Submitted:
26 September 2025
Posted:
09 October 2025
You are already at the latest version
Abstract
Background: Tinnitus has been increasingly reported during the COVID-19 pandemic, following both infection and vaccination. While these reports suggest that pandemic-related factors may influence the onset or worsening of tinnitus, the perceptual characteristics and functional consequences of such cases remain poorly understood. This study examined the nature, severity, and communication-related impact of self-reported tinnitus following COVID-19 vaccination. Methods: A total of 770 adults who self-reported new or worsened tinnitus after vaccination completed a structured online survey between August 2021 and May 2023. Standardized instruments included the Tinnitus Functional Index (TFI), selected items from the Tinnitus Handicap Inventory (THI), the Speech, Spatial and Qualities of Hearing Scale (SSQ), and visual analog scales assessing loudness discomfort and hyperacusis-related symptoms. Analyses included descriptive statistics, chi-square tests, t-tests, and correlation matrices. Results: Respondents reported moderate to severe tinnitus-related distress, with the greatest impacts on emotional control, sleep, and relaxation. Many described communication difficulties in noisy or multi-talker environments, despite relatively preserved spatial hearing and sound quality. A substantial subset endorsed hyperacusis symptoms, including sound-induced annoyance, fear, and intolerance. Correlation analyses revealed strong associations between emotional, cognitive, and auditory domains, underscoring the multidimensional burden of tinnitus in this population. Conclusions: Self-reported tinnitus after COVID-19 vaccination was associated with distress, sleep disruption, and communication difficulties that mirror patterns commonly observed in tinnitus more broadly. Although causality cannot be determined, these findings highlight the importance of comprehensive audiological and psychological assessment for individuals reporting auditory complaints after vaccination and support the inclusion of functional hearing outcomes in tinnitus evaluation protocols.
Keywords:
1. Introduction
2. Methods
2.1. Survey Design
2.2. Data Collection
2.3. Data Cleaning
