Submitted:
01 September 2025
Posted:
02 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
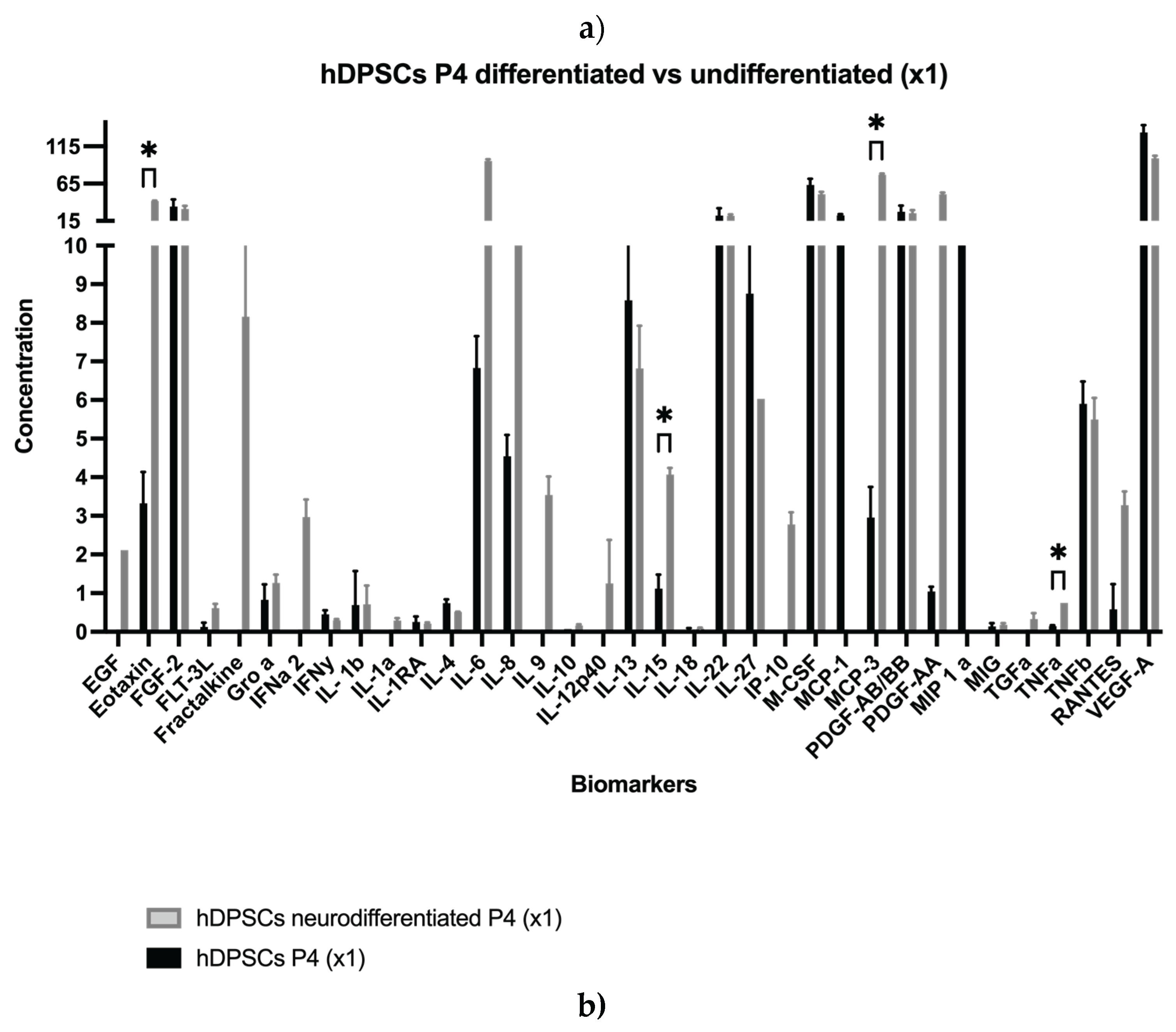
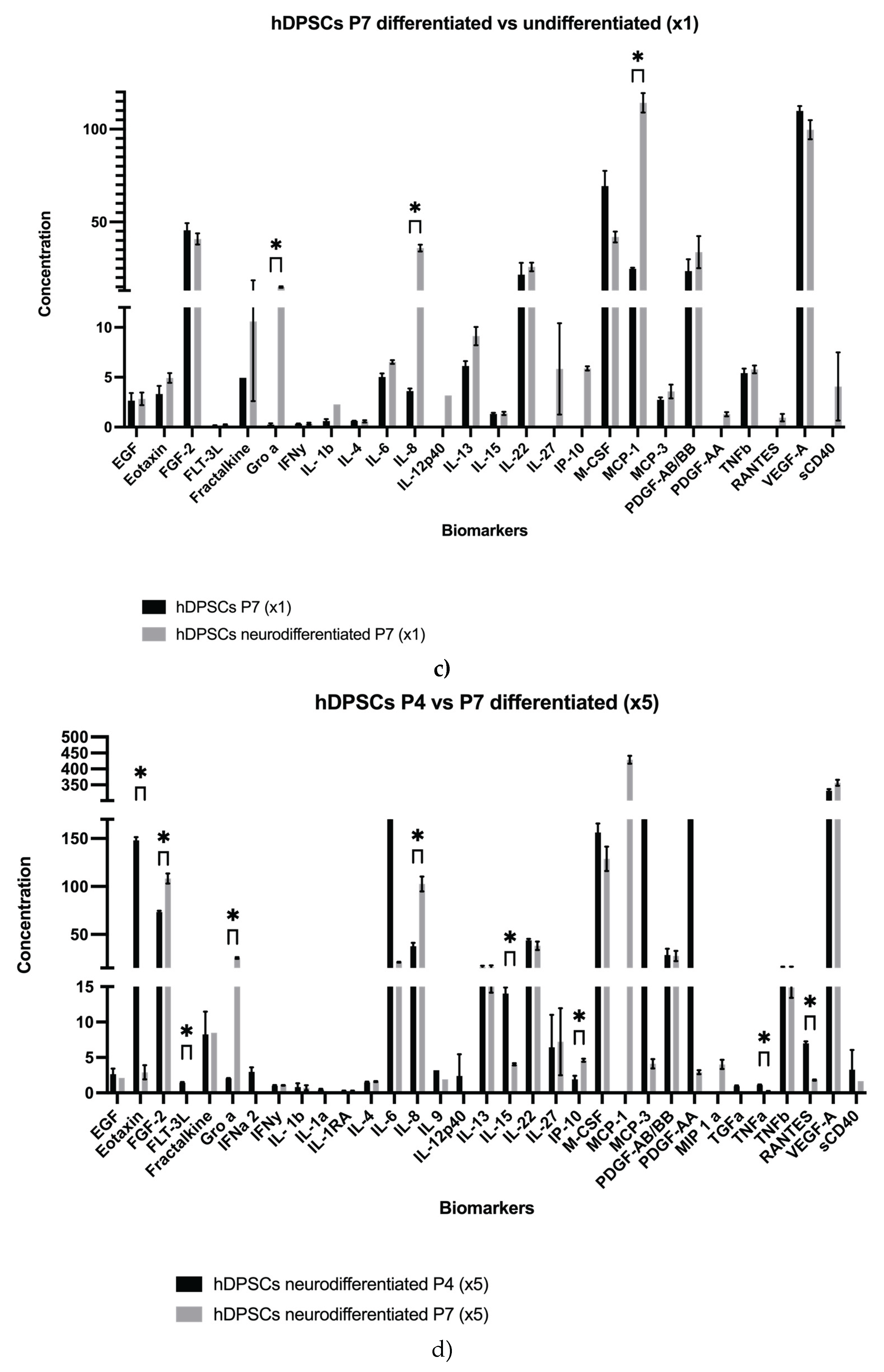
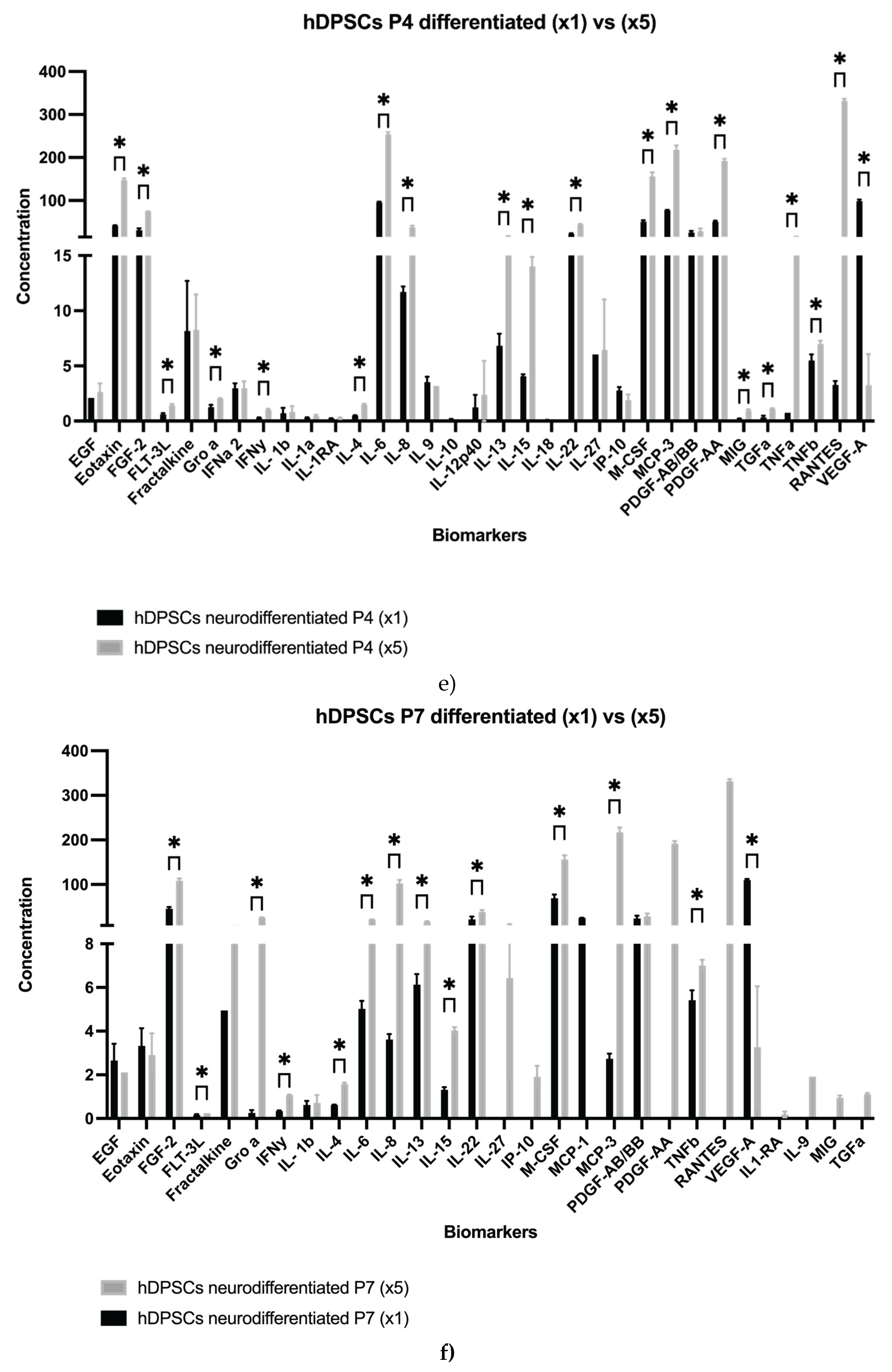
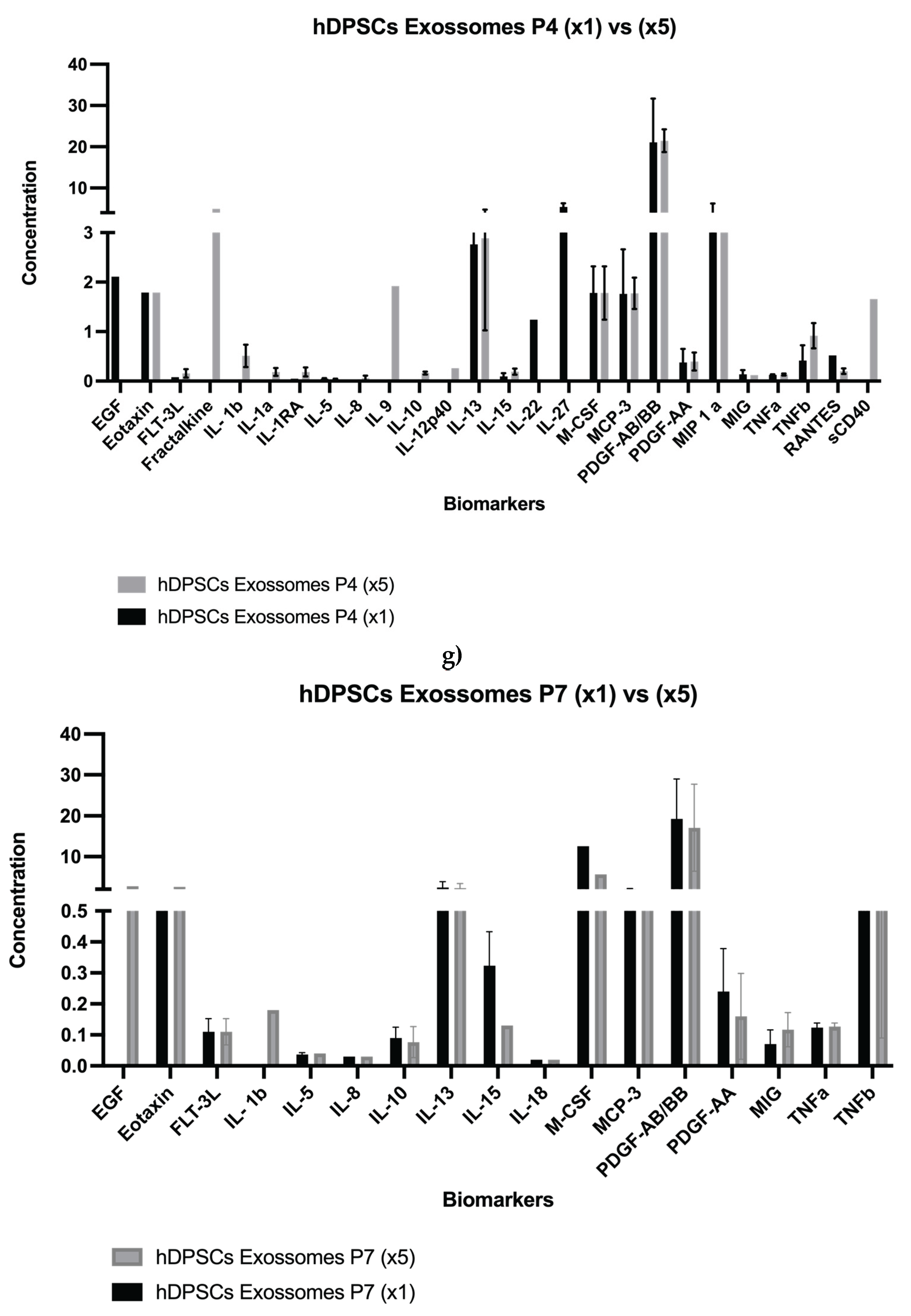
2.1. Analysis of hDPSCs Conditioned Medium and hDPSCs Exosomes
| Biomolecule | Mean ± SEM (P4) (x1) |
Mean ± SEM (P4) (x5) |
Mean ± SEM (P7) (x1) |
Mean ± SEM (P7) (x5) |
| EGF Eotaxin |
1.40 ± 0.00 41.66 ± 0.65 |
1.77 ± 0.77 148.06 ± 3.26 |
2.84 ± 0.63 4.93 ± 0.49 |
0.70 ± 0.00 2.90 ± 1.00 |
| FGF-2 | 31.24 ± 4.03 | 73.33 ± 1.42 | 40.83 ± 3.00 | 108.31 ± 5.20 |
| FLT-3L | 0.61 ± 0.11 | 1.42 ± 0.13 | 0.23 ± 0.06 | 0.25 ± 0.00 |
| Fractalkine Groα |
5.44 ±4.54 1.26 ±0.21 |
8.27 ± 3.21 2.03 ± 0.06 |
7.06 ± 7.98 14.93 ± 0.41 |
2.83 ± 0.00 25.46 ± 0.91 |
| IFN α 2 | 2.96 ±0.45 | 1.98 ± 0.64 | nd | nd |
| IFNγ | 0.3 ± 0.03 | 1.01 ± 0.09 | 0.35 ± 0.10 | 1.06 ± 0.03 |
| IL-1α IL-1b |
0.29 ± 0.06 0.71 ±0.49 |
0.46 ± 0.12 0.81 ± 0.55 |
nd 0.76 ± 0.00 |
nd 0.72 ± 0.36 |
| IL-1RA | 0.21 ± 0.03 | 0.25 ± 0.06 | nd | 0.19 ± 0.13 |
| IL-4 | 0.51 ± 0.01 | 1.48 ± 0.09 | 0.58 ± 0.11 | 1.58 ± 0.06 |
| IL-6 | 95.44 ± 2.04 | 254.07 ± 5.16 | 6.53 ± 0.19 | 21.07 ± 0.44 |
| IL-8 IL-9 |
11.7 ± 0.51 2.36 ±0.48 |
37.67 ± 3.48 2.13 ± 0.00 |
35.89 ± 1.83 nd |
102.60 ± 7.78 0.64 ± 0.00 |
| IL-10 | 0.16 ± 0.03 | nd | nd | nd |
| IL-12p40 | 1.25 ± 1.21 | 2.39 ± 3.06 | 2.11 ± 0.00 | nd |
| IL-13 | 6.82 ± 1.10 | 15.56 ± 1.74 | 9.13 ± 0.92 | 15.93 ± 1.77 |
| IL-15 IL-18 |
4.07 ± 0.17 0.08 ± 0.02 |
14.02 ± 0.87 nd |
1.37 ± 0.17 nd |
15.93 ± 1.77 nd |
| IL-22 IL-27 |
21.07 ± 2.09 2.01 ± 0.00 |
43.86 ± 1.52 4.29 ± 4.59 |
17.17 ± 2.32 5.83 ± 4.58 |
38.18 ± 4.45 7.21 ± 4.73 |
