Submitted:
23 September 2025
Posted:
25 September 2025
You are already at the latest version
Abstract
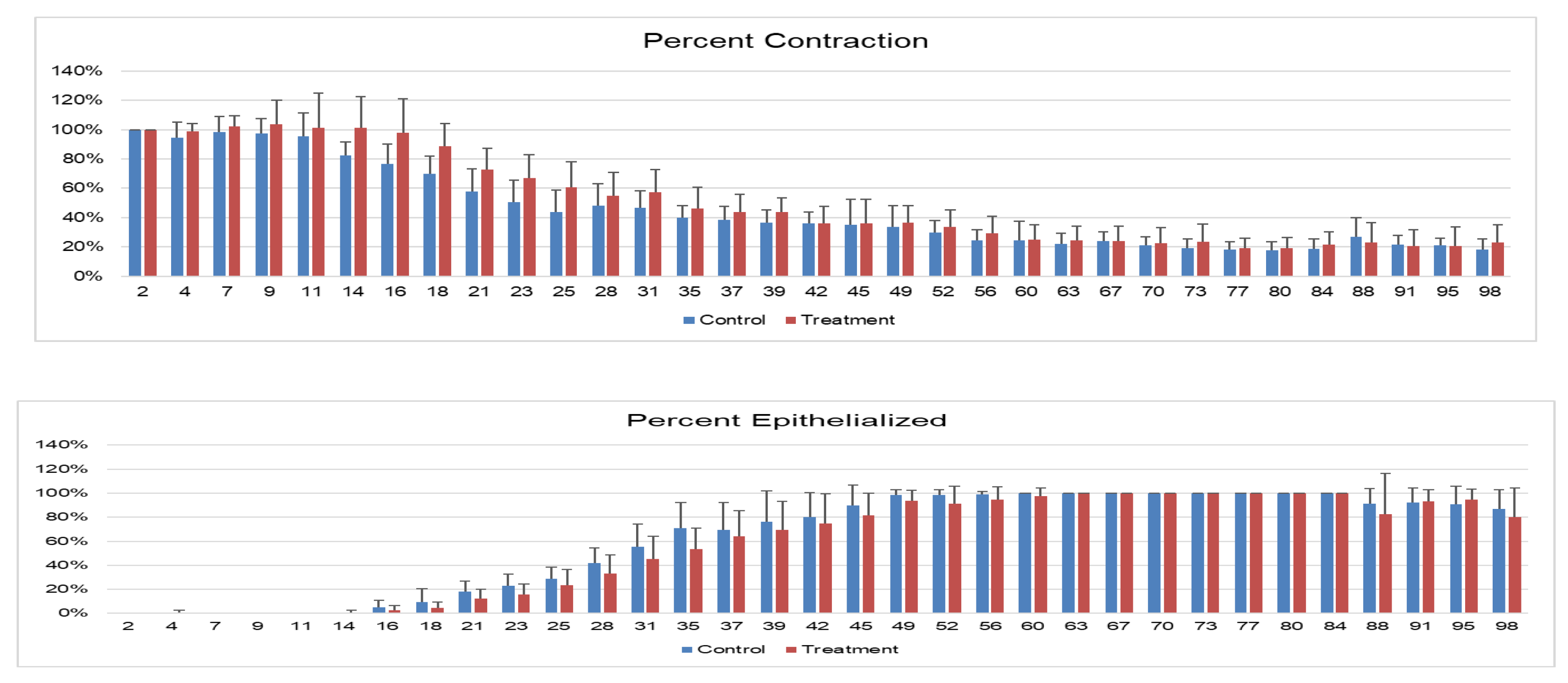
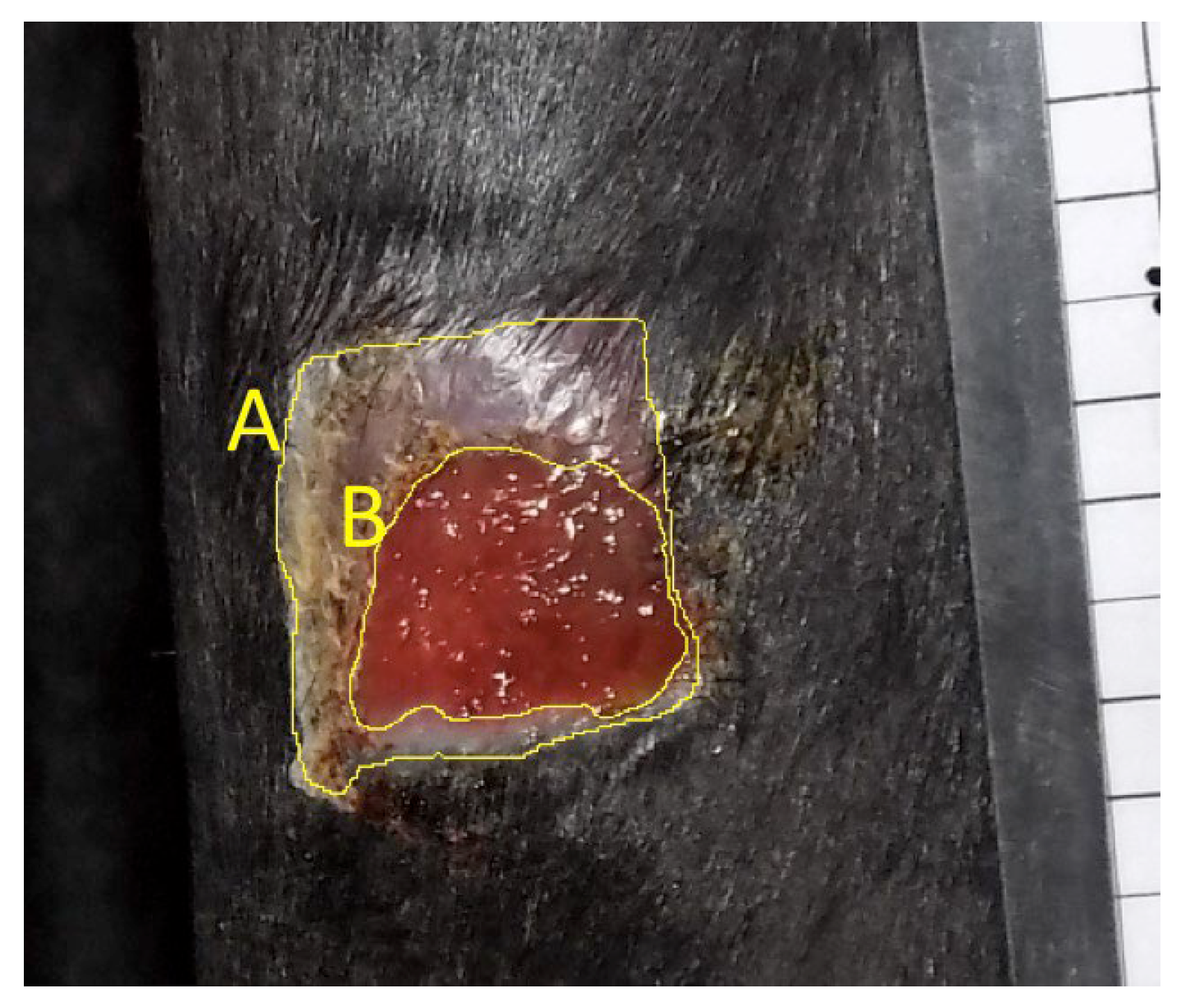
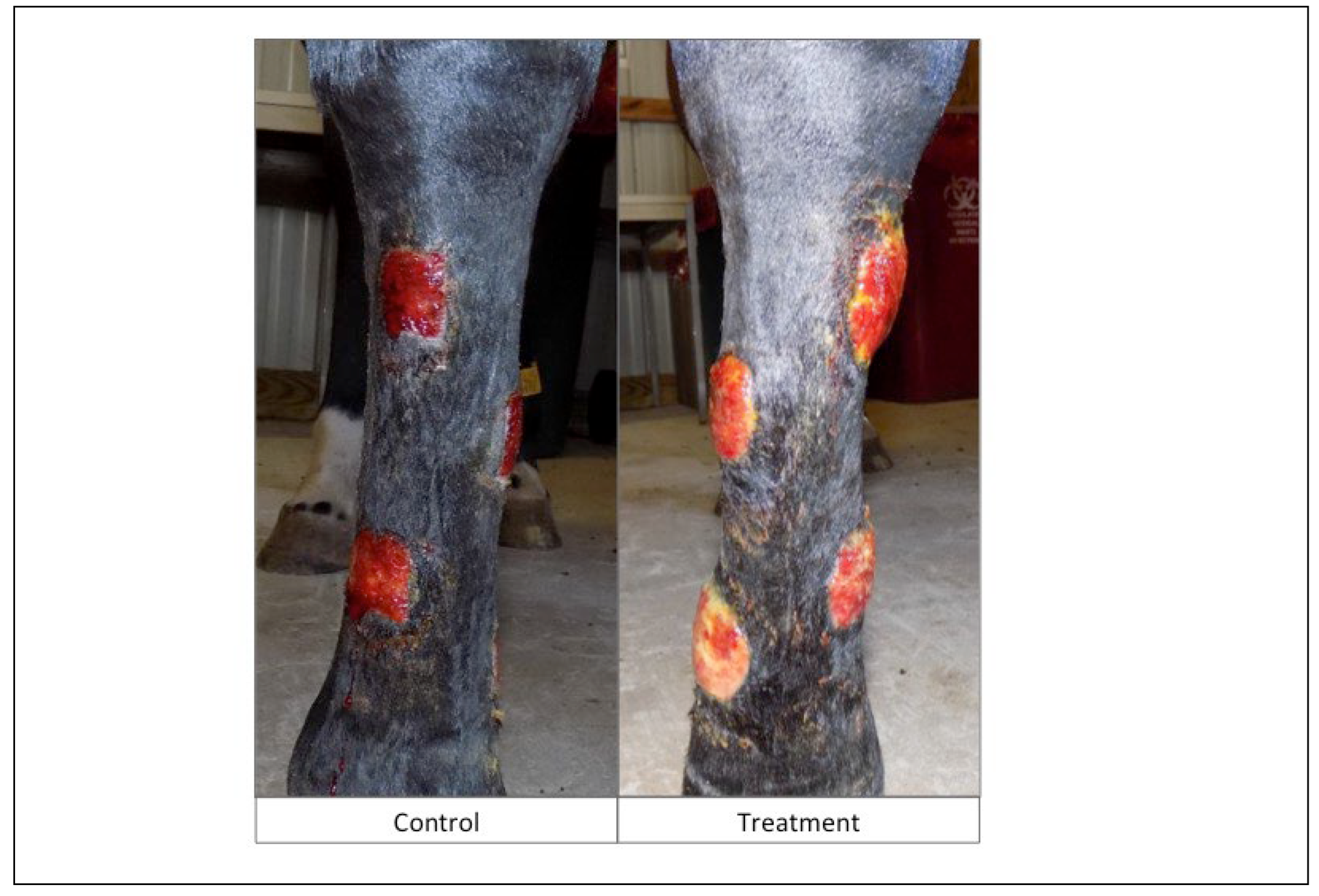
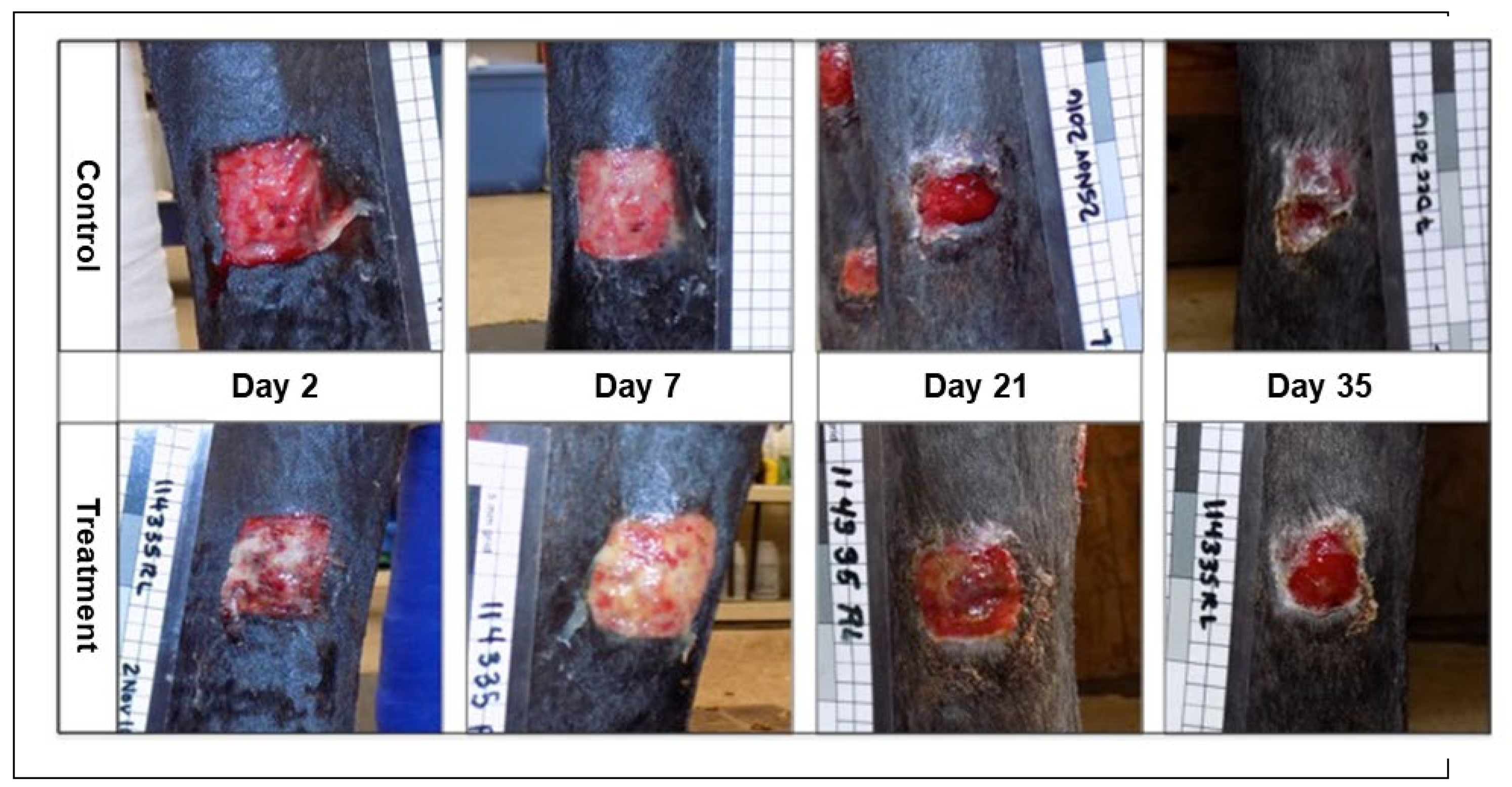
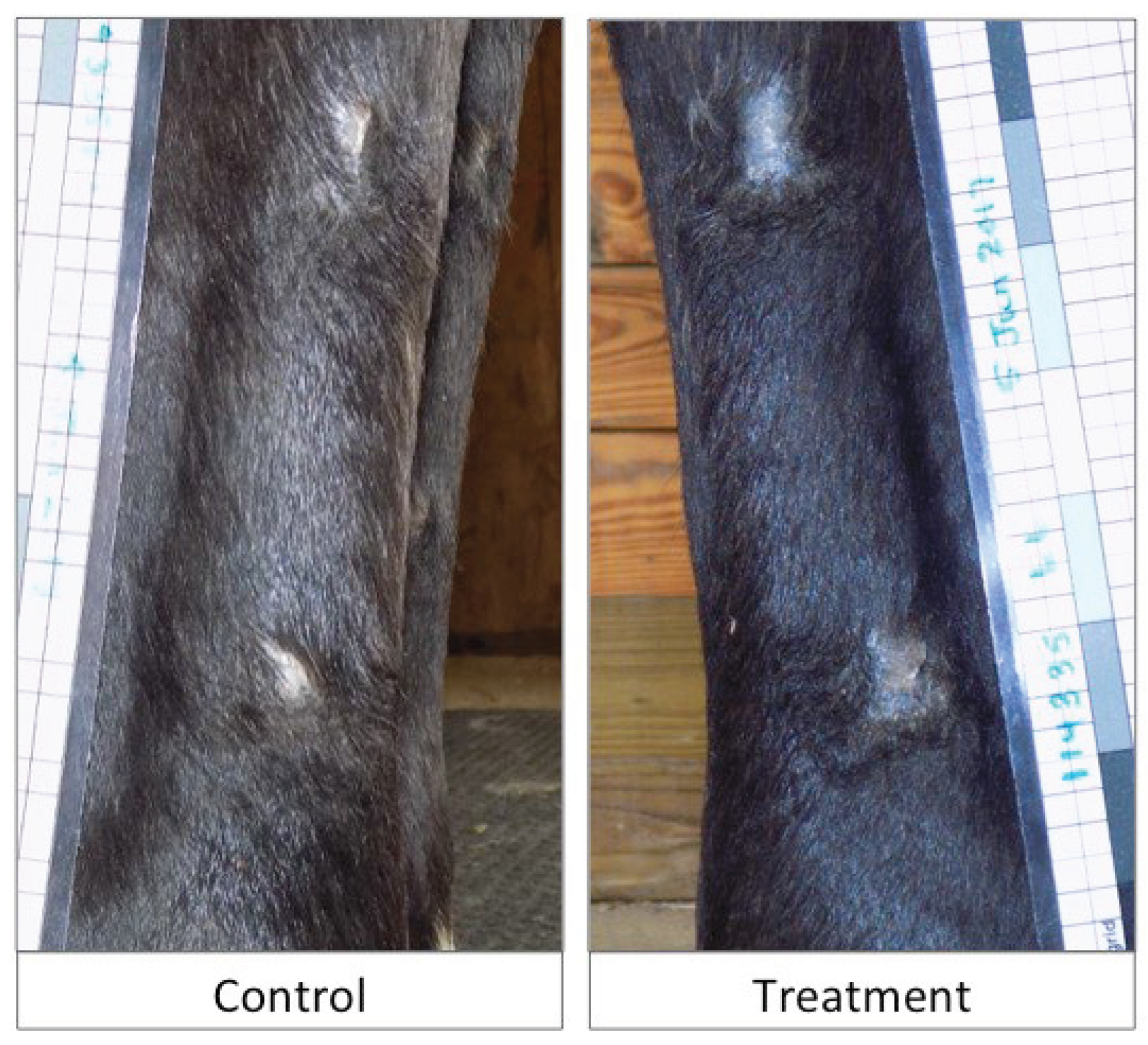
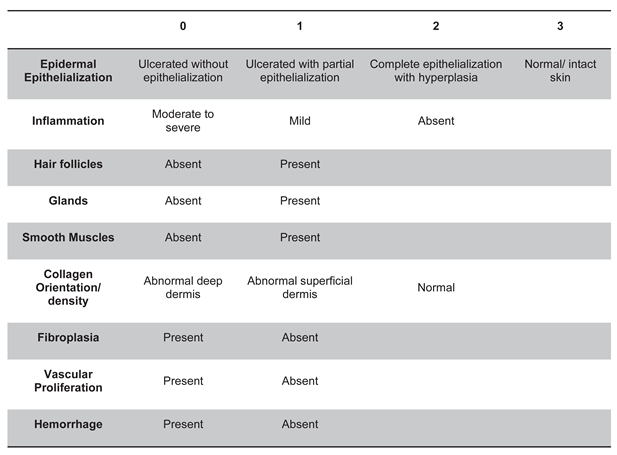
Objective: To assess the safety and efficacy of lyophilized, milled, human amnion on wound healing in an equine distal limb wound model in a blinded controlled study. Animals: Four clinically normal adult horses (3 Thoroughbred