Submitted:
08 August 2025
Posted:
13 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Model
2.2. Viral Stock
2.3. Depletion Treatment, Viral Infection, and Sample Collection
2.4. Immunophenotyping
2.5. qRT- PCR
2.6. ELISAs for Detection of DENV NS1 Antigen and DENV Anti-NS1 IgG
2.7. ELISAs for DENV and ZIKV Antibodies
2.8. Endpoint Dilution Binding Assay
2.9. EDIII and NS1 Antigen Production
2.10. Coupling of EDIII Antigens to Beads via Avidin-Biotin Interaction
2.11. Coupling of Biotinylated NS1 Antigens to Beads via Avidin-His-Tag Ab Interaction
2.12. EDIII and NS1 Multiplex Assay
2.13. In Vitro ADE Assay in K562 Cells
2.14. DENV Neutralization Assays
2.15. ZIKV Neutralization Assays
2.16. BAFF ELISA
2.17. Serum Cytokines Assay
2.18. Use of AI
2.19. Quantification and Statistical Analysis
3. Results
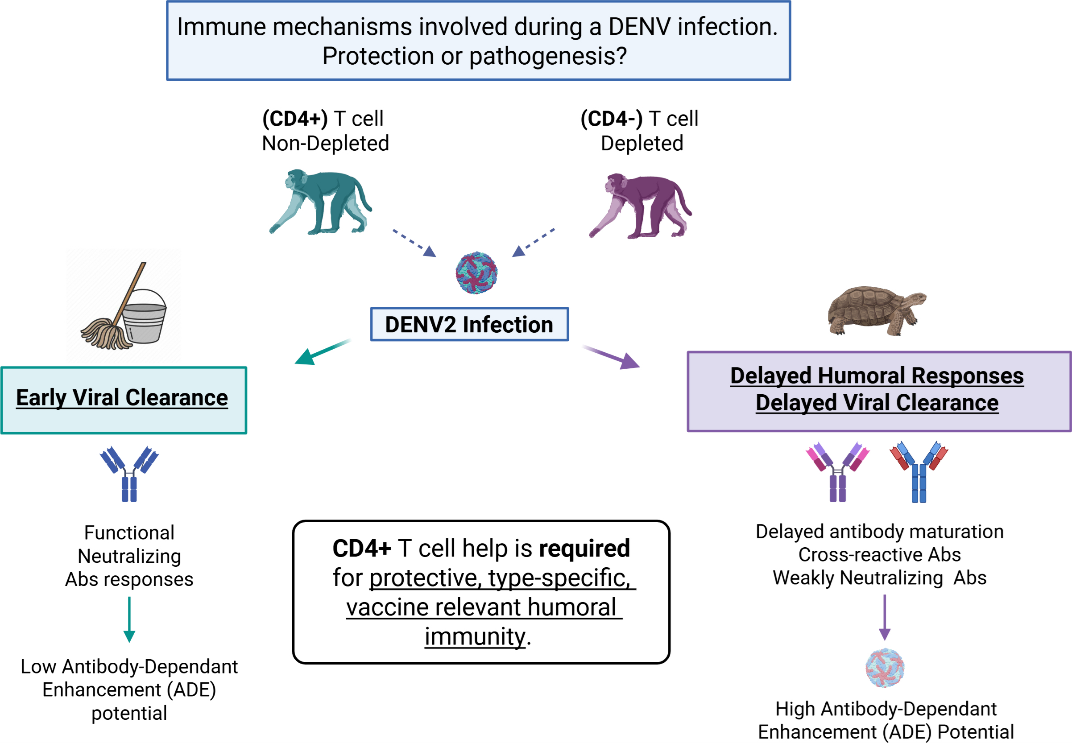
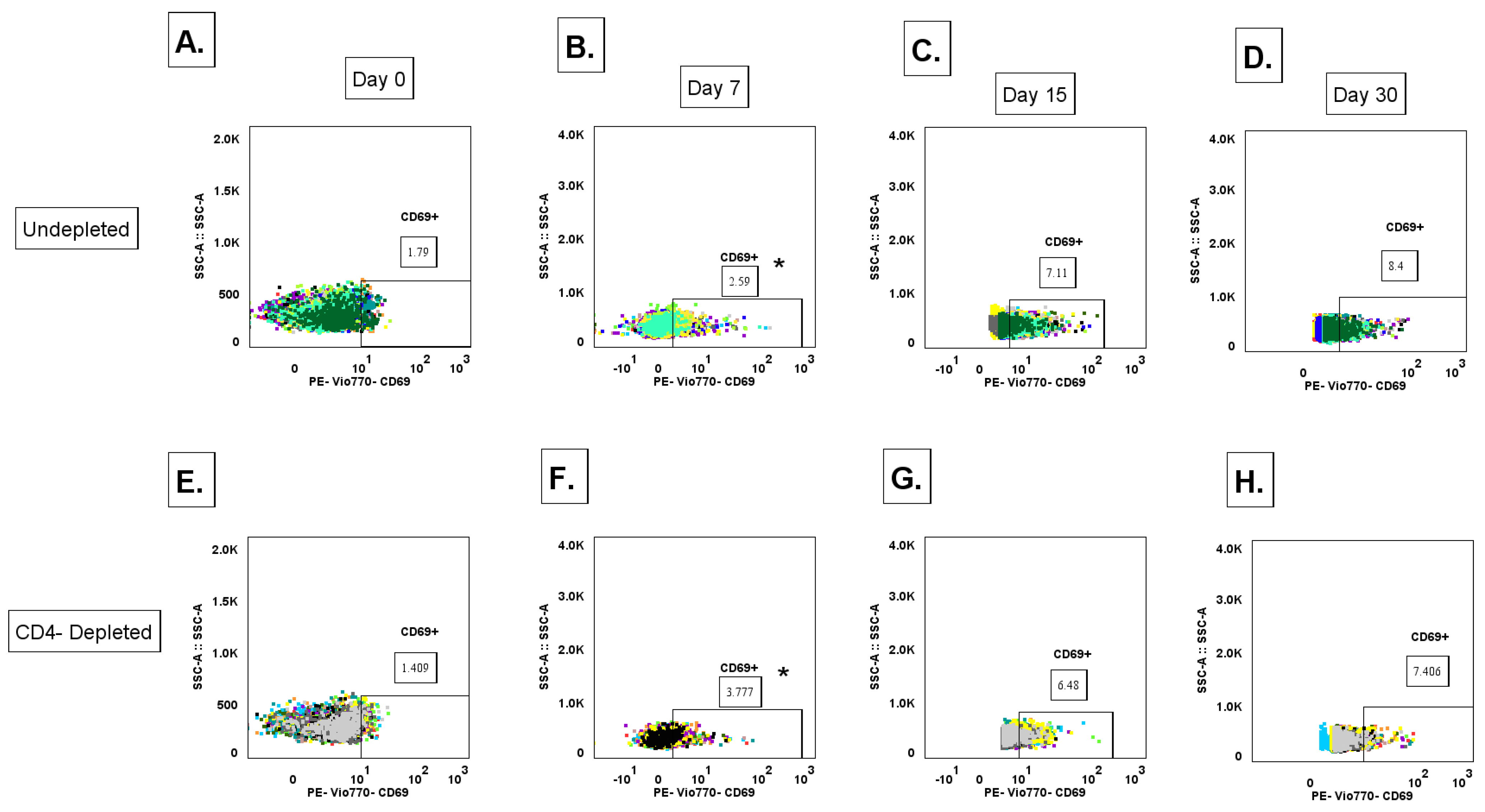
3.1. Assessment of CD4+ T Cell Depletion Treatment in Rhesus macaques
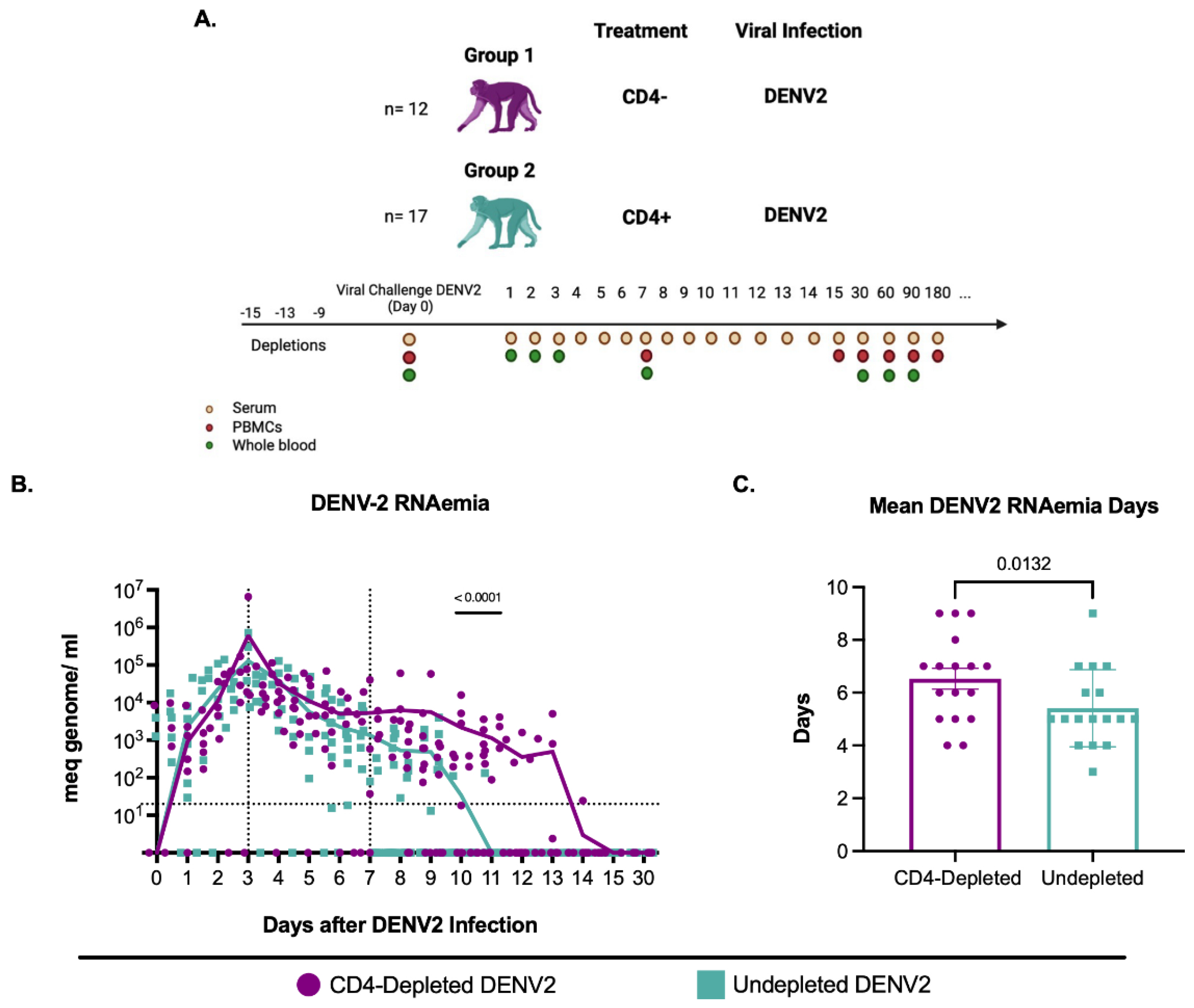
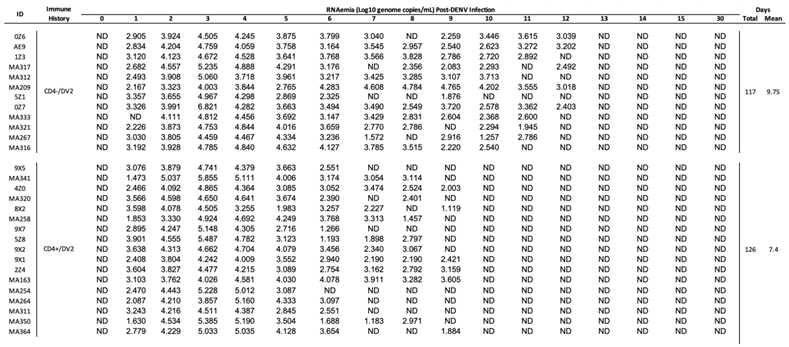
3.2. CD4+ T Cell Depletion Impairs DENV2 Viral Clearance in Flavivirus-Naïve Macaques
3.3. CD4+ T Cell Depletion Is Associated to Increased Monocytosis
3.4. CD4+ T Cell Depletion Results in Higher Detection of DENV NS1 Protein
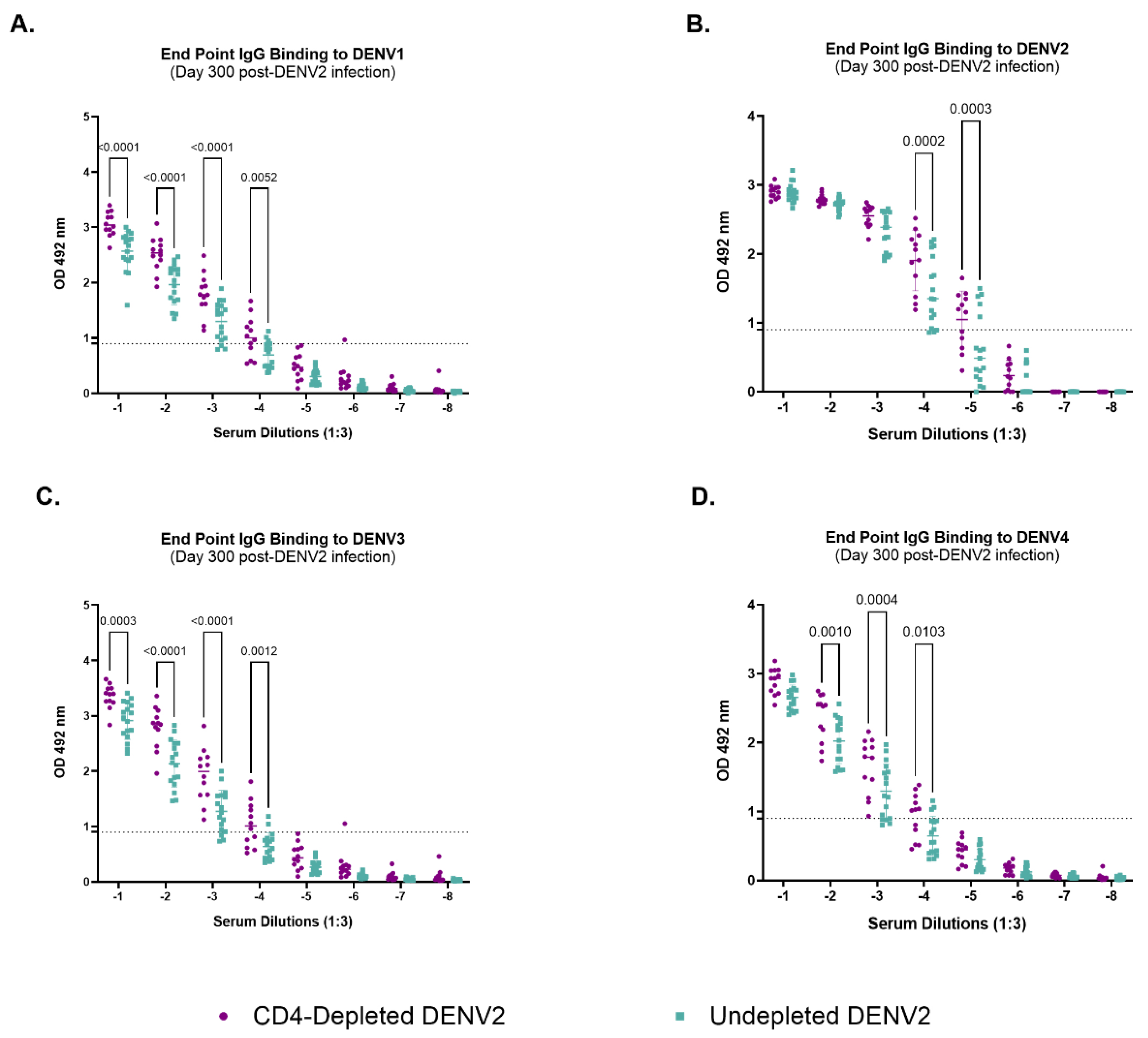
3.5. Lack of CD4+ T Cells Constrains Anti-DENV2 Antibody Induction
3.6. DENV Anti-NS1 Abs Are Modified by a Lack of CD4+ T Cells
3.7. Depletion of CD4+ T Cells Modifies IgG-Binding Capacities of Antibodies Against Heterologous Dengue After Primary DENV2 Infection
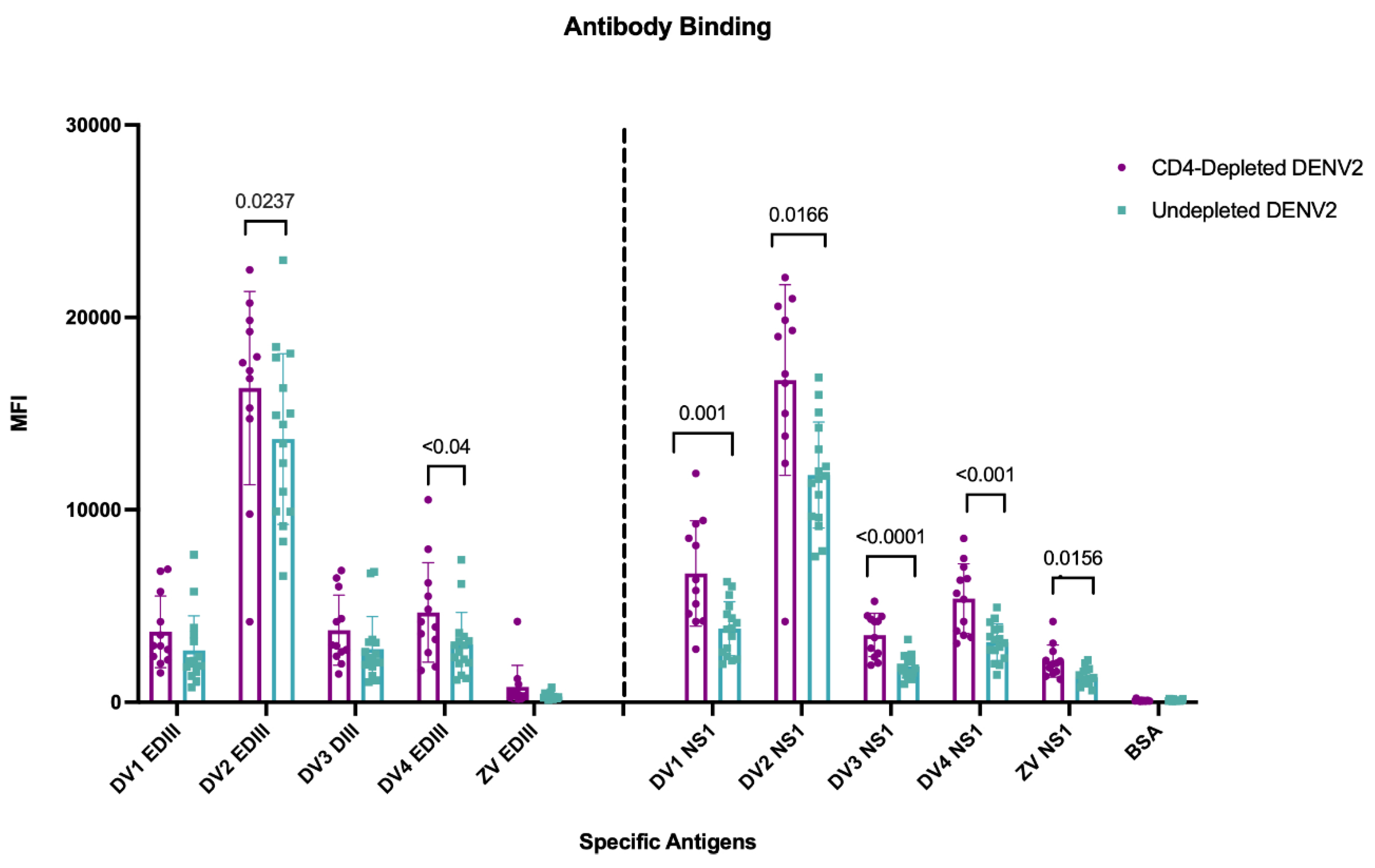
3.8. Lack of CD4+ T Cells During a Primary DENV2 Infection Reshapes Abs Binding to the Envelope EDIII Domain and to the NS1 Protein
3.9. Cross-Reactivity Is Increased by Lack of CD4 T Cells
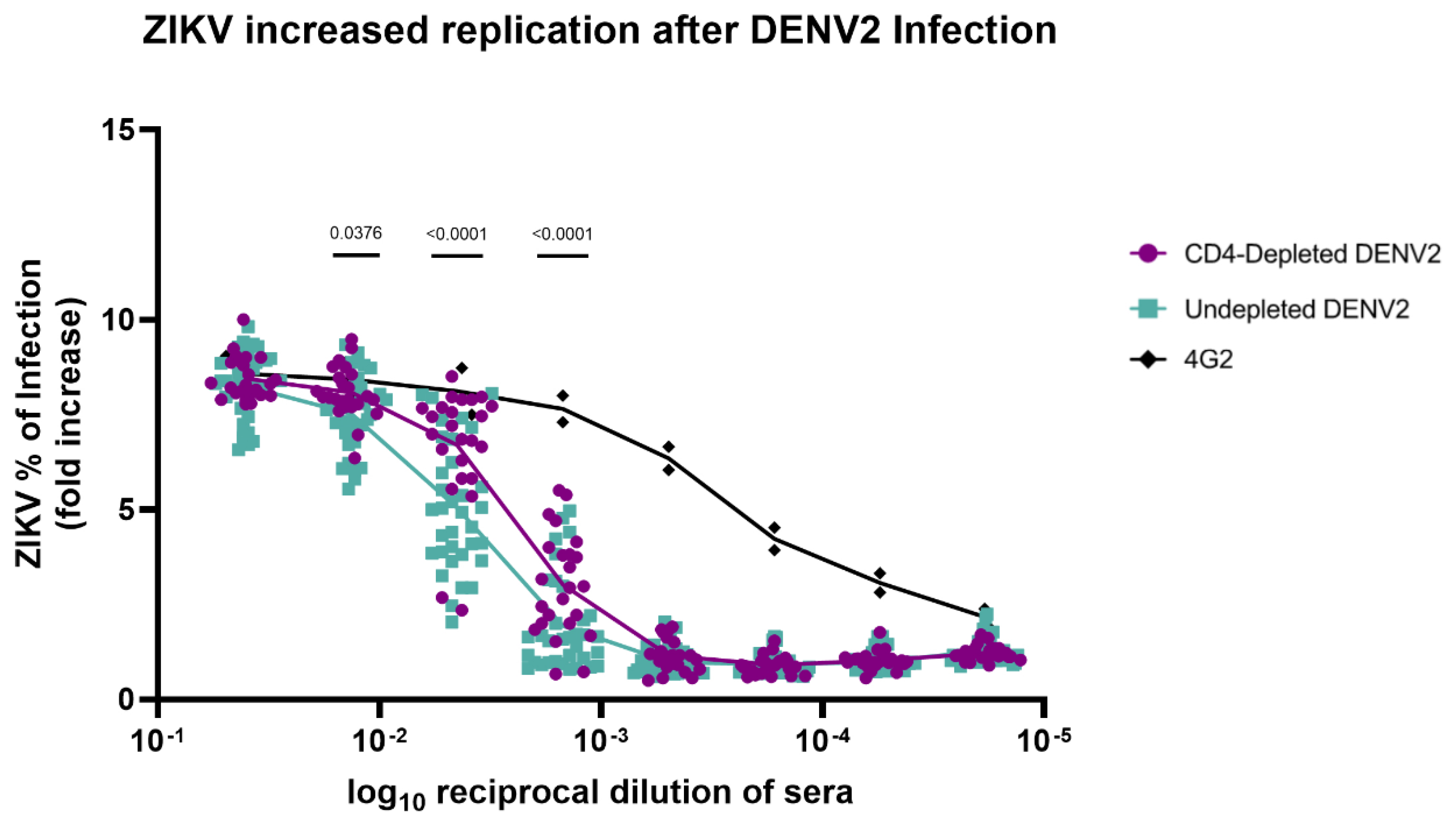
3.10. Absence of CD4+ T Cells Modifies Antibody Repertoire Properties Increasing ZIKV Replication
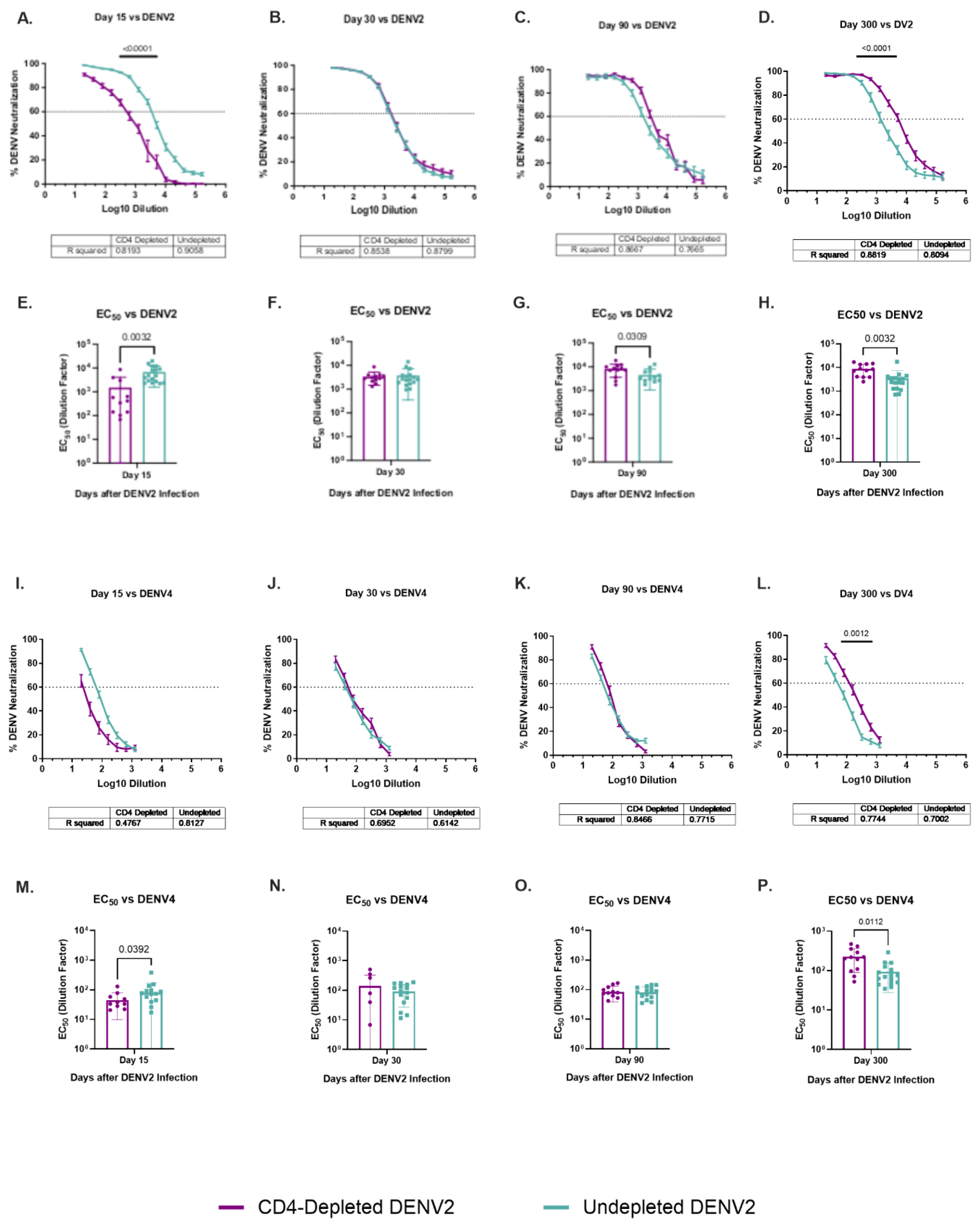
3.11. Dynamic of Specific DENV2 and Heterologous DENV4 Neutralizations Are Modulated by a Lack of CD4+ T Cells
3.12. Lack of CD4+ T Cell Priming Promotes Increased IgG+ MBC Polyclonal Activation During a Primary DENV2 Infection
3.13. CD4 T Cell Depletion Before DENV2 Infection Results in a Delay of Cytokine Profiles in Rhesus macaques
4. Discussion
5. Conclusions
6. Limitations
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tian, N.; Zheng, J.X.; Guo, Z.Y.; Li, L.H.; Xia, S.; Lv, S.; Zhou, X.N. Dengue incidence trends and its burden in major endemic regions from 1990 to 2019. Trop Med Infect Dis 2022, 7. [Google Scholar] [CrossRef]
