Submitted:
10 September 2025
Posted:
11 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients and Materials
2.2. Histological Analysis
2.3. Immunohistochemstry (IHC)
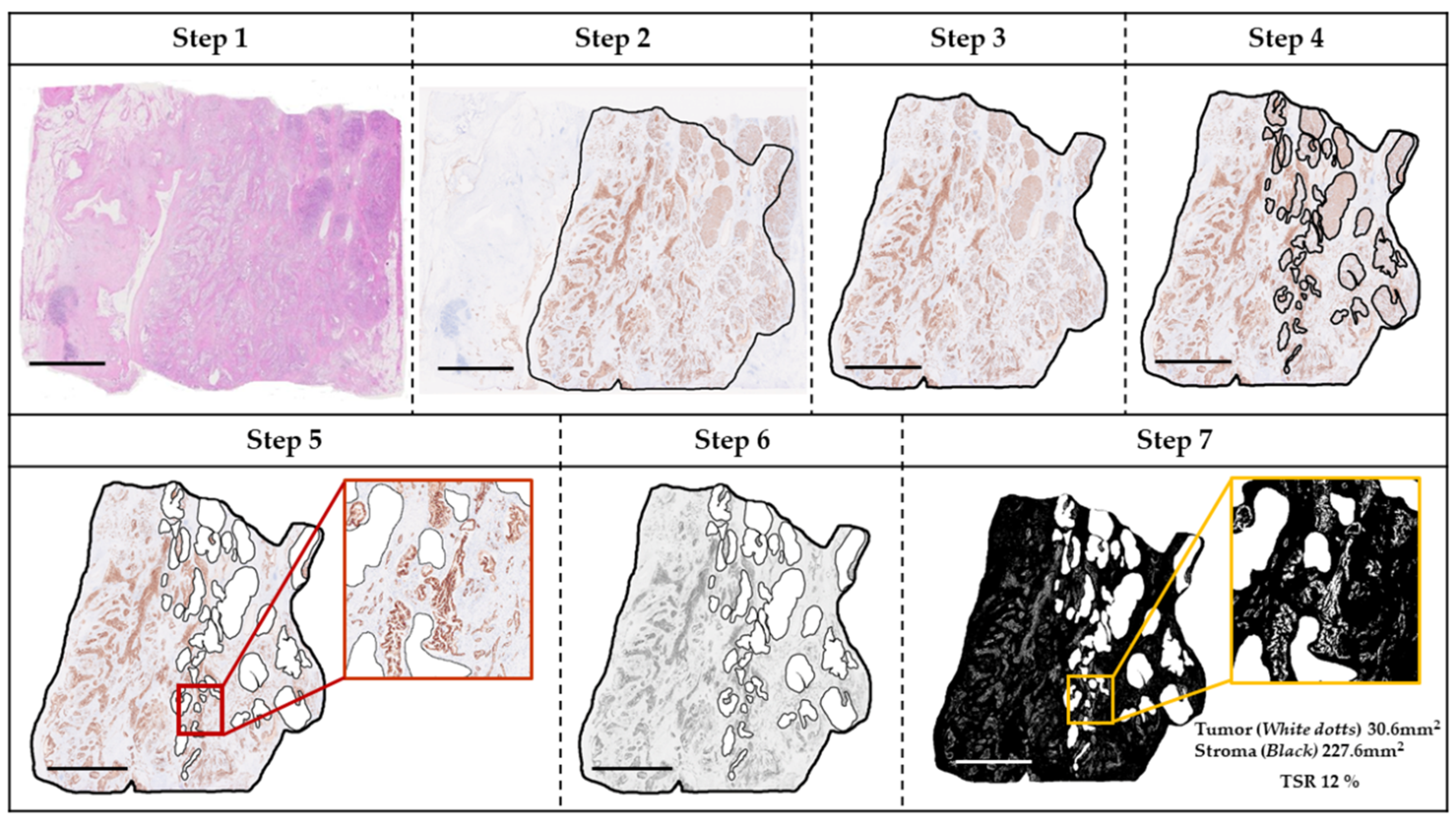
2.4. TSR
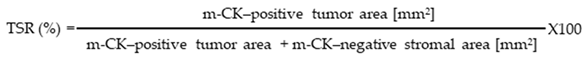
2.5. Assessment of β-CTN IHC

2.6. TB
2.7. Statistical Analysis
3. Results
3.1. Clinicopathological Differences Between NAC and UFS
3.2. β-CTN and m-CK IHC
3.3. Baseline Values of TSR, β-CTN/m-CK Index, and TB
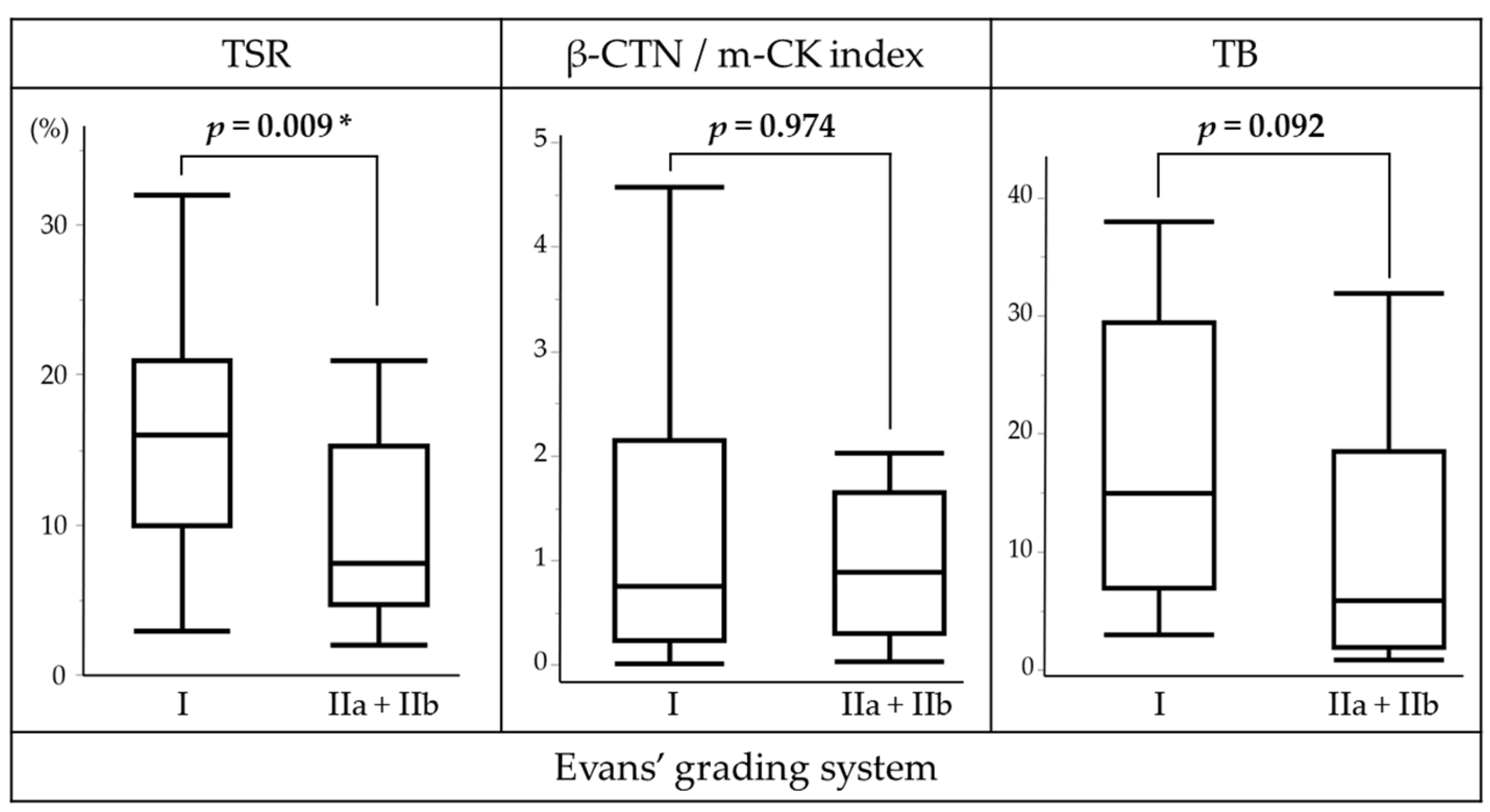
