Submitted:
08 August 2025
Posted:
12 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Accuracy
2.2. Precision
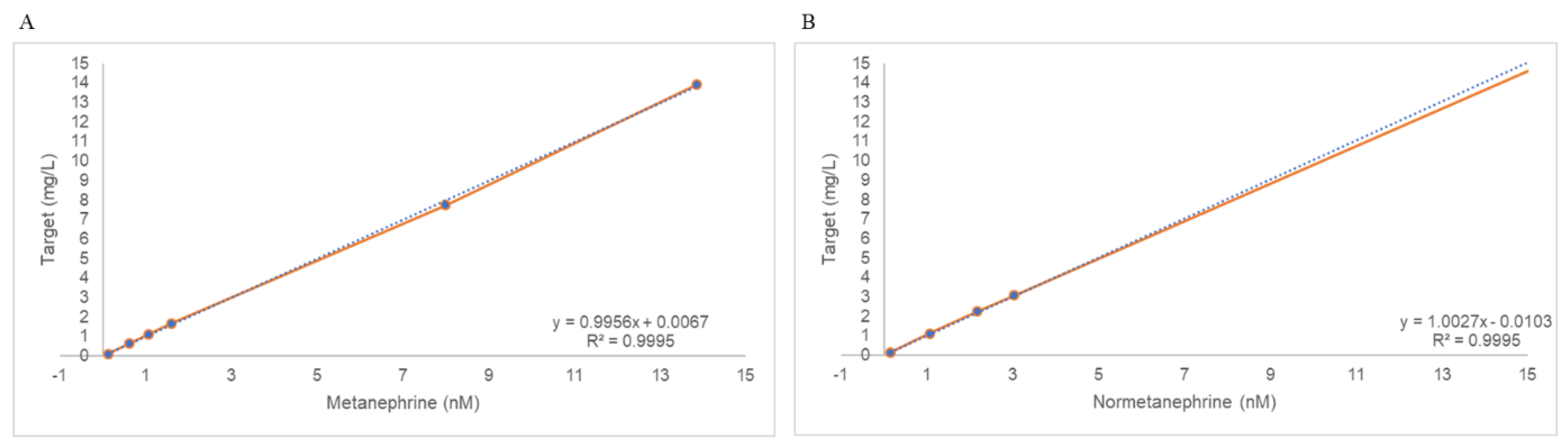
2.3. Linearity
2.4. Carryover
2.5. Lower Limit of Quantification
| Analyte | Expected concentration (nmol/L) | Average concentration (nmol/L, n = 5) | CV (%) |
Accuracy (%) |
| Metanephrine | 0.307 | 0.301 | 3.9 | 98.1 |
| 0.245 | 0.247 | 2.8 | 100.7 | |
| 0.184 | 0.185 | 2.9 | 100.4 | |
| 0.123 | 0.126 | 5.5 | 102.8 | |
| 0.061 | 0.072 | 7.3 | 118.1 | |
| Normetanephrine | 0.541 | 0.502 | 10.9 | 92.8 |
| 0.432 | 0.409 | 11.0 | 94.6 | |
| 0.324 | 0.272 | 6.4 | 84.0 | |
| 0.216 | 0.150 | 37.1 | 69.6 | |
| 0.108 | 0.072 | 27.1 | 66.8 |
2.6. Ion Suppression
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Sample Preparation
4.3. SPE
4.4. Analytical Procedure
4.5. Method Validation
4.5.1. Accuracy
4.5.2. Precision
4.5.3. Linearity
4.5.4. Carryover
4.5.5. LLOQ
4.5.6. Ion Suppression
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CV | Coefficient of variation |
| DOPA | Dihydroxyphenylalanine |
| HPLC | High-performance liquid chromatography |
| IS | Internal standard |
| LC-MS/MS | Liquid chromatography and tandem mass spectrometry |
| LLOQ | Lower limit of quantification |
| SPE | Solid-phase extraction |
References
- De Silva, D.C.; Wijesiriwardene, B. The adrenal glands and their functions. Ceylon Medical Journal. 2009, 52. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Lattke, P.; Herberg, M.; Siegert, G.; Qin, N.; Därr, R.; Hoyer, J.; Villringer, A.; Prejbisz, A.; Januszewicz, A.; et al. Reference intervals for plasma free metanephrines with an age adjustment for normetanephrine for optimized laboratory testing of phaeochromocytoma. Ann Clin Biochem. 2013, 50, 62–69. [Google Scholar] [CrossRef]
- Chan, E.C.; Ho, P.C. Chromatographic measurements of catecholamines and metanephrines. Chromatographic Methods in Clinical Chemistry and Toxicology. 2007, 101–126. [Google Scholar]
- Meiser, J.; Weindl, D.; Hiller, K. Complexity of dopamine metabolism. Cell Communication and Signaling. 2013, 11, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Pacak, K.; Linehan, W.M.; Eisenhofer, G.; Walther, M.M.; Goldstein, D.S. Recent advances in genetics, diagnosis, localization, and treatment of pheochromocytoma. Annals of internal medicine. 2001, 134, 315–329. [Google Scholar] [CrossRef]
- Knudson Jr, A.G.; Strong, L. Mutation and cancer: neuroblastoma and pheochromocytoma. American journal of human genetics. 1972, 24, 514. [Google Scholar] [PubMed]
- Goldenberg, M.; Serlin, I.; Edwards, T.; Rapport, M.M. Chemical screening methods for the diagnosis of pheochromocytoma: I. Nor-epinephrine and epinephrine in human urine. The American Journal of Medicine. 1954, 16, 310–327. [Google Scholar] [CrossRef]
- Smythe, G.; Edwards, G.; Graham, P.; Lazarus, L. Biochemical diagnosis of pheochromocytoma by simultaneous measurement of urinary excretion of epinephrine and norepinephrine. Clinical chemistry. 1992, 38, 486–492. [Google Scholar] [CrossRef]
