1. Introduction
Hypertension affects approximately 31.1% of the global adult population, highlighting the importance of exploring different strategies to regulate blood pressure (BP) as a key public health priority [
1]. Data from the Non-Communicable Diseases (NCD) Risk Factor Collaboration [
2] show that the prevalence of hypertension among individuals 30–79 years has doubled between 1990 and 2019, increasing to 331 million women. Hypertension is a leading risk factor for cardiovascular disease [
3], and it remains a critical global health challenge [
4]. Acute reductions in systolic BP ranging from 5 to 8 mmHg have been associated with clinically meaningful cardiovascular risk attenuation, representing a relevant non-pharmacological strategy for early hemodynamic modulation [
5]. Thus, strategies to acutely reduce the BP have been explored. For instance, Monteiro et al. [
6] reported a reduction in systolic BP of 13±7 mmHg among normotensive individuals following an upper cervical spinal manipulation (CM).
Although no direct relationship has been established between the CM and the reduction of the BP, there appears to be a compensatory interplay between hemodynamic and autonomic responses following sympathetic-vagal stimulation. The autonomic nervous system (ANS), which includes the parasympathetic and sympathetic divisions, is an integral part of the nervous system's role in maintaining homeostasis by regulating the functions of cells, tissues, and organs [
7]. Control of this system is exerted by higher brain centers, including the limbic system, hypothalamus, and specific brainstem nuclei [
7]. Cardiac ANS activity is commonly assessed indirectly using non-invasive markers such as heart rate variability (HRV) [
8]. In this context, Lastova et al. [
9], Picchiottino et al. [
10], and Jupin et al. [
11] demonstrated changes in sympatho-vagal balance after myofascial release and joint manipulation, respectively.
Physical therapists, chiropractors, and osteopaths frequently utilize spinal manipulations to restore musculoskeletal function [
12,
13]. Nevertheless, tissue mobilization techniques (e.g., sliding manual massage (MM)) are often used as an adjunct to treatment protocols. Previous literature suggests distinct neurophysiological effects in response to CM [
14,
15,
16] and MM [
17]. However, their effects are not yet fully understood, especially regarding BP and HRV responses. A limited studies examining the effects of upper CM on BP responses [
18,
19,
20,
21,
22] and its influence on cardiac electrophysiology, HRV and electrocardiogram parameters [
19,
21]. The MM literature includes a few studies examining the impact of tissue mobilization on BP responses [
23,
24]. Notably, only one of these studies specifically examined women [
23], and none examined HRV as an outcome.
Thus, this study aimed to compare the acute effects of CM or to simulated CM (Sham) and Control on HRV and BP responses in asymptomatic individuals. The present study had two initial hypotheses. The first hypothesis suggests that the CM would demonstrate better control of the sympatho-vagal balance compared to the Control and Sham. This hypothesis is based on the anatomical and physiological context of vagus nerve stimulation [
25], specifically the proximal branch that passes bilaterally through the cervical region [
26]. The second hypothesis proposes a higher magnitude of systolic BP reduction through the MM condition compared to the Control and Sham. This hypothesis is based on physiological mechanisms previously proposed for a similar technique performed with foam rolling, which showed higher concentrations of nitric oxide [
27], improved arterial perfusion in the lateral thigh region [
28], and promoted reductions in systolic BP [
29,
30].
2. Materials and Methods
2.1. Study Design
A double-blind, three-arm, parallel-group, randomized, crossover, placebo-controlled trial reported accordance with the Consolidated Standards of Reporting Trials (CONSORT) [
31] and Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) [
32] guidelines. The protocol received approval from the Research Ethics Committee of the Augusto Motta University Center (IRB No. 7.058.455) and was prospectively registered in the Brazilian Registry of Clinical Trials (RBR-47274gx; WHO Universal Trial Number: U1111-1252-3077). All the participants signed the informed consent form before the participation.
2.2. Participants
The trial was conducted at the Musculoskeletal Performance Laboratory, Postgraduate Program in Rehabilitation Science, Rio de Janeiro, Brazil. Participants were recruited by invitation through an announcement in the Augusto Motta University Center (undergraduate and postgraduate program) via social networks. Participants were included if they (1) aged between 19 and 44 years, (2) only women, (3) presented rest systolic and diastolic BP values within the normative range of <120–139 mmHg and <70–89 mmHg, and (4) engaging in at least 300 minutes of physical activity per week. Participants were excluded if they (1) demonstrate any clinical or functional alteration of the basilar artery during the initial vertebrobasilar integrity screening and/or (2) report habitual intake of caffeine-based supplements, or consume any other substances known to influence basal metabolic rate or autonomic function. These criteria ensure participant safety and minimize potential confounding factors during the application of spinal manipulative procedures. Participation in the study were entirely voluntary. Individuals who decline participation or are unable to complete the study for any reason were classified as dropouts.
2.3. Procedures
Participants attended four laboratory sessions, each separated by a 48-hour interval to minimize potential carryover effects. A Latin square randomization approach was employed to assign participants to one of four experimental conditions: (1) CM: bilateral high-velocity, low-amplitude manipulation of the upper cervical spine (C0–C2); (2) MM: a single 120-second session of MM release applied unilaterally to the anterior and posterior thigh, posterior lower leg, and lumbar musculature; (3) Sham: mimicking the positioning used in CM without the application of thrust manipulation; or (4) Control: rest without intervention. Participants were informed that they would undergo a CM, a Sham, or an MM, and that all procedures were potentially therapeutic. Following this briefing, participants were directed to the licensed physical therapist responsible for administering the assigned intervention. Both participants and the researcher responsible for data analysis were blinded to the intervention each participant received, thereby maintaining a double-blind design throughout the study. Conversely, the investigator administering the interventions was aware of the specific condition assigned to each participant on each experimental day.
