Submitted:
17 August 2025
Posted:
18 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction: Fungi as Master Builders of Programmable Matter
2. The Fungal Cell Wall: A Hierarchical Nanocomposite Blueprint
2.1. The Chitin-Glucan Scaffold: The Structural Backbone
2.2. The Glycoprotein Matrix: A Functional and Adaptive Interface
2.3. Hydrophobins: Nature's Amphipathic Surfactants
2.4. Architectural Plasticity and Interspecies Variation
3. Biochemical Levers for Tuning Mycelium Material Properties
3.1. Substrate-Driven Morphogenesis and Composition
3.2. The Fungal Secretome as an In-Situ Modification Toolkit
3.3. Environmental Signaling as a Control Mechanism: The Role of pH
4. A Synthetic Biology Toolkit for Designing "Smart" Mycelium Materials
4.1. Rational Design Through Genetic Engineering of Cell Wall Architecture
4.2. Chemical Genetics: Dynamic and Reversible Control of Material Properties
4.3. Programming Functionality: Engineering Surfaces and Bioreceptivity
4.4. Inducible Systems and Biosensors: Towards Living Functional Materials (LFMs)
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AGS | α-1,3-glucan synthase |
| BFI | Bacterial-Fungal Interactions |
| CAZymes | Carbohydrate-Active enzymes |
| CHS | Chitin Synthases |
| DNMT | DNA Methyltransferase |
| DOAJ | Directory of Open Access Journals |
| ECM | Extracellular Matrix |
| FKS / GLS | Genes encoding β-(1,3)-glucan synthase |
| GAG | Galactosaminogalactan |
| GlcNAc | N-acetyl-D-glucosamine |
| HDAC | Histone Deacetylase |
| LD | Linear Dichroism |
| LFM | Living Functional Materials |
| MDPI | Multidisciplinary Digital Publishing Institute |
| NaOH | Sodium Hydroxide |
| PDB | Potato Dextrose Broth |
| PSK | Polysaccharide-K |
| PSP | Polysaccharopeptide |
| SAHA | Suberanilohydroxamic acid |
| TLA | Three Letter Acronym |
| TSA | Trichostatin A |
| WCA | Water Contact Angle |
References
- Antinori, M.E.; Contardi, M.; Suarato, G.; Armirotti, A.; Bertorelli, R.; Mancini, G.; Debellis, D.; Athanassiou, A. Advanced mycelium materials as potential self-growing biomedical scaffolds. Sci. Rep. 2021, 11, 12630. [Google Scholar] [CrossRef] [PubMed]
- Roth, M.G.; Westrick, N.M.; Baldwin, T.T. Fungal biotechnology: From yesterday to tomorrow. Front. Fungal Biol. 2023, 4. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V. Connecting materials sciences with fungal biology: a sea of possibilities. Fungal Biol. Biotechnol. 2022, 9, 5. [Google Scholar] [CrossRef]
- Goldman, G.H. New Opportunities for Modern Fungal Biology. Front. fungal Biol. 2020, 1, 596090. [Google Scholar] [CrossRef]
- Corbu, V.M.; Gheorghe-Barbu, I.; Dumbravă, A. Ștefania; Vrâncianu, C.O.; Șesan, T.E. Current Insights in Fungal Importance—A Comprehensive Review. Microorganisms 2023, 11, 1384. [Google Scholar] [CrossRef]
- Daâssi, D.; Bouassida, M.; Almaghrabi, F.; Chamkha, M. Mycoremediation: An Innovative and Sustainable Approach. In; 2025.
- Madusanka, C.; Udayanga, D.; Nilmini, R.; Rajapaksha, S.; Hewawasam, C.; Manamgoda, D.; Vasco-Correa, J. A review of recent advances in fungal mycelium based composites. Discov. Mater. 2024, 4, 13. [Google Scholar] [CrossRef]
- Elsacker, E.; Vandelook, S.; Brancart, J.; Peeters, E.; De Laet, L. Mechanical, physical and chemical characterisation of mycelium-based composites with different types of lignocellulosic substrates. PLoS One 2019, 14, e0213954. [Google Scholar] [CrossRef] [PubMed]
- Alemu, D.; Tafesse, M.; Mondal, A.K. Mycelium-Based Composite: The Future Sustainable Biomaterial. Int. J. Biomater. 2022, 2022, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Javadian, A.; Le Ferrand, H.; E. Hebel, D.; Saeidi, N. Application of Mycelium-Bound Composite Materials in Construction Industry: A Short Review. SOJ Mater. Sci. Eng. 2020, 7, 1–9. [Google Scholar] [CrossRef]
- Yang, L.; Park, D.; Qin, Z. Material Function of Mycelium-Based Bio-Composite: A Review. Front. Mater. 2021, 8. [Google Scholar] [CrossRef]
- Antinori, M.E.; Ceseracciu, L.; Mancini, G.; Heredia-Guerrero, J.A.; Athanassiou, A. Fine-Tuning of Physicochemical Properties and Growth Dynamics of Mycelium-Based Materials. ACS Appl. Bio Mater. 2020, 3, 1044–1051. [Google Scholar] [CrossRef]
- Gow, N.A.R.; Latge, J.-P.; Munro, C.A. The Fungal Cell Wall: Structure, Biosynthesis, and Function. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rubio, R.; de Oliveira, H.C.; Rivera, J.; Trevijano-Contador, N. The Fungal Cell Wall: Candida, Cryptococcus, and Aspergillus Species. Front. Microbiol. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Billerbeck, S.; Oliveira, A.G.; Gonçalves, A.P. Editorial: Fungi as cell factories: Genetic engineering and applications. Front. Bioeng. Biotechnol. 2022, 10. [Google Scholar] [CrossRef]
- Zou, G.; Li, T.; Mijakovic, I.; Wei, Y. Synthetic biology enables mushrooms to meet emerging sustainable challenges. Front. Microbiol. 2024, 15. [Google Scholar] [CrossRef]
- Jo, C.; Zhang, J.; Tam, J.M.; Church, G.M.; Khalil, A.S.; Segrè, D.; Tang, T.-C. Unlocking the magic in mycelium: Using synthetic biology to optimize filamentous fungi for biomanufacturing and sustainability. Mater. Today Bio 2023, 19, 100560. [Google Scholar] [CrossRef]
- Kirtika Padalia, B. BIOLOGY AND DIVERSITY OF VIRUSES, BACTERIA AND FUNGI (PAPER CODE: BOT 501).