| IP-10 | 2.77 ± 0.31 | 1.91 ± 0.50 | 5.89 ± 0.21 | 4.60 ± 0.21 |
| M-CSF | 50.78 ± 3.15 | 156.36 ± 9.17 | 41.92 ± 2.91 | 128.81 ± 12.65 |
| MCP-1 | nd | nd | 114.2 ± 5.22 | 428.88 ± 12.34 |
| MCP-3 | 76.98 ± 1.59 | 217.32 ± 10.82 | 3.57 ± 0.69 | 4.11 ± 0.66 |
| MIG MIP PDGF-AA PDGF-AB/BB |
0.18 ± 0.05 nd 50.84 ± 2.28 25.45 ± 3.93 |
nd nd 192.09 ± 5.22 28.45 ± 6.69 |
nd nd 1.29 ± 0.21 33.69 ± 8.64 |
nd 4.02 ± 0.65 2.92 ± 0.28 27.52 ± 5.32 |
| TGFα | 0.22 ± 0.15 | nd | nd | 0.23 ± 0.04 |
| TNFα | 0.75 ± 0.00 | 1.11 ± 0.60 | nd | 14.90 ± 1.49 |
| TNFβ RANTES VEGF-A sCD40 |
5.49 ± 0.56 3.27 ± 0.35 98.89 ± 3.39 nd |
15.2 ± 1.30 7.00 ± 0.27 331.81 ± 4.57 3.27 ± 2.79 |
5.79 ± 0.39 0.95 ± 0.37 99.72 ± 5.17 2.72 ± 3.41 |
1.80 ± 0.07 356.41 ± 9.34 0.55 ± 0.00 nd |
| Biomolecule | Mean ± SEM (Exosomes P4) (x1) |
Mean ± SEM (Exosomes P4) (x5) |
Mean ± SEM (Exosomes P7) (x1) |
Mean ± SEM (Exosomes P7) (x5) |
| EGF Eotaxin |
0.70 ±0.00 0.60 ± 0.65 |
nd 0.60 ± 0.00 |
nd 0.41 ± 0.00 |
0.91 ± 0.00 0.86 ± 0.00 |
| FGF-2 | 0.08 ± 0.00 | nd | nd | Nd |
| FLT-3L | nd | 0.16 ± 0.09 | 0.07 ± 0.04 | 0.07 ± 0.04 |
| Fractalkine | nd | 3.3 ± 0.00 | nd | nd |
| IL-1α IL-1b |
nd nd |
0.12 ± 0.07 0.34 ± 0.23 |
nd nd |
nd 0.06 ± 0.00 |
| IL-1RA | 0.03 ± 0.00 | 0.12 ± 0.09 | nd | 0.19 ± 0.13 |
| IL-5 | 0.05 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.00 |
| IL-8 IL-9 |
0.01 ± 0.00 nd |
0.06 ± 0.05 0.64 ± 0.00 |
0.01 ± 0.00 nd |
0.01 ± 0.00 nd |
| IL-10 | 0.03 ± 0.00 | 0.16 ± 0.03 | 0.09 ± 0.03 | 0.08 ±0.05 |
| IL-12p40 | nd | 0.09 ± 0.00 | nd | nd |
| IL-13 | 2.76 ± 1.17 | 2.89 ± 1.86 | 2.44 ± 1.42 | 2.29 ± 1.12 |
| IL-15 IL-18 |
0.09 ± 0.07 nd |
0.19 ± 0.06 nd |
0.32 ± 0.11 nd |
0.04 ± 0.00 nd |
| IL-22 IL-27 |
