Submitted:
17 September 2025
Posted:
18 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
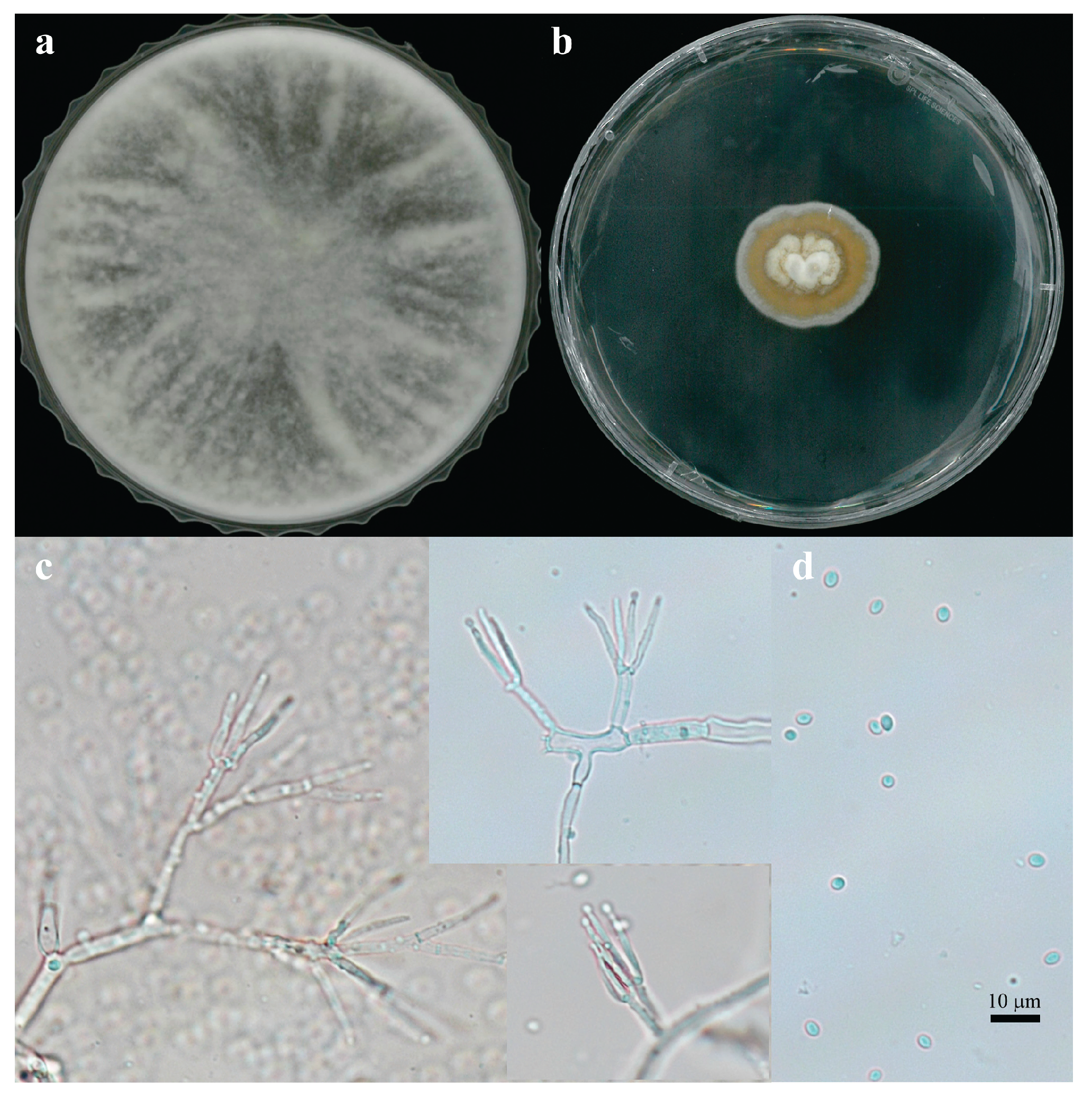
2.1. Isolation and Identification of FBCC-F1632
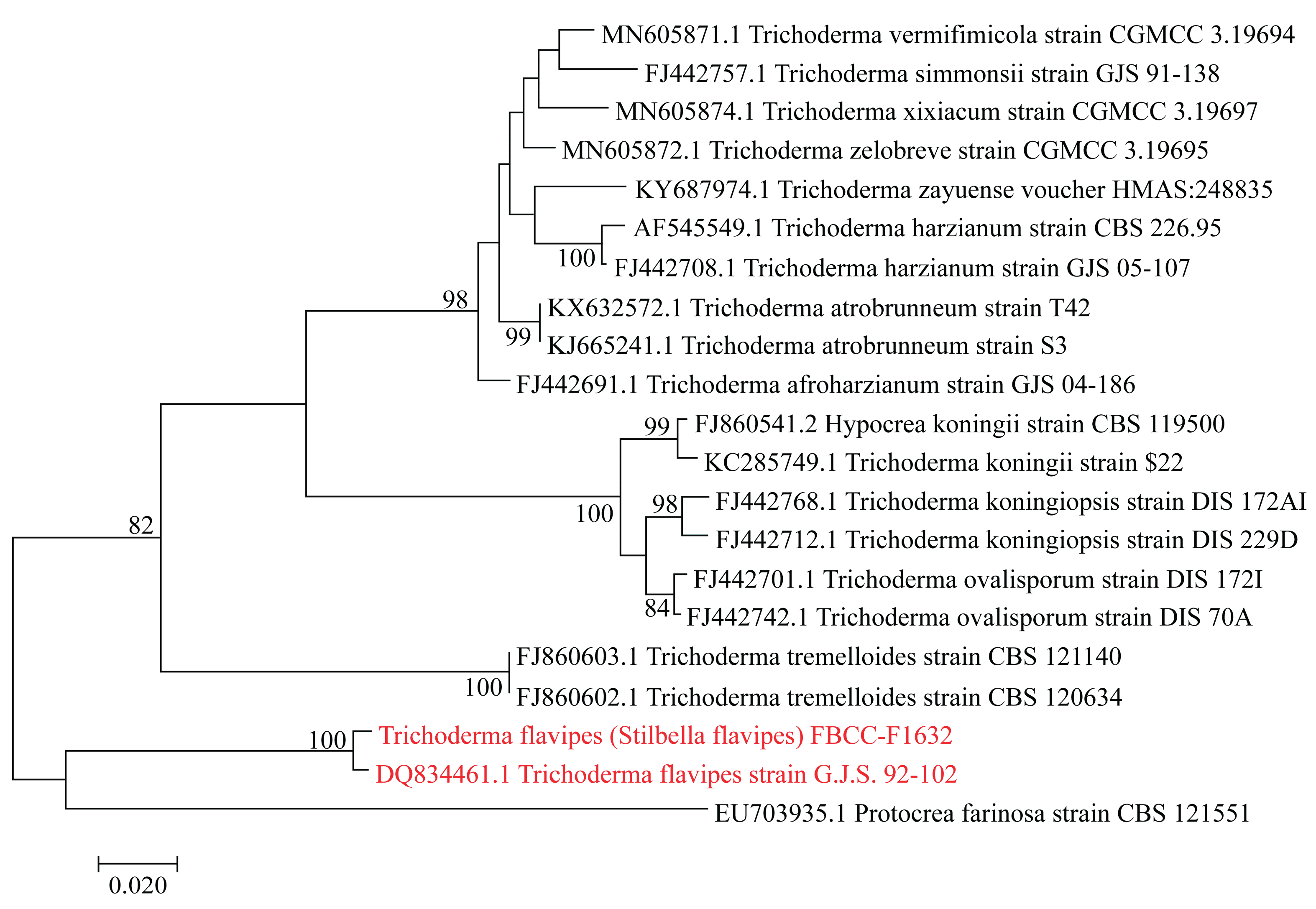
2.2. DNA Extraction and Phylogenetic Analysis
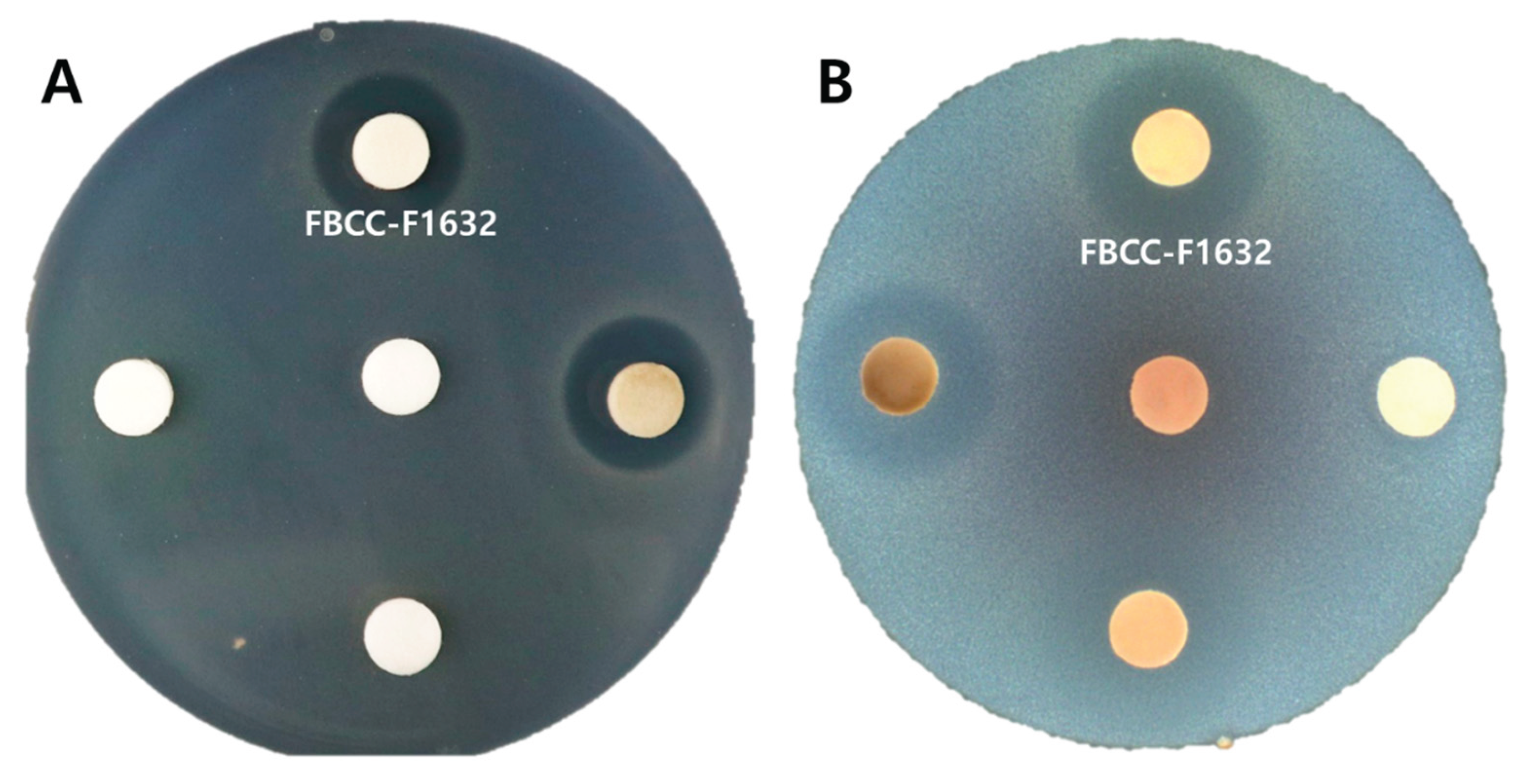
2.3. Antimicrobial Activity
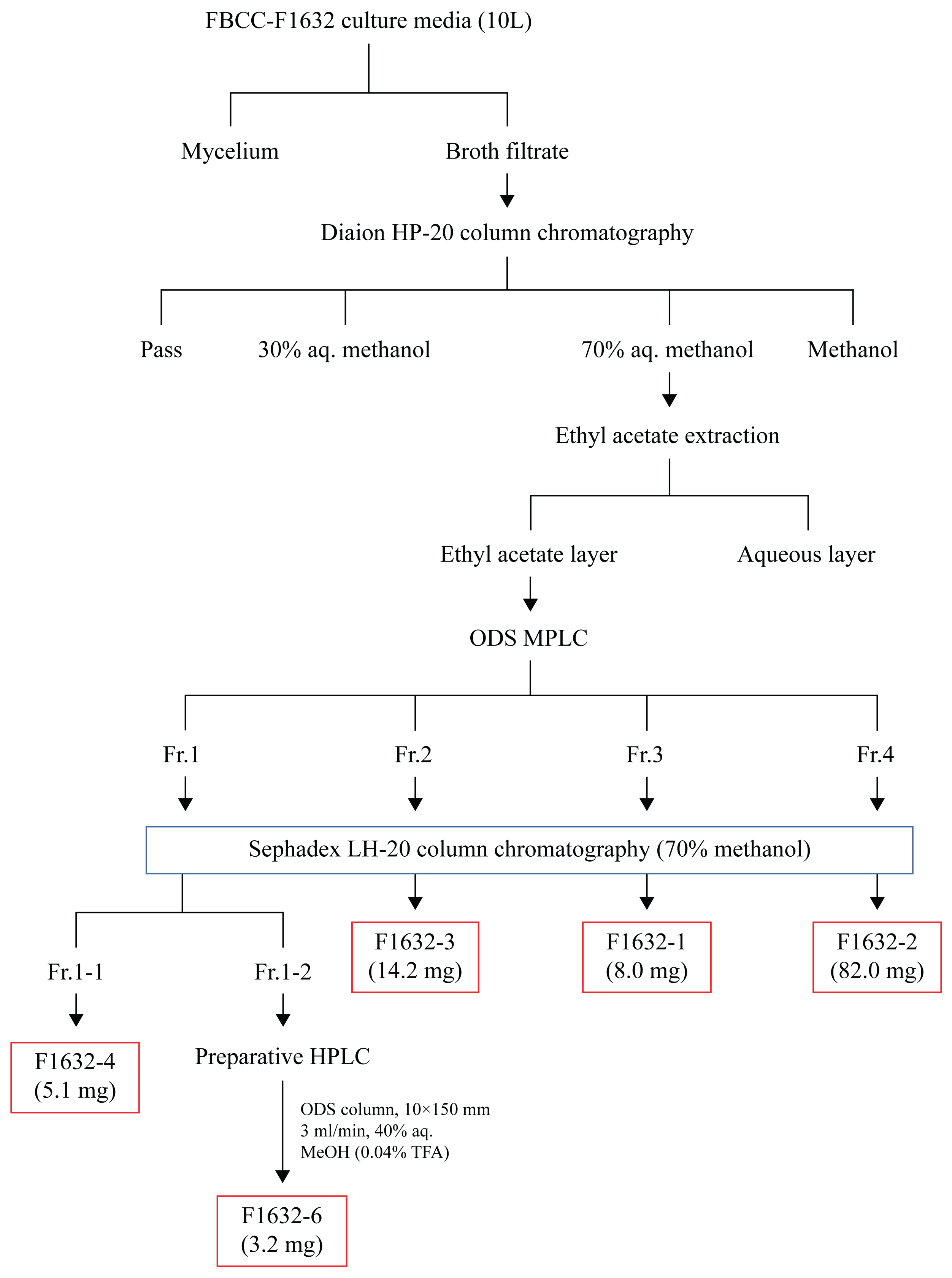
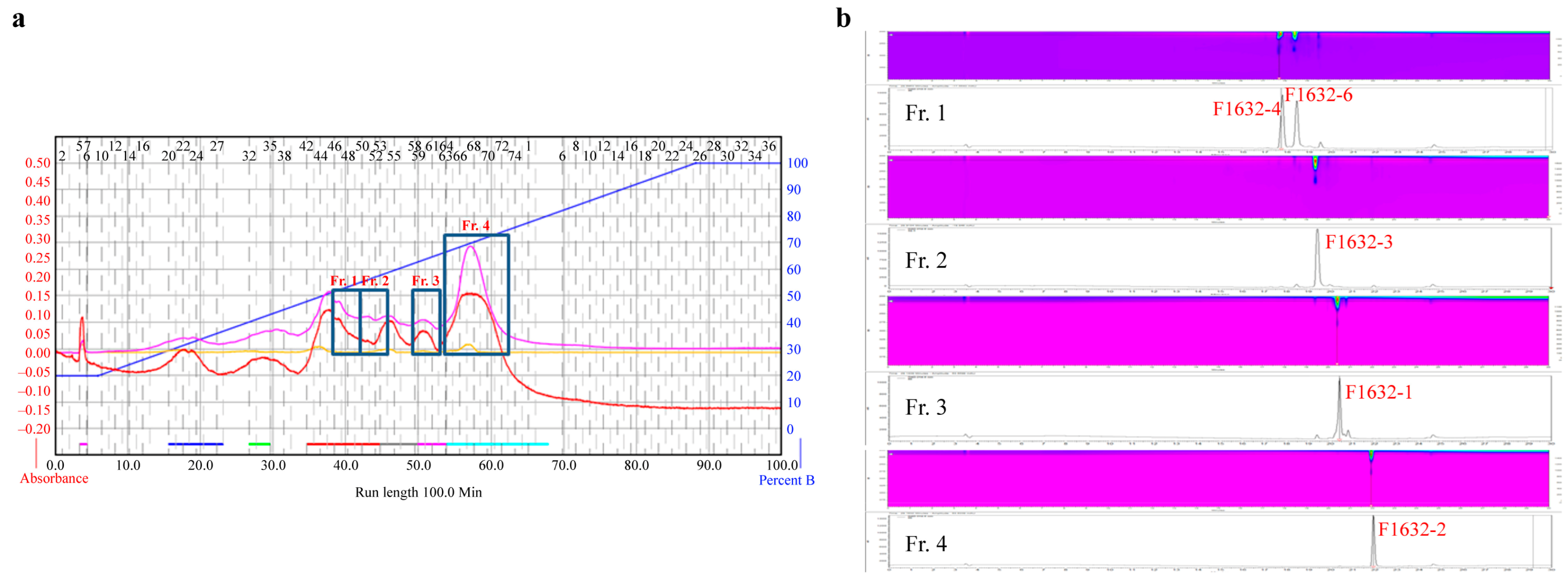
2.4. Material Separation and Purification
2.5. Structure Analysis
3. Results
3.1. Isolation and Identification of FBCC-F1632
3.2. Antimicrobial Activity of FBCC-F1632
3.3. Compound Extraction from FBCC-F1632
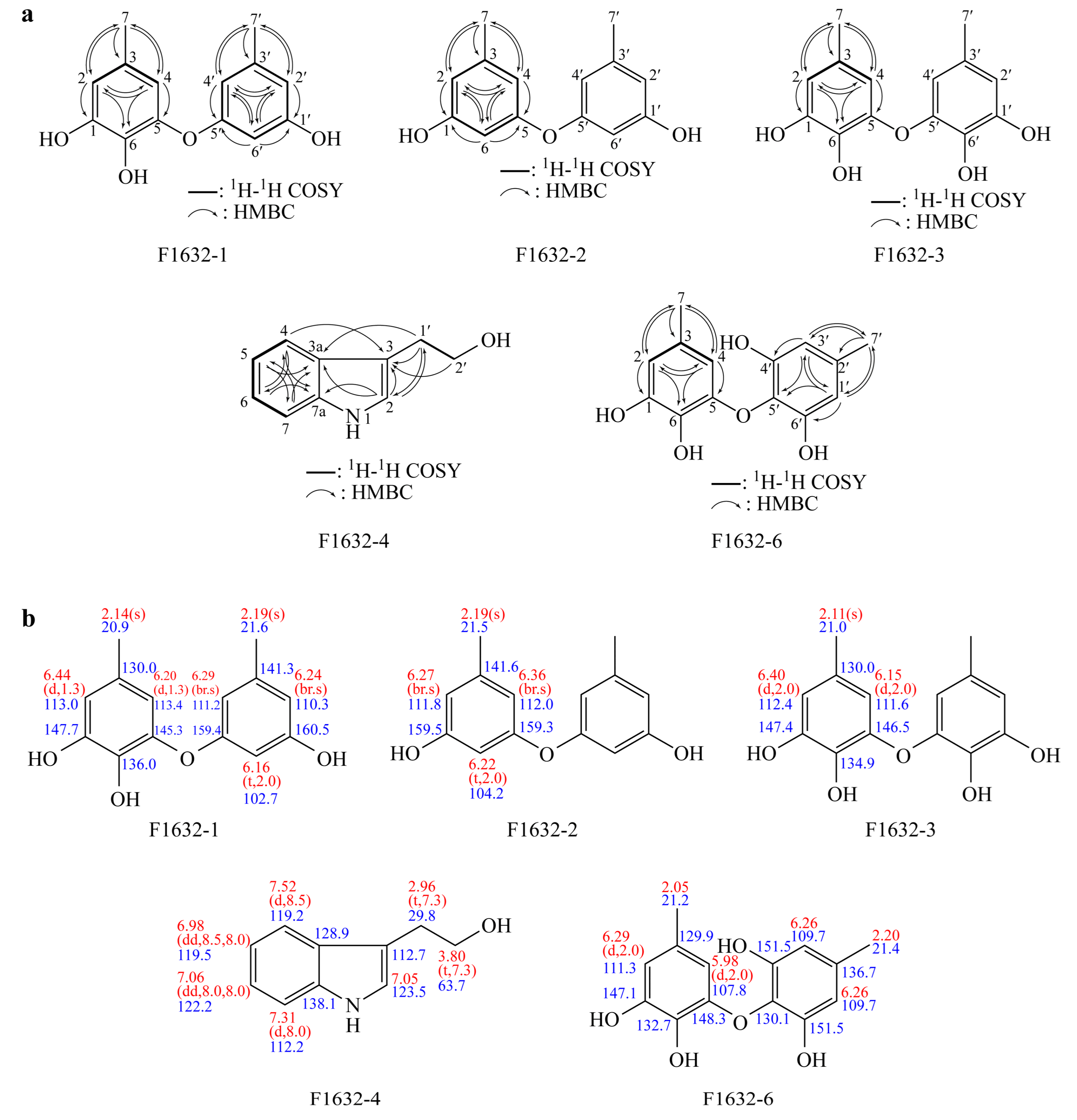
3.4. Structure Analysis
3.4.1. Structure of F1632-1
3.4.2. Structure of F1632-2
3.4.3. Structure of F1632-3
3.4.4. Structure of F1632-4
3.4.5. Structure of F1632-6
3.5. Antimicrobial Activity of Compounds Isolated from FBCC-F1632
