Submitted:
08 August 2025
Posted:
11 August 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Tissue Preparation and Immunohistochemistry
2.3. Fluorescence Detection and Image Processing
2.4. Multielectrode array recordings
3. Results
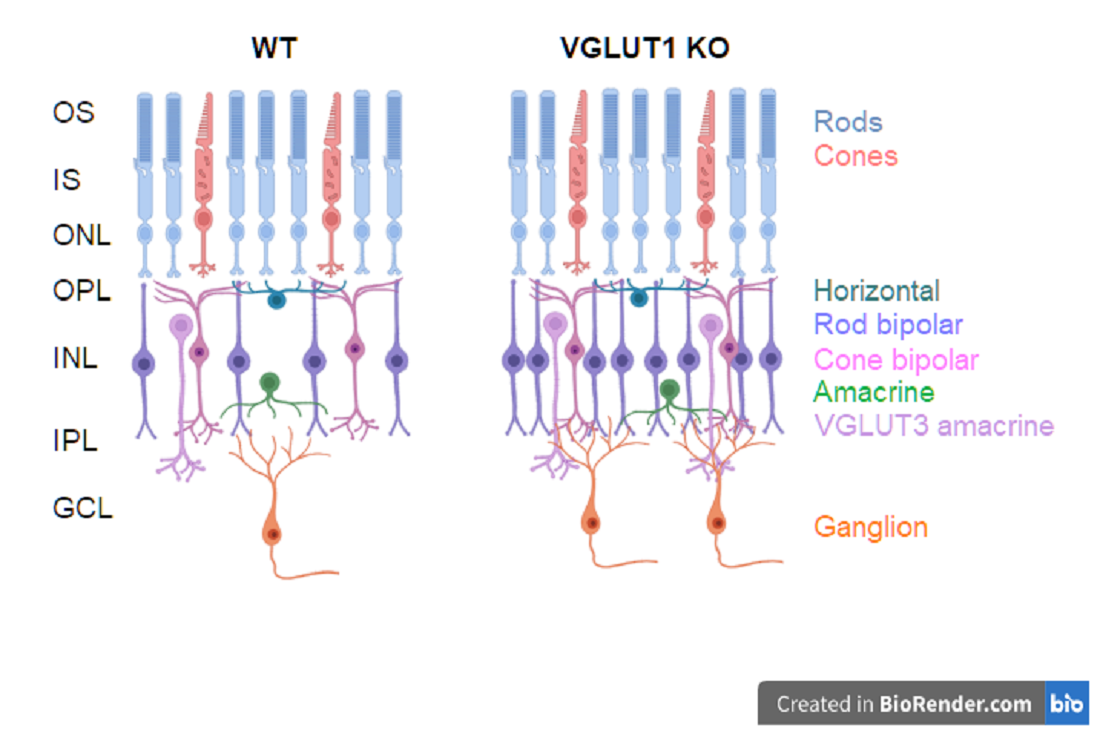
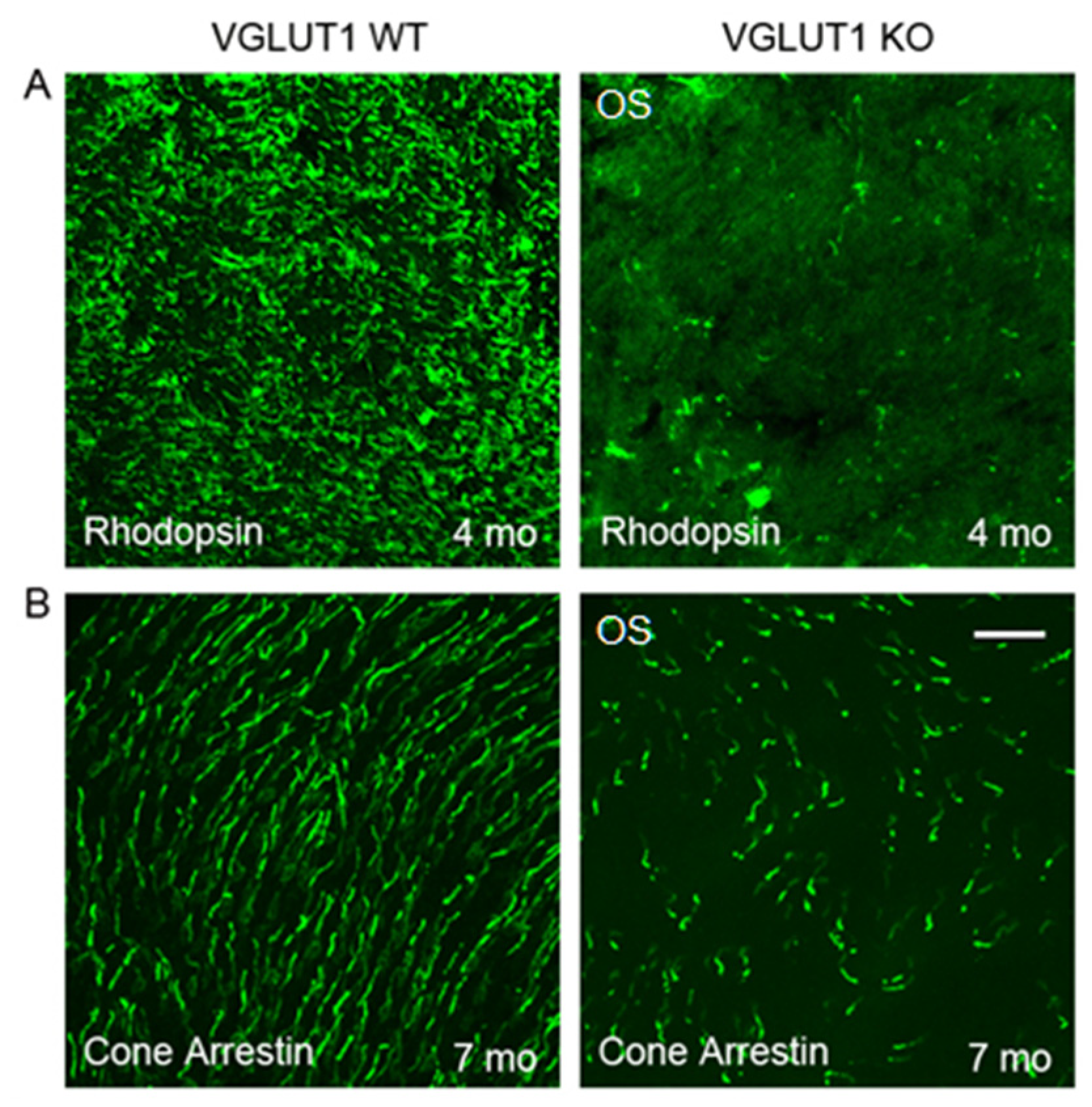
