Submitted:
07 October 2025
Posted:
08 October 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Composition of Hydrogel Films
2.1. Natural Biopolymers: Chitosan, Alginate, Gelatin, and Others
2.2. Synthetic and Semi-Synthetic Polymers in Hydrogel Formulations
2.3. Composite and Multifunctional Hydrogel Films
3. Synthesis and Fabrication Techniques of Hydrogel Films
3.1. Chemical Crosslinking and Physical Gelation Methods
3.2. Fabrication Methods of Hydrogel Films
3.2.1. Film Formation
3.2.2. Preparation Methods
- Solvent Casting [92]: This straightforward technique involves dissolving the polymer in a solvent, casting the solution onto a substrate, and allowing the solvent to evaporate. The evaporation process leads to film formation and, in the case of hydrogels, is followed by gelation.
- Dip Coating [93]: The substrate is immersed in a polymer solution and withdrawn at a controlled rate, forming a thin film as the solution adheres to the surface.
- Spin Coating [14]: A small volume of polymer solution is dropped onto the center of a substrate, which is then rotated at high speed. Centrifugal force spreads the solution evenly, forming a uniform thin film.
- Spray Coating [94]: The polymer solution is atomized and sprayed onto the substrate, allowing for rapid and scalable film deposition.
- Blade Coating [95]: A blade is used to spread the polymer solution across the substrate, controlling film thickness through blade height and solution viscosity.
- Bar Coating [96]: Similar to blade coating, but uses a cylindrical bar wrapped with wire to distribute the solution evenly across the substrate.
- Slot Die Coating [97]: The polymer solution is dispensed through a narrow slit (die) directly onto the moving substrate, allowing for precise control over film thickness and uniformity.
- Photolithography [98]: A photosensitive polymer is exposed to UV light through a patterned mask, enabling the creation of microstructured hydrogel films with high spatial resolution.
- 3D Printing [99]: Hydrogel structures are built layer-by-layer using techniques such as extrusion-based printing or stereolithography (SLA), which uses light to polymerize photosensitive resins with high precision.
Solvent Casting Method
Dip Coating Method
Spin Coating Method
- Spin speed: Higher speeds generally produce thinner films.
- Viscosity of the solution: More viscous solutions tend to yield thicker coatings.
- Solvent evaporation rate: Faster evaporation can lead to quicker solidification and thinner films.
- Volume of the applied solution: Larger volumes may result in thicker layers.
Spray Coating and Blade Coating Methods
Blade Coating and Bar Coating Methods
- A 90° blade angle generates high shear, ideal for producing thin and uniform coatings.
- A 45° angle offers a balance between removing excess material and retaining some on the surface, yielding medium-thickness films.
- Angles between 15° and 30° reduce shear pressure, allowing for thicker coatings, which are beneficial when working with high-viscosity formulations.
Bar Coating Methods
Slot Die Coating Methods
Photolithography Methods
3D Printing Techniques
4. Unique Properties of Hydrogel Films
4.1. Thin-Film Architecture and Flexibility
4.2. High Water Content
4.4. Surface Adhesion and Conformability to Tissues
4.5. Biocompatibility of Hydrogel Films for Biomedical Applications
4.6. Biodegradability of Hydrogel Films for Biomedical Applications
- Biomedical Applications of Hydrogel Films
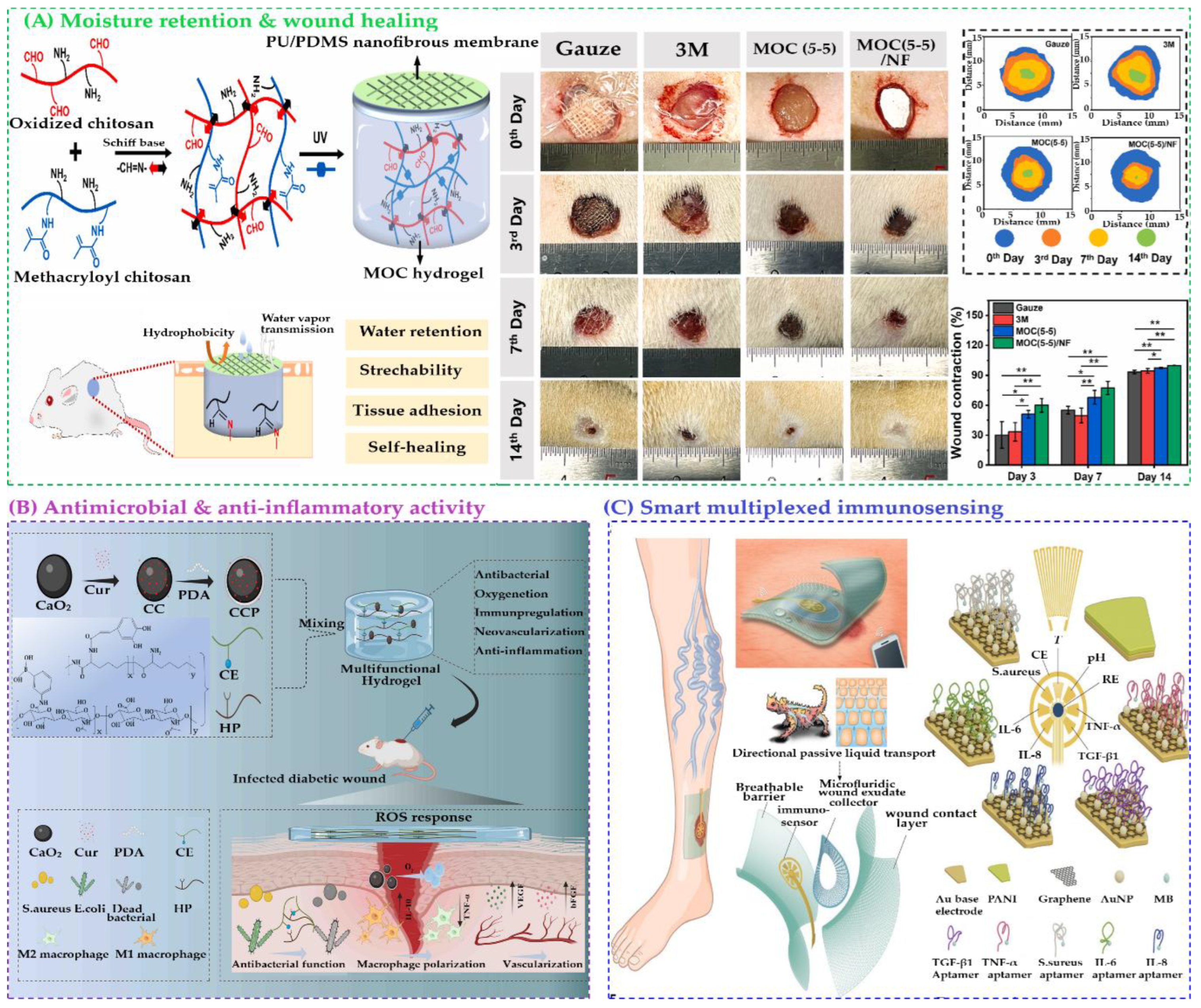
4.7. Wound Dressings: Moisture Retention, Antimicrobial, Anti-Inflammatory Incorporation, and Smart Monitoring
4.8. Hydrogel Films as Cell Culture
- Advanced Substrates for Cell Culture
- Hydrogel Films Mimicking Basement Membrane for Cell Culture
4.9. Drug Delivery Systems via Hydrogel Films
- Controlled and Sustained Release
- Transdermal and Mucosal Delivery Platforms
- Hydrogel Film-Based Multi-Drug Loading and Release Kinetics
4.10. Tissue Engineering
- Hydrogel Films as Barrier Layers in Wound Healing
- Hydrogel Films for Hemostasis and Anti-Adhesion in Wound Healing
- Structural Scaffolding & Mechanical Reinforcement
4.11. Ophthalmic Applications
- Contact Lenses and Corneal Patches
- Regenerative and Bioactive Potential
4.12. Ocular Drug Delivery Films
- Challenges in Conventional Ocular Delivery
- Hydrogel Film Technologies
- Nanoparticle-Enhanced Hydrogel Films
- Clinical Applications and Innovations
- Dry eye syndrome: Films loaded with lubricants and anti-inflammatory agents [235].
- Glaucoma: Sustained release of prostaglandin analogs to reduce intraocular pressure [238].
- Post-surgical care: Antibiotic-loaded films to prevent infection and promote healing [240].
- Retinal diseases: Intravitreal hydrogel implants for long-term drug delivery [239].
- Bioelectronic hydrogel films for real-time monitoring and feedback-controlled release
- Personalized hydrogel formulations using AI-guided design and bioprinting
- Stem cell and exosome-loaded hydrogel films for regenerative ophthalmology [232].
4.13. Implant Coatings and Biosensors
- Implant Coatings
4.14. Hydrogel Films Integrated Biosensors
- Self-healing hydrogel coatings for long-term implant durability
- Stimuli-responsive biosensors for dynamic health monitoring
- 3D-printed hydrogel interfaces for personalized implant design
- Bioelectronic hydrogel platforms for integrated sensing and stimulation
- These innovations are paving the way for next-generation biomedical devices that are minimally invasive, highly adaptive, and clinically effective.
4.15. Anti-fouling and Biointegration
- Anti-fouling Properties
- Biointegration and Tissue Compatibility
4.16. Responsive Films for Diagnostics
- Mechanisms of Responsiveness
- Colorimetric sensors: DNAzyme-crosslinked hydrogels enable visual detection of hydrogen peroxide (H2O2) through peroxidase-like activity, offering a simple and regenerable platform for environmental and biomedical monitoring [260].
- Electrochemical biosensors: Hydrogel films embedded with aptamers or antibodies can detect analytes such as glucose, lactate, and pathogens with high precision [260].
- Optical biosensors: Holographic hydrogel sensors diffract light in response to analyte-induced changes in refractive index, enabling label-free and real-time detection [261].
- Recent innovations include aptamer-functionalized hydrogels for continuous plasmonic biomonitoring, capable of detecting small molecules like vancomycin with high sensitivity and stability in physiological fluids [262].
- Wearable and Implantable Diagnostics
- Real-time sensing and feedback
- Wireless data transmission
- Integration with therapeutic platforms
- AI-guided signal interpretation
5. Recent Advances in Hydrogel Films for Biomedical Applications
5.1. Stimuli-Responsive Hydrogel Films
- Enzyme-responsive systems have shown promise in site-specific drug delivery and diagnostic imaging. For example, hydrogels composed of chitosan, hyaluronic acid, PEGDA, and GelMA degrade selectively in the presence of MMP-2 and hyaluronidase, releasing doxorubicin at tumor sites while sparing healthy cells. These systems also incorporate fluorescent dyes and superparamagnetic iron oxide nanoparticles (SPIONs) for dual optical and MRI-based diagnostics, demonstrating their theranostic potential [265].
- Multi-stimuli-responsive hydrogels react to combinations of triggers such as pH, temperature, light, and magnetic fields, offering precise control over therapeutic actions. These systems are being applied in cancer therapy, wound healing, and biosensing [266,267]. For instance, hydrogels that respond to acidic pH and elevated temperatures—common features of tumor microenvironments—can release chemotherapeutics only at diseased sites, reducing systemic toxicity [266]. Similarly, light-responsive hydrogels allow spatiotemporal control of drug release or activation of therapeutic agents using external light sources [148].
- In diagnostics, these hydrogels convert environmental changes into optical, electrochemical, or mechanical signals. They can detect biomarkers such as glucose, lactate, or inflammatory enzymes, and are being integrated into wearable devices and implantable sensors [268].