For each experimental session, HRV, systolic and diastolic BP were measured at rest (Baseline) and every 15 minutes for 60 minutes after each intervention (Post-0, Post-15, Post-30, Post-45, and Post-60). All procedures were performed in the morning to avoid any confounding effect of circadian rhythm on HRV and BP. Regarding temperature, ambient temperature was kept constant at levels unlikely to activate thermoregulatory responses, thereby minimizing potential interference with HRV measurements.
Participants were recommended to maintain their eating and sleeping habits during the ten days of data collection. Prior to the study, all participants received verbal and written explanations of all study procedures and completed the Physical Activity Readiness Questionnaire [
33]. Following the completion of the baseline assessment, the primary evaluator (Examiner 1) exited the testing area to maintain blinding regarding condition allocation. Subsequently, a licensed physical therapist (Examiner 2), trained and experienced entered to administer the CM or the corresponding Sham or MM intervention, as determined by the randomization condition. Upon completion of the intervention, Examiner 2 left the room, allowing Examiner 1 to re-enter and conduct the identical post-intervention assessments using the same standardized protocols implemented at baseline.
2.4. Randomization
Randomization and allocation concealment were performed using sequentially numbered, opaque, sealed envelopes (1–15), each containing a pre-determined assignment card. An independent researcher, who had no involvement in the assessment or delivery of interventions, was responsible for implementing the allocation. Envelopes were opened only after participants completed informed consent and baseline testing. Each participant was assigned a unique study ID to ensure anonymity. To assess the effectiveness of blinding between the CM and Sham conditions, participants were asked to indicate which intervention they believed they had received following post-intervention HRV and BP assessments. Additionally, they were asked to report their perception of the intervention’s efficacy by answering the question: “Do you believe the intervention you received has a therapeutic effect?”. These assessments were conducted by a blinded research assistant uninvolved in any other stage of the trial.
2.5. Interventions
Cervical Manipulation (CM): Initially, two tests were performed to assess the sensitivity of various vascular and neural branches in the neck region, which served to determine whether to proceed with CM. The first test conducted was the vertebral artery sensitivity test (basilar artery) (34). For this test, the participant was positioned in the supine position while the physical therapist held the participant's head off the treatment table and performed a full backward tilt and cervical rotation to one side with the participant's eyes open for 20 seconds. The test was considered positive if the participant reported headache, nausea, nystagmus, or any event related to hypoxia. Next, the Adson test was performed, in which the physical therapist carried out combined movements of extension, external rotation, and abduction in a stretched position while palpating the participant’s radial pulse [
35]. Both tests were considered positive if the magnitude of the radial pulse decreased.
CM was performed using the Toggle Recoil technique, a specialized method characterized by a high-velocity and low-amplitude thrust applied manually between the C0 and C2 vertebrae [
36], without any rotational movement. A physical therapist, with more than three years of clinical experience with this specific technique, was responsible for applying the CM technique to all participants. The participants were positioned on the treatment table in the lateral decubitus position, with the side to be treated facing upward (the technique was applied unilaterally). The physical therapist stood behind the participant and gently placed the pisiform area of one hand on the skin between the C0 and C2 vertebrae, while using the other hand to stabilize the wrist. A rapid triceps contraction was then performed by the physical therapist, resulting in a rapid elbow extension (toggle), immediately followed by a recoil (recoil). Randomization of the side to receive the CM condition followed a Latin square strategy, which ensured that the participant remained blinded to the treated side.
Manual Massage (MM): For the MM condition, a design adapted by Monteiro et al. [
23] was used but adapted to the muscles of the lower limbs. A physical therapist with more than eight years of clinical experience with this specific technique, was responsible for applying the MM technique to all participants. The physical therapist performed a caudal to cranial MM release slip (palmar region) and applied, unilaterally, in random order, a single 120-second set to the anterior (i.e., quadriceps) and posterior (i.e., hamstrings) thigh, posterior (i.e., calf) leg, and trunk (i.e., multifidus) regions. Randomization of the segment to receive the MM condition followed a Latin square strategy, which ensured that the participant remained blinded to the treated side. Anterior thigh positions were performed lying in a supine position between the acetabulum and the quadriceps tendon. Posterior thigh and leg were performed in lying (prone) position between the ischial tuberosity and popliteal fossa, and Achilles tendon and popliteal fossa, respectively. The multifidus region was treated with the participant in a prone position, targeting the area between the sacral base and the proximal transitional zone of the spine, specifically between C7 and T1. The physical therapist would slide the fingers or hands over the skin in the target region while the participant remained lying and relaxed. MM was applied at different angles to target all areas with controlled pressure by a pain level scale score of 6 out of 10. Participants were instructed to maintain their usual respiratory pattern throughout all MM protocols. MM protocols were performed at the same time of day to avoid possible diurnal variations.
Simulated CM (SHAM): SHAM condition was performed similarly to the CM, except for the absence of CM. During this condition, the participants were positioned laterally on the treatment table with the side to be manipulated facing upward. The physical therapist stood behind the participant, gently placing the pisiform area of one hand on the skin over the C0 to C2 region and the other hand on the cervical drop mechanism of the table. The intentional disengagement of the cervical drop was then executed and lightly accompanied by the other hand, allowing the participants to hear the drop mechanism while being unaware of the absence of manipulation.