2.4. Statistical Analysis
3. Results
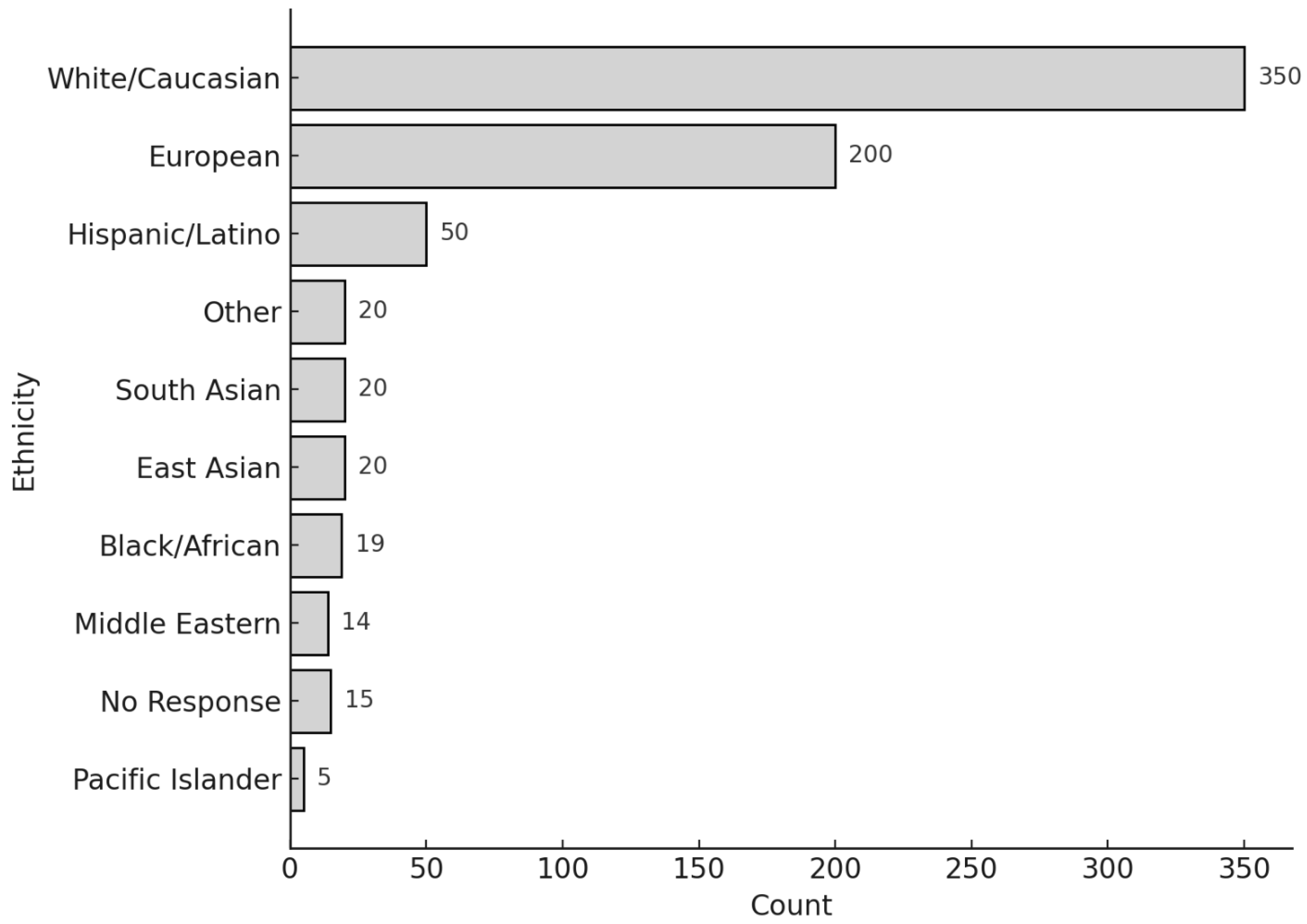
3.1. Participant Characteristics
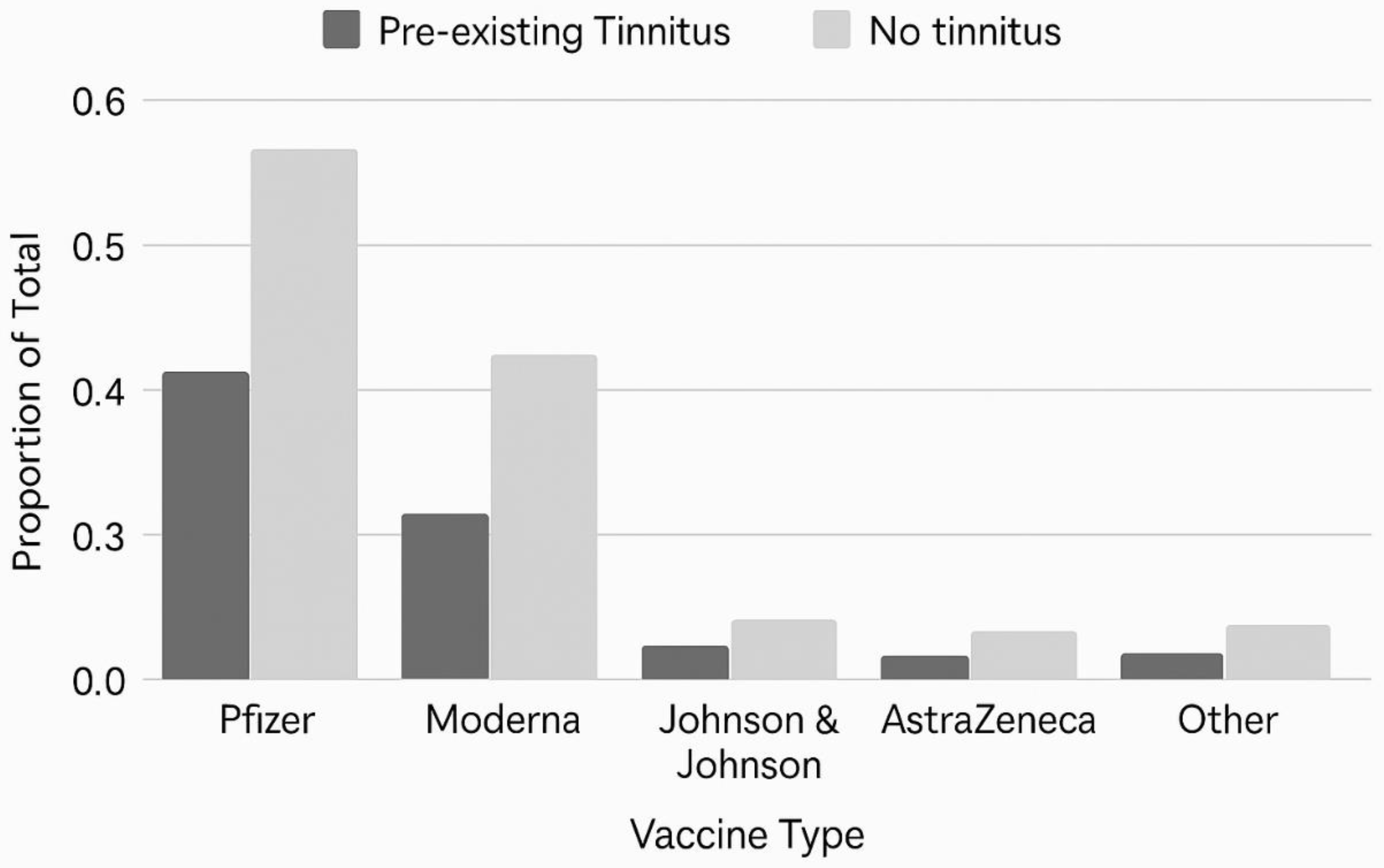
3.2. Vaccination Profiles
3.3. Medical and Audiological Follow-Up
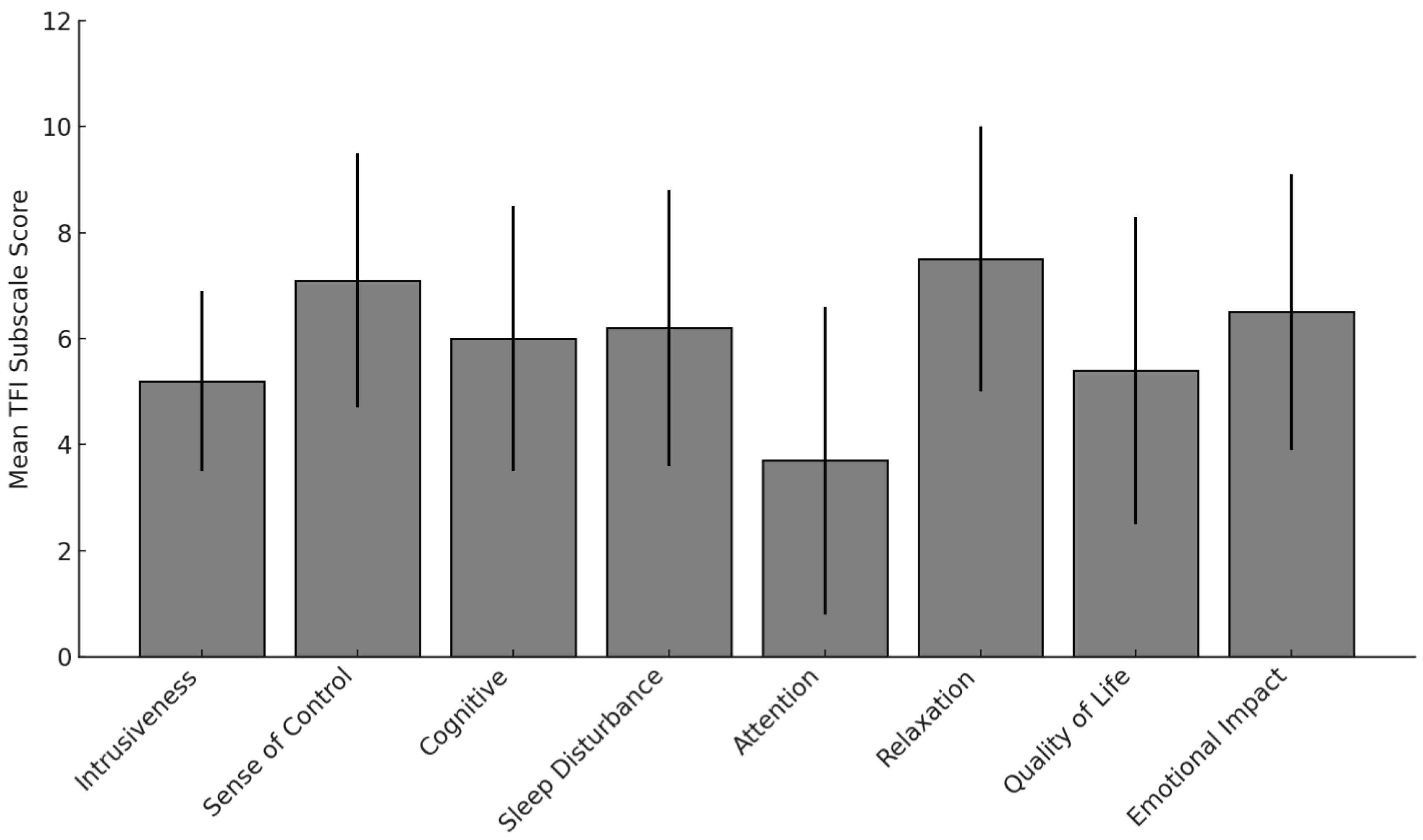
3.3.1. Tinnitus Functional Index (TFI)
3.3.2. Intrusiveness and Sense of Control
3.3.2.1. Cognitive, Sleep, and Emotional Burden
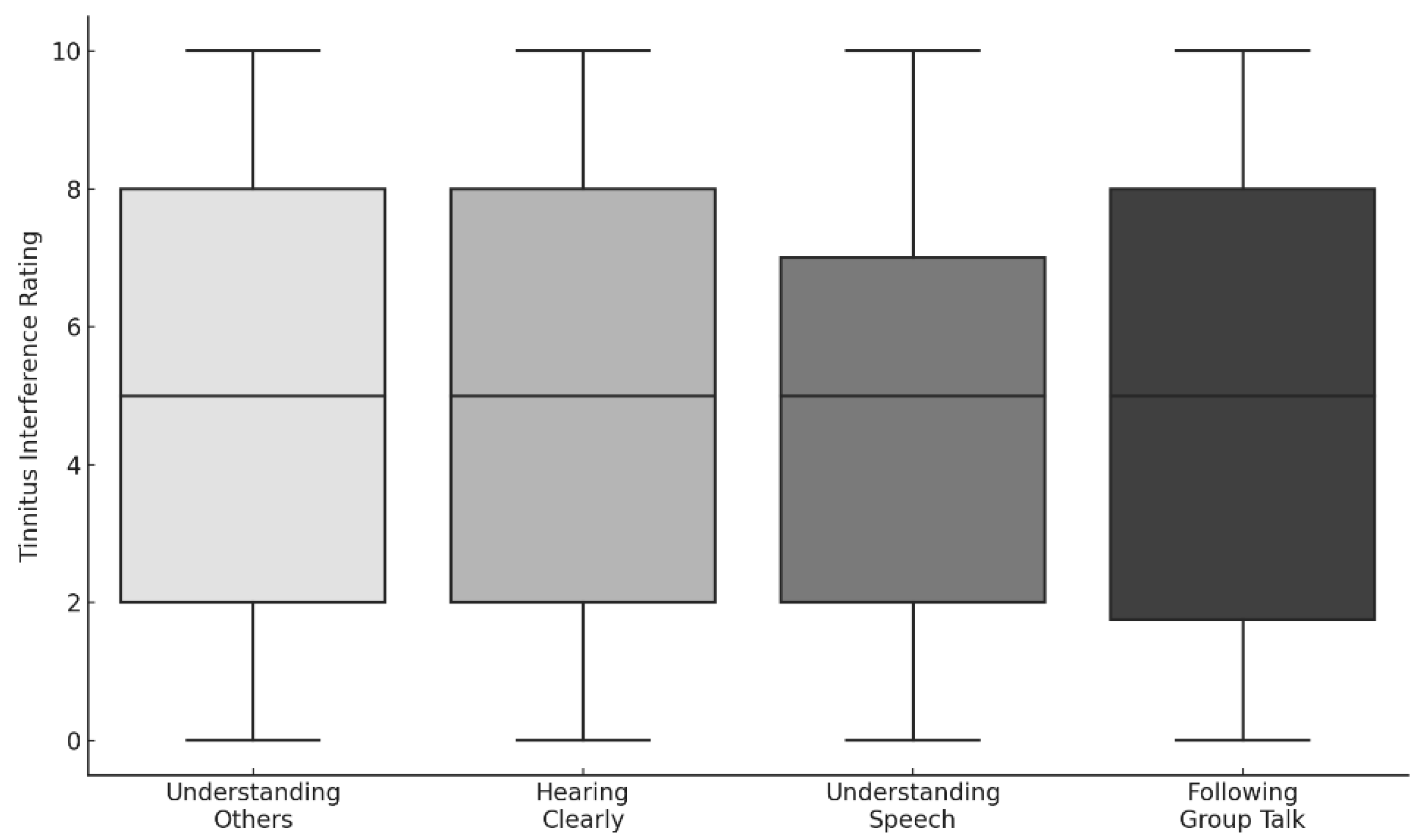
3.3.2.2. Auditory and Communication Interference
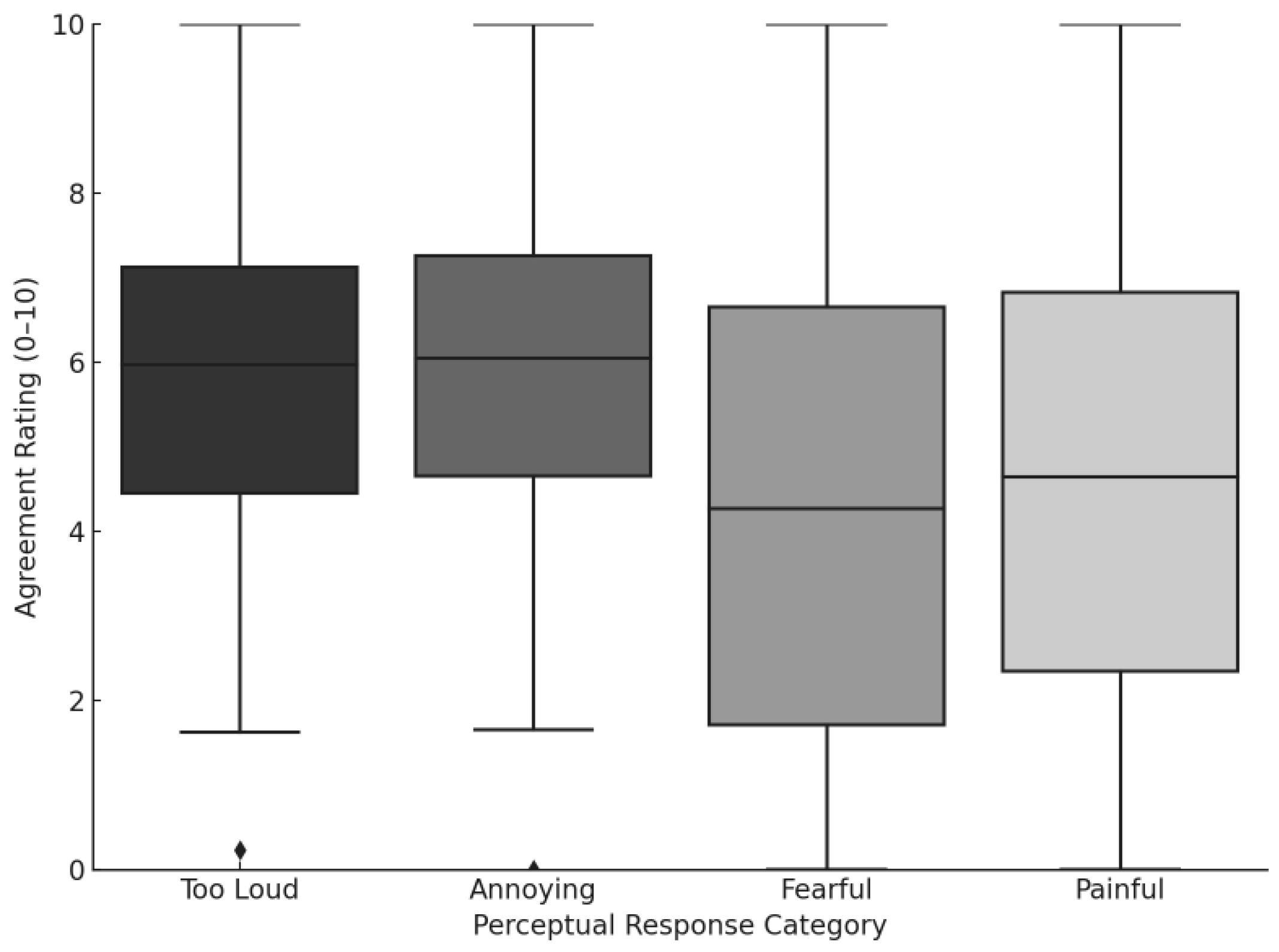
3.3.3. Hyperacusis and Loudness Sensitivity
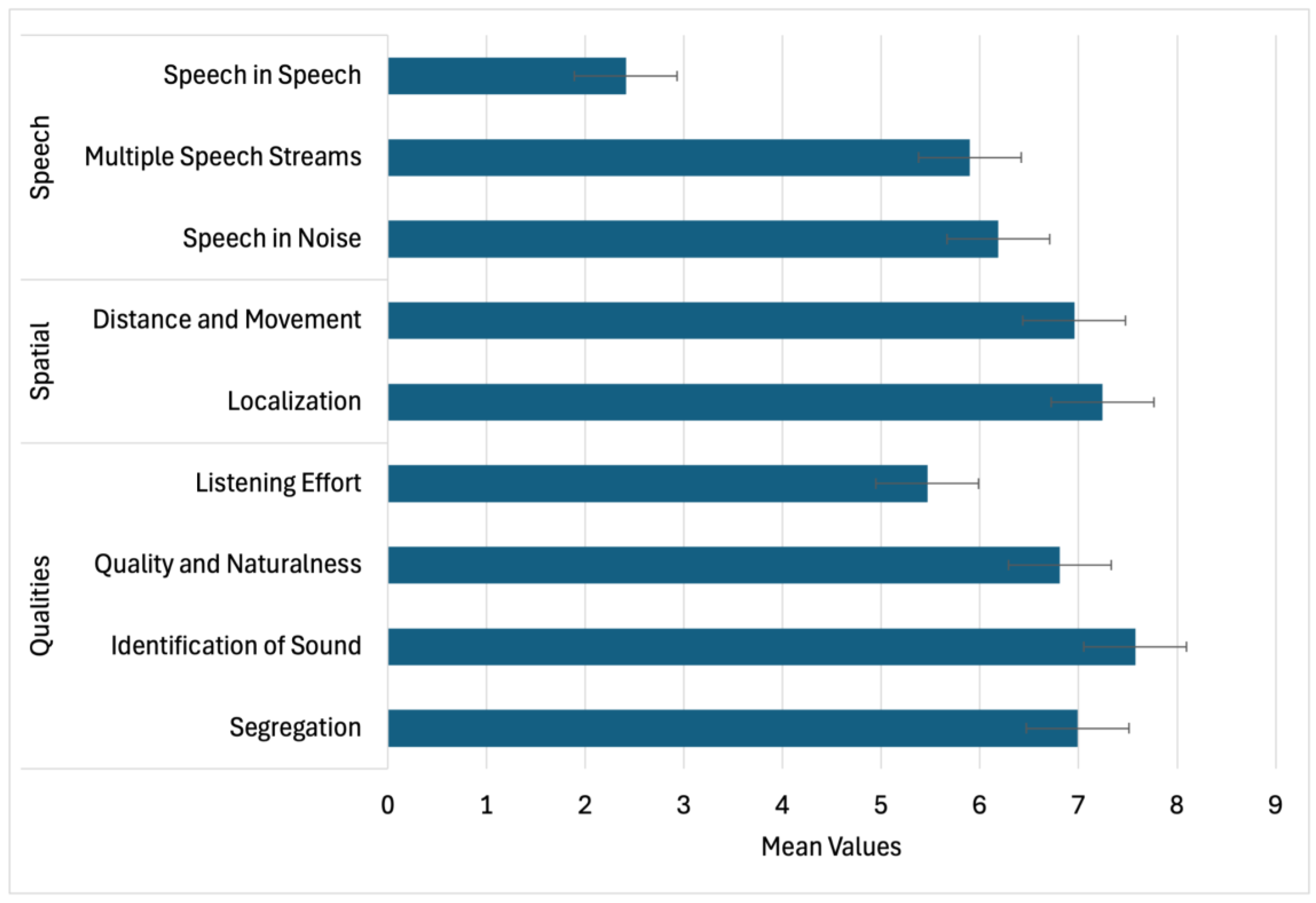
3.3.4. Functional Hearing Outcomes (SSQ)
3.3.5. Loudness Ratings and Lateralization
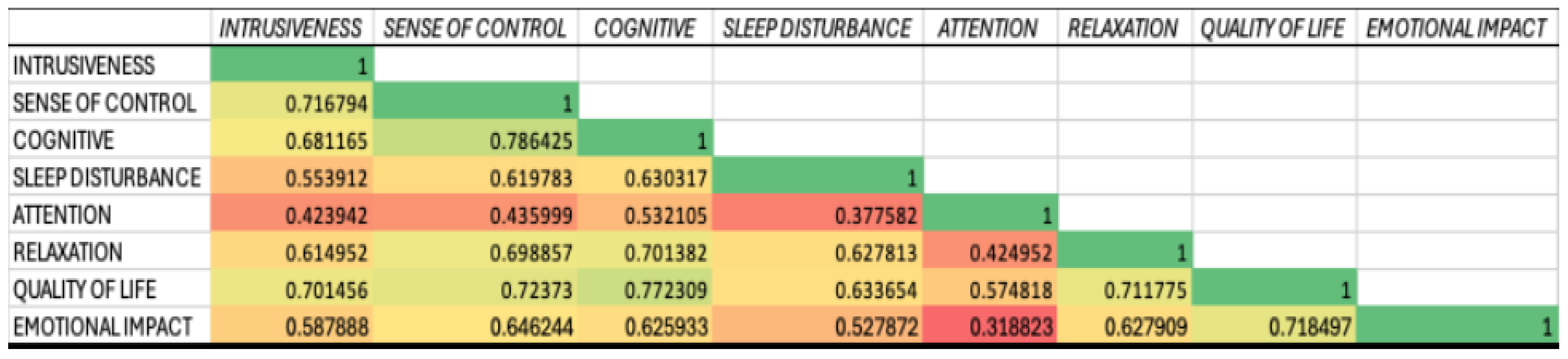
3.3.6. Contextualizing Symptom Burden
4. Discussion
4.1. Tinnitus-Related Impairments Across Functional Domains
4.2. Speech Perception and Listening Effort in Complex Environments
4.3. Perceived Loudness and Lateralization
4.4. Pandemic-Related Factors and Post-Vaccination Reports
4.5. Clinical and Research Implications
4.6. Limitations and Future Directions
5. Conclusions
Declaration of the Use of Artificial Intelligence (AI) in Manuscript Preparation