5.2. Nanocomposite and Hybrid Hydrogel Films
5.3. 3D and 4D Printing of Hydrogel Films
5.4. Biofunctionalization and Smart Materials
5.5. Integration with Wearable and Flexible Electronics
6. Challenges and Limitations of Hydrogel Films for Biomedical Applications
6.1. Mechanical Durability and Tear Resistance
6.2. Sterilization and Storage Stability
6.3. Regulatory and Clinical Translation
6.4. Cost and Scalability of Production
7. Future Perspectives of Hydrogel Films for Biomedical Applications
7.1. Personalized and Patient-Specific Hydrogel Films
7.2. AI-Guided Design and Optimization
7.3. Sustainable and Biodegradable Materials
7.4. Clinical Trials and Commercialization Pathways
8. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Declaration of generative AI and AI-assisted technologies in the writing process
References
- Gajurel, B.; Tamang, K.B.; Das, D.; Adhikari, R. Advances in synthetic strategies and applications of polymeric hydrogels. Polymer Engineering & Science 2025, 65, 2803–2840. [Google Scholar] [CrossRef]
- Segneanu, A.E.; Bejenaru, L.E.; Bejenaru, C.; Blendea, A.; Mogosanu, G.D.; Bita, A.; Boia, E.R. Advancements in Hydrogels: A Comprehensive Review of Natural and Synthetic Innovations for Biomedical Applications. Polymers (Basel) 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Choi, W.S.; Jeong, J.O. A Review of Advanced Hydrogel Applications for Tissue Engineering and Drug Delivery Systems as Biomaterials. Gels 2024, 10. [Google Scholar] [CrossRef]
- Ubaldini, A.; Calistri, S. Advances in Hydrogel Film Fabrication and Functional Applications Across Biomedical and Environmental Fields. Applied Sciences 2025, 15, 9579. [Google Scholar] [CrossRef]
- Le, H.H.; Tran, V.T.; Mredha, M.T.I.; Na, J.Y.; Seon, J.-K.; Jeon, I. Thin-film hydrogels with superior stiffness, strength, and stretchability. Extreme Mechanics Letters 2020, 37, 100720. [Google Scholar] [CrossRef]
- Choi, S.M.; Shin, E.J.; Zo, S.M.; Kummara, M.R.; Kim, C.M.; Kumar, A.; Bae, H.J.; Sood, A.; Han, S.S. Development of Scalable Elastic Gelatin Hydrogel Films Crosslinked with Waterborne Polyurethane for Enhanced Mechanical Properties and Strain Recovery. Gels 2025, 11, 49. [Google Scholar] [CrossRef]
- Raeisi, A.; Farjadian, F. Commercial hydrogel product for drug delivery based on route of administration. Front Chem 2024, 12, 1336717. [Google Scholar] [CrossRef]
- Labie, H.; Blanzat, M. Hydrogels for dermal and transdermal drug delivery. Biomaterials Science 2023, 11, 4073–4093. [Google Scholar] [CrossRef]
- Alavi, S.E.; Panah, N.; Page, F.; Gholami, M.; Dastfal, A.; Ajay Sharma, L.; Ebrahimi, H. Hydrogel-based therapeutic coatings for dental implants. European Polymer Journal 2022, 181, 111652. [Google Scholar] [CrossRef]
- Yuk, H.; Wu, J.; Zhao, X. Hydrogel interfaces for merging humans and machines. Nature Reviews Materials 2022, 7. [Google Scholar] [CrossRef]
- Periyasamy, T.; Asrafali, S.P.; Lee, J. Hydrogels for Translucent Wearable Electronics: Innovations in Materials, Integration, and Applications. Gels 2025, 11. [Google Scholar] [CrossRef]
- Khan, S.; Maryam, L.; Gulzar, A.; Mansoor, M.A.; Iqbal, M. smart and active hydrogels in biotechnology—synthetic techniques and applications. Journal of Materials Science 2024, 59, 16449–16471. [Google Scholar] [CrossRef]
- Xu, Z.; Deng, J.; Gao, D.; Du, Y.; Zhang, Y.; Lai, Y. Stimuli-responsive biomedical polymeric films for tissue regeneration. Microstructures 2025, 5, 2025055. [Google Scholar] [CrossRef]
- Zheng, S.Y.; Tian, Y.; Zhang, X.N.; Du, M.; Song, Y.; Wu, Z.L.; Zheng, Q. Spin-coating-assisted fabrication of ultrathin physical hydrogel films with high toughness and fast response. Soft Matter 2018, 14, 5888–5897. [Google Scholar] [CrossRef] [PubMed]
- Yola, A.M.; Campbell, J.; Volodkin, D. Microfluidics meets layer-by-layer assembly for the build-up of polymeric scaffolds. Applied Surface Science Advances 2021, 5, 100091. [Google Scholar] [CrossRef]
- Bagheri, A.; Jin, J. Photopolymerization in 3D Printing. ACS Applied Polymer Materials 2019, 1, 593–611. [Google Scholar] [CrossRef]
- Uysal, B.; Madduma-Bandarage, U.S.K.; Jayasinghe, H.G.; Madihally, S. 3D-Printed Hydrogels from Natural Polymers for Biomedical Applications: Conventional Fabrication Methods, Current Developments, Advantages, and Challenges. Gels 2025, 11, 192. [Google Scholar] [CrossRef]
- Liu, J.; Du, C.; Huang, W.; Lei, Y. Injectable smart stimuli-responsive hydrogels: pioneering advancements in biomedical applications. Biomaterials Science 2024, 12, 8–56. [Google Scholar] [CrossRef]
- Li, X.; Li, M.; Tang, L.; Shi, D.; Lam, E.; Bae, J. 3D shape morphing of stimuli-responsive composite hydrogels. Materials Chemistry Frontiers 2023, 7, 5989–6034. [Google Scholar] [CrossRef]
- Neumann, M.; di Marco, G.; Iudin, D.; Viola, M.; van Nostrum, C.F.; van Ravensteijn, B.G.; Vermonden, T. Stimuli-responsive hydrogels: the dynamic smart biomaterials of tomorrow. Macromolecules 2023, 56, 8377–8392. [Google Scholar] [CrossRef]
- Angaria, N.; Saini, S.; Hussain, M.S.; Sharma, S.; Singh, G.; Khurana, N.; Kumar, R. Natural polymer-based hydrogels: versatile biomaterials for biomedical applications. International Journal of Polymeric Materials and Polymeric Biomaterials 2024, 73, 1550–1568. [Google Scholar] [CrossRef]
- Wang, C.; Yokota, T.; Someya, T. Natural Biopolymer-Based Biocompatible Conductors for Stretchable Bioelectronics. Chemical Reviews 2021, 121, 2109–2146. [Google Scholar] [CrossRef]
- Ebhodaghe, S.O. A short review on chitosan and gelatin-based hydrogel composite polymers for wound healing. Journal of Biomaterials Science, Polymer Edition 2022, 33, 1595–1622. [Google Scholar] [CrossRef] [PubMed]
- Varghese, R.; Dalvi, Y.B.; Lochana, P.; Achinthya, S.; Somani, B.O.; Karnaver, P.; Thomas, N.G.; Rupesh, S.; Varghese, N.; VP, J. Physiochemical and Biomedical Properties of Hydrogels: From Fundamentals to Applications. Hydrogels and Nanogels-Applications in Medicine 2024. [Google Scholar]
- Wang, M.; Bai, J.; Shao, K.; Tang, W.; Zhao, X.; Lin, D.; Huang, S.; Chen, C.; Ding, Z.; Ye, J. Poly(vinyl alcohol) Hydrogels: The Old and New Functional Materials. International Journal of Polymer Science 2021, 2021, 2225426. [Google Scholar] [CrossRef]
- Kumar, A.C.; Erothu, H. Synthetic Polymer Hydrogels. In Biomedical Applications of Polymeric Materials and Composites; 2016; pp. 141–162.
- Rana, M.M.; De la Hoz Siegler, H. Evolution of Hybrid Hydrogels: Next-Generation Biomaterials for Drug Delivery and Tissue Engineering. Gels 2024, 10, 216. [Google Scholar] [CrossRef]
- Liu, Z.; Ma, X.; Liu, J.; Zhang, H.; Fu, D. Advances in the application of natural/synthetic hybrid hydrogels in tissue engineering and delivery systems: A comprehensive review. International Journal of Pharmaceutics 2025, 672, 125323–125323. [Google Scholar] [CrossRef]
- Sevinc Ozdemir, N.; Kenar, H. Properties and Preparation Techniques of Hydrogels. In Hydrogels and Bioinks in Tissue Engineering; Springer: 2025; pp. 57–74.
- Yammine, P.; El Safadi, A.; Kassab, R.; El-Nakat, H.; Obeid, P.J.; Nasr, Z.; Tannous, T.; Sari-Chmayssem, N.; Mansour, A.; Chmayssem, A. Types of Crosslinkers and Their Applications in Biomaterials and Biomembranes. Chemistry 2025, 7, 61. [Google Scholar] [CrossRef]
- Ribeiro, M.M.; Simões, M.; Vitorino, C.; Mascarenhas-Melo, F. Physical crosslinking of hydrogels: The potential of dynamic and reversible bonds in burn care. Coordination Chemistry Reviews 2025, 542, 216868. [Google Scholar] [CrossRef]
- Kaur, H.; Gogoi, B.; Sharma, I.; Das, D.K.; Azad, M.A.; Pramanik, D.D.; Pramanik, A. Hydrogels as a Potential Biomaterial for Multimodal Therapeutic Applications. Mol Pharm 2024, 21, 4827–4848. [Google Scholar] [CrossRef]
- Norahan, M.H.; Pedroza-González, S.C.; Sánchez-Salazar, M.G.; Álvarez, M.M.; Trujillo de Santiago, G. Structural and biological engineering of 3D hydrogels for wound healing. Bioactive Materials 2023, 24, 197–235. [Google Scholar] [CrossRef]
- Khattak, S.; Ullah, I.; Yousaf, M.T.; Ullah, S.; Yousaf, H.; Li, Y.; Jin, H.; Shen, J.; Xu, H.-T. Advancements in hydrogels: A comprehensive review of natural, synthetic, and hybrid innovations for wound healing. International Journal of Biological Macromolecules 2025, 327, 147270. [Google Scholar] [CrossRef]
- Olteanu, G.; Neacșu, S.M.; Joița, F.A.; Musuc, A.M.; Lupu, E.C.; Ioniță-Mîndrican, C.B.; Lupuliasa, D.; Mititelu, M. Advancements in Regenerative Hydrogels in Skin Wound Treatment: A Comprehensive Review. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Gounden, V.; Singh, M. Hydrogels and Wound Healing: Current and Future Prospects. Gels 2024, 10. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Luo, S.; Huang, W.; Lu, X.; Kankala, R.K.; Wang, S.; Xu, P.; Chen, A. ECM-inspired stem cell secretome sustained releasing composite nanofibrous membranes for accelerated wound healing. Mater Today Bio 2025, 34, 102141. [Google Scholar] [CrossRef] [PubMed]
- Sawadkar, P.; Lali, F.; Garcia-Gareta, E.; Garrido, B.G.; Chaudhry, A.; Matharu, P.; Kyriakidis, C.; Greco, K. Innovative hydrogels in cutaneous wound healing: current status and future perspectives. Frontiers in Bioengineering and Biotechnology 2025, 13, 1454903. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.Y.; Kang, L.; Li, Z.M.; Tseng, S.L.; Wang, L.Q.; Li, T.H.; Li, Z.J.; Huang, J.Z.; Yu, N.Z.; Long, X. Biological scaffold as potential platforms for stem cells: Current development and applications in wound healing. World J Stem Cells 2024, 16, 334–352. [Google Scholar] [CrossRef]
- Bogadi, S.; Malayandi, R.; Vasanth Raj, P.; Suresh Kumar, A.; Parvathaneni, M.; Kumar Kundu, M.; Rabiul Islam, M.; Khan, F.S.; Tagde, P.; Kumar Mondal, T.; et al. Silk fibroin and sericin: Multifunctional formulations for treating diabetic wound healing. European Polymer Journal 2024, 220, 113465. [Google Scholar] [CrossRef]
- Nifontova, G.; Safaryan, S.; Khristidis, Y.; Smirnova, O.; Vosough, M.; Shpichka, A.; Timashev, P. Advancing wound healing by hydrogel-based dressings loaded with cell-conditioned medium: a systematic review. Stem cell research & therapy 2024, 15, 371. [Google Scholar]
- Qutub, M.; Tatode, A.; Taksande, J.; Premchandani, T.; Umekar, M.; Hussain, U.M.; Biyani, D.; Mane, D. Stimuli-responsive supramolecular hydrogels for paclitaxel delivery: Progress and prospects. Aspects of Molecular Medicine 2025, 5, 100062. [Google Scholar] [CrossRef]
- Davodabadi, F.; Sargazi, S.; Baino, F. Recent advances in hydrogel-based drug delivery systems for enhanced cancer therapy: A review. Materials Today Communications 2025, 48, 113615. [Google Scholar] [CrossRef]
- Yaghoubi, A.; Ramazani, A.; Sillanpaa, M.; Ghasemzadeh, H.; Mohammadi, E. Biocompatible porous PAM/CNT nanocomposite hydrogel films for sustained drug delivery and cancer therapy. Scientific Reports 2025, 15, 22387. [Google Scholar] [CrossRef] [PubMed]
- Sanati, M.; Amin Yavari, S. Liposome-integrated hydrogel hybrids: Promising platforms for cancer therapy and tissue regeneration. Journal of Controlled Release 2024, 368, 703–727. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, B.M. Current advances in stimuli-responsive hydrogels as smart drug delivery carriers. Gels 2023, 9, 838. [Google Scholar] [CrossRef]
- Nasseri, R.; Bouzari, N.; Huang, J.; Golzar, H.; Jankhani, S.; Tang, X.; Mekonnen, T.H.; Aghakhani, A.; Shahsavan, H. Programmable nanocomposites of cellulose nanocrystals and zwitterionic hydrogels for soft robotics. Nature Communications 2023, 14, 6108. [Google Scholar] [CrossRef]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices 2011, 8, 607–626. [Google Scholar] [CrossRef]
- Spicer, C. Hydrogel scaffolds for tissue engineering: The importance of polymer choice. Polymer Chemistry 2019, 11. [Google Scholar] [CrossRef]
- Ayala-Ham, A.; López-Gutierrez, J.; Bermúdez, M.; Aguilar-Medina, M.; Sarmiento-Sánchez, J.I.; López-Camarillo, C.; Sanchez-Schmitz, G.; Ramos-Payan, R. Hydrogel-Based Scaffolds in Oral Tissue Engineering. Frontiers in Materials 2021, Volume 8 - 2021. [CrossRef]
- Zhang, B.; Zhang, M.; Jiang, C.; Yan, W.; Pan, Y.; Meng, F. Engineered polysaccharide scaffolds for cartilage regeneration: Mechanisms, functionalization, and clinical prospects. Colloids and Surfaces B: Biointerfaces 2026, 257, 115134. [Google Scholar] [CrossRef]
- Lu, J.; Gao, Y.; Cao, C.; Wang, H.; Ruan, Y.; Qin, K.; Liu, H.; Wang, Y.; Yang, P.; Liu, Y.; et al. 3D bioprinted scaffolds for osteochondral regeneration: advancements and applications. Materials Today Bio 2025, 32, 101834. [Google Scholar] [CrossRef]
- Bai, L.; Zhou, D.; Li, G.; Liu, J.; Chen, X.; Su, J. Engineering bone/cartilage organoids: strategy, progress, and application. Bone Res 2024, 12, 66. [Google Scholar] [CrossRef]
- Baishya, G.; Parasar, B.; Limboo, M.; Kumar, R.; Dutta, A.; Hussain, A.; Phukan, M.M.; Saikia, D. Advancements in nanocomposite hydrogels: A comprehensive review of biomedical applications. Discover Materials 2024, 4, 40. [Google Scholar] [CrossRef]
- Gong, W.; Kim, J.; Kim, C.; Chang, H.; Ahn, Y.; Schaffer, D.V.; Baek, J. Hydrogel Fabrication Techniques for Advanced Artificial Sensory Systems. International Journal of Extreme Manufacturing 2025. [Google Scholar] [CrossRef]
- Gorantla, A.; Hall, J.T.V.E.; Troidle, A.; Janjic, J.M. Biomaterials for Protein Delivery: Opportunities and Challenges to Clinical Translation. Micromachines 2024, 15, 533. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, X.; Xu, W.; Li, Y.; Lai, R.; Qiu, X.; Chen, X.; Chen, Z.; Mi, B.; Wu, M.; et al. Translational Challenges and Prospective Solutions in the Implementation of Biomimetic Delivery Systems. Pharmaceutics 2023, 15, 2623. [Google Scholar] [CrossRef]