0.41 ± 0.00 3.60 ± 0.88 |
nd nd |
nd nd |
nd nd |
| M-CSF | 1.78 ± 0.54 | 1.78 ± 0.54 | 12.58 ± 0.00 | 5.66 ± 0.00 |
| MCP-3 | 1.76 ± 0.90 | 1.77 ± 0.32 | 1.34 ± 0.18 | 0.27 ± 0.00 |
| MIG MIP PDGF-AA PDGF-AB/BB |
0.14 ± 0.09 2.10 ± 3.07 0.38 ± 0.27 21.07 ± 10.62 |
0.08 ± 0.00 1.31 ± 0.00 0.40 ± 0.18 21.45 ± 2.77 |
0.07 ± 0.05 nd 0.24 ± 0.14 12.84 ± 9.70 |
0.12 ± 0.05 nd 0.16 ± 0.14 17.06 ± 10.61 |
| TNFα | 0.13 ± 0.01 | 0.13 ± 0.02 | 0.12 ± 0.01 | 0.13 ± 0.01 |
| TNFβ RANTES sCD40 |
0.42 ± 0.31 0.17 ± 0.00 nd |
0.92 ± 0.25 0.13 ± 0.06 0.55 ± 0.00 |
0.56 ± 0.38 nd nd |
0.63 ± 0.54 nd nd |
| Cytokine / Factor | P4 | P7 | Function in Nerve Regeneration |
| FGF-2 | ✓ | ✓ | Angiogenesis, Schwann cell proliferation, axon outgrowth |
| VEGF-A | ✓ | — | Angiogenesis, neuroprotection |
| PDGF-AA | ✓ | — | Schwann cell support, tissue remodeling |
| IL-6 | ✓ | — | Inflammation resolution, glial modulation |
| IL-8 | ✓ | ✓ | Angiogenesis, chemotaxis |
| IL-22 | ✓ | ✓ | Tissue protection, neurotrophic signaling |
| M-CSF | ✓ | ✓ | Macrophage activation and polarization |
| MCP-3 | ✓ | — | Monocyte/macrophage recruitment |
| MCP-1 | — | ✓ | Monocyte chemotaxis, immune modulation |
| Eotaxin | ✓ | — | Chemokine signaling, potential glial effects |
| RANTES | — | ✓ | T-cell chemotaxis, axon growth, remyelination |
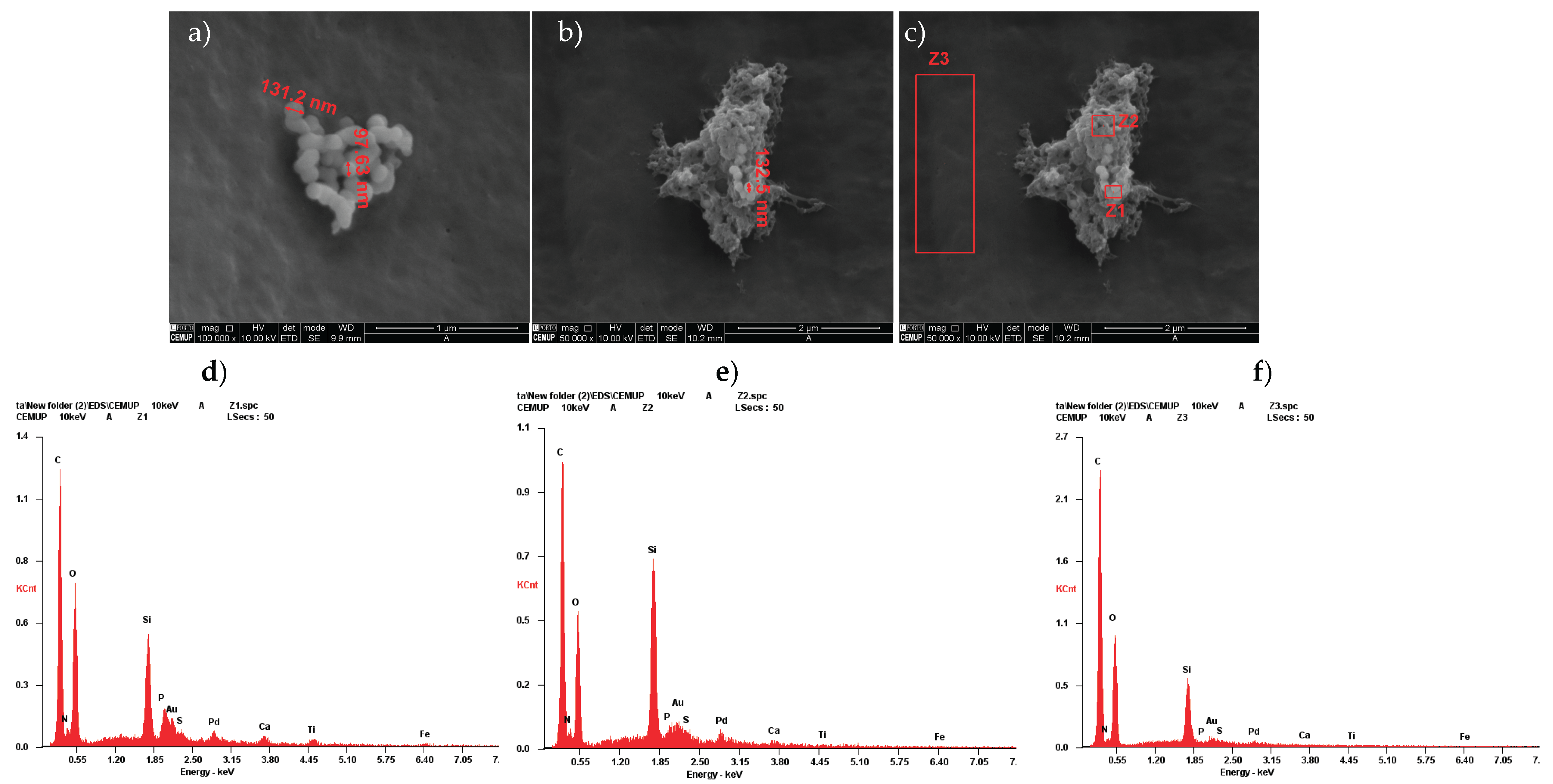
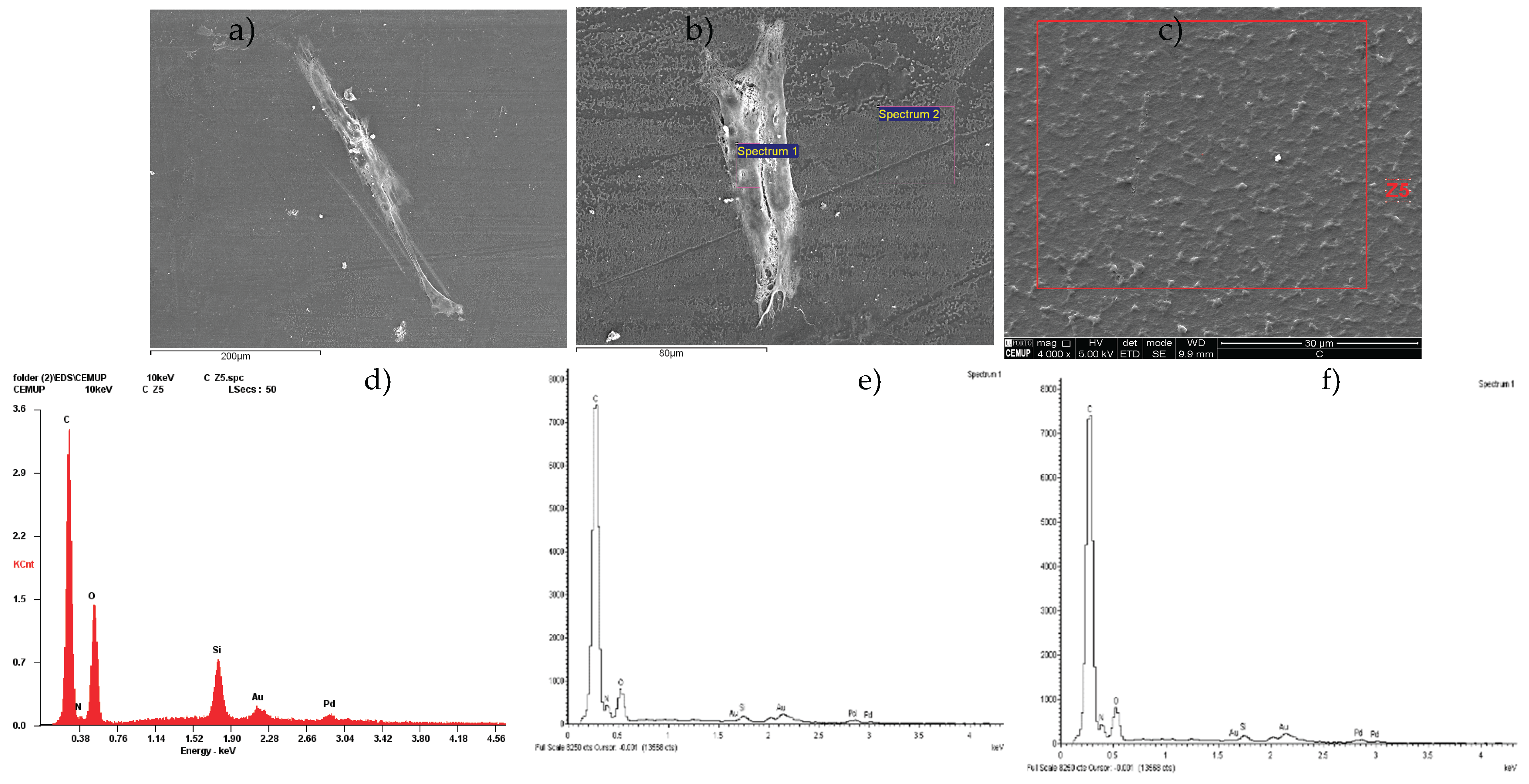
2.2. Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray Spectroscopy (EDS) Analysis
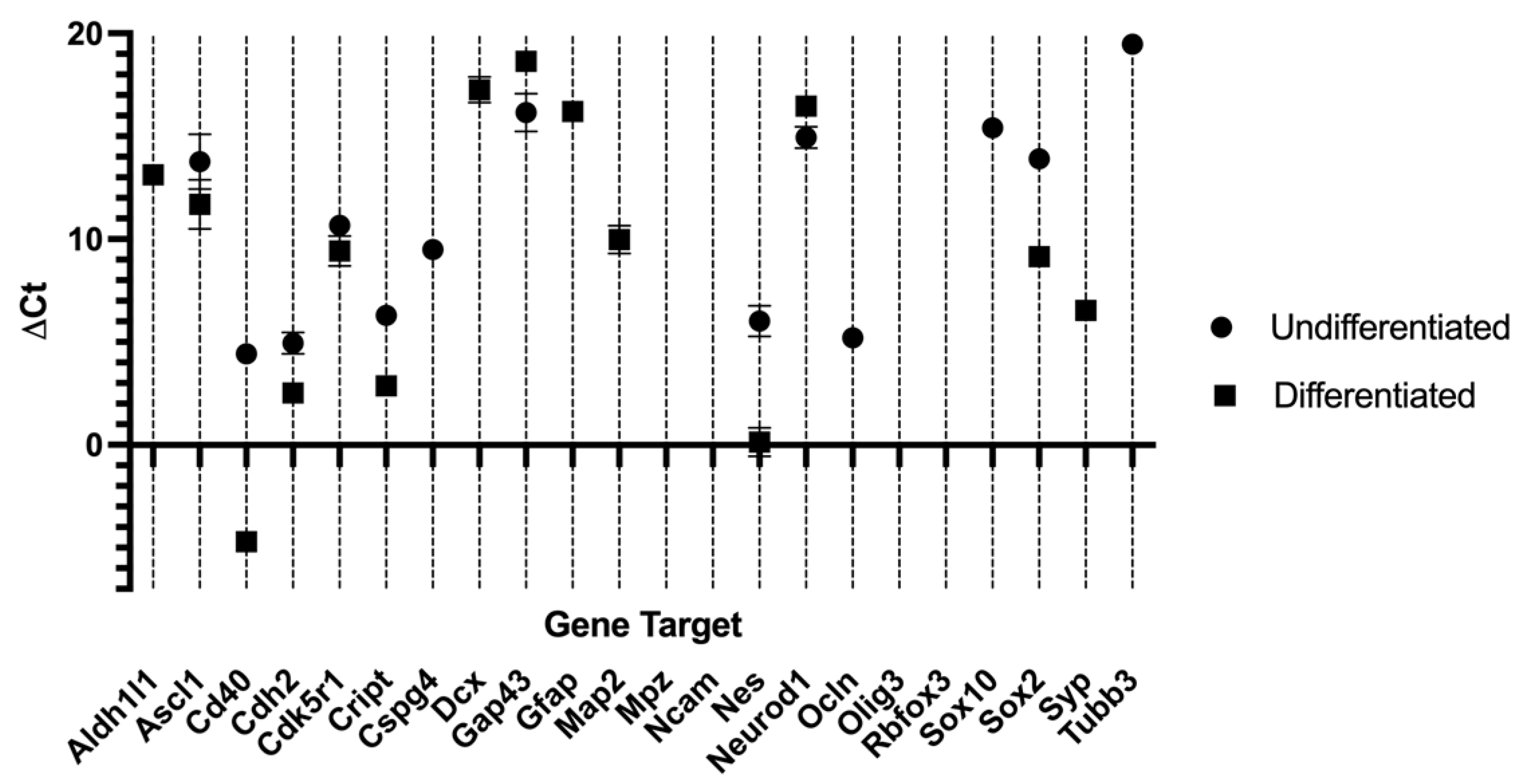
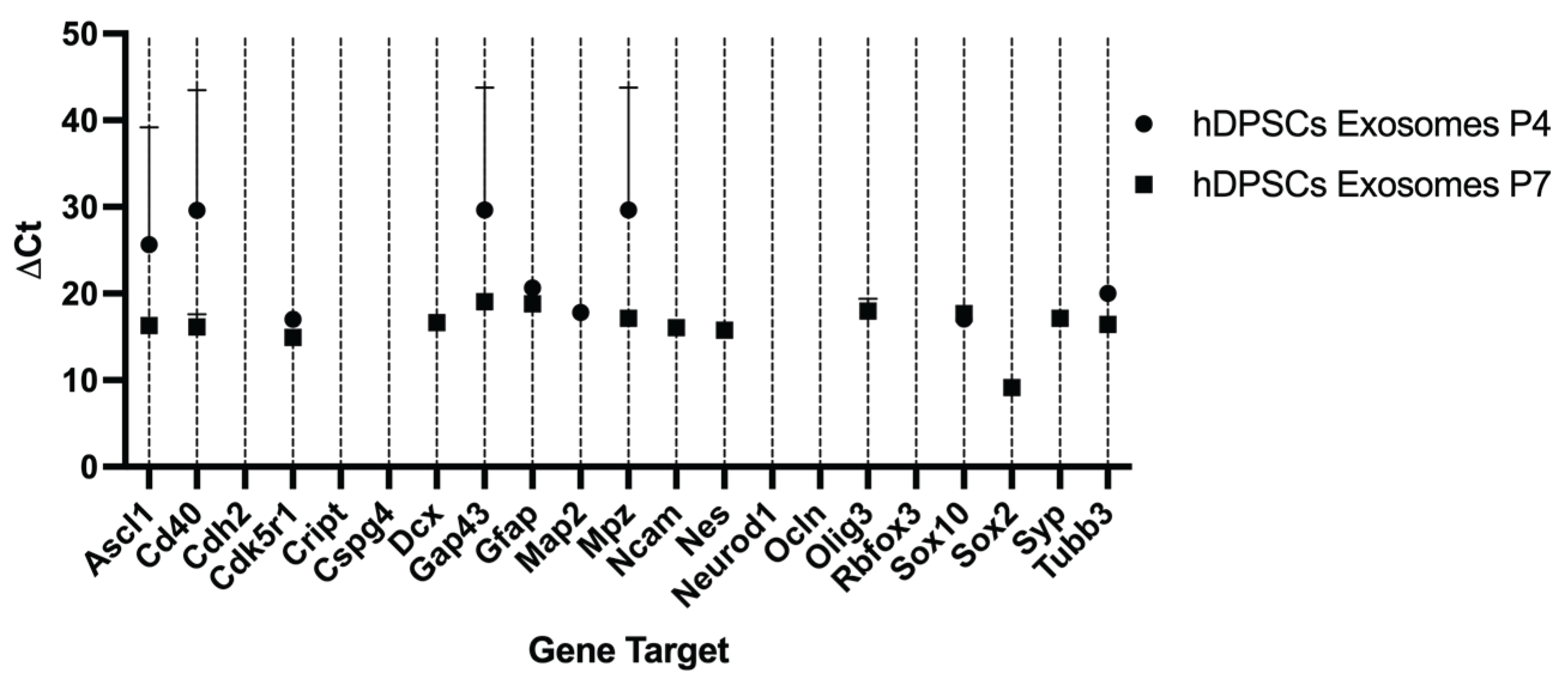
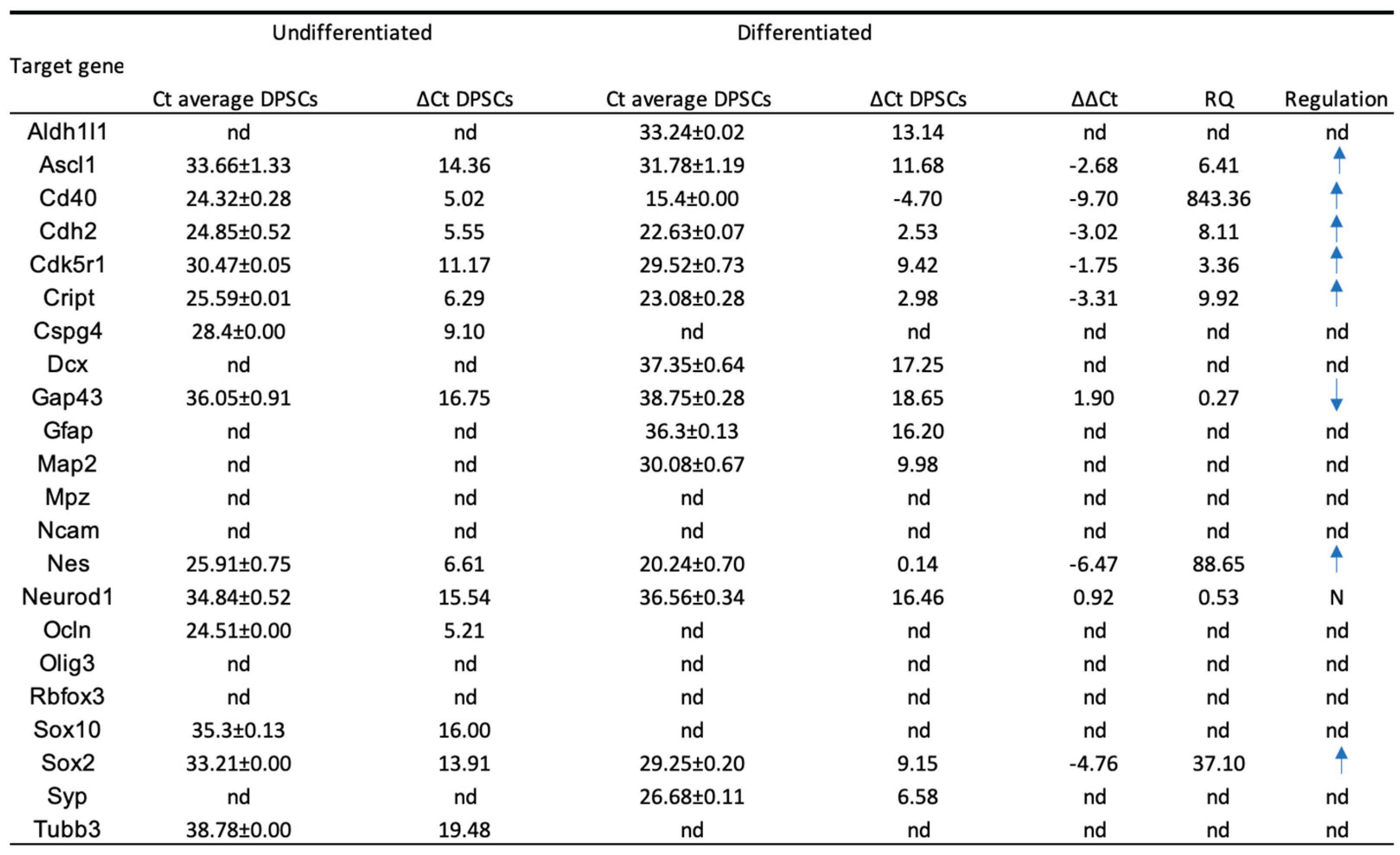
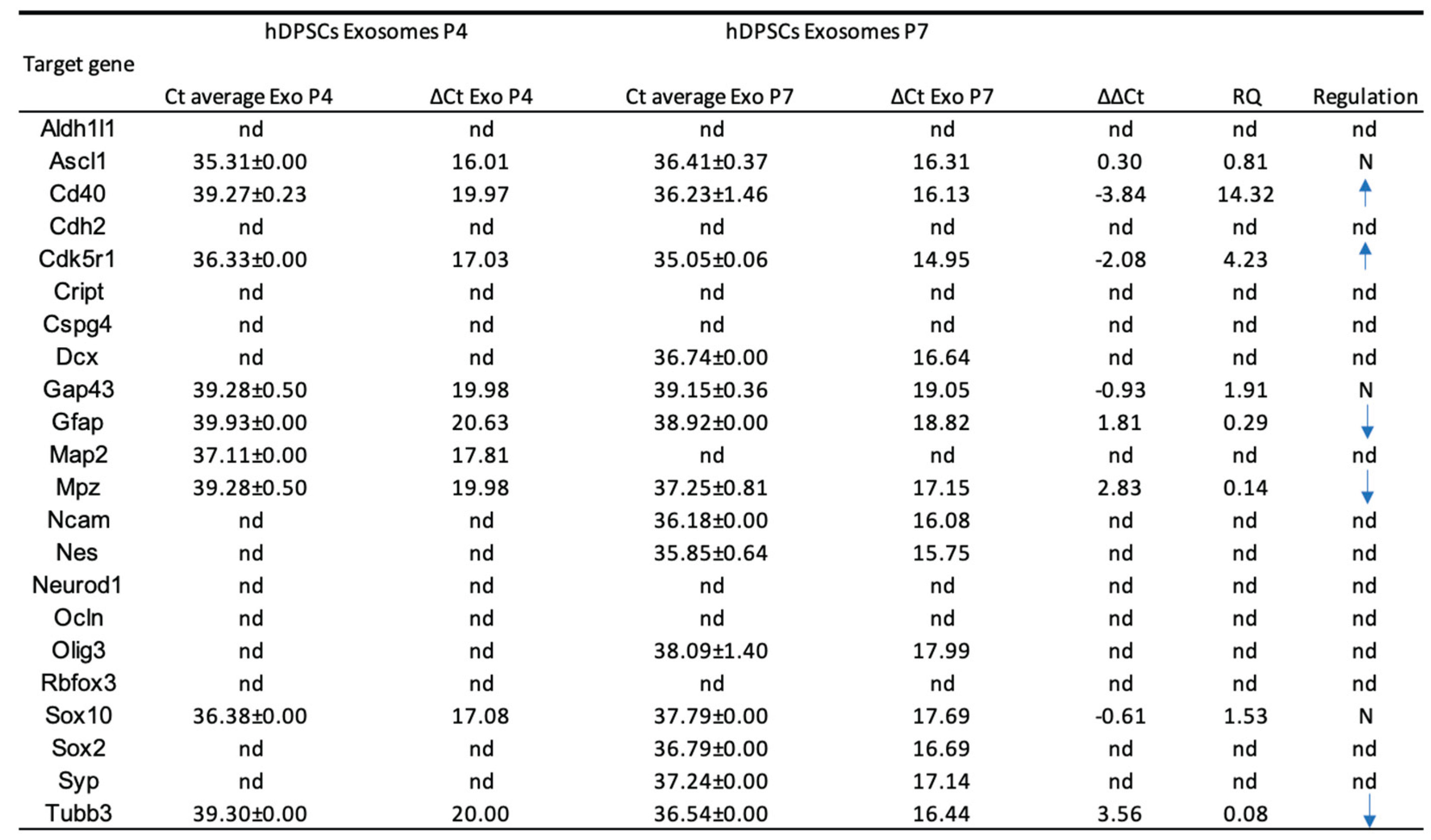
2.3. Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