and 1 Paint, age 6-19 years) obtained via donation. Institutional Animal Care and Use Committee approval was obtained for this study. Procedures: One forelimb of each horse was randomly assigned to the treatment group, and the contralateral limb was assigned as the control. Full-thickness skin wounds were created on each metacarpus. Treatment limb wounds were treated with lyophilized, milled, human-derived amnion material delivered on triple antibiotic ointment on non-adherent dressing. Control wounds were treated with triple antibiotic ointment on non-adherent dressing. Distal limb bandages were applied. Digital photographs of the wounds every 2-4 days throughout a 98-day study period were blinded and analyzed for wound area and epithelialization. Histopathology was performed on samples from days 7, 21, 35, and 84. Results: All treatment limbs exhibited an inflammatory response characterized by focal edema and discharge from the wounds, and this was not seen in control limbs. One horse developed unilateral cellulitis in the treatment limb, and not the control limb that resolved with additional treatment. Wounds were completely epithelialized in control limbs sooner than treatment limbs in each horse (p = 0.011). Mean wound size (junction of neoepithelium and wound margin) was smaller in control wounds than treatment wounds at all time points, with a significant difference on days 23 and 39, with trends toward significance on days 18 and 35. Overall histologic scores were better in control wounds than in amnion-treated wounds at all time points, only reaching significance on day 84 (p = 0.039). Conclusions and Clinical Relevance: In this pilot study, lyophilized human amnion did not