- Xu, F.; Dawson, C.; Lamb, M.; Mueller, E.; Stefanek, E.; Akbari, M.; Hoare, T. Hydrogels for Tissue Engineering: Addressing Key Design Needs Toward Clinical Translation. Frontiers in Bioengineering and Biotechnology 2022, Volume 10 - 2022. [CrossRef]
- Han, I.K.; Chung, T.; Han, J.; Kim, Y.S. Nanocomposite hydrogel actuators hybridized with various dimensional nanomaterials for stimuli responsiveness enhancement. Nano Convergence 2019, 6, 18. [Google Scholar] [CrossRef]
- Banerjee, H.; Suhail, M.; Ren, H. Hydrogel actuators and sensors for biomedical soft robots: brief overview with impending challenges. Biomimetics 2018, 3, 15. [Google Scholar] [CrossRef]
- Lee, H.K.; Yang, Y.J.; Koirala, G.R.; Oh, S.; Kim, T.-i. From lab to wearables: Innovations in multifunctional hydrogel chemistry for next-generation bioelectronic devices. Biomaterials 2024, 310, 122632. [Google Scholar] [CrossRef]
- Zöller, K.; To, D.; Bernkop-Schnürch, A. Biomedical applications of functional hydrogels: Innovative developments, relevant clinical trials and advanced products. Biomaterials 2025, 312, 122718. [Google Scholar] [CrossRef]
- Fan, M.-H.; Pi, J.-K.; Zou, C.-Y.; Jiang, Y.-L.; Li, Q.-J.; Zhang, X.-Z.; Xing, F.; Nie, R.; Han, C.; Xie, H.-Q. Hydrogel-exosome system in tissue engineering: A promising therapeutic strategy. Bioactive Materials 2024, 38, 1–30. [Google Scholar] [CrossRef]
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Wathoni, N.; Yuniarsih, N.; Cahyanto, A.; Muhctaridi, M. α-Mangostin Hydrogel Film Based Chitosan–Alginate for Recurrent Aphthous Stomatitis. Applied Sciences 2019, 9, 5235. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Ng, T.B.; Wong, J.H.; Chan, W.Y. Chitosan: an update on potential biomedical and pharmaceutical applications. Marine drugs 2015, 13, 5156–5186. [Google Scholar] [CrossRef]
- Aderibigbe, B.A.; Buyana, B. Alginate in wound dressings. Pharmaceutics 2018, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Stubbe, B.; Mignon, A.; Declercq, H.; Van Vlierberghe, S.; Dubruel, P. Development of gelatin-alginate hydrogels for burn wound treatment. Macromolecular bioscience 2019, 19, 1900123. [Google Scholar] [CrossRef]
- Salamon, A.; Van Vlierberghe, S.; Van Nieuwenhove, I.; Baudisch, F.; Graulus, G.-J.; Benecke, V.; Alberti, K.; Neumann, H.-G.; Rychly, J.; Martins, J.C. Gelatin-based hydrogels promote chondrogenic differentiation of human adipose tissue-derived mesenchymal stem cells in vitro. Materials 2014, 7, 1342–1359. [Google Scholar] [CrossRef] [PubMed]
- Paula, C.T.; Madeira, A.B.; Pereira, P.; Branco, R.; Morais, P.V.; Coelho, J.F.; Fonseca, A.C.; Serra, A.C. ROS-degradable PEG-based wound dressing films with drug release and antibacterial properties. European Polymer Journal 2022, 177, 111447. [Google Scholar] [CrossRef]
- Chopra, H.; Bibi, S.; Kumar, S.; Khan, M.S.; Kumar, P.; Singh, I. Preparation and evaluation of chitosan/PVA based hydrogel films loaded with honey for wound healing application. Gels 2022, 8, 111. [Google Scholar] [CrossRef]
- Mahardian, A. Biocompatible hydrogel film of polyethylene oxide-polyethylene glycol dimetacrylate for wound dressing application. In Proceedings of the IOP Conference Series: Materials Science and Engineering; 2018; p. 012076. [Google Scholar]
- Jeong, H.I.; An, D.H.; Lim, J.W.; Oh, T.; Lee, H.; Park, S.-M.; Jeong, J.H.; Chung, J.W. Hydrogel surface-modified polyurethane copolymer film with water permeation resistance and biocompatibility for implantable biomedical devices. Micromachines 2021, 12, 447. [Google Scholar] [CrossRef]
- Miroshnichenko, D.; Lebedeva, K.; Cherkashina, A.; Lebedev, V.; Tsereniuk, O.; Krygina, N. Study of hybrid modification with humic acids of environmentally safe biodegradable hydrogel films based on hydroxypropyl methylcellulose. C 2022, 8, 71. [Google Scholar] [CrossRef]
- Mali, K.; Dhawale, S.; Dias, R.; Dhane, N.; Ghorpade, V. Citric Acid Crosslinked Carboxymethyl Cellulose-based Composite Hydrogel Films for Drug Delivery. Indian Journal of Pharmaceutical Sciences 2018, 80. [Google Scholar] [CrossRef]
- Sharma, G.; George Joy, J.; Sharma, A.R.; Kim, J.-C. Accelerated full-thickness skin wound tissue regeneration by self-crosslinked chitosan hydrogel films reinforced by oxidized CNC-AgNPs stabilized Pickering emulsion for quercetin delivery. Journal of Nanobiotechnology 2024, 22, 323. [Google Scholar] [CrossRef]
- Kouser, R.; Vashist, A.; Zafaryab, M.; Rizvi, M.A.; Ahmad, S. Na-montmorillonite-dispersed sustainable polymer nanocomposite hydrogel films for anticancer drug delivery. ACS omega 2018, 3, 15809–15820. [Google Scholar] [CrossRef]
- Motoyama, K.; Higashi, T.; Okajima, M.K.; Kaneko, T.; Arima, H. Potential use of sacran hydrogels as wound dressing material. Yakugaku zasshi: Journal of the Pharmaceutical Society of Japan 2018, 138, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Huang, Y.; Wang, Y.; Xu, H.; Xing, M.; Zhong, W. Mussel-inspired dopamine and carbon nanotube leading to a biocompatible self-rolling conductive hydrogel film. Materials 2017, 10, 964. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Xue, Y.; Hartley, R.; Sant, V.; Eles, J.R.; Cui, X.T.; Stolz, D.B.; Sant, S. Hierarchically aligned fibrous hydrogel films through microfluidic self-assembly of graphene and polysaccharides. Biotechnology and bioengineering 2018, 115, 2654–2667. [Google Scholar] [CrossRef]
- Ghauri, Z.H.; Islam, A.; Qadir, M.A.; Gull, N.; Haider, B.; Khan, R.U.; Riaz, T. Development and evaluation of pH-sensitive biodegradable ternary blended hydrogel films (chitosan/guar gum/PVP) for drug delivery application. Scientific Reports 2021, 11, 21255. [Google Scholar] [CrossRef]
- Wong, R.S.H.; Dodou, K. Effect of drug loading method and drug physicochemical properties on the material and drug release properties of poly (ethylene oxide) hydrogels for transdermal delivery. Polymers 2017, 9, 286. [Google Scholar] [CrossRef]
- Jantrawut, P.; Bunrueangtha, J.; Suerthong, J.; Kantrong, N. Fabrication and characterization of low methoxyl pectin/gelatin/carboxymethyl cellulose absorbent hydrogel film for wound dressing applications. Materials 2019, 12, 1628. [Google Scholar] [CrossRef]
- Aman, J.; Shahi, N.C.; Lohani, U.C.; Balodhi, D.; Singh, R.; Kumar, N.; Bhat, M.I.; Kumar, A.P. [Retracted] Process Optimization for Development of Guar Gum-Based Biodegradable Hydrogel Film Using Response Surface Methodology. Bioinorganic Chemistry and Applications 2022, 2022, 9180000. [Google Scholar] [CrossRef]
- Park, K.M.; Park, K.D. In situ cross-linkable hydrogels as a dynamic matrix for tissue regenerative medicine. Tissue Engineering and Regenerative Medicine 2018, 15, 547–557. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Islam, M.; Hasan, M.K.; Nam, K.-W. A Comprehensive review of radiation-induced hydrogels: Synthesis, properties, and multidimensional applications. Gels 2024, 10, 381. [Google Scholar] [CrossRef]
- Wolfel, A.; Romero, M.R.; Igarzabal, C.I.A. Post-synthesis modification of hydrogels. Total and partial rupture of crosslinks: Formation of aldehyde groups and re-crosslinking of cleaved hydrogels. Polymer 2017, 116, 251–260. [Google Scholar] [CrossRef]
- Xiao, Z.; Li, Q.; Liu, H.; Zhao, Q.; Niu, Y.; Zhao, D. Adhesion mechanism and application progress of hydrogels. European Polymer Journal 2022, 173, 111277. [Google Scholar] [CrossRef]
- Cook, J.P.; Goodall, G.W.; Khutoryanskaya, O.V.; Khutoryanskiy, V.V. Microwave-assisted hydrogel synthesis: a new method for crosslinking polymers in aqueous solutions. Macromolecular rapid communications 2012, 33, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Larraneta, E.; Lutton, R.E.; Brady, A.J.; Vicente-Pérez, E.M.; Woolfson, A.D.; Thakur, R.R.S.; Donnelly, R.F. Microwave-assisted preparation of hydrogel-forming microneedle arrays for transdermal drug delivery applications. Macromolecular materials and engineering 2015, 300, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Thongsuksaengcharoen, S.; Samosorn, S.; Songsrirote, K. A facile synthesis of self-catalytic hydrogel films and their application as a wound dressing material coupled with natural active compounds. Acs Omega 2020, 5, 25973–25983. [Google Scholar] [CrossRef]
- Pargaonkar, S.S.; Ghorpade, V.S.; Mali, K.K.; Dias, R.J.; Havaldar, V.D.; Kadam, V.J.; Pargaonkar, M.S.S. Hydrogel films of citric acid cross-linked hydroxypropyl methylcellulose/methylcellulose for hydrophilic drug delivery. Indian J. Pharm. Educ. Res 2023, 57, 718–727. [Google Scholar] [CrossRef]
- Brinker, C.; Hurd, A.; Schunk, P.; Frye, G.; Ashley, C. Review of sol-gel thin film formation. Journal of Non-Crystalline Solids 1992, 147, 424–436. [Google Scholar] [CrossRef]
- Wang, L.; Xue, Y.; Li, S.; Zhang, X.; Miao, Z.; Zeng, Z.; Ruan, D.; Shen, Y.; Yuan, H.; Zhao, Y.; et al. Tough and Functional Hydrogel Coating by Electrostatic Spraying. Small 2025, 21, 2408780. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, H.J.; Xi, S.; Zhang, Y.; Rao, P.; You, X.; Qu, S. Lignin-Based Ultrathin Hydrogel Coatings with Strong Substrate Adhesion Enabled by Hydrophobic Association. Advanced Functional Materials 2025, 35, 2413464. [Google Scholar] [CrossRef]
- Yan, Y.; Cui, J.; Qiu, X.; Liu, H.; Liu, X.; Yao, P.; Huang, J.; Cui, X.; Liang, X.; Huang, C. Towards Large-Scale Fabrication of Self-Healable Functional Hydrogel Coatings for Anti-Fog/Frost Surfaces and Flexible Sensors. Advanced Materials Technologies 2021, 6, 2001267. [Google Scholar] [CrossRef]
- Pemble, O.J.; Bardosova, M.; Povey, I.M.; Pemble, M.E. A Slot-Die Technique for the Preparation of Continuous, High-Area, Chitosan-Based Thin Films. Polymers 2021, 13, 1566. [Google Scholar] [CrossRef]
- Li, C.Y.; Hao, X.P.; Wu, Z.L.; Zheng, Q. Photolithographically Patterned Hydrogels with Programmed Deformations. Chemistry – An Asian Journal 2019, 14, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, C. Recent advances in 3D printing hydrogel for topical drug delivery. MedComm – Biomaterials and Applications 2022, 1. [Google Scholar] [CrossRef]
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian Journal of Pharmaceutical Sciences 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Bauer, M.; Duerkop, A.; Baeumner, A.J. Critical review of polymer and hydrogel deposition methods for optical and electrochemical bioanalytical sensors correlated to the sensor’s applicability in real samples. Analytical and Bioanalytical Chemistry 2023, 415, 83–95. [Google Scholar] [CrossRef]
- Li, Y.; Ni, C.; Cao, R.; Jiang, Y.; Xia, L.; Ren, H.; Chen, Y.; Xie, T.; Zhao, Q. Sprayable porous hydrogel coating for efficient and sustainable evaporative cooling. Matter 2024, 7, 4270–4280. [Google Scholar] [CrossRef]
- Butt, M.A. Thin-Film Coating Methods: A Successful Marriage of High-Quality and Cost-Effectiveness—A Brief Exploration. Coatings 2022, 12, 1115. [Google Scholar] [CrossRef]
- Jeong, T.-J.; Yu, X.; Harris, T.A.L. Scaled Production of Functionally Gradient Thin Films Using Slot Die Coating on a Roll-to-Roll System. ACS Applied Materials & Interfaces 2024, 16, 9264–9274. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Ke, L.-Y.; Wei, S.-Y.; Poddar, M.S.; Liu, C.-H. Optofluidic thin-film lithography for photocrosslinking hydrogel-based microarchitectures and the assembling of modular cell-embedded microarchitectures. Sensors and Actuators B: Chemical 2022, 352, 131048. [Google Scholar] [CrossRef]
- Park, S.; Shou, W.; Makatura, L.; Matusik, W.; Fu, K. 3D printing of polymer composites: Materials, processes, and applications. Matter 2022, 5, 43–76. [Google Scholar] [CrossRef]
- Priya, A.S.; Premanand, R.; Ragupathi, I.; Rao Bhaviripudi, V.; Aepuru, R.; Kannan, K.; Shanmugaraj, K. Comprehensive Review of Hydrogel Synthesis, Characterization, and Emerging Applications. 2024. [CrossRef]
- Dong, M.; Jiao, D.; Zheng, Q.; Wu, Z.L. Recent progress in fabrications and applications of functional hydrogel films. Journal of Polymer Science 2023, 61, 1026–1039. [Google Scholar] [CrossRef]
- Akhtar, M.F.; Hanif, M.; Ranjha, N.M. Methods of synthesis of hydrogels … A review. Saudi Pharmaceutical Journal 2016, 24, 554–559. [Google Scholar] [CrossRef]
- Gong, J.P. Why are double network hydrogels so tough? Soft Matter 2010, 6, 2583–2590. [Google Scholar] [CrossRef]
- Nguyen, V.N.; Tran, T.V.; Trai, V.K. Facile fabrication route of stretchable thin-film hydrogels with high strength. MRS Advances 2024, 9, 1672–1677. [Google Scholar] [CrossRef]
- Maulvi, F.A.; Soni, T.G.; Shah, D.O. A review on therapeutic contact lenses for ocular drug delivery. Drug delivery 2016, 23, 3017–3026. [Google Scholar] [CrossRef]
- Ngo, H.V.; Tran, P.H.L.; Lee, B.-J.; Tran, T.T.D. Development of film-forming gel containing nanoparticles for transdermal drug delivery. Nanotechnology 2019, 30, 415102. [Google Scholar] [CrossRef]
- Sun, W.; Wu, W.; Dong, X.; Yu, G. Frontier and hot topics in the application of hydrogel in the biomedical field: a bibliometric analysis based on CiteSpace. Journal of Biological Engineering 2024, 18, 40. [Google Scholar] [CrossRef]
- Asadikorayem, M.; Weber, P.; Surman, F.; Puiggalí-Jou, A.; Zenobi-Wong, M. Foreign body immune response to zwitterionic and hyaluronic acid granular hydrogels made with mechanical fragmentation. Advanced Healthcare Materials 2025, 14, 2402890. [Google Scholar] [CrossRef]
- Costa, A.L.R.; Willerth, S.M.; de la Torre, L.G.; Han, S.W. Trends in hydrogel-based encapsulation technologies for advanced cell therapies applied to limb ischemia. Materials Today Bio 2022, 13, 100221. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Huang, J. Recent Progress in Hydrogel Synthesis and Biomedical Applications. Gels 2025, 11. [Google Scholar] [CrossRef] [PubMed]
