Submitted:
11 August 2025
Posted:
12 August 2025
You are already at the latest version
Abstract
The study provides valuable insights into the sustainable utilization of edible tuber peels from high-mountain regions, which constitute promising reserves of bioactive phenolic compounds with the potential to enhance the biotechnological properties of lactic acid bacteria. Thirty-two extracts derived from peels of different varieties of tubers, such as Oxalis tuberosa Mol. (Oxalidaceae), Ullucus tuberosus Caldas (Basellaceae) and Solanum tuberosum L. subsp. andigena (Solanaceae) from the Argentinian Puna were incorporated into lactobacilli cultures and individually evaluated. These selectively enhance the development of the probiotic strain Lactiplantibacillus plantarum ATCC 10241 and of Lacticaseibacillus paracasei CO1 from ovine origin, without promoting the growth of pathogenic bacteria when small amounts of each extract are incorporated into the culture media. To determine the main compounds concentrated in the phytoextracts, a bio-guided study was conducted. The most significant results were obtained by O. tuberosa phytochemicals added to the culture medium, yielding promising increases in biofilm formation and biosurfactant activity. These adaptive strategies developed by bacteria possess key biotechnological significance. Furthermore, the bio-detoxification capacity of mutagenic substances, particularly of the novel strain CO1, along with its mode of action and genetic identification are reported for the first time. In conclusion, lactobacilli strains have potential as fermentation starters, and natural product-based culture media contribute to multiple bacterial biotechnological applications in both health and the environment.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Natural Product Source