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Calabon, M.S.; Hyde, K.D.; Jones, E.B.G.; Luo, Z.-L.; Dong, W.; Hurdeal, V.G.; Gentekaki, E.; Rossi, W.; Leonardi, M.; Thiyagaraja, V.; Lestari, A.S.; Shen, H.-W.; Bao, D.-F.; Boonyuen, N.; Zeng, M. Freshwater fungal numbers. Fungal Divers. 2022, 114, 3–235. [Google Scholar] [CrossRef]
- El-Elimat, T.; Raja, H.A.; Figueroa, M.; Al Sharie, A.H.; Bunch, R.L.; Oberlies, N.H. Freshwater fungi as a source of chemical diversity: A review. J. Nat. Prod. 2021, 84, 898–916. [Google Scholar] [CrossRef]
- Calabon, M.S.; Hyde, K.D.; Jones, E.B.G.; Bao, D.; Bhunjun, C.; Phukhamsakda, C.; Shen, H.; Gentekaki, E.; Al Sharie, A.; Barros, J.; Chandrasiri, K.; Hu, D.; Hurdeal, V.; Rossi, W.; Valle, L.; Zhang, H.; Figueroa, M.; Raja, H.; Seena, S.; Song, H.; Dong, W.; El-Elimat, T.; Leonardi, M.; Li, Y.; Li, Y.; Luo, Z.; Ritter, C.; Strongman, D.; Wei, M.; Balasuriya, A. Freshwater fungal biology. Mycosphere 2023, 14, 195–413. [Google Scholar] [CrossRef]