Conflicts of Interest Statement
Ethics Statement
References
- Baguley, D., McFerran, D., & Hall, D. (2013). Tinnitus. The Lancet, 382(9904), 1600–1607. [CrossRef]
- Beukes, E. W., Manchaiah, V., & Allen, P. M. (2021). The impact of COVID-19 and the associated restrictions on people with pre-existing chronic tinnitus. International Journal of Audiology, 60(9), 707–714. [CrossRef]
- Beukes, E. W., Baguley, D. M., Jacquemin, L., & Andersson, G. (2020). Changes in tinnitus experiences during the COVID-19 pandemic. Frontiers in Public Health, 8, 592878. [CrossRef]
- Figueiredo, R. R., Azevedo, A. A., de Oliveira Penido, N., & Canto, G. (2022). Tinnitus and COVID-19: An overview of preexisting evidence and perspectives for future research. Journal of Otology, 17(2), 77–82. [CrossRef]
- Gatehouse, S., & Noble, W. (2004). The Speech, Spatial and Qualities of Hearing Scale (SSQ). International Journal of Audiology, 43(2), 85–99. [CrossRef]
- Hiller, W., & Goebel, G. (2006). Factors influencing tinnitus loudness and annoyance. Archives of Otolaryngology–Head & Neck Surgery, 132(12), 1323–1330. [CrossRef]
