Submitted:
12 August 2025
Posted:
13 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
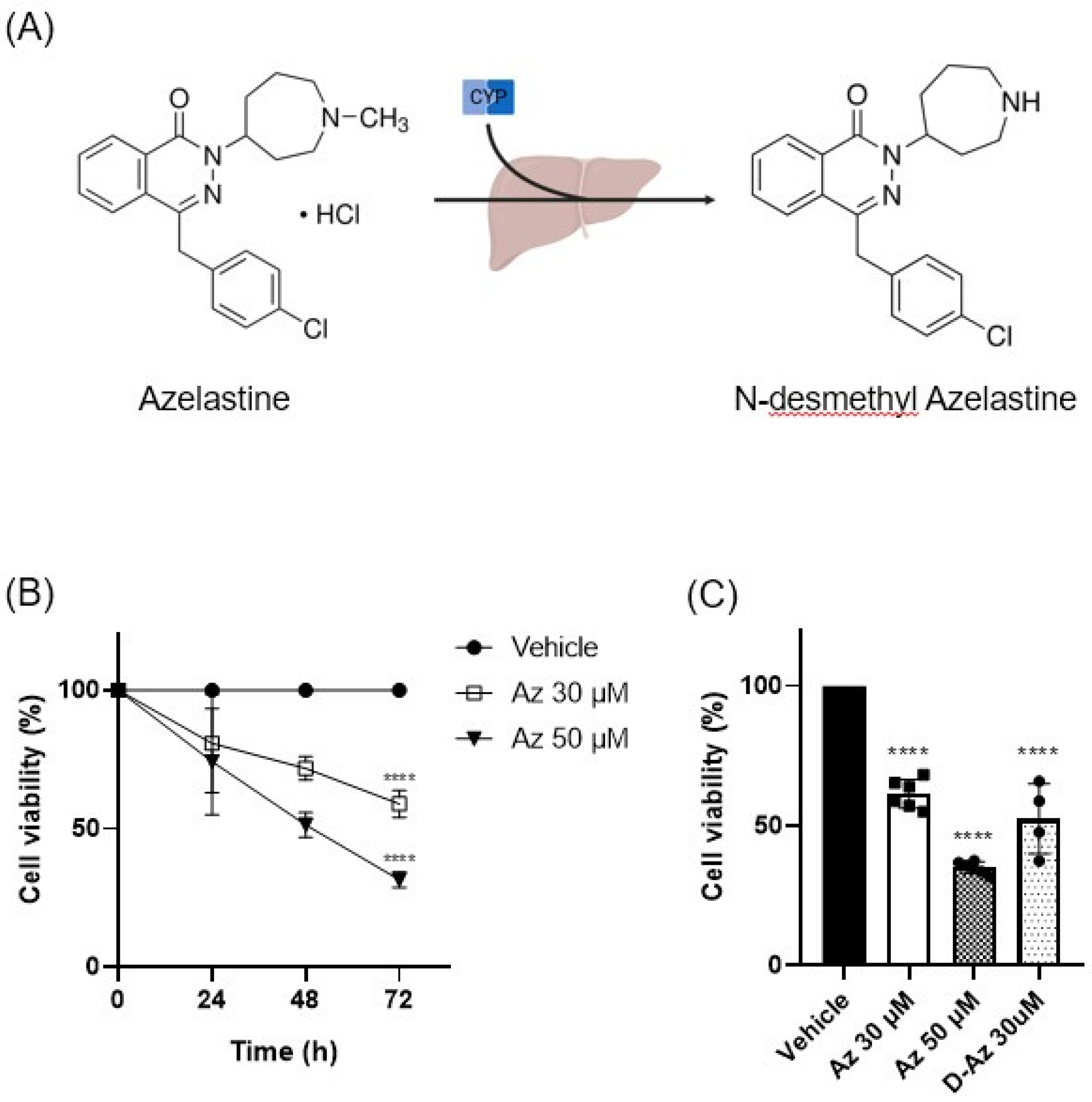
2.1. Azelastine Decreases TNBC Cell Viability in a Dose- and Time-Dependent Manner
2.2. Downregulated HRH1 Expression in Breast Tumors Does Not Correlate with Patient Survival
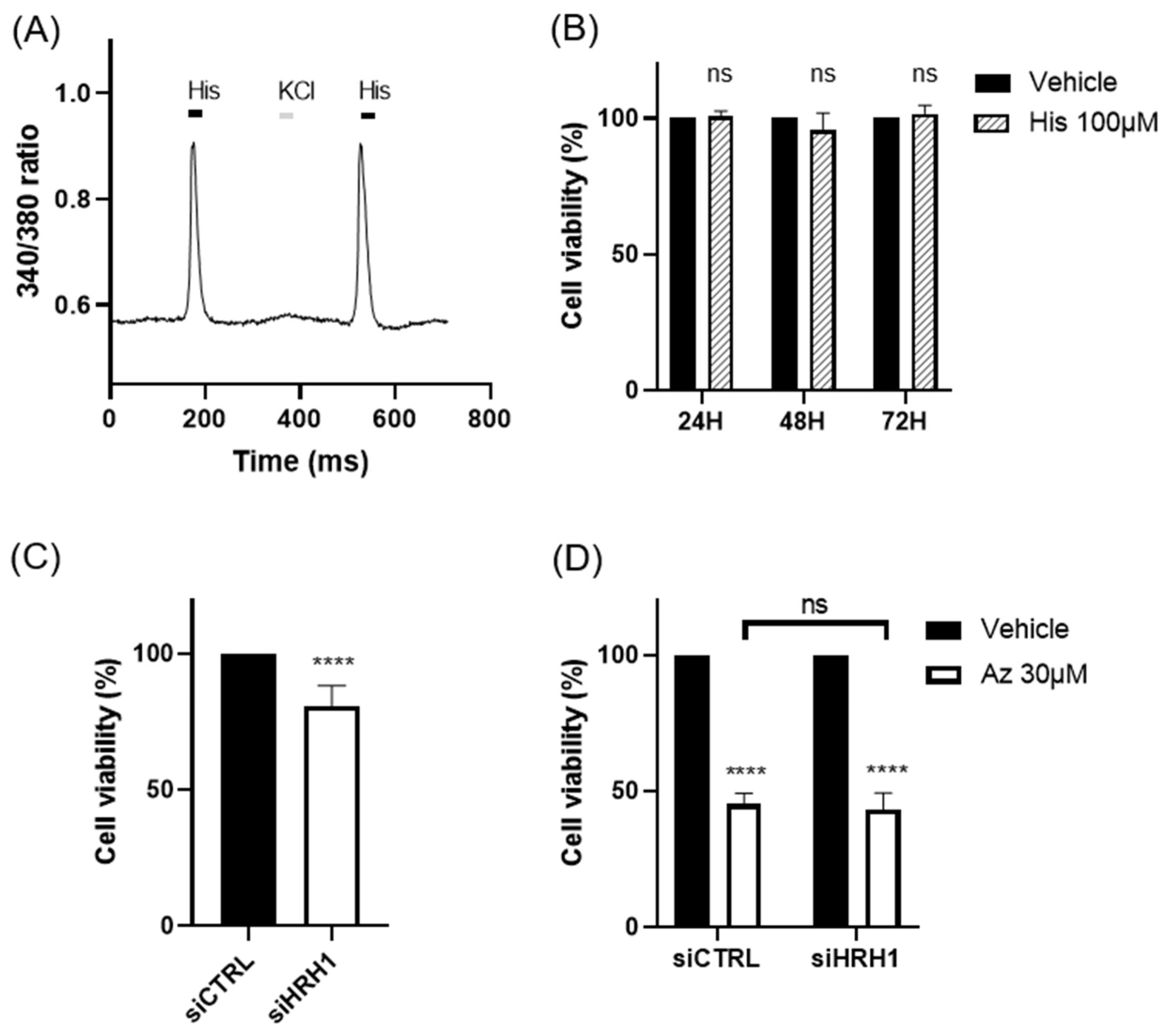
2.3. HRH1 Is Functionally Expressed in TNBC Cells But Does Not Mediate Azelastine Cytotoxicity
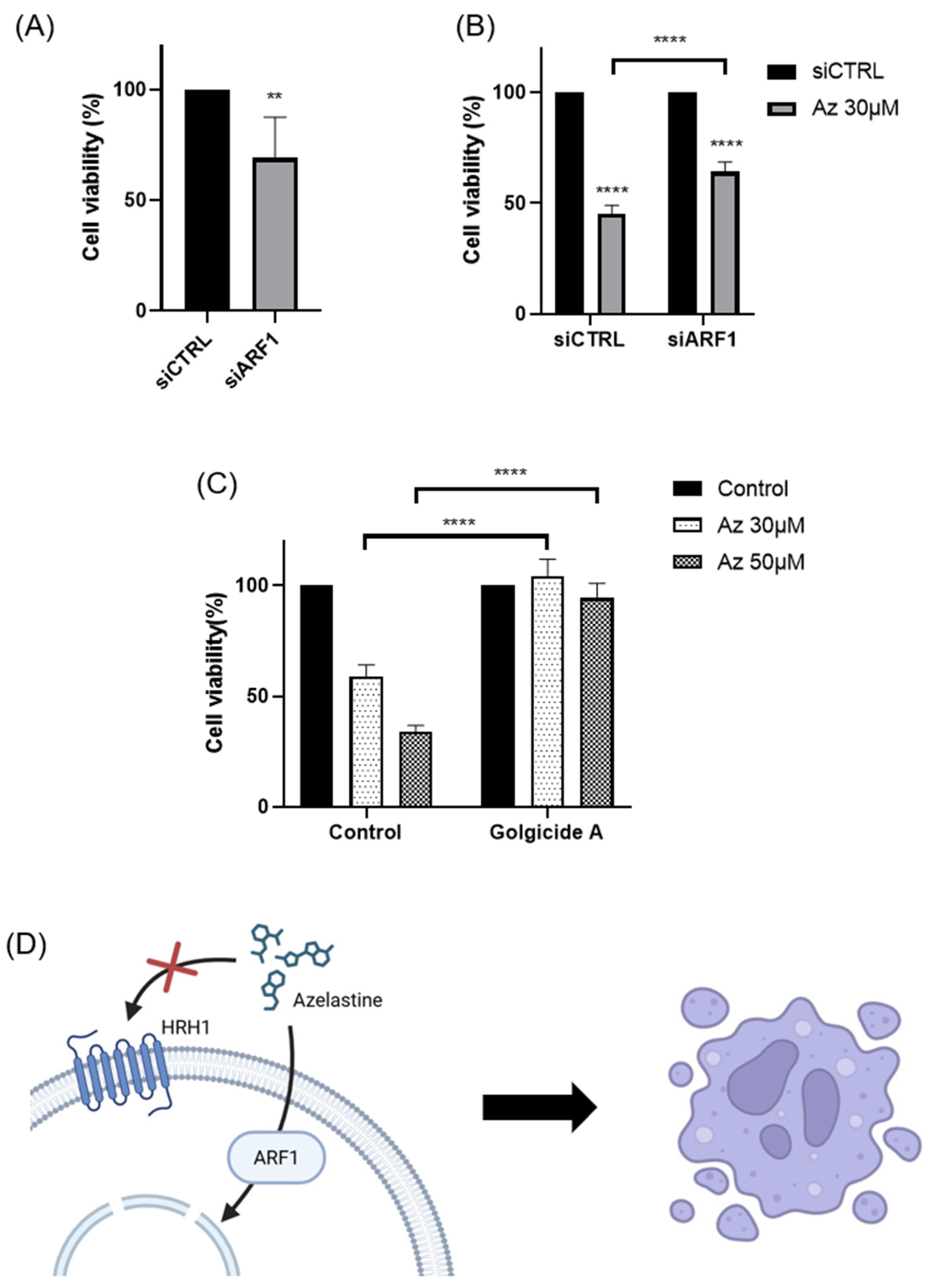
2.4. ARF1 Is Overexpressed in TNBC and Associated with Poor Patient Survival
2.5. ARF1 Inhibition Rescues Cells from Azelastine-Induced Death
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Drug Treatment and Reagents
4.3. Cell Viability Assay
4.4. Calcium Imaging
4.5. RNA Interference
4.6. Bioinformatic Analysis of Patient Datasets
4.7. Statistical Analysis
4.8. Ethics Statement
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ARF1 | ADP-ribosylation Factor 1 |
| HRH1 | Histamine Receptor 1 |
| DFS | Disease-Free Survival |
| OS | Overall Survival |
| TNBC | Triple-Negative Breast Cancer |
| GCA | Golgicide A |
| CTGS | Cancer Target Gene Screening |
| TCGA | The Cancer Genome Atlas |
References
- Loibl, S.; Poortmans, P.; Morrow, M.; Denkert, C.; Curigliano, G. Breast cancer. Lancet 2021, 397, 1750–1769. [Google Scholar] [CrossRef]
- Bianchini, G.; De Angelis, C.; Licata, L.; Gianni, L. Treatment landscape of triple-negative breast cancer—expanded options, evolving needs. Nat. Rev. Clin. Oncol. 2022, 19, 91–113. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.M.; Renshaw, L. Triple-negative breast cancer: Challenges and treatment options. Breast 2018, 38, 1–10. [Google Scholar]