- Lenders, J.W.; Keiser, H.R.; Goldstein, D.S.; Willemsen, J.J.; Friberg, P.; Jacobs, M.-C.; Kloppenborg, P.W.; Thien, T.; Eisenhofer, G. Plasma metanephrines in the diagnosis of pheochromocytoma. Annals of internal medicine. 1995, 123, 101–109. [Google Scholar] [CrossRef]
- Raber, W.; Raffesberg, W.; Bischof, M.; Scheuba, C.; Niederle, B.; Gasic, S.; Waldhäusl, W.; Roden, M. Diagnostic efficacy of unconjugated plasma metanephrines for the detection of pheochromocytoma. Archives of Internal Medicine. 2000, 160, 2957–2963. [Google Scholar] [CrossRef]
- Eisenhofer, G. biochemical diagnosis of pheochromocytoma—is it time to switch to plasma-free metanephrines? The Journal of Clinical Endocrinology & Metabolism. 2003, 88, 550–552. [Google Scholar]
- Eisenhofer, G.; Walther, M.; Keiser, H.; Lenders, J.; Friberg, P.; Pacak, K. Plasma metanephrines: a novel and cost-effective test for pheochromocytoma. Brazilian Journal of Medical and Biological Research. 2000, 33, 1157–1169. [Google Scholar] [CrossRef]
- Grossman, E.; Goldstein, D.S.; Hoffman, A.; Keiser, H.R. Glucagon and clonidine testing in the diagnosis of pheochromocytoma. Hypertension. 1991, 17, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Pappachan, J.M.; Tun, N.N.; Arunagirinathan, G.; Sodi, R.; Hanna, F.W. Pheochromocytomas and hypertension. Current hypertension reports. 2018, 20, 1–13. [Google Scholar] [CrossRef] [PubMed]
- de Jong, W.H.; Graham, K.S.; van der Molen, J.C.; Links, T.P.; Morris, M.R.; Ross, H.A.; de Vries, E.G.; Kema, I.P. Plasma free metanephrine measurement using automated online solid-phase extraction HPLC–tandem mass spectrometry. Clinical chemistry. 2007, 53, 1684–1693. [Google Scholar] [CrossRef] [PubMed]
- Petteys, B.J.; Graham, K.S.; Parnás, M.L.; Holt, C.; Frank, E.L. Performance characteristics of an LC–MS/MS method for the determination of plasma metanephrines. Clinica Chimica Acta. 2012, 413, 1459–1465. [Google Scholar] [CrossRef]
- de Jong, W.H.; de Vries, E.G.; Kema, I.P. Current status and future developments of LC-MS/MS in clinical chemistry for quantification of biogenic amines. Clinical biochemistry. 2011, 44, 95–103. [Google Scholar] [CrossRef]
- Peaston, R.T.; Graham, K.S.; Chambers, E.; van der Molen, J.C.; Ball, S. Performance of plasma free metanephrines measured by liquid chromatography–tandem mass spectrometry in the diagnosis of pheochromocytoma. Clinica chimica acta. 2010, 411, 546–552. [Google Scholar] [CrossRef]