Control: The Control condition included no interventions (CM, MM, or Sham); only HRV and BP measurements were taken. Participants arrived at the laboratory and were positioned supine on the treatment table for 10 minutes to acclimatize to the temperature, lighting, and noise of the environment. Following this acclimatization period, baseline and "post-condition" values were collected, with an 8-minute interval (equivalent to the duration of the MM intervention) between these two time points.
2.6. Outcome Measures
Blood Pressure: Systolic and diastolic BP were measured using an automatic oscillometric device (Omron Hem 7113, São Paulo, Brazil) [
30]. Measurements were taken on the left arm, following the recommendations of the American Heart Association [
37]. For optimal analysis and comparison of BP responses, the following time points were established: a) Resting (Baseline); b) immediately post-session (1st time point); c) 15 minutes post-session (2nd time point); d) 30 minutes post-session (3rd time point); e) 45 minutes post-session (4th time point); f) 60 minutes post-session (5th time point).
Heart Rate Variability (HRV): HRV recordings were performed with participants lying in the supine position. The Polar H10 heart rate monitor was connected to a chest strap, secured firmly yet comfortably around the thorax, with the sensor and electrode positioned at the lower third of the sternum (approximately over the xiphoid process). The heart rate monitor was paired via Bluetooth with the Elite HRV app installed on an iPhone [
38,
39]. Data were collected for 15 minutes at rest (Baseline) and up to 60 minutes (Post-0, Post-15, Post-30, Post-45, Post-60) after the experimental condition of the day.
Measurements were taken in a quiet room with participants in a supine position, minimal external noise, and temperature controlled between 20 and 22.8°C. Raw iR-R data from the Elite HRV app were exported and subsequently analyzed using specialized software (Kubios, V.2.0, Kuopio, Finland). Time-domain and frequency-domain analyses were performed. Fast Fourier transform was used for frequency domain analysis. HRV parameters assessed were RMSSDms, SDNNms, LNms, pNN50%, Mean iR-Rms, Total Powerms2, LF Power ms2, HF Power ms2, and LF/HF Ratio. These indexes provide insights into sympathetic and parasympathetic activities of the central nervous system (CNS) [
8]. For optimal analysis and comparison of HRV responses, the following time points were established: a) Resting (Baseline); b) immediately post-session (1st time point); c) 15 minutes post-session (2nd time point); d) 30 minutes post-session (3rd time point); e) 45 minutes post-session (4th time point); f) 60 minutes post-session (5th time point).
2.7. Data Analysis
Fifteen healthy [
40] women with normal BP were recruited based on an a priori sample size calculation (effect size = 0.50; 1-β = 0.85; α = 0.05; nonsphericity correction = 1.0) [
41], based values on BP Monteiro et al. [
23] study, using G*Power [
42] indicated that fourteen participants would be adequate to achieve the statistical power.
The normality of the data distribution was assessed using the Shapiro-Wilk test and graphical inspection of histogram and QQ-plots. Normality was rejected for all variables examined. In this scenario, median and interquartile range were used to as measures of central tendency and dispersion, respectively and inferential analyses were conducted using the Friedman Test [
43]. The significance level was set at 5%. Adjusted pairwise comparisons were automatically conducted by the software in case of a statistically significant difference. When no statistically significant difference was found, Wilcoxon signed rank tests were conducted aiming to assess effect sizes. Effect sizes were used to interpret within condition results according to the following Cohen´s guidelines for r: .1 (small effect), .3 (medium effect), and .5 (large effect) [
