Submitted:
21 October 2025
Posted:
22 October 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Results
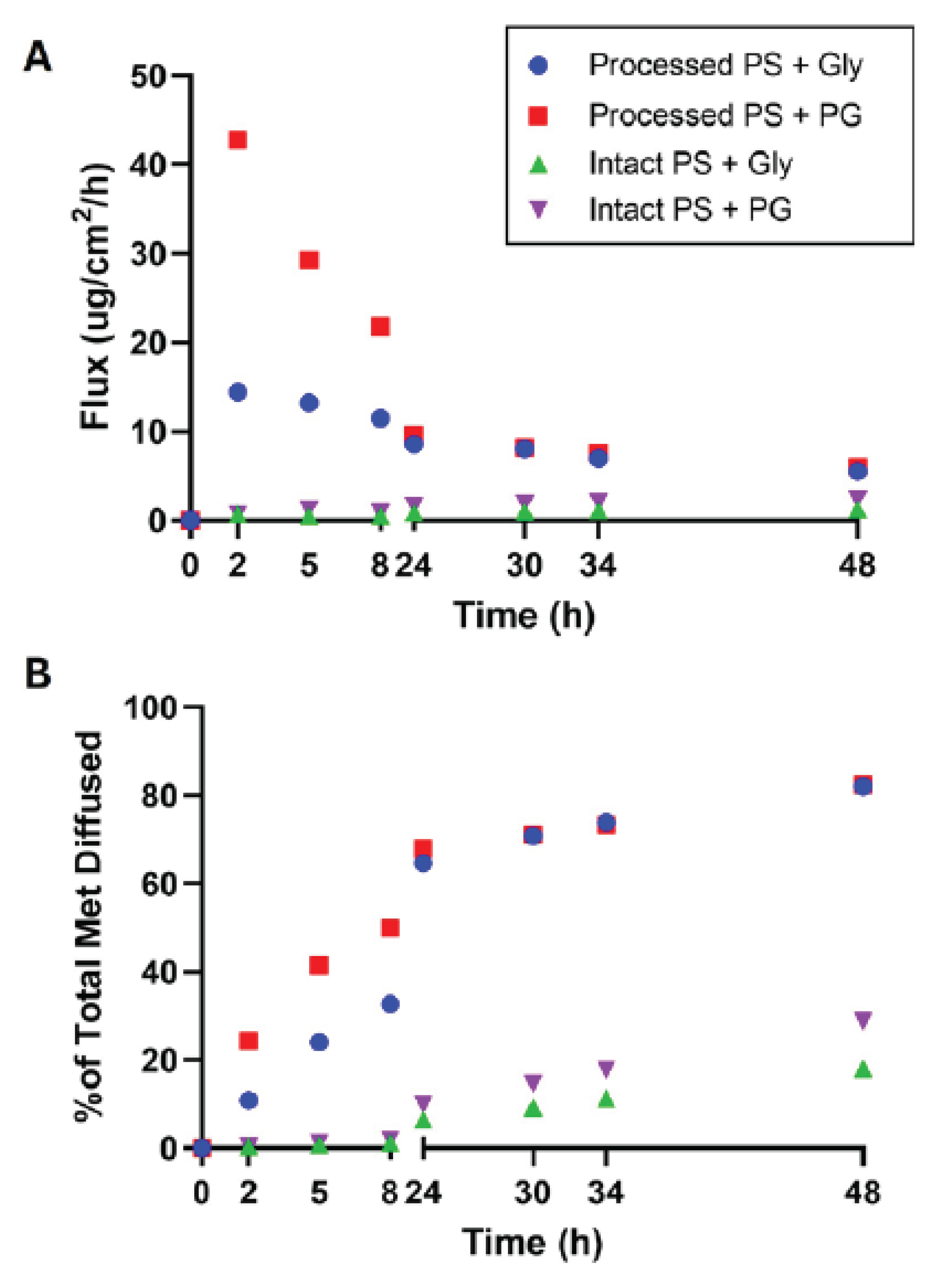
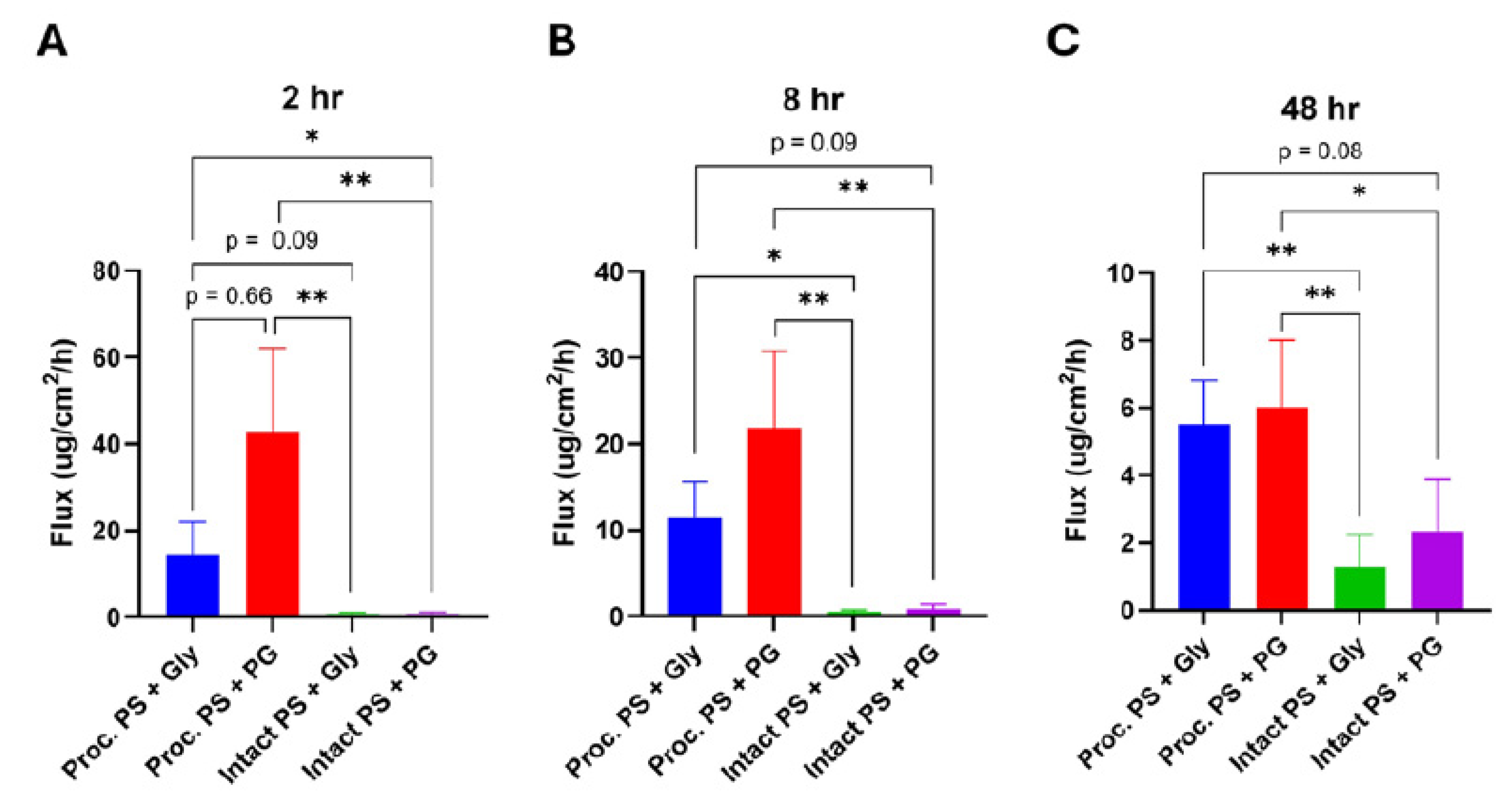
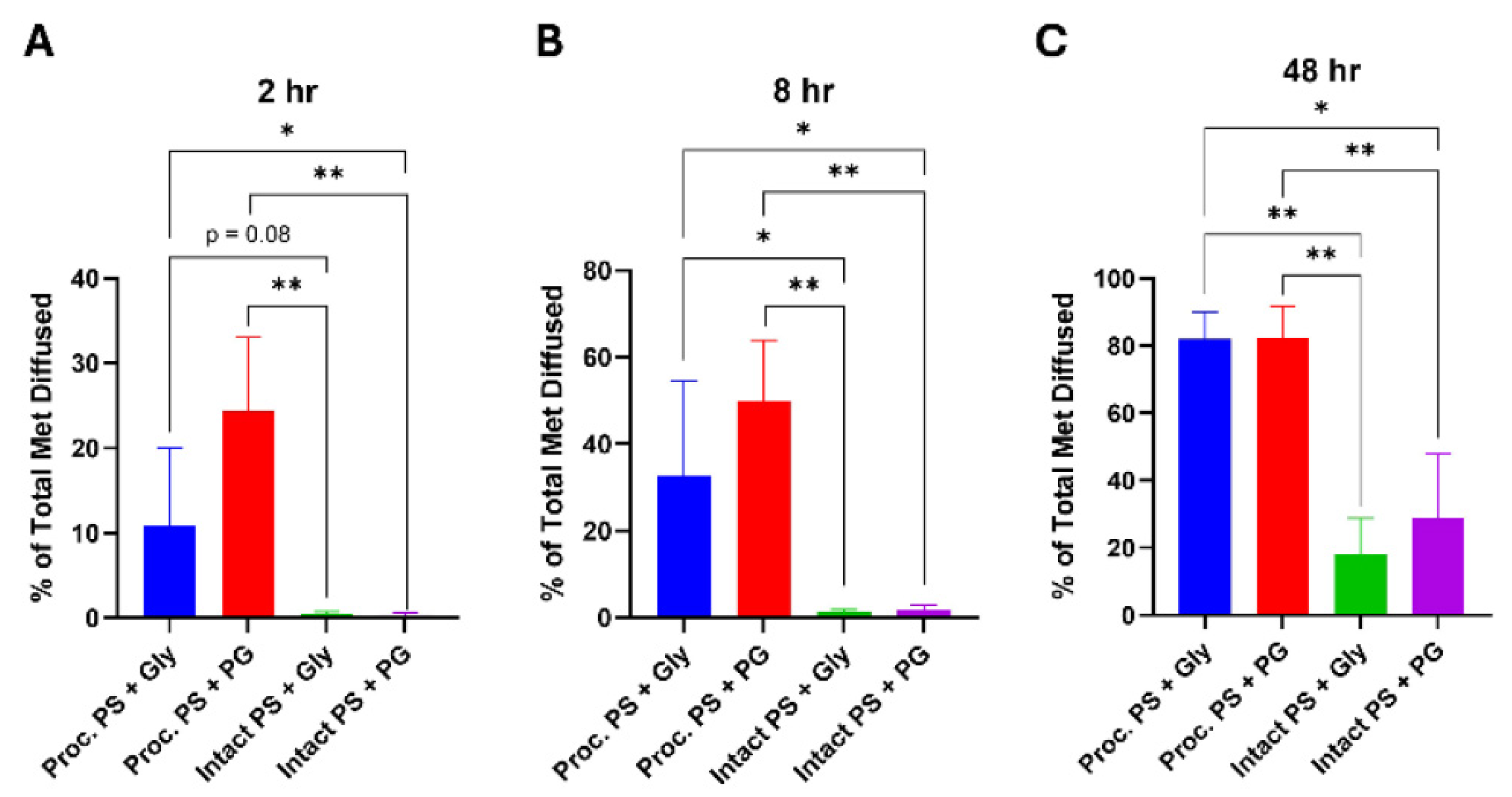
Processed Porcine Skin Exhibits Dramatically Higher Permeation
Intact Porcine Skin Maintains Barrier Properties
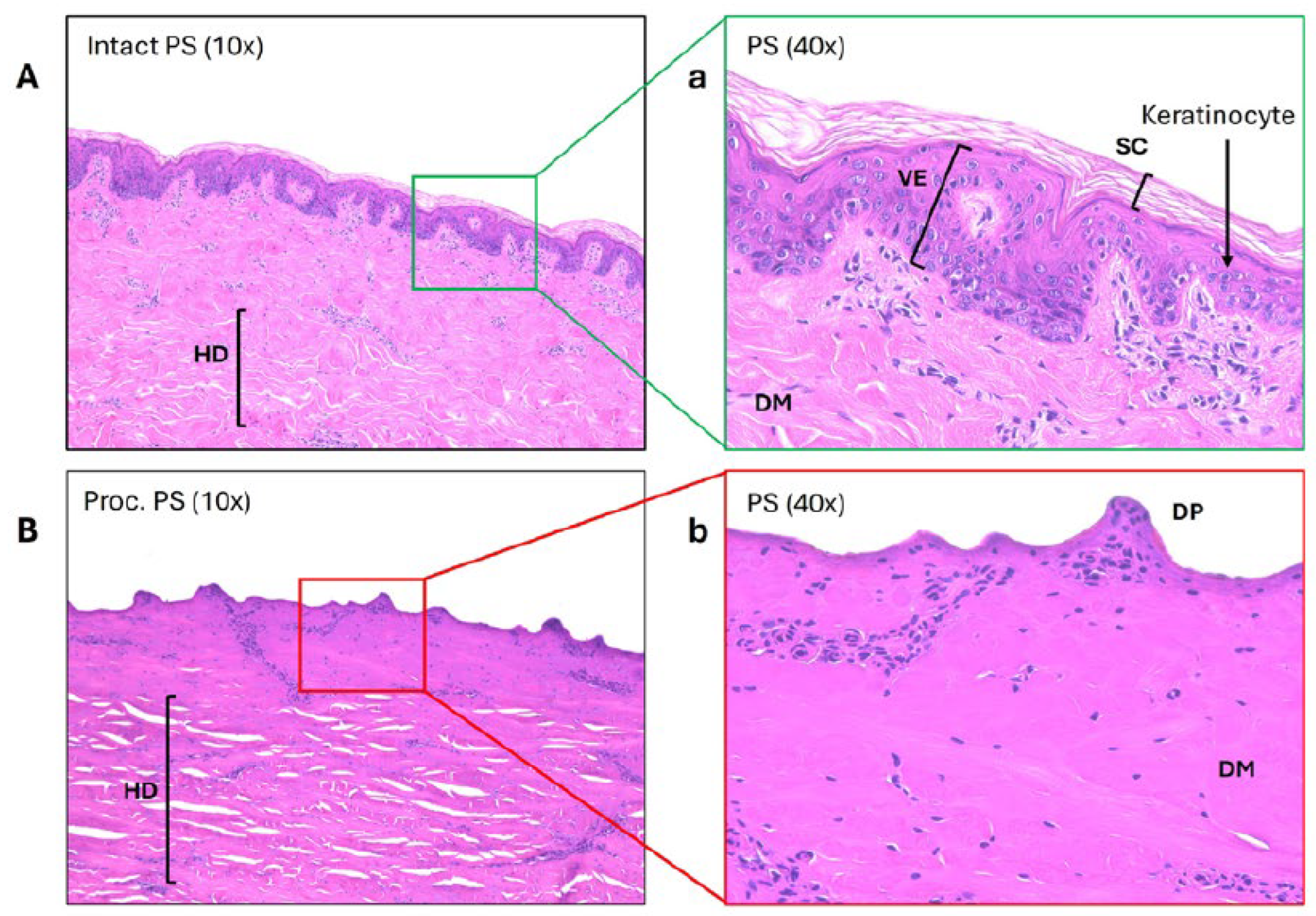
Histological Evidence Confirms Barrier Loss in Processed Skin