2.1.1. Natural Product Extraction
2.2. Bacterial Strains
2.2.1. Probiotic and Environmental Bacteria
2.2.2. Pathogenic Bacteria
2.3. Genotypic Characterization of Novel Isolate
2.4. Bacterial Growth
2.5. Biofilm Biomass Quantification
2.5.1. Specific Biofilm Index Determination
2.6. Surfactant Activity
2.7. Chemical and Spectrochemical Analysis
2.8. Lactobacilli Fermentation into Modified Media
2.9. Specific Enzyme Inhibition
2.10. Electron Microscopy
2.11. TLC and GC-MS Analysis
3. Results and Discussion
3.1. Identification of a Novel Lacticaseibacillus Paracasei Strain
3.1.1. Origin
3.2. Effects of Tuber Peel Extracts Added into Culture Media on the Non-Pathogenic Bacteria Development
3.2.1. Food Origin-Probiotic Lactiplantibacillus Plantarum ATCC 10241
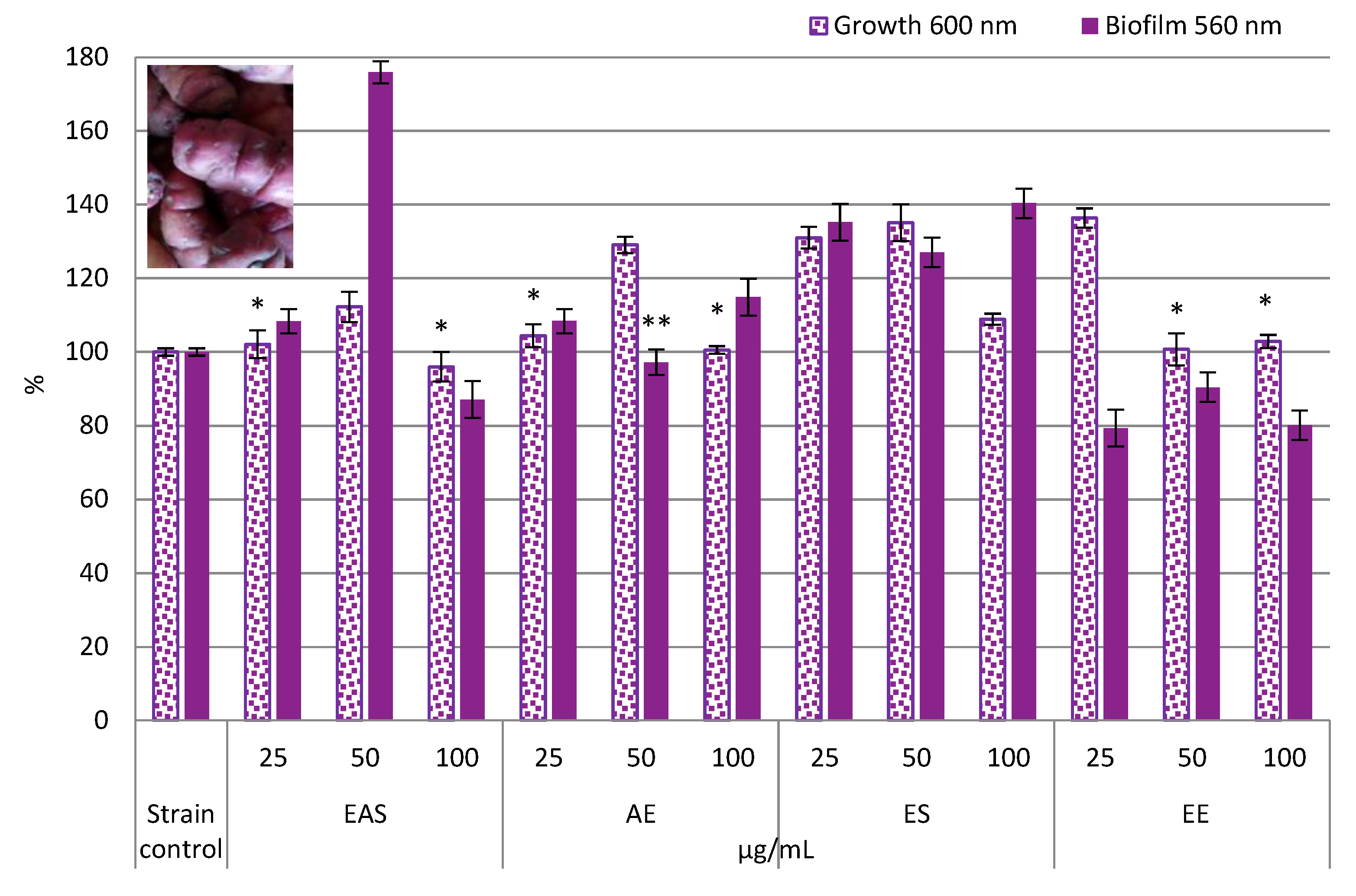
3.2.2. Lacticaseibacillus Paracasei CO1
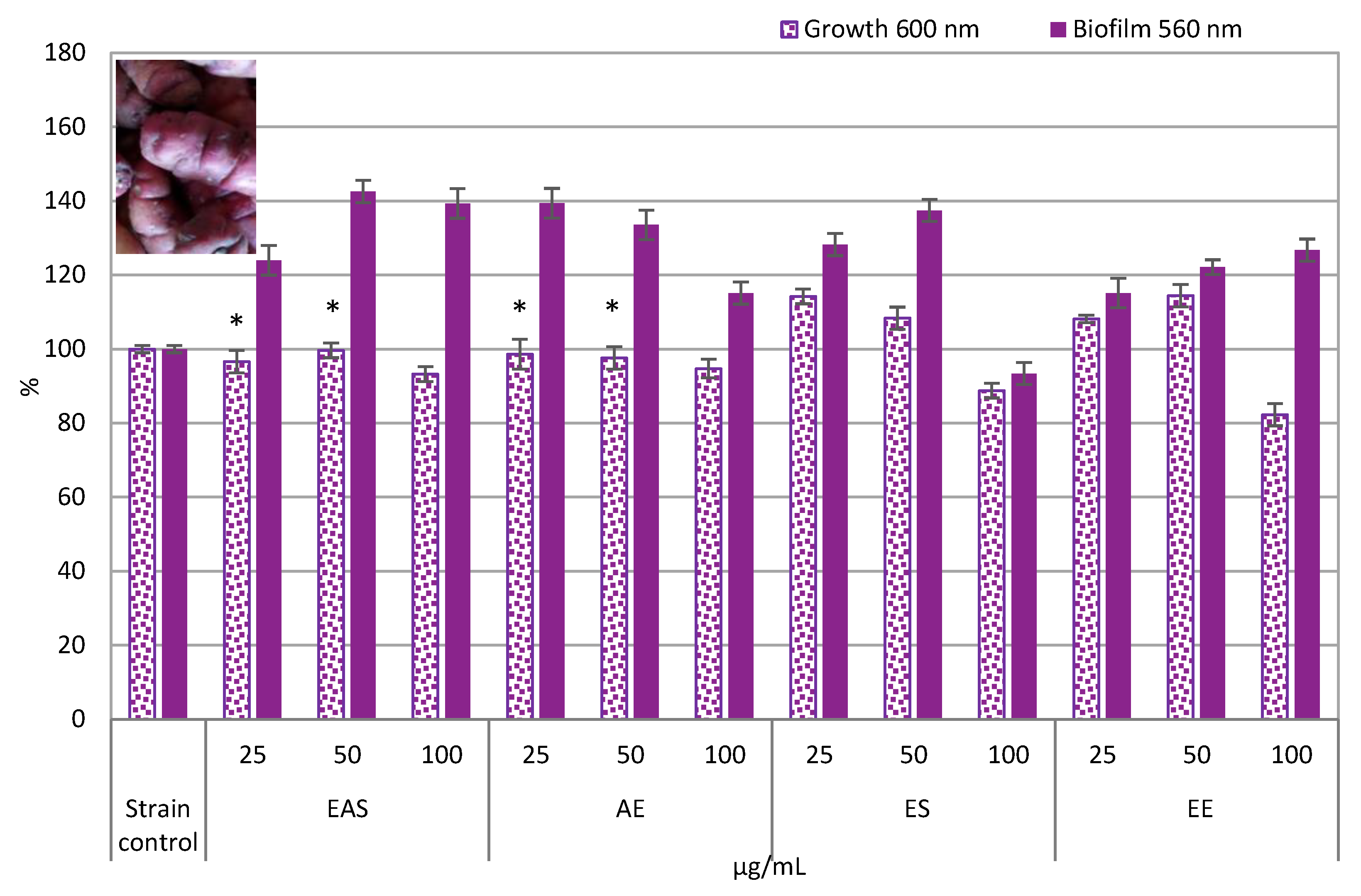