43]. IBM SPSS Statistics 20 software was used for analysis (SPSS, Chicago, III).
3. Results
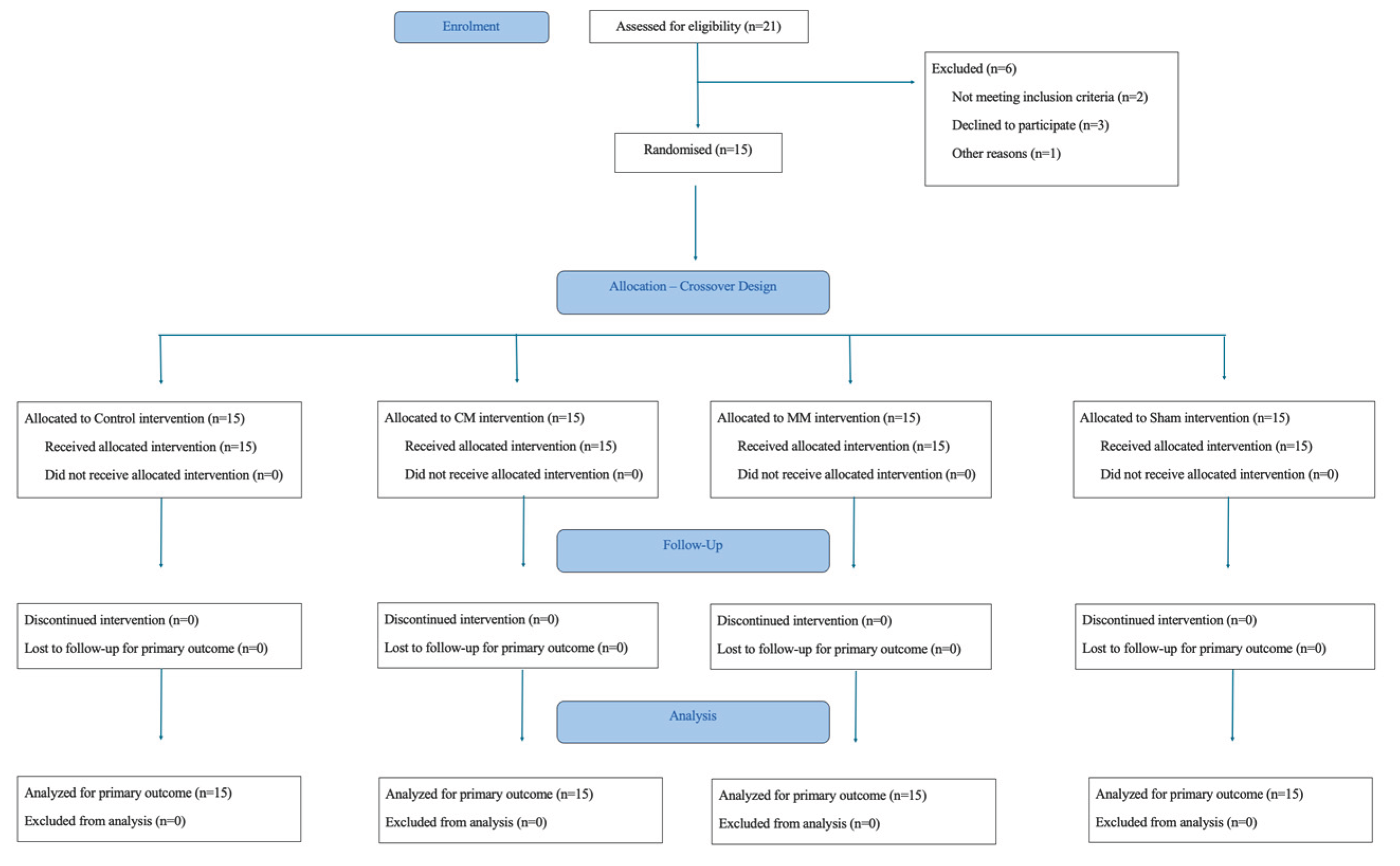
Figure 1 presents a flowchart showing the number of eligible participants excluded and the reason for their exclusion. All participants completed the study and provided data on all outcome measures.
The participants' characteristics are described in
Table 1.
3.1. Blood Pressure
No statistically significant differences were observed in Systolic BP across any of the time points (
Table 2). The only statistically significant difference observed was in Diastolic BP within the Control condition (Post-45 vs Baseline; p=0.004) (
Table 3).
3.2. Heart Rate Variability – Time-Domain
Within condition evaluations of RMSSD
ms index significantly increased in the Control condition between Post-0 and Baseline (p=0.032; r=0.56) and between Post-15 and Baseline (p=0.023; r=0.57) (
Table 4). Similarly, in the Sham condition, RMSSD
ms index significantly increased between Post-15 and Baseline (p=0.014; r=0.60) (
Table 4). Lastly, in the CM condition, RMSSD
ms index significantly increased between Post-15 and Baseline (p=0.027; r=0.60) and between Post-60 and Baseline (p=0.010; r=0.17) (
Table 4). A statistically significant difference was observed between CM and MM conditions at the Post-15 time point (p=0.049); however, this difference was no longer significant following pairwise comparisons (
Table 4).
Significant increases in the SDNN
ms index were observed in the Control condition between Post-45 and Baseline (p=0.037; r=0.55) (
Table 4). In the CM condition, significant increases were noted between Post-45 and Baseline (p=0.014; r=0.60) and between Post-60 and Baseline (p=0.019; r=0.58) (
Table 4). Similarly, in the MM condition, increases were observed between Post-60 and Baseline (p=0.019; r=0.58) (
Table 4). No statistically significant differences were found between conditions at any time point (
Table 4). Regarding the analyses of pNN50% index, no statistically significant differences were observed between any time points in the Control, Sham, and MM conditions after pairwise comparisons. However, within condition analyses in the CM condition revealed statistically significant increases between Post-0 and Baseline (p=0.044; r=0.54), Post-15 and Baseline (p=0.044; r=0.54), and Post-60 and Baseline (p=0.019; r=0.58) (
Table 4). No statistically significant differences between protocols were identified at any time point (
Table 4).
Within condition analyses of LN(RMSSD) index revealed statistically significant differences in all conditions except for the MM condition (
Table 4). In the Control condition, significant increases were observed between Post-0 and Baseline (p=0.027; r=0.57). In the Sham condition, significant increases occurred between Post-15 and Baseline (p=0.011; r=0.61). For the CM condition, significant increases were identified between Post-15 and Baseline (p=0.027; r=0.57) and between Post-60 and Baseline (p=0.010; r=0.62) (
Table 4). No statistically significant differences were found between protocols at any time point. For the mean iR-R, within condition analyses revealed statistically significant differences across all conditions (
Table 4). In the Control condition, differences were observed at all time points (p<0.05;
Table 4). In the Sham condition, significant increases were identified between Post-0 and Baseline (p=0.027; r=0.57) and between Post-15 and Baseline (p=0.010; r=0.62) (
Table 4). For the CM condition, significant increases were observed at all investigated time points (p<0.05;
Table 4), except between Post-0 and Baseline (p=0.051) (
Table 4). Lastly, in the MM condition, significant increases were noted between Post-45 and Baseline (p<0.001; r=0.73) and between Post-60 and Baseline (p=0.019; r=0.58) (
Table 4). No statistically significant differences were observed between protocols at any time point.