- Pandi, A.; Nakka, M.; Maturi, K.; Basha, J. Triple-negative breast cancer: New insights into pathogenesis and therapeutic strategies. Cancer Treat. Rev. 2021, 91, 102108. [Google Scholar]
- Migliorini, D.; Campana, L.; Zazzeroni, F.; Ceccarelli, C.; Soverini, S.; Giannini, A.; et al. Histamine receptors as therapeutic targets in cancer. Cancer Res. 2019, 79, 4596–4604. [Google Scholar]
- Kim, H.; Lee, J.Y.; Park, S.J.; Lee, D.; Youn, H. Histamine receptor expression in breast cancer and its prognostic significance. Oncol. Rep. 2021, 45, 1497–1505. [Google Scholar]
- Choi, S.; Park, M.; Lee, E.; Kim, K. Histamine receptor 1 expression in triple-negative breast cancer. Breast Cancer Res. 2020, 22, 67. [Google Scholar]
- El-Mowafy, A.M.; White, K. Terfenadine, a H1-receptor antagonist, demonstrates a new class of pro-apoptotic activity in human breast cancer cells, which involves protein kinase C-dependent and -independent pathways. Br. J. Pharmacol. 1999, 126, 575–585. [Google Scholar]
- Sarkar, D.; Bouker, K.B.; Sengupta, S.; Sharma, A.; Toth, K.; Koomen, J.M.; et al. Repositioning antihistamines as cancer therapeutics. J. Exp. Med. 2021, 218, e20210453. [Google Scholar]
- Hill, S.J.; Ganellin, C.R.; Timmerman, H.; Schwartz, J.C.; Shankley, N.P.; Young, J.M.; et al. International Union of Pharmacology. XIII. Classification of histamine receptors. Pharmacol. Rev. 1997, 49, 253–278. [Google Scholar] [CrossRef]