- Wessels, J.G.H. DEVELOPMENTAL REGULATION OF FUNGAL CELL WALL FORMATION. Annu. Rev. Phytopathol. 1994, 32, 413–437. [Google Scholar] [CrossRef]
- Fuertes-Rabanal, M.; Rebaque, D.; Largo-Gosens, A.; Encina, A.; Mélida, H. Cell walls: a comparative view of the composition of cell surfaces of plants, algae, and microorganisms. J. Exp. Bot. 2025, 76, 2614–2645. [Google Scholar] [CrossRef]
- Nawawi, W.M.F.B.W.; Jones, M.; Murphy, R.J.; Lee, K.-Y.; Kontturi, E.; Bismarck, A. Nanomaterials Derived from Fungal Sources—Is It the New Hype? Biomacromolecules 2020, 21, 30–55. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Hsueh, P.-H.; Ulfadillah, S.A.; Wang, S.-T.; Tsai, M.-L. Exploring the Sustainable Utilization of Deep Eutectic Solvents for Chitin Isolation from Diverse Sources. Polymers (Basel). 2024, 16, 3187. [Google Scholar] [CrossRef]
- Jones, M.; Huynh, T.; Dekiwadia, C.; Daver, F.; John, S. Mycelium Composites: A Review of Engineering Characteristics and Growth Kinetics. J. Bionanoscience 2017, 11, 241–257. [Google Scholar] [CrossRef]
- Guerriero, G.; Avino, M.; Zhou, Q.; Fugelstad, J.; Clergeot, P.-H.; Bulone, V. Chitin Synthases from Saprolegnia Are Involved in Tip Growth and Represent a Potential Target for Anti-Oomycete Drugs. PLoS Pathog. 2010, 6, e1001070. [Google Scholar] [CrossRef]
- Joo, S.-M.; Kim, Y.-G.; Kwak, Y.-J.; Yoo, D.J.; Jeong, C.-U.; Park, J.; Oh, M.-S. Enhanced Long-Term Reliability of Seal DeltaSpot Welded Dissimilar Joint between 6061 Aluminum Alloy and Galvannealed Steel via Excimer Laser Irradiation. Materials (Basel). 2021, 14, 6756. [Google Scholar] [CrossRef] [PubMed]
- Cortina-Escribano, M.; Pihlava, J.-M.; Miina, J.; Veteli, P.; Linnakoski, R.; Vanhanen, H. Effect of Strain, Wood Substrate and Cold Treatment on the Yield and β-Glucan Content of Ganoderma lucidum Fruiting Bodies. Molecules 2020, 25, 4732. [Google Scholar] [CrossRef]
- Fazli Wan Nawawi, W.M.; Lee, K.-Y.; Kontturi, E.; Murphy, R.J.; Bismarck, A. Chitin Nanopaper from Mushroom Extract: Natural Composite of Nanofibers and Glucan from a Single Biobased Source. ACS Sustain. Chem. Eng. 2019, 7, 6492–6496. [Google Scholar] [CrossRef]
- Jiménez-Ortigosa, C.; Aimanianda, V.; Muszkieta, L.; Mouyna, I.; Alsteens, D.; Pire, S.; Beau, R.; Krappmann, S.; Beauvais, A.; Dufrêne, Y.F.; et al. Chitin Synthases with a Myosin Motor-Like Domain Control the Resistance of Aspergillus fumigatus to Echinocandins. Antimicrob. Agents Chemother. 2012, 56, 6121–6131. [Google Scholar] [CrossRef]
- Kleijburg, F.E.L.; Safeer, A.A.; Baldus, M.; Wösten, H.A.B. Binding of micro-nutrients to the cell wall of the fungus Schizophyllum commune. Cell Surf. 2023, 10, 100108. [Google Scholar] [CrossRef]
- Free, S.J. Fungal Cell Wall Organization and Biosynthesis. In; 2013; pp. 33–82.
- Book of abstracts of all the posters : Barcelona, Spain, 29th June - 3rd July 2025 : precision agriculture: a reality for everyone; Universitat Politècnica de Catalunya, 2025; ISBN 979138761357.