3.4. Association of TSR with Clinicopathological Features
3.5. Association of β-CTN/m-CK Index with Clinicopathological Features
3.6. Association of TB with Clinicopathological Features
3.7. Interrelationships Among β-CTN/m-CK Index, TSR, and TB in PDAC
3.8. NAC Response in Relation to TSR, β-CTN/m-CK Index, and TB
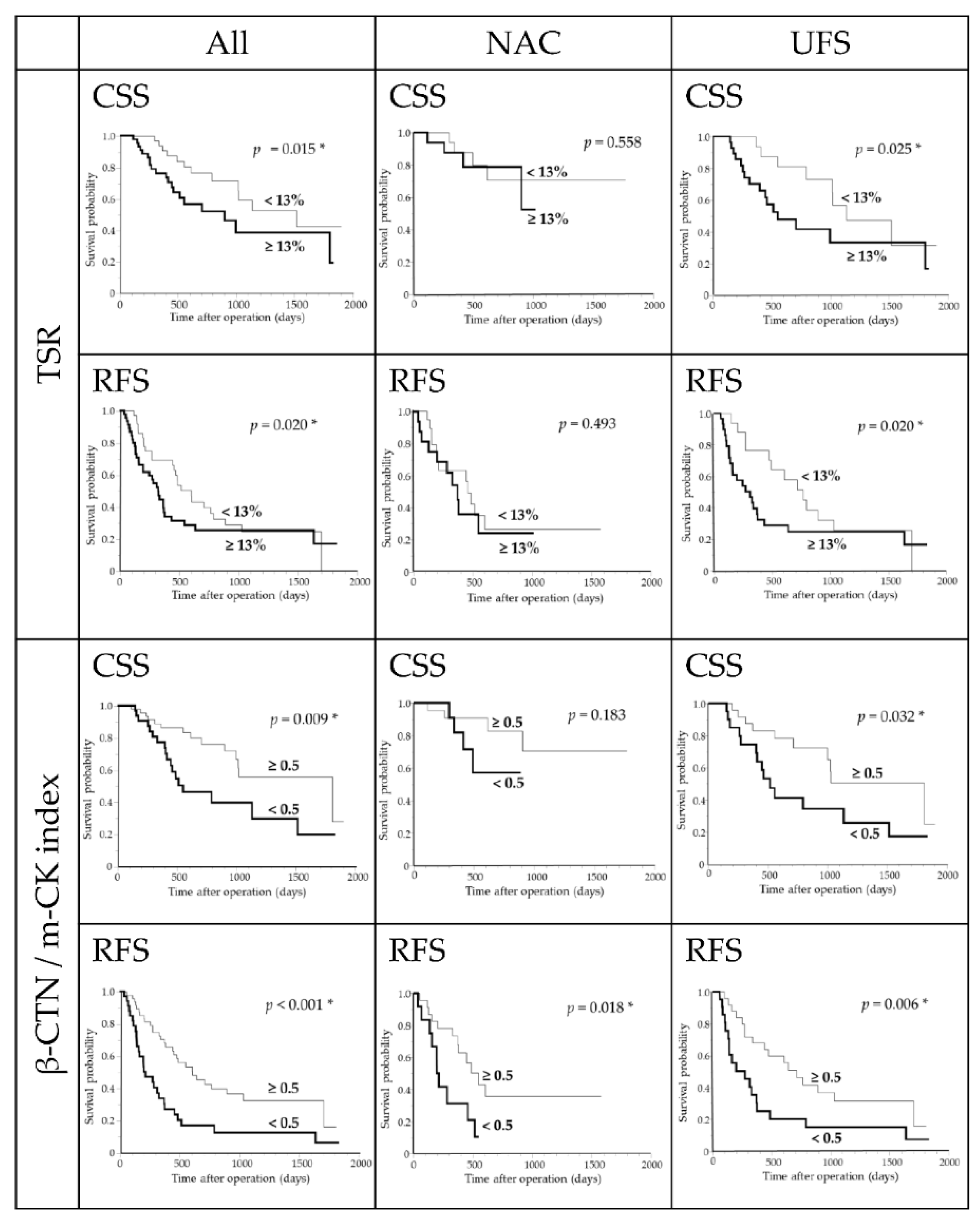
3.9. Survival Analysis in the Overall Cohort
3.10. Survival Analysis in the NAC Subgroup
3.11. Survival Analysis in the UFS Subgroup
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| β-CTN | β-catenin |
| CI | confidence interval |
| CSS | cancer-specific survival |
| IHC | Immunohistochemistry |
| m-CK | multi-cytokeratin |
| NAC | neoadjuvant chemotherapy |
| PC | pancreatic cancer |
| PDAC | pancreatic ductal adenocarcinoma |
| RFS | relapse-free survival |
| TB | tumor budding |
| TSR | tumor–stroma ratio |
| UFS | upfront surgery |
| WSI | whole-slide image |
References
- Zhao, Z.; Liu, W. Pancreatic cancer: A review of risk factors, diagnosis, and treatment. Technol. Cancer Res. Treat. 2020, 19, 1533033820962117.
