Submitted:
13 September 2025
Posted:
15 September 2025
You are already at the latest version
Abstract
Keywords:
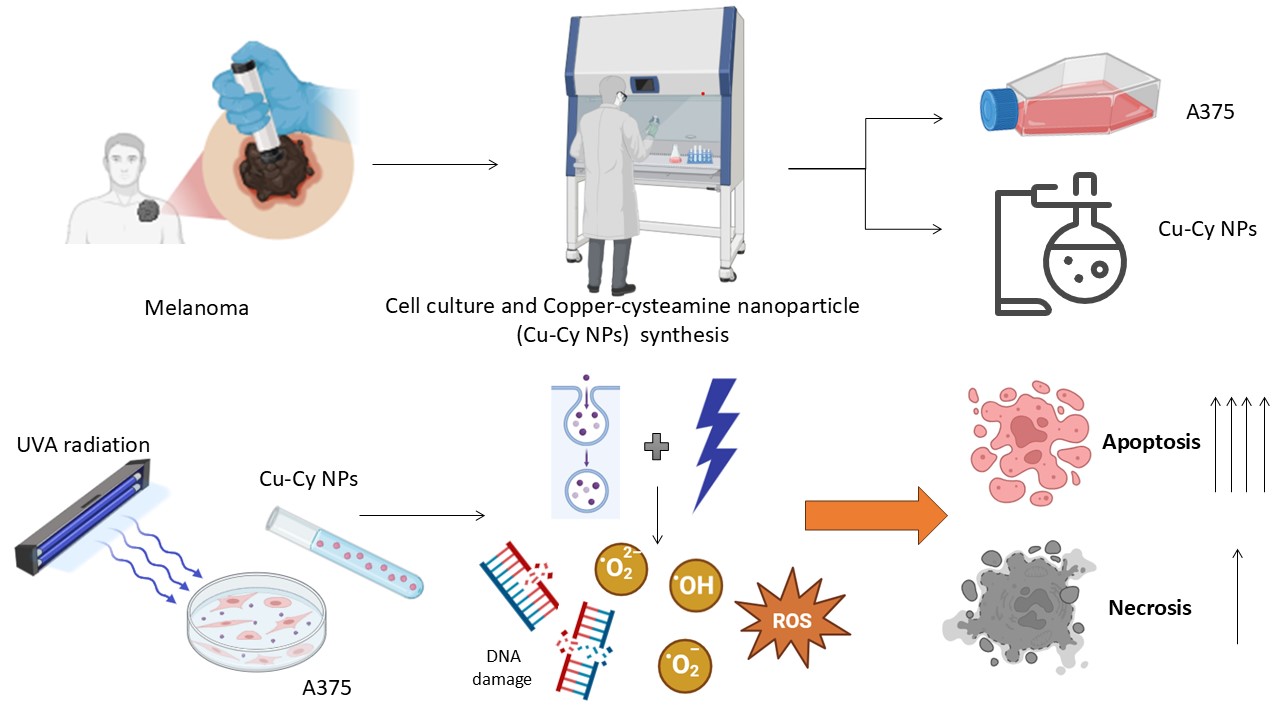
1. Introduction
2. Materials and Methods
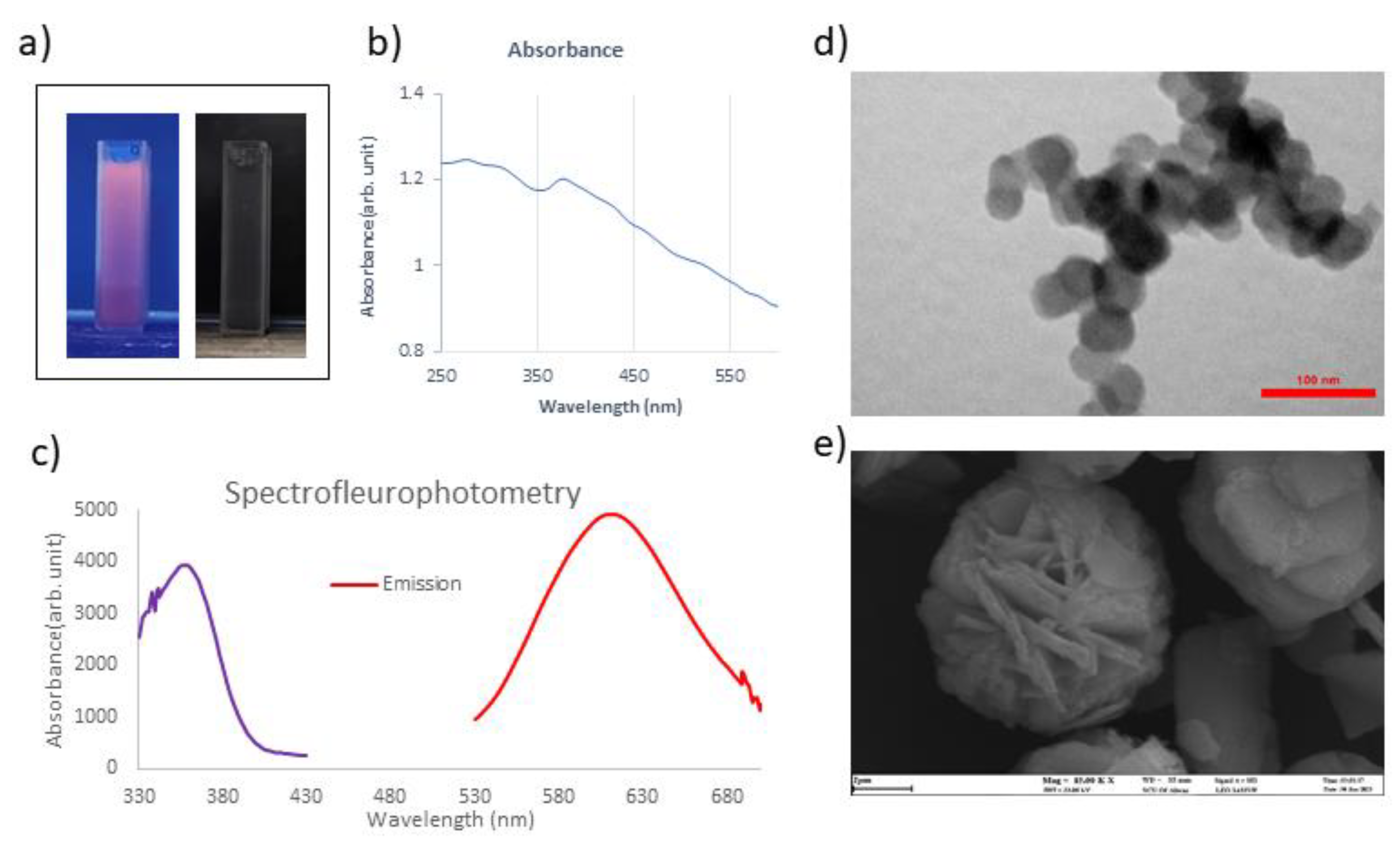
2.1. Synthesis and Characterization of Cu-Cy NPs:
2.2. Exploring the Synergistic Effect of Cu-Cy NPs and UV
2.2.1. Cell Culture
2.2.2. UV Irradiation
2.2.3. MTT Viability Assay
2.2.4. Assessment of Intracellular ROS Generation Using the NBT Assay
2.2.5. Apoptosis Assay Using Annexin V-FITC/PI Staining
2.3. Statistical Analysis
3. Result
3.1. Synthesis and Characterization of Cu-Cy NPs
3.2. Synergistic Effect of Cu-Cy NPs and UVA Irradiation
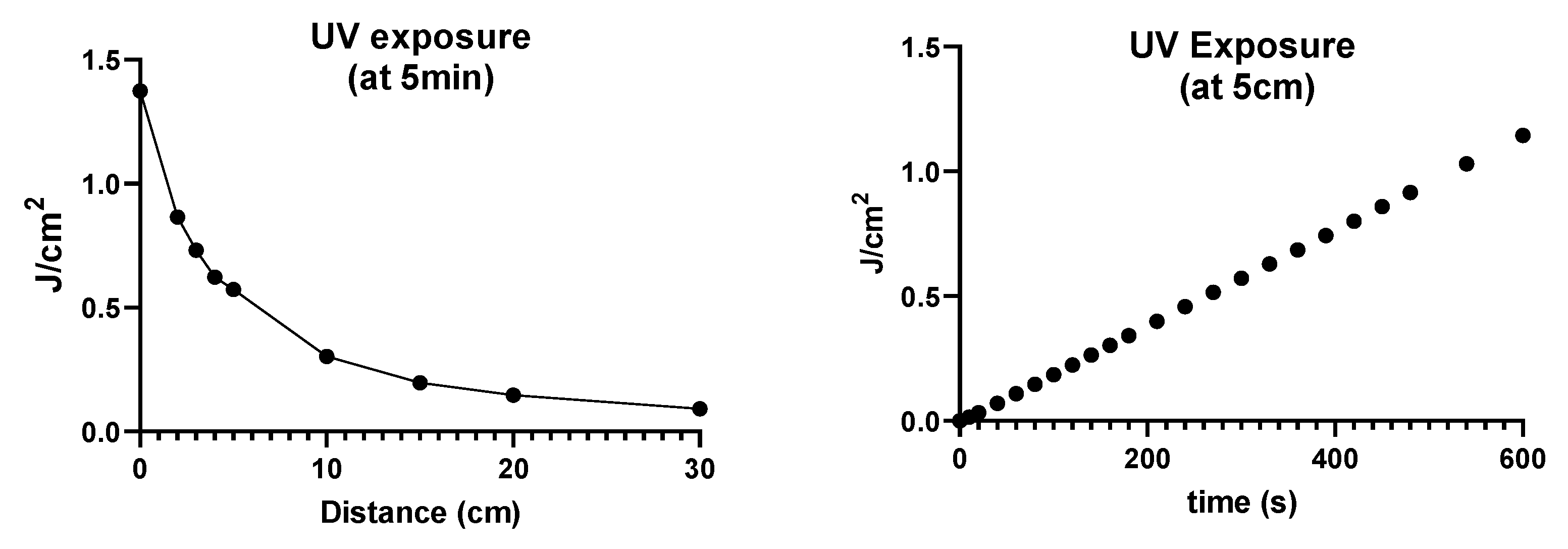
3.2.1. UVA Irradiation Dosimetry
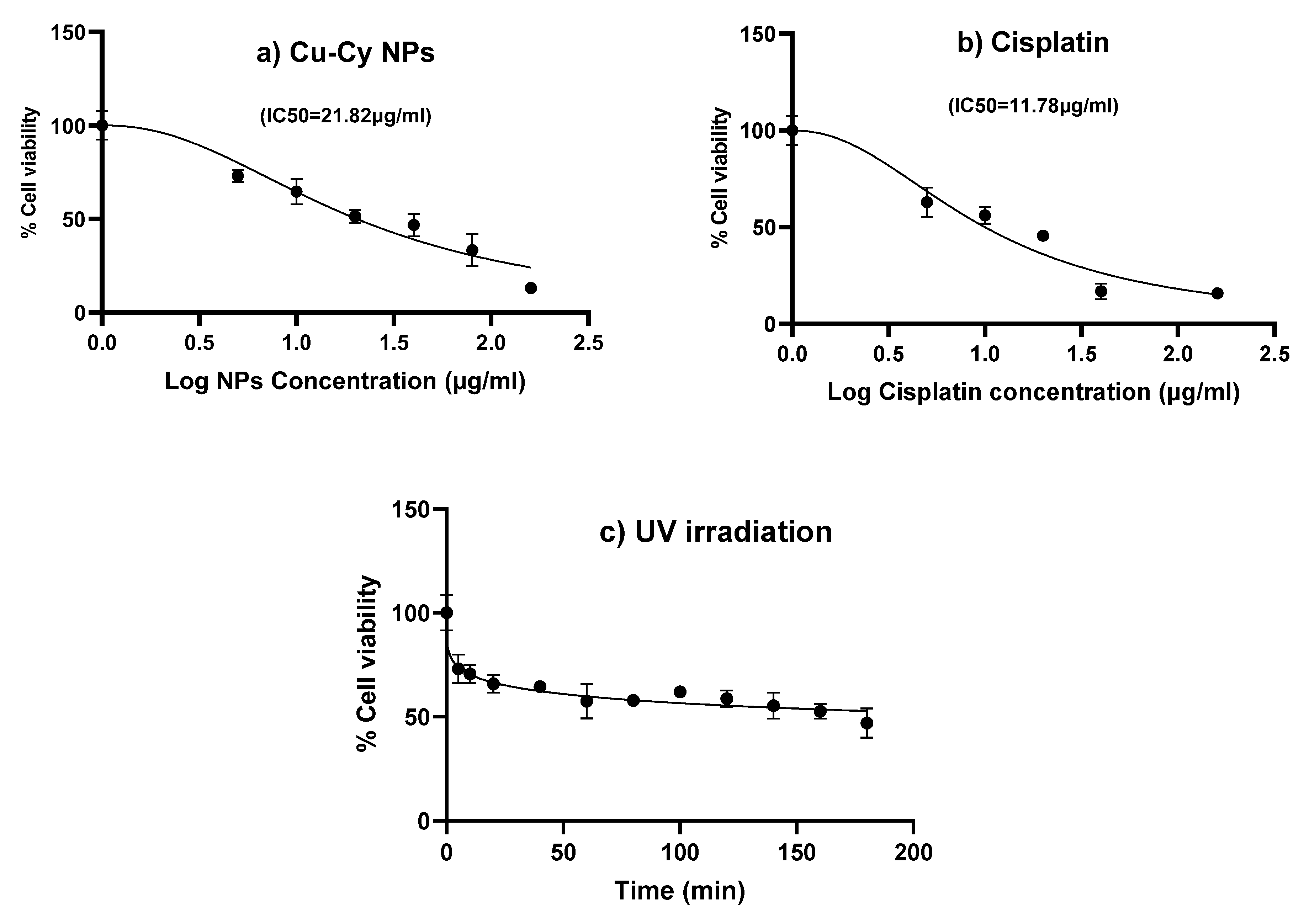
3.2.2. Cytotoxicity Assessment by MTT Assay
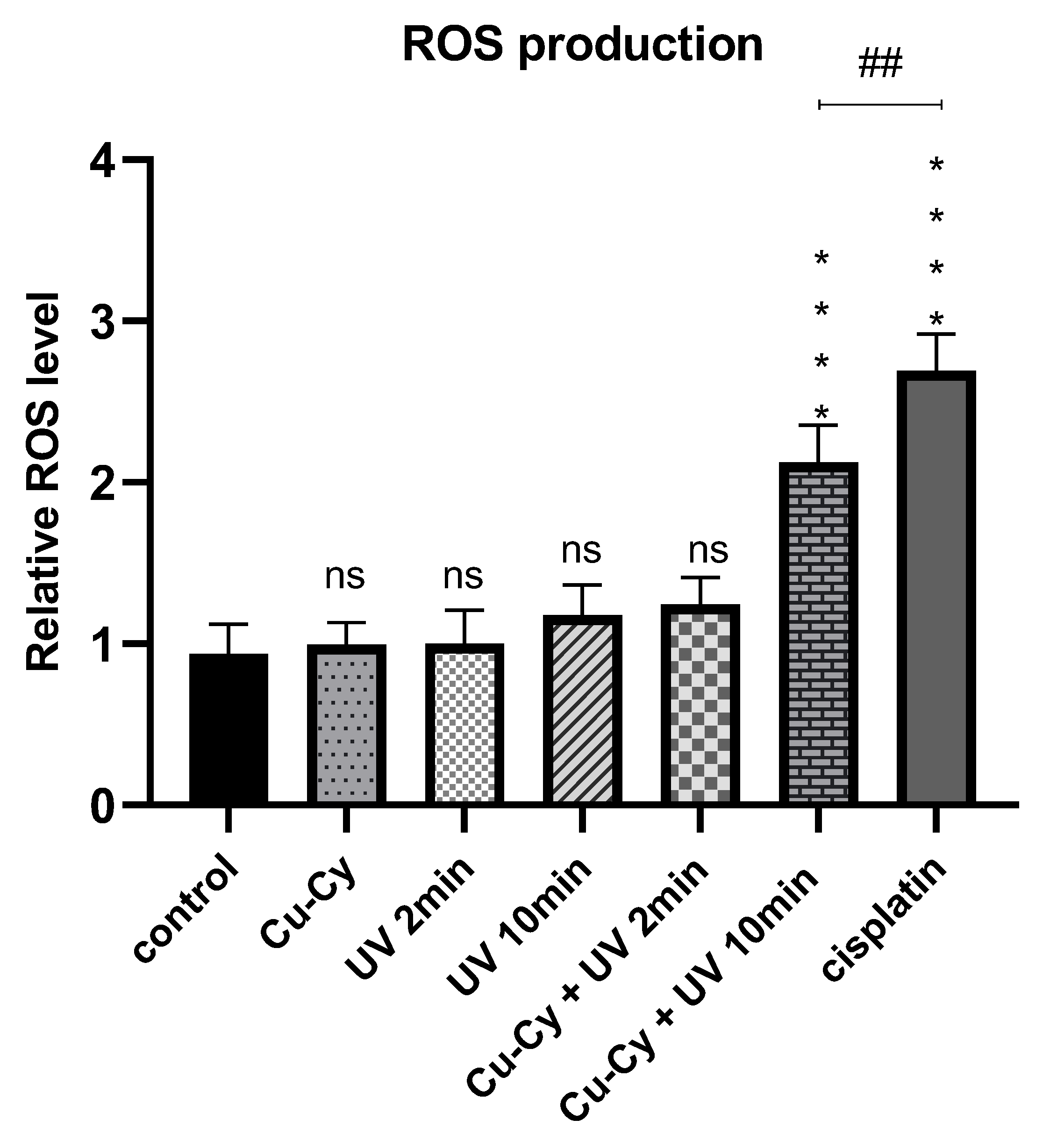