- Li, Q.; Liu, K.Y.; Liu, Q.; Wang, G.; Jiang, W.; Meng, Q.; et al. Antihistamine drug Ebastine inhibits cancer growth by targeting Polycomb group protein EZH2. Mol. Cancer Ther. 2020, 19, 2023–2033. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Liu, D.; Wei, L.; Ye, X.; Zhang, L.; Zhang, X.; et al. Ebastine impairs metastatic spread in triple-negative breast cancer by targeting focal adhesion kinase. Cancer Lett. 2023, 561, 216125. [Google Scholar]
- Hill, R.L.; Davis, M.A.; Kim, J.; et al. Investigating the effects of antihistamines on tumor cell proliferation. J. Cancer Res. Clin. Oncol. 2020, 146, 1515–1527. [Google Scholar]
- Simons, F.E. Azelastine: a review of its pharmacodynamics, pharmacokinetics, and clinical efficacy. J. Allergy Clin. Immunol. 1999, 103, 359–367. [Google Scholar]
- Anderson, N.; Borlak, J. Drug-induced phospholipidosis. FEBS Lett. 2006, 580, 5533–5540. [Google Scholar] [CrossRef]
- Hochhaus, G.; Derendorf, H. Pharmacokinetics and metabolism of azelastine. Drugs 1994, 48, 694–703. [Google Scholar]
- Halliwell, W.H. Cationic amphiphilic drug-induced phospholipidosis. Toxicol. Pathol. 1997, 25, 53–60. [Google Scholar] [CrossRef]
- Reasor, M.J.; Hastings, K.L. Drug-induced phospholipidosis: characterization and relevance to the clinical situation. Toxicol. Appl. Pharmacol. 2003, 192, 282–291. [Google Scholar]
- Lee, K.; Bae, Y.; Kim, H.; et al. The involvement of ARF-1 in tumor suppression and its modulation by pharmacological agents. Cancer Lett. 2022, 519, 20–28. [Google Scholar]