- Zottl, J.; Sebesta, C.G.; Tomosel, E.; Sebesta, M.C.; Sebesta, C. Unraveling the burden of pancreatic cancer in the 21st century: Trends in incidence, mortality, survival, and key contributing factors. Cancers (Basel) 2025, 17, 1607. [CrossRef]
- Klein, A.P. Pancreatic cancer epidemiology: Understanding the role of lifestyle and inherited risk factors. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 493–502. [CrossRef]
- Ei, S.; Takahashi, S.; Ogasawara, T.; Mashiko, T.; Masuoka, Y.; Nakagohri, T. Neoadjuvant and adjuvant treatments for resectable and borderline resectable pancreatic ductal adenocarcinoma: The current status of pancreatic ductal adenocarcinoma treatment in Japan. Gut Liver 2023, 17, 698–710. [CrossRef]
- Ye, M.; Zhang, Q.; Chen, Y.; Fu, Q.; Li, X.; Bai, X.; Liang, T. Neoadjuvant chemotherapy for primary resectable pancreatic cancer: A systematic review and meta-analysis. HPB (Oxford) 2020, 22, 821–832. [CrossRef]
- Sugawara, T.; Rodriguez Franco, S.; Sherman, S.; Torphy, R.J.; Colborn, K.; Franklin, O.; Ishida, J.; Grandi, S.; Al-Musawi, M.H.; Gleisner, A.; et al. Neoadjuvant chemotherapy versus upfront surgery for resectable pancreatic adenocarcinoma: An updated nationwide study. Ann. Surg. 2024, 279, 331–339.
- Sudo, K.; Nakamura, K.; Yamaguchi, T. S-1 in the treatment of pancreatic cancer. World J. Gastroenterol. 2014, 20, 15110–15118.
- Unno, M.; Motoi, F.; Matsuyama, Y.; Satoi, S.; Toyama, H.; Matsumoto, I.; Aosasa, S.; Shirakawa, H.; Wada, K.; Fujii, T.; et al. Neoadjuvant chemotherapy with gemcitabine and S-1 versus upfront surgery for resectable pancreatic cancer: Results of the randomized phase II/III Prep-02/JSAP05 trial. Ann. Surg. 2025. Online ahead of print.
- Okusaka, T.; Nakamura, M.; Yoshida, M.; Kitano, M.; Ito, Y.; Mizuno, N.; et al. Clinical practice guidelines for pancreatic cancer 2022 from the Japan Pancreas Society: A synopsis. Int. J. Clin. Oncol. 2023, 28, 493–511. [CrossRef]
- Winther, S.B.; Bjerregaard, J.K.; Schonnemann, K.R.; Ejlsmark, M.W.; Krogh, M.; Jensen, H.A.; Pfeiffer, P. S-1 (Teysuno) and gemcitabine in Caucasian patients with unresectable pancreatic adenocarcinoma. Cancer Chemother. Pharmacol. 2018, 81, 573–578. [CrossRef]
- McDonald, O.G. The biology of pancreatic cancer morphology. Pathology 2022, 54, 236–247. [CrossRef]
- biophysics in pancreatic ductal adenocarcinoma: Can we learn from breast cancer? Pancreas 2020, 49, 313–325.
- Pyo, J.S.; Kim, N.Y.; Min, K.W.; Kang, D.W. Significance of tumor–stroma ratio (TSR) in predicting outcomes of malignant tumors. Medicina (Kaunas) 2023, 59, 1258. [CrossRef]
- Chen, X.; Sun, S.; Zhao, J.; Yu, S.; Chen, J.; Chen, X. Tumor–stroma ratio combined with PD-L1 identifies pancreatic ductal adenocarcinoma patients at risk for lymph node metastases. Br. J. Cancer 2025, 132, 1131–1140. [CrossRef]
- Liao, H.; Yuan, J.; Liu, C.; Zhang, J.; Yang, Y.; Liang, H.; Jiang, S.; Chen, S.; Li, Y.; Liu, Y.; et al. Feasibility and effectiveness of automatic deep learning network and radiomics models for differentiating tumor–stroma ratio in pancreatic ductal adenocarcinoma. Insights Imaging 2023, 14, 223. [CrossRef]
- Lu, M.; Zou, Y.; Fu, P.; Li, Y.; Wang, P.; Li, G.; Luo, S.; Chen, Y.; Guan, G.; Zhang, S.; et al. The tumor–stroma ratio and the immune microenvironment improve the prognostic prediction of pancreatic ductal adenocarcinoma. Discov. Oncol. 2023, 14, 124. [CrossRef]
- Meng, Y.; Zhang, H.; Li, Q.; Liu, F.; Fang, X.; Li, J.; Yu, J.; Feng, X.; Lu, J.; Bian, Y.; et al. Magnetic resonance radiomics and machine-learning models: An approach for evaluating tumor–stroma ratio in patients with pancreatic ductal adenocarcinoma. Acad. Radiol. 2022, 29, 523–535. [CrossRef]