- Langguth, B., Kreuzer, P. M., Kleinjung, T., & De Ridder, D. (2013). Tinnitus: Causes and clinical management. The Lancet Neurology, 12(9), 920–930. [CrossRef]
- Maihoub, S., Mavrogeni, P., Molnár, V., & Molnár, A. (2025). Tinnitus and its comorbidities: a comprehensive analysis of their relationships. Journal of Clinical Medicine, 14(4), 1285. [CrossRef]
- Maihoub, S., Mavrogeni, P., Répássy, G. D., & Molnár, A. (2025). Exploring how blood cell levels influence subjective tinnitus: A cross-sectional case-control study. Audiology Research, 15(3), 72. [CrossRef]
- Meikle, M. B., Henry, J. A., Griest, S. E., Stewart, B. J., Abrams, H. B., McArdle, R.,... & Vernon, J. A. (2012). The tinnitus functional index: Development of a new clinical measure for chronic, intrusive tinnitus. Ear and Hearing, 33(2), 153–176. [CrossRef]
- Moon, I. J., Shim, H. J., Park, H. J., Jung, J. Y., Lee, S. H., & Lee, W. S. (2015). Tinnitus and speech perception in noise: The role of hearing loss and cognition. Journal of Audiology & Otology, 19(2), 82–87. [CrossRef]