- Wong, J.M.; Adams, L.E.; Durbin, A.P.; Muñoz-Jordán, J.L.; Poehling, K.A.; Sánchez-González, L.M.; Volkman, H.R.; Paz-Bailey, G. Dengue: A growing problem with new interventions. Pediatrics 2022, 149. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, J.; Johnson, B.A.; Xia, H.; Ku, Z.; Schindewolf, C.; Widen, S.G.; An, Z.; Weaver, S.C.; Menachery, V.D. , et al. Delta spike p681r mutation enhances sars-cov-2 fitness over alpha variant. bioRxiv 2021. [Google Scholar]
- Paixao, E.S.; Cardim, L.L.; Costa, M.C.N.; Brickley, E.B.; de Carvalho-Sauer, R.C.O.; Carmo, E.H.; Andrade, R.F.S.; Rodrigues, M.S.; Veiga, R.V.; Costa, L.C. , et al. Mortality from congenital zika syndrome - nationwide cohort study in brazil. N Engl J Med 2022, 386, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Martelli, C.M.T.; Raja, A.I.; Sanchez Clemente, N.; de Araùjo, T.V.B.; Ximenes, R.A.A.; Miranda-Filho, D.B.; Ramond, A.; Brickley, E.B. Epidemic preparedness: Prenatal zika virus screening during the next epidemic. BMJ Glob Health 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O. , et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef]
- Frieden, T.R. Six components necessary for effective public health program implementation. American Journal of Public Health 2014, 104, 17–22. [Google Scholar] [CrossRef]
- Akinsulie, O.C.; Idris, I. Global re-emergence of dengue fever: The need for a rapid response and surveillance. The Microbe 2024, 4, 100107. [Google Scholar] [CrossRef]
- Ebi, K.L.; Nealon, J. Dengue in a changing climate. Environmental Research 2016, 151, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.G.; Alvarez, M.; Halstead, S.B. Secondary infection as a risk factor for dengue hemorrhagic fever/dengue shock syndrome: An historical perspective and role of antibody-dependent enhancement of infection. Archives of virology 2013, 158, 1445–1459. [Google Scholar] [CrossRef]
- Halstead, S. Neutralization and antibody-dependent enhancement of dengue viruses. Advances in Viruses Research 2003, 60. [Google Scholar]
- Halstead, S.B.; Rojanasuphot, S.; Sangkawibha, N. Original antigenic sin in dengue. Am J Trop Med Hyg 1983, 32, 154–156. [Google Scholar] [CrossRef]
- Katzelnick, L.C.; Gresh, L.; Halloran, M.E.; Mercado, J.C.; Kuan, G.; Gordon, A.; Balmaseda, A.; Harris, E. Antibody-dependent enhancement of severe dengue disease in humans. Science 2017, 358, 929–932. [Google Scholar] [CrossRef]
- Ricciardi, M.J.; Magnani, D.M.; Grifoni, A.; Kwon, Y.C.; Gutman, M.J.; Grubaugh, N.D.; Gangavarapu, K.; Sharkey, M.; Silveira, C.G.T.; Bailey, V.K. , et al. Ontogeny of the b- and t-cell response in a primary zika virus infection of a dengue-naive individual during the 2016 outbreak in miami, fl. PLoS neglected tropical diseases 2017, 11, e0006000. [Google Scholar] [CrossRef] [PubMed]
- Wahala, W.M.; Silva, A.M. The human antibody response to dengue virus infection. Viruses 2011, 3, 2374–2395. [Google Scholar] [CrossRef]