- Kawahara, K.; Takano, S.; Furukawa, K.; Takayashiki, T.; Kuboki, S.; Ohtsuka, M. The effect of the low stromal ratio induced by neoadjuvant chemotherapy on recurrence patterns in borderline resectable pancreatic ductal adenocarcinoma. Clin. Exp. Metastasis 2022, 39, 311–322. [CrossRef]
- Li, B.; Wang, Y.; Jiang, H.; Li, B.; Shi, X.; Gao, S.; Ni, C.; Zhang, Z.; Guo, S.; Xu, J.; et al. Pros and cons: High proportion of stromal component indicates better prognosis in patients with pancreatic ductal adenocarcinoma—A research based on the evaluation of whole-mount histological slides. Front. Oncol. 2020, 10, 1472. [CrossRef]
- Leppänen, J.; Lindholm, V.; Isohookana, J.; Haapasaari, K.M.; Karihtala, P.; Lehenkari, P.P.; Saarnio, J.; Kauppila, J.H.; Karttunen, T.J.; Helminen, O.; et al. Tenascin C, fibronectin, and tumor–stroma ratio in pancreatic ductal adenocarcinoma. Pancreas 2019, 48, 43–48. [CrossRef]
- Matsuda, Y.; Inoue, Y.; Hiratsuka, M.; Kawakatsu, S.; Arai, T.; Matsueda, K.; Saiura, A.; Takazawa, Y. Encapsulating fibrosis following neoadjuvant chemotherapy is correlated with outcomes in patients with pancreatic cancer. PLoS One 2019, 14, e0222155. [CrossRef]
- Koay, E.J.; Lee, Y.; Cristini, V.; Lowengrub, J.S.; Kang, Y.; Lucas, F.A.S.; Hobbs, B.P.; Ye, R.; Elganainy, D.; Almahariq, M.; et al. Visually apparent quantifiable CT imaging and feature identifies biophysical subtypes of pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2018, 24, 5883–5894. [CrossRef]
- Vendittelli, P.; Bokhorst, J.M.; Smeets, E.M.M.; Kryklyva, V.; Brosens, L.A.A.; Verbeke, C.; Litjens, G. Automatic quantification of tumor–stroma ratio as a prognostic marker for pancreatic cancer. PLoS One 2024, 19, e0301969. [CrossRef]
- Shi, S.; Liang, C.; Xu, J.; Meng, Q.; Hua, J.; Yang, X.; et al. The strain ratio as obtained by endoscopic ultrasonography elastography correlates with the stroma proportion and the prognosis of local pancreatic cancer. Ann. Surg. 2020, 271, 559–565. [CrossRef]
- Bever, K.M.; Sugar, E.A.; Bigelow, E.; Sharma, R.; Laheru, D.; Wolfgang, C.L.; Jaffee, E.M.; Anders, R.A.; De Jesus-Acosta, A.; Zheng, L.; et al. The prognostic value of stroma in pancreatic cancer in patients receiving adjuvant therapy. HPB (Oxford) 2015, 17, 292–298. [CrossRef]
- Liang, C.; Shi, S.; Meng, Q.; Liang, D.; Ji, S.; Zhang, B.; Qin, Y.; Xu, J.; Ni, Q.; Yu, X.; et al. Complex roles of the stroma in the intrinsic resistance to gemcitabine in pancreatic cancer: Where we are and where we are going. Exp. Mol. Med. 2017, 49, e406. [CrossRef]
- Koay, E.J.; Truty, M.J.; Cristini, V.; Thomas, R.M.; Chen, R.; Chatterjee, D.; Kang, Y.; Bhosale, P.R.; Tamm, E.P.; Crane, C.H.; et al. Transport properties of pancreatic cancer describe gemcitabine delivery and response. J. Clin. Invest. 2014, 124, 1525–1536. [CrossRef]
- Olive, K.P.; Jacobetz, M.A.; Davidson, C.J.; et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 2009, 324, 1457–1461. [CrossRef]
- Suenaga, M.; Yamada, S.; Fujii, T.; Tanaka, C.; Kanda, M.; Nakayama, G.; Sugimoto, H.; Koike, M.; Fujiwara, M.; Kodera, Y. S-1 plus nab-paclitaxel is a promising regimen for pancreatic cancer in a preclinical model. J. Surg. Oncol. 2016, 113, 413–419. [CrossRef]
- Morimoto, Y.; Takada, K.; Nakano, A.; Takeuchi, O.; Watanabe, K.; Hirohara, M.; Masuda, Y. Combination of S-1 and the oral ATR inhibitor ceralasertib is effective against pancreatic cancer cells. Cancer Chemother. Pharmacol. 2024, 94, 763–774. [CrossRef]
- Lin, W.H.; Cooper, L.M.; Anastasiadis, P.Z. Cadherins and catenins in cancer: Connecting cancer pathways and tumor microenvironment. Front. Cell Dev. Biol. 2023, 11, 1137013. [CrossRef]
- Borcherding, N.; Cole, K.; Kluz, P.; Jorgensen, M.; Kolb, R.; Bellizzi, A.; Zhang, W. Re-evaluating E-cadherin and β-catenin: A pan-cancer proteomic approach with an emphasis on breast cancer. Am. J. Pathol. 2018, 188, 1910–1920.
