Submitted:
19 September 2025
Posted:
22 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Bioresources
2.1. Classification of bioresources
2.1.1. Plant-based
2.1.2. Animal-based
2.1.3. Microbial
2.1.4. Marine
2.1.5. Algal
2.2. Physical properties
2.2.1. Plant-based
2.2.2. Animal-based
2.2.3. Microbial
2.2.4. Marine
2.2.5. Algal
2.3. Chemical properties
2.3.1. Plant-based
2.3.2. Animal-based
2.3.3. Microbial
2.3.4. Marine
2.3.5. Algal
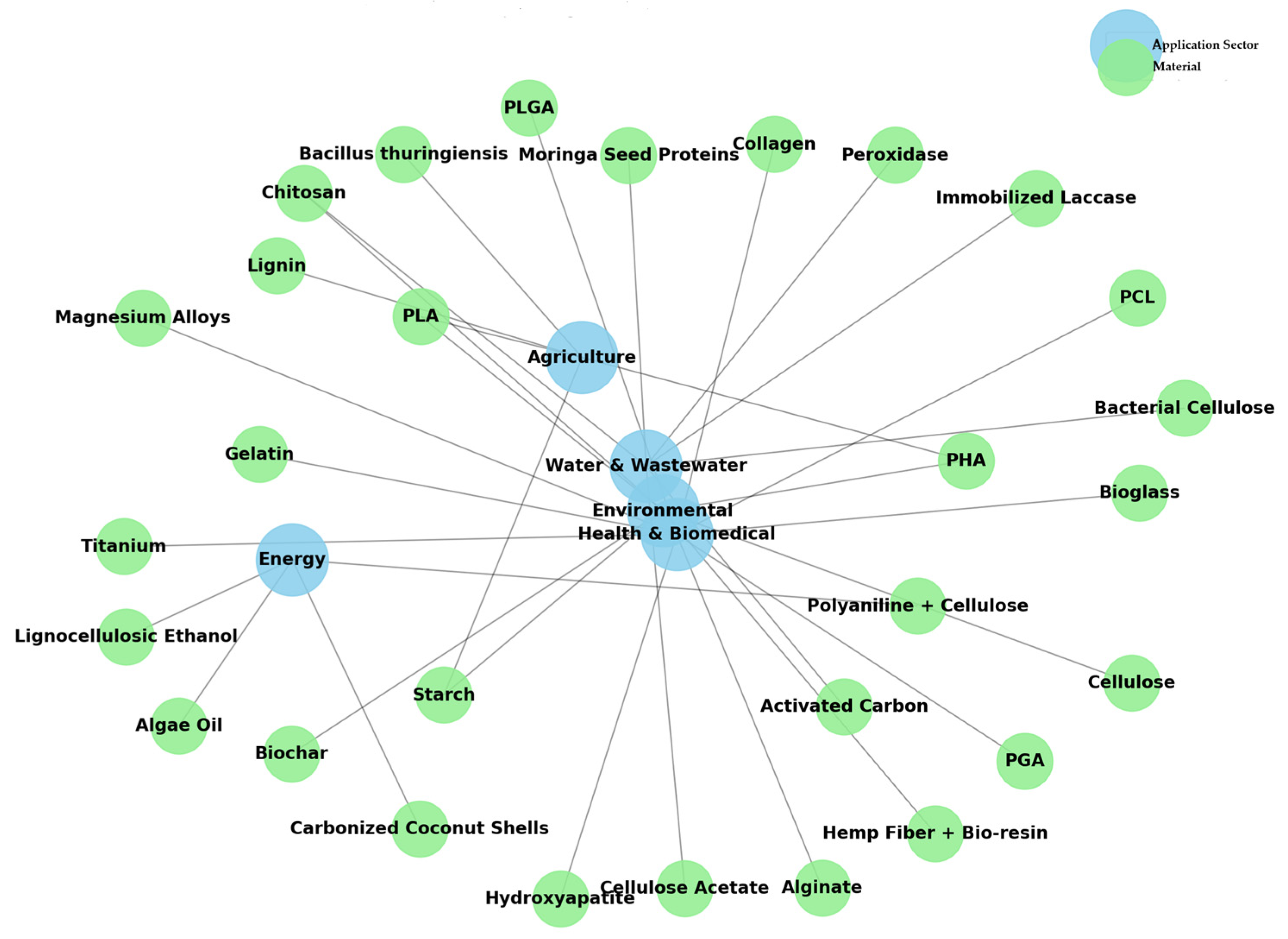
3. Biomaterials
3.1. Biocarbon
3.1.1. Biochar
3.1.2. Bioactivated carbon
3.2. Biomaterials
3.2.1. Catalytic biomaterials
3.2.2. Dental biomaterials
3.2.3. Electroactive biomaterials
3.2.4. Extracellular matrix biomaterials
3.2.5. Injectable biomaterials
3.2.6. Nanocomposite biomaterials
3.2.7. Piezoelectric biomaterials
| Biowaste source | Processing | Piezoelectric material | Potential applications | References |
|---|---|---|---|---|
| Fish scales | Collagen extraction (acid/enzymatic) | Collagen-based films/scaffolds | Tissue engineering, wearable sensors | [109,110] |
| Crustacean shells (shrimp/crab) | Chitin extraction → deacetylation → chitosan | Chitosan films/nanofibers | Wound dressings, flexible piezoelectric sensors | [111] |
| Silk cocoons (sericulture waste) | Degumming → fibroin isolation → electrospinning | Silk fibroin nanofibers/films | Neural scaffolds, biosensors | [103] |
| Keratin from feathers/wool | Reduction/hydrolysis → film casting or electrospinning | Keratin-based piezoelectric films | Flexible sensors, tissue engineering | [102] |
| Eggshell membranes | Demineralization → membrane isolation | Collagen-rich membrane | Bone tissue scaffolds, biosensors | [112] |
| Algae biomass | Polysaccharide extraction → hydrogel formation | Alginate/carrageenan composites | Soft piezoelectric hydrogels for drug delivery | [108] |
3.2.8. Smart biomaterials
3.3. Biometals
3.3.1. Bioiron
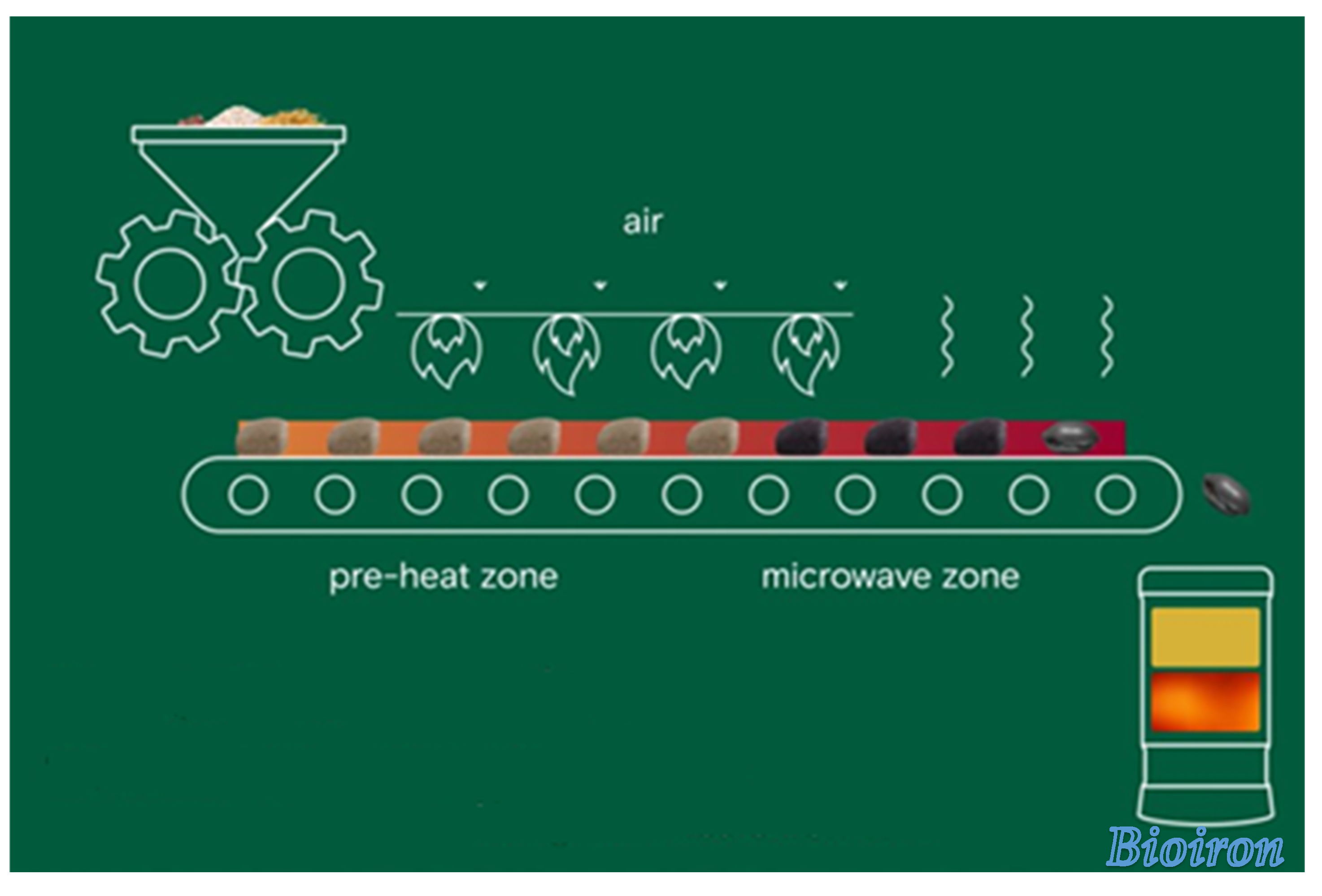
3.3.2. Bio-based green steel
3.3.3. Biotitanium
3.4. Bioplastics
3.4. Building biomaterials
3.5. eTextiles
4. Effect of bioresources properties on biomaterials development and applications
4.1. Effect of plant-based
4.2. Effect of animal-based
4.3. Effect of marine based bioresources properties on biomaterails
4.4. Effect of microbial
4.5. Effect of algal
5. Recent advances in biomaterials production
| Bioproduct | Biowaste source | Key advances | Benefits | Challenges | References |
|---|---|---|---|---|---|
| Polyhydroxyalkanoates (PHAs) and other bioplastics | Food waste, agricultural residues | Fermentation of heterogeneous waste streams (e.g., to lactic acid) feeding Cupriavidus necator; modular bioprocesses; higher yields | Sustainable plastic alternatives, valorisation of organic waste | Feedstock variability, scale-up costs, compostability standards | [142] |
| Nanocellulose and bio-based composites | Crop residues (straw, husks, stalks) | Improved pretreatments and isolation; surface functionalisation; incorporation into composites | Lightweight, high-strength, biodegradable packaging and structural materials | High energy consumption in pretreatment; supply chain logistics | [143] |
| Carbonaceous materials (biochar, activated carbon, nanocarbons) | Agro-wastes, food residues, organic by-products | Low-temperature pyrolysis; hybrid plasma-thermal methods; enhanced structural control | Useful in adsorption, catalysis, energy storage | Energy inputs, reproducibility, economic viability | [144] |
| Chitosan, collagen, hydroxyapatite | Shellfish shells, animal bones, hides | Advanced extraction and purification; composite and coating development | Biomedical, packaging and environmental applications | Regulatory approval, batch consistency, cost of extraction | [145] |
| Single-Cell Protein (SCP) | Food/agro-waste, industrial by-products, syngas from waste | Growth on lignocellulosic hydrolysates; metabolic engineering; “full gas” route (CO, CO2, H2 → protein); integrated biorefineries | Sustainable alternative to soy/fishmeal - High protein yield - Can valorize diverse wastes |
High production cost - Regulatory hurdles for food/feed safety - Scale-up and consumer acceptance |
[146,147] |