- Britton, D.; Almanzar, D.; Xiao, Y.; Shih, H.-W.; Legocki, J.; Rabbani, P.; Montclare, J.K. Exosome Loaded Protein Hydrogel for Enhanced Gelation Kinetics and Wound Healing. ACS Applied Bio Materials 2024, 7, 5992–6000. [Google Scholar] [CrossRef] [PubMed]
- Lavrentev, F.V.; Shilovskikh, V.V.; Alabusheva, V.S.; Yurova, V.Y.; Nikitina, A.A.; Ulasevich, S.A.; Skorb, E.V. Diffusion-Limited Processes in Hydrogels with Chosen Applications from Drug Delivery to Electronic Components. Molecules 2023, 28, 5931. [Google Scholar] [CrossRef]
- Kanduč, M.; Kim, W.K.; Roa, R.; Dzubiella, J. How the shape and chemistry of molecular penetrants control responsive hydrogel permeability. ACS nano 2020, 15, 614–624. [Google Scholar] [CrossRef]
- Narayana, S.; Gowda, B.J.; Hani, U.; Ahmed, M.G.; Asiri, Z.A.; Paul, K. Smart poly (N-isopropylacrylamide)-based hydrogels: a tour D’horizon of biomedical applications. Gels 2025, 11, 207. [Google Scholar] [CrossRef]
- Vegad, U.; Patel, M.; Khunt, D.; Zupančič, O.; Chauhan, S.; Paudel, A. pH stimuli-responsive hydrogels from non-cellulosic biopolymers for drug delivery. Frontiers in Bioengineering and Biotechnology 2023, 11, 1270364. [Google Scholar] [CrossRef]
- El-Sherbiny, I.M.; Yacoub, M.H. Hydrogel scaffolds for tissue engineering: Progress and challenges. Global Cardiology Science and Practice 2013, 2013, 38. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Y.; Hu, Z.; Lu, W.; Li, Z.; Gao, N.; Liu, N.; Li, Y.; He, J.; Gao, Q. Gelatin-based metamaterial hydrogel films with high conformality for ultra-soft tissue monitoring. Nano-Micro Letters 2024, 16, 34. [Google Scholar] [CrossRef]
- Bovone, G.; Dudaryeva, O.Y.; Marco-Dufort, B.; Tibbitt, M.W. Engineering hydrogel adhesion for biomedical applications via chemical design of the junction. ACS Biomaterials Science & Engineering 2021, 7, 4048–4076. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Sun, K.; Seong, M.; Hwang, I.; Jang, H.; Park, S.; Choi, G.; Lee, S.-H.; Kim, J.; Jeong, H.E. Applications of bioinspired reversible dry and wet adhesives: A review. Frontiers in Mechanical Engineering 2021, 7, 668262. [Google Scholar] [CrossRef]
- Yuk, H.; Varela, C.E.; Nabzdyk, C.S.; Mao, X.; Padera, R.F.; Roche, E.T.; Zhao, X. Dry double-sided tape for adhesion of wet tissues and devices. Nature 2019, 575, 169–174. [Google Scholar] [CrossRef]
- Nie, L.; Muñoz-Camargo, C.; Ganguly, S.; Bahsis, L.; Cruz, J.C.; Mohammadinejad, R.; Nicosia, A.; Reyes, L.H.; Wang, X. Editorial: Biocompatible hydrogels: properties, synthesis and applications in biomedicine. Frontiers in Chemistry 2024, Volume 12 - 2024. [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. Journal of Advanced Research 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Ren, P.; Yang, L.; Wei, D.; Liang, M.; Xu, L.; Zhang, T.; Hu, W.; Zhang, Z.; Zhang, Q. Alginate/polyacrylamide host-guest supramolecular hydrogels with enhanced adhesion. International Journal of Biological Macromolecules 2023, 242, 124885. [Google Scholar] [CrossRef]
- Ollier, R.C.; Webber, M.J. Mechanoresponsive Hydrogels Emerging from Dynamic and Non-Covalent Interactions. Advanced Materials 2025. [Google Scholar] [CrossRef]
- Feliciano, A.J.; van Blitterswijk, C.; Moroni, L.; Baker, M.B. Realizing tissue integration with supramolecular hydrogels. Acta Biomaterialia 2021, 124, 1–14. [Google Scholar] [CrossRef]
- Scherer, W.F.; Syverton, J.T.; Gey, G.O. Studies on the propagation in vitro of poliomyelitis viruses : iv. Viral multiplication in a stable strain of human malignant epithelial cells (strain hela) derived from an epidermoid carcinoma of the cervix. Journal of Experimental Medicine 1953, 97, 695–710. [Google Scholar] [CrossRef]
- Yi, B.; Wang, X.; Yu, J.; Diao, J.; Wang, G.; Li, S.; Bo, J.; Zhang, X.; Zhang, C.; Guimarães, C.F.; et al. Biomimetic hydrogel micro-/nanofibers for in situ soft tissue repair and regeneration. Bioactive Materials 2026, 55, 485–502. [Google Scholar] [CrossRef]
- Wang, Y.; Kankala, R.K.; Ou, C.; Chen, A.; Yang, Z. Advances in hydrogel-based vascularized tissues for tissue repair and drug screening. Bioactive Materials 2022, 9, 198–220. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Y.; Hu, Z.; Lu, W.; Li, Z.; Gao, N.; Liu, N.; Li, Y.; He, J.; Gao, Q.; et al. Gelatin-Based Metamaterial Hydrogel Films with High Conformality for Ultra-Soft Tissue Monitoring. Nano-Micro Letters 2023, 16, 34. [Google Scholar] [CrossRef]
- Jia, B.; Li, G.; Cao, E.; Luo, J.; Zhao, X.; Huang, H. Recent progress of antibacterial hydrogels in wound dressings. Materials Today Bio 2023, 19, 100582. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Lin, C.; Hu, H.; Zhao, Y.; Liao, J.; Al-Smadi, F.; Mi, B.; Hu, Y.; Liu, G. Recent advances and challenges in hydrogel-based delivery of immunomodulatory strategies for diabetic wound healing. Theranostics 2026, 16, 516–544. [Google Scholar] [CrossRef]
- Karthikeyan, L.; Kang, H.W. Recent progress in multifunctional theranostic hydrogels: the cornerstone of next-generation wound care technologies. Biomater. Sci. 2025, 13, 4358–4389. [Google Scholar] [CrossRef]
- Han, Y.; Cao, J.; Li, M.; Ding, P.; Yang, Y.; Okoro, O.V.; Sun, Y.; Jiang, G.; Shavandi, A.; Nie, L. Fabrication and characteristics of multifunctional hydrogel dressings using dopamine modified hyaluronic acid and phenylboronic acid modified chitosan. Frontiers in Chemistry 2024, Volume 12 - 2024. [CrossRef]
- Delgado-Pujol, E.J.; Martínez, G.; Casado-Jurado, D.; Vázquez, J.; León-Barberena, J.; Rodríguez-Lucena, D.; Torres, Y.; Alcudia, A.; Begines, B. Hydrogels and Nanogels: Pioneering the Future of Advanced Drug Delivery Systems. Pharmaceutics 2025, 17. [Google Scholar] [CrossRef]
- Naranđa, J.; Bračič, M.; Maver, U.; Trojner, T. Recent Advancements in Smart Hydrogel-Based Materials in Cartilage Tissue Engineering. Materials 2025, 18, 2576. [Google Scholar] [CrossRef]
- Sánchez-Cid, P.; Jiménez-Rosado, M.; Romero, A.; Pérez-Puyana, V. Novel Trends in Hydrogel Development for Biomedical Applications: A Review. Polymers 2022, 14, 3023. [Google Scholar] [CrossRef]
- Ho, T.-C.; Chang, C.-C.; Chan, H.-P.; Chung, T.-W.; Shu, C.-W.; Chuang, K.-P.; Duh, T.-H.; Yang, M.-H.; Tyan, Y.-C. Hydrogels: Properties and Applications in Biomedicine. Molecules 2022, 27, 2902. [Google Scholar] [CrossRef]
- Tang, S.; Zhou, C.; and Zou, H. Current Status and Prospects of Biomaterials (Review). Journal of Jinan University (Natural Science Edition) 2000, 21, 122–125. [Google Scholar]
- Chiellini, E.; Solaro, R. Multifunctional bioerodible/biodegradable polymeric materials. Macromolecular Symposia 1995, 98, 803–824. [Google Scholar] [CrossRef]
- Protsak, I.S.; Morozov, Y.M. Fundamentals and Advances in Stimuli-Responsive Hydrogels and Their Applications: A Review. Gels 2025, 11, 30. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, A.; Muñana-González, S.; Lanceros-Mendez, S.; Ruiz-Rubio, L.; Alvarez, L.P.; Vilas-Vilela, J.L. Biodegradable Natural Hydrogels for Tissue Engineering, Controlled Release, and Soil Remediation. Polymers 2024, 16, 2599. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M. Enzyme-Responsive Hydrogels as Potential Drug Delivery Systems—State of Knowledge and Future Prospects. International Journal of Molecular Sciences 2022, 23, 4421. [Google Scholar] [CrossRef]
- Ding, K.; Liao, M.; Wang, Y.; Lu, J.R. Advances in Composite Stimuli-Responsive Hydrogels for Wound Healing: Mechanisms and Applications. Gels 2025, 11, 420. [Google Scholar] [CrossRef]
- Zhang, X.; Zu, Q.; Deng, C.; Gao, X.; Liu, H.; Jin, Y.; Yang, X.; Wang, E. Biodegradable Double-Layer Hydrogels with Sequential Drug Release for Multi-Phase Collaborative Regulation in Scar-Free Wound Healing. Journal of Functional Biomaterials 2025, 16, 164. [Google Scholar] [CrossRef]
- Hu, B.; Gao, J.; Lu, Y.; Wang, Y. Applications of Degradable Hydrogels in Novel Approaches to Disease Treatment and New Modes of Drug Delivery. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Santhamoorthy, M.; Kim, S.-C. A Review of the Development of Biopolymer Hydrogel-Based Scaffold Materials for Drug Delivery and Tissue Engineering Applications. Gels 2025, 11, 178. [Google Scholar] [CrossRef] [PubMed]
- Aljeboree, A.M.; Hasan, I.T.; Jwaid, M.M.; Dawood, A.H.; Jawad, M.A. Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite. Engineering Proceedings 2023, 59, 219. [Google Scholar]
- Mukhopadhyay, P.; Eid, N.; Abdelmegeed, M.A.; Sen, A. Interplay of Oxidative Stress, Inflammation, and Autophagy: Their Role in Tissue Injury of the Heart, Liver, and Kidney. Oxidative Medicine and Cellular Longevity 2018, 2018, 2090813. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, S.M.; Bakaic, E.; Stewart, S.A.; Hoare, T.; Adronov, A. Properties of Poly(ethylene glycol) Hydrogels Cross-Linked via Strain-Promoted Alkyne–Azide Cycloaddition (SPAAC). Biomacromolecules 2016, 17, 1093–1100. [Google Scholar] [CrossRef]
- Liew, K.-F.; Hanapi, N.A.; Chan, K.-L.; Yusof, S.R.; Lee, C.-Y. Assessment of the Blood-Brain Barrier Permeability of Potential Neuroprotective Aurones in Parallel Artificial Membrane Permeability Assay and Porcine Brain Endothelial Cell Models. Journal of Pharmaceutical Sciences 2017, 106, 502–510. [Google Scholar] [CrossRef]
- Stidl, R.; Denne, M.; Goldstine, J.; Kadish, B.; Korakas, K.I.; Turecek, P.L. Polyethylene Glycol Exposure with Antihemophilic Factor (Recombinant), PEGylated (rurioctocog alfa pegol) and Other Therapies Indicated for the Pediatric Population: History and Safety. Pharmaceuticals 2018, 11, 75. [Google Scholar] [CrossRef]
- Fu, S.; Zhu, X.; Huang, F.; Chen, X. Anti-PEG Antibodies and Their Biological Impact on PEGylated Drugs: Challenges and Strategies for Optimization. Pharmaceutics 2025, 17, 1074. [Google Scholar] [CrossRef]
- Muktar, M.Z.; Bakar, M.A.A.; Amin, K.A.M.; Che Rose, L.; Wan Ismail, W.I.; Razali, M.H.; Abd Razak, S.I.; in het Panhuis, M. Gellan gum hydrogels filled edible oil microemulsion for biomedical materials: Phase diagram, mechanical behavior, and in vivo studies. Polymers 2021, 13, 3281. [Google Scholar] [CrossRef]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin wound healing process and new emerging technologies for skin wound care and regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Jang, K.-I.; Chung, H.U.; Xu, S.; Lee, C.H.; Luan, H.; Jeong, J.; Cheng, H.; Kim, G.-T.; Han, S.Y.; Lee, J.W. Soft network composite materials with deterministic and bio-inspired designs. Nature communications 2015, 6, 6566. [Google Scholar] [CrossRef]
- Alberts, A.; Moldoveanu, E.-T.; Niculescu, A.-G.; Grumezescu, A.M. Hydrogels for Wound Dressings: Applications in Burn Treatment and Chronic Wound Care. Journal of Composites Science 2025, 9, 133. [Google Scholar] [CrossRef]
- Ribeiro, M.; Simões, M.; Vitorino, C.; Mascarenhas-Melo, F. Hydrogels in Cutaneous Wound Healing: Insights into Characterization, Properties, Formulation and Therapeutic Potential. Gels 2024, 10, 188. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Wei, Y.; Xu, K. Hydrogel-Based Treatment of Diabetic Wounds: From Smart Responsive to Smart Monitoring. Gels 2025, 11, 647. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Y.P.; Kirsner, R.S. Angiogenesis in wound repair: angiogenic growth factors and the extracellular matrix. Microscopy research and technique 2003, 60, 107–114. [Google Scholar] [CrossRef]
- Huang, C.; Dong, L.; Zhao, B.; Lu, Y.; Huang, S.; Yuan, Z.; Luo, G.; Xu, Y.; Qian, W. Anti-inflammatory hydrogel dressings and skin wound healing. Clinical and Translational Medicine 2022, 12, e1094. [Google Scholar] [CrossRef]
- Hasan, N.; Jiafu, C.; Mustopa, A.Z.; Himawan, A.; Umami, R.N.; Ullah, M.; Wathoni, N.; Yoo, J.-W. Recent advancements of nitric oxide-releasing hydrogels for wound dressing applications. Journal of Pharmaceutical Investigation 2023, 53, 781–801. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Zabermawi, N.M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; Abd El-Hakim, Y.M.; Al-Sagheer, A.A. Antimicrobial and antioxidant properties of chitosan and its derivatives and their applications: A review. International journal of biological macromolecules 2020, 164, 2726–2744. [Google Scholar] [CrossRef]
- Li, M.; Dong, Y.; Wang, M.; Lu, X.; Li, X.; Yu, J.; Ding, B. Hydrogel/nanofibrous membrane composites with enhanced water retention, stretchability and self-healing capability for wound healing. Composites Part B: Engineering 2023, 257, 110672. [Google Scholar] [CrossRef]
- Cheng, H.; Newton, M.A.A.; Rajib, M.; Zhang, Q.; Gao, W.; Lu, Z.; Zheng, Y.; Dai, Z.; Zhu, J. A ZIF-8-encapsulated interpenetrated hydrogel/nanofiber composite patch for chronic wound treatment. Journal of Materials Chemistry B 2024, 12, 2042–2053. [Google Scholar] [CrossRef]
- Ruan, L.; Pan, C.; Ran, X.; Wen, Y.; Lang, R.; Peng, M.; Cao, J.; Yang, J. Dual-Delivery Temperature-Sensitive Hydrogel with Antimicrobial and Anti-Inflammatory Brevilin A and Nitric Oxide for Wound Healing in Bacterial Infection. Gels 2024, 10, 219. [Google Scholar] [CrossRef]
- Ullah, I.; Hussain, Z.; Ullah, S.; Zahra, Q.u.a.; Zhang, Y.; Mehmood, S.; Liu, X.; Kamya, E.; Waseem Ghani, M.; Mansoorianfar, M.; et al. An osteogenic, antibacterial, and anti-inflammatory nanocomposite hydrogel platform to accelerate bone reconstruction. Journal of Materials Chemistry B 2023, 11, 5830–5845. [Google Scholar] [CrossRef]