3.1. VGLUT1 Null Retina Exhibits Loss of Function Induced Progressive Loss of Photoreceptors Similar to Mouse Models of Retinitis Pigmentosa
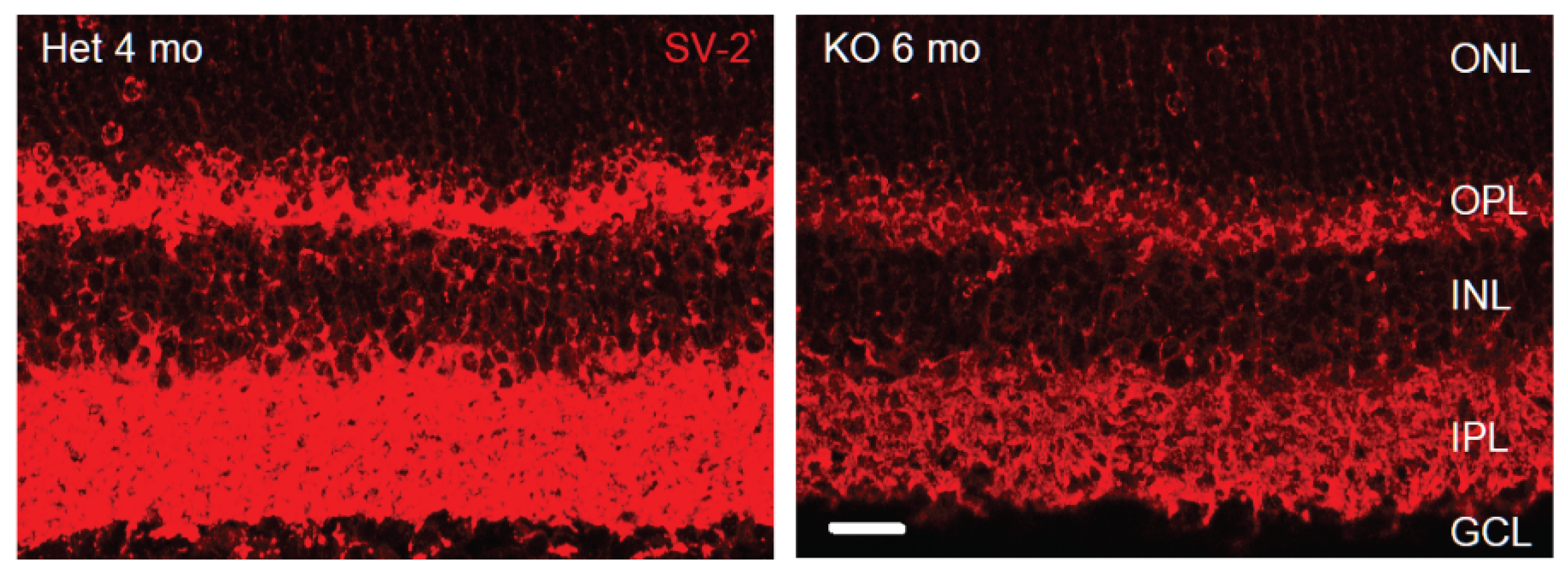