| Biofuels & Platform Chemicals | Lignocellulose, food waste, MSW, crude glycerol | Pretreatment & hydrolysis improvements; syngas fermentation; metabolic engineering for organic acids/diols; biomass-derived catalysts | Reduces reliance on fossil fuels - Broad product range (ethanol, succinic acid, etc.) - Integrates into circular bioeconomy |
Energy-intensive pretreatments - Low yields with mixed wastes - High separation/purification costs |
[148,149] |
| Mycelium-based composites and bio-designed materials | Agricultural residues, paper waste, mixed organic waste | Scalable cultivation on waste substrates; moulded panels and packaging | Biodegradable foams, packaging, building materials | Moisture resistance, durability | [150,151] |
6. Future perspectives on sustainability and the circular bioeconomy
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- M. a. P. S. Hemakumara and D. M. K. T. Dissanayake, “The Impact of Industrialization on Environmental Sustainability: A case study in Gampaha district,” NSBM J. Manag., vol. 6, no. 1, Jan. 2020. [CrossRef]
- H. Irfan, “Air pollution and cardiovascular health in South Asia: A comprehensive review,” Curr. Probl. Cardiol., vol. 49, no. 2, p. 102199, Feb. 2024. [CrossRef]
- ”United Nations Framework Convention on Climate Change: Adoption of the Paris Agreement, Paris.” Accessed: Aug. 09, 2025. [Online]. Available: https://unfccc.int/sites/default/files/resource/parisagreement_publication.pdf.
- Department of Biophysics and Biochemistry, Baku State University, Baku, Azerbaijan and R. K. Khalilov, “FUTURE PROSPECTS OF BIOMATERIALS IN NANOMEDICINE,” Adv. Biol. Earth Sci., vol. 9, no. Special Issue, pp. 5–10, Apr. 2024. [CrossRef]
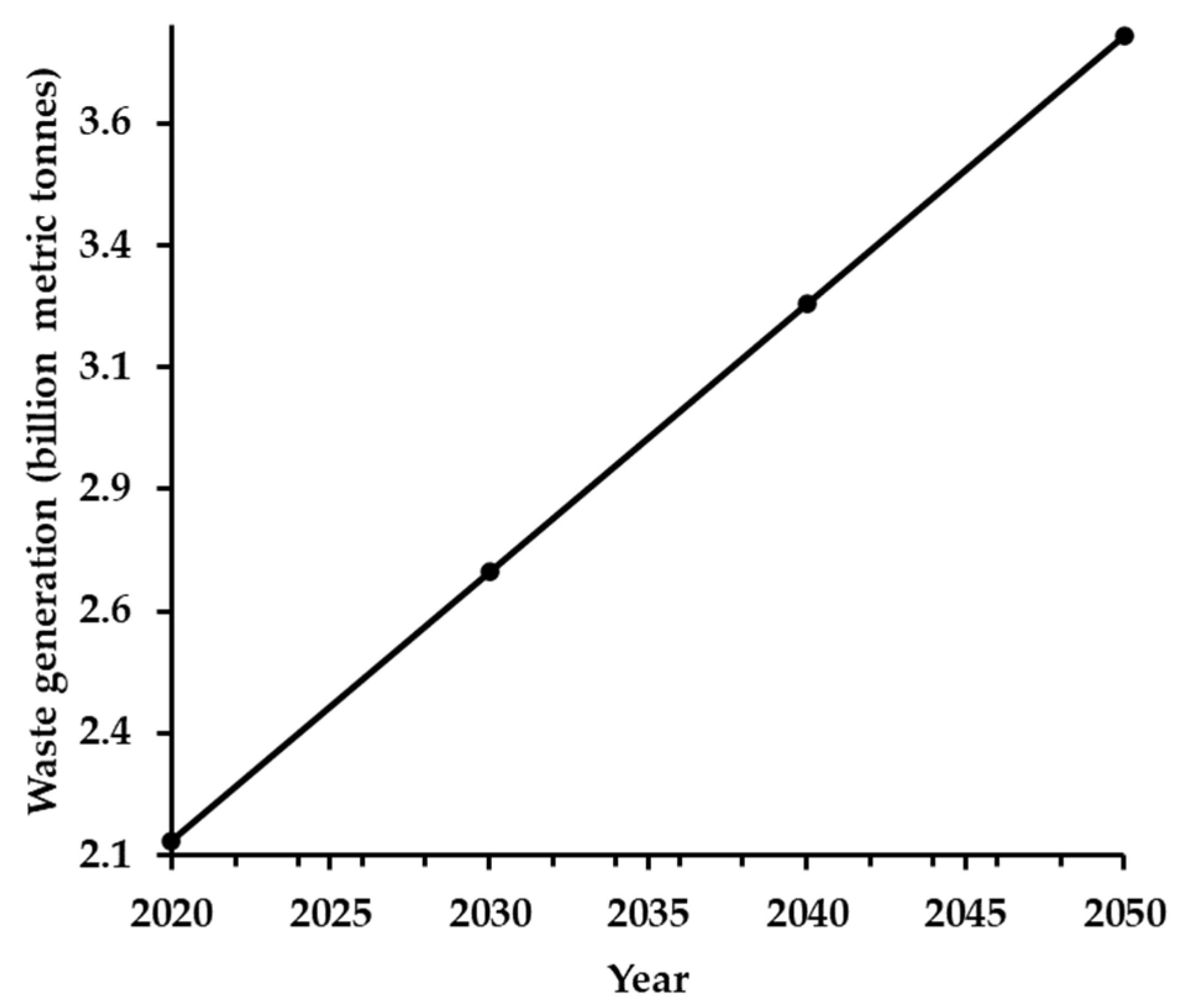
- ”Municipal solid waste generation worldwide in 2020, and projections from 2030 to 2050,” Statista. Accessed: Aug. 08, 2025. [Online]. Available: https://www.statista.com/statistics/916625/global-generation-of-municipal-solid-waste-forecast/.
- S. Dey, M. Santra, M. Choudhury, A. R. Ghosh, and P. Samanta, “Food waste generation and its industrial utilization: An overview,” Environ. Sci. Pollut. Res., vol. 32, no. 12, pp. 7493–7512, Mar. 2025. [CrossRef]
- ”Greenhouse gas emissions worldwide—statistics & facts,” Statista. Accessed: Aug. 08, 2025. [Online]. Available: https://www.statista.com/topics/5770/global-greenhouse-gas-emissions/#topicOverview.
- ”Coal emissions worldwide—statistics & facts,” Statista. Accessed: Aug. 08, 2025. [Online]. Available: https://www.statista.com/topics/10428/coal-emissions-worldwide/.
- L. Chen et al., “Biomaterials technology and policies in the building sector: a review,” Environ. Chem. Lett., vol. 22, no. 2, pp. 715–750, Apr. 2024. [CrossRef]
- H. B. M⊘ller, S. G. Sommer, and B. K. Ahring, “Biological Degradation and Greenhouse Gas Emissions during Pre-Storage of Liquid Animal Manure,” J. Environ. Qual., vol. 33, no. 1, pp. 27–36, 2004. [CrossRef]
- T. Tesfaye, B. Sithole, D. Ramjugernath, and V. Chunilall, “Valorisation of chicken feathers: Characterisation of chemical properties,” Waste Manag., vol. 68, pp. 626–635, Oct. 2017. [CrossRef]
- 12. A. M. King`ori, “A Review of the Uses of Poultry Eggshells and Shell Membranes,” Int. J. Poult. Sci., vol. 10, no. 11, pp. 908–912, Oct. 2011. [CrossRef]
- I.S. Arvanitoyannis and A. Kassaveti, “Fish industry waste: treatments, environmental impacts, current and potential uses,” Int. J. Food Sci. Technol., vol. 43, no. 4, pp. 726–745, Apr. 2008. [CrossRef]
- C. F. Bustillo-Lecompte and M. Mehrvar, “Slaughterhouse wastewater characteristics, treatment, and management in the meat processing industry: A review on trends and advances,” J. Environ. Manage., vol. 161, pp. 287–302, Sept. 2015. [CrossRef]
- M. E. Dimambro, R. D. Lillywhite, and C. R. Rahn, “The Physical, Chemical and Microbial Characteristics of Biodegradable Municipal Waste Derived Composts,” Compost Sci. Util., vol. 15, no. 4, pp. 243–252, Sept. 2007. [CrossRef]
- “Principles of microbe and cell cultivation by S. J. Pirt | Open Library.” Accessed: Sept. 02, 2025. [Online]. Available: https://openlibrary.org/books/OL5063023M/Principles_of_microbe_and_cell_cultivation.