- Pratinthong, K.; Punyodom, W.; Jantrawut, P.; Jantanasakulwong, K.; Tongdeesoontorn, W.; Sriyai, M.; Panyathip, R.; Thanakkasaranee, S.; Worajittiphon, P.; Tanadchangsaeng, N.; et al. Modification of a Carboxymethyl Cellulose/Poly(vinyl alcohol) Hydrogel Film with Citric Acid and Glutaraldehyde Crosslink Agents to Enhance the Anti-Inflammatory Effectiveness of Triamcinolone Acetonide in Wound Healing. Polymers 2024, 16, 1798. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Hu, L.; Chen, X.; Zuo, L.; Bai, X.; Du, W.; Xu, N. Antibacterial and Anti-Inflammatory Polysaccharide from Fructus Ligustri Lucidi Incorporated in PVA/Pectin Hydrogels Accelerate Wound Healing. Molecules 2024, 29, 1423. [Google Scholar] [CrossRef] [PubMed]
- Chuysinuan, P.; Pengsuk, C.; Lirdprapamongkol, K.; Thanyacharoen, T.; Techasakul, S.; Svasti, J.; Nooeaid, P. Turmeric Herb Extract-Incorporated Biopolymer Dressings with Beneficial Antibacterial, Antioxidant and Anti-Inflammatory Properties for Wound Healing. Polymers 2023, 15, 1090. [Google Scholar] [CrossRef] [PubMed]
- Ahmady, A.R.; Razmjooee, K.; Saber-Samandari, S.; Toghraie, D. Fabrication of chitosan-gelatin films incorporated with thymol-loaded alginate microparticles for controlled drug delivery, antibacterial activity and wound healing: in-vitro and in-vivo studies. International journal of biological macromolecules 2022, 223, 567–582. [Google Scholar] [CrossRef]
- Tang, Y.; Zhao, R.; Yi, M.; Ge, Z.; Wang, D.; Wang, G.; Deng, X. Multifunctional Hydrogel Enhances Inflammatory Control, Antimicrobial Activity, and Oxygenation to Promote Healing in Infectious Wounds. Biomacromolecules 2024, 25, 2423–2437. [Google Scholar] [CrossRef]
- Gao, Y.; Nguyen, D.T.; Yeo, T.; Lim, S.B.; Tan, W.X.; Madden, L.E.; Jin, L.; Long, J.Y.K.; Aloweni, F.A.B.; Liew, Y.J.A. A flexible multiplexed immunosensor for point-of-care in situ wound monitoring. Science Advances 2021, 7, eabg9614. [Google Scholar]
- Cadinoiu, A.N.; Rata, D.M.; Daraba, O.M.; Ichim, D.L.; Popescu, I.; Solcan, C.; Solcan, G. Silver Nanoparticles Biocomposite Films with Antimicrobial Activity: In Vitro and In Vivo Tests. International Journal of Molecular Sciences 2022, 23, 10671. [Google Scholar] [CrossRef]
- Hashempur, M.H.; Sabili, A.; Karami, F.; Zomorodian, K.; Shenavari, S.; Vaez, A.; Sahraeian, K.; Zareshahrabadi, Z. Synthesize, antioxidant and antimicrobial properties of a chitosan xerogel film with Nigella Sativa extract. Scientific Reports 2025, 15, 24635. [Google Scholar] [CrossRef]
- Boateng, J.; Mani, J.; Kianfar, F. Improving drug loading of mucosal solvent cast films using a combination of hydrophilic polymers with amoxicillin and paracetamol as model drugs. BioMed Research International 2013, 2013, 198137. [Google Scholar] [CrossRef]
- Xin, H.; Maruf, D.A.A.; Akin-Ige, F.; Amin, S. Stimuli-responsive hydrogels for skin wound healing and regeneration. Emergent Materials 2025, 8, 1339–1356. [Google Scholar] [CrossRef]
- Wang, S.; Wu, W.Y.; Yeo, J.C.C.; Soo, X.Y.D.; Thitsartarn, W.; Liu, S.; Tan, B.H.; Suwardi, A.; Li, Z.; Zhu, Q. Responsive hydrogel dressings for intelligent wound management. BMEMat 2023, 1, e12021. [Google Scholar] [CrossRef]
- Gamerith, C.; Luschnig, D.; Ortner, A.; Pietrzik, N.; Guse, J.-H.; Burnet, M.; Haalboom, M.; van der Palen, J.; Heinzle, A.; Sigl, E. pH-responsive materials for optical monitoring of wound status. Sensors and Actuators B: Chemical 2019, 301, 126966. [Google Scholar] [CrossRef]
- Eskilson, O.; Zattarin, E.; Berglund, L.; Oksman, K.; Hanna, K.; Rakar, J.; Sivlér, P.; Skog, M.; Rinklake, I.; Shamasha, R. Nanocellulose composite wound dressings for real-time pH wound monitoring. Materials Today Bio 2023, 19, 100574. [Google Scholar]
- Rahimi, R.; Brener, U.; Chittiboyina, S.; Soleimani, T.; Detwiler, D.A.; Lelièvre, S.A.; Ziaie, B. Laser-enabled fabrication of flexible and transparent pH sensor with near-field communication for in-situ monitoring of wound infection. Sensors and Actuators B: Chemical 2018, 267, 198–207. [Google Scholar] [CrossRef]
- Han, Z.; Yuan, M.; Liu, L.; Zhang, K.; Zhao, B.; He, B.; Liang, Y.; Li, F. pH-Responsive wound dressings: advances and prospects. Nanoscale Horizons 2023, 8, 422–440. [Google Scholar] [CrossRef]
- Mariani, F.; Serafini, M.; Gualandi, I.; Arcangeli, D.; Decataldo, F.; Possanzini, L.; Tessarolo, M.; Tonelli, D.; Fraboni, B.; Scavetta, E. Advanced Wound Dressing for Real-Time pH Monitoring. ACS Sensors 2021, 6, 2366–2377. [Google Scholar] [CrossRef]
- Kaewpradub, K.; Veenuttranon, K.; Jantapaso, H.; Mittraparp-arthorn, P.; Jeerapan, I. A Fully-Printed Wearable Bandage-Based Electrochemical Sensor with pH Correction for Wound Infection Monitoring. Nano-Micro Letters 2024, 17, 71. [Google Scholar] [CrossRef]
- Du, M.; Jin, J.; Zhou, F.; Chen, J.; Jiang, W. Dual drug-loaded hydrogels with pH-responsive and antibacterial activity for skin wound dressing. Colloids and Surfaces B: Biointerfaces 2023, 222, 113063. [Google Scholar] [CrossRef]
- Zhao, Y.; Huang, L.; Lin, G.; Tong, M.; Xie, Y.; Pan, H.; Shangguan, J.; Yao, Q.; Xu, S.; Xu, H. Skin-adaptive film dressing with smart-release of growth factors accelerated diabetic wound healing. International journal of biological macromolecules 2022, 222, 2729–2743. [Google Scholar] [CrossRef]
- Moeinipour, A.; Afkhami, A.; Madrakian, T. Stimuli-responsive polymeric film based on hydrogen-bonded organic framework designing as a smart wound dressing. Iranian Polymer Journal 2025, 34, 1387–1397. [Google Scholar] [CrossRef]
- Li, X.; Xue, X.; Xie, P. Smart dressings and their applications in chronic wound management. Cell Biochemistry and Biophysics 2024, 82, 1965–1977. [Google Scholar] [CrossRef] [PubMed]
- Brunsen, A.; Ritz, U.; Mateescu, A.; Hoefer, I.; Frank, P.; Menges, B.; Hofmann, A.; Rommens, P.; Knoll, W.; Jonas, U. Photocrosslinkable dextran hydrogel films as substrates for osteoblast and endothelial cell growth. Journal of Materials Chemistry 2012, 22, 19590–19604. [Google Scholar] [CrossRef]
- Zhu, F.; Chen, Y.; Yang, S.; Wang, Q.; Liang, F.; Qu, X.; Hu, Z. Surface patterned hydrogel film as a flexible scaffold for 2D and 3D cell co-culture. RSC Advances 2016, 6, 61185–61189. [Google Scholar] [CrossRef]
- Moreau, D.; Chauvet, C.; Etienne, F.; Rannou, F.P.; Corté, L. Hydrogel films and coatings by swelling-induced gelation. Proceedings of the National Academy of Sciences 2016, 113, 13295–13300. [Google Scholar] [CrossRef]
- Garland, S.P.; McKee, C.T.; Chang, Y.-R.; Raghunathan, V.K.; Russell, P.; Murphy, C.J. A cell culture substrate with biologically relevant size-scale topography and compliance of the basement membrane. Langmuir 2014, 30, 2101–2108. [Google Scholar] [CrossRef]
- Hameed, H.; Faheem, S.; Paiva-Santos, A.C.; Sarwar, H.S.; Jamshaid, M. A Comprehensive Review of Hydrogel-Based Drug Delivery Systems: Classification, Properties, Recent Trends, and Applications. AAPS PharmSciTech 2024, 25, 64. [Google Scholar] [CrossRef]
- Kim, S.; Kim, D.-D.; Karmakar, M.; Cho, H.-J. Injectable hydrogel systems for local cancer therapy. Journal of Pharmaceutical Investigation 2024, 54, 555–591. [Google Scholar] [CrossRef]
- Hu, X.; Zhang, C.; Xiong, Y.; Ma, S.; Sun, C.; Xu, W. A review of recent advances in drug loading, mathematical modeling and applications of hydrogel drug delivery systems. Journal of Materials Science 2024, 59, 15077–15116. [Google Scholar] [CrossRef]
- Abou-Okeil, A.; Taha, G.M. Investigation and kinetics of hydrogel scaffold with sustained release ciprofloxacin hydrochloride. Polymer Bulletin 2024, 81, 17393–17411. [Google Scholar] [CrossRef]
- Kang, J.; Li, Y.; Qin, Y.; Huang, Z.; Wu, Y.; Sun, L.; Wang, C.; Wang, W.; Feng, G.; Qi, Y. In Situ Deposition of Drug and Gene Nanoparticles on a Patterned Supramolecular Hydrogel to Construct a Directionally Osteochondral Plug. Nano-Micro Letters 2023, 16, 18. [Google Scholar] [CrossRef]
- Özakar, E.; Sevinç-Özakar, R.; Yılmaz, B. Preparation, characterization, and evaluation of cytotoxicity of fast dissolving hydrogel based oral thin films containing pregabalin and methylcobalamin. Gels 2023, 9, 147. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, P.; Ahmed, K.A.; Shirsand, S.B.; Raikar, P.K.; Hiraskar, A. Transdermal Patches: Design, Evaluation, and Potential Applications in Modern Therapeutics. Biomedical Materials & Devices 2025, 1–19. [Google Scholar] [CrossRef]
- Abedini, A.A.; Pircheraghi, G.; Kaviani, A.; Hosseini, S. Exploration of curcumin-incorporated dual anionic alginate-quince seed gum films for transdermal drug delivery. International Journal of Biological Macromolecules 2023, 248, 125798. [Google Scholar] [CrossRef]
- Hosseini, M.S.; Kamali, B.; Nabid, M.R. Multilayered mucoadhesive hydrogel films based on Ocimum basilicum seed mucilage/thiolated alginate/dopamine-modified hyaluronic acid and PDA coating for sublingual administration of nystatin. International Journal of Biological Macromolecules 2022, 203, 93–104. [Google Scholar] [CrossRef]
- Yoon, M.S.; Lee, J.M.; Jo, M.J.; Kang, S.J.; Yoo, M.K.; Park, S.Y.; Bong, S.; Park, C.-S.; Park, C.-W.; Kim, J.-S. Dual-Drug Delivery Systems Using Hydrogel–Nanoparticle Composites: Recent Advances and Key Applications. Gels 2025, 11, 520. [Google Scholar] [CrossRef]
- Manghnani, P.N.; Nelson, A.Z.; Wong, K.; Lee, Y.W.; Khan, S.A.; Doyle, P.S. From burst to controlled release: Using hydrogel crosslinking chemistry to tune release of micro-crystalline active pharmaceutical ingredients. RSC Pharmaceutics 2025, 2, 94–101. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, J.; Luo, J.; Cui, Y.; Chen, J.; Zeng, B.; Deng, Z.; Shao, L. “Double-sided protector” Janus hydrogels for skin and mucosal wound repair: applications, mechanisms, and prospects. Journal of Nanobiotechnology 2025, 23, 387. [Google Scholar] [CrossRef]
- Taboada, G.M.; Yang, K.; Pereira, M.J.; Liu, S.S.; Hu, Y.; Karp, J.M.; Artzi, N.; Lee, Y. Overcoming the translational barriers of tissue adhesives. Nature Reviews Materials 2020, 5, 310–329. [Google Scholar] [CrossRef]
- Shirzaei Sani, E.; Kheirkhah, A.; Rana, D.; Sun, Z.; Foulsham, W.; Sheikhi, A.; Khademhosseini, A.; Dana, R.; Annabi, N. Sutureless repair of corneal injuries using naturally derived bioadhesive hydrogels. Science advances 2019, 5, eaav1281. [Google Scholar] [CrossRef]
- Lin, X.; Liu, Y.; Bai, A.; Cai, H.; Bai, Y.; Jiang, W.; Yang, H.; Wang, X.; Yang, L.; Sun, N. A viscoelastic adhesive epicardial patch for treating myocardial infarction. Nature biomedical engineering 2019, 3, 632–643. [Google Scholar] [CrossRef]
- Stapleton, L.M.; Steele, A.N.; Wang, H.; Lopez Hernandez, H.; Yu, A.C.; Paulsen, M.J.; Smith, A.A.; Roth, G.A.; Thakore, A.D.; Lucian, H.J. Use of a supramolecular polymeric hydrogel as an effective post-operative pericardial adhesion barrier. Nature biomedical engineering 2019, 3, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Wu, T.; Chen, X.; Liu, Y.; Li, Y.; Xu, Z.; Fan, C.; Liu, W. A janus hydrogel wet adhesive for internal tissue repair and anti-postoperative adhesion. Advanced Functional Materials 2020, 30, 2005689. [Google Scholar] [CrossRef]
- Fang, Y.; Zheng, Y.; Chi, C.; Jiang, S.; Qin, W.; Zhang, Y.; Liu, H.; Chen, Q. PAA-PU Janus Hydrogels Stabilized by Janus Particles and its Interfacial Performance During Hemostatic Processing. Advanced Healthcare Materials 2024, 13, 2303802. [Google Scholar] [CrossRef] [PubMed]
- Tamo, A.K. Nanocellulose-based hydrogels as versatile materials with interesting functional properties for tissue engineering applications. Journal of Materials Chemistry B 2024, 12, 7692–7759. [Google Scholar] [CrossRef]
- Prakashini, R.S.; Thangam, T.; Hemamalani, A.U.; Parthasarathy, K. Role of Natural Polymers in Hydrogel-Based Scaffolds for Tissue Engineering Applications. Regenerative Engineering and Translational Medicine 2025. [Google Scholar] [CrossRef]
- Ragab, A.; El-Badry, N.; Tamer, N.; Naas, A.; Hamdy, A.; Tawakey, S.H.; Hassan, A.H.; Salim, A.I. Biodegradable chitosan/PVA-based hydrogel incorporating green synthesized silver nanoparticles for wound healing applications. BMC chemistry 2025, 19, 1–16. [Google Scholar] [CrossRef]
- Guillén-Carvajal, K.; Valdez-Salas, B.; Beltrán-Partida, E.; Salomón-Carlos, J.; Cheng, N. Chitosan, Gelatin, and Collagen Hydrogels for Bone Regeneration. Polymers 2023, 15, 2762. [Google Scholar] [CrossRef]
- Li, J.; Chen, G.; Xu, X.; Abdou, P.; Jiang, Q.; Shi, D.; Gu, Z. Advances of injectable hydrogel-based scaffolds for cartilage regeneration. Regenerative biomaterials 2019, 6, 129–140. [Google Scholar] [CrossRef]
- Li, C.-S.; Xu, Y.; Li, J.; Qin, S.-H.; Huang, S.-W.; Chen, X.-M.; Luo, Y.; Gao, C.-T.; Xiao, J.-H. Ultramodern natural and synthetic polymer hydrogel scaffolds for articular cartilage repair and regeneration. BioMedical Engineering OnLine 2025, 24, 13. [Google Scholar] [CrossRef]
- Amiryaghoubi, N.; Fathi, M.; Barar, J.; Omidi, Y. Hydrogel-based scaffolds for bone and cartilage tissue engineering and regeneration. Reactive and Functional Polymers 2022, 177, 105313. [Google Scholar] [CrossRef]
- Hajirasouliha, E.; Zandi, M.; Hashemi Tabatabaei, M.; Zarrinbakhsh, P. Ocular contact lenses: smart materials for biomedical applications. Polymer Bulletin 2024, 81, 7791–7832. [Google Scholar] [CrossRef]
- Adrus, N.; bin Mohd Farizal, M.A.; Jamaluddin, J.; bin Syaiful Azim, F.S.; Mizi, F.M.; Nanda Kumar, S.; Govindasamy, J.J. Hydrogel as a Foundational Material for Contact Lens. In Contact Lenses: Research, Industry, and User Perspectives, Adrus, N., bin Mohd Farizal, M.A., Jamaluddin, J., bin Syaiful Azim, F.S., Mizi, F.M., Nanda Kumar, S., Govindasamy, J.J., Eds.; Springer Nature Singapore: Singapore, 2025; pp. 13–42.