- Martinez, D.R.; Metz, S.W.; Baric, R.S. Dengue vaccines: The promise and pitfalls of antibody-mediated protection. Cell Host Microbe 2021, 29, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Gallichotte, E.N.; Widman, D.G.; Yount, B.L.; Wahala, M.W.; Durbin, A.; Whitehead, S.; Sariol, C.A.; Crowe, J.E., Jr.; de Silva, A.M.; Baric, R.S. A new quaternary structure epitope on dengue virus serotype 2 is the target of durable type-specific neutralizing antibodies. MBio 2015, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Kurane, I.; Rothman, A.L.; Livingston, P.G.; Green, S.; Gagnon, S.J.; Janus, J.; Innis, B.L.; Nimmannitya, S.; Nisalak, A.; Ennis, F.A. Immunopathologic mechanisms of dengue hemorrhagic fever and dengue shock syndrome. Arch Virol Suppl 1994, 9, 59–64. [Google Scholar]
- Mapalagamage, M.; Weiskopf, D.; Sette, A.; De Silva, A.D. Current understanding of the role of t cells in chikungunya, dengue and zika infections. Viruses 2022, 14. [Google Scholar] [CrossRef]
- Mathew, A.; Rothman, A.L. Understanding the contribution of cellular immunity to dengue disease pathogenesis. Immunological reviews 2008, 225, 300–313. [Google Scholar] [CrossRef]
- Rivino, L. Understanding the human t cell response to dengue virus. Advances in experimental medicine and biology 2018, 1062, 241–250. [Google Scholar]
- de Alwis, R.; Bangs, D.J.; Angelo, M.A.; Cerpas, C.; Fernando, A.; Sidney, J.; Peters, B.; Gresh, L.; Balmaseda, A.; de Silva, A.D. , et al. Immunodominant dengue virus-specific cd8+ t cell responses are associated with a memory pd-1+ phenotype. Journal of virology 2016, 90, 4771–4779. [Google Scholar] [CrossRef]
- Elong Ngono, A.; Chen, H.W.; Tang, W.W.; Joo, Y.; King, K.; Weiskopf, D.; Sidney, J.; Sette, A.; Shresta, S. Protective role of cross-reactive cd8 t cells against dengue virus infection. EBioMedicine 2016, 13, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Weiskopf, D.; Angelo, M.A.; Bangs, D.J.; Sidney, J.; Paul, S.; Peters, B.; de Silva, A.D.; Lindow, J.C.; Diehl, S.A.; Whitehead, S. , et al. The human cd8+ t cell responses induced by a live attenuated tetravalent dengue vaccine are directed against highly conserved epitopes. Journal of virology 2015, 89, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Weiskopf, D.; Cerpas, C.; Angelo, M.A.; Bangs, D.J.; Sidney, J.; Paul, S.; Peters, B.; Sanches, F.P.; Silvera, C.G.; Costa, P.R. , et al. Human cd8+ t-cell responses against the 4 dengue virus serotypes are associated with distinct patterns of protein targets. The Journal of infectious diseases 2015, 212, 1743–1751. [Google Scholar] [CrossRef]