- A. V. S. Silva, G. M. da Costa, and G. Cruz, “Thermal Behavior and Physicochemical Properties of Fish Scales for the Generation of Value-Added Products,” in Fish Waste to Valuable Products, S. Maqsood, M. N. Naseer, S. Benjakul, and A. A. Zaidi, Eds., Singapore: Springer Nature, 2024, pp. 375–411. [CrossRef]
- S. Bentov, S. Abehsera, and A. Sagi, “The Mineralized Exoskeletons of Crustaceans,” in Extracellular Composite Matrices in Arthropods, E. Cohen and B. Moussian, Eds., Cham: Springer International Publishing, 2016, pp. 137–163. [CrossRef]
- E. Delgado, D. J. Valles-Rosales, N. C. Flores, and D. Reyes-Jáquez, “Evaluation of fish oil content and cottonseed meal with ultralow gossypol content on the functional properties of an extruded shrimp feed,” Aquac. Rep., vol. 19, p. 100588, Mar. 2021. [CrossRef]
- A. Demirbas, “Potential applications of renewable energy sources, biomass combustion problems in boiler power systems and combustion related environmental issues,” Prog. Energy Combust. Sci., vol. 31, no. 2, pp. 171–192, Jan. 2005. [CrossRef]
- L. Brennan and P. Owende, “Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products,” Renew. Sustain. Energy Rev., vol. 14, no. 2, pp. 557–577, Feb. 2010. [CrossRef]
- Singh and, S. Gu, “Commercialization potential of microalgae for biofuels production,” Renew. Sustain. Energy Rev., vol. 14, no. 9, pp. 2596–2610, Dec. 2010. [Google Scholar] [CrossRef]
- S. Kaniapan, H. Suhaimi, Y. Hamdan, and J. Pasupuleti, “Experiment analysis on the characteristic of empty fruit bunch, palm kernel shell, coconut shell, and rice husk for biomass boiler fuel,” J. Mech. Eng. Sci., vol. 15, no. 3, pp. 8300–8309, 2021.
- ”Ultimate Analysis—an overview | ScienceDirect Topics.” Accessed: Aug. 20, 2025. [Online]. Available: https://www.sciencedirect.com/topics/engineering/ultimate-analysis?
- W. Spencer, “Advancing Cleaner Technologies for Production of Synthetic Rutile and Activated Carbon,” Murdoch University, Perth, 2025.
- School of Physics, Universiti Sains Malaysia, 11800 USM Pulau Pinang, Malaysia et al., “Corn Cob as a Potential Feedstock for Slow Pyrolysis of Biomass,” J. Phys. Sci., vol. 27, no. 2, pp. 123–137, 2016. [CrossRef]
- K. Sarkar and Q. Wang, “Characterization of Pyrolysis Products and Kinetic Analysis of Waste Jute Stick Biomass,” Processes, vol. 8, no. 7, p. 837, July 2020. [CrossRef]
- N. Daimary, K. S. H. Eldiehy, P. Boruah, D. Deka, U. Bora, and B. K. Kakati, “Potato peels as a sustainable source for biochar, bio-oil and a green heterogeneous catalyst for biodiesel production,” J. Environ. Chem. Eng., vol. 10, no. 1, p. 107108, Feb. 2022. [CrossRef]
- O. G. Banadkoki, S. Sokhansanj, A. Lau, J. S. Lee, S. Arunachalam, and D. Smith, “The Study of Post-Harvest Processing and Handling of Residues From Plants Grown Primarily For Agronomics: Soybean Stalk, Corn Stover, Tomato Vine, Cucumber, Eggplant, and Summer Squash,” Eur. J. Agric. Food Sci., vol. 6, no. 5, pp. 44–53, Oct. 2024. [CrossRef]
- A. G. Adeniyi, K. O. Iwuozor, E. C. Emenike, O. J. Ajala, S. Ogunniyi, and K. B. Muritala, “Thermochemical co-conversion of biomass-plastic waste to biochar: a review,” Green Chem. Eng., vol. 5, no. 1, pp. 31–49, Mar. 2024. [CrossRef]
- W. Spencer, D. Ibana, P. Singh, and A. N. Nikoloski, “Biofuels as renewable reductants for the processing of ilmenite to produce synthetic rutile,” Miner. Eng., vol. 187, p. 107808, Sept. 2022. [CrossRef]
- P. Muthu, G. Muniappan, and R. B. Jeyakumar, “Efficacious Utilization of Food Waste for Bioenergy Generation through the Anaerobic Digestion Method,” Processes, vol. 11, no. 3, p. 702, Feb. 2023. [CrossRef]
- V. Chilla and S. Suranani, “Thermogravimetric and kinetic analysis of orange peel using isoconversional methods,” Mater. Today Proc., vol. 72, pp. 104–109, Jan. 2023. [CrossRef]
- N. Ozbay, A. S. Yargic, R. Z. Yarbay Sahin, and E. Yaman, “Valorization of banana peel waste via in-situ catalytic pyrolysis using Al-Modified SBA-15,” Renew. Energy, vol. 140, pp. 633–646, Sept. 2019. [CrossRef]
- J. M. Costa, L. C. Ampese, H. D. D. Ziero, W. G. Sganzerla, and T. Forster-Carneiro, “Apple pomace biorefinery: Integrated approaches for the production of bioenergy, biochemicals, and value-added products—An updated review,” J. Environ. Chem. Eng., vol. 10, no. 5, p. 108358, Oct. 2022. [CrossRef]
- Alkhader, S. Khan, R. A. Shawabkeh, and I. Janajreh, “Co-gasification of Jordanian olive mill solid waste and RTC coal for thermochemical treatment: characterization and equilibrium modelling,” Biomass Convers. Biorefinery, June 2024. [CrossRef]
- A. F. Ferreira, J. P. Ribau, and M. Costa, “A decision support method for biochars characterization from carbonization of grape pomace,” Biomass Bioenergy, vol. 145, p. 105946, Feb. 2021. [CrossRef]
- N. Voća, N. Bilandžija, V. Jurisic, A. Matin, T. Kricka, and I. Sedak, “Proximate, Ultimate, and Energy Values Analysis of Plum Biomass By-products Case Study: Croatia’s Potential,” J. Agric. Sci., vol. 18: 1655-1666, 2016.
- M. Ashraf, N. Ramzan, R. U. Khan, and A. K. Durrani, “Analysis of mixed cattle manure: Kinetics and thermodynamic comparison of pyrolysis and combustion processes,” Case Stud. Therm. Eng., vol. 26, p. 101078, Aug. 2021. [CrossRef]
- R. Wnetrzak, D. J. M. Hayes, L. S. Jensen, J. J. Leahy, and W. Kwapinski, “Determination of the Higher Heating Value of Pig Manure,” Waste Biomass Valorization, vol. 6, no. 3, pp. 327–333, June 2015. [CrossRef]
- T. Tesfaye, B. Sithole, D. Ramjugernath, and V. Chunilall, “Valorisation of chicken feathers: Characterisation of chemical properties,” Waste Manag., vol. 68, pp. 626–635, Oct. 2017. [CrossRef]
- G. Murugan, K. Ahilan, V. P. A. Prakasam, J. Malreddy, S. Benjakul, and M. Nagarajan, “Fish Waste Composition and Classification,” in Fish Waste to Valuable Products, S. Maqsood, M. N. Naseer, S. Benjakul, and A. A. Zaidi, Eds., in Sustainable Materials and Technology., Singapore: Springer Nature Singapore, 2024, pp. 1–26. [CrossRef]
- A. V. S. Silva, G. M. da Costa, and G. Cruz, “Thermal Behavior and Physicochemical Properties of Fish Scales for the Generation of Value-Added Products,” in Fish Waste to Valuable Products, S. Maqsood, M. N. Naseer, S. Benjakul, and A. A. Zaidi, Eds., Singapore: Springer Nature, 2024, pp. 375–411. [CrossRef]
- A. V. S. Silva, L. D. M. Torquato, and G. Cruz, “Potential application of fish scales as feedstock in thermochemical processes for the clean energy generation,” Waste Manag., vol. 100, pp. 91–100, Dec. 2019. [CrossRef]
- R. H. Rødde, A. Einbu, and K. M. Vårum, “A seasonal study of the chemical composition and chitin quality of shrimp shells obtained from northern shrimp (Pandalus borealis),” Carbohydr. Polym., vol. 71, no. 3, pp. 388–393, Feb. 2008. [CrossRef]
- ”Nutritive value of fish meal. Online J. Anim. Feed Res., 6(1): 14-19.”.
- ”Fish meal, protein 65% | Tables of composition and nutritional values of feed materials INRA CIRAD AFZ.” Accessed: Aug. 28, 2025. [Online]. Available: https://feedtables.com/content/fish-meal-protein-65.
- ”Empirical elemental formula for biomass—Bacteria Escherichia coli—BNID 101800.” Accessed: Aug. 28, 2025. [Online]. Available: https://bionumbers.hms.harvard.edu/bionumber.aspx.
- W. Wu et al., “An approach for carbon content measurement in marine sediment: Application of organic and elemental carbon analyzer,” Mar. Environ. Res., vol. 188, p. 106000, June 2023. [CrossRef]
- ”AquaTech: Fish Farming—Systems & Equipment.” Accessed: Aug. 31, 2025. [Online]. Available: https://www.aqua-tech.eu/.