improve wound healing compared to control, and may have hindered wound healing. We terminated the study after one cohort of 4 horses due to these results. Because wounds treated with amnion in this pilot study exhibited an inflammatory response that resulted in delayed time to wound closure, human lyophilized milled amnion is not recommended for use in equine wound management. Xenogeneic amnion may not be appropriate for wound healing in equines, but further study is needed to strengthen these conclusions.
Keywords:
Introduction
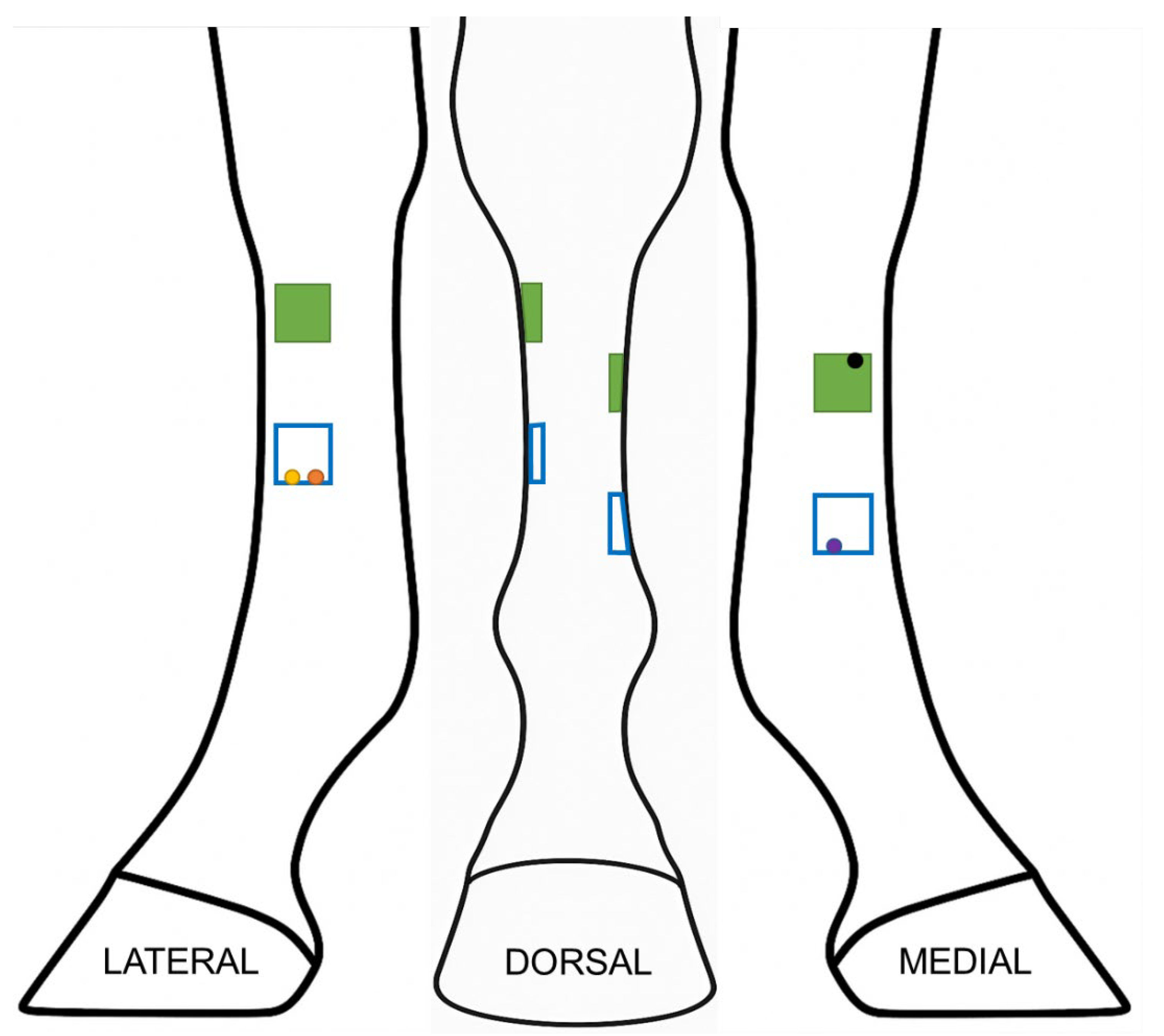