3.2.3. Intracellular ROS Generation Assessed by NBT Assay
3.2.4. Apoptosis Analysis by Flow Cytometry
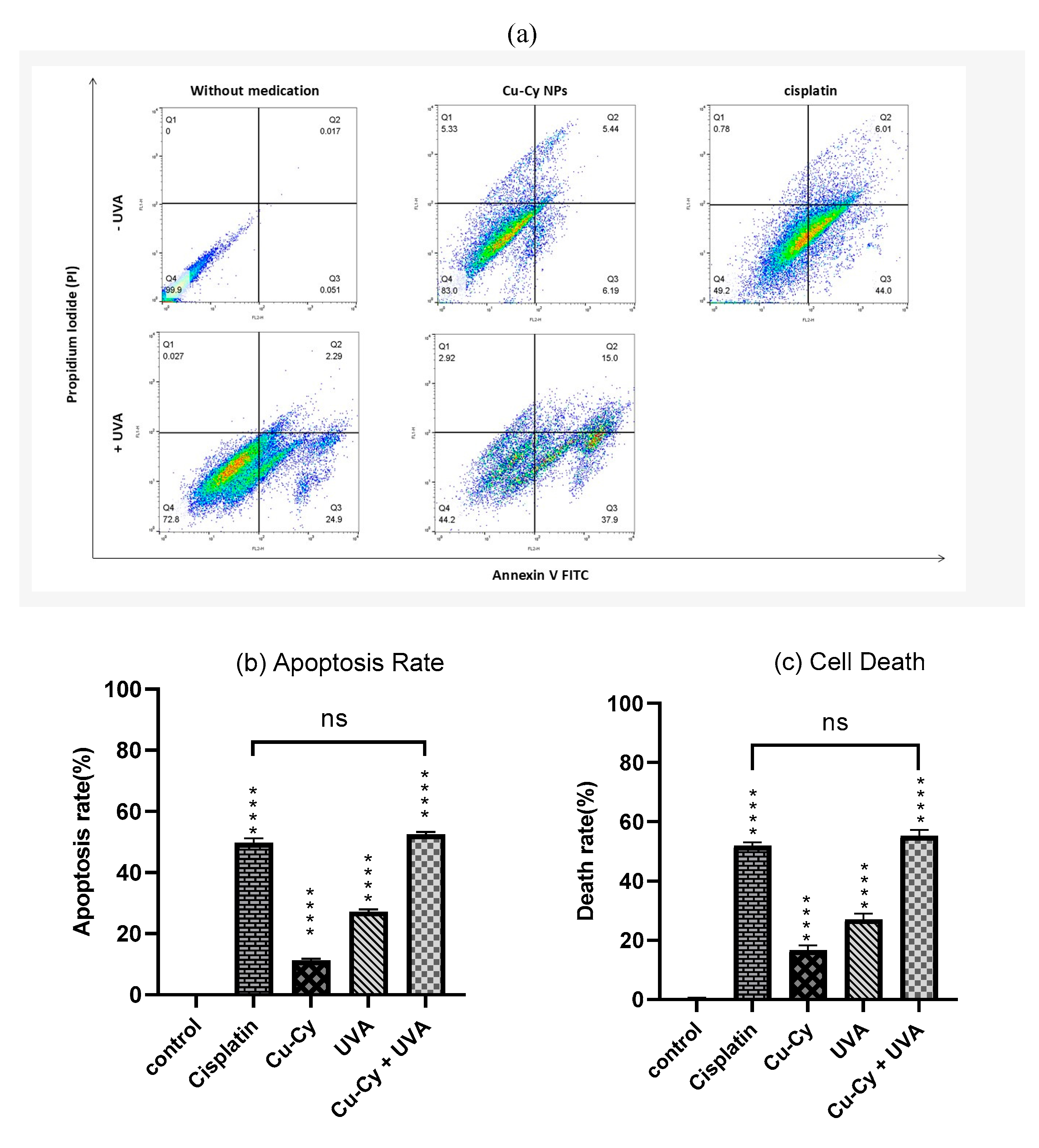
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- B. Ahmed, M.I. Qadir, S. Ghafoor, Malignant Melanoma: Skin Cancer-Diagnosis, Prevention, and Treatment, Crit. Rev. Eukaryot. Gene Expr. 30 (2020) 291–297. [CrossRef]
- M.F. Jojoa Acosta, L.Y. Caballero Tovar, M.B. Garcia-Zapirain, W.S. Percybrooks, Melanoma diagnosis using deep learning techniques on dermatoscopic images, BMC Med. Imaging 21 (2021) 6. [CrossRef]
- Y.-Y. Lin, C.-Y. Chen, D.-L. Ma, C.-H. Leung, C.-Y. Chang, H.-M.D. Wang, Cell-derived artificial nanovesicle as a drug delivery system for malignant melanoma treatment, Biomed. Pharmacother. 147 (2022) 112586. [CrossRef]
- L. Zeng, B.H.J. Gowda, M.G. Ahmed, M.A.S. Abourehab, Z.-S. Chen, C. Zhang, J. Li, P. Kesharwani, Advancements in nanoparticle-based treatment approaches for skin cancer therapy, Mol. Cancer 22 (2023) 10. [CrossRef]