- S. Jabeen, X. Gao, M. Altarawneh, J. Hayashi, M. Zhang, and B. Z. Dlugogorski, “Analytical Procedure for Proximate Analysis of Algal Biomass: Case Study for Spirulina platensis and Chlorella vulgaris,” Energy Fuels, vol. 34, no. 1, pp. 474–482, Jan. 2020. [CrossRef]
- A. Aboulkas, H. Hammani, M. El Achaby, E. Bilal, A. Barakat, and K. El Harfi, “Valorization of algal waste via pyrolysis in a fixed-bed reactor: Production and characterization of bio-oil and bio-char,” Bioresour. Technol., vol. 243, pp. 400–408, Nov. 2017. [CrossRef]
- W. Han, W. Clarke, and S. Pratt, “Composting of waste algae: A review,” Waste Manag., vol. 34, no. 7, pp. 1148–1155, July 2014. [CrossRef]
- A. Molino, V. Larocca, S. Chianese, and D. Musmarra, “Biofuels Production by Biomass Gasification: A Review,” Energies, vol. 11, no. 4, p. 811, Apr. 2018. [CrossRef]
- Joseph, Stephen, “Biochar for environmental management: science, technology and implementation,” Routledge.
- W. A. Rasaq, M. Golonka, M. Scholz, and A. Białowiec, “Opportunities and Challenges of High-Pressure Fast Pyrolysis of Biomass: A Review,” Energies, vol. 14, no. 17, p. 5426, Jan. 2021. [CrossRef]
- J. A. Libra et al., “Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis,” Biofuels, vol. 2, no. 1, pp. 71–106, Jan. 2011. [CrossRef]
- W.-H. Chen et al., “Progress in biomass torrefaction: Principles, applications and challenges,” Prog. Energy Combust. Sci., vol. 82, p. 100887, Jan. 2021. [CrossRef]
- Y.-F. Huang, P.-T. Chiueh, and S.-L. Lo, “A review on microwave pyrolysis of lignocellulosic biomass,” Sustain. Environ. Res., vol. 26, no. 3, pp. 103–109, May 2016. [CrossRef]
- W. Spencer, G. Senanayake, M. Altarawneh, D. Ibana, and A. N. Nikoloski, “Review of the effects of coal properties and activation parameters on activated carbon production and quality,” Miner. Eng., vol. 212, p. 108712, July 2024. [CrossRef]
- K. S. Ukanwa, K. Patchigolla, R. Sakrabani, E. Anthony, and S. Mandavgane, “A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass,” Sustainability, vol. 11, no. 22, p. 6204, Jan. 2019. [CrossRef]
- W. Spencer, D. Ibana, P. Singh, and A. N. Nikoloski, “Effect of Surface Area, Particle Size and Acid Washing on the Quality of Activated Carbon Derived from Lower Rank Coal by KOH Activation,” Sustainability, vol. 16, no. 14, p. 5876, July 2024. [CrossRef]
- N. A. Ali, N. Khairuddin, T. S. M. Tengku Azmi, and M. B. M. Siddique, “The Preparation of CaO Catalyst from Eggshells and Its Application in Biodiesel Production from Waste Cooking Oil,” Arab. J. Sci. Eng., vol. 48, no. 1, pp. 383–388, Jan. 2023. [CrossRef]
- M. Lu, F. Ge, W. Xu, J. Jiang, and M. Zhou, “N, P-doped coconut shell activated carbon supported Mo2C catalysts for alkaline water electrolysis hydrogen evolution,” J. Alloys Compd., vol. 1036, p. 181783, July 2025. [CrossRef]
- R. Zou et al., “Biochar: From by-products of agro-industrial lignocellulosic waste to tailored carbon-based catalysts for biomass thermochemical conversions,” Chem. Eng. J., vol. 441, p. 135972, Aug. 2022. [CrossRef]
- S. Zahedi, J. Safaei Ghomi, and H. Shahbazi-Alavi, “Preparation of chitosan nanoparticles from shrimp shells and investigation of its catalytic effect in diastereoselective synthesis of dihydropyrroles,” Ultrason. Sonochem., vol. 40, pp. 260–264, Jan. 2018. [CrossRef]
- M. Bilal et al., “Ecofriendly synthesis of hydroxyapatite from fish scales and its application toward adsorptive removal of Pb(II),” J. Indian Chem. Soc., vol. 101, no. 8, p. 101175, Aug. 2024. [CrossRef]
- K. Heyl, B. Garske, and F. Ekardt, “Using bone char as phosphate recycling fertiliser: an analysis of the new EU Fertilising Products Regulation,” Environ. Sci. Eur., vol. 35, no. 1, p. 109, Dec. 2023. [CrossRef]
- Meriatna, H. Husin, M. Riza, M. Faisal, Ahmadi, and Sulastri, “Biodiesel production using waste banana peel as renewable base catalyst,” Mater. Today Proc., vol. 87, pp. 214–217, 2023. [CrossRef]
- S. F. Basumatary et al., “Advances in CaO-based catalysts for sustainable biodiesel synthesis,” Green Energy Resour., vol. 1, no. 3, p. 100032, Sept. 2023. [CrossRef]
- C. Yogin, Soodesh; et al., “Carbonaceous catalysts (biochar and activated carbon) from agricultural residues and their application in production of biodiesel: A review,” Chem. Eng. Res. Des., vol. 203, pp. 759–788, Mar. 2024. [CrossRef]
- M. N. Soltani Rad, S. Behrouz, M. Mohammad-Javadi, and E. Zarenezhad, “Synthesis of fish scale derived hydroxyapatite silica propyl bis aminoethoxy ethane cuprous complex (HASPBAEECC) as a novel hybrid nano-catalyst for highly efficient synthesis of new benzimidazole-1,2,3-triazole hybrid analogues as antifungal agents,” Mol. Divers., vol. 26, no. 5, pp. 2503–2521, Oct. 2022. [CrossRef]
- J. S. Swarup, R. Thomas, J. Rucharitha, V. R. Arunkumar, and V. V, “Eggshell-derived hydroxyapatite as a biomaterial in dentistry: a scoping review of synthesis, properties and applications,” Evid. Based Dent., May 2025. [CrossRef]
- G. Thomas, M. G. Varghese, P. K. John, S. S. Raj, R. S. Jacob, and A. M. Francis, “Application of nanocomposites in dentistry,” in Nanostructured Materials for Biomedical Applications, Elsevier, 2024, pp. 313–337. [CrossRef]
- F. Kurtuldu, H. Kaňková, A. M. Beltrán, L. Liverani, D. Galusek, and A. R. Boccaccini, “Anti-inflammatory and antibacterial activities of cerium-containing mesoporous bioactive glass nanoparticles for drug-free biomedical applications,” Mater. Today Bio, vol. 12, p. 100150, Sept. 2021. [CrossRef]
- A. Baidya, M. Kharaziha, and N. Annabi, “Hydrogels for dental applications,” in Hydrogels for Tissue Engineering and Regenerative Medicine, Elsevier, 2024, pp. 725–748. [CrossRef]
- W. Yuan, L. D. A. Q. Ferreira, B. Yu, S. Ansari, and A. Moshaverinia, “Dental-derived stem cells in tissue engineering: the role of biomaterials and host response,” Regen. Biomater., vol. 11, p. rbad100, Jan. 2024. [CrossRef]
- S. K. Ghosh and D. Mandal, “High-performance bio-piezoelectric nanogenerator made with fish scale,” Appl. Phys. Lett., vol. 109, no. 10, p. 103701, Sept. 2016. [CrossRef]
- S. Divya, S. Ramasundaram, K. Aruchamy, T. H. Oh, T. Levingstone, and N. Dunne, “Piezoelectric nanogenerators from sustainable biowaste source: Power harvesting and respiratory monitoring with electrospun crab shell powder-poly(vinylidene fluoride) composite nanofibers,” J. Colloid Interface Sci., vol. 679, pp. 324–334, Feb. 2025. [CrossRef]
- P. Manasa, S. Sambasivam, and F. Ran, “Recent progress on biomass waste derived activated carbon electrode materials for supercapacitors applications—A review,” J. Energy Storage, vol. 54, p. 105290, Oct. 2022. [CrossRef]
- M. A. Bhakare, P. H. Wadekar, R. V. Khose, M. P. Bondarde, and S. Some, “Eco-friendly biowaste-derived graphitic carbon as black pigment for conductive paint,” Prog. Org. Coat., vol. 147, p. 105872, Oct. 2020. [CrossRef]
- B. Mishra et al., “Valorization of agro-industrial biowaste to biomaterials: An innovative circular bioeconomy approach,” Circ. Econ., vol. 2, no. 3, p. 100050, Sept. 2023. [CrossRef]
- S. Chand et al., “Sustainable synthesis and multifunctional applications of biowaste-derived carbon nanomaterials and metal oxide composites: A review,” Chemosphere, vol. 385, p. 144540, Sept. 2025. [CrossRef]
- H. Luze, S. P. Nischwitz, C. Smolle, R. Zrim, and L.-P. Kamolz, “The Use of Acellular Fish Skin Grafts in Burn Wound Management—A Systematic Review,” Medicina (Mex.), vol. 58, no. 7, p. 912, July 2022. [CrossRef]
- L. Chen, G. Cheng, S. Meng, and Y. Ding, “Collagen Membrane Derived from Fish Scales for Application in Bone Tissue Engineering,” Polymers, vol. 14, no. 13, p. 2532, June 2022. [CrossRef]
- A. Torres-Mansilla et al., “Eggshell Membrane as a Biomaterial for Bone Regeneration,” Polymers, vol. 15, no. 6, p. 1342, Mar. 2023. [CrossRef]
- S. Ahmed and S. Ikram, “Chitosan Based Scaffolds and Their Applications in Wound Healing,” Achiev. Life Sci., vol. 10, no. 1, pp. 27–37, June 2016. [CrossRef]
- C. Ruiz-Cañada, Á. Bernabé-García, S. Liarte, M. Rodríguez-Valiente, and F. J. Nicolás, “Chronic Wound Healing by Amniotic Membrane: TGF-β and EGF Signaling Modulation in Re-epithelialization,” Front. Bioeng. Biotechnol., vol. 9, p. 689328, July 2021. [CrossRef]