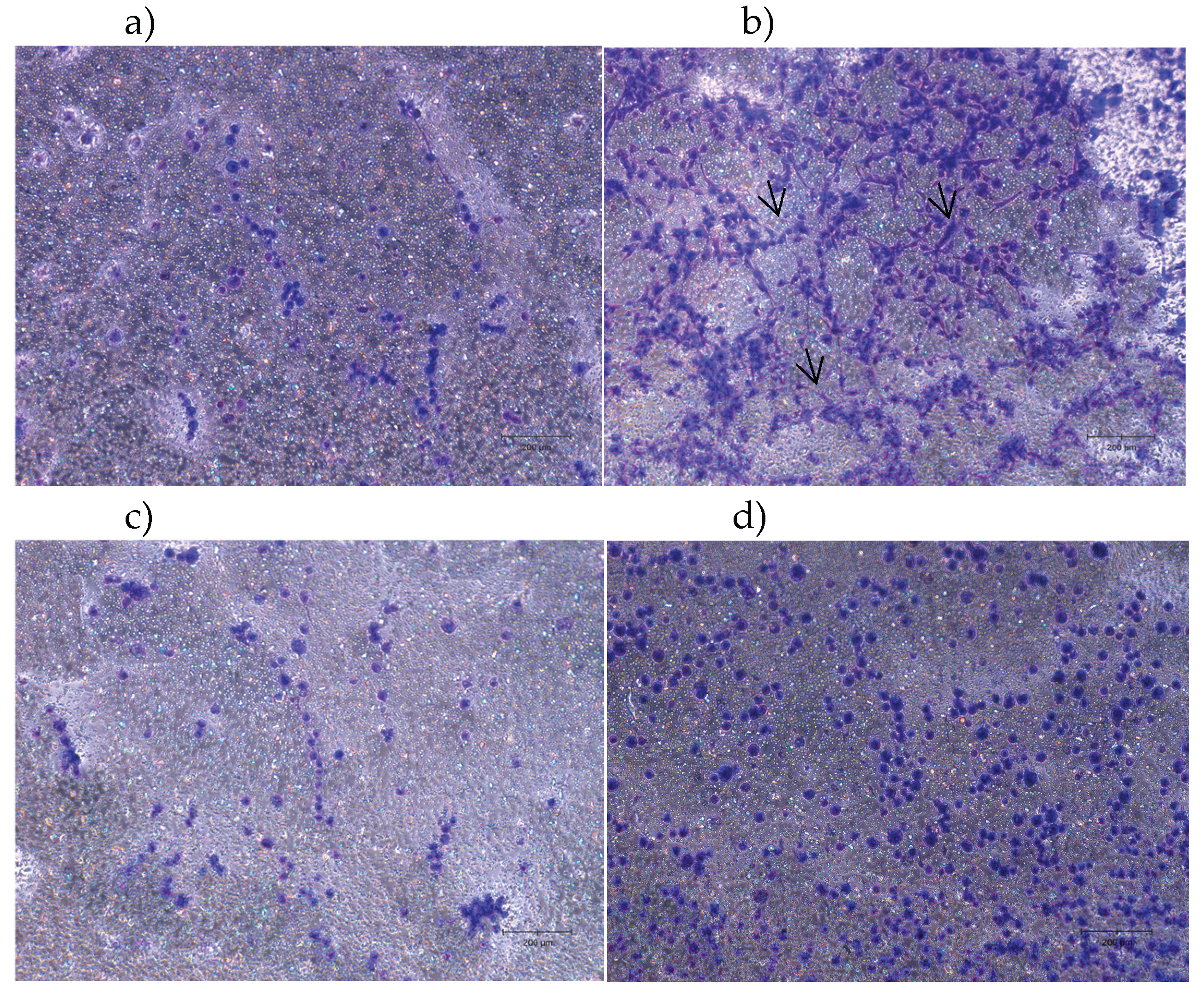
2.4. Neurite Outgrowth Assay
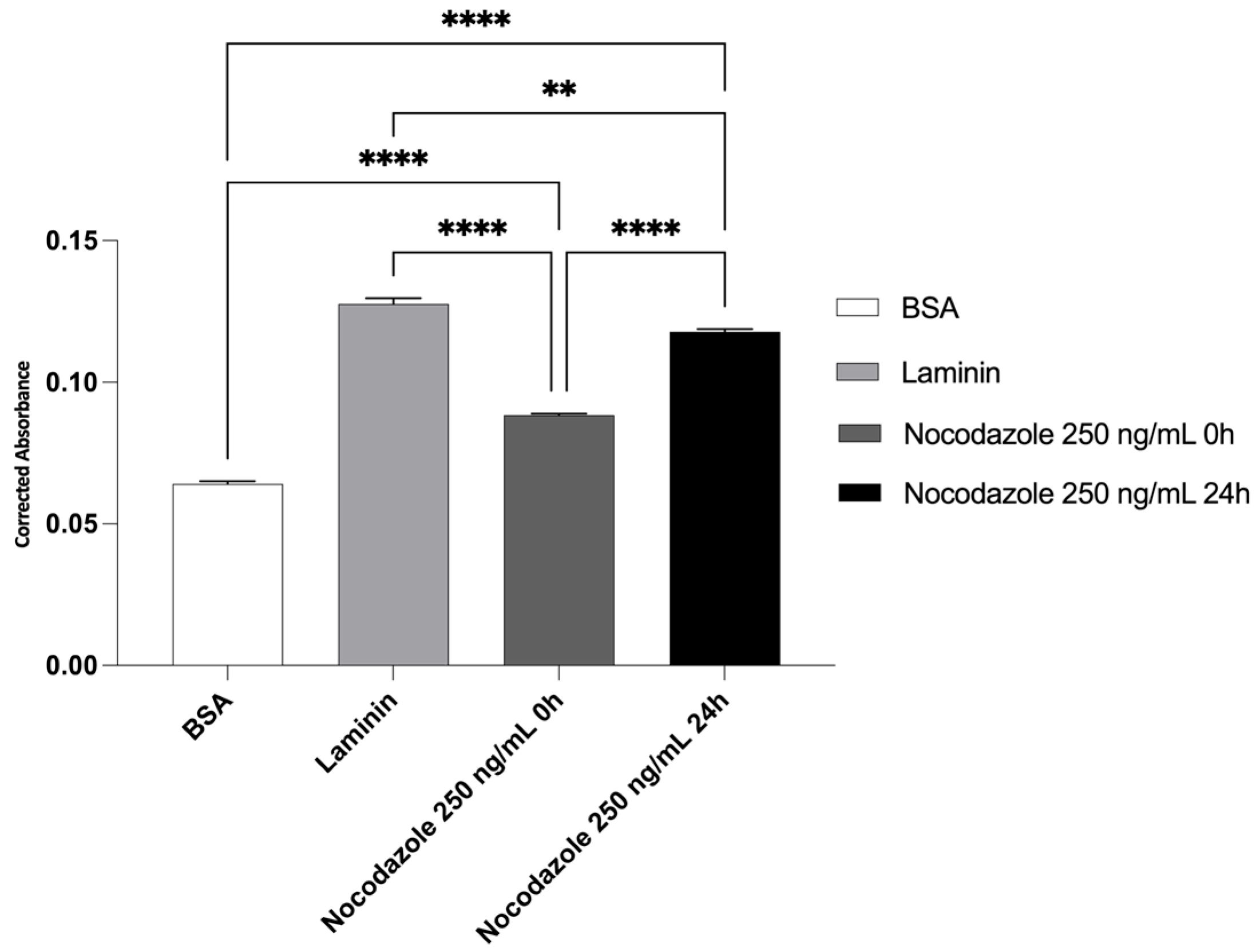
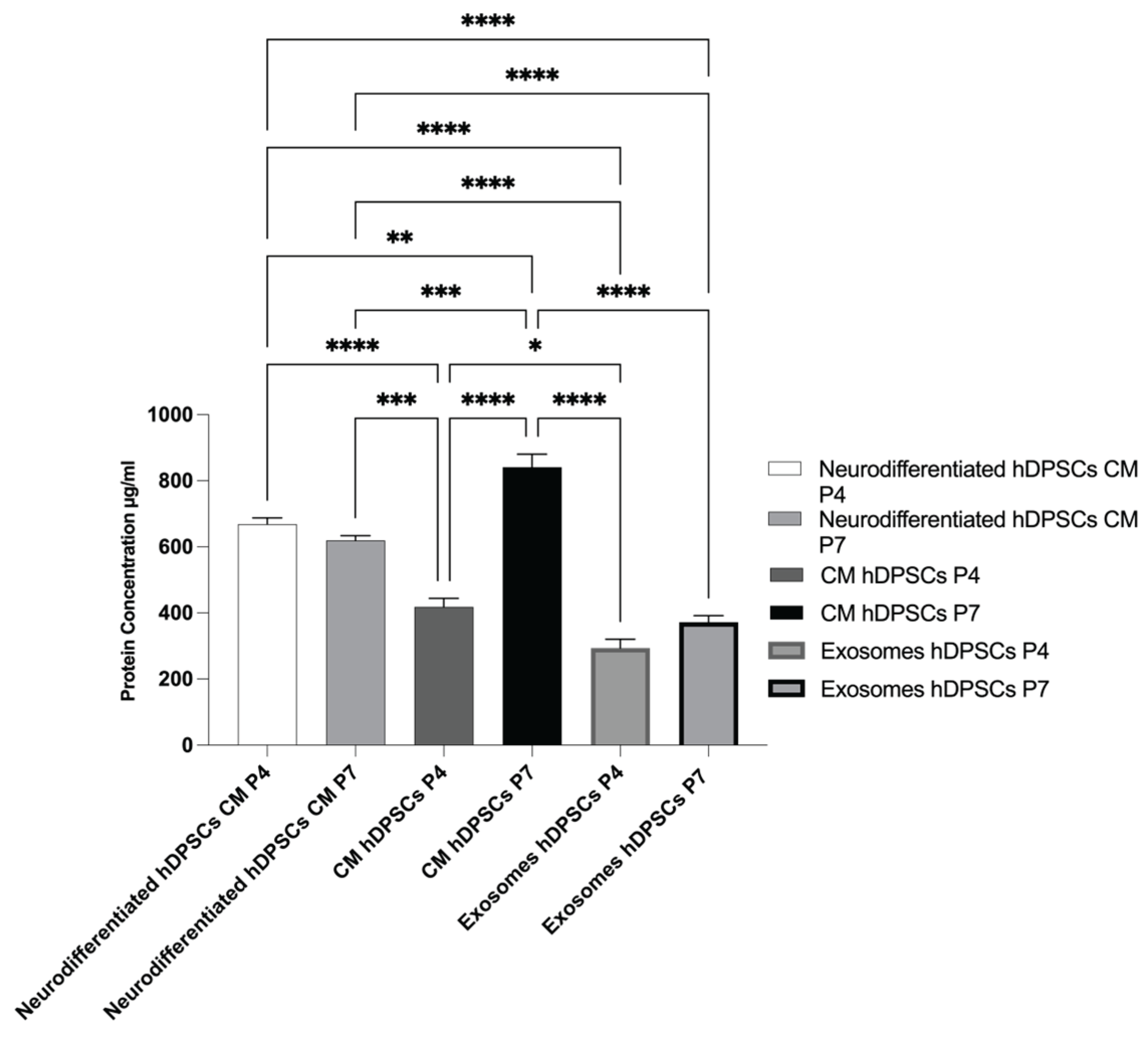
2.5. Total Protein Quantification
3. Discussion
4. Materials and Methods
4.1. Preparation of hDPSCs and Conditioned Medium
- -
- Undifferentiated hDPSCs at P7 and P4, both with unconcentrated CM (1×).
- -
- Neurodifferentiated hDPSCs at P4 and P7 with unconcentrated CM (1×).
- -
- Neurodifferentiated hDPSCs at P4 and P 7 with concentrated CM (5×).
- -
- Exosomes isolated from hDPSCs at P4 and P7 with unconcentrated CM (1×).
- -
- Exosomes isolated from hDPSCs at P4 and P7 with concentrated CM (5×).
4.2. Analysis of hDPSCs Conditioned Medium
4.3. Exosome Isolation from Cell Culture Media
4.4. Neurogenic Differentiation Assay
4.5. Scanning Electron Microscopy
4.6. RT-PCR
4.6.1. RNA Isolation and cDNA Synthesis from hDPSCs
4.6.2. RNA Isolation and cDNA Synthesis from Exosomes Derived from hDPSCs
4.7. Quantitative RT-PCR Assay
- -
- Ct < 29: Strong positive reaction, indicating a high abundance of the target nucleic acid.
- -
- 30 < Ct < 39: Moderate gene expression, with a detectable but lower quantity of the target nucleic acid.
- -
- Ct > 39: Weak signal, suggesting minimal target nucleic acid presence or possible environmental contamination.
4.8. Neurite Outgrowth Assay
4.9. Total Protein Quantification
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CM | Conditioned Medium |
| EDS | Energy Dispersive X-ray spectroscopy |
| EVs | Extracellular vesicles |
| hDPSCs | Human Dental Pulp Stem Cells |
| hDPSCs-CM | Human Dental Pulp Stem Cells Conditioned Medium |
| ISEV | International Society for Extracellular Vesicles |
| MSCs | Mesenchymal Stem Cells |