Discussion
Material and Methods
Chemicals and Reagents
Acquisition of Porcine Skin
Preparation of Porcine Skin for Permeation Testing
Franz Diffusion Cell Permeation Testing
Histology
Experimental Groups
Instrumentation and Chromatographic Conditions
Statistical Analysis
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Simon, G.A.; Maibach, H.I. The pig as an experimental animal model of percutaneous permeation in man: qualitative and quantitative observations--an overview. Skin Pharmacol Appl Skin Physiol 2000, 13, 229-234. [CrossRef]
- Gallagher, S.; Kruger, U.; Josyula, K.; Rahul; Gong, A.; Song, A.; Sweet, R.; Makled, B.; Parsey, C.; Norfleet, J.; et al. Thermally damaged porcine skin is not a surrogate mechanical model of human skin. Sci Rep 2022, 12, 4565. [CrossRef]
- Silva, I.R.; Lima, F.A.; Reis, E.C.O.; Ferreira, L.A.M.; Goulart, G.A.C. Stepwise Protocols for Preparation and Use of Porcine Ear Skin for in Vitro Skin Permeation Studies Using Franz Diffusion Cells. Curr Protoc 2022, 2, e391. [CrossRef]
- Beck, H.; Bracher, M. Standard Protocol: Percutaneous Absorption/Penetration In Vitro Pig Skin. Colipa Guidelines: Guidelines for Percutaneous Absorption/Penetration. Rte de Chesalles 21, CH 1723 Marly 1. 1995.