- Ślusarczyk, J.; Adamska, E.; Czerwik-Marcinkowska, J. Fungi and algae as sources of medicinal and other biologically active compounds: A review. Nutrients 2021, 13, 3178. [Google Scholar] [CrossRef] [PubMed]
- Hammerschmidt, S.; Hacker, J.; Klenk, H.-D. Threat of infection: Microbes of high pathogenic potential – strategies for detection, control and eradication. Int. J. Med. Microbiol. 2005, 295, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Huemer, M.; Mairpady Shambat, S.; Brugger, S.D.; Zinkernagel, A.S. Antibiotic resistance and persistence-Implications for human health and treatment perspectives. E.M.B.O. Rep. 2020, 21, e51034. [Google Scholar] [CrossRef] [PubMed]
- Jaklitsch, W.M.; Voglmayr, H. New combinations in Trichoderma (Hypocreaceae, Hypocreales). Mycotaxon 2013, 126, 143–156. [Google Scholar] [CrossRef]
- Zin, N.A.; Badaluddin, N.A. Biological functions of Trichoderma spp for agriculture applications. Ann. Agric. Sci. 2020, 65, 168–178. [Google Scholar] [CrossRef]
- Guo, R.; Li, G.; Zhang, Z.; Peng, X. Structures and biological activities of secondary metabolites from Trichoderma harzianum. Mar. Drugs 2022, 20, 701. [Google Scholar] [CrossRef]