- M.R. Hamblin, Photodynamic Therapy for Cancer: What’s Past is Prologue, Photochem. Photobiol. 96 (2020) 506–516. [CrossRef]
- C.-N. Lee, R. Hsu, H. Chen, T.-W. Wong, Daylight Photodynamic Therapy: An Update., Molecules 25 (2020). [CrossRef]
- B. Halliwell, Free radicals and antioxidants - quo vadis?, Trends Pharmacol. Sci. 32 (2011) 125–130. [CrossRef]
- G.M.F. Calixto, J. Bernegossi, L.M. de Freitas, C.R. Fontana, M. Chorilli, Nanotechnology-Based Drug Delivery Systems for Photodynamic Therapy of Cancer: A Review., Molecules 21 (2016) 342. [CrossRef]
- I. Yoon, J.Z. Li, Y.K. Shim, Advance in photosensitizers and light delivery for photodynamic therapy., Clin. Endosc. 46 (2013) 7–23. [CrossRef]
- B.C. Wilson, Photodynamic therapy for cancer: principles., Can. J. Gastroenterol. 16 (2002) 393–396. [CrossRef]
- K. Kalka, H. Merk, H. Mukhtar, Photodynamic therapy in dermatology, J. Am. Acad. Dermatol. 42 (2000) 389–413. [CrossRef]
- H. Abrahamse, M.R. Hamblin, New photosensitizers for photodynamic therapy, Biochem. J. 473 (2016) 347–364. [CrossRef]
- J.C.S. Simões, S. Sarpaki, P. Papadimitroulas, B. Therrien, G. Loudos, Conjugated Photosensitizers for Imaging and PDT in Cancer Research, J. Med. Chem. 63 (2020) 14119–14150. [CrossRef]
- X. Huang, F. Wan, L. Ma, J.B. Phan, R.X. Lim, C. Li, J. Chen, J. Deng, Y. Li, W. Chen, M. He, Investigation of copper-cysteamine nanoparticles as a new photosensitizer for anti-hepatocellular carcinoma, Cancer Biol. Ther. 20 (2019) 812–825. [CrossRef]
- S. Shrestha, J. Wu, B. Sah, A. Vanasse, L.N. Cooper, L. Ma, G. Li, H. Zheng, W. Chen, M.P. Antosh, X-ray induced photodynamic therapy with copper-cysteamine nanoparticles in mice tumors, Proc. Natl. Acad. Sci. 116 (2019) 16823–16828. [CrossRef]