- Zhang, S.; Wu, M.; Wang, H.; Bao, Y.; Liu, H. The role of ARF-1 in cancer progression and its therapeutic potential. Front. Cell Dev. Biol. 2022, 10, 835246. [Google Scholar]
- Schlienger, S.; Campbell, S.; Claing, A. ARF1 regulates proliferation and the actin cytoskeleton in breast cancer cells. J. Biol. Chem. 2009, 284, 3956–3967. [Google Scholar]
- Donaldson, J.G.; Jackson, C.L. ARF1 and its GEFs in cancer cell migration and invasion. Small GTPases 2018, 9, 17–25. [Google Scholar]
- Roush, W.; Wang, Y.; Wang, H.; et al. ARF-1 activation and its role in tumor cell migration and survival. J. Cell Sci. 2016, 129, 1211–1221. [Google Scholar]
- Tanaka, A.; Satoh, T.; Okada, Y.; et al. Targeting ARF-1 for cancer therapy: Insights from preclinical studies. Mol. Cancer 2019, 18, 99. [Google Scholar]
- Hu, P.; Liu, Q.; Li, J.; Ma, Y.; Wei, W.; Zhang, Y.; Zhang, P. Azelastine directly targets ARF1 to inhibit colorectal cancer cell growth. Cell Death Dis. 2022, 13, 237. [Google Scholar]
- Xie, H.; Sun, L.; Zhao, Y.; Liu, Y.; Lu, Y.; Cui, Y.; Guo, L. ARF1 is amplified in breast cancer and promotes tumorigenesis through regulation of the MEK/ERK pathway. Cancer Lett. 2018, 438, 1–10. [Google Scholar]
- Koch, R.L.; Jensen, H.A.; Engelman, J.A.; et al. The CAD–ARF1 pathway as a therapeutic target in cancer. Mol. Cancer Ther. 2021, 20, 2105–2116. [Google Scholar]
- Zhang, L.; Wang, C.; Liu, Q.; Hu, P. The therapeutic potential of targeting ARF-1 in cancer therapy. Nat. Rev. Cancer 2022, 22, 253–266. [Google Scholar]