- Siscar-Lewin, S.; Hube, B.; Brunke, S. Emergence and evolution of virulence in human pathogenic fungi. Trends Microbiol. 2022, 30, 693–704. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Schmoll, M.; Esquivel-Ayala, B.A.; González-Esquivel, C.E.; Rocha-Ramírez, V.; Larsen, J. Mechanisms for plant growth promotion activated by Trichoderma in natural and managed terrestrial ecosystems. Microbiol. Res. 2024, 281, 127621. [Google Scholar] [CrossRef]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among ascomycetes: Evidence from an RNA polymerse II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Samuels, G.J. Trichoderma: Systematics, the sexual state, and ecology. Phytopathology 2006, 96, 195–206. [Google Scholar] [CrossRef]
- Kubiak, A.; Wolna-Maruwka, A.; Pilarska, A.A.; Niewiadomska, A.; Piotrowska-Cyplik, A. Fungi of the Trichoderma genus: Future perspectives of benefits in sustainable agriculture. Appl. Sci. 2023, 13, 6434. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Nei, M.; Kumar, S. Molecular Evolution and Phylogenetics; Oxford University Press: New York, 2000. [Google Scholar]
- Yamazaki, M.; Maebayashi, Y. Structure determination of violaceol-I and -II, new fungal metabolites from a strain of Emericella violacea. Chem. Pharm. Bull. 1982, 30, 514–518. [Google Scholar] [CrossRef]
- Nuankeaw, K.; Chaiyosang, B.; Suebrasri, T.; Kanokmedhakul, S.; Lumyong, S.; Boonlue, S. First report of secondary metabolites, Violaceol I and Violaceol II produced by endophytic fungus, Trichoderma polyalthiae and their antimicrobial activity. Mycoscience 2020, 61, 16–21. [Google Scholar] [CrossRef]
- Bottone, E.J. Bacillus cereus, a volatile human pathogen. Clin. Microbiol. Rev. 2010, 23, 382–398. [Google Scholar] [CrossRef]
- Liu, Q.; Meng, X.; Li, Y.; Zhao, C.-N.; Tang, G.-Y.; Li, H.-B. Antibacterial and antifungal activities of spices. Int. J. Mol. Sci. 2017, 18, 1283. [Google Scholar] [CrossRef]