- Lenders, J.; Eisenhofer, G.; Armando, I.; Keiser, H.; Goldstein, D.; Kopin, I. Determination of metanephrines in plasma by liquid chromatography with electrochemical detection. Clinical chemistry. 1993, 39, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Lenders, J.W.; Duh, Q.Y.; Eisenhofer, G.; Gimenez-Roqueplo, A.P.; Grebe, S.K.; Murad, M.H.; Naruse, M.; Pacak, K.; Young, W.F., Jr.; Endocrine, S. Pheochromocytoma and paraganglioma: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2014, 99, 1915–1942. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Prejbisz, A.; Peitzsch, M.; Pamporaki, C.; Masjkur, J.; Rogowski-Lehmann, N.; Langton, K.; Tsourdi, E.; Pęczkowska, M.; Fliedner, S.; et al. Biochemical Diagnosis of Chromaffin Cell Tumors in Patients at High and Low Risk of Disease: Plasma versus Urinary Free or Deconjugated O-Methylated Catecholamine Metabolites. Clin Chem. 2018, 64, 1646–1656. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xiao, H.; Zhou, X.; Huang, X.; Li, Y.; Xiao, H.; Cao, X. ACCURACY OF PLASMA FREE METANEPHRINES IN THE DIAGNOSIS OF PHEOCHROMOCYTOMA AND PARAGANGLIOMA: A SYSTEMATIC REVIEW AND META-ANALYSIS. Endocr Pract. 2017, 23, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Luo, X.; Li, H.; Guan, Q.; Cheng, L. A simple and robust liquid chromatography tandem mass spectrometry assay for determination of plasma free metanephrines and its application to routine clinical testing for diagnosis of pheochromocytoma. Biomed Chromatogr. 2019, 33, e4622. [Google Scholar] [CrossRef]
- Weismann, D.; Peitzsch, M.; Raida, A.; Prejbisz, A.; Gosk, M.; Riester, A.; Willenberg, H.S.; Klemm, R.; Manz, G.; Deutschbein, T.; et al. Measurements of plasma metanephrines by immunoassay vs liquid chromatography with tandem mass spectrometry for diagnosis of pheochromocytoma. Eur J Endocrinol. 2015, 172, 251–260. [Google Scholar] [CrossRef]
- Clarke, M.W.; Cooke, B.; Hoad, K.; Glendenning, P. Improved plasma free metadrenaline analysis requires mixed mode cation exchange solid-phase extraction prior to liquid chromatography tandem mass spectrometry detection. Ann Clin Biochem. 2011, 48, 352–357. [Google Scholar] [CrossRef]
- Dubbelman, A.C.; van Wieringen, B.; Roman Arias, L.; van Vliet, M.; Vermeulen, R.; Harms, A.C.; Hankemeier, T. Strategies for Using Postcolumn Infusion of Standards to Correct for Matrix Effect in LC-MS-Based Quantitative Metabolomics. J Am Soc Mass Spectrom. 2024, 35, 3286–3295. [Google Scholar] [CrossRef]
- Xiong, X.; Zhang, Y.; Zhao, R. Quantitative Measurement of Plasma Free Metanephrines by a Simple and Cost-Effective Microextraction Packed Sorbent with Porous Graphitic Carbon and Liquid Chromatography-Tandem Mass Spectrometry. J Anal Methods Chem. 2021, 2021, 8821276. [Google Scholar] [CrossRef]
- Fu, Y.; Li, W.; Picard, F. Assessment of matrix effect in quantitative LC-MS bioanalysis. Bioanalysis. 2024, 16, 631–634. [Google Scholar] [CrossRef]
- van Faassen, M.; Bischoff, R.; Eijkelenkamp, K.; de Jong, W.H.A.; van der Ley, C.P.; Kema, I.P. In Matrix Derivatization Combined with LC-MS/MS Results in Ultrasensitive Quantification of Plasma Free Metanephrines and Catecholamines. Anal Chem. 2020, 92, 9072–9078. [Google Scholar] [CrossRef]