- Kuştaş, S.; Gezer, E.D. Physical and mechanical properties of mycelium-based insulation materials produced from desilicated wheat straws - Part A. BioResources 2024, 19, 1330–1347. [Google Scholar] [CrossRef]
- Terauchi, Y.; Nagayama, M.; Tanaka, T.; Tanabe, H.; Yoshimi, A.; Nanatani, K.; Yabu, H.; Arita, T.; Higuchi, T.; Kameda, T.; et al. Adsorption Kinetics and Self-Assembled Structures of Aspergillus oryzae Hydrophobin RolA on Hydrophobic and Charged Solid Surfaces. Appl. Environ. Microbiol. 2022, 88. [Google Scholar] [CrossRef] [PubMed]
- Zangi, R.; de Vocht, M.L.; Robillard, G.T.; Mark, A.E. Molecular Dynamics Study of the Folding of Hydrophobin SC3 at a Hydrophilic/Hydrophobic Interface. Biophys. J. 2002, 83, 112–124. [Google Scholar] [CrossRef]
- Chau, H.W.; Si, B.C.; Goh, Y.K.; Vujanovic, V. A novel method for identifying hydrophobicity on fungal surfaces. Mycol. Res. 2009, 113, 1046–1052. [Google Scholar] [CrossRef]
- Morris, V.K.; Ren, Q.; Macindoe, I.; Kwan, A.H.; Byrne, N.; Sunde, M. Recruitment of Class I Hydrophobins to the Air:Water Interface Initiates a Multi-step Process of Functional Amyloid Formation. J. Biol. Chem. 2011, 286, 15955–15963. [Google Scholar] [CrossRef]
- Gunaratnam, G.; Dudek, J.; Jung, P.; Becker, S.L.; Jacobs, K.; Bischoff, M.; Hannig, M. Quantification of the Adhesion Strength of Candida albicans to Tooth Enamel. Microorganisms 2021, 9, 2213. [Google Scholar] [CrossRef] [PubMed]
- Sousa, I.C.G.; Teixeira, S.C.; Souza, M.V. de; Conde, M.B.M.; Bailon, G.R.; Cardoso, S.H.S.; Araújo, L.D.; Oliveira, E.B. de; Ferreira, S.O.; Oliveira, T.V. de; et al. Sustainable Extraction and Multimodal Characterization of Fungal Chitosan from Agaricus bisporus. Foods 2025, 14, 2785. [Google Scholar] [CrossRef]
- Habtemariam, S. Trametes versicolor (Synn. Coriolus versicolor) Polysaccharides in Cancer Therapy: Targets and Efficacy. Biomedicines 2020, 8, 135. [Google Scholar] [CrossRef]
- Tovar-Herrera, O.E.; Martha-Paz, A.M.; Pérez-LLano, Y.; Aranda, E.; Tacoronte-Morales, J.E.; Pedroso-Cabrera, M.T.; Arévalo-Niño, K.; Folch-Mallol, J.L.; Batista-García, R.A. Schizophyllum commune : An unexploited source for lignocellulose degrading enzymes. Microbiologyopen 2018, 7. [Google Scholar] [CrossRef]