- Rosova, B.; Proks, J.; Filipova, A.; Hadzi Nikolov, D.; Chloupkova, R.; Richter, I.; Szabo, A.; Rozsypalova, A.; Matej, R.; Melichar, B.; et al. Favorable prognostic significance of membranous β-catenin expression and negative prognostic significance of cytoplasmic β-catenin expression in pancreatic cancer. Neoplasma 2023, 70, 796–803. [CrossRef]
- Eurola, A.; Ristimäki, A.; Mustonen, H.; Nurmi, A.M.; Hagström, J.; Kallio, P.; Alitalo, K.; Haglund, C.; Seppänen, H. β-catenin plus PROX1 immunostaining stratifies disease progression and patient survival in neoadjuvant-treated pancreatic cancer. Tumour Biol. 2022, 44, 69–84. [CrossRef]
- Saukkonen, K.; Hagström, J.; Mustonen, H.; Juuti, A.; Nordling, S.; Kallio, P.; Alitalo, K.; Seppänen, H.; Haglund, C. PROX1 and β-catenin are prognostic markers in pancreatic ductal adenocarcinoma. BMC Cancer 2016, 16, 472. [CrossRef]
- Wang, Z.; Ma, Q.; Li, P.; Sha, H.; Li, X.; Xu, J. Aberrant expression of CXCR4 and β-catenin in pancreatic cancer. Anticancer Res. 2013, 33, 4103–4110.
- Zhong, R.L.; Li, Y.; Fang, Z.; Fang, K.F.; Wang, L. PKR2 and β-catenin genes regulates pancreatic cancer chemosensitivity. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 48–54.
- Eurola, A.; Ristimäki, A.; Mustonen, H.; Nurmi, A.M.; Hagström, J.; Haglund, C.; Seppänen, H. Impact of histological response after neoadjuvant therapy on podocalyxin as a prognostic marker in pancreatic cancer. Sci. Rep. 2021, 11, 9896. [CrossRef]
- Kokumai, T.; Omori, Y.; Ishida, M.; Ohtsuka, H.; Mizuma, M.; Nakagawa, K.; Maeda, C.; Ono, Y.; Mizukami, Y.; Miura, S.; et al. GATA6 and CK5 stratify the survival of patients with pancreatic cancer undergoing neoadjuvant chemotherapy. Mod. Pathol. 2023, 36, 100102. [CrossRef]
- Lugli, A.; Zlobec, I.; Berger, M.D.; Kirsch, R.; Nagtegaal, I.D. Tumour budding in solid cancers. Nat. Rev. Clin. Oncol. 2021, 18, 101–115. [CrossRef]
- Lugli, A.; Kirsch, R.; Ajioka, Y.; Bosman, F.; Cathomas, G.; Dawson, H.; et al. Recommendations for reporting tumor budding in colorectal cancer based on the International Tumor Budding Consensus Conference (ITBCC) 2016. Mod. Pathol. 2017, 30, 1299–1311. [CrossRef]
- Kohler, I.; Bronsert, P.; Timme, S.; Werner, M.; Brabletz, T.; Hopt, U.T.; et al. Detailed analysis of epithelial–mesenchymal transition and tumor budding identifies predictors of long-term survival in pancreatic ductal adenocarcinoma. J. Gastroenterol. Hepatol. 2015, 30 (Suppl. 1), 78–84. [CrossRef]
- Zhou, T.; Man, Q.; Li, X.; Xie, Y.; Hou, X.; Wang, H.; et al. Artificial intelligence-based comprehensive analysis of immune-stemness-tumor budding profile to predict survival of patients with pancreatic adenocarcinoma. Cancer Biol. Med. 2023, 20, 196–217. [CrossRef]
- Hayasaki, A.; Mizuno, S.; Usui, M.; Kaluba, B.; Komatsubara, H.; Sakamoto, T.; et al. Tumor budding is an independent adverse prognostic factor of pancreatic ductal adenocarcinoma patients treated by resection after preoperative chemoradiotherapy. Pancreas 2025, 54, e340–e348. [CrossRef]