- Haltaufderhyde, K.; Srikiatkhachorn, A.; Green, S.; Macareo, L.; Park, S.; Kalayanarooj, S.; Rothman, A.L.; Mathew, A. Activation of peripheral t follicular helper cells during acute dengue virus infection. The Journal of infectious diseases 2018, 218, 1675–1685. [Google Scholar] [CrossRef]
- Sánchez-Vargas, L.A.; Mathew, A. Peripheral follicular helper t cells in acute viral diseases: A perspective on dengue. Future Virol 2019, 14, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Helmold Hait, S.; Vargas-Inchaustegui, D.A.; Musich, T.; Mohanram, V.; Tuero, I.; Venzon, D.J.; Bear, J.; Rosati, M.; Vaccari, M.; Franchini, G. , et al. Early t follicular helper cell responses and germinal center reactions are associated with viremia control in immunized rhesus macaques. Journal of virology 2019, 93. [Google Scholar] [CrossRef]
- Crotty, S. A brief history of t cell help to b cells. Nat Rev Immunol 2015, 15, 185–189. [Google Scholar] [CrossRef]
- Ma, C.S.; Deenick, E.K.; Batten, M.; Tangye, S.G. The origins, function, and regulation of t follicular helper cells. J Exp Med 2012, 209, 1241–1253. [Google Scholar] [CrossRef]
- Koh, B.; Ulrich, B.J.; Nelson, A.S.; Panangipalli, G.; Kharwadkar, R.; Wu, W.; Xie, M.M.; Fu, Y.; Turner, M.J.; Paczesny, S. , et al. Bcl6 and blimp1 reciprocally regulate st2(+) treg-cell development in the context of allergic airway inflammation. J Allergy Clin Immunol 2020, 146, 1121–1136.e1129. [Google Scholar] [CrossRef]
- Lu, Y.; Jiang, R.; Freyn, A.W.; Wang, J.; Strohmeier, S.; Lederer, K.; Locci, M.; Zhao, H.; Angeletti, D.; O'Connor, K.C. , et al. Cd4+ follicular regulatory t cells optimize the influenza virus-specific b cell response. J Exp Med 2021, 218. [Google Scholar] [CrossRef]
- Balakrishnan, T.; Bela-Ong, D.B.; Toh, Y.X.; Flamand, M.; Devi, S.; Koh, M.B.; Hibberd, M.L.; Ooi, E.E.; Low, J.G.; Leo, Y.S. , et al. Dengue virus activates polyreactive, natural igg b cells after primary and secondary infection. PLoS One 2011, 6, e29430. [Google Scholar] [CrossRef]
- Fink, K. Origin and function of circulating plasmablasts during acute viral infections. Front Immunol 2012, 3, 78. [Google Scholar] [CrossRef]
- Bernasconi, N.L.; Onai, N.; Lanzavecchia, A. A role for toll-like receptors in acquired immunity: Up-regulation of tlr9 by bcr triggering in naive b cells and constitutive expression in memory b cells. Blood 2003, 101, 4500–4504. [Google Scholar] [CrossRef] [PubMed]
- Pantoja, P.; Perez-Guzman, E.X.; Rodriguez, I.V.; White, L.J.; Gonzalez, O.; Serrano, C.; Giavedoni, L.; Hodara, V.; Cruz, L.; Arana, T. , et al. Zika virus pathogenesis in rhesus macaques is unaffected by pre-existing immunity to dengue virus. Nature communications 2017, 8, 15674. [Google Scholar] [CrossRef] [PubMed]