- Adav, S.S.; Sze, S.K. Fungal Secretome for Biorefinery: Recent Advances in Proteomic Technology. Mass Spectrom. Lett. 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Ballen Sierra, L.A.; Mendes-Pereira, T.; García, G.J.Y.; Werkhaizer, C.Q.; de Rezende, J.B.; Rodrigues, T.A.B.; Badotti, F.; Cardoso, E.S. de C.; da Costa, A.M.; Uetanabaro, A.P.; et al. Current situation and future perspectives for the use of fungi in the biomaterial industry and proposal for a new classification of fungal-derived materials. PeerJ Mater. Sci. 2023, 5, e31. [Google Scholar] [CrossRef]
- Mattern, D.J.; Valiante, V.; Unkles, S.E.; Brakhage, A.A. Synthetic biology of fungal natural products. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Varriale, L.; Ulber, R. Fungal-Based Biorefinery: From Renewable Resources to Organic Acids. ChemBioEng Rev. 2023, 10, 272–292. [Google Scholar] [CrossRef]
- Vieira, R.I.M.; Peixoto, A. da S.; Monclaro, A.V.; Ricart, C.A.O.; Filho, E.X.F.; Miller, R.N.G.; Gomes, T.G. Fungal Coculture: Unlocking the Potential for Efficient Bioconversion of Lignocellulosic Biomass. J. Fungi 2025, 11, 458. [Google Scholar] [CrossRef]
- van den Brandhof, J.G.; Hansen, N.; Hou, C.; Broers, S.C.; Tegelaar, M.; Wösten, H.A.B. Characterization of pure mycelium materials from different mushroom-forming fungi. Antonie Van Leeuwenhoek 2025, 118, 121. [Google Scholar] [CrossRef]
- Peñalva, M.A.; Tilburn, J.; Bignell, E.; Arst, H.N. Ambient pH gene regulation in fungi: making connections. Trends Microbiol. 2008, 16, 291–300. [Google Scholar] [CrossRef]
- Lara-Martínez, D.; Tristán-Flores, F.E.; Cervantes-Montelongo, J.A.; Silva-Martínez, G.A. Fungal Stress Responses and the Importance of GPCRs. J. Fungi 2025, 11, 213. [Google Scholar] [CrossRef]
- Cornet, M.; Gaillardin, C. pH Signaling in Human Fungal Pathogens: a New Target for Antifungal Strategies. Eukaryot. Cell 2014, 13, 342–352. [Google Scholar] [CrossRef]
- Kováčová, K.; Degani, G.; Stratilová, E.; Farkaš, V.; Popolo, L. Catalytic properties of Phr family members of cell wall glucan remodeling enzymes: implications for the adaptation of Candida albicans to ambient pH. FEMS Yeast Res. 2015, 15. [Google Scholar] [CrossRef]
- Brown, H.E.; Telzrow, C.L.; Saelens, J.W.; Fernandes, L.; Alspaugh, J.A. Sterol-Response Pathways Mediate Alkaline Survival in Diverse Fungi. MBio 2020, 11. [Google Scholar] [CrossRef]
- Jeennor, S.; Anantayanon, J.; Panchanawaporn, S.; Chutrakul, C.; Laoteng, K. Morphologically engineered strain of Aspergillus oryzae as a cell chassis for production development of functional lipids. Gene 2019, 718, 144073. [Google Scholar] [CrossRef]
- Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A.; Athanassiou, A. Advanced Materials From Fungal Mycelium: Fabrication and Tuning of Physical Properties. Sci. Rep. 2017, 7, 41292. [Google Scholar] [CrossRef]
- Appels, F.V.W.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.B.; Dijksterhuis, J.; Krijgsheld, P.; Wösten, H.A.B. Fabrication factors influencing mechanical, moisture- and water-related properties of mycelium-based composites. Mater. Des. 2019, 161, 64–71. [Google Scholar] [CrossRef]
- Sayfutdinova, A.; Samofalova, I.; Barkov, A.; Cherednichenko, K.; Rimashevskiy, D.; Vinokurov, V. Structure and Properties of Cellulose/Mycelium Biocomposites. Polymers (Basel). 2022, 14, 1519. [Google Scholar] [CrossRef]
- Manan, S.; Ullah, M.W.; Ul-Islam, M.; Atta, O.M.; Yang, G. Synthesis and applications of fungal mycelium-based advanced functional materials. J. Bioresour. Bioprod. 2021, 6, 1–10. [Google Scholar] [CrossRef]
- Guarro, J.; Gené, J.; Stchigel, A.M. Developments in Fungal Taxonomy. Clin. Microbiol. Rev. 1999, 12, 454–500. [Google Scholar] [CrossRef] [PubMed]
- Deveau, A.; Bonito, G.; Uehling, J.; Paoletti, M.; Becker, M.; Bindschedler, S.; Hacquard, S.; Hervé, V.; Labbé, J.; Lastovetsky, O.A.; et al. Bacterial–fungal interactions: ecology, mechanisms and challenges. FEMS Microbiol. Rev. 2018, 42, 335–352. [Google Scholar] [CrossRef]
- Steffan, B.N.; Venkatesh, N.; Keller, N.P. Let’s Get Physical: Bacterial-Fungal Interactions and Their Consequences in Agriculture and Health. J. Fungi 2020, 6, 243. [Google Scholar] [CrossRef]
- Nogueira, F.; Sharghi, S.; Kuchler, K.; Lion, T. Pathogenetic Impact of Bacterial–Fungal Interactions. Microorganisms 2019, 7, 459. [Google Scholar] [CrossRef]
- Corrêa-Moreira, D.; Baptista, B. de O.; Giosa, D.; Oliveira, M.M.E. Editorial: Emerging fungal pathogens: perspectives. Front. Fungal Biol. 2024, 5. [Google Scholar] [CrossRef]
- El-Gendi, H.; Saleh, A.K.; Badierah, R.; Redwan, E.M.; El-Maradny, Y.A.; El-Fakharany, E.M. A Comprehensive Insight into Fungal Enzymes: Structure, Classification, and Their Role in Mankind’s Challenges. J. Fungi 2021, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Alapan, D.; Bisweswar, O.; Prasenjit, S.; Prasanjit, D.; Arkapal, B. Recent advances in the clinical development of antifungal vaccines: a narrative review. Front. Trop. Dis. 2024, 5. [Google Scholar] [CrossRef]
- Eagan, J.L.; Keller, N.P. Fungal secondary metabolism. Curr. Biol. 2025, 35, R503–R508. [Google Scholar] [CrossRef]