3.3. Heart Rate Variability – Frequency-Domain
Significant within-condition increases in Total Power (ms
2) index were observed in the Control, CM, and MM conditions (
Table 5). In the Control condition, the significant difference was noted between Post-45 and Baseline (p=0.027; r=0.57). In the CM condition, differences were observed between Post-30 and Baseline (p=0.037; r=0.55) and between Post-60 and Baseline (p=0.001; r=0.74). In contrast, for the MM condition, the observed differences were diluted after pairwise comparisons. No statistically significant differences were found between protocols at any time point.
Analyses of the LF Power (ms
2) index revealed a statistically significant difference in the CM condition between Post-60 and Baseline (p=0.001; r=0.71) (
Table 5). No statistically significant differences were observed between protocols at any time point. Regarding the HF Power (ms
2) index, a statistically significant difference was noted in the Sham condition, but this difference was diluted after pairwise comparisons (
Table 5). Similarly, no statistically significant differences between protocols were found at any time point. For the LF/HF ratio, a within-condition statistically significant difference was identified in the MM condition between Post-60 and Baseline (p=0.022; r=0.55). Additionally, statistically significant differences were observed between the MM and Control conditions (p=0.014) and between the MM and CM conditions (p=0.043).
4. Discussion
The main results were significant increases (p<0.05) within conditions, with a large effect size for RMSSD
ms, SDNN
ms, PNN50%, LF Power (ms
2), and LF/HF ratio. The results of the present study partially confirm the first hypothesis, which suggested that the CM condition would present better sympatho-vagal control compared to the Control and Sham conditions. In the within-condition analysis, only the PNN50% index (
Table 4) and LN Power (ms
2) (
Table 5) showed significant increases for CM, with no significant changes observed for Control and Sham. These results indicate an increase in both parasympathetic control (PNN50%) and sympathetic control (LF power (ms
2)), but without predominance between them, which was reflected in the lack of significance in the LF/HF ratio (
Table 5). Even without active intervention, factors such as the placebo effect and natural adaptive physiological processes can lead to improvements or noticeable changes in participants' conditions [44-46]. However, the fact that significant increases were observed in both the Control and Sham conditions, with no difference compared to the CM condition, prevents the inference of better effects for the experimental condition.
Nevertheless, Win et al. [
21] investigated the effect of upper CM on sympatho-vagal modulation by HRV measurement and found different results from the present investigation. These authors observed an increase in HRV parameters, which may indicate a greater influx of vagal nerve activity. In addition, there was a decrease in LF
nu parameters (p=0.01) and in the sympatho-vagal balance (HF/LF ratio), confirming the positive effects of upper CM on autonomic modulation. On the other hand, Picchiottino et al. [
10] compared the effects of manipulation of the mid-thoracic region (T5 vertebra) and the sham effect on HRV responses. Picchiottino et al. [
10] found an increase in the iR-R, as well as in the HF
nu (vagal pathway) and LFnu (vagal and sympathetic pathway) parameters, LF/HF ratio, RMSSD
ms (vagal pathway), and[44–46 SDNN
ms (vagal and sympathetic pathway). The results by Picchiottino et al. [
10] suggest that there is no relationship between T5 manipulation and autonomic modulation, as both parameters (parasympathetic and sympathetic) increased, thus reinforcing the mixed action of the vagus nerve and supporting the findings of the present study. The results described here suggest a possible relationship between manipulation and vagus nerve stimulation for predominantly parasympathetic input, regardless of the location (anatomical height) of this stimulus, a fact that was not predominantly observed in the results of the present study.
The second hypothesis proposes a higher magnitude of systolic BP reduction through the MM condition compared to the Control and Sham. This hypothesis is based on physiological mechanisms previously proposed for a similar technique performed with foam rolling, which demonstrated higher concentrations of nitric oxide [
27] and improved arterial perfusion in the lateral thigh region [
28]. The results of the present study do not support this hypothesis, as no significant reduction in BP values was observed (
Table 2 and
Table 3). Contrary to the results of the present study, but without comparing experimental conditions to placebo, Liao et al. [
47] conducted a systematic review with meta-analysis and found that massage techniques significantly contributed to reductions in systolic (-7.39 mmHg; effect size = -0.728) and diastolic (-5.04 mmHg; effect size = -0.334) BP. Regarding the results related to sliding MM in women, the literature only points to the study by Monteiro et al. [
23], which aimed to evaluate this. In contrast to the results of the present study, Monteiro et al. [
23] observed reduction in systolic BP at 50 (p=0.011; r=2.61; ∆=-4.0 mmHg) and 60 (p=0.011; r=-2.74; ∆=-4.0 mmHg) minutes after application of MM as an experimental condition.