- I. Belviso et al., “Decellularized Human Dermal Matrix as a Biological Scaffold for Cardiac Repair and Regeneration,” Front. Bioeng. Biotechnol., vol. 8, p. 229, Mar. 2020. [CrossRef]
- J. R. Gershlak et al., “Crossing kingdoms: Using decellularized plants as perfusable tissue engineering scaffolds,” Biomaterials, vol. 125, pp. 13–22, May 2017. [CrossRef]
- S. Ahmed and S. Ikram, “Chitosan Based Scaffolds and Their Applications in Wound Healing,” Achiev. Life Sci., vol. 10, no. 1, pp. 27–37, June 2016. [CrossRef]
- C. I. Codrea, D. Baykara, R.-A. Mitran, A. C. Ç. Koyuncu, O. Gunduz, and A. Ficai, “3D-Bioprinted Gelatin Methacryloyl-Strontium-Doped Hydroxyapatite Composite Hydrogels Scaffolds for Bone Tissue Regeneration,” Polymers, vol. 16, no. 13, p. 1932, July 2024. [CrossRef]
- V. Kattimani, K. P. Lingamaneni, P. S. Chakravarthi, T. S. S. Kumar, and A. Siddharthan, “Eggshell-Derived Hydroxyapatite: A New Era in Bone Regeneration,” J. Craniofac. Surg., vol. 27, no. 1, pp. 112–117, Jan. 2016. [CrossRef]
- V. Trakoolwannachai, P. Kheolamai, and S. Ummartyotin, “Characterization of hydroxyapatite from eggshell waste and polycaprolactone (PCL) composite for scaffold material,” Compos. Part B Eng., vol. 173, p. 106974, Sept. 2019. [CrossRef]
- B. Mondal, S. Mondal, A. Mondal, and N. Mandal, “Fish scale derived hydroxyapatite scaffold for bone tissue engineering,” Mater. Charact., vol. 121, pp. 112–124, Nov. 2016. [CrossRef]
- S. Vaezifar, M. Molaei, F. Dibaei, and P. Heydari, “Antibacterial Behavior of Chitosan Nanoparticles Prepared from Shrimp Shells Against Bacteria Causing Skin Infections: Usability in Skin Tissue Engineering,” BioNanoScience, vol. 15, no. 3, p. 511, Sept. 2025. [CrossRef]
- Y. Shen, “Rice husk silica derived nanomaterials for sustainable applications,” Renew. Sustain. Energy Rev., vol. 80, pp. 453–466, Dec. 2017. [CrossRef]
- Y. Yang, H. Liu, M. Wu, J. Ma, and P. Lu, “Bio-based antimicrobial packaging from sugarcane bagasse nanocellulose/nisin hybrid films,” Int. J. Biol. Macromol., vol. 161, pp. 627–635, Oct. 2020. [CrossRef]
- V. Kumar et al., “Potential of banana based cellulose materials for advanced applications: A review on properties and technical challenges,” Carbohydr. Polym. Technol. Appl., vol. 6, p. 100366, Dec. 2023. [CrossRef]
- Y. Leow et al., “Coconut husk-derived nanocellulose as reinforcing additives in thermal-responsive hydrogels,” Carbohydr. Polym., vol. 323, p. 121453, Jan. 2024. [CrossRef]
- Z. Xiong et al., “Biocompatible and stretchable chitosan piezoelectric gel with antibacterial capability and motion monitoring function for Achilles tendon rupture treatment,” Carbohydr. Polym., vol. 352, p. 123149, Mar. 2025. [CrossRef]
- R. Wang and H. Tong, “Preparation Methods and Functional Characteristics of Regenerated Keratin-Based Biofilms,” Polymers, vol. 14, no. 21, p. 4723, Nov. 2022. [CrossRef]
- R. Dutta, S. Chowdhury, K. Kar, and K. Mazumder, “Silk Fibroin–Based Biomaterial Scaffold in Tissue Engineering: Present Persuasive Perspective,” Regen. Eng. Transl. Med., Dec. 2024. [CrossRef]
- S. K. Ghosh and D. Mandal, “High-performance bio-piezoelectric nanogenerator made with fish scale,” Appl. Phys. Lett., vol. 109, no. 10, p. 103701, Sept. 2016. [CrossRef]
- L. Jin, Y. Tai, and J. Nam, “Piezoelectric silk fibroin nanofibers: Structural optimization to enhance piezoelectricity and biostability for neural tissue engineering,” Nano Energy, vol. 132, p. 110367, Dec. 2024. [CrossRef]
- X. Yue et al., “Silk fibroin-based piezoelectric nanofibrous scaffolds for rapid wound healing,” Biomater. Sci., vol. 11, no. 15, pp. 5232–5239, 2023. [CrossRef]
- X. Liang et al., “Robust Piezoelectric Biomolecular Membranes from Eggshell Protein for Wearable Sensors,” ACS Appl. Mater. Interfaces, vol. 15, no. 48, pp. 55790–55802, Dec. 2023. [CrossRef]
- R. Chen et al., “Sodium alginate based piezoelectric hydrogel for promoting healing of infected wounds at movable parts,” Int. J. Biol. Macromol., vol. 285, p. 138287, Jan. 2025. [CrossRef]
- C. Ribeiro, V. Sencadas, D. M. Correia, and S. Lanceros-Méndez, “Piezoelectric polymers as biomaterials for tissue engineering applications,” Colloids Surf. B Biointerfaces, vol. 136, pp. 46–55, Dec. 2015. [CrossRef]
- M. Andonegi et al., “Piezoelectric properties of collagen films: Insights into their potential for electroactive biomedical applications,” Int. J. Biol. Macromol., vol. 309, p. 142799, May 2025. [CrossRef]
- G. De Marzo et al., “Chitosan-Based Piezoelectric Flexible and Wearable Patch for Sensing Physiological Strain,” in The 8th International Symposium on Sensor Science, MDPI, May 2021, p. 12. [CrossRef]
- X. Liang, H. Cong, G. Jiang, and H. He, “Enhancing Physicochemical and Piezoelectric Properties of Eggshell Membrane Proteins by Ultrasonic-Assisted Enzymes for Food and Sensor Applications,” Int. J. Mol. Sci., vol. 26, no. 5, p. 2190, Feb. 2025. [CrossRef]
- B. Mishra et al., “Valorization of agro-industrial biowaste to biomaterials: An innovative circular bioeconomy approach,” Circ. Econ., vol. 2, no. 3, p. 100050, Sept. 2023. [CrossRef]
- B. Tian and J. Liu, “Smart stimuli-responsive chitosan hydrogel for drug delivery: A review,” Int. J. Biol. Macromol., vol. 235, p. 123902, Apr. 2023. [CrossRef]
- F. Hong et al., “Chitosan-based hydrogels: From preparation to applications, a review,” Food Chem. X, vol. 21, p. 101095, Mar. 2024. [CrossRef]
- S. Gavrilaș, M. Raț, and F.-D. Munteanu, “Biowaste Valorisation and Its Possible Perspectives Within Sustainable Food Chain Development,” Processes, vol. 13, no. 7, p. 2085, July 2025. [CrossRef]
- P. R. Yaashikaa, P. Senthil Kumar, and S. Varjani, “Valorization of agro-industrial wastes for biorefinery process and circular bioeconomy: A critical review,” Bioresour. Technol., vol. 343, p. 126126, Jan. 2022. [CrossRef]
- W. Spencer, D. Ibana, P. Singh, and A. N. Nikoloski, “Biofuels as renewable reductants for the processing of ilmenite to produce synthetic rutile,” Miner. Eng., vol. 187, p. 107808, Sept. 2022. [CrossRef]
- ”Rio Tinto says a new way of processing iron ore in steelmaking could change the world. Do its claims stack up?” Accessed: Sept. 09, 2025. [Online]. Available: https://www.abc.net.au/news/2024-06-05/rio-tinto-bioiron-explainer-net-zero-carbon-emissions/103935180.
- W. Spencer, D. Ibana, P. Singh, and A. N. Nikoloski, “Sustainable Production of Activated Carbon from Waste Wood Using Goethite Iron Ore,” Sustainability, vol. 17, no. 2, p. 681, Jan. 2025. [CrossRef]
- D. Gleeson, “Rio Tinto ups the BioIron ante with new pilot plant in Western Australia,” International Mining. Accessed: Sept. 10, 2025. [Online]. Available: https://im-mining.com/2024/06/04/rio-tinto-ups-the-bioiron-ante-with-new-pilot-plant-in-western-australia/.
- ”Australia forges a future made from green steel.” [Online]. Available: https://international.austrade.gov.au/en/news-and-analysis/news/australia-forges-a-future-made-from-green-steel.
- F. Rosner et al., “Green steel: design and cost analysis of hydrogen-based direct iron reduction,” Energy Environ. Sci., vol. 16, no. 10, pp. 4121–4134, 2023. [CrossRef]
- W. Spencer, D. Ibana, P. Singh, and A. N. Nikoloski, “Effect of ilmenite properties on synthetic rutile quality,” Miner. Eng., vol. 177, p. 107365, Feb. 2022. [CrossRef]
- ”In 2024, The Global Plastic Production Will Exceed 400 Million Tons—MARKET NEWS—News.” Accessed: Aug. 17, 2025. [Online]. Available: https://www.kitechrecycle.com/news/in-2024-the-global-plastic-production-will-ex-84661069.html.
- ”New market data: The positive trend for the bioplastics industry remains stable,” Renewable Carbon News. Accessed: Aug. 17, 2025. [Online]. Available: https://renewable-carbon.eu/news/new-market-data-the-positive-trend-for-the-bioplastics-industry-remains-stable/.
- ”From sugarcane plantation to supermarket shelves: sustainability throughout the production chain and in the daily life of people.” Accessed: Sept. 04, 2025. [Online]. Available: https://www.braskem.com/usa/news-detail/from-sugarcane-plantation-to-supermarket-shelves-sustainability-throughout-the-production-chain-and-in-the-daily-life-of-people.