- Kordana, N.; Johnson, A.; Quinn, K.; Obar, J.J.; Cramer, R.A. Recent developments in Aspergillus fumigatus research: diversity, drugs, and disease. Microbiol. Mol. Biol. Rev. 2025, 89. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I.; Mohamed, A.A. A Comprehensive Review on the Biological, Agricultural and Pharmaceutical Properties of Secondary Metabolites Based-Plant Origin. Int. J. Mol. Sci. 2023, 24, 3266. [Google Scholar] [CrossRef]
- Miyazawa, K.; Yoshimi, A.; Sano, M.; Tabata, F.; Sugahara, A.; Kasahara, S.; Koizumi, A.; Yano, S.; Nakajima, T.; Abe, K. Both Galactosaminogalactan and α-1,3-Glucan Contribute to Aggregation of Aspergillus oryzae Hyphae in Liquid Culture. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Chauhan, M.; Shivarathri, R.; Aptekmann, A.A.; Chowdhary, A.; Kuchler, K.; Desai, J. V.; Chauhan, N. The Gcn5 lysine acetyltransferase mediates cell wall remodeling, antifungal drug resistance, and virulence of Candida auris. mSphere 2025, 10. [Google Scholar] [CrossRef]
- Mapplebeck, J.C.S.; Lorenzo, L.-E.; Lee, K.Y.; Gauthier, C.; Muley, M.M.; De Koninck, Y.; Prescott, S.A.; Salter, M.W. Chloride Dysregulation through Downregulation of KCC2 Mediates Neuropathic Pain in Both Sexes. Cell Rep. 2019, 28, 590–596.e4. [Google Scholar] [CrossRef] [PubMed]
- Krüger, W.; Vielreicher, S.; Kapitan, M.; Jacobsen, I.; Niemiec, M. Fungal-Bacterial Interactions in Health and Disease. Pathogens 2019, 8, 70. [Google Scholar] [CrossRef]
- Baert, K.; de Geest, B.G.; de Rycke, R.; da Fonseca Antunes, A.B.; de Greve, H.; Cox, E.; Devriendt, B. β-glucan microparticles targeted to epithelial APN as oral antigen delivery system. J. Control. Release 2015, 220, 149–159. [Google Scholar] [CrossRef]
- Caruso, D.J.; Palombo, E.A.; Moulton, S.E.; Zaferanloo, B. Exploring the Promise of Endophytic Fungi: A Review of Novel Antimicrobial Compounds. Microorganisms 2022, 10, 1990. [Google Scholar] [CrossRef]
- Vadivel, D.; Cartabia, M.; Scalet, G.; Buratti, S.; Di Landro, L.; Benedetti, A.; Auricchio, F.; Babbini, S.; Savino, E.; Dondi, D. Innovative chitin-glucan based material obtained from mycelium of wood decay fungal strains. Heliyon 2024, 10, e28709. [Google Scholar] [CrossRef]
- de Lima Batista, A.C.; de Souza Paiva, W.; de Souza Neto, F.E. Chitosan. In Polysaccharides of Microbial Origin; Springer International Publishing: Cham, 2021; pp. 1–18. [Google Scholar]
- Izadi, H.; Asadi, H.; Bemani, M. Chitin: a comparison between its main sources. Front. Mater. 2025, 12. [Google Scholar] [CrossRef]