- Perez-Guzman, E.X.; Pantoja, P.; Serrano-Collazo, C.; Hassert, M.A.; Ortiz-Rosa, A.; Rodriguez, I.V.; Giavedoni, L.; Hodara, V.; Parodi, L.; Cruz, L. , et al. Time elapsed between zika and dengue virus infections affects antibody and t cell responses. Nature communications 2019, 10, 4316. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Collazo, C.; Pérez-Guzmán, E.X.; Pantoja, P.; Hassert, M.A.; Rodríguez, I.V.; Giavedoni, L.; Hodara, V.; Parodi, L.; Cruz, L.; Arana, T. , et al. Effective control of early zika virus replication by dengue immunity is associated to the length of time between the 2 infections but not mediated by antibodies. PLoS neglected tropical diseases 2020, 14, e0008285. [Google Scholar] [CrossRef]
- Marzan-Rivera, N.; Serrano-Collazo, C.; Cruz, L.; Pantoja, P.; Ortiz-Rosa, A.; Arana, T.; Martinez, M.I.; Burgos, A.G.; Roman, C.; Mendez, L.B. , et al. Infection order outweighs the role of cd4(+) t cells in tertiary flavivirus exposure. iScience 2022, 25, 104764. [Google Scholar] [CrossRef]
- Adams, C.; Jadi, R.; Segovia-Chumbez, B.; Daag, J.; Ylade, M.; Medina, F.A.; Sharp, T.M.; Munoz-Jordan, J.L.; Yoon, I.K.; Deen, J. , et al. Novel assay to measure seroprevalence of zika virus in the philippines. Emerg Infect Dis 2021, 27, 3073–3081. [Google Scholar] [CrossRef]
- Duong, V.; Ly, S.; Lorn Try, P.; Tuiskunen, A.; Ong, S.; Chroeung, N.; Lundkvist, A.; Leparc-Goffart, I.; Deubel, V.; Vong, S. , et al. Clinical and virological factors influencing the performance of a ns1 antigen-capture assay and potential use as a marker of dengue disease severity. PLoS neglected tropical diseases 2011, 5, e1244. [Google Scholar] [CrossRef]
- Chau, T.N.; Anders, K.L.; Lien le, B.; Hung, N.T.; Hieu, L.T.; Tuan, N.M.; Thuy, T.T.; Phuong le, T.; Tham, N.T.; Lanh, M.N. , et al. Clinical and virological features of dengue in vietnamese infants. PLoS neglected tropical diseases 2010, 4, e657. [Google Scholar]
- Avirutnan, P.; Punyadee, N.; Noisakran, S.; Komoltri, C.; Thiemmeca, S.; Auethavornanan, K.; Jairungsri, A.; Kanlaya, R.; Tangthawornchaikul, N.; Puttikhunt, C. , et al. Vascular leakage in severe dengue virus infections: A potential role for the nonstructural viral protein ns1 and complement. The Journal of infectious diseases 2006, 193, 1078–1088. [Google Scholar] [CrossRef]
- Perera, D.R.; Ranadeva, N.D.; Sirisena, K.; Wijesinghe, K.J. Roles of ns1 protein in flavivirus pathogenesis. ACS Infect Dis 2024, 10, 20–56. [Google Scholar] [CrossRef] [PubMed]
- Podestà, M.A.; Cavazzoni, C.B.; Hanson, B.L.; Bechu, E.D.; Ralli, G.; Clement, R.L.; Zhang, H.; Chandrakar, P.; Lee, J.M.; Reyes-Robles, T. , et al. Stepwise differentiation of follicular helper t cells reveals distinct developmental and functional states. Nature communications 2023, 14, 7712. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Moutaftsi, M.; Moyron-Quiroz, J.; McCausland, M.M.; Davies, D.H.; Johnston, R.J.; Peters, B.; Rafii-El-Idrissi Benhnia, M.; Hoffmann, J.; Su, H.P. , et al. Selective cd4+ t cell help for antibody responses to a large viral pathogen: Deterministic linkage of specificities. Immunity 2008, 28, 847–858. [Google Scholar] [CrossRef]