3.3. Impact on Gram-Negative Bacteria
3.4. Chemical and Spectrochemical Data
| Andean plant tubers (Names) |
Peel extracts (Codes) |
Extract yields (%) | Phenolic compounds (FeCl3 and AlCl3 reagents) |
UV spectroscopy |
Assignments |
|
|---|---|---|---|---|---|---|
| λ (nm) | Abs | |||||
|
Oxalis tuberosa var. oca rosa |
1AE |
1.19 |
Positive |
348.5 | 1.003 | Flavonoids (cinnamoyl group) |
| 325.5 | 1.120 |
Ferulic acid and coumarins |
||||
| 303.5 | 1.225 | |||||
| 1EAS |
0.50 |
Positive | 323.0 | 0.774 | Flavonoids (cinnamoyl group) | |
| 282.4 | 1.243 | Flavonoids (benzoyl group) Ferulic acid and coumarins |
||||
|
Ullucus tuberosus |
3AE |
7.29 |
Positive | 348.5 | 0.631 | Flavonoids (cinnamoyl group) |
| 267.5 | 1.147 | Flavonoids (benzoyl group) | ||||
|
3EAS |
0.11 |
Positive | 351.2 | 0.704 | Flavonoids (cinnamoyl group) | |
| 277.2 | 1.191 | Flavonoids (benzoyl group) Ferulic acid and coumarins | ||||