- Li, Z.; Cheng, H.; Ke, L.; Liu, M.; Wang, C.; Jun Loh, X.; Li, Z.; Wu, Y. Recent advances in new copolymer hydrogel-formed contact lenses for ophthalmic drug delivery. ChemNanoMat 2021, 7, 564–579. [Google Scholar] [CrossRef]
- Wei, S.; Yin, R.; Tang, T.; Wu, Y.; Liu, Y.; Wang, P.; Wang, K.; Mei, M.; Zou, R.; Duan, X. Gas-permeable, irritation-free, transparent hydrogel contact lens devices with metal-coated nanofiber mesh for eye interfacing. ACS nano 2019, 13, 7920–7929. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, S.; Chen, X.; Yung, A.; Naderi, A.; Ghovvati, M.; Liu, Y.; Farzad, A.; Mostafavi, A.; Dana, R.; Annabi, N. Development and optimization of an ocular hydrogel adhesive patch using definitive screening design (DSD). Biomaterials Science 2023, 11, 1318–1334. [Google Scholar] [CrossRef] [PubMed]
- Tighsazzadeh, M.; Boateng, J. Matrix hyaluronic acid and bilayer poly-hydroxyethyl methacrylate-hyaluronic acid films as potential ocular drug delivery platforms. International Journal of Biological Macromolecules 2024, 260, 129496. [Google Scholar] [CrossRef] [PubMed]
- Manjeri, A.; George, S.D. Hydrogel-embedded polydimethylsiloxane contact lens for ocular drug delivery. ACS Applied Bio Materials 2024, 7, 7324–7331. [Google Scholar] [CrossRef]
- Kulbay, M.; Wu, K.Y.; Truong, D.; Tran, S.D. Smart molecules in ophthalmology: Hydrogels as responsive systems for ophthalmic applications. Smart molecules 2024, 2, e20230021. [Google Scholar] [CrossRef]
- Wei, R.; Wang, Y.; Feng, Z.; Liu, R.; Liu, C.; Hu, X.; Liu, Y.; Kong, B.; Zhou, X.; Li, M. Self-healing adhesive oxidized guar gum hydrogel loaded with mesenchymal stem cell exosomes for corneal wound healing. Journal of Nanobiotechnology 2025, 23, 321. [Google Scholar] [CrossRef]
- Safi, S.Z.; Fazil, S.; Saeed, L.; Shah, H.; Arshad, M.; Alobaid, H.M.; Rehman, F.; Sharif, F.; Selvaraj, C.; Orakzai, A.H. Chitosan-and heparin-based advanced hydrogels: their chemistry, structure and biomedical applications. Chemical Papers 2024, 78, 9287–9309. [Google Scholar] [CrossRef]
- Truong, D.; Wu, K.Y.; Nguyen, L.; Tran, S.D. Advancements in hydrogel technology for ocular drug delivery. Exploration of BioMat-X 2024, 1, 331–352. [Google Scholar] [CrossRef]
- Lynch, C.R.; Kondiah, P.P.D.; Choonara, Y.E.; du Toit, L.C.; Ally, N.; Pillay, V. Hydrogel Biomaterials for Application in Ocular Drug Delivery. Frontiers in Bioengineering and Biotechnology 2020, Volume 8 - 2020. [CrossRef]
- Chattopadhyay, D.; Orasugh, J.T.; Adhikari, A.; Ray, S.S. Stimuli-Responsive Hydrogels for Ophthalmic Drug Delivery; Elsevier: 2024.
- Arabpour, Z.; Salehi, M.; An, S.; Moghtader, A.; Anwar, K.N.; Baharnoori, S.M.; Shah, R.J.; Abedi, F.; Djalilian, A.R. Exploring Hydrogel Nanoparticle Systems for Enhanced Ocular Drug Delivery. Gels 2024, 10, 589. [Google Scholar] [CrossRef]
- Akbari, E.; Imani, R.; Shokrollahi, P.; Jarchizadeh, R. Hydrogel-based formulations for drug delivery to the anterior segment of the eye. Journal of Drug Delivery Science and Technology 2023, 81, 104250. [Google Scholar] [CrossRef]
- Yan, K.; Zhang, Q.; Liu, Q.; Han, Y.; Liu, Z. Advances in adhesive hydrogels applied for ophthalmology: An overview focused on the treatment. Theranostics 2025, 15, 915. [Google Scholar] [CrossRef] [PubMed]
- A Phase 3, Multicenter, Randomized, Double-Masked, Active-Controlled Study to Evaluate the Efficacy and Safety of Treatment With CsA-PG Ophthalmic Gel in Dry Eye Patients. 2025.
- Ullah, A.; Kim, D.Y.; Lim, S.I.; Lim, H.-R. Hydrogel-Based Biointerfaces: Recent Advances, Challenges, and Future Directions in Human–Machine Integration. Gels 2025, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, A.; Haag, R.; Schedler, U. Hydrogels and their role in biosensing applications. Advanced healthcare materials 2021, 10, 2100062. [Google Scholar] [CrossRef]
- Sagdic, K.; Fernández-Lavado, E.; Mariello, M.; Akouissi, O.; Lacour, S.P. Hydrogels and conductive hydrogels for implantable bioelectronics. MRS Bulletin 2023, 48, 495–505. [Google Scholar] [CrossRef]
- Wang, M.; Zheng, Y.; Yin, C.; Dai, S.; Fan, X.; Jiang, Y.; Liu, X.; Fang, J.; Yi, B.; Zhou, Q.; et al. Recent Progress in antibacterial hydrogel coatings for targeting biofilm to prevent orthopedic implant-associated infections. Frontiers in Microbiology 2023, Volume 14 - 2023. [CrossRef]
- Leng, J.; He, Y.; Yuan, Z.; Tao, B.; Li, K.; Lin, C.; Xu, K.; Chen, M.; Dai, L.; Li, X.; et al. Enzymatically-degradable hydrogel coatings on titanium for bacterial infection inhibition and enhanced soft tissue compatibility via a self-adaptive strategy. Bioactive Materials 2021, 6, 4670–4685. [Google Scholar] [CrossRef]
- Gao, Q.; Sun, F.; Li, Y.; Li, L.; Liu, M.; Wang, S.; Wang, Y.; Li, T.; Liu, L.; Feng, S.; et al. Biological Tissue-Inspired Ultrasoft, Ultrathin, and Mechanically Enhanced Microfiber Composite Hydrogel for Flexible Bioelectronics. Nano-Micro Letters 2023, 15, 139. [Google Scholar] [CrossRef]
- Jensen, M.J.; Peel, A.; Horne, R.; Chamberlain, J.; Xu, L.; Hansen, M.R.; Guymon, C.A. Antifouling and Mechanical Properties of Photografted Zwitterionic Hydrogel Thin-Film Coatings Depend on the Cross-Link Density. ACS Biomaterials Science & Engineering 2021, 7, 4494–4502. [Google Scholar] [CrossRef]
- Si, Y.; Xu, L.; Wang, N.; Zheng, J.; Yang, R.; Li, J. Target microRNA-responsive DNA hydrogel-based surface-enhanced Raman scattering sensor arrays for microRNA-marked cancer screening. Analytical chemistry 2020, 92, 2649–2655. [Google Scholar] [CrossRef]
- Tavakoli, J.; Tang, Y. Hydrogel Based Sensors for Biomedical Applications: An Updated Review. Polymers 2017, 9, 364. [Google Scholar] [CrossRef]
- Saeidi, M.; Chenani, H.; Orouji, M.; Adel Rastkhiz, M.; Bolghanabadi, N.; Vakili, S.; Mohamadnia, Z.; Hatamie, A.; Simchi, A. Electrochemical Wearable Biosensors and Bioelectronic Devices Based on Hydrogels: Mechanical Properties and Electrochemical Behavior. Biosensors 2023, 13, 823. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Afshari, R.; Jain, S.; Zheng, Y.; Lin, M.-H.; Zenkar, S.; Yin, J.; Chen, J.; Peppas, N.A.; Annabi, N. Advances in conducting nanocomposite hydrogels for wearable biomonitoring. Chemical Society Reviews 2025, 54, 2595–2652. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.; Chien, J.C.; Axpe, E.; Blankemeier, L.; Baker, S.W.; Swaminathan, S.; Piunova, V.A.; Zubarev, D.Y.; Maikawa, C.L.; Grosskopf, A.K. Combinatorial polyacrylamide hydrogels for preventing biofouling on implantable biosensors. Advanced Materials 2022, 34, 2109764. [Google Scholar] [CrossRef] [PubMed]
- Roh, S.; Jang, S.Y.; Jung, Y.; Lee, K.; Yoo, J. Development of Substrate-Independent Antifouling and Bactericidal Surfaces Using Visible Light Cross-Linked Hydrogel Coatings for Biomedical Applications. Advanced Healthcare Materials 2025, 14, 2402565. [Google Scholar] [CrossRef]
- Rong, H.; Sun, S.; Lu, M.; Zhang, Y.; Liu, L.; Guo, Z.; Zhang, Z.; Ye, Z.; Zhang, J.; Chen, B. Super-hydrophilic and super-lubricating Zwitterionic hydrogel coatings coupled with polyurethane to reduce postoperative dura mater adhesions and infections. Acta Biomaterialia 2025, 192, 206–217. [Google Scholar] [CrossRef]
- Pan, F.; Zhang, S.; Altenried, S.; Zuber, F.; Chen, Q.; Ren, Q. Advanced antifouling and antibacterial hydrogels enabled by controlled thermo-responses of a biocompatible polymer composite. Biomaterials Science 2022, 10, 6146–6159. [Google Scholar] [CrossRef]
- Wancura, M.; Nkansah, A.; Robinson, A.; Toubbeh, S.; Talanker, M.; Jones, S.; Cosgriff-Hernandez, E. PEG-Based Hydrogel Coatings: Design Tools for Biomedical Applications. Annals of Biomedical Engineering 2024, 52, 1804–1815. [Google Scholar] [CrossRef]
- Pan, M.; Shui, T.; Zhao, Z.; Xiang, L.; Yan, B.; Gu, N.; Zeng, H. Engineered Janus hydrogels: biomimetic surface engineering and biomedical applications. National Science Review 2024, 11, nwae316. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hu, Y.; Ju, Y.; Yang, P.; Shen, N.; Yang, A.; Wu, R.; Fang, B.; Liu, L. Immunomodulatory hydrogels for tissue repair and regeneration. APL Materials 2024, 12. [Google Scholar] [CrossRef]
- Quazi, M.Z.; Hwang, J.; Song, Y.; Park, N. Hydrogel-Based Biosensors for Effective Therapeutics. Gels 2023, 9, 545. [Google Scholar] [CrossRef] [PubMed]
- Lucío, M.I.; Cubells-Gómez, A.; Maquieira, Á.; Bañuls, M.-J. Hydrogel-based holographic sensors and biosensors: past, present, and future. Analytical and Bioanalytical Chemistry 2022, 414, 993–1014. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Gerber, A.; Santa, C.; Aktug, G.; Hengerer, B.; Clark, H.A.; Jonas, U.; Dostalek, J.; Sergelen, K. Molecularly responsive aptamer-functionalized hydrogel for continuous plasmonic biomonitoring. Journal of the American Chemical Society 2025, 147, 11485–11500. [Google Scholar] [CrossRef]
- Chen, B.; Zhu, Y.; Yu, R.; Feng, Y.; Han, Z.; Liu, C.; Zhu, P.; Lu, L.; Mao, Y. Recent Progress of Biomaterial-Based Hydrogels for Wearable and Implantable Bioelectronics. Gels 2025, 11, 442. [Google Scholar] [CrossRef]
- Yang, G.; Qiu, Y.; Pang, B.; Guo, W.; Liu, S.; Zheng, Q.; Zhou, S.; Tian, J.; Liu, W.; Xie, B. A reusable hydrogel biosensor array with electrically responsive hydrogel interfaces for noninvasive locating of perforating arteries. Science Advances 2025, 11, eadw6166. [Google Scholar] [CrossRef]
- Zahid, S.; Ali, R.; Kousar, S.; Yousafzai, M.A.Z.; Muhammad, R.K.; Iqbal, K.; Mahmood, W.; Hasan, T.; Khan, M. ENZYME-RESPONSIVE HYDROGELS FOR TARGETED THERAPEUTIC DELIVERY AND DIAGNOSTIC APPLICATIONS.