- Wang, J.-F.; Lin, X.-P.; Qin, C.; Liao, S.-R.; Wan, J.-T.; Zhang, T.-Y.; Liu, J.; Fredimoses, M.; Chen, H.; Yang, B.; Zhou, X.-F.; Yang, X.-W.; Tu, Z.-C.; Liu, Y.-H. Antimicrobial and antiviral sesquiterpenoids from sponge-associated fungus, Aspergillus sydowii ZSDS1-F6. J. Antibiot. (Tokyo) 2014, 67, 581–583. [Google Scholar] [CrossRef]
- Bunyapaiboonsri, T.; Yoiprommarat, S.; Intereya, K.; Kocharin, K. New diphenyl ethers from the insect pathogenic fungus Cordyceps sp BCC 1861. Chem. Pharm. Bull. (Tokyo) 2007, 55, 304–307. [Google Scholar] [CrossRef]
- Xu, L.; Meng, W.; Cao, C.; Wang, J.; Shan, W.; Wang, Q. Antibacterial and antifungal compounds from marine fungi. Mar. Drugs 2015, 13, 3479–3513. [Google Scholar] [CrossRef] [PubMed]
- Itabashi, T.; Nozawa, K.; Nakajima, S.; Kawai, K. A new azaphilone, falconensin H, from Emericella falconensis. Chem. Pharm. Bull. 1993, 41, 2040–2041. [Google Scholar] [CrossRef]