2. Materials and Methods
3. Results
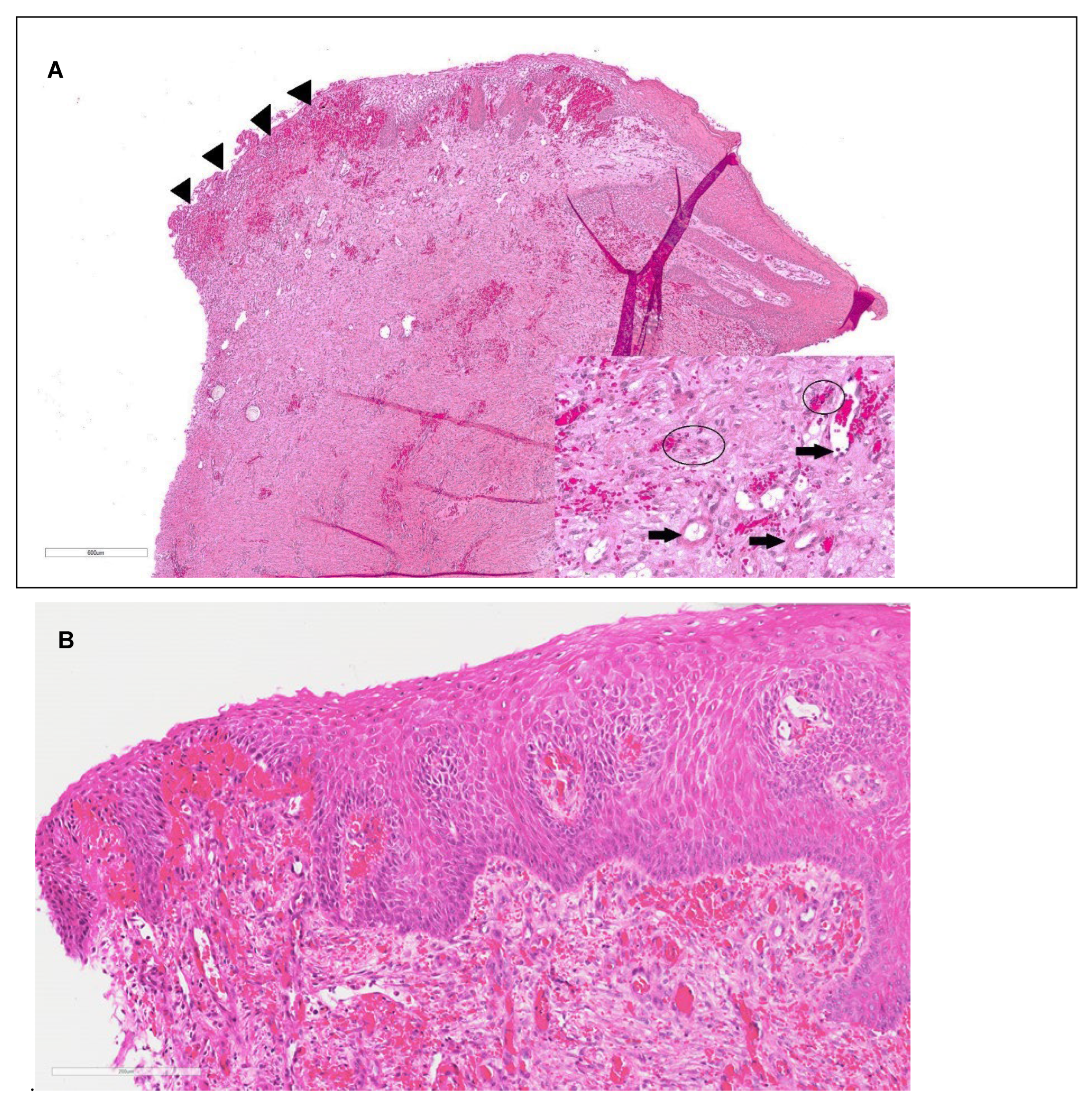
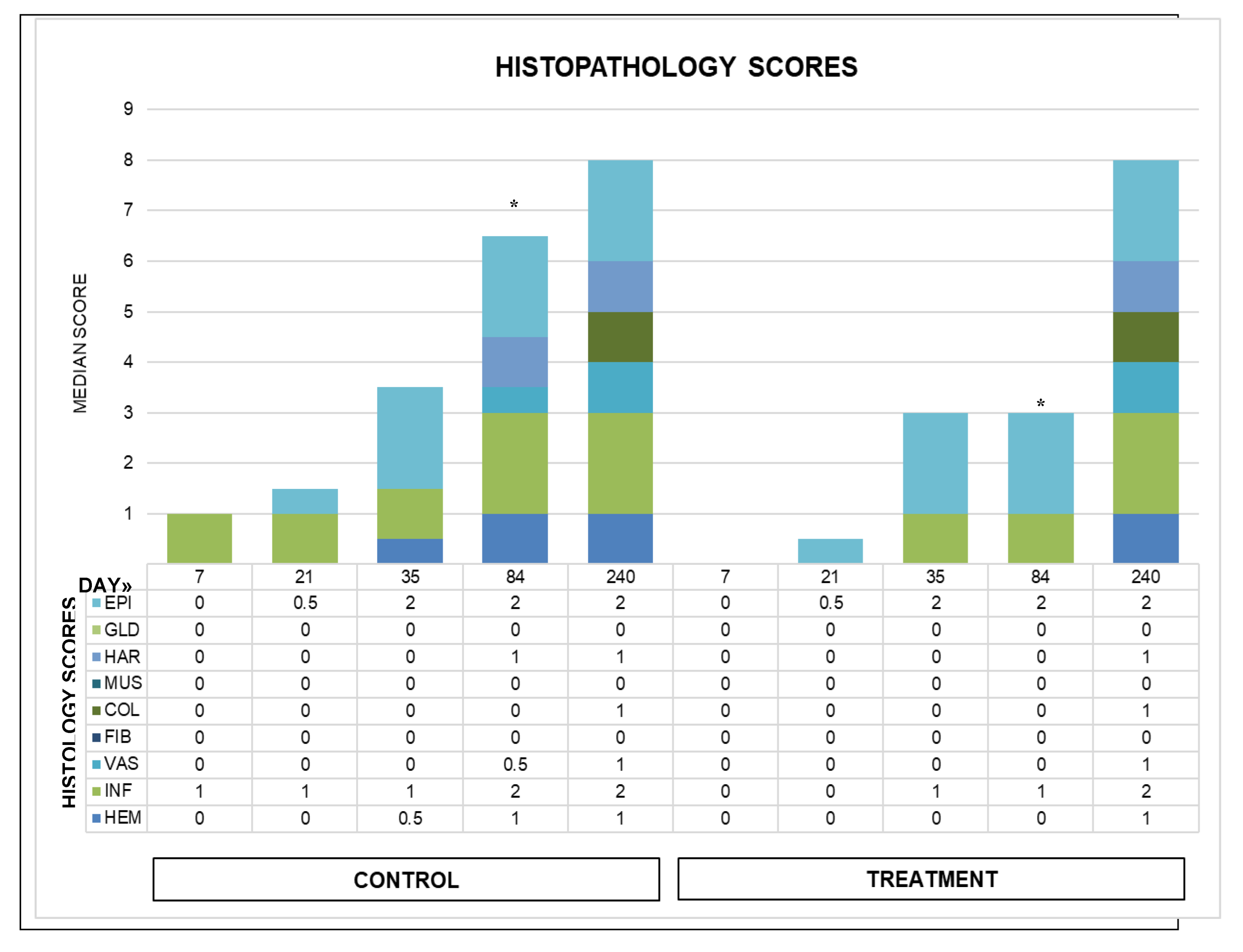
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
IACUC Approval
References
- Bigbie RB, Schumacher J, Swaim SF, et al: Effects of amnion and live yeast cell derivative on second-intention healing in horses. Am J Vet Res 52:1376-1382, 1991. [CrossRef]
- Goodrich LR, Moll HD, Crisman MV, et al: Comparison of equine amnion and a nonadherent wound dressing material for bandaging pinch-grafted wounds in ponies. Am J Vet Res 61:326-329, 2000. [CrossRef]
- Favaron PO, Carvalho RC, Borghesi J, et al: The Amniotic Membrane: Development and Potential Applications - A Review. Reprod Domest Anim 50:881-892, 2015. [CrossRef]
- Kakabadze Z, Mardaleishvili K, Loladze G, et al: Clinical application of decellularized and lyophilized human amnion/chorion membrane grafts for closing post-laryngectomy pharyngocutaneous fistulas. J Surg Oncol 113:538-543, 2016. [CrossRef]
- Murphy SV: 2017 TERMIS - Americas Conference & Exhibition Charlotte, NC December 3-6, 2017, Proceedings, Tissue Eng Part A, Dec, 2017 (available from.