- Solanki, R.; Bhatia, D. Stimulus-Responsive Hydrogels for Targeted Cancer Therapy. Gels 2024, 10, 440. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Heydarpour, R.; Tehrani, Z.M. Multi-stimuli-responsive hydrogels and their medical applications. New Journal of Chemistry 2021, 45, 15705–15717. [Google Scholar] [CrossRef]
- Toews, P.M.; Velraj, A.; Bates, J.S. Stimuli-responsive hydrogels, their mass transfer, intermolecular interactions, and applications in biomedical devices. Journal of Materials Science: Materials in Engineering 2025, 20, 66. [Google Scholar] [CrossRef]
- Sojdeh, S.; Panjipour, A.; Yaghmour, A.; Arabpour, Z.; Djalilian, A.R. Click Chemistry-Based Hydrogels for Tissue Engineering. Gels 2025, 11, 724. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Zhao, Q.; Xie, T.; Wu, J. DLP 3D printing of electrically conductive hybrid hydrogels via polymerization-induced phase separation and subsequent in situ assembly of polypyrrole. Journal of Materials Chemistry A 2024, 12, 5348–5356. [Google Scholar] [CrossRef]
- Chakrapani, G.; Zare, M.; Ramakrishna, S. Intelligent hydrogels and their biomedical applications. Materials Advances 2022, 3, 7757–7772. [Google Scholar] [CrossRef]
- Bordbar-Khiabani, A.; Gasik, M. Smart Hydrogels for Advanced Drug Delivery Systems. International Journal of Molecular Sciences 2022, 23, 3665. [Google Scholar] [CrossRef]
- Cao, J.; Zhang, Z.; Li, K.; Ma, C.; Zhou, W.; Lin, T.; Xu, J.; Liu, X. Self-Healable PEDOT:PSS-PVA Nanocomposite Hydrogel Strain Sensor for Human Motion Monitoring. Nanomaterials 2023, 13, 2465. [Google Scholar] [CrossRef]
- Ahmad Ruzaidi, D.A.; Maurya, M.R.; Yempally, S.; Abdul Gafoor, S.; Geetha, M.; Che Roslan, N.; Cabibihan, J.-J.; Kumar Sadasivuni, K.; Mahat, M.M. Revealing the improved sensitivity of PEDOT:PSS/PVA thin films through secondary doping and their strain sensors application. RSC Advances 2023, 13, 8202–8219. [Google Scholar] [CrossRef]
- Jurin, F.E.; Buron, C.C.; Frau, E.; del Rossi, S.; Schintke, S. The Electrical and Mechanical Characteristics of Conductive PVA/PEDOT:PSS Hydrogel Foams for Soft Strain Sensors. Sensors 2024, 24, 570. [Google Scholar]
- Rumon, M.M.H.; Rahman, M.S.; Akib, A.A.; Sohag, M.S.; Rakib, M.R.A.; Khan, M.A.R.; Yesmin, F.; Shakil, M.S.; Rahman Khan, M.M. Progress in hydrogel toughening: addressing structural and crosslinking challenges for biomedical applications. Discover Materials 2025, 5, 5. [Google Scholar] [CrossRef]
- Lin, X.; Zhao, X.; Xu, C.; Wang, L.; Xia, Y. Progress in the mechanical enhancement of hydrogels: Fabrication strategies and underlying mechanisms. Journal of polymer science 2022, 60, 2525–2542. [Google Scholar] [CrossRef]
- Li, X.; Gong, J.P. Design principles for strong and tough hydrogels. Nature Reviews Materials 2024, 9, 380–398. [Google Scholar] [CrossRef]
- Heinemann, C.; Buchner, F.; Lee, P.S.; Bernhardt, A.; Kruppke, B.; Wiesmann, H.-P.; Hintze, V. Effects of Gamma Irradiation and Supercritical Carbon Dioxide Sterilization on Methacrylated Gelatin/Hyaluronan Hydrogels. Journal of Functional Biomaterials 2023, 14, 317. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, R.; Espinosa-Andrews, H.; García-Carvajal, Z.Y. Stimuli-Responsive Hydrogels in Drug Delivery. In Functional Biomaterials: Drug Delivery and Biomedical Applications, Jana, S., Jana, S., Eds.; Springer Singapore: Singapore, 2022; pp. 75–103. [Google Scholar]
- Khonakdar, H.; Ehsani, M.; Naderi, G.; Shokrolahi, F.; Khonakdar, H.A. Recent Advances in Injectable Stimuli-Responsive Hydrogels for Biomedical Applications. Journal of Polymers and the Environment 2025, 33, 3512–3554. [Google Scholar] [CrossRef]
- Fang, Z.; Chen, P.; Ji, Q.; Yan, C.; Gong, A. Stimuli-responsive hydrogel for disease therapy. Polymer Bulletin 2024, 81, 1981–2000. [Google Scholar] [CrossRef]
- Fam, S.B. Regulations for Hydrogels and Bioinks in Clinical Trials. In Hydrogels and Bioinks in Tissue Engineering, Hasirci, N., Hasirci, V., Eds.; Springer Nature Switzerland: Cham, 2025; pp. 359–367.
- Clegg, J.R.; Adebowale, K.; Zhao, Z.; Mitragotri, S. Hydrogels in the clinic: An update. Bioengineering & Translational Medicine 2024, 9, e10680. [Google Scholar] [CrossRef]