| NGC | Nerve Guide Conduit |
| PNIs | Peripheral Nerve Injuries |
| RT-PCR | Reverse Transcriptase Polymerase Chain Reaction |
| SEM | Scanning Eletron Microscopy |
References
- Lopes, B., et al., Peripheral Nerve Injury Treatments and Advances: One Health Perspective. Int J Mol Sci, 2022. 23(2).
- Lopes, B., et al., In vitro evaluation of dental pulp stem cells for sciatic nerve regeneration: foundations for future in vivo applications. Front Cell Dev Biol, 2025. 13: p. 1528213.
- Alvites, R., et al., Peripheral nerve injury and axonotmesis: State of the art and recent advances. Cogent Medicine, 2018. 5(1).
- Xu, G., et al., Advancements in autologous peripheral nerve transplantation care: a review of strategies and practices to facilitate recovery. Front Neurol, 2024. 15: p. 1330224.
- Zhu, J., et al., Advances in Peripheral Nerve Injury Repair with the Application of Nanomaterials. Journal of Nanomaterials, 2022. 2022: p. 1-22.
- Alvites, R.D., et al., Effects of Olfactory Mucosa Stem/Stromal Cell and Olfactory Ensheating Cells Secretome on Peripheral Nerve Regeneration. Biomolecules, 2022. 12(6).
- Shen, X., et al., Repairing sciatic nerve injury with self-assembling peptide nanofiber scaffold-containing chitosan conduit. Front Neurol, 2022. 13: p. 867711.
- Alvites, R.D., et al., Combined Use of Chitosan and Olfactory Mucosa Mesenchymal Stem/Stromal Cells to Promote Peripheral Nerve Regeneration In Vivo. Stem Cells Int, 2021. 2021: p. 6613029.
- Sousa, A.C., et al., Hybrid scaffolds for bone tissue engineering: Integration of composites and bioactive hydrogels loaded with hDPSCs. Biomater Adv, 2024. 166: p. 214042.
- Lopes, B., et al., Animal models in peripheral nerve transection studies: a systematic review on study design and outcomes assessment. Regen Med, 2023.