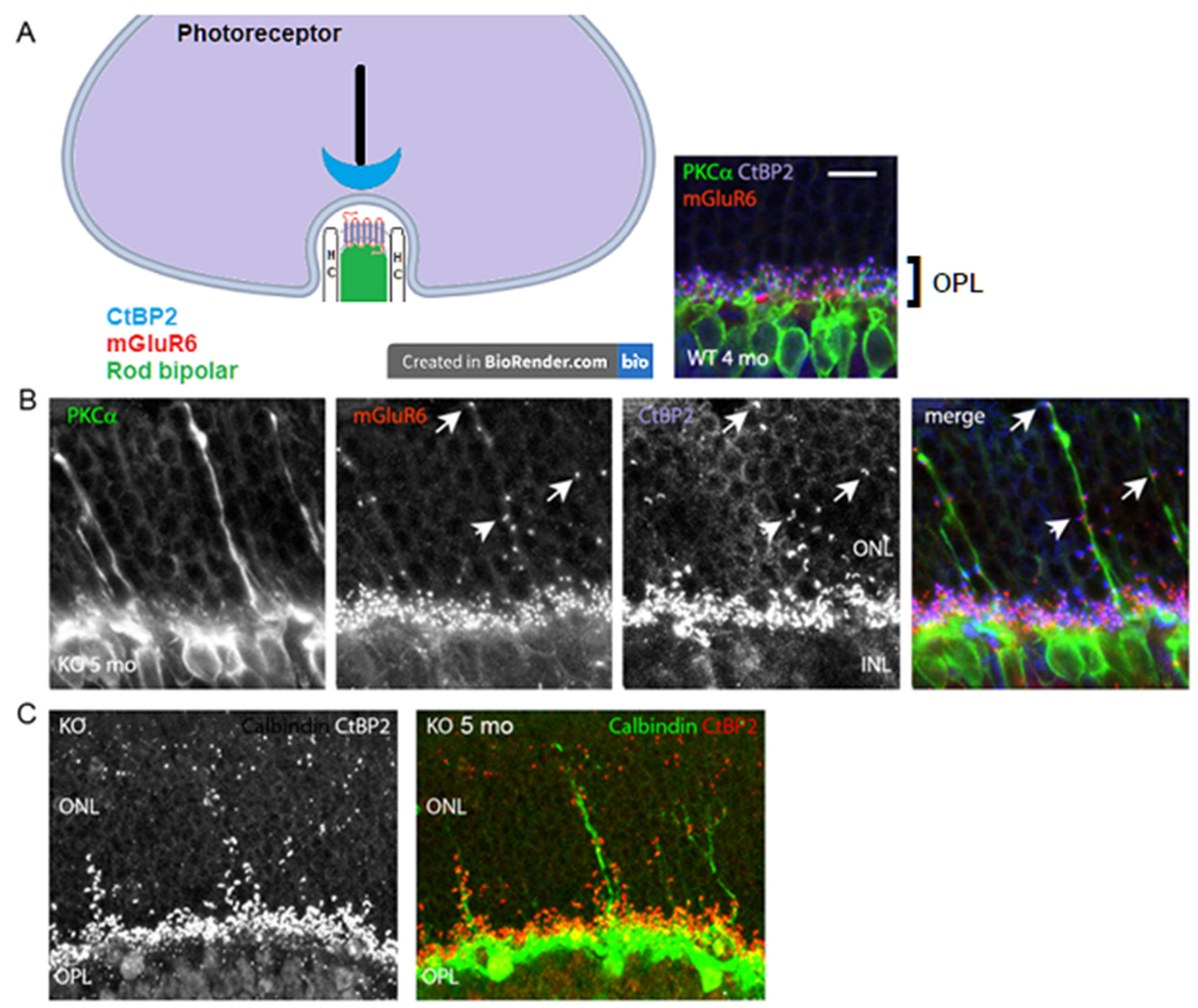
3.2. Loss of Glutamatergic Neurotransmission in the OPL Triggers Synaptic Disorganization and Ectopic Synaptic Formations in the ONL