- Newman, C. W., Jacobson, G. P., & Spitzer, J. B. (1996). Development of the Tinnitus Handicap Inventory. Archives of Otolaryngology–Head & Neck Surgery, 122(2), 143–148.
- Noreña, A. J. (2011). An integrative model of tinnitus based on a central gain controlling neural sensitivity. Neuroscience & Biobehavioral Reviews, 35(5), 1089–1109. [CrossRef]
- Rauschecker, J. P., Leaver, A. M., & Mühlau, M. (2010). Tuning out the noise: Limbic-auditory interactions in tinnitus. Neuron, 66(6), 819–826. [CrossRef]
- Roberts, L. E., Eggermont, J. J., Caspary, D. M., Shore, S. E., Melcher, J. R., & Kaltenbach, J. A. (2010). Ringing ears: The neuroscience of tinnitus. The Journal of Neuroscience, 30(45), 14972–14979. [CrossRef]
- Tai, Y., Jain, N., Kim, G., & Husain, F. T. (2024). Tinnitus and COVID-19: Effect of infection, vaccination, and the pandemic. Frontiers in Public Health, 12, 1508607. [CrossRef]
- Tan, B. K. J., Ng, J. Y., & Puvanendran, R. (2022). Post-COVID-19 tinnitus: A review. American Journal of Otolaryngology, 43(5), 103418. [CrossRef]
- Taziki Balajelini, M., Sadoughi, M., & Mirzaei, M. (2022). Evaluation of tinnitus prevalence and characteristics in healthcare workers after COVID-19 vaccination. Noise & Health, 24(115), 17–22. [CrossRef]