- Appanna, R.; Kg, S.; Xu, M.H.; Toh, Y.X.; Velumani, S.; Carbajo, D.; Lee, C.Y.; Zuest, R.; Balakrishnan, T.; Xu, W. , et al. Plasmablasts during acute dengue infection represent a small subset of a broader virus-specific memory b cell pool. EBioMedicine 2016, 12, 178–188. [Google Scholar] [CrossRef]
- Duangchinda, T.; Dejnirattisai, W.; Vasanawathana, S.; Limpitikul, W.; Tangthawornchaikul, N.; Malasit, P.; Mongkolsapaya, J.; Screaton, G. Immunodominant t-cell responses to dengue virus ns3 are associated with dhf. Proc Natl Acad Sci U S A 2010, 107, 16922–16927. [Google Scholar] [CrossRef]
- Rouers, A.; Chng, M.H.Y.; Lee, B.; Rajapakse, M.P.; Kaur, K.; Toh, Y.X.; Sathiakumar, D.; Loy, T.; Thein, T.L.; Lim, V.W.X. , et al. Immune cell phenotypes associated with disease severity and long-term neutralizing antibody titers after natural dengue virus infection. Cell Rep Med 2021, 2, 100278. [Google Scholar] [CrossRef]
- Rivino, L.; Kumaran, E.A.; Jovanovic, V.; Nadua, K.; Teo, E.W.; Pang, S.W.; Teo, G.H.; Gan, V.C.; Lye, D.C.; Leo, Y.S. , et al. Differential targeting of viral components by cd4+ versus cd8+ t lymphocytes in dengue virus infection. Journal of virology 2013, 87, 2693–2706. [Google Scholar] [CrossRef] [PubMed]
- Mangada, M.M.; Rothman, A.L. Altered cytokine responses of dengue-specific cd4+ t cells to heterologous serotypes. J Immunol 2005, 175, 2676–2683. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.D.; Wang, H.; da Silva Antunes, R.; Tian, Y.; Tippalagama, R.; Alahakoon, S.U.; Premawansa, G.; Wijewickrama, A.; Premawansa, S.; De Silva, A.D. , et al. A population of cd4(+)cd8(+) double-positive t cells associated with risk of plasma leakage in dengue viral infection. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.; Eisenhauer, P.; Diehl, S.A.; Pierce, K.K.; Whitehead, S.S.; Durbin, A.P.; Kirkpatrick, B.D.; Sette, A.; Weiskopf, D.; Boyson, J.E. , et al. Rapid induction and maintenance of virus-specific cd8(+) t(emra) and cd4(+) t(em) cells following protective vaccination against dengue virus challenge in humans. Front Immunol 2020, 11, 479. [Google Scholar] [CrossRef] [PubMed]
- Weiskopf, D.; Bangs, D.J.; Sidney, J.; Kolla, R.V.; De Silva, A.D.; de Silva, A.M.; Crotty, S.; Peters, B.; Sette, A. Dengue virus infection elicits highly polarized cx3cr1+ cytotoxic cd4+ t cells associated with protective immunity. Proc Natl Acad Sci U S A 2015, 112, E4256–4263. [Google Scholar] [CrossRef]