- Ibuki, E.; Kadota, K.; Kimura, N.; Ishikawa, R.; Oshima, M.; Okano, K.; et al. Prognostic significance of tumor budding in patients with pancreatic invasive ductal carcinoma who received neoadjuvant therapy. Heliyon 2023, 10, e23928. [CrossRef]
- Argon, A.; Öz, Ö.; Kebat, T.A. Evaluation and prognostic significance of tumor budding in pancreatic ductal adenocarcinomas. Indian J. Pathol. Microbiol. 2023, 66, 38–43. [CrossRef]
- Karamitopoulou, E.; Wartenberg, M.; Zlobec, I.; Cibin, S.; Worni, M.; Gloor, B.; et al. Tumour budding in pancreatic cancer revisited: Validation of the ITBCC scoring system. Histopathology 2018, 73, 137–146. [CrossRef]
- Lohneis, P.; Sinn, M.; Klein, F.; Bischoff, S.; Striefler, J.K.; Wislocka, L.; et al. Tumour buds determine prognosis in resected pancreatic ductal adenocarcinoma. Br. J. Cancer 2018, 118, 1485–1491. [CrossRef]
- Oba, A.; Del Chiaro, M.; Fujii, T.; Okano, K.; Stoop, T.F.; Wu, Y.H.A.; et al. “Conversion surgery” for locally advanced pancreatic cancer: A position paper by the study group at the joint meeting of the International Association of Pancreatology (IAP) & Japan Pancreas Society (JPS) 2022. Pancreatology 2023, 23, 712–720. [CrossRef]
- Isaji, S.; Mizuno, S.; Windsor, J.A.; Bassi, C.; Fernández-Del Castillo, C.; Hackert, T.; et al. International consensus on definition and criteria of borderline resectable pancreatic ductal adenocarcinoma 2017. Pancreatology 2018, 18, 2–11. [CrossRef]
- Evans, D.B.; Rich, T.A.; Byrd, D.R.; Cleary, K.R.; Connelly, J.H.; Levin, B.; Charnsangavej, C.; Fenoglio, C.J.; Ames, F.C. Preoperative chemoradiation and pancreaticoduodenectomy for adenocarcinoma of the pancreas. Arch. Surg. 1992, 127, 1335–1339. [CrossRef]
- Neumann, C.C.M.; von Hörschelmann, E.; Reutzel-Selke, A.; Seidel, E.; Sauer, I.M.; Pratschke, J.; Bahra, M.; Schmuck, R.B. Tumor–stromal cross-talk modulating the therapeutic response in pancreatic cancer. Hepatobiliary Pancreat. Dis. Int. 2018, 17, 461–472. [CrossRef]
- Gore, J.; Korc, M. Pancreatic cancer stroma: Friend or foe? Cancer Cell 2014, 25, 711–712. [CrossRef]
- Chen, Y.; Kim, J.; Yang, S.; Wang, H.; Wu, C.J.; Sugimoto, H.; LeBleu, V.S.; Kalluri, R. Type I collagen deletion in αSMA+ myofibroblasts augments immune suppression and accelerates progression of pancreatic cancer. Cancer Cell 2021, 39, 548–565.e6. [CrossRef]
- Kim, S.W.; Roh, J.; Park, C.S. Immunohistochemistry for pathologists: Protocols, pitfalls, and tips. J. Pathol. Transl. Med. 2016, 50, 411–418. [CrossRef]
- Kubota, Y.; Kawakami, H.; Natsuizaka, M.; Kawakubo, K.; Marukawa, K.; Kudo, T.; Abe, Y.; Kubo, K.; Kuwatani, M.; Hatanaka, Y.; et al. CTNNB1 mutational analysis of solid-pseudopapillary neoplasms of the pancreas using endoscopic ultrasound-guided fine-needle aspiration and next-generation deep sequencing. J. Gastroenterol. 2015, 50, 203–210. [CrossRef]
- Dolled-Filhart, M.; McCabe, A.; Giltnane, J.; Cregger, M.; Camp, R.L.; Rimm, D.L. Quantitative in situ analysis of β-catenin expression in breast cancer shows decreased expression is associated with poor outcome. Cancer Res. 2006, 66, 5487–5494. [CrossRef]