|
Solanum tuberosum subsp. andigena var. miskila colorada |
5AE |
2.00 |
Positive |
343.0 | 0.791 | Flavonoids (cinnamoyl group) |
| 300.5 | 0.461 | Ferulic acid and coumarins | ||||
|
5EAS |
0.16 |
Positive |
341.5 | 0.617 | Flavonoids (cinnamoyl group) | |
| 321.5 | 0.964 | Ferulic acid and coumarins | ||||
| 300.5 | 0.447 | |||||
| 5EE |
2.47 |
Positive |
322.0 | 1.110 | Flavonoids (cinnamoyl group) | |
| 305.5 | 0.965 | Ferulic acid and coumarins | ||||
| 294.5 | 0.956 | Flavonoids (benzoyl group) | ||||
|
Solanum tuberosum subsp. andigena var. cuarentona |
7EE |
3.15 |
Positive |
322.0 | 1.843 | Flavonoids (cinnamoyl group) |
| 305.5 | 1.619 | Ferulic acid and coumarins | ||||
3.5. Mutagenic Compounds Biodegradation
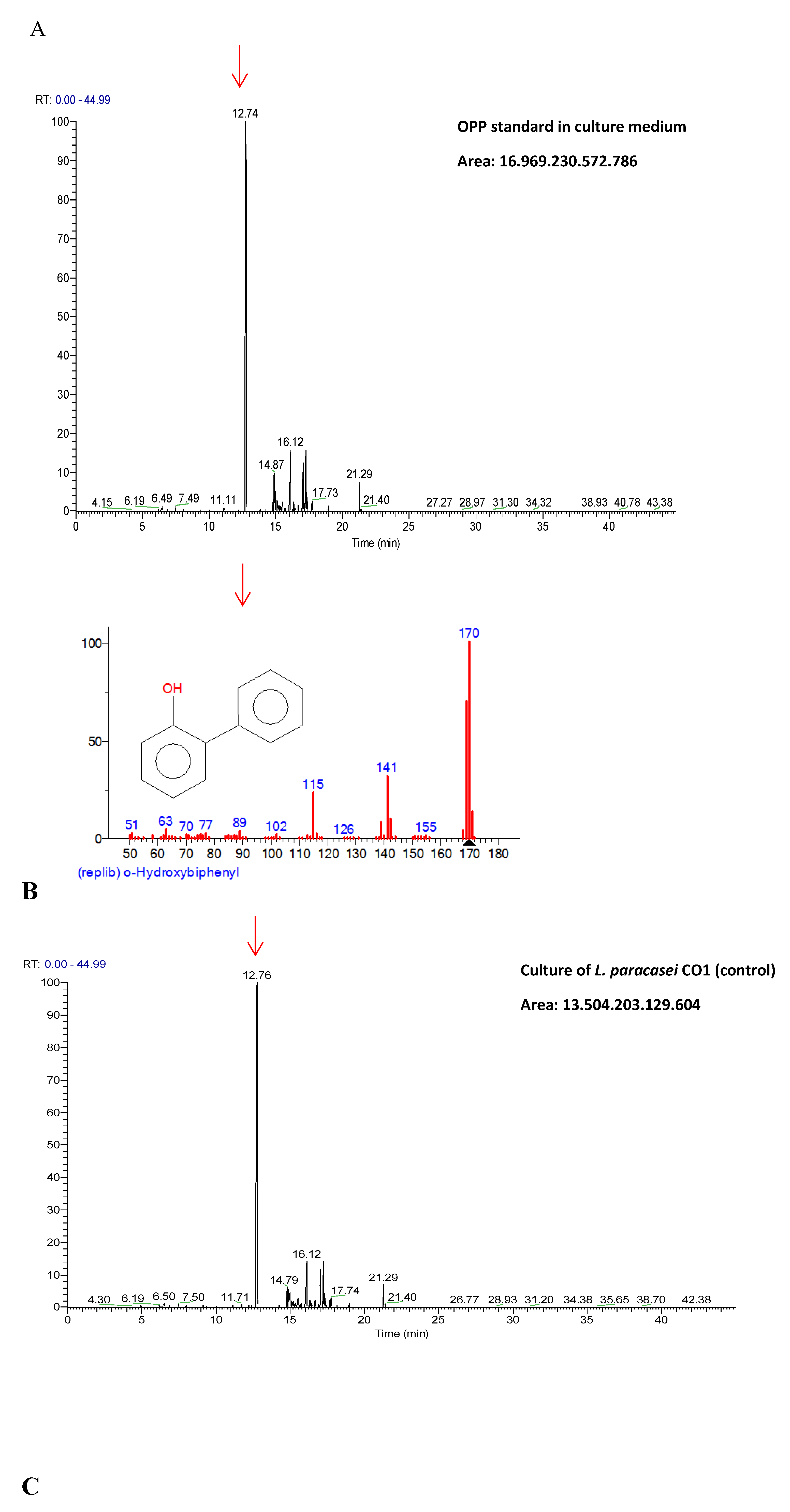
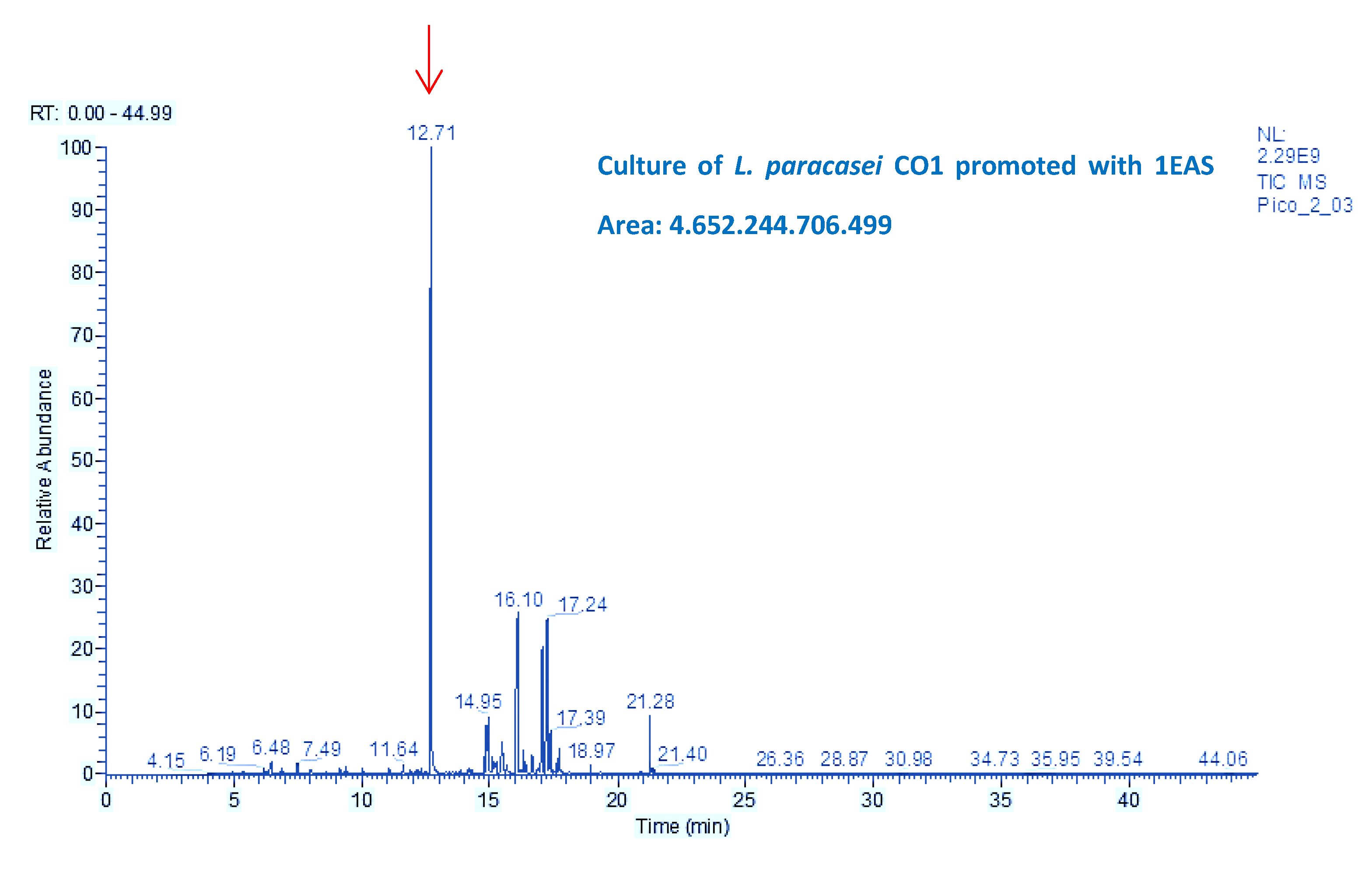
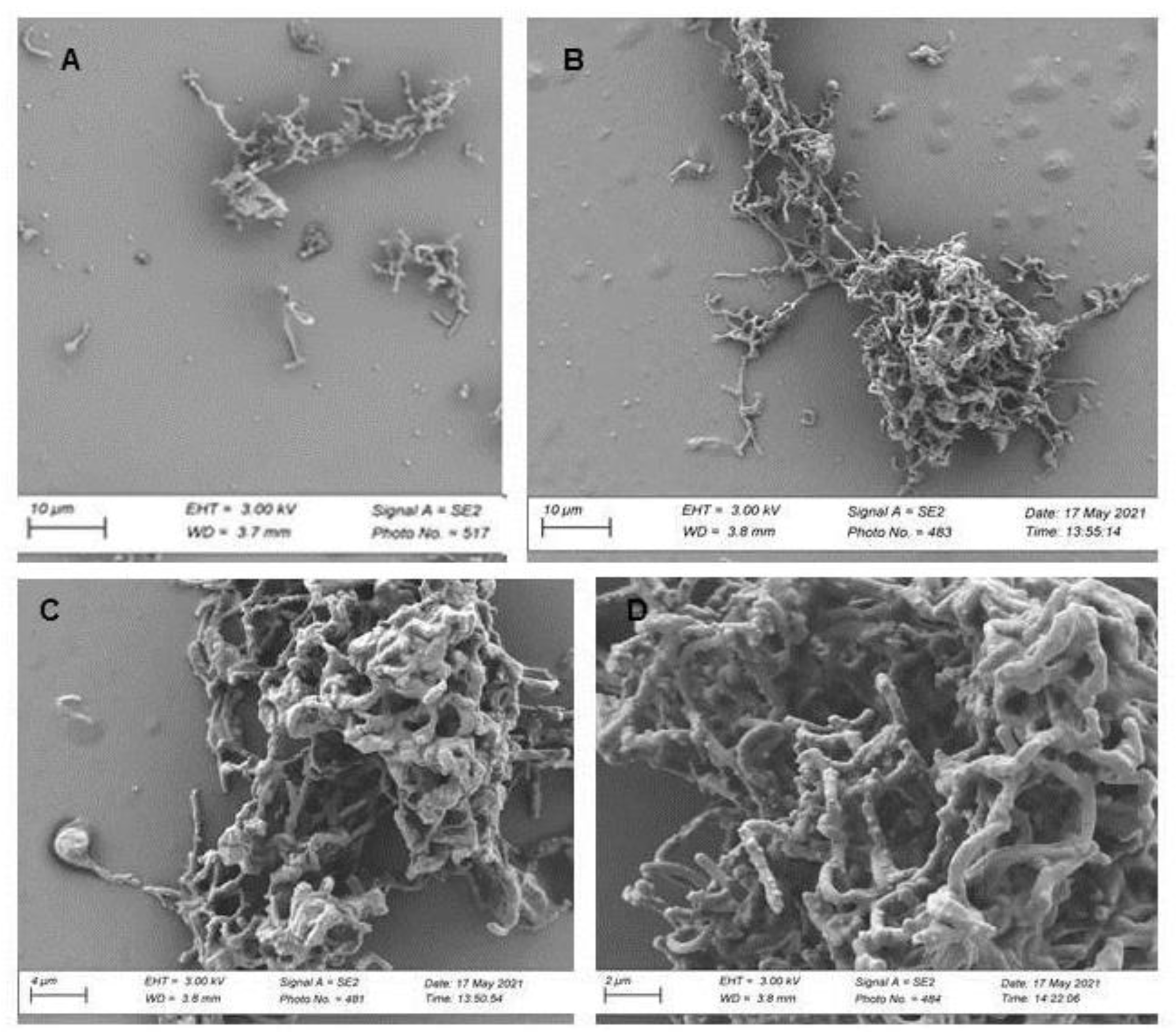
3.6. Air-liquid Surface Activity
