 |
| 7 | -1 | -1 | 1 | Fructose | 1 | 70 | Y7 |
| 8 | 0 | -1 | 1 | Glycerol | 1 | 70 | Y8 |
| 9 | 1 | -1 | 1 | Glucose | 1 | 70 | Y9 |
| 10 | -1 | 1 | 1 | Fructose | 2 | 70 | Y10 |
| 11 | 0 | 1 | 1 | Glycerol | 2 | 70 | Y11 |
| 12 | 1 | 1 | 1 | Glucose | 2 | 70 | Y12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