- Report. Skin Absorption: In vitro Method, OECD Guideline for the Testing of Chemicals (428). 2004.
- Diembeck, W.; Grimmert, A. Standard Protocol: Percutaneous Absorption/Penetration In Vitro Excised Pig Skin Colipa Guidelines (Hamburg, Germany) 1995.
- Iliopoulos, F.; Sil, B.C.; Moore, D.J.; Lucas, R.A.; Lane, M.E. 3-O-ethyl-l-ascorbic acid: Characterisation and investigation of single solvent systems for delivery to the skin. Int J Pharm X 2019, 1, 100025. [CrossRef]
- Makuch, E.; Nowak, A.; Gunther, A.; Pelech, R.; Kucharski, L.; Duchnik, W.; Klimowicz, A. The Effect of Cream and Gel Vehicles on the Percutaneous Absorption and Skin Retention of a New Eugenol Derivative With Antioxidant Activity. Front Pharmacol 2021, 12, 658381. [CrossRef]
- Nowak, A.; Muzykiewicz-Szymanska, A.; Peruzynska, M.; Kucharska, E.; Kucharski, L.; Jakubczyk, K.; Niedzwiedzka-Rystwej, P.; Stefanowicz-Hajduk, J.; Drozdzik, M.; Majtan, J. Assessment of in vitro skin permeation and accumulation of phenolic acids from honey and honey-based pharmaceutical formulations. BMC Complement Med Ther 2025, 25, 43. [CrossRef]
- Keck, C.M.; Abdelkader, A.; Pelikh, O.; Wiemann, S.; Kaushik, V.; Specht, D.; Eckert, R.W.; Alnemari, R.M.; Dietrich, H.; Brussler, J. Assessing the Dermal Penetration Efficacy of Chemical Compounds with the Ex-Vivo Porcine Ear Model. Pharmaceutics 2022, 14. [CrossRef]
- Afshar, K.; Adibfard, S.; Nikbakht, M.H.; Rastegarnasab, F.; Pourmahdi-Boroujeni, M.; Abtahi-Naeini, B. A Systematic Review on Clinical Evidence for Topical Metformin: Old Medication With New Application. Health Sci Rep 2024, 7, e70281. [CrossRef]
- Agarwal, S.; Krishnamurthy, K. Histology, Skin. StatPearls, StatPearls Publishing: Treasure Island (FL) 2023.
- Berg, E.P. In Critical Points Affecting Fresh Pork Quality within the Packing Plant. Pork Information Gateway, Eilert, S., Ed. 2006.
- Brashears, M.; Miller, M.; Brooks, T. Pork Harvest Process, International Center for Food Industry Excellence (ICFIE) ,Texas Tech University: Lubbock, Texas, USA.
- Andrews, S.N.; Jeong, E.; Prausnitz, M.R. Transdermal delivery of molecules is limited by full epidermis, not just stratum corneum. Pharm Res 2013, 30, 1099-1109. [CrossRef]
- Park, J.H.; Lee, J.W.; Kim, Y.C.; Prausnitz, M.R. The effect of heat on skin permeability. Int J Pharm 2008, 359, 94-103. [CrossRef]
- Cioce, A.; Cavani, A.; Cattani, C.; Scopelliti, F. Role of the Skin Immune System in Wound Healing. Cells 2024, 13. [CrossRef]
- Sintov, A.C. Cumulative evidence of the low reliability of frozen/thawed pig skin as a model for in vitro percutaneous permeation testing. Eur J Pharm Sci 2017, 102, 261-263. [CrossRef]
- Meira, A.S.; Battisel, A.P.; Teixeira, H.F.; Volapato, N.M. Evaluation of porcine skin layers separation methods, freezing storage and anatomical site in in vitro percutaneous absorption studies using penciclovir formulations. Journal of Drug Delivery Science and Technology 2020, 60, 101926.
- FDA. In Vitro Permeation Test Studies for Topical Drug Products Submitted in ANDAs: Guidance for Industry. 2022.
- Huttunen, K.M.; Rautio, J.; Leppanen, J.; Vepsalainen, J.; Keski-Rahkonen, P. Determination of metformin and its prodrugs in human and rat blood by hydrophilic interaction liquid chromatography. J Pharm Biomed Anal 2009, 50, 469-474. [CrossRef]
- PermeGear, I. Diffusion testing fundamentals. 2015, PermeGear, Inc, 1-8.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