- Li, L.; et al. ARF-1’s role in modulating cellular trafficking and cancer progression. Cancer Res. 2020, 80, 987–996. [Google Scholar]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2019, 18, 41–58. [Google Scholar] [CrossRef]
- Lorenz, W.; Doenicke, A.; Weber, D.; Neugebauer, E.; Schmal, A.; Backhaus, B. Histamine and histamine antagonists in experimental and clinical oncology. Pharmacol. Ther. 1985, 27, 319–354. [Google Scholar]
- Panula, P.; Chazot, P.L.; Cowart, M.; Gutzmer, R.; Leurs, R.; Liu, W.L.; et al. International Union of Basic and Clinical Pharmacology. XCVIII. Histamine Receptors. Pharmacol. Rev. 2015, 67, 601–655. [Google Scholar] [CrossRef]
- Lee, M.; Park, S.; Kim, J. Histamine receptor 1 is a modulator of angiogenesis in breast cancer. J. Clin. Oncol. 2020, 38, 1158–1166. [Google Scholar]
- Liu, Y.; Zhang, Y.; Li, J.; Wei, W.; Ma, Y.; Zhang, P.; et al. Azelastine inhibits melanoma cell growth by regulating histamine receptors. Cancer Res. 2019, 79, 3024–3032. [Google Scholar]
- Zhang, J.; Zhao, Z.; Li, W.; et al. Mechanisms of azelastine in cancer treatment: A novel approach. Cancer Chemother. Pharmacol. 2021, 88, 847–857. [Google Scholar]
- Choi, H.; Kim, Y.; Kim, K.; et al. ARF1 promotes tumorigenesis by regulating PI3K-AKT signaling in liver cancer. Exp. Mol. Med. 2020, 52, 821–832. [Google Scholar]
- Jin, S.; et al. Role of ARF-1 in cellular homeostasis and cancer progression. Cell. Mol. Life Sci. 2018, 75, 2761–2773. [Google Scholar]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