| Supernatants | Oil spreading halos (mm) | Surfactant activity* |
|---|---|---|
| Lacticaseibacillus paracasei CO1 | 80 ± 5 | 100% |
| L. paracasei CO1 + OPP | 160 ± 0 | 200% |
| L. paracasei CO1-EAS + OPP | 320 ± 0 | 400% |
| L. paracasei CO1 + PhOH | 88 ± 1 | 110% |
| L. paracasei CO1- EAS + PhOH | 169 ± 2 | 212% |
| Lactiplantibacillus plantarum ATCC 10241 | 105 ± 2 | 100% |
| Lp. plantarum ATCC 10241 + PhOH | 130 ± 1 | 124% |
| Tween 80 | 50 ± 3 | 62.5% |
3.7. Phenol Oxidase Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Erginkaya, Z.; Konuray-Altun, G. Potential biotherapeutic properties of lactic acid bacteria in foods. Food Biosci. 2022, 46, 101544. [CrossRef]
- Abedin, M.M.; Chourasia, R.; Phukon, L.C.; Sarkar, P.; Ray, R.C.; Singh, S.P.; Rai, A.K. Lactic acid bacteria in the functional food industry: Biotechnological properties and potential applications. Crit. Rev. Food Sci. Nutr. 2023, 5, 1–9.