- E. T. H. Vink, D. A. Glassner, J. J. Kolstad, R. J. Wooley, and R. P. O’Connor, “The eco-profiles for current and near-future NatureWorks® polylactide (PLA) production,” https://home.liebertpub.com/ind. Accessed: Sept. 04, 2025. [Online]. Available: https://www.liebertpub.com/doi/10.1089/ind.2007.3.058.
- ”Mater-Bi—biodegradable and compostable bioplastics—Novamont.” Accessed: Sept. 04, 2025. [Online]. Available: https://www.novamont.com/eng/mater-bi.
- ”Corbion-PLA.pdf.” Accessed: Sept. 05, 2025. [Online]. Available: https://www.europabio.org/wp-content/uploads/2024/09/Corbion-PLA.pdf.
- ”Biopolymers.” Accessed: Sept. 05, 2025. [Online]. Available: https://plastics-rubber.basf.com/global/en/performance_polymers/fpgs/fpg_biodegradable_plastics.
- ”Product ranges,” Biome Bioplastics. Accessed: Sept. 05, 2025. [Online]. Available: https://biomebioplastics.com/product-ranges/.
- ”PHA: Beginning of Life—Danimer Scientific.” Accessed: Sept. 05, 2025. [Online]. Available: https://danimerscientific.com/pha-beginning-of-life/.
- ”[TORAY],” TORAY. Accessed: Sept. 05, 2025. [Online]. Available: https://www.toray.com.
- ”Mitsubishi Chemical.” Accessed: Sept. 05, 2025. [Online]. Available: https://www.m-chemical.co.jp/en/products/bioplastics/.
- ”Home | Plantic Technologies | Changing the nature of plastics | Bioplastics,” Plantic. Accessed: Sept. 05, 2025. [Online]. Available: https://plantic.com.au/.
- H. Y. Jaramillo, O. Vasco-Echeverri, R. López-Barrios, and R. A. García-León, “Optimization of Bio-Brick Composition Using Agricultural Waste: Mechanical Properties and Sustainable Applications,” Sustainability, vol. 17, no. 5, Art. no. 5, Jan. 2025. [CrossRef]
- S. Guridi, M. Iannacchero, and E. Pouta, “Towards More Sustainable Interactive Textiles: A Literature Review on The Use of Biomaterials for eTextiles.,” in Proceedings of the CHI Conference on Human Factors in Computing Systems, Honolulu HI USA: ACM, May 2024, pp. 1–19. [CrossRef]
- S. Li, “High-Performance Bio-Based Fibers for Sustainable Textile Applications,” J. Text. Sci. Fash. Technol., vol. 11, no. 3, May 2025. [CrossRef]
- Yang, Z. Zhang, J. Yuan, J. Xu, Q. Ji, and Y. Bai, “Eco-friendly production of leather-like material from bacterial cellulose and waste resources,” J. Clean. Prod., vol. 476, p. 143700, Oct. 2024. [CrossRef]
- C. Var and S. Palamutcu, “Man-Made Bio-based and Biodegradable Fibers for Textile Applications,” in Sustainable Manufacturing Practices in the Textiles and Fashion Sector, S. S. Muthu, Ed., in Sustainable Textiles: Production, Processing, Manufacturing & Chemistry., Cham: Springer Nature Switzerland, 2024, pp. 229–280. [CrossRef]
- T. Liu, K. Ye, and S. Jin, “Global transcriptomics reveals carbon footprint of food waste in the bioconversion of ecofriendly polymers,” Bioresour. Technol., vol. 433, p. 132719, Oct. 2025. [CrossRef]
- H. Ren et al., “Emerging nanocellulose from agricultural waste: Recent advances in preparation and applications in biobased food packaging,” Int. J. Biol. Macromol., vol. 277, p. 134512, Oct. 2024. [CrossRef]
- F. Li, G. Li, B. G. Lougou, Q. Zhou, B. Jiang, and Y. Shuai, “Upcycling biowaste into advanced carbon materials via low-temperature plasma hybrid system: applications, mechanisms, strategies and future prospects,” Waste Manag., vol. 189, pp. 364–388, Dec. 2024. [CrossRef]
- B. Mishra et al., “Valorization of agro-industrial biowaste to biomaterials: An innovative circular bioeconomy approach,” Circ. Econ., vol. 2, no. 3, p. 100050, Sept. 2023. [CrossRef]
- P. T. Sekoai, Y. Roets-Dlamini, F. O’Brien, S. Ramchuran, and V. Chunilall, “Valorization of Food Waste into Single-Cell Protein: An Innovative Technological Strategy for Sustainable Protein Production,” Microorganisms, vol. 12, no. 1, p. 166, Jan. 2024. [CrossRef]
- S. Matassa et al., “Upcycling of biowaste carbon and nutrients in line with consumer confidence: the ‘full gas’ route to single cell protein,” Green Chem., vol. 22, no. 15, pp. 4912–4929, 2020. [CrossRef]
- J Y. Kim, Y.-J. Ahn, J. A. Lee, and S. Y. Lee, “Recent advances in the production of platform chemicals using metabolically engineered microorganisms,” Curr. Opin. Green Sustain. Chem., vol. 40, p. 100777, Apr. 2023. [CrossRef]
- J. Lee, W.-H. Chen, and Y.-K. Park, “Recent achievements in platform chemical production from food waste,” Bioresour. Technol., vol. 366, p. 128204, Dec. 2022. [CrossRef]
- C. Madusanka et al., “A review of recent advances in fungal mycelium based composites,” Discov. Mater., vol. 4, no. 1, p. 13, May 2024. [CrossRef]
- D.-G. Barta, I. Simion, A.-E. Tiuc, and O. Vasile, “Mycelium-Based Composites as a Sustainable Solution for Waste Management and Circular Economy,” Materials, vol. 17, no. 2, p. 404, Jan. 2024. [CrossRef]
| Type of waste | Fruits |
|---|---|
| Peel, cores, spoiled/blemished fruit | Apple |
| Peelings, rejected or overripe fruit | Banana |
| Peel (including pith), seeds, spoiled fruit | Orange |
| Peel, seed kernel, spoiled fruit | Mango |
| Peel, core, leaves | Pineapple |
| Peel, seeds, spoiled fruit | Papaya, watermelon, melon (cantaloupe, honeydew), pomegranate, lemon, lime, grapefruit, kiwi, guava, lychee, persimmon |
| Peels, pits, spoiled fruit | Peach, plum, apricot |
| Peels, spoiled fruit | Starfruit |
| pits, spoiled fruit | Date |
| pods, seeds, husks | Tamarind |
| stems, skins, spoiled/raisined fruit | Grapes |
| stems, spoiled berries | Blueberry, raspberry, blackberry, cranberry, mulberry, elderberry, fig |
| stems, pits, damaged or unmarketable fruit | Cherry |
| husk, shell, coconut water, spoiled fruit | Coconut |
| leaves, stems, spoiled berries | Strawberry |
| Peel, seed, spoiled fruit | Avocado |
| Peel, core, seeds, fibrous strands | Jackfruit |
| Peel, spoiled fruit | Dragon fruit |
| Type of waste generated | Vegetables |
|---|---|
| Peelings | Potato, carrot, tomato, cucumber, bell pepper, zucchini, eggplant, pumpkin, squash, radish, beetroot, sweet potato, turnip, parsnip, kohlrabi, taro, cassava |
| Trimmings/tops | Potato, carrot, tomato, cucumber, radish, parsnip, brussels sprouts |
| Damaged tubers/roots | Potato, sweet potato, beetroot, turnip, parsnip |
| Spoiled/overripe fruit | Tomato, cucumber, bell pepper, zucchini, eggplant, okra |
| Outer skins/outer leaves | Onion, garlic, lettuce, cabbage, leek, brussels sprouts, artichoke, shallots, bamboo shoots |
| Seeds/stems | Bell pepper, pumpkin, squash, green beans, peas |
| Stems/stalks | Spinach, kale, cauliflower, broccoli, celery, fennel, chard, watercress, arugula, artichoke |
| Wilted or yellowed leaves | Lettuce, spinach, kale, chard, arugula, watercress |
| Pods | Green beans, peas, okra |
| Husk/cob | Corn (maize), bamboo shoots |
| Roots/sprouted parts | Onion, garlic, shallots |
| Category | Cereals | Legumes |
| Field residues | Straw, stalks, husks, leaves | Stalks, pods, leaves |
| Post-harvest waste | Bran, broken grains, milling dust, husks | Seed coats/hulls, broken seeds, pods |
| Rejected/spoiled produce | Moldy grains, insect-damaged seeds | Spoiled pods/seeds |
| Category | Type of waste |
| Logging residues | Branches, tops, bark, leaves, twigs left after tree felling |
| Sawmill and processing waste | Sawdust, wood chips, bark, slabs, shavings |
| Pulp and paper industry waste | Bark, black liquor, sludge, paper trim, recycled fiber rejects |
| Rejected/spoiled wood | Rotten wood, insect-damaged timber, off-spec lumber |
| Category | Bamboo waste | Hemp waste | Oilseed species waste |
| Field Residues | Bamboo leaves, branches, culm tops | Stalks, leaves, and seeds left after harvest | Seed pods, stalks, leaves |
| Processing Waste | Bamboo dust, shaved strips | Hemp hurds (woody core), fibre waste | Seed cake (after oil extraction), seed hulls |
| Rejected/Spoiled Produce | Broken culms, insect-damaged parts | Moldy or insect-damaged hemp stalks | Spoiled seeds or pods |
| Type of waste | Description |
| Manure | Cow dung, pig manure, poultry litter, sheep manure |
| Manure slurry/liquid manure | More liquid form of manure, often collected in farms |