- Tyler, R. S., Pienkowski, M., Roncancio, E. R., Jun, H. J., Brozoski, T., Dauman, N.,... & Moore, B. C. J. (2014). A review of hyperacusis and future directions: Part I. Definitions and manifestations. American Journal of Audiology, 23(4), 402–419. [CrossRef]
- Viola, P., Ralli, M., Pisani, D., & Petrucci, A. G. (2021). Tinnitus and equilibrium disorders in COVID-19 patients: Preliminary results. European Archives of Oto-Rhino-Laryngology, 278(10), 3725–3730. [CrossRef]
- Wang, H., Zhou, Y., Lin, H., & Ma, X. (2022). A review of SARS-CoV-2 infection and vaccine-associated otologic symptoms. Frontiers in Neurology, 13, 928099. [CrossRef]
- Wang, W., Yellamsetty, A., Edmonds, R. M., Barcavage, S. R., & Bao, S. (2024). COVID-19 vaccination-related tinnitus is associated with pre-vaccination metabolic disorders. Frontiers in Pharmacology. [CrossRef]
- Wilson, P. H., Henry, J., Bowen, M., & Haralambous, G. (1991). Tinnitus reaction questionnaire: psychometric properties of a measure of distress associated with tinnitus. Journal of Speech and Hearing Research, 34(1), 197–201.