- M. Yao, L. Ma, L. Li, J. Zhang, R.X. Lim, W. Chen, Y. Zhang, A New Modality for Cancer Treatment—Nanoparticle Mediated Microwave Induced Photodynamic Therapy, J. Biomed. Nanotechnol. 12 (2016) 1835–1851. [CrossRef]
- P. Wang, X. Wang, L. Ma, S. Sahi, L. Li, X. Wang, Q. Wang, Y. Chen, W. Chen, Q. Liu, Nanosonosensitization by Using Copper–Cysteamine Nanoparticles Augmented Sonodynamic Cancer Treatment, Part. Part. Syst. Charact. 35 (2018) 1700378. [CrossRef]
- L. Chudal, N.K. Pandey, J. Phan, O. Johnson, L. Lin, H. Yu, Y. Shu, Z. Huang, M. Xing, J.P. Liu, M.-L. Chen, W. Chen, Copper-Cysteamine Nanoparticles as a Heterogeneous Fenton-Like Catalyst for Highly Selective Cancer Treatment, ACS Appl. Bio Mater. 3 (2020) 1804–1814. [CrossRef]
- Q. Zhang, X. Guo, Y. Cheng, L. Chudal, N.K. Pandey, J. Zhang, L. Ma, Q. Xi, G. Yang, Y. Chen, X. Ran, C. Wang, J. Zhao, Y. Li, L. Liu, Z. Yao, W. Chen, Y. Ran, R. Zhang, Use of copper-cysteamine nanoparticles to simultaneously enable radiotherapy, oxidative therapy and immunotherapy for melanoma treatment, Signal Transduct. Target. Ther. 5 (2020) 58. [CrossRef]
- N.R. York, H.T. Jacobe, UVA1 phototherapy: a review of mechanism and therapeutic application., Int. J. Dermatol. 49 (2010) 623–630. [CrossRef]
- N.K. Pandey, L. Chudal, J. Phan, L. Lin, O. Johnson, M. Xing, J.P. Liu, H. Li, X. Huang, Y. Shu, W. Chen, A facile method for the synthesis of copper–cysteamine nanoparticles and study of ROS production for cancer treatment, J. Mater. Chem. B 7 (2019) 6630–6642. [CrossRef]
- M. Ejtema, N. Chegeni, A. Zarei-Ahmady, Z. Salehnia, M. Shamsi, S. Razmjoo, Exploring the combined impact of cisplatin and copper-cysteamine nanoparticles through Chemoradiation: An in-vitro study, Toxicol. Vitr. 99 (2024) 105878. [CrossRef]