| Poultry litter | Mixture of poultry manure, feathers, and bedding material |
| Feathers | Poultry feathers collected during processing |
| Bones | Animal bones from slaughter waste |
| Gelatin & collagen waste | Derived from bones, skins, and connective tissue during meat processing |
| Offal | Internal organs and other slaughterhouse waste |
| Hair | Animal hair from hides and slaughter processes |
| Fish waste | Fish scales, guts, bones discarded during fish processing |
| Crustacean shells | Shells from shrimp, crab, lobster |
| Milk processing waste | Whey and sludge from dairy processing |
| Wool waste | Wool trimmings and dust from shearing |
| Eggshells | Waste from poultry farms or egg processing |
| Tallow & animal fats | Rendered fats from slaughterhouses |
| Slaughterhouse wastewater | Organic-rich effluent from meat processing plants |
| Animal fat trimmings | Fat removed from meat cuts |
| Category | Type of solid waste |
| Microbial biomass residue | Leftover cell pellets after fermentation or microbial growth processes (e.g., yeast, bacteria biomass) |
| Spent culture media | Solid residues or dehydrated microbial growth media components after use |
| Used petri dishes and plates | Plastic or glass plates contaminated with microbial cultures |
| Contaminated disposable labware | Pipette tips, swabs, inoculating loops, gloves, masks contaminated with microbes |
| Filter membranes and papers | Used filtration materials capturing microbial cells or spores |
| Solid fermentation residues | Insoluble residues from solid-state fermentation processes |
| Packaging waste | Bags, containers, and wrappers of microbial media and reagents |
| Category | Type of solid waste |
| Fish processing waste | Fish bones, scales, skin, fins, offal, and carcasses |
| Shellfish and crustacean waste | Shells, exoskeletons, and carapaces from shrimp, crab, lobster, mollusca |
| Fishmeal and fish oil residues | Solid residues from fishmeal production plants |
| Marine microbial biomass | Residues of microbial biomass cultured from marine environments |
| Marine sediment waste | Organic-rich sediments and detritus collected during marine resource processing |
| Packaging and plastic waste | Plastic containers, nets, ropes, and other materials used in marine bioresource handling |
| Category | Type of solid waste |
| Algal biomass residues | Leftover algal biomass after extraction of bioactive compounds, oils, or pigments |
| Harvesting waste | Broken or damaged algal fronds and filaments discarded during harvesting |
| De-watered algal sludge | Concentrated algal paste or sludge after dewatering processes |
| Spent growth media solids | Residual solids or precipitates from algal cultivation media |
| Algal cell walls and debris | Insoluble cell wall materials remaining after extraction or processing |
| Packaging waste | Bags, containers, and materials used to store or transport algal biomass |
| Agricultural residues | Carbon (%) | Hydrogen (%) | Nitrogen (%) | Oxygen (%) | Reference |
|---|---|---|---|---|---|
| Rice husk | 42.5 | 5.4 | 0.4 | 34.6 | [23] |
| Sugarcane bagasse | 44.8 | 5.4 | 0.4 | 39.6 | [24] |
| Gumtree sticks | 46.4 | 6.6 | - | 46.0 | [25] |
| Corncob | 43.8 | 6.5 | 0.8 | 48.2 | [26] |
| Jute stick | 43.4 | 5.8 | 7.8 | 43.0 | [27] |
| Potato peelings | 43.8 | 5.8 | 3.5 | 46.8 | [28] |
| Cucumber | 35.7 | 4.7 | 3.3 | 55.8 | [29] |
| Eggplant | 41.6 | 5.3 | 2.9 | 49.8 | [29] |
| Corn stover | 45.8 | 5.7 | 0.5 | 48.1 | [29] |
| Soybean stalk | 46.0 | 5.8 | 0.5 | 47.8 | [29] |
| Tomato vine | 41.3 | 5.2 | 1.2 | 51.9 | [29] |
| Sawdust | 50.4 | 6.1 | 0.0 | 43.5 | [29] |
| Wood | 45.7 | 7.57 | 1.89 | 1.01 | [30] |
| Wood kindling | 57.8 | 5.3 | 1.1 | 34.6 | [31] |
| Pine bark | 48.5 | 5.9 | 0.17 | 0.03 | [30] |
| Pine wood | 47.9 | 5.5 | 0.6 | 0.1 | [30] |
| Alder wood | 49.5 | 6.43 | 0.07 | 0.06 | [30] |
| Rice bran wax | 76.8 | 15.1 | 0.02 | 0.68 | [30] |
| Hazelnut | 51.5 | 5.2 | 0.2 | - | [30] |
| Corn stalks | 43.3 | 6.12 | 2.12 | - | [30] |
| Wheat straw | 43.7 | 6.11 | 0.52 | 0.1 | [30] |
| Pinecone | 42.6 | 5.56 | 0.76 | 0.05 | [30] |
| Okra | 39.3 | 5.4 | 3.2 | 35.7 | [32] |
| Food waste | 41.8 | 5.06 | 2.01 | [32] |
| Fruit waste | Carbon (%) | Hydrogen (%) | Nitrogen (%) | Oxygen (%) | Reference |
|---|---|---|---|---|---|
| Orange peel | 45.1 | 8.8 | 0.5 | 42.3 | [33] |
| Banana peel | 40.0 | 7.1 | 0.7 | 52.3 | [34] |
| Apple pomace | 46.8 | 6.4 | 0.6 | 43.6 | [35] |
| Olive mill solid waste | 47.5 | 6.7 | 0.7 | 40.1 | [36] |
| Grape pomace | 51.1 | 6.7 | 1.9 | 40.1 | [37] |
| Bistrica | 48.1 | 6.5 | 0.84 | 44.35 | [38] |
| Cacanska lepotica | 55.3 | 6.3 | 1.6 | 36.8 | [38] |
| President | 47.9 | 6.7 | 0.8 | 44.7 | [38] |
| Stanley | 48.8 | 6.6 | 0.9 | 44.8 | [38] |
| Bistrica | 54.6 | 6.8 | 0.8 | 37.7 | [38] |
| Empty fruit bunch | 46.0 | 6.4 | 0.5 | 31.8 | [23] |
| Palm kernel shell | 48.4 | 6.5 | 1.3 | 0.4 | [23] |
| Coconut shell | 50.2 | 5.7 | 0.7 | 42.6 | [23] |
| Waste Type | C (%) | H (%) | N (%) | S (%) | O (%) | References |
|---|---|---|---|---|---|---|
| Cow dung (manure) | 34.4 | 34.4 | 34.4 | 34.4 | 34.4 | [39] |
| Pig manure | 33.2 | 5.4 | 1.2 | 0.1 | 60.1 | [40] |
| Chicken feathers | 47.5 | 6 | 0.4 | 0.1 | 46 | [41] |
| Fish waste | 35.0 | 5.5 | 1.5 | 0.1 | 58.0 | [42] |
| Crustacean shells | 30.0 | 5.0 | 0.2 | 0.1 | 64.7 | [42] |
| Group | Item | Material/notes | C (%) | H (%) | N (%) | O (%) | S (%) |
|---|---|---|---|---|---|---|---|
| Biomass-rich | Microbial biomass residue | Cell pellets | 45–55 | 6–8 | 6–8 | 30–40 | 0.5–1 |
| Biomass-rich | Spent culture media | Residual biomass + salts | 40–50 | 5–7 | 4–6 | 35–45 | 0.2–0.5 |
| Biomass-rich | Solid fermentation residues | Insoluble residues | 50–60 | 6–8 | 5–7 | 25–35 | 0.3–0.6 |
| Cellulose-rich | Swab tip | Cotton | 44–45 | 6–6.5 | 0 | 48–50 | 0 |
| Cellulose-rich | Filter paper | Cellulose | 44–45 | 6–6.5 | 0 | 48–50 | 0 |
| Plastic-rich | Petri dish/Plate | Polystyrene | 92.3 | 7.7 | 0 | 0 | 0 |
| Plastic-rich | Pipette tip | Polypropylene | 85–86 | 14–15 | 0 | 0 | 0 |
| Plastic-rich | Swab tip | Polyester | ~70 | ~10 | trace | ~20 | 0 |
| Plastic-rich | Gloves | Latex | 85–88 | 10–12 | 0–1 | 0–2 | 0.5–2 |
| Plastic-rich | Gloves | Nitrile | 81–84 | 10–12 | 1–2 | 0–1 | 0.1–0.5 |
| Plastic-rich | Masks | Polypropylene | 85–86 | 14–15 | 0 | 0–1 | 0 |
| Plastic-rich | Filter membranes | PVDF | ~33 | ~4 | 0 | 0 | 0 |
| Plastic-rich | Filter membranes | Nylon | ~55 | ~6 | 9–10 | ~30 | 0 |
| Plastic-rich | Packaging waste | PE, PP, PS | 85–93 | 7–15 | 0 | 0–1 | 0–2 |
| Elemental composition values are estimates based on commonly used materials; actual values may vary. | |||||||
| Type of solid waste | Carbon (%) | Hydrogen (%) | Nitrogen (%) | Oxygen (%) | References |
|---|---|---|---|---|---|
| Fish bones, scales, skin, fins, offal, carcasses (general fish tissue/processing waste, dry basis) | 40–52 | 6–8 | 8–13 | 25–35 | [43] |
| Fish scales (dried sample) | 20 | 3 | 6 | 69 | [44] |
| Shells/exoskeletons (shrimp, crab, lobster — chitin organic fraction) | 47.3 | 6.45 | 6.89 | 39.37 | [45] |
| Solid residues from fishmeal production (fishmeal, dry basis) | 45–52 | 6–8 | 9.5–12 | 25–35 | [46,47] |
| Residues of microbial biomass (marine microbes, dry organic fraction) | 50–55 | 6–8 | 10–14 | 20–25 | [48] |
| Organic-rich sediments & detritus (marine organic fraction) | 40–55 | 5–8 | 2–8 | 30–45 | [49] |
| Plastics—Polyethylene (PE) | 85.63 | 14.37 | [50] | ||
| Plastics—Polypropylene (PP) | 85.63 | 14.37 | [50] | ||
| Plastics—Polyethylene terephthalate (PET) | 62.5 | 4.2 | 33.3 | [50] | |
| Plastics—Nylon-6 (polyamide) | 63.69 | 9.8 | 12.38 | 14.14 | [50] |
| Material | Carbon (%) | Hydrogen (%) | Nitrogen (%) | Sulfur (%) | Oxygen (%) | Reference |
|---|---|---|---|---|---|---|
| Chlorella | 52.1 ± 0.7 | 6.5 ± 0.2 | 9.95 ± 0.15 | 0.55 ± 0.02 | 30.9 ± 1.2 | [51] |
| Spirulina | 49.4 ± 0.6 | 7.2 ± 0.4 | 10.70 ± 0.12 | 0.60 ± 0.01 | 32.1 ± 0.3 | [51] |
| Enteromorpha clathrata | 32.7 | 5.38 | 4.85 | 2.01 | 51.9 | [30] |
| Algal Waste | 35.27 | 4.71 | 4.44 | 0.73 | 54.85 | [52] |
| Ulva sp. | 5.78 | 0.68 | [53] | |||