- Yellamsetty, A. (2023). COVID-19 vaccination effects on tinnitus and hyperacusis: Longitudinal case study. The International Tinnitus Journal, 27(2), 253–258. [CrossRef]
- Yellamsetty, A., & Gonzalez, V. (2024). Understanding COVID-19-related tinnitus: Emerging evidence and implications. Tinnitus Today.
- Yellamsetty, A., Egbe-Etu, E., & Bao, S. (2024). Impact of COVID-19 vaccination on tinnitus onset and severity: A comprehensive survey study. Frontiers in Audiology and Otology, 3, 1509444. [CrossRef]
| Vaccine Manufacturer | All Survey Cases | U.S. Survey Cases |
|---|---|---|
| Pfizer | 427 | 328 |
| Moderna | 256 | 239 |
| Johnson & Johnson | 55 | 48 |
| AstraZeneca | 28 | 2 |
| Other | 4 | 1 |
| Vaccine Manufacturer | 1 Dose | 2 Doses | 3 Doses | 4+ Doses | Total Cases | Pre-existing Tinnitus (n) |
|---|---|---|---|---|---|---|
| Pfizer | 16 | 187 | 177 | 47 | 427 | 113 |
| Moderna | 7 | 94 | 122 | 33 | 256 | 67 |
| Johnson & Johnson | 31 | 10 | 10 | 4 | 55 | 17 |
| AstraZeneca | 18 | 8 | 2 | 0 | 28 | 4 |
| Type of Evaluation | Total (n=373) | Male (n=162) | Female (n=199) | Other/Unspecified (n=12) |
|---|---|---|---|---|
| Audiometric Testing | 161 | 73 | 85 | 3 |
| ENT Consultation | 105 | 52 | 50 | 3 |
| MRI | 89 | 40 | 46 | 3 |
| Blood Tests | 54 | 26 | 27 | 1 |
| Vestibular (VNG, Balance) Tests | 27 | 12 | 14 | 1 |
| Neurologist Evaluation | 26 | 13 | 12 | 1 |
| Other (e.g., TMJ, Cardiology) | 18 | 7 | 10 | 1 |
| SSQ Domain | Item Description | χ² (df) | p-value |
|---|---|---|---|
| Speech | Speech in Noise (TV on) | 125.61 | < 0.0001 |
| Multiple Streams (TV & person) | 57.89 | < 0.0001 | |
| Speech in Speech (Crowd) | 67.16 | < 0.0001 | |
| Group Speech (Restaurant) | 55.33 | < 0.0001 | |
| Switching Speakers | 88.26 | < 0.0001 | |
| Spatial | Localization (Dog barking) | 248.56 | < 0.0001 |
| Distance Judgement | 168.11 | < 0.0001 | |
| Directionality (Coming vs. Going) | 188.84 | < 0.0001 | |
| Qualities | Segregation (Jumbled Sounds) | 272.88 | < 0.0001 |
| Identifying Instruments | 311.27 | < 0.0001 | |
| Sound Naturalness (Clarity) | 165.83 | < 0.0001 | |
| Listening Effort | 46.94 | < 0.0001 |
| Speech | Spatial | Qualities | |
|---|---|---|---|
| Speech | 1.00 | 0.73*** | 0.81*** |
| Spatial | — | 1.00 | 0.77*** |
| Qualities | — | — | 1.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