- Martínez, J.P.; Gil, M.L.; López-Ribot, J.L.; Chaffin, W.L. Serologic Response to Cell Wall Mannoproteins and Proteins of Candida albicans. Clin. Microbiol. Rev. 1998, 11, 121–141. [Google Scholar] [CrossRef]
- Güler, P.; Kutluer, F.; Kunduz, İ. Screening to Mycelium Specifications of Ganoderma lucidum (Fr.) Karst (Reishi). 2011.
- Xue, M.; Hou, X.; Fu, J.; Zhang, J.; Wang, J.; Zhao, Z.; Xu, D.; Lai, D.; Zhou, L. Recent Advances in Search of Bioactive Secondary Metabolites from Fungi Triggered by Chemical Epigenetic Modifiers. J. Fungi 2023, 9, 172. [Google Scholar] [CrossRef]
- Angelova, G.; Brazkova, M.; Mihaylova, D.; Slavov, A.; Petkova, N.; Blazheva, D.; Deseva, I.; Gotova, I.; Dimitrov, Z.; Krastanov, A. Bioactivity of Biomass and Crude Exopolysaccharides Obtained by Controlled Submerged Cultivation of Medicinal Mushroom Trametes versicolor. J. Fungi 2022, 8, 738. [Google Scholar] [CrossRef]
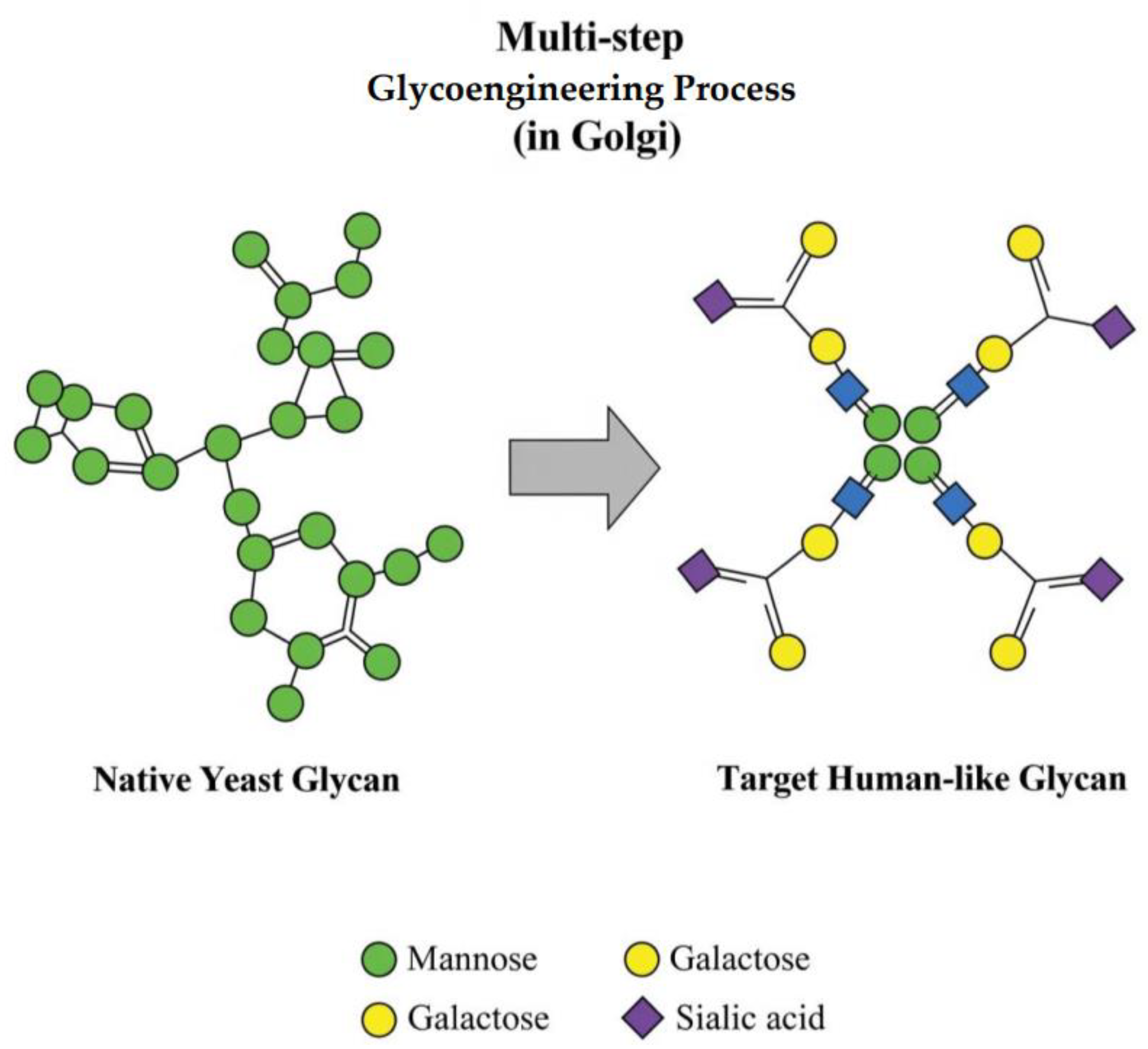
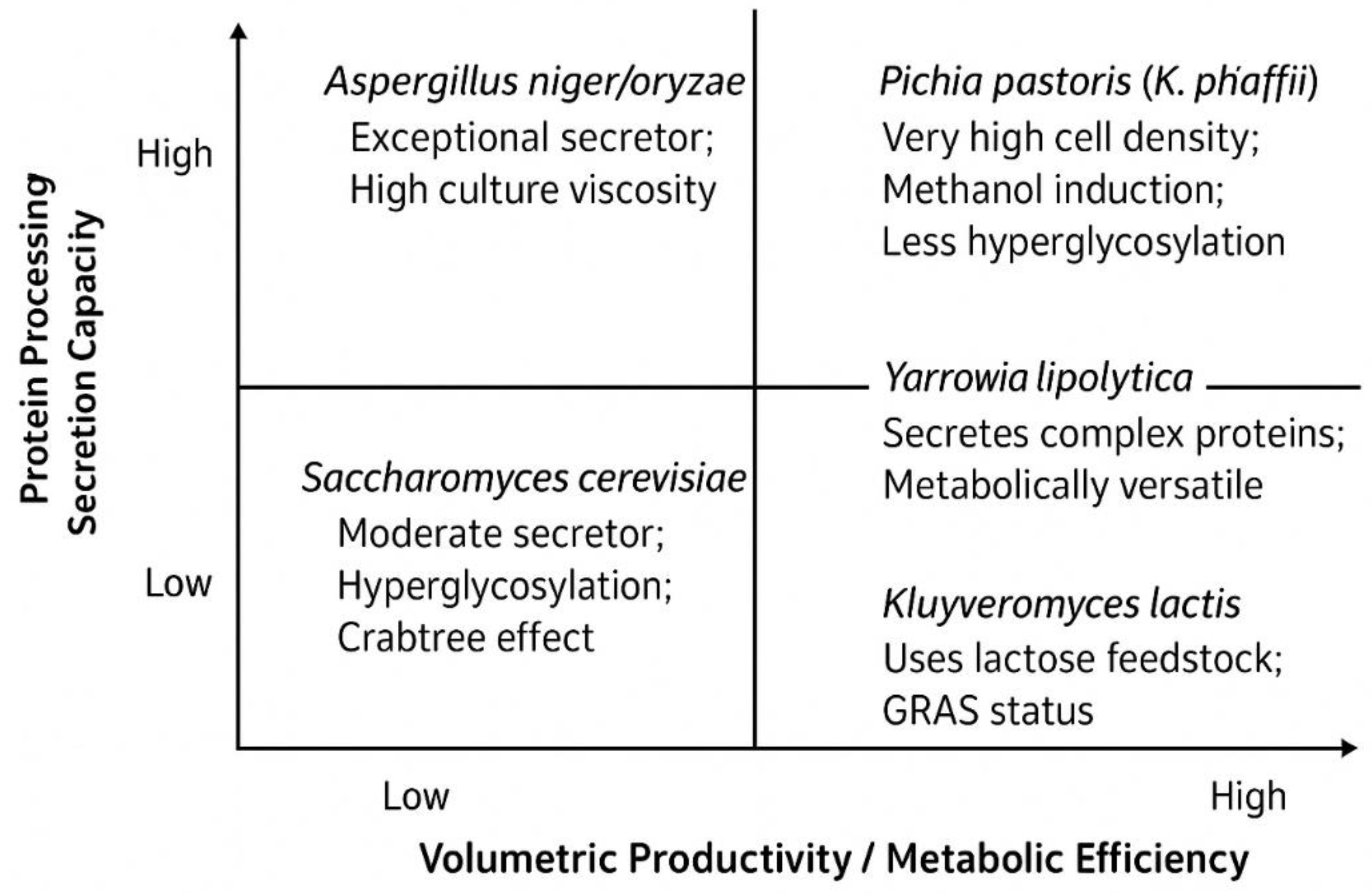
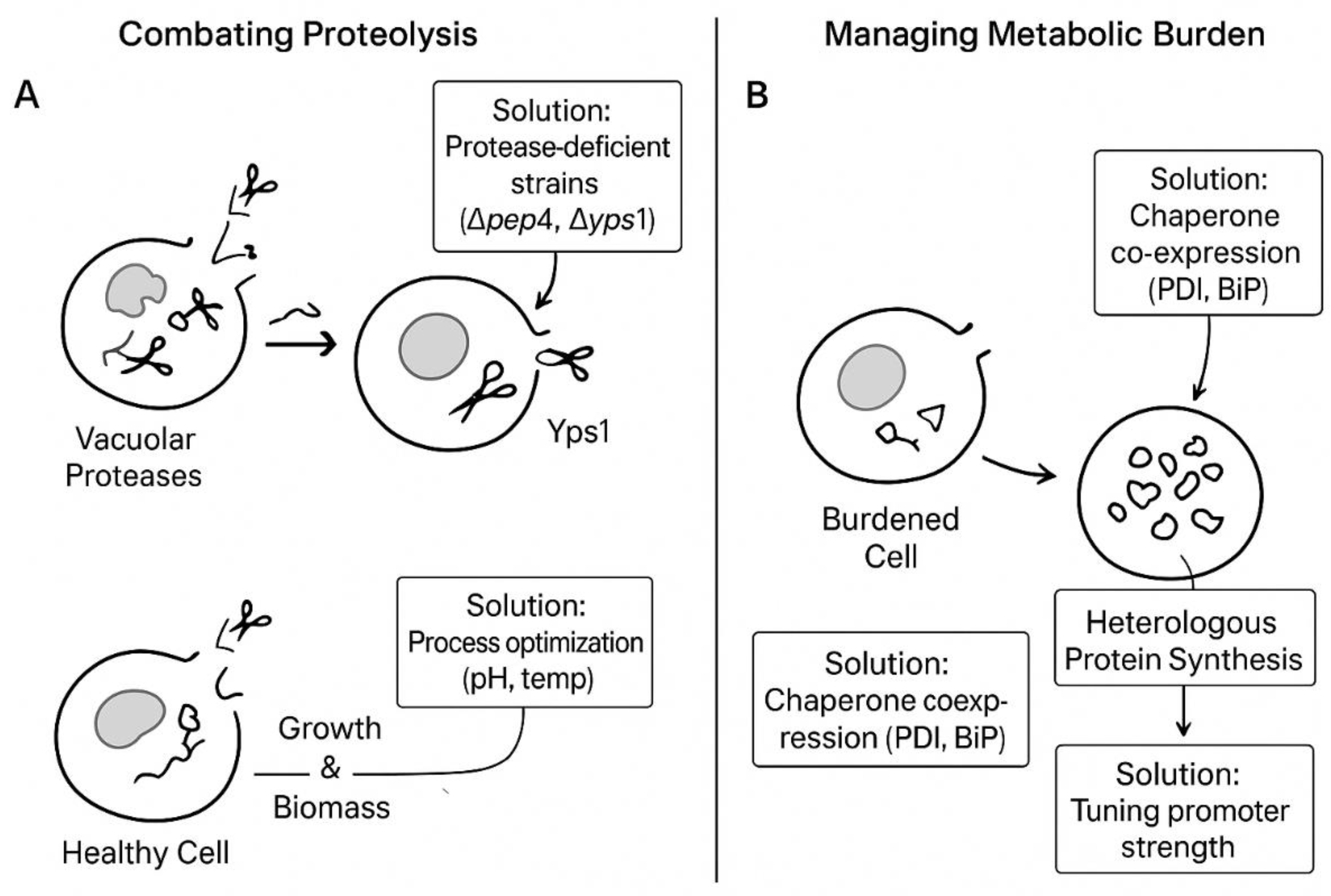
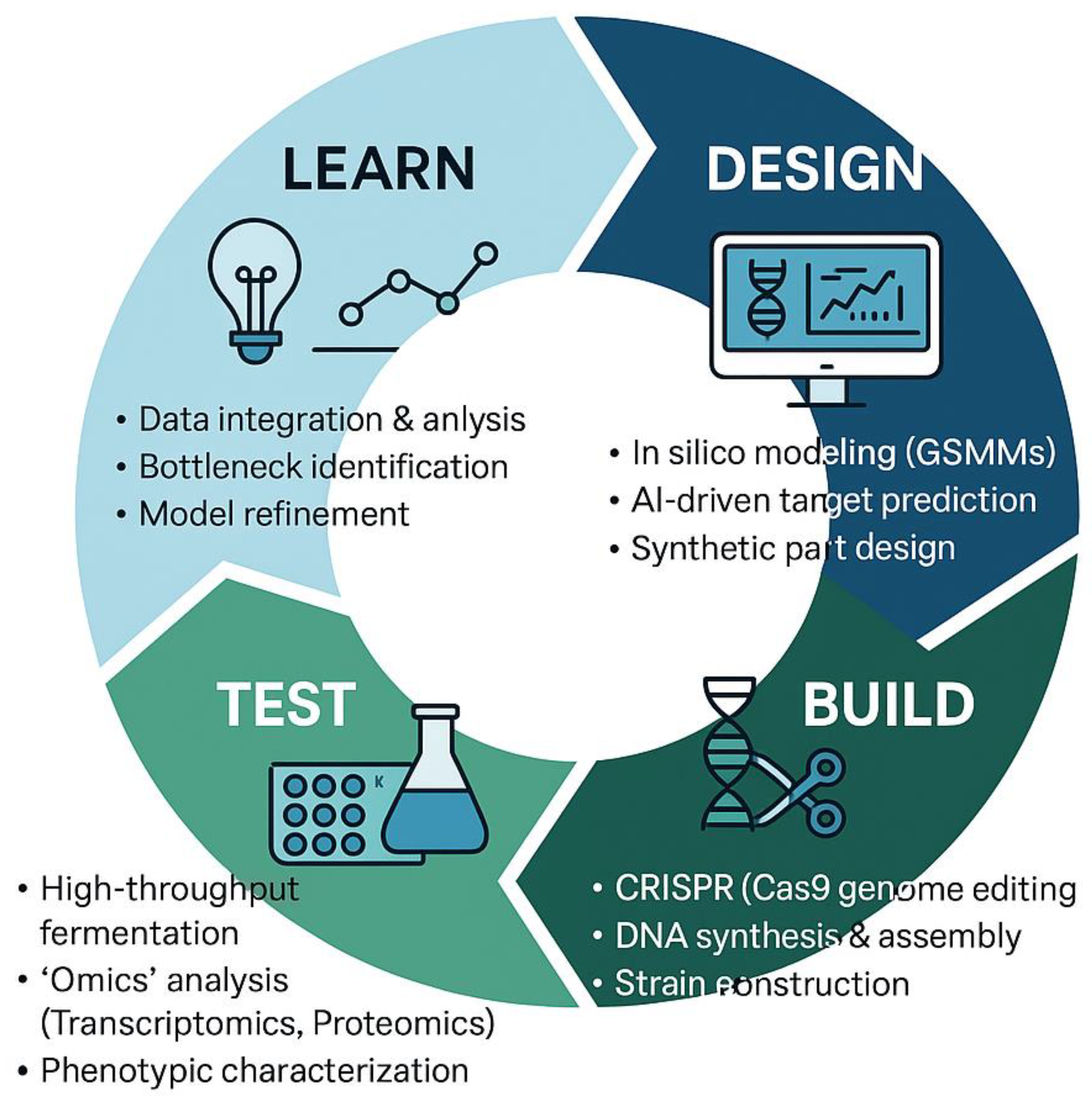
| Fungal Species | Primary Hyphal System | Key Cell Wall Components | Reported Mechanical Properties (Example Values) | Reported Physical/Thermal Properties (Example Values) | Key References |
|---|---|---|---|---|---|
| Ganoderma lucidum | Dimitic/Trimitic | High β-glucan content, complex branching; chitin; triterpenoids. | Superior physical and mechanical properties compared to P. ostreatus. Compressive strength can be tuned by substrate. | Hydrophobic (WCA ~120°). Mycelium mats can be tuned for porosity and density. | [12,32,53] |
| Pleurotus ostreatus | Monomitic | β-1,3 and β-1,6 glucans; chitin; high density of surface functional groups. | Lower mechanical properties than G. lucidum. Compressive strength of composites: 0.03–0.3 MPa | High water absorption capacity. Effective for biosorption of heavy metals. | [9,32] |
| Trametes versicolor | Trimitic | Polysaccharide-K (PSK) and Polysaccharopeptide (PSP); high glucan content. | Trimitic system implies high intrinsic strength. Used for strong composites. Flexural modulus (pressed): ~34-80 MPa depending on substrate. | Good insulation and fire-retardant properties. | [23,54] |
| Schizophyllum commune | Monomitic | High levels of secreted hydrolytic enzymes (xylanases, glucanases). Cell wall binds various micronutrients. | Tensile strength (pure sheet): ~9.5 MPa. Mechanical properties depend heavily on substrate and processing. | Cell wall acts as a pH-dependent ion-exchange material. | [29,46] |