| Ulva sp. | 24.34 | 2.77 | [53] | |||
| Green algae | 26.3 | 3.6 | [53] | |||
| Green algae | 19.5 | 3.6 | [53] | |||
| Blue-green algae | 36.3 | 7.4 | [53] | |||
| Blue-green algae | 44.9 | 7.4 | [53] | |||
| Undaria pinnatifida | 31.32 | 2.67 | [53] | |||
| Undaria pinnatifida | 36.95 | 4.14 | [53] |
| Reaction No. | Description | Reaction |
|---|---|---|
| R1 | Pyrolysis | Biomass → CO + H2 + CO2 + CH4 + H2O + Tar + Char |
| R2 | Char oxidation | Char + O2 → CO2 |
| R3 | Partial oxidation | C + ½ O2 → CO |
| R4 | Hydrogen oxidation | H2 + ½ O2 → H2O |
| R5 | Boudouard reaction | C + CO2 → 2CO |
| R6 | Reforming of char | C + H2O → CO + H2 |
| R7 | Water gas shift | CO + H2O → CO2 + H2 |
| R8 | Methanation reaction | C + 2H2 → CH4 |
| R9 | Steam reforming of methane | CH4 + H2O → CO + 3H2 |
| R10 | Dry reforming of methane | CH4 + CO2 → 2CO + 2H2 |
| R11 | Steam reforming of tar | Tar + H2O → H2 + CO2 + CO + CₓHy |
| Method | Process description | Temperature range (°c) | Residence time | Comments | References |
|---|---|---|---|---|---|
| Slow Pyrolysis | Thermal decomposition of biomass in low-oxygen conditions at moderate heat. | 400–600 | Hours to days | High biochar yield; commonly used for soil amendment. | [55] |
| Fast Pyrolysis | Rapid heating of biomass with limited oxygen; focus on bio-oil production. | 500–600 | Seconds to minutes | Lower biochar yield; higher liquid (bio-oil) yield. | [56] |
| Gasification | Partial oxidation of biomass produces syngas; biochar is a by-product. | 700–1,000 | Minutes to hours | Produces energy-rich gases; biochar yield is moderate. | [54] |
| Hydrothermal Carbonisation (HTC) | Biomass treated in hot, pressurized water (wet biomass). | 180–250 | Hours | Suitable for wet feed stocks; produces hydrochar. | [57] |
| Torrefaction | Mild pyrolysis at lower temperatures to produce a coal-like biochar. | 200–300 | Hours | Improves biomass energy density; easier to grind and store. | [58] |
| Microwave Pyrolysis | Biomass heated using microwaves in oxygen-limited conditions. | 400–600 | Minutes | Fast, uniform heating; energy-efficient; scalable. | [59] |
| Flash Carbonisation | Very rapid pyrolysis under high temperatures. | 700–1,000 | Seconds | Produces highly porous char for specialised industrial applications. | [55] |
| Reaction No | Description | Reactant/activating agent | Reaction |
|---|---|---|---|
| R1 | Carbonisation | Biomass | Biomass → Char + Volatiles + Gases (CO, CO2, H2, CH4, H2O) |
| R2 | Partial oxidation | Char + Air (O2) | C + ½ O2 → CO |
| R3 | Char oxidation | Char + O2 | C + O2 → CO2 |
| R4 | Boudouard reaction | Char + CO2 | C + CO2 → 2CO |
| R5 | Steam-carbon reaction | Char + H2O | C + H2O → CO + H2 |
| R6 | Water-gas shift | CO + H2O | CO + H2O → CO2 + H2 |
| R7 | Methanation | Char + H2 | C + 2H2 → CH4 |
| R8 | Chemical activation | KOH | 6KOH + 2C → 2K + 3H2 + 2K2CO3 → Activated Carbon + Gases |
| R9 | Chemical activation | NaOH | 6NaOH + 2C → 2Na + 3H2 + 2Na2CO3 → Activated Carbon + Gases |
| R10 | Chemical activation | ZnCl2 | Biomass + ZnCl2 → ZnCl2–Biomass → Heat → Activated Carbon + Zn + HCl + Gases |
| R11 | Chemical activation | H3PO4 | Biomass + H3PO4 → C–O–P + H2O → Activated Carbon with Phosphate Groups |
| Applications | Uses |
|---|---|
| Automobile industry | Capture fuel vapours from tanks and carburettors; remove odours and pollutants from cabin air. |
| Cigarette industry | Filter harmful smoke components like ammonia, cyanide, and formaldehyde. |
| Heavy metals removal | Remove toxic metals (lead, mercury, copper, chromium, nickel, zinc) from water and wastewater. |
| Food processing | Purify oils, juices, honey, MSG, and meat extracts; remove colour, taste, and odour impurities. |
| Hospitals and laboratories | Clean indoor air by removing odours, volatile compounds, and pollutants. |
| Manufacturing and industrial processes | Treat effluent gases and remove pollutants and odours before release. |
| Nuclear power plants | Capture radioactive gases and vapours to protect the environment and workers. |
| Petrochemical, rubber, paints, plastics | Remove chemicals, pesticides, and toxic materials from wastewater. |
| Precious metals recovery | Recover gold, silver, and other metals from solutions or waste streams. |
| Pyrometallurgy and process industries | Remove gases (SO2, NH3), heavy metals, halogens, and organic pollutants from flue gases. |
| Sugar refining | Remove color, molasses, and impurities from raw sugar. |
| Water purification | Remove organic compounds, taste, odor, and micro-pollutants from drinking water. |
| Wastewater treatment | Remove chlorine, solids, cloudiness, organic matter, and odor from industrial or process water. |
| Pharmaceuticals | Purify drugs, vitamins, and antibiotics; remove impurities and unwanted chemicals. |
| Gas separation and storage | Capture CO2, H2S, and other gases; store gases for industrial use. |
| Energy storage | Used in supercapacitors and batteries to store energy efficiently. |
| Air purification | Remove smoke, dust, VOCs, and odors from indoor and outdoor air. |
| Cosmetics | Remove impurities from creams, lotions, and personal care products. |
| Odor control | Control smells in landfills, wastewater plants, and animal farms. |
| Environmental remediation | Clean soil and water contaminated with oils, chemicals, or heavy metals. |
| Biowaste source | Catalytic material | Typical application | References |
|---|---|---|---|
| Eggshell | CaO (alkaline catalyst) | Biodiesel transesterification, base-catalysed organic reactions | [63] |
| Coconut/nutshells | Activated carbon, doped carbon | Catalyst support, electrocatalysis (ORR/OER, HER) | [64] |
| Agricultural residues | Biochar (carbon catalyst) | Biodiesel production, pollutant removal, tar reduction | [65] |
| Shrimp/crab shells | Chitosan & chitosan-metal complexes | Organocatalysis, pollutant degradation, catalyst support | [66] |
| Fish scales | Hydroxyapatite (HAp) | Heterogeneous catalysis, support for metal catalysts | [67] |
| Animal bone | Bone char (carbon + HAp) | Environmental catalysis, phosphate recovery, catalyst support | [68] |
| Banana/citrus peels | K/Ca-rich ash or char | Biodiesel transesterification, green base catalyst | [69] |
| Mollusc shells | CaO (calcined shell catalyst) | Biodiesel, condensation reactions | [70] |
| Lignocellulosic agri waste | Functionalized/doped carbon | Electrocatalysis, hydrogen evolution, supported catalysts | [71] |
| Mixed biowaste | Biochar/HAp composites with metals | Reforming, hydrogen production, environmental remediation | [72] |
| Producer | Bioplastic products | Primary raw materials | Conversion process | References |
|---|---|---|---|---|
| Braskem | Bio-based polyethylene (bio-PE) | Sugarcane ethanol | Fermentation → Ethanol → Ethylene → Polymerisation | [127] |
| NatureWorks LLC | Ingeo™ PLA | Corn starch/dextrose | Wet milling → Dextrose → Fermentation → Lactic acid → Polymerisation | [128] |
| Novamont | Mater-Bi® | Corn starch, cellulose, vegetable oils | Blending, esterification, polymerisation | [129] |
| Corbion | PLA, PHA | Corn glucose, sugarcane | Fermentation → Lactic acid or PHA → Polymerisation | [130] |
| BASF SE | Ecoflex®, Ecovio® | Starch, cellulose, renewable monomers | Chemical modification and polymerisation | [131] |
| Biome Bioplastics | Biodegradable and compostable plastics | Potato starch, corn starch, cellulose | Blending, polymerisation, compounding | [132] |
| Danimer Scientific | Nodax® PHA | Canola oil, soy oil | Microbial fermentation → PHA polymer accumulation → Extraction & processing | [133] |
| Toray Industries | PLA, PHA | Corn, sugarcane, plant-derived sugars | Fermentation → Lactic acid or PHA → Polymerisation | [134] |
| Mitsubishi Chemical | Bio-PE, PLA | Sugarcane ethanol, corn | Fermentation → Ethanol → Ethylene → Polymerization; or → Lactic acid → PLA | [135] |
| Plantic Technologies | Biodegradable and compostable plastics | Corn starch, plant-based resins | Gelatinisation, blending, and compounding | [136] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




