Submitted:
29 August 2025
Posted:
29 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
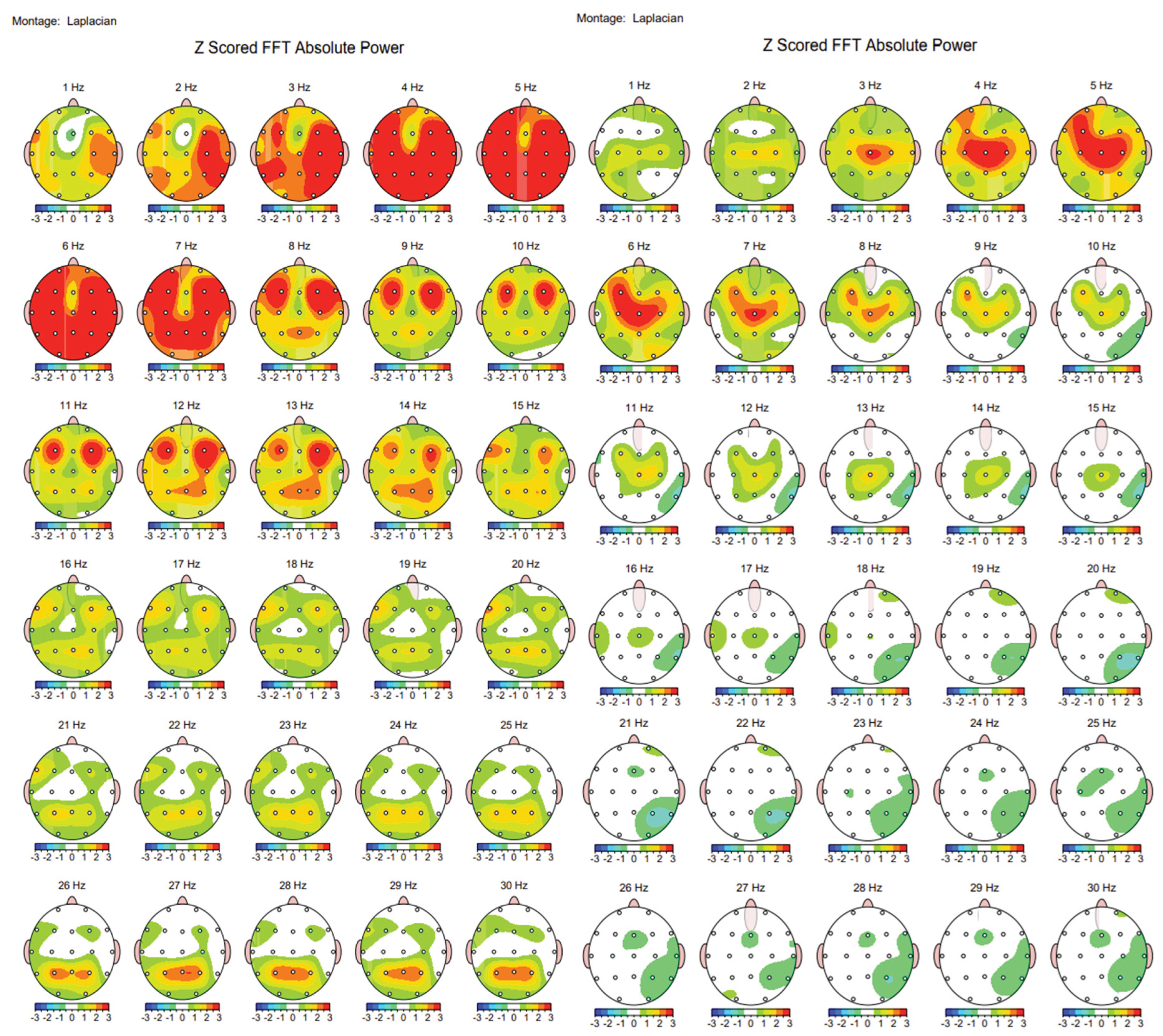
Case Description
Discussion
Acknowledgment
References
- Bredesen, D.E.; Sharlin, K.; Jenkins, D.; Okuno, M.; Youngberg, W.; Cohen, S.H.; Stefanko, A.; Brown, R.L.; Chung, H.; Chwa, W.J.; Jarrett, M.; Hathaway, A.; Toups, K.; Wilkinson, J.; Mellington, M.; Henneberg, A. Reversal of cognitive decline: 100 patients. Journal of Alzheimer's Disease & Parkinsonism 2018, 8, 450. [Google Scholar] [CrossRef]
- Toups, K.; Hathaway, A.; Gordon, D.; Chung, H.; Raji, C.; Boyd, A.; Hill, B.D.; Hausman-Cohen, S.; Attarha, M.; Chwa, W.J.; Jarrett, M.; Bredesen, D.E. Precision medicine approach to Alzheimer's disease: Successful pilot project. Journal of Alzheimer's Disease 2022, 88, 1411–1421. [Google Scholar] [CrossRef] [PubMed]
- Sandison, H.; Callan, N.G.L.; Rao, R.V.; Phipps, J.; Bradley, R. Observed improvement in cognition during a personalized lifestyle intervention in people with cognitive decline. Journal of Alzheimer's Disease 2023, 94, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Bredesen, D.E.; Ross, M.K.; Ross, S. Sustained cognitive improvement in Alzheimer's disease patients following a precision medicine protocol: Case series. Biomedicines 2024, 12, 1776. [Google Scholar] [CrossRef] [PubMed]
- Mayo Clinic Staff. (2023, December 22). Corticobasal degeneration (corticobasal syndrome) - Symptoms and causes. Mayo Clinic. Retrieved from https://www.mayoclinic.org/diseases-conditions/corticobasal-degeneration/symptoms-causes/syc-20354767.
- Kouri, N.; Whitwell, J.L.; Josephs, K.A.; Rademakers, R.; Dickson, D.W. Corticobasal degeneration: a pathologically distinct 4R tauopathy. Nature Reviews Neurology 2011, 7, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Walsh, L.; Hill, C.; Ross, R.P. Impact of glyphosate (Roundup™) on the composition and functionality of the gut microbiome. Gut Microbes 2023, 15, 2263935. [Google Scholar] [CrossRef] [PubMed]
- Caballero, M.; Amiri, S.; Denney, J.T.; Monsivais, P.; Hystad, P.; Amram, O. Estimated residential exposure to agricultural chemicals and premature mortality by Parkinson's disease in Washington State. International Journal of Environmental Research and Public Health 2018, 15, 2885. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.F.; Kunkle, G.A.; Stepien, R.L. Cytotoxicity of 4-hydroxyanisole and tyrosinase activity in variant cell lines of B16 melanoma. Pigment Cell Research 1989, 2, 525–530. [Google Scholar] [CrossRef]
- Terre'Blanche, G.; Heyer, N.; Bergh, J.J.; Mienie, L.J.; van der Schyf, C.J.; Harvey, B.H. The styrene metabolite, phenylglyoxylic acid, induces striatal-motor toxicity in the rat: influence of dose escalation/reduction over time. Neurotoxicity Research 2011, 20, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huo, W.-B.; Deng, J.-Y.; Tang, Q.-P.; Wang, J.-X.; Liao, Y.-L.; Gou, D.; Pei, D.-S. Neurotoxicity and the potential molecular mechanisms of mono-2-ethylhexyl phthalic acid (MEHP) in zebrafish. Ecotoxicology and Environmental Safety 2023, 265, 115516. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