| Agaricus bisporus | Monomitic | Chitin-glucan complexes. Chitosan derived from it has lower crystallinity than crustacean source. | Nanopapers from its chitin-glucan have tensile strength >200 MPa. | Chitosan films show good film-forming ability and porosity. | [27] |
| Modulator Category | Specific Target | Agent/Method | Observed Biochemical Effect on Cell Wall | Potential Impact on Material Properties | Key References |
|---|---|---|---|---|---|
| Genetic | Gcn5 Lysine Acetyltransferase | Gene Deletion (gcn5Δ) | ↑ β-glucan exposure, ↑ chitin content, altered expression of FKS1 and adhesins. | Altered adhesion, stress response, and potentially flexibility/strength. Broad-spectrum control. | [69] |
| α-1,3-Glucan Synthase | Gene Deletion (agsΔ) | ↓ α-1,3-glucan content. | Altered morphology (e.g., smaller pellets or dispersed growth, species-dependent) ; ↓ culture viscosity, improved bioprocessing. | [52,67] | |
| Chitin Synthase (Class V/VII) | Gene Deletion (csmΔ) | Disorganization of wall structure, altered surface rodlet layer. | Altered surface properties (e.g., hydrophilicity), modified mechanical integrity. | [28] | |
| pH-Sensing Pathway (PacC/Rim101) | Gene Deletion (pacCΔ) | Inability to adapt wall structure to ambient pH, defective expression of pH-regulated enzymes. | Loss of pH-dependent programmability, defects in material formation under specific pH. | [61] | |
| Chemical | Chitin Synthases (all classes) | Nikkomycin Z, Polyoxins | Competitive inhibition of chitin synthesis at hyphal tips. | Localized growth inhibition, creation of zones of weakness/flexibility, patterned growth. | [71] |
| Histone Deacetylases (HDACs) | SAHA, Trichostatin A (TSA) | Chromatin de-repression, activation of silent biosynthetic gene clusters. | Induction of novel secondary metabolites (pigments, polymers), adding new functionalities. | [78] | |
| DNA Methyltransferases (DNMTs) | 5-Azacytidine | DNA demethylation, activation of silent gene clusters. | Similar to HDAC inhibitors; induction of novel chemical functionalities in the material. | [78] | |
| β-1,3-Glucan Synthase | Echinocandins (e.g., Caspofungin) | Inhibition of β-1,3-glucan synthesis. | Weakened cell wall, increased sensitivity to stress, potential for controlled lysis or softening. | [79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