- El Sayed, M.M. Production of polymer hydrogel composites and their applications. Journal of Polymers and the Environment 2023, 31, 2855–2879. [Google Scholar] [CrossRef]
- Li, B.; Wang, Z.; Huang, C.; Xu, L.; Huang, S.; Qu, M.; Xu, Z.; Zhang, D.; Guo, B.; Ji, C. Optimization of 3D Printing Process for Hydrogel-Based Thermal Management Patches in Engineering Machinery. In Proceedings of the The 9th International Conference on Advances in Construction Machinery and Vehicle Engineering, Singapore, 2026//, 2026; pp. 235–247.
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal transduction and targeted therapy 2021, 6, 426. [Google Scholar] [CrossRef]
- Huang, X.; Fu, D.; Zha, X.; Ling, T.; Huang, J. High-precision 3D printing of hydrogel: Material innovations, process breakthroughs, and translational applications in regenerative medicine. APL Materials 2025, 13. [Google Scholar] [CrossRef]
- Musuc, A.M.; Chelu, M. Biomaterials-Based Hydrogels for Therapeutic Applications. In Biomaterials in Microencapsulation, Sharma, A., Ed.; IntechOpen: London, 2024.
- Negut, I.; Bita, B. Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review. Gels 2023, 9, 845. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Chen, X.; Wang, S.; Chen, Z.; Pan, P.; Huang, Q. Integrating machine learning for the optimization of polyacrylamide/alginate hydrogel. Regenerative Biomaterials 2024, 11, rbae109. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Song, P.; Li, G.; Han, Y.; Ren, X.; Bai, L.; Su, J. AI energized hydrogel design, optimization and application in biomedicine. Materials Today Bio 2024, 25, 101014. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Yazdanpanah, S.; Petillo, O.; Conte, R.; Sepe, F.; Peluso, G.; Calarco, A. Sustainable Hydrogels for Medical Applications: Biotechnological Innovations Supporting One Health. Gels 2025, 11, 559. [Google Scholar] [CrossRef]
- Nanda, D.; Behera, D.; Pattnaik, S.S.; Behera, A.K. Advances in natural polymer-based hydrogels: synthesis, applications, and future directions in biomedical and environmental fields. Discover Polymers 2025, 2, 6–6. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Janarthanan, G.; Kang, J.; Noh, I. Status for Commercialization of FDA-approved Hydrogels and Their Intellectual Properties. 2021.
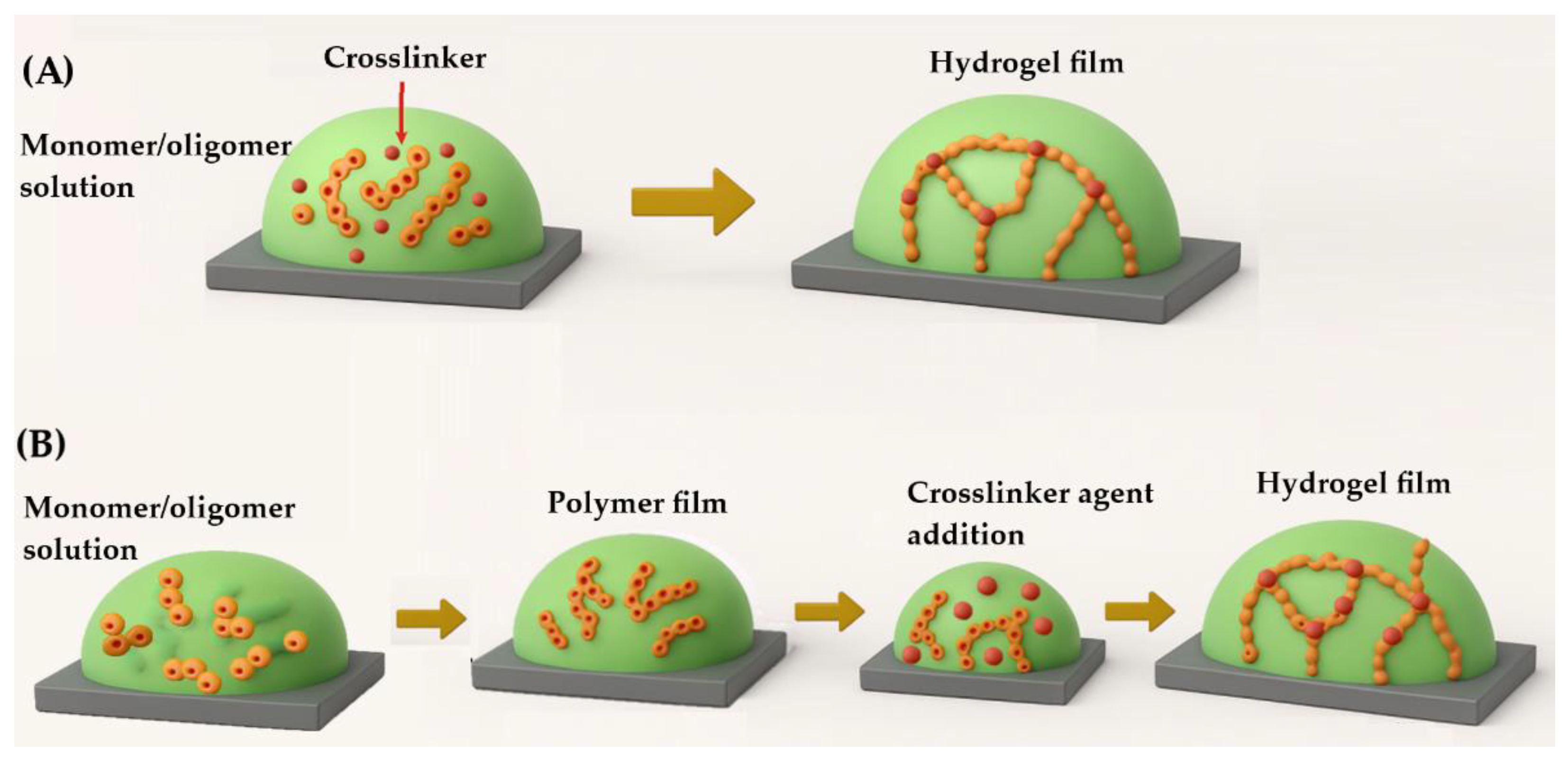
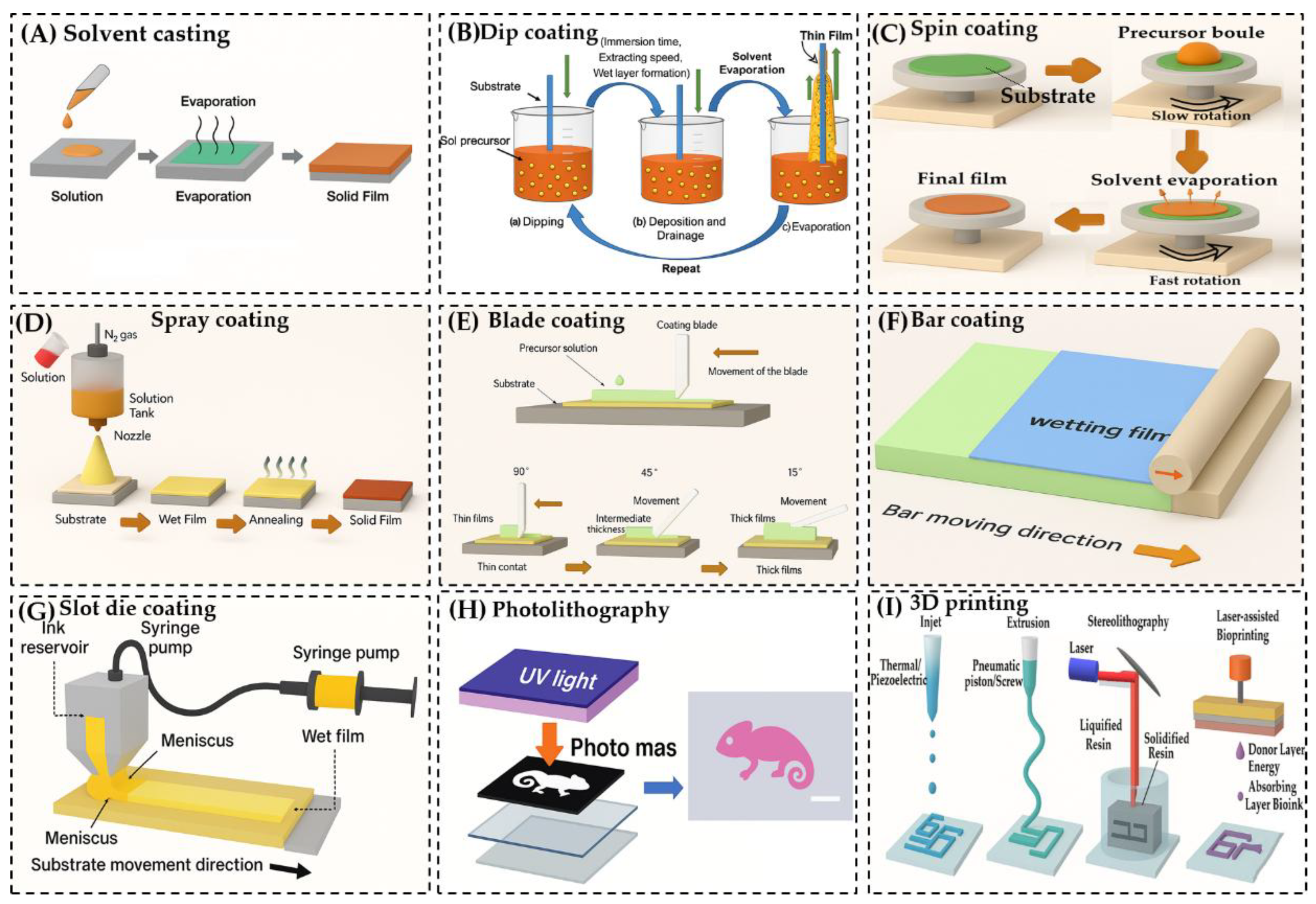
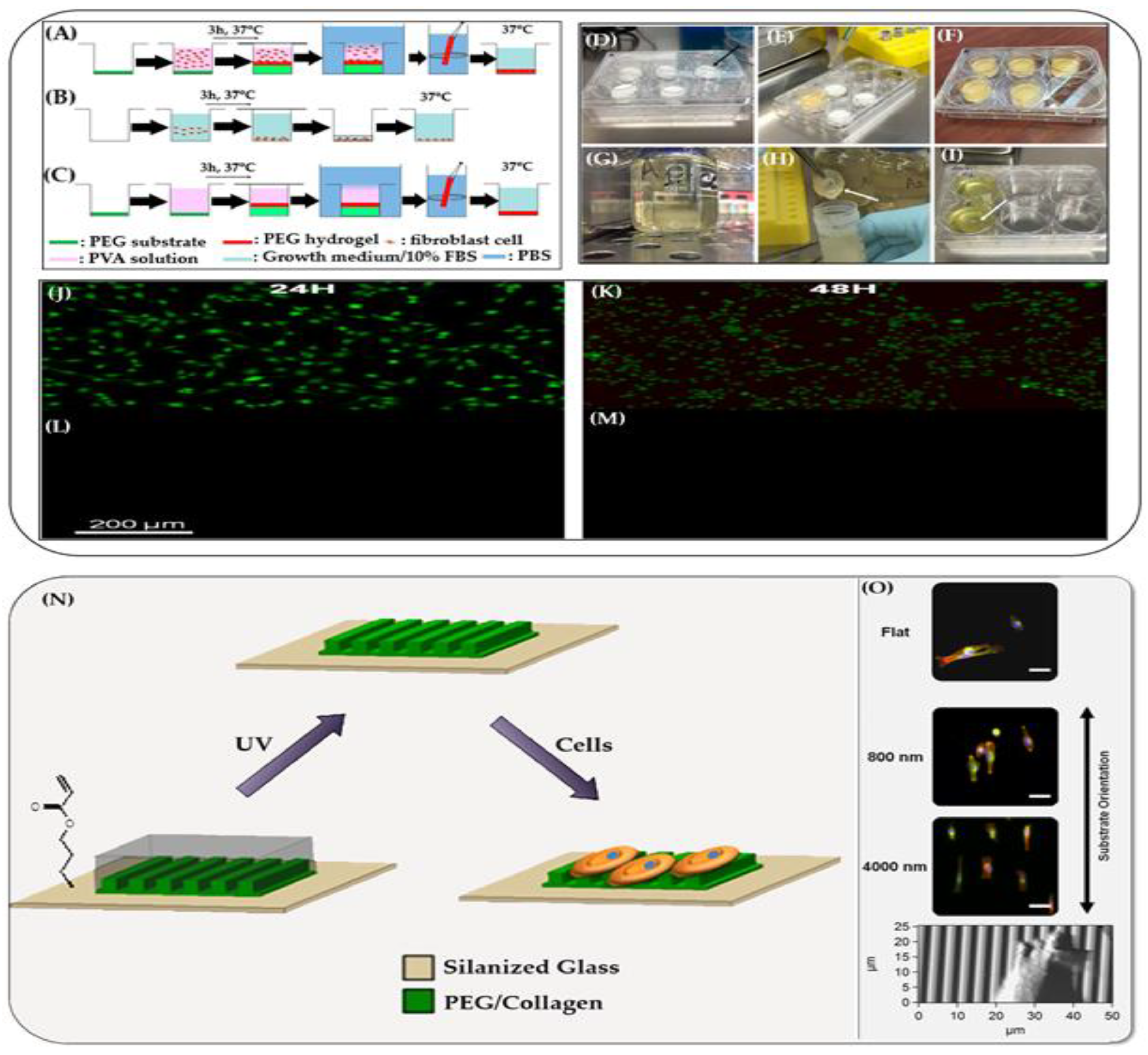


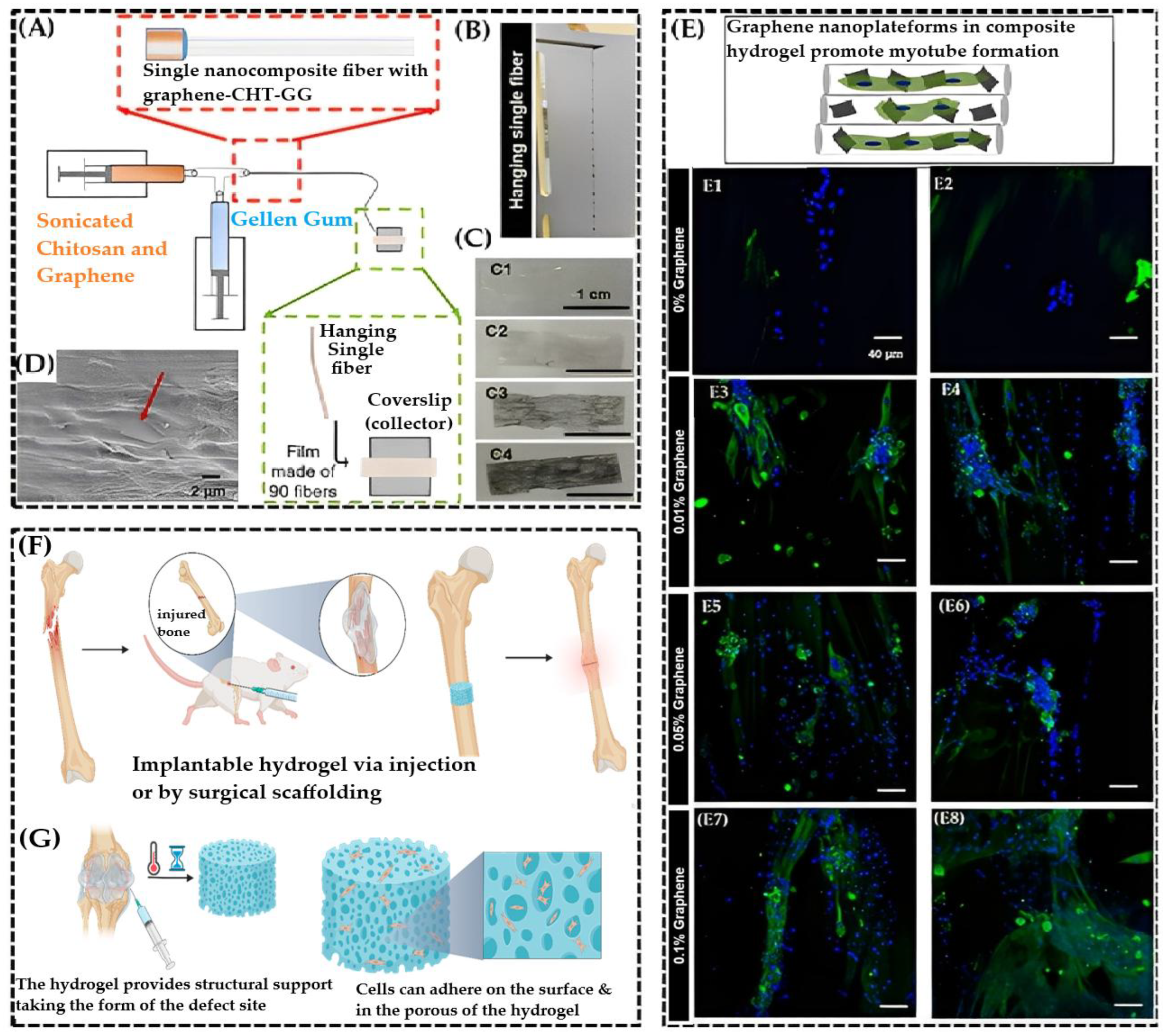
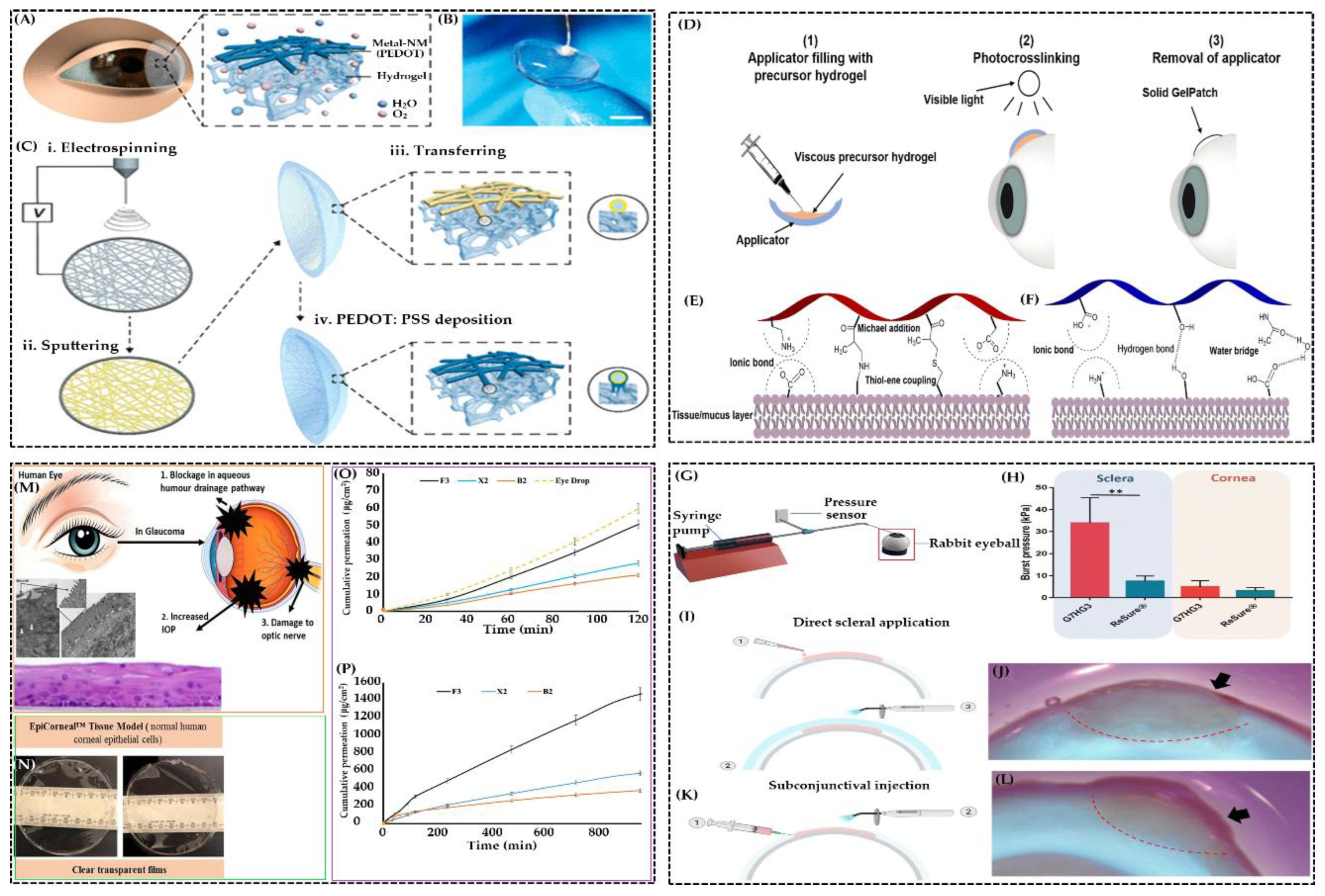
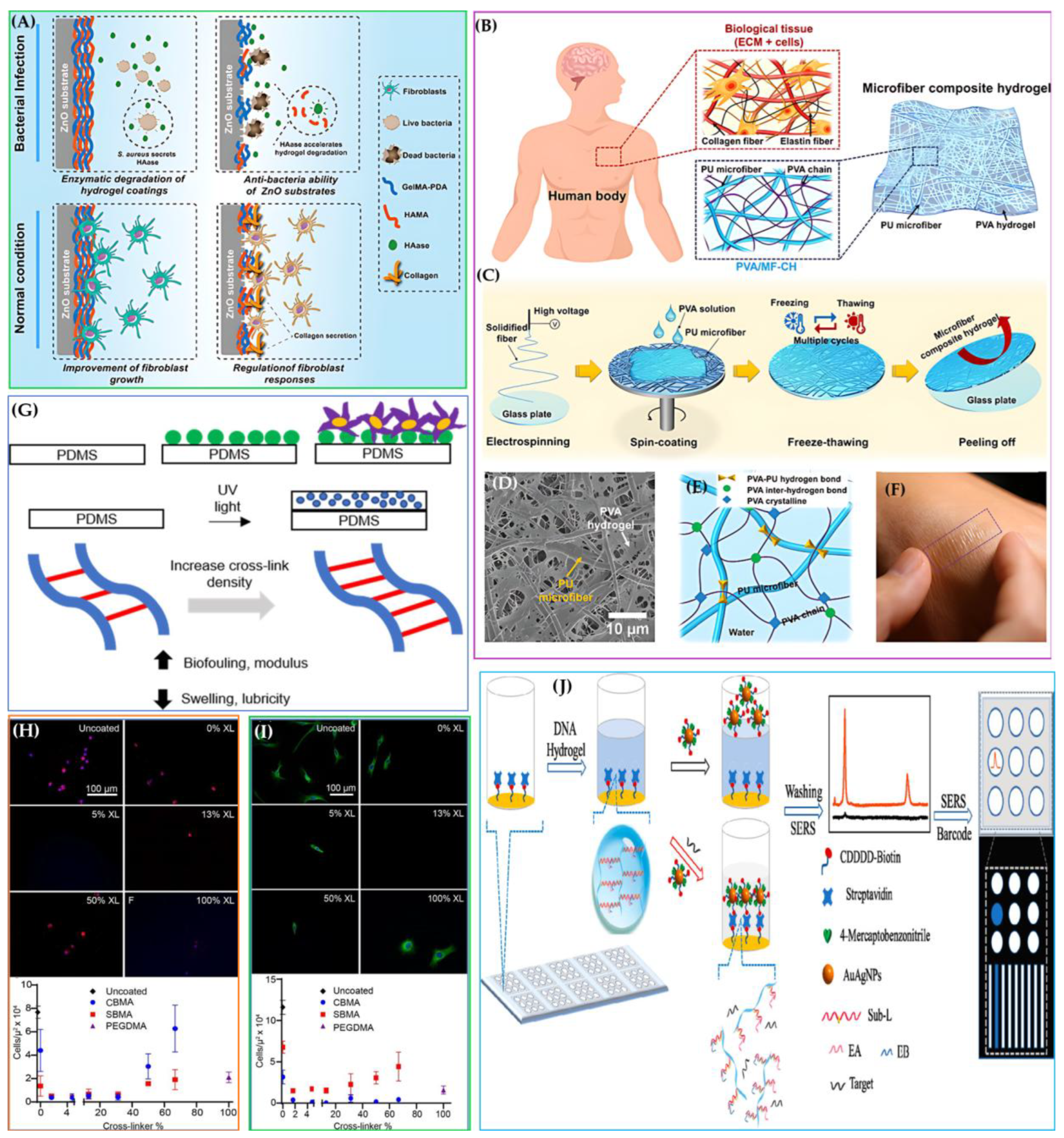
| Technique | Cost | Scalability | Film Uniformity | Min. Thickness | Patterning Ability | Surface Compatibility | Material Waste | Drying Time | Speed | Process Complexity |
| Solvent Casting | Low | Limited | Moderate | Tens of micrometers | Yes | Smooth, flat surfaces | High | Long | Slow | Simple |
| Dip Coating | Low | Not scalable | High | Nanometers | Yes | Rigid, complex shapes | Moderate | Slow | Slow | Simple |
| Spin Coating | Medium | Potential | High | Nanometers | No | Small, flat substrates | Moderate | Fast | Very slow | Moderate |
| Spray Coating | High | Potential | Low | Tens–hundreds of nm | No | Flat or curved, flexible/rigid | Moderate | Fast | Fast | Moderate |
| Blade Coating | Medium | Limited | Moderate | Tens of micrometers | Yes | Flexible/rigid substrates | Moderate | Slow | Fast | Simple |
| Bar Coating | Medium | Scalable | Moderate | Nanometers | No | Flexible/rigid substrates | Moderate | Slow | Medium | Moderate |
| Slot Die Coating | High | Scalable | Moderate | Tens–hundreds of nm | Limited | Flexible/rigid substrates | Moderate | Slow | Fast | Complex |
| Photolithography | Very High | Not scalable | Very High | Micrometers | Yes | Glass, polymers, inorganic (needs pretreatment) | Moderate | Medium | Medium | High |
| 3D Printing | High | Limited | Moderate | Micrometers | Yes | Glass, polymers, inorganic (needs pretreatment) | Moderate | Medium | Medium | Complex |
| Electrospinning | Medium | Scalable | High | Nanometers | Limited | Flat or 3D surfaces, flexible/rigid | Low | Fast | Fast | Moderate–High |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