Similarly, but using foam rolling as an intervention strategy, Lastova et al. [
9] observed a reduction in systolic BP, which differs from the findings of the present study. Lastova et al. [
9] investigated BP response 10 and 30 minutes after a foam rolling session for the thigh (adductors, posterior, anterior, and lateral), calf (gastrocnemius), and back (upper and lower). The authors observed a significant decrease in systolic BP with a concomitant increase in vagal modulation up to 30 minutes after foam rolling. It is worth noting that Lastova et al. [
9] measured BP only 30 minutes after foam rolling, leaving a gap in interpretation as to whether this reduction may persist beyond that time point. The specific physiological mechanisms underlying the BP response has been previously studied. The first to investigate potential effects related to BP control were Okamoto et al. [
27], who observed a higher concentration of nitric oxide after foam rolling, indicating a greater vasodilatory effect that could reduce systolic BP and heart rate. Hotfiel et al. [
28] observed increased local arterial perfusion in the lateral thigh region after foam rolling. The authors also associate these changes with vasodilation caused by increased nitric oxide after FR. Therefore, the fact that different techniques (e.g., MM and foam rolling) elicit the same central responses suggests that these effects may be attributed to therapeutic touch [
48,
49,
50], which triggers responses at the level of the ANS. It is hypothesized that mechanoreceptors located in muscle and fascia, when activated, reduce muscle tone, thereby promoting an increase in parasympathetic response and the release of neuropeptides and endocannabinoids, leading to a subsequent reduction in blood pressure [
17].
An important consideration in interpreting the results of this study is the paced nature of the MM slide condition, both within and between individuals. This design feature can be viewed as both a limitation and a strength. On the one hand, the lack of strict control may reduce the internal validity of the results, as the number and duration of slides could influence the outcomes. On the other hand, allowing the slide to be freely paced increases ecological validity and generalizability and better reflects real-world rehabilitation scenarios. All participants in this study were women. Notably, resting cardiovascular parameters do not appear to vary significantly with different phases of the menstrual cycle. Interestingly, Queiroz et al. [
51] highlighted those certain phases of the menstrual cycle, such as menstrual bleeding, may influence the magnitude of BP reduction but not its overall occurrence. Although caution is warranted in generalizing these findings, it is essential to emphasize that the primary aim of this study was not to investigate underlying mechanisms but rather to assess BP. We intentionally chose not to control for menstrual cycle phases in order to better represent real-world conditions, thereby enhancing the ecological validity of the findings. In this context, the inclusion of women participants does not pose significant challenges in extrapolating the results to men populations. The main differences between the sexes are primarily in the underlying physiological mechanisms rather than in the BP responses themselves [
51].
5. Conclusions
In conclusion, the effect size findings suggest that both SM and MM may promote autonomic modulation in normotensive women by maintaining a balanced sympathovagal response. However, these outcomes were not consistently accompanied by statistically significant differences. Therefore, caution will be warranted when interpreting and applying these results, as they reveal an emerging gap in the literature that warrants further investigation. Testing was performed in apparently healthy women with normal BP; therefore, it may not be generalizable to the men and/or the hypertensive population.
Author Contributions
Conceptualization, E.R.M. and D.M-G.; methodology, E.R.M, R.C.deS., and D.M-G.; validation, E.R.M., L.S.P., G.H.G., F.deO., R.C.deS., L.A.C.N., A.J.L., and D.M-G.; formal analysis, E.R.M. and F.de.O.; investigation, E.R.M., G.H.G., F.deO. and R.C.deS.; resources, E.R.M., G.H.G., F.deO. and R.C.deS.; data curation, E.R.M. and F.deO..; writing—original draft preparation, E.R.M., L.S.P., G.H.G., F.deO., R.C.deS., L.A.C.N., A.J.L., and D.M-G.; writing—review and editing, E.R.M., L.S.P., G.H.G., F.deO., R.C.deS., L.A.C.N., A.J.L., and D.M-G.; visualization, E.R.M., L.S.P., G.H.G., F.deO., R.C.deS., L.A.C.N., A.J.L., and D.M-G.; supervision, A.J.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of the Augusto Motta University Center (IRB No. 7.058.455; approved on September 6, 2024) and was prospectively registered in the Brazilian Registry of Clinical Trials (RBR-47274gx; WHO Universal Trial Number: U1111-1252-3077).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
No acknowledgment is required. No artificial intelligence tools were used in the preparation of this manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ANS |
Autonomic Nervous System |
| BP |
Blood Pressure |
| CONSORT |
Consolidated Standards of Reporting Trials |
| CM |
Cervical Spinal Manipulation |
| HRV |
Heart Rate Variability |
| MM |
Manual Massage |
| NCD |
Non-Communicable Diseases |
| SHAM |