- Elong Ngono, A.; Young, M.P.; Bunz, M.; Xu, Z.; Hattakam, S.; Vizcarra, E.; Regla-Nava, J.A.; Tang, W.W.; Yamabhai, M.; Wen, J. , et al. Cd4+ t cells promote humoral immunity and viral control during zika virus infection. PLoS Pathog 2019, 15, e1007474. [Google Scholar]
- Henriques, H.R.; Rampazo, E.V.; Gonçalves, A.J.; Vicentin, E.C.; Amorim, J.H.; Panatieri, R.H.; Amorim, K.N.; Yamamoto, M.M.; Ferreira, L.C.; Alves, A.M. , et al. Targeting the non-structural protein 1 from dengue virus to a dendritic cell population confers protective immunity to lethal virus challenge. PLoS neglected tropical diseases 2013, 7, e2330. [Google Scholar] [CrossRef]
- Pinto, P.B.A.; Assis, M.L.; Vallochi, A.L.; Pacheco, A.R.; Lima, L.M.; Quaresma, K.R.L.; Pereira, B.A.S.; Costa, S.M.; Alves, A.M.B. T cell responses induced by DNA vaccines based on the denv2 e and ns1 proteins in mice: Importance in protection and immunodominant epitope identification. Front Immunol 2019, 10, 1522. [Google Scholar] [CrossRef]
- Goncalves, A.J.; Oliveira, E.R.; Costa, S.M.; Paes, M.V.; Silva, J.F.; Azevedo, A.S.; Mantuano-Barradas, M.; Nogueira, A.C.; Almeida, C.J.; Alves, A.M. Cooperation between cd4+ t cells and humoral immunity is critical for protection against dengue using a DNA vaccine based on the ns1 antigen. PLoS neglected tropical diseases 2015, 9, e0004277. [Google Scholar] [CrossRef]
- Dias, A.G., Jr.; Atyeo, C.; Loos, C.; Montoya, M.; Roy, V.; Bos, S.; Narvekar, P.; Singh, T.; Katzelnick, L.C.; Kuan, G. , et al. Antibody fc characteristics and effector functions correlate with protection from symptomatic dengue virus type 3 infection. Sci Transl Med 2022, 14, eabm3151. [Google Scholar] [CrossRef]
- Sanchez-Vargas, L.A.; Mathew, A.; Salje, H.; Sousa, D.; Casale, N.A.; Farmer, A.; Buddhari, D.; Anderson, K.; Iamsirithaworn, S.; Kaewhiran, S. , et al. Protective role of ns1-specific antibodies in the immune response to dengue virus through antibody-dependent cellular cytotoxicity. The Journal of infectious diseases 2024, 230, 1147–1156. [Google Scholar] [CrossRef]
- Nivarthi, U.K.; Kose, N.; Sapparapu, G.; Widman, D.; Gallichotte, E.; Pfaff, J.M.; Doranz, B.J.; Weiskopf, D.; Sette, A.; Durbin, A.P. , et al. Mapping the human memory b cell and serum neutralizing antibody responses to dengue virus serotype 4 infection and vaccination. Journal of virology 2017, 91. [Google Scholar] [CrossRef]
- Priyamvada, L.; Cho, A.; Onlamoon, N.; Zheng, N.Y.; Huang, M.; Kovalenkov, Y.; Chokephaibulkit, K.; Angkasekwinai, N.; Pattanapanyasat, K.; Ahmed, R. , et al. B cell responses during secondary dengue virus infection are dominated by highly cross-reactive, memory-derived plasmablasts. Journal of virology 2016, 90, 5574–5585. [Google Scholar] [CrossRef]