- McQuilling JP, Vines JB, Mowry KC: In vitro assessment of a novel, hypothermically stored amniotic membrane for use in a chronic wound environment. Int Wound J 14:993-1005, 2017. [CrossRef]
- Rodriguez-Ares MT, Lopez-Valladares MJ, Tourino R, et al: Effects of lyophilization on human amniotic membrane. Acta Ophthalmol 87:396-403, 2009. [CrossRef]
- Russo A, Bonci P, Bonci P: The effects of different preservation processes on the total protein and growth factor content in a new biological product developed from human amniotic membrane. Cell Tissue Bank 13:353-361, 2012. [CrossRef]
- Tehrani FA, Ahmadiani A, Niknejad H: The effects of preservation procedures on antibacterial property of amniotic membrane. Cryobiology 67:293-298, 2013. [CrossRef]
- Duddy HR, Schoonover MJ, Williams MR, et al: Healing time of experimentally induced distal limb wounds in horses is not reduced by local injection of equine-origin liquid amnion allograft. Am J Vet Res 83, 2022. [CrossRef]
- Fowler AW, Gilbertie JM, Watson VE, et al: Effects of acellular equine amniotic allografts on the healing of experimentally induced full-thickness distal limb wounds in horses. Vet Surg 48:1416-1428, 2019. [CrossRef]
- Moyer W, Moyer W: Equine joint injection and regional anesthesia (ed 5th). Chadds Ford, PA, Academic Veterinary Solutions, LLC, 2011.
- Kelleher ME, Kilcoyne I, Dechant JE, et al: A preliminary study of silver sodium zirconium phosphate polyurethane foam wound dressing on wounds of the distal aspect of the forelimb in horses. Vet Surg 44:359-365, 2015. [CrossRef]
- Harmon CCG, Hawkins JF, Li J, et al: Effects of topical application of silver sulfadiazine cream, triple antimicrobial ointment, or hyperosmolar nanoemulsion on wound healing, bacterial load, and exuberant granulation tissue formation in bandaged full-thickness equine skin wounds. Am J Vet Res 78:638-646, 2017. [CrossRef]
- Howard RD, Stashak TS, Baxter GM: Evaluation of occlusive dressings for management of full-thickness excisional wounds on the distal portion of the limbs of horses. Am J Vet Res 54:2150-2154, 1993. [CrossRef]
- Berry DB, 2nd, Sullins KE: Effects of topical application of antimicrobials and bandaging on healing and granulation tissue formation in wounds of the distal aspect of the limbs in horses. Am J Vet Res 64:88-92, 2003.
- Badylak SF, Gilbert TW: Immune response to biologic scaffold materials. Semin Immunol 20:109-116, 2008.
- Auer JA, Stick JA: Equine surgery (ed 4th). St. Louis, Mo., Elsevier/Saunders, 2012.
- Jorgensen E, Bay L, Bjarnsholt T, et al: The occurrence of biofilm in an equine experimental wound model of healing by secondary intention. Vet Microbiol 204:90-95, 2017. [CrossRef]
- Wilmink JM, Stolk PW, van Weeren PR, et al: Differences in second-intention wound healing between horses and ponies: macroscopic aspects. Equine Vet J 31:53-60, 1999. [CrossRef]
- Carter CA, Jolly DG, Worden CE, Sr., et al: Platelet-rich plasma gel promotes differentiation and regeneration during equine wound healing. Exp Mol Pathol 74:244-255, 2003. [CrossRef]
- Labens R, Raidal S, Borgen-Nielsen C, et al: Wound healing of experimental equine skin wounds and concurrent microbiota in wound dressings following topical propylene glycol gel treatment. Front Vet Sci 10:1294021, 2023. [CrossRef]
- Charlotte CP, Benoit B, Olivier ML: The effects of a synthetic epidermis spray on secondary intention wound healing in adult horses. PLoS One 19:e0299990, 2024.
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