- Stocco, E., et al., Bridging Gaps in Peripheral Nerves: From Current Strategies to Future Perspectives in Conduit Design. Int J Mol Sci, 2023. 24(11).
- Redolfi Riva, E., et al., Beyond the limiting gap length: peripheral nerve regeneration through implantable nerve guidance conduits. Biomaterials Science, 2024. 12(6): p. 1371-1404.
- Smolinska, V., M. Bohac, and L. Danisovic, Current status of the applications of conditioned media derived from mesenchymal stem cells for regenerative medicine. Physiol Res, 2023. 72(S3): p. S233-S245.
- Stefanska, K., et al., Dental pulp stem cells - A basic research and future application in regenerative medicine. Biomed Pharmacother, 2024. 178: p. 116990.
- Fujii, Y., et al., Application of Dental Pulp Stem Cells for Bone and Neural Tissue Regeneration in Oral and Maxillofacial Region. Stem Cells Int, 2023. 2023: p. 2026572.
- Mattei, V. and S. Delle Monache, Dental Pulp Stem Cells (DPSCs) and Tissue Regeneration: Mechanisms Mediated by Direct, Paracrine, or Autocrine Effects. Biomedicines, 2023. 11(2).
- Tsuruta, T., et al., Dental pulp-derived stem cell conditioned medium to regenerate peripheral nerves in a novel animal model of dysphagia. PLoS One, 2018. 13(12): p. e0208938.
- Wang, W., et al., Prospective applications of extracellular vesicle-based therapies in regenerative medicine: implications for the use of dental stem cell-derived extracellular vesicles. Front Bioeng Biotechnol, 2023. 11: p. 1278124.
- Alvites, R., et al., Mesenchymal Stem/Stromal Cells and Their Paracrine Activity-Immunomodulation Mechanisms and How to Influence the Therapeutic Potential. Pharmaceutics, 2022. 14(2).