- Nuñez, I.M.; Verni, M.C.; Argañaraz Martinez, F.E.; Babot, J.D.; Terán, V.; Danilovich, M.E.; Cartagena, E.; Alberto, M.R.; Arena, M.E. Novel lactic acid bacteria strains from regional peppers with health-promoting potential. Fermentation 2024, 10, 209. [CrossRef]
- Dable-Tupas, G.; Otero, M.C.B.; Bernolo, L., Eds.; Functional foods and nutraceuticals: bioactive components, formulations, and innovations. Springer Nature, Berlin, 2020.
- Reque, P.M.; Brandelli, A.. Encapsulation of probiotics and nutraceuticals: Applications in functional food industry. Trends Food Sci. Technol. 2021, 114, 1–10. [CrossRef]
- Chantre-López, A.R.; Solanilla-Duque, J.F.; Ascacio-Valdes, J.A.; Salazar-Sánchez, M. del R.; Flores-Gallegos, A.C.; Nery-Flores, S.D., et al. Andean tubers: traditional medicine and other applications. Plant Sci. Today 2024, 11, 342–352. [CrossRef]
- Leidi, E.O.; Altamirano, A.M.;, Mercado, G.; Rodriguez, J.P.; Ramos, A.; Alandia, G.; Sørensen, M.; Jacobsen, S.E.,. Andean roots and tubers crops as sources of functional foods. J. Funct. Foods 2018, 51, 86–93. [CrossRef]
- Camire, M.E.; Kubow, S.; Donnelly, D.J. Potatoes and human health. Crit. Rev. Food Sci. Nutr. 2009, 49, 823–840. [CrossRef]
- Rosas-Cruz, G.P.; Silva-Correa, C.R.; Calderón-Peña, A.A.; Villarreal-La Torre, V.E.; Aspajo-Villalaz, C.L.; CruzadoRazco, J.L.; Rosario-Chávarri, J.; Rodríguez-Soto, J.; Pretel-Sevillano, O.; Sagástegui-Guarniz, W.; González Siccha, A. Wound healing activity of an ointment from Solanum tuberosum L. “Tumbay yellow potato” on Mus musculus Balb/c. Pharmacogn J. 2020, 12, 1268–1275. [CrossRef]
- Campos, D.; Noratto, G.; Chirinos, R.; Arbizu, C.; Roca, W.; Cisneros-Zevallos, L., Antioxidant capacity and secondary metabolites in four species of Andean tuber crops: native potato (Solanum sp.), mashua (Tropaeolum tuberosum Ruiz & Pavon), oca (Oxalis tuberosa Molina) and ulluco (Ullucus tuberosus Caldas). J. Sci. Food Agric. 2006, 86, 1481–1488. [CrossRef]
- Andre, C.M.; Ghislain, M.; Bertin, P.; Oufir, M.; Herrera, M.R.; Hoffmann, L.; Hausman, J.F.; Larondelle, Y.; Evers, D. Andean potato cultivars (Solanum tuberosum L.) as a source of antioxidant and mineral micronutrients. J. Agric. Food Chem. 2007, 55, 366–378. [CrossRef]
- Vescovo, D.; Manetti, C.; Ruggieri, R.; Spizzirri, U.G.; Aiello, F.; Martuscelli, M.; Restuccia, D. The Valorization of potato peels as a functional ingredient in the food industry: A comprehensive review. Foods 2025, 14, 1333. [CrossRef]
- Mis-Solval, K.E.; Jiang, N.; Yuan, M.; Joo, K.H.; Cavender, G.A. The Effect of the ultra-high-pressure homogenization of protein encapsulants on the survivability of probiotic cultures after spray drying. Foods 2019, 8, 689. [CrossRef]
- Lane, D.J. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds; Wiley: New York, 1991; pp. 115–175.
- O'Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [CrossRef]
- Amaya, S; Pereira, J.A.; Borkosky, S.A.; Valdez, J.C.; Bardón, A.; Arena, M.E. Inhibition of quorum sensing in Pseudomonas aeruginosa by sesquiterpene lactones. Phytomedicine 2012, 19,1173–1177. [CrossRef]
- Cartagena, E.; Orphèe, C.H.; Verni, M.C.; Arena, M.E.; González, S.N.; Argañaraz, M.I.; Bardón, A. Patent: Medio de Cultivo Promotor y Bacterias No Patógenas Detoxificantes de Compuestos Mutagénicos/Carcinogénicos. Instituto Nacional de la Propiedad Industrial-INPI Nº 20190102418, Argentina, 2021.