- Helms, E.; Onate, M.K.; Sherman, M.H. Fibroblast heterogeneity in the pancreatic tumor microenvironment. Cancer Discov. 2020, 10, 648–656. [CrossRef]

| Variable | NAC | UFS | p-Value |
| Number of cases | 35 | 49 | |
| Age (years) | |||
| Mean / Median (range) | 69.5 / 71 (39-85) | 74.8 / 75 (50-91) | 0.040 * |
| <75 | 22 | 24 | 0.268 |
| ≥75 | 13 | 25 | |
| Sex | 0.664 | ||
| Male | 19 | 24 | |
| Female | 16 | 25 | |
| Tumor location | 1.000 | ||
| Head | 21 | 29 | |
| Body / Tail | 14 | 20 | |
| Adjuvant therapy | 0.264 | ||
| Yes | 23 | 25 | |
| No | 12 | 24 | |
| pT category | 0.060 | ||
| 1 | 13 | 10 | |
| 2 | 13 | 31 | |
| 3 | 9 | 8 | |
| pN category | 0.564 | ||
| N0 | 18 | 21 | |
| N1 | 11 | 15 | |
| N2 | 6 | 13 | |
| Tumor differentiation | 0.787 | ||
| Well | 14 | 23 | |
| Moderate | 18 | 23 | |
| Poor | 3 | 3 | |
| Lymphovascular invasion | 0.468 | ||
| Absent | 8 | 15 | |
| Present | 27 | 34 | |
| Perineural invasion | 0.199 | ||
| Absent | 11 | 9 | |
| Present | 24 | 40 | |
| Peripancreatic fat invasion | 0.409 | ||
| Absent | 9 | 8 | |
| Present | 26 | 41 | |
| Resection margin | 0.067 | ||
| Negative | 33 | 39 | |
| Positive | 2 | 10 | |
| Variable | CSS | RFS | ||||
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Adjuvant therapy (Yes vs No) | 5.395 | 2.233 - 13.031 | <0.001 * | 2.210 | 1.230 - 3.972 | 0.008 * |
| pT category (1 + 2 vs 3) | N/A | N/A | N/A | 1.785 | 0.909 - 3.505 | 0.093 |
| pN category (N0 vs N1 + N2) | 1.477 | 0.651 - 3.350 | 0.350 | 1.953 | 1.093 - 3.492 | 0.024 * |
| Tumor differentiation (Well vs Moderate + Poor) | 7.776 | 2.832 - 21.349 | <0.001 * | 2.394 | 1.260 - 4.551 | 0.008 * |
| Lymphovascular invasion (Absent vs Present) | N/A | N/A | N/A | 2.546 | 1.218 - 5.321 | 0.013 * |
| Peripancreatic fat invasion (Absent vs Present) | 6.972 | 1.911 - 25.428 | 0.003 * | N/A | N/A | N/A |
| TSR (<13% vs ≥13%) | 2.414 | 1.071 - 5.439 | 0.034 * | N/A | N/A | N/A |
| β-CTN/m-CK index (≥0.5 vs <0.5) | N/A | N/A | N/A | 2.028 | 1.120 - 3.670 | 0.020 * |
| Variable | CSS | RFS | ||||
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Sex (Female vs Male) | 18.054 | 1.614 - 201.926 | 0.019 * | N/A | N/A | N/A |
| Resection margin (Negative vs Positive) | 63.446 | 2.871 - 1402.272 | 0.009 * | 4.033 | 0.817 - 19.922 | 0.087 |
| β-CTN/m-CK index (≥0.5 vs <0.5) | N/A | N/A | N/A | 2.516 | 1.031 - 6.138 | 0.043 * |
| TB (Low vs High) | 6.008 | 0.847 - 42.627 | 0.073 | N/A | N/A | N/A |
| Variable | CSS | RFS | ||||
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Adjuvant therapy (Yes vs No) | 5.539 | 1.882 - 16.306 | 0.002 * | 2.013 | 0.980 - 4.136 | 0.057 |
| Tumor differentiation (Well vs Moderate + Poor) | 7.632 | 2.471 - 23.575 | <0.001 * | 5.006 | 2.178 - 11.508 | <0.001 * |
| Peripancreatic fat invasion (Absent vs Present) | 4.933 | 1.104 - 22.033 | 0.037 * | N/A | N/A | N/A |
| TSR (<13% vs ≥13%) | 2.536 | 0.947 - 6.790 | 0.064 | N/A | N/A | N/A |
| β-CTN/m-CK index (≥0.5 vs <0.5) | N/A | N/A | N/A | 2.230 | 1.106 - 4.500 | 0.025 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