- M. Kolarikova, B. Hosikova, H. Dilenko, K. Barton-Tomankova, L. Valkova, R. Bajgar, L. Malina, H. Kolarova, Photodynamic therapy: Innovative approaches for antibacterial and anticancer treatments., Med. Res. Rev. 43 (2023) 717–774. [CrossRef]
- B.W. Henderson, T.J. Dougherty, How does photodynamic therapy work?, Photochem. Photobiol. 55 (1992) 145–157. [CrossRef]
- Z. Liu, L. Xiong, G. Ouyang, L. Ma, S. Sahi, K. Wang, L. Lin, H. Huang, X. Miao, W. Chen, Y. Wen, Investigation of Copper Cysteamine Nanoparticles as a New Type of Radiosensitiers for Colorectal Carcinoma Treatment, Sci. Rep. 7 (2017) 9290. [CrossRef]
- T.L. de Jager, A.E. Cockrell, S.S. Du Plessis, Ultraviolet Light Induced Generation of Reactive Oxygen Species, in: Ultrav. Light Hum. Heal. Dis. Environ., Springer, 2017: pp. 15–23. [CrossRef]
- C.-H. Lee, S.-B. Wu, C.-H. Hong, H.-S. Yu, Y.-H. Wei, Molecular Mechanisms of UV-Induced Apoptosis and Its Effects on Skin Residential Cells: The Implication in UV-Based Phototherapy, Int. J. Mol. Sci. 14 (2013) 6414–6435. [CrossRef]
- Z. He, L. Zhang, C. Zhuo, F. Jin, Y. Wang, Apoptosis inhibition effect of Dihydromyricetin against UVA-exposed human keratinocyte cell line, J. Photochem. Photobiol. B Biol. 161 (2016) 40–49. [CrossRef]
- V. Carneiro Leite, R. Ferreira Santos, L. Chen Chen, L. Andreu Guillo, Psoralen derivatives and longwave ultraviolet irradiation are active in vitro against human melanoma cell line, J. Photochem. Photobiol. B Biol. 76 (2004) 49–53. [CrossRef]
- M.T. Bilkan, Z. Çiçek, A.G.C. Kurşun, M. Özler, M.A. Eşmekaya, Investigations on effects of titanium dioxide (TiO2) nanoparticle in combination with UV radiation on breast and skin cancer cells, Med. Oncol. 40 (2022) 60. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