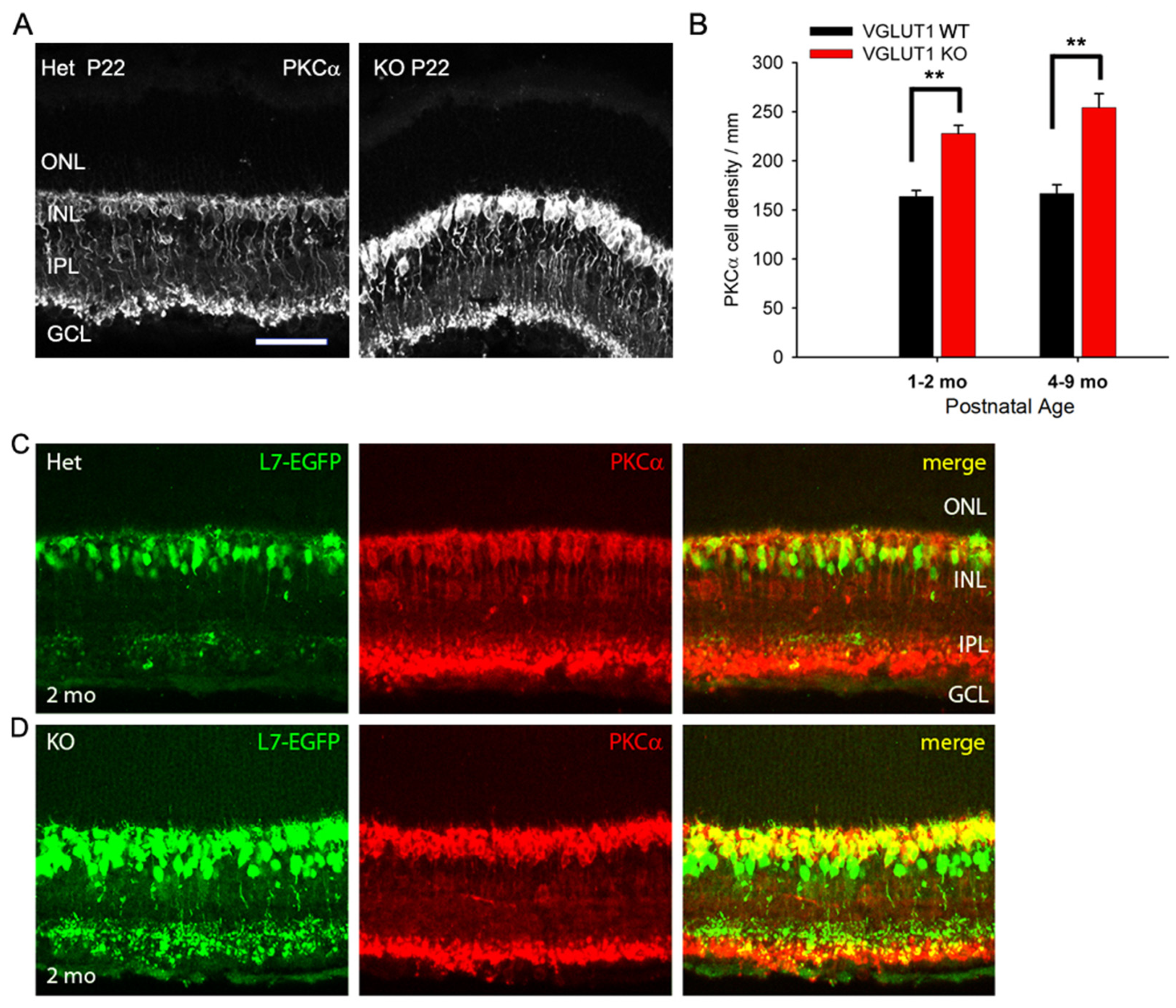
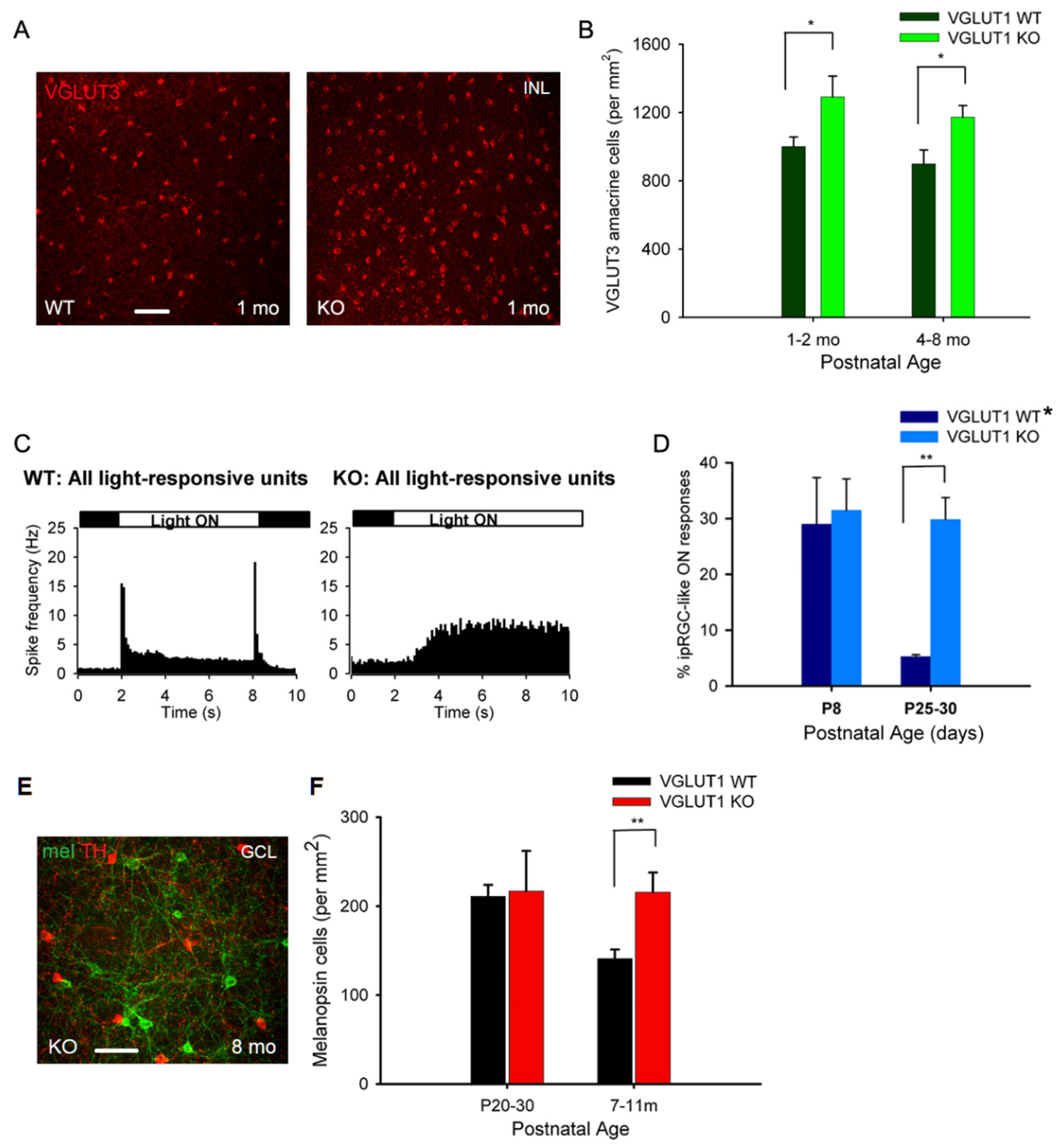
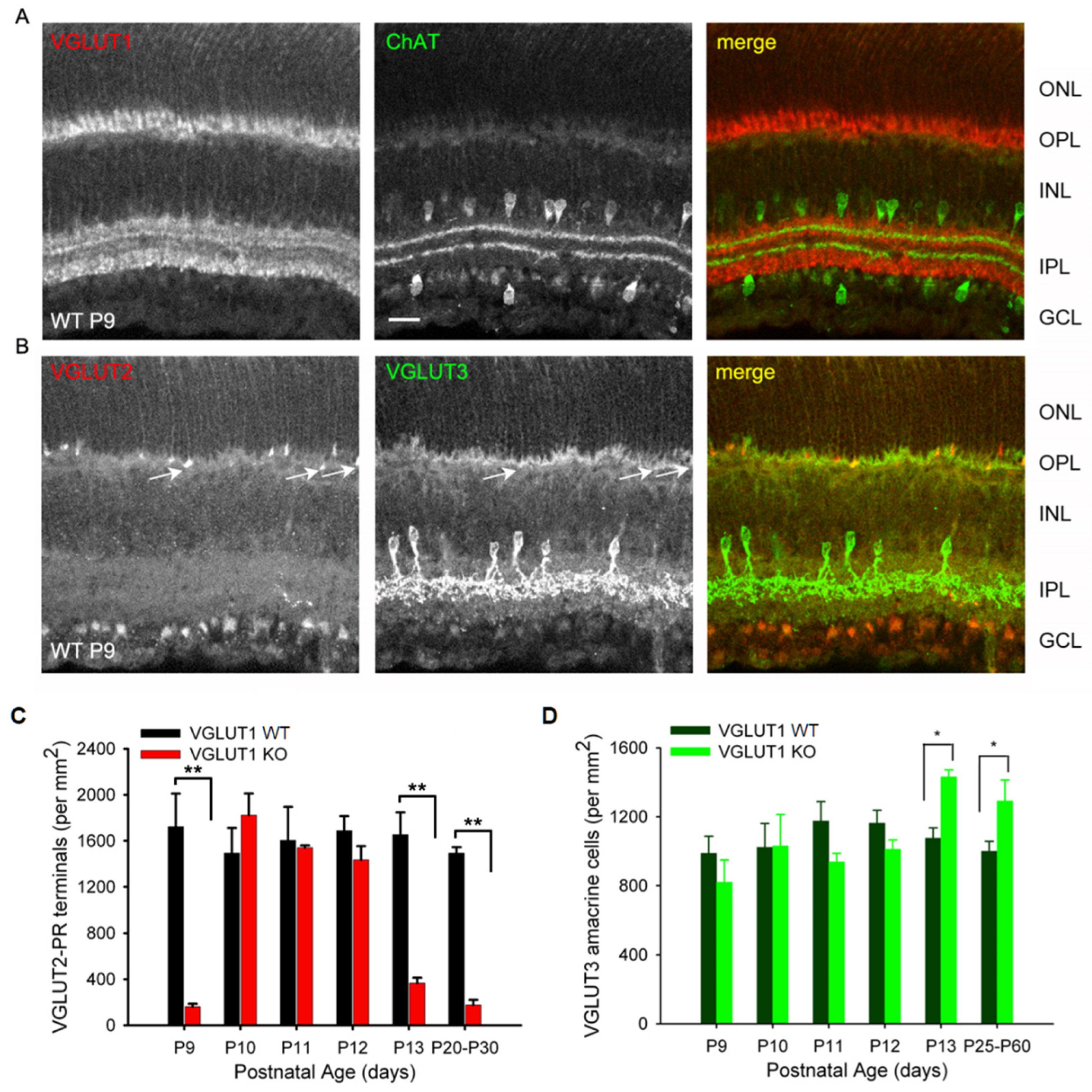
3.3. In Contrast to Progressive Decrease in Photoreceptor Densities, Bipolar Cells, VGLUT3 Amacrine Cells and Melanopsin ipRGCs Have Higher Densities in Juvenile, Adult and Aged VGLUT1 Null Retina