- Van Nguyen, M.V.; Han, J.W.; Kim, H.; Choi, G.J. Phenyl ethers from the marine-derived fungus Aspergillus tabacinus and their antimicrobial activity against plant pathogenic fungi and bacteria. A.C.S. Omega 2022, 7, 33273–33279. [Google Scholar] [CrossRef] [PubMed]
| Position | F1632-1 | F1632-2 | F1632-3 | F1632-4 | F1632-6 | |||||
| δ13C(mult.) | δ1H(mult.) | δ13C(mult.) | δ1H(mult.) | δ13C(mult.) | δ1H(mult.) | δ13C(mult.) | δ1H(mult.) | δ13C(mult.) | δ1H(mult.) | |
| 1 | 147.7 | 159.5 | 147.4 | 147.1 | ||||||
| 2 | 113.0 | 6.44 (1H, d, J=1.3 Hz) | 111.8 | 6.27 (2H, br. s) | 112.4 | 6.40 (2H, d, J=2.0 Hz) | 123.5 | 7.05 (1H, s) | 111.3 | 6.29 (1H, d, J=2.0 Hz) |
| 3 | 130.0 | 141.6 | 130.0 | 112.7 | 129.9 | |||||
| 3a | 128.9 | |||||||||
| 4 | 113.4 | 6.20 (1H, d, J=1.3 Hz) | 112 | 6.36 (2H, br. s) | 111.6 | 6.15 (2H, d, J=2.0 Hz) | 119.2 | 7.52 (1H, d, J=8.5 Hz) | 107.8 | 5.98 (1H, d, J=2.0 Hz) |
| 5 | 145.3 | 159.3 | 146.5 | 119.5 | 6.98 (1H, dd, J=8.5, 8.0 Hz) | 148.3 | ||||
| 6 | 136.0 | 104.2 | 6.22 (2H, t, J=2.0 Hz) | 134.9 | 122.2 | 7.06 (1H, dd, J=8.0, 8.0 Hz) | 132.7 | |||