- Bos, S.; Graber, A.L.; Cardona-Ospina, J.A.; Duarte, E.M.; Zambrana, J.V.; Ruíz Salinas, J.A.; Mercado-Hernandez, R.; Singh, T.; Katzelnick, L.C.; de Silva, A. , et al. Protection against symptomatic dengue infection by neutralizing antibodies varies by infection history and infecting serotype. Nature communications 2024, 15, 382. [Google Scholar] [CrossRef]
- Katzelnick, L.C.; Bos, S.; Harris, E. Protective and enhancing interactions among dengue viruses 1-4 and zika virus. Curr Opin Virol 2020, 43, 59–70. [Google Scholar] [CrossRef]
- Zambrana, J.V.; Hasund, C.M.; Aogo, R.A.; Bos, S.; Arguello, S.; Gonzalez, K.; Collado, D.; Miranda, T.; Kuan, G.; Gordon, A. , et al. Primary exposure to zika virus is linked with increased risk of symptomatic dengue virus infection with serotypes 2, 3, and 4, but not 1. Sci Transl Med 2024, 16, eadn2199. [Google Scholar] [CrossRef]
- Crooks, C.M.; Weiler, A.M.; Rybarczyk, S.L.; Bliss, M.I.; Jaeger, A.S.; Murphy, M.E.; Simmons, H.A.; Mejia, A.; Fritsch, M.K.; Hayes, J.M. , et al. Previous exposure to dengue virus is associated with increased zika virus burden at the maternal-fetal interface in rhesus macaques. PLoS neglected tropical diseases 2021, 15, e0009641. [Google Scholar] [CrossRef] [PubMed]
- Elsner, R.A.; Shlomchik, M.J. Coordinated regulation of extrafollicular b cell responses by il-12 and ifnγ. Immunological reviews 2025, 331, e70027. [Google Scholar] [CrossRef]
- Correa, A.R.; Berbel, A.C.; Papa, M.P.; Morais, A.T.; Peçanha, L.M.; Arruda, L.B. Dengue virus directly stimulates polyclonal b cell activation. PLoS One 2015, 10, e0143391. [Google Scholar] [CrossRef] [PubMed]
- Ruffin, N.; Thang, P.H.; Rethi, B.; Nilsson, A.; Chiodi, F. The impact of inflammation and immune activation on b cell differentiation during hiv-1 infection. Front Immunol 2011, 2, 90. [Google Scholar] [CrossRef]
- Kwissa, M.; Nakaya, H.I.; Onlamoon, N.; Wrammert, J.; Villinger, F.; Perng, G.C.; Yoksan, S.; Pattanapanyasat, K.; Chokephaibulkit, K.; Ahmed, R. , et al. Dengue virus infection induces expansion of a cd14(+)cd16(+) monocyte population that stimulates plasmablast differentiation. Cell Host Microbe 2014, 16, 115–127. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Qiao, X.; Klasse, P.J.; Chiu, A.; Chadburn, A.; Knowles, D.M.; Moore, J.P.; Cerutti, A. Hiv-1 envelope triggers polyclonal ig class switch recombination through a cd40-independent mechanism involving baff and c-type lectin receptors. J Immunol 2006, 176, 3931–3941. [Google Scholar] [CrossRef] [PubMed]
- Provine, N.M.; Badamchi-Zadeh, A.; Bricault, C.A.; Penaloza-MacMaster, P.; Larocca, R.A.; Borducchi, E.N.; Seaman, M.S.; Barouch, D.H. Transient cd4+ t cell depletion results in delayed development of functional vaccine-elicited antibody responses. Journal of virology 2016, 90, 4278–4288. [Google Scholar] [CrossRef] [PubMed]
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