- Sagaradze, G., et al., Conditioned Medium from Human Mesenchymal Stromal Cells: Towards the Clinical Translation. Int J Mol Sci, 2019. 20(7).
- Ledesma-Martinez, E., V.M. Mendoza-Nunez, and E. Santiago-Osorio, Mesenchymal Stem Cells Derived from Dental Pulp: A Review. Stem Cells Int, 2016. 2016: p. 4709572.
- Gwam, C., N. Mohammed, and X. Ma, Stem cell secretome, regeneration, and clinical translation: a narrative review. Ann Transl Med, 2021. 9(1): p. 70.
- Bar, J.K., A. Lis-Nawara, and P.G. Grelewski, Dental Pulp Stem Cell-Derived Secretome and Its Regenerative Potential. Int J Mol Sci, 2021. 22(21).
- Younes, R., et al., The Secretome of Human Dental Pulp Stem Cells and Its Components GDF15 and HB-EGF Protect Amyotrophic Lateral Sclerosis Motoneurons against Death. Biomedicines, 2023. 11(8).
- Mohd Nor, N.H., et al., From Teeth to Therapy: A Review of Therapeutic Potential within the Secretome of Stem Cells from Human Exfoliated Deciduous Teeth. Int J Mol Sci, 2023. 24(14).
- Liu, Y., et al., Application of dental pulp stem cells for bone regeneration. Front Med (Lausanne), 2024. 11: p. 1339573.
- Valadi, H., et al., Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol, 2007. 9(6): p. 654-9.
- Vilaca-Faria, H., A.J. Salgado, and F.G. Teixeira, Mesenchymal Stem Cells-derived Exosomes: A New Possible Therapeutic Strategy for Parkinson’s Disease? Cells, 2019. 8(2).
- Odehnalova, N., et al., The potential of exosomes in regenerative medicine and in the diagnosis and therapies of neurodegenerative diseases and cancer. Front Med (Lausanne), 2025. 12: p. 1539714.
- Chansaenroj, J., et al., Potential of Dental Pulp Stem Cell Exosomes: Unveiling miRNA-Driven Regenerative Mechanisms. Int Dent J, 2025. 75(2): p. 415-425.
- Shafiei, M., et al., A Comprehensive Review on the Applications of Exosomes and Liposomes in Regenerative Medicine and Tissue Engineering. Polymers (Basel), 2021. 13(15).
- Gugliandolo, A. and E. Mazzon, Dental Mesenchymal Stem Cell Secretome: An Intriguing Approach for Neuroprotection and Neuroregeneration. Int J Mol Sci, 2021. 23(1).
- Lotvall, J., et al., Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles, 2014. 3: p. 26913.
- Dilsiz, N., A comprehensive review on recent advances in exosome isolation and characterization: Toward clinical applications. Transl Oncol, 2024. 50: p. 102121.
- Li, Q., et al., Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes as Nanomedicine for Peripheral Nerve Injury. Int J Mol Sci, 2024. 25(14).
- Lai, J.J., et al., Exosome Processing and Characterization Approaches for Research and Technology Development. Adv Sci (Weinh), 2022. 9(15): p. e2103222.
- Zhang, S., et al., MSC exosomes alleviate temporomandibular joint osteoarthritis by attenuating inflammation and restoring matrix homeostasis. Biomaterials, 2019. 200: p. 35-47.
- Wei, H., et al., Mesenchymal stem cell-derived exosomal miR-223 regulates neuronal cell apoptosis. Cell Death Dis, 2020. 11(4): p. 290.
- Chai, Y., et al., Study on the Role and Mechanism of Exosomes Derived from Dental Pulp Stem Cells in Promoting Regeneration of Myelin Sheath in Rats with Sciatic Nerve Injury. Mol Neurobiol, 2024. 61(9): p. 6175-6188.
- Yavuz, B., et al., Applications of Stem Cell-Derived Extracellular Vesicles in Nerve Regeneration. Int J Mol Sci, 2024. 25(11).
- Namini, M.S., et al., Cell-free therapy based on extracellular vesicles: a promising therapeutic strategy for peripheral nerve injury. Stem Cell Res Ther, 2023. 14(1): p. 254.
- Ezdakova, M.I. and E.R. Andreeva, Impaired Communication through Gap Junctions Reduces the Angiogenic Potential of the Secretome in Mesenchymal Stromal Cell—Endothelial Cell Interactions In Vitro. Bulletin of Experimental Biology and Medicine, 2024. 178(1): p. 139-144.
- Liu, J., et al., Senescence in Mesenchymal Stem Cells: Functional Alterations, Molecular Mechanisms, and Rejuvenation Strategies. Front Cell Dev Biol, 2020. 8: p. 258.
- Liu, Y., et al., Application of dental pulp stem cell-conditioned medium combined with deep cryopreservation of autologous cranial flaps. Stem Cell Res Ther, 2025. 16(1): p. 272.
- Harrell, C.R., et al., Therapeutic Use of Mesenchymal Stem Cell-Derived Exosomes: From Basic Science to Clinics. Pharmaceutics, 2020. 12(5).
- Rastogi, S., et al., The Evolving Landscape of Exosomes in Neurodegenerative Diseases: Exosomes Characteristics and a Promising Role in Early Diagnosis. Int J Mol Sci, 2021. 22(1).
- Chen, G.Y., et al., Effects of exosomes from human dental pulp stem cells on the biological behavior of human fibroblasts. Sci Rep, 2025. 15(1): p. 1134.
- Wu, S.H., et al., Adipose-Derived Mesenchymal Stem Cells From a Hypoxic Culture Improve Neuronal Differentiation and Nerve Repair. Front Cell Dev Biol, 2021. 9: p. 658099.
- Caseiro, A.R., et al., Mesenchymal Stem/ Stromal Cells metabolomic and bioactive factors profiles: A comparative analysis on the umbilical cord and dental pulp derived Stem/ Stromal Cells secretome. PLoS One, 2019. 14(11): p. e0221378.
- Gao, J., et al., Dental pulp stem cells conditioned medium-functionalized microspheres for endodontic regeneration. Frontiers in Cell and Developmental Biology, 2025. 13.
- Gharaei, M.A., et al., Human dental pulp stromal cell conditioned medium alters endothelial cell behavior. Stem Cell Res Ther, 2018. 9(1): p. 69.
- El Moshy, S., et al., Dental Stem Cell-Derived Secretome/Conditioned Medium: The Future for Regenerative Therapeutic Applications. Stem Cells Int, 2020. 2020: p. 7593402.
- Maccaferri, M., et al., Human dental pulp stem cells modulate pro-inflammatory macrophages both through cell-to-cell contact and paracrine signaling. Front Immunol, 2024. 15: p. 1440974.
- Barone, L., et al., Dental pulp mesenchymal stem cell (DPSCs)-derived soluble factors, produced under hypoxic conditions, support angiogenesis via endothelial cell activation and generation of M2-like macrophages. J Biomed Sci, 2024. 31(1): p. 99.
- Li, P., et al., Immunomodulatory properties of mesenchymal stem cells/dental stem cells and their therapeutic applications. Cell Mol Immunol, 2023. 20(6): p. 558-569.
- Alraies, A., et al., Variation in human dental pulp stem cell ageing profiles reflect contrasting proliferative and regenerative capabilities. BMC Cell Biol, 2017. 18(1): p. 12.
- Saito, Y., S. Yamamoto, and T.S. Chikenji, Role of cellular senescence in inflammation and regeneration. Inflamm Regen, 2024. 44(1): p. 28.
- Lin, C.Y., et al., The Exosomes of Stem Cells from Human Exfoliated Deciduous Teeth Suppress Inflammation in Osteoarthritis. Int J Mol Sci, 2024. 25(16).
- Hua, S., et al., Periodontal and Dental Pulp Cell-Derived Small Extracellular Vesicles: A Review of the Current Status. Nanomaterials (Basel), 2021. 11(7).
- Mai, Z., et al., Translational and Clinical Applications of Dental Stem Cell-Derived Exosomes. Front Genet, 2021. 12: p. 750990.
- Arimura, Y., et al., Peripheral-neuron-like properties of differentiated human dental pulp stem cells (hDPSCs). PLOS ONE, 2021. 16(5): p. e0251356.
- Sousa, A.C., et al., Assessment of 3D-Printed Polycaprolactone, Hydroxyapatite Nanoparticles and Diacrylate Poly(ethylene glycol) Scaffolds for Bone Regeneration. Pharmaceutics, 2022. 14(12).
- Brunello, G., et al., Exosomes Derived from Dental Pulp Stem Cells Show Different Angiogenic and Osteogenic Properties in Relation to the Age of the Donor. Pharmaceutics, 2022. 14(5).
- Harley-Troxell, M.E., et al., Interactions of Cells and Biomaterials for Nerve Tissue Engineering: Polymers and Fabrication. Polymers (Basel), 2023. 15(18).
- Pardo-Rodriguez, B., et al., Functional differentiation of human dental pulp stem cells into neuron-like cells exhibiting electrophysiological activity. Stem Cell Res Ther, 2025. 16(1): p. 10.
- Gao, Y., et al., Neuronal Cell Differentiation of Human Dental Pulp Stem Cells on Synthetic Polymeric Surfaces Coated With ECM Proteins. Front Cell Dev Biol, 2022. 10: p. 893241.
- Seonwoo, H., et al., Neurogenic Differentiation of Human Dental Pulp Stem Cells on Graphene-Polycaprolactone Hybrid Nanofibers. Nanomaterials (Basel), 2018. 8(7).
- Ellis, P., et al., SOX2, a persistent marker for multipotential neural stem cells derived from embryonic stem cells, the embryo or the adult. Dev Neurosci, 2004. 26(2-4): p. 148-65.
- Suzuki, S., et al., The neural stem/progenitor cell marker nestin is expressed in proliferative endothelial cells, but not in mature vasculature. J Histochem Cytochem, 2010. 58(8): p. 721-30.
- Vainorius, G., et al., Ascl1 and Ngn2 convert mouse embryonic stem cells to neurons via functionally distinct paths. Nat Commun, 2023. 14(1): p. 5341.
- Alimperti, S. and S.T. Andreadis, CDH2 and CDH11 act as regulators of stem cell fate decisions. Stem Cell Res, 2015. 14(3): p. 270-82.
- Al-Maswary, A.A., et al., Exploring the neurogenic differentiation of human dental pulp stem cells. PLOS ONE, 2022. 17(11): p. e0277134.
- Luzuriaga, J., et al., Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies. Int J Mol Sci, 2021. 22(7).
- Geuna, S., et al., In vitro models for peripheral nerve regeneration. European Journal of Neuroscience, 2016. 43(3): p. 287-296.