| 7 | 20.9 | 2.14 (3H, s) | 21.5 | 2.19 (6H, s) | 21.0 | 2.11 (6H, s) | 112.2 | 7.31 (1H, d, J=8.0 Hz) | 21.2 | 2.05 (3H, s) |
| 7a | 138.1 | |||||||||
| 1’ | 160.5 | 29.8 | 2.96 (2H, t, J=7.3 Hz) | 109.7 | ||||||
| 2’ | 110.3 | 6.24 (1H, br. s) | 63.7 | 3.80 (2H, t, J=7.3 Hz) | 136.7 | |||||
| 3’ | 141.3 | 109.7 | 6.26 (2H, s) | |||||||
| 4’ | 111.2 | 6.29 (1H, br. s) | 151.5 | |||||||
| 5’ | 159.4 | 130.1 | ||||||||
| 6’ | 102.7 | 6.16 (1H, t, J=2.0 Hz) | 151.5 | |||||||
| 7’ | 21.6 | 2.19 (3H, s) | 21.4 | 2.20 (3H, s) | ||||||
| No. | Compound | Name | MIC concentration | MBC concetration | ||
| S. aureus | B. cereus | S. aureus | B. cereus | |||
| 1 | F1632-1 | Cordyol C | 50 | 12.5 | 50 | 25 |
| 2 | F1632-2 | Diorcinol | 50 | 25 | - | 50 |
| 3 | F1632-3 | Violaceol I | 12.5 | 25 | 25 | 50 |
| 4 | F1632-4 | Tryprophol | - | - | - | - |
| 5 | F1632-6 | Violaceol II | 25 | 25 | 50 | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