- Walter, V.; Syldatk, C.; Hausmann, R. Screening Concepts for the Isolation of Biosurfactant Producing Microorganisms. In Biosurfactants. Advances in Experimental Medicine and Biology; Sen, R., Ed.; Springer: New York, NY, 2010; pp. 1–13.
- Sambanthamoorthy, K.; Feng, X.; Patel, R.; Patel, S.; Paranavitana, C. Antimicrobial and antibiofilm potential of biosurfactants isolated from lactobacilli against multi-drug-resistant pathogens. BMC Microbiol. 2014, 14, 1–9. [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [CrossRef]
- Chen, Z. Application of UV-vis spectroscopy in the detection and analysis of substances. Transactions on Materials, Biotechnology and Life Sciences 2024, 3, 131-136. [CrossRef]
- Verni, M.C.; Orphèe, C.H.; González, S.N.; Bardón, A.; Arena, M.E.; Cartagena, E. Flourensia fiebrigii SF Blake in combination with Lactobacillus paracasei subsp. paracasei CE75. A novel antipathogenic and detoxifying strategy. LWT 2022, 156, 113023. [CrossRef]
- ECHA European Chemicals Agency Documents, 2021. https://echa.europa.eu/documents/10162/de108693-1d4f-abfe-818e-9cea812ab4c2.
- Lachman, J.; Hamouz, K. Red and purple coloured potatoes as a significant antioxidant source in human nutrition – a review. Plant Soil Environ. 2005, 51, 477–482. [CrossRef]
- Campos, D.; Betalleluz-Pallardel, I.; Chirinos, R.; Aguilar-Galvez, A.; Noratto, G.; Pedreschi, R. Prebiotic effects of yacon (Smallanthus sonchifolius Poepp. & Endl.), a source of fructooligosaccharides and phenolic compounds with antioxidant activity. Food Chem. 2012, 135, 1592–1599. [CrossRef]
- Verediano, T.A.; Viana, M.L.; Vaz-Tostes, M.D.G.; Costa, N. The potential prebiotic effects of yacon (Smallanthus sonchifolius) in colorectal cancer. Curr. Nutr. Food Sci. 2021, 17, 167–175. [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; Verbeke, K.; Reid, G.. Expert consensus document: The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [CrossRef]
- Palai, S.; Derecho, C.M.P.; Kesh, S.S.; Egbuna, C.; Onyeike, P.C. Prebiotics, probiotics, synbiotics and its importance in the management of diseases. In Functional Foods and Nutraceuticals: Bioactive Components, Formulations and Innovations 2020. Cham: Springer International Publishing, pp. 173–196.
- Berlanga, M.; Guerrero, R. Living together in biofilms: the microbial cell factory and its biotechnological implications. Microb. Cell Fact. 2016, 15, 165–176. [CrossRef]
- Frassinetti, S.M.G.; Moccia, E.; Longo, V.; Di Gioia, D. Antimicrobial and antibiofilm activity of Cannabis sativa L. seeds extract against Staphylococcus aureus and growth effects on probiotic Lactobacillus spp. LWT 2020, 124, 109149. [CrossRef]
- Cartagena, E.; Arena, M.E.; Verni, M.C.; Bardón, A.. Natural terpenoids as a valuable resource of selective health-beneficial biofilm promoters, in Rowland, S. (Ed.), Biofilms: Advances in Research and Applications. 2021. Nova Science Publishers, New York, pp. 159–186.
- Santonoceta, G.D.G.; Sgarlata, C.. pH-Responsive Cobalt(II)-coordinated assembly containing quercetin for antimicrobial applications. Molecules 2023, 28, 5581. [CrossRef]
- Holser, R. Principal component analysis of phenolic acid spectra. ISRN spectroscopy 2012. [CrossRef]
- ECHA, 2023. https://echa.europa.eu/substance-information/-/substanceinfo/100.001.812.
- Parlindungan, E.; Dekiwadia, C.; Tran, K.T.; Jones, O.A.; May, B.K. Morphological and ultrastructural changes in Lactobacillus plantarum B21 as an indicator of nutrient stress. LWT 2018, 92, 556–563. [CrossRef]
- Matthews, A.; Grimaldi, A.; Walker, M.; Bartowsky, E.; Grbin, P.; Jiranek, V. Lactic acid bacteria as a potential source of enzymes for use in vinification. Appl. Environ. Microbiol. 2004, 70, 5715–5731. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