- Deng, Y.; Liu, Q.; Liu, Z.; Zhao, H.; Zhou, W.; Zhang, C. Commutability Assessment of Processed Human Plasma Samples for Normetanephrine and Metanephrine Measurements Based on the Candidate Reference Measurement Procedure. Ann Lab Med. 2022, 42, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Yim, J.; Cha, J.H.; Kim, J.; Kim, K.J.; Nam, M.; Nam, M.H.; Lee, C.K.; Cho, Y.; Yun, S.G. HPLC-MS/MS Method Validation for Antifungal Agents in Human Serum in Clinical Application. Clinical Laboratory. 2023, 69. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Choi, H.; Cha, J.H.; Kim, J.; Nam, M.-H.; Nam, M.; Lee, C.K.; Cho, Y.; Yun, S.G. Development and Validation of a U-HPLC-MS/MS Method for the Concurrent Measurement of four Immunosuppressants in Whole Blood. Clinical Laboratory. 2022, 68. [Google Scholar] [CrossRef] [PubMed]
| Analyte | Nominal concentration (mg/L) | Mean | SD | Accuracy (%) |
| Metanephrine | 0.30 | 0.30 | 0.01 | 101.5 |
| 0.93 | 0.90 | 0.02 | 96.5 | |
| 4.94 | 4.76 | 0.06 | 96.3 | |
| Normetanephrine | 0.67 | 0.65 | 0.02 | 98.1 |
| 1.50 | 1.44 | 0.06 | 96.2 | |
| 8.26 | 7.91 | 0.20 | 95.7 |
| Analyte | Nominal concentration (mg/L) | Within run (n = 20) | Between run (n = 5) | |||||||
| Mean | CV (%) |
SD | Accuracy (%) | Mean | CV (%) |
SD | Accuracy (%) | |||
| Metanephrine | 0.30 | 0.30 | 1.4 | 0.00 | 99.8 | 0.30 | 1.7 | 0.01 | 100.7 | |
| 4.94 | 4.77 | 1.4 | 0.07 | 96.5 | 4.77 | 1.5 | 0.07 | 96.5 | ||
| Normetanephrine | 0.67 | 0.66 | 4.2 | 0.03 | 98.0 | 0.62 | 7.0 | 0.04 | 93.1 | |
| 8.26 | 8.13 | 1.8 | 0.15 | 98.4 | 7.95 | 3.0 | 0.24 | 96.3 | ||
| Analyte | Low 1 (n = 10) |
Low 3 (n = 10) |
F test p-value |
t-test p-value |
||
| Mean | SD | Mean | SD | |||
| Metanephrine | 0.31 | 0.03 | 0.30 | 0.03 | 0.34 | 0.29 |
| Normetanephrine | 0.69 | 0.13 | 0.62 | 0.04 | 0.45 | 0.07 |
| Analyte | Analyte concentration (nmol/L) | Recovery | Matrix Factor |
Process efficiency | IS-normalized Recovery |
Is-normalized Matrix Factor |
Is-normalized Process efficiency |
| A/C (%) | C/B (%) | A/B (%) | (Aanalytes/Canalytes)/ (AIS/CIS) (%) |
(Canalytes/Banalytes)/ (CIS/BIS) (%) |
(Aanalytes/Banalytes)/ (AIS/BIS) (%) |
||
| Metanephrine | 0.6 | 122% | 94% | 11% | 108% | 112% | 121% |
| 1.3 | 122% | 88% | 12% | 107% | 102% | 110% | |
| 2.5 | 116% | 97% | 11% | 111% | 101% | 112% | |
| Normetanephrine | 1.3 | 170% | 58% | 2% | 86% | 106% | 92% |
| 2.5 | 140% | 91% | 3% | 101% | 111% | 111% | |
| 5.0 | 129% | 89% | 4% | 112% | 98% | 110% |
| Analyte | Precursor ion (m/z) | Product ion (m/z) | RT (min) | DP (V) | CE (V) | CXP (V) | |
| Metanephrine | 180.036 | 148.2 | 2.16 | 136 | 23 | 10 | |
| Metanephrine2 | 180.036 | 165.2 | 2.16 | 136 | 30 | 10 | |
| IS_Metanephrine | 183.051 | 151.2 | 2.16 | 141 | 23 | 8 | |
| Normetanephrine | 166.100 | 134.1 | 2.68 | 100 | 26 | 7 | |
| Normetanephrine2 | 166.100 | 106.2 | 2.68 | 100 | 31 | 8 | |
| IS_Normetanephrine | 169.400 | 137.2 | 2.68 | 100 | 26 | 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