- Li, D., et al., Human Dental Pulp Stem Cells and Gingival Mesenchymal Stem Cells Display Action Potential Capacity In Vitro after Neuronogenic Differentiation. Stem Cell Reviews and Reports, 2019. 15(1): p. 67-81.
- Kucharova, K. and W.B. Stallcup, The NG2 proteoglycan promotes oligodendrocyte progenitor proliferation and developmental myelination. Neuroscience, 2010. 166(1): p. 185-94.
- Fujiwara, S., et al., SOX10 transactivates S100B to suppress Schwann cell proliferation and to promote myelination. PLoS One, 2014. 9(12): p. e115400.
- Pummi, K.P., et al., Tight junction proteins ZO-1, occludin, and claudins in developing and adult human perineurium. J Histochem Cytochem, 2004. 52(8): p. 1037-46.
- Latremoliere, A., et al., Neuronal-Specific TUBB3 Is Not Required for Normal Neuronal Function but Is Essential for Timely Axon Regeneration. Cell Rep, 2018. 24(7): p. 1865-1879 e9.
- Gusel’nikova, V.V. and D.E. Korzhevskiy, NeuN As a Neuronal Nuclear Antigen and Neuron Differentiation Marker. Acta Naturae, 2015. 7(2): p. 42-7.
- LeBlanc, S.E., et al., Direct regulation of myelin protein zero expression by the Egr2 transactivator. J Biol Chem, 2006. 281(9): p. 5453-60.
- Delle Monache, S., et al., Hypoxia Induces DPSC Differentiation versus a Neurogenic Phenotype by the Paracrine Mechanism. Biomedicines, 2022. 10(5).
- Holahan, M.R., A Shift from a Pivotal to Supporting Role for the Growth-Associated Protein (GAP-43) in the Coordination of Axonal Structural and Functional Plasticity. Front Cell Neurosci, 2017. 11: p. 266.
- Grasselli, G. and P. Strata, Structural plasticity of climbing fibers and the growth-associated protein GAP-43. Front Neural Circuits, 2013. 7: p. 25.
- Libberecht, K., et al., The Influence of Lysosomal Stress on Dental Pulp Stem Cell-Derived Schwann Cells. Biomolecules, 2024. 14(4).
- Candelise, N., et al., The Importance of Stem Cells Isolated from Human Dental Pulp and Exfoliated Deciduous Teeth as Therapeutic Approach in Nervous System Pathologies. Cells, 2023. 12(13).
- Bassett, C., et al., Differential Expression of MicroRNA (MiR-27, MiR-145) among Dental Pulp Stem Cells (DPSCs) Following Neurogenic Differentiation Stimuli. Biomedicines, 2023. 11(11).
- Radwitz, J., et al., Tubb3 expression levels are sensitive to neuronal activity changes and determine microtubule growth and kinesin-mediated transport. Cell Mol Life Sci, 2022. 79(11): p. 575.
- Jurga, A.M., et al., Beyond the GFAP-Astrocyte Protein Markers in the Brain. Biomolecules, 2021. 11(9).
- Konnova, E.A., et al., Characterisation of GFAP-Expressing Glial Cells in the Dorsal Root Ganglion after Spared Nerve Injury. Int J Mol Sci, 2023. 24(21).
- Mester, T., et al., CD40 Expression in Fibrocytes Is Induced by TSH: Potential Synergistic Immune Activation. PLoS One, 2016. 11(9): p. e0162994.
- Xia, L., et al., CDK5R1 promotes Schwann cell proliferation, migration, and production of neurotrophic factors via CDK5/BDNF/TrkB after sciatic nerve injury. Neuroscience Letters, 2023. 817: p. 137514.
- Ao, C., et al., The role of Cdk5 in neurological disorders. Front Cell Neurosci, 2022. 16: p. 951202.
- Sugimori, M., et al., Ascl1 is required for oligodendrocyte development in the spinal cord. Development, 2008. 135(7): p. 1271-81.
- Carney, T.J., et al., A direct role for Sox10 in specification of neural crest-derived sensory neurons. Development, 2006. 133(23): p. 4619-30.
- Tordoff, E., et al., A novel multiplexed immunoassay for surface-exposed proteins in plasma extracellular vesicles. J Extracell Vesicles, 2024. 13(11): p. e70007.
- Ahmad, P., et al., Isolation methods of exosomes derived from dental stem cells. Int J Oral Sci, 2025. 17(1): p. 50.
- Tang, Y., Y. Zhou, and H.J. Li, Advances in mesenchymal stem cell exosomes: a review. Stem Cell Res Ther, 2021. 12(1): p. 71.
- O’Connor, C., et al., Neurotrophic extracellular matrix proteins promote neuronal and iPSC astrocyte progenitor cell- and nano-scale process extension for neural repair applications. J Anat, 2025. 246(4): p. 585-601.
- Deng, L.X., et al., Laminin-coated multifilament entubulation, combined with Schwann cells and glial cell line-derived neurotrophic factor, promotes unidirectional axonal regeneration in a rat model of thoracic spinal cord hemisection. Neural Regen Res, 2021. 16(1): p. 186-191.
- Speer, J., et al., Integrin-mediated interactions with a laminin-presenting substrate modulate biosynthesis and phenotypic expression for cells of the human nucleus pulposus. Eur Cell Mater, 2021. 41: p. 793-810.
- Arimori, T., et al., Structural mechanism of laminin recognition by integrin. Nat Commun, 2021. 12(1): p. 4012.
- Nolan, A.M., et al., The neurite growth inhibitory effects of soluble TNFα on developing sympathetic neurons are dependent on developmental age. Differentiation, 2014. 88(4): p. 124-130.
- Popova, D. and S.O.P. Jacobsson, A fluorescence microplate screen assay for the detection of neurite outgrowth and neurotoxicity using an antibody against βIII-tubulin. Toxicology in Vitro, 2014. 28(3): p. 411-418.
- Sierra-Fonseca, J.A., et al., Nerve growth factor induces neurite outgrowth of PC12 cells by promoting Gbetagamma-microtubule interaction. BMC Neurosci, 2014. 15: p. 132.
- Gagnon, J., V. Caron, and A. Tremblay, SUMOylation of nuclear receptor Nor1/NR4A3 coordinates microtubule cytoskeletal dynamics and stability in neuronal cells. Cell Biosci, 2024. 14(1): p. 91.
- DeVault, L., et al., The response of Dual-leucine zipper kinase (DLK) to nocodazole: Evidence for a homeostatic cytoskeletal repair mechanism. PLOS ONE, 2024. 19(4): p. e0300539.
- Plessis-Belair, J., et al., Nuclear Import Defects Drive Cell Cycle Dysregulation in Neurodegeneration. Aging Cell, 2025: p. e70091.
- Verkest, C., et al., Intrinsically disordered intracellular domains control key features of the mechanically-gated ion channel PIEZO2. Nat Commun, 2022. 13(1): p. 1365.
- Borghi, R., et al., Altered cytoskeleton dynamics in patient-derived iPSC-based model of PCDH19 clustering epilepsy. Front Cell Dev Biol, 2024. 12: p. 1518533.
- Lasser, M., J. Tiber, and L.A. Lowery, The Role of the Microtubule Cytoskeleton in Neurodevelopmental Disorders. Front Cell Neurosci, 2018. 12: p. 165.
- Sanchez-Huertas, C. and E. Herrera, With the Permission of Microtubules: An Updated Overview on Microtubule Function During Axon Pathfinding. Front Mol Neurosci, 2021. 14: p. 759404.
- Verstraelen, P., et al., Dysregulation of Microtubule Stability Impairs Morphofunctional Connectivity in Primary Neuronal Networks. Front Cell Neurosci, 2017. 11: p. 173.
- Sferra, A., F. Nicita, and E. Bertini, Microtubule Dysfunction: A Common Feature of Neurodegenerative Diseases. Int J Mol Sci, 2020. 21(19).
- Harvey, A., et al., Proteomic analysis of the extracellular matrix produced by mesenchymal stromal cells: implications for cell therapy mechanism. PLoS One, 2013. 8(11): p. e79283.
- Abd-elwahab, S.A.-e., et al., Mesenchymal-Stem Cell-Derived Conditioned Media Versus Exosomes in the Treatment of Rat Model of Polycystic Ovary: An Attempt to Understand the Underlying Mechanisms (Biochemical and Histological Study). Microscopy and Microanalysis, 2023. 29(3): p. 1244-1257.
- Novais, A., et al., Priming Dental Pulp Stem Cells from Human Exfoliated Deciduous Teeth with Fibroblast Growth Factor-2 Enhances Mineralization Within Tissue-Engineered Constructs Implanted in Craniofacial Bone Defects. Stem Cells Transl Med, 2019. 8(8): p. 844-857.
- Zhou, T., et al., Conditioned medium derived from 3D tooth germs: A novel cocktail for stem cell priming and early in vivo pulp regeneration. Cell Prolif, 2021. 54(11): p. e13129.
- Sousa, A.C., et al., Production, Characterisation, and In Vitro Evaluation of 3D Printed PCL/HANp/PEGDA Scaffold for Bone Regeneration, in Materiais 2022. 2022.
- Caseiro, A.R., et al., Human umbilical cord blood plasma as an alternative to animal sera for mesenchymal stromal cells in vitro expansion - A multicomponent metabolomic analysis. PLoS One, 2018. 13(10): p. e0203936.
- Machado, A., et al., Dextrin hydrogel loaded with a macroporous Bonelike® scaffold and dental pulp stem cells for critical-sized defect repair. Materialia, 2023. 30.
- Sousa, P., et al., Rat Hair Follicle Stem Cell-Derived Exosomes: Isolation, Characterization and Comparative Analysis of Their In Vitro Wound Healing Potential. Int J Mol Sci, 2025. 26(11).
- Alvites, R.D., et al., Rat Olfactory Mucosa Mesenchymal Stem/Stromal Cells (OM-MSCs): A Characterization Study. International Journal of Cell Biology, 2020. 2020: p. 1-21.
- Clerici, M., et al., Pitfalls of Accurate Protein Determination inside PLGA Nanoparticles Using the Micro BCA Assay, in The 1st International Electronic Conference on Pharmaceutics. 2020.
 |
 |
| Neurodifferentiated hDPSCs CM P4 | Neurodifferentiated hDPSCs CM P7 | CM hDPSCs P4 | CM hDPSCs P7 | Exosomes hDPSCs P4 | Exosomes hDPSCs P7 | |
| Neurodifferentiated hDPSCs CM P4 | ns | **** | ** | **** | **** | |
| Neurodifferentiated hDPSCs CM P7 | *** | *** | **** | **** | ||
| CM hDPSCs P4 | **** | * | ns | |||
| CM hDPSCs P7 | **** | **** | ||||
| Exosomes hDPSCs P4 | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