Simulated Cervical Spinal Manipulation |
| SPIRIT |
Standard Protocol Items: Recommendations for Interventional Trials |
References
- Mills, K.T.; Stefanescu, H.J. The global epidemiology of hypertension. Nat Rev Nephrol 2020, 16, 223–237. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet 2021, 398, 957–980. [Google Scholar] [CrossRef]
- Schiffrin, E.L. Immune mechanisms in hypertension and vascular injury. Clin Sci (Lond) 2014, 126, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ringwald-de Meyer, S.; de La Harpe, R.; Vollenweider, P.; Marques-Vidal, P.; Vaucher, J. Factor associated with and risk of cardiovascular disease in people with uncontrolled hypertension. Sci Rep 2025, 15, 375. [Google Scholar] [CrossRef]
- Pescatello, L.S.; Buchner, D.M.; Jakicic, J.M.; et al. Physical activity to prevent and treat hypertension: a systematic review. Med Sci Sports Exerc 2019, 51, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, E.R.; Pescatello, L.S.; Leitão, L.; et al. Muscular performance and blood pressure after different pre-strength training strategies in recreationally strength-trained women: cross-over trial. J Cardiovasc Dev Dis 2025, 12, 7. [Google Scholar] [CrossRef]
- Jänig, W. The integrative action of the autonomic nervous system: neurobiology of homeostasis. Cambridge, UK.; New York: Cambridge University Press, 2006. [Google Scholar]
- Vanderlei, L.C.M.; Pastre, C.M.E.; Hoshi, R.A.; De Carvalho, T.D.; De Godoy, M.F. Basic notions of heart rate variability and its clinical applicability. Rev Bras Cir Cardiovasc 2009, 24, 205–217. [Google Scholar] [CrossRef]
- Lastova, K.; Nordvall, M.; Walters-Edwards, M.; Allnutt, A.; Wong, A. Cardiac autonomic and blood pressure responses to an acute foam rolling session. J Strength Cond Res 2018, 32, 2825–2830. [Google Scholar] [CrossRef] [PubMed]
- Picchiottino, M.; Honoré, M.; Leboeuf-Y, de C. ; Gagey, O.; Cottin, F.; Hallman, D.M. The effect of a single spinal manipulation on cardiovascular autonomic activity and the relationship to pressure pain threshold: a randomized, cross-over, sham-controlled trial. Chiropr Man Therap 2020, 28, 7. [Google Scholar] [CrossRef]
- Jupin, C.; Aibar, V.B.; Sarhan, F.R. Short-Term Effects of Spinal Manual Therapy on the Nervous System in Managing Musculoskeletal Pain: A Systematic Review. J Clin Med 2025, 14, 3830. [Google Scholar] [CrossRef]
- Bell, S.; D’Angelo, K.; Kawchuk, G.N.; Triano, J.J.; Howarth, S.J. Procedure selection and patient positioning influence spine kinematics during high-velocity, low-amplitude spinal manipulation applied to the low back. J Manip Physiol Ther 2017, 40, 147–155. [Google Scholar] [CrossRef]
- Hidalgo, B.; Hall, T.; Bossert, J.; Dugeny, A.; Cagnie, B.; Pitance, L. The efficacy of manual therapy and exercise for treating non-specific neck pain: a systematic review. J Back Musculoskelet Rehabil 2017, 30, 1149–1169. [Google Scholar] [CrossRef]
- Gross, A.; Langevin, P.; Burnie, S.J.; et al. Manipulation and mobilisation for neck pain contrasted against an inactive control or another active treatment. Cochrane Database Syst Rev 2015, CD004249. [Google Scholar] [CrossRef] [PubMed]
- Coulter, I.D.; Crawford, C.; Hurwitz, E.L.; et al. Manipulation and mobilization for treating chronic low back pain: a systematic review and meta-analysis. Spine J 2018, 18, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, S.M.; de Zoete, A.; Van Middelkoop, M.; Assendelft, W.J.J.; De Boer, M.R.; Van Tulder, M.W. Benefits and harms of spinal manipulative therapy for the treatment of chronic low back pain: systematic review and meta-analysis of randomised controlled trials. BMJ 2019, 364, l689. [Google Scholar] [CrossRef]
- Beardsley, C.; Škarabot, J. Effects of self-myofascial release: A systematic review. J Bodyw Mov Ther 2015, 19, 747–758. [Google Scholar] [CrossRef]
- Knutson, G.A. Significant changes in systolic blood pressure post vectored upper cervical adjustment vs resting control groups: a possible effect of the cervico sympathetic and/or pressor reflex. J Manipulative Physiol Ther 2001, 24, 101–109. [Google Scholar] [CrossRef]
- Ward, J.; Tyer, K.; Coats, J.; Williams, G.; Weigand, S.; Cockburn, D. Immediate effects of atlas manipulation on cardiovascular physiology. Clinical Chiropractic 2012, 15, 147–157. [Google Scholar] [CrossRef]
- Yung, E.; Wong, M.; Williams, H.; Mache, K. Blood pressure and heart rate response to posteriorly directed pressure applied to the cervical spine in young, pain-free individuals: a randomized, repeated-measures, double-blind, placebo-controlled study. J Orthop Sports Phys Ther 2014, 2014 44, 622–626. [Google Scholar] [CrossRef]
- Win, N.N.; Jorgensen, A.M.S.; Chen, Y.S.; Haneline, M.T. Effects of Upper and Lower Cervical Spinal Manipulative Therapy on Blood Pressure and Heart Rate Variability in Volunteers and Patients With Neck Pain: A Randomized Controlled, Cross-Over, Preliminary Study. J Chiropr Med 2015, 14, 1–9. [Google Scholar] [CrossRef]
- Goertz, C.M.; Salsbury, S.A.; Vining, R.D.; et al. Effect of Spinal Manipulation of Upper Cervical Vertebrae on Blood Pressure: Results of a Pilot Sham-Controlled Trial. J Manipulative Physiol Ther 2016, 39, 369–380. [Google Scholar] [CrossRef]
- Monteiro, E.R.; Pescatello, L.S.; Winchester, J.B.; et al. Effects of Manual Therapies and Resistance Exercise on Postexercise Hypotension in Women With Normal Blood Pressure. J Strength Cond Res 2022, 36, 948–954. [Google Scholar] [CrossRef]
- Monteiro, E.R.; Budde, H.; Silva, J.G.; Oliveira, A.; Novaes, J.S.; de Jesus, I.R.T. Enhance range-of-motion and hypotensive effect in elderly submitted to three manual therapy techniques: Cross-over study. J Bodyw Mov Ther 2024, 37, 238–245. [Google Scholar] [CrossRef]
- Courties, A.; Berenbaum, F.; Sellam, J. Vagus nerve stimulation in musculoskeletal diseases. Joint Bone Spine 2021, 88, 105149. [Google Scholar] [CrossRef]
- Cunningham, C.J.; Martínez, J.L. The Wandering Nerve: Positional Variations of the Cervical Vagus Nerve and Neurosurgical Implications. World Neurosurg 2021, 156, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Masuhara, M.; Ikuta, K. Acute effects of self-myofascial release using a foam roller on arterial function. J Strength Cond Res 2014, 28, 69–73. [Google Scholar] [CrossRef]
- Hotfiel, T.; Swoboda, B.; Krinner, S.; et al. Acute effects of lateral thigh foam rolling on arterial tissue perfusion determined by spectral doppler and power doppler ultrasound. J Strength Cond Res 2017, 31, 893–900. [Google Scholar] [CrossRef]
- Monteiro, E.R.; Vingren, J.L.; Pescatello, L.S.; et al. Effects of foam rolling and strength training on post exercise hypotension in normotensive women: a cross-over study. J Bodyw Mov Therapies 2023, 34, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, E.R.; Aguilera, L.M.; Ruá-Alonso, M.; et al. Effect of Manual Massage, Foam Rolling, and Strength Training on Hemodynamic and Autonomic Responses in Adults: A Scoping Review. Healthcare (Basel) 2025, 13, 1371. [Google Scholar] [CrossRef] [PubMed]
- Hopewell, S.; Chan, A-W. ; Collins, G.S. CONSORT 2025 statement: updated guideline for reporting randomised trials. BMJ 2025, 389, e081123. [Google Scholar] [CrossRef] [PubMed]
- Chan, A-W. ; Bourton, I.; Hopewell, S.; et al. SPIRIT 2025 statement: Updated guideline for protocols of randomised trials. PLoS Med 2025, 22, e1004589. [Google Scholar]
- Shepard, R.J. PAR-Q, Canadian Home Fitness Test and exercise screening alternatives. Sports Med 1988, 5, 185–195. [Google Scholar] [CrossRef]
- Gorrell, L.M.; Sawatsky, A.; Edwards, W.B.; Herzog, W. Vertebral arteries do not experience tensile force during manual cervical spine manipulation applied to human cadavers. J Man Manip Ther 2022, 31, 261–269. [Google Scholar] [CrossRef]
- Hixson, K.M.; Horris, H.B.; McLeod, T.C.V.; Bacon, C.E.W. The Diagnostic Accuracy of Clinical Diagnostic Tests for Thoracic Outlet Syndrome. J Sport Rehabil 2017, 26, 459–465. [Google Scholar] [CrossRef]
- Strazewski, J. The essentials of toggle recoil. 2008.
- 2020 ACC/AHA Guidelines for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021, 143, 143–227.
- Chhetri, P.; Shrestha, L.; Mahatra, N.B. Validity of elite-hrv smartphone application for measuring heart rate variability compared to polar v800 heart rate monitor. J Nepal Health Res Counc 2021, 19, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Himariotis, A.T.; Coffey, K.F.; Noel, S.E.; Cornell, D.J. Validity of a smartphone application in calculating measures of heart rate variability. Sensors 2022, 22, 9883–2022. [Google Scholar] [CrossRef] [PubMed]
- McEvoy JW, McCarthy CP, Bruno RM, et al. 2024 ESC Guidelines for the management of elevated blood pressure and hypertension. Eur Heart J 2024, 45, 3912–4018. [Google Scholar] [CrossRef] [PubMed]
- Beck, T.W. The importance of a priori sample size estimation in strength and conditioning research. J Strength Cond Res 2013, 27, 2323–2337. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Bucher, A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Pallant, J. SPSS Survival Manual: A Step By Step Guide to Data Analysis Using SPSS Program (6th ed.). London, UK: McGraw-Hill Education, 2016.
- Gould, B.A.; Mann, S.; Davies, A.B.; Altman, D.G.; Raftery, E.B. Does placebo lower blood-pressure? Lancet 1981, 2, 1377–1381. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F.; Carlino, E.; Pollo, A. How placebos change the patient's brain. Neuropsychopharmacology 2011, 36, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, M.; Winkler, A.; Rief, V.; Doering, B.K. Effect of placebo groups on blood pressure in hypertension: a meta-analysis of beta-blocker trials. J Am Soc Hypertens 2016, 10, 917–929. [Google Scholar] [CrossRef]
- Liao, I.C.; Chen, S.L.; Wang, M.Y.; Tsai, P.S. Effects of massage on blood pressure in patients with hypertension and prehy-pertension: a meta-analysis of randomized controlled trials. J Cardiovasc Nurs 2016, 31, 73–83. [Google Scholar] [CrossRef]
- Berkley, K.J.; Hubscher, C.H. Are there separate central nervous system pathways for touch and pain? Nat Med 1995, 1, 766–773. [Google Scholar] [CrossRef]
- McGlone, F.; Valbo, A.B.; Olausson, H.; Loken, L.; Wessberg, J. Discriminative touch and emotional touch. Can J Exp Psychol 2007, 61, 173–183. [Google Scholar] [CrossRef]
- Preusser, S.; Thiel, S.D.; Rook, C.; et al. The perception of touch and the ventral somatosensory pathway. Brain 2015, 138, 540–548. [Google Scholar] [CrossRef]
- Queiroz, A.C.C.; Rezk, C.C.; Teixeira, L.; Tinucci, T.; Mion, D.; Forjaz, C.L.M. Gender influence on post-resistance exercise hypotension and hemodynamics. Int J Sports Med 2013, 34, 939–944. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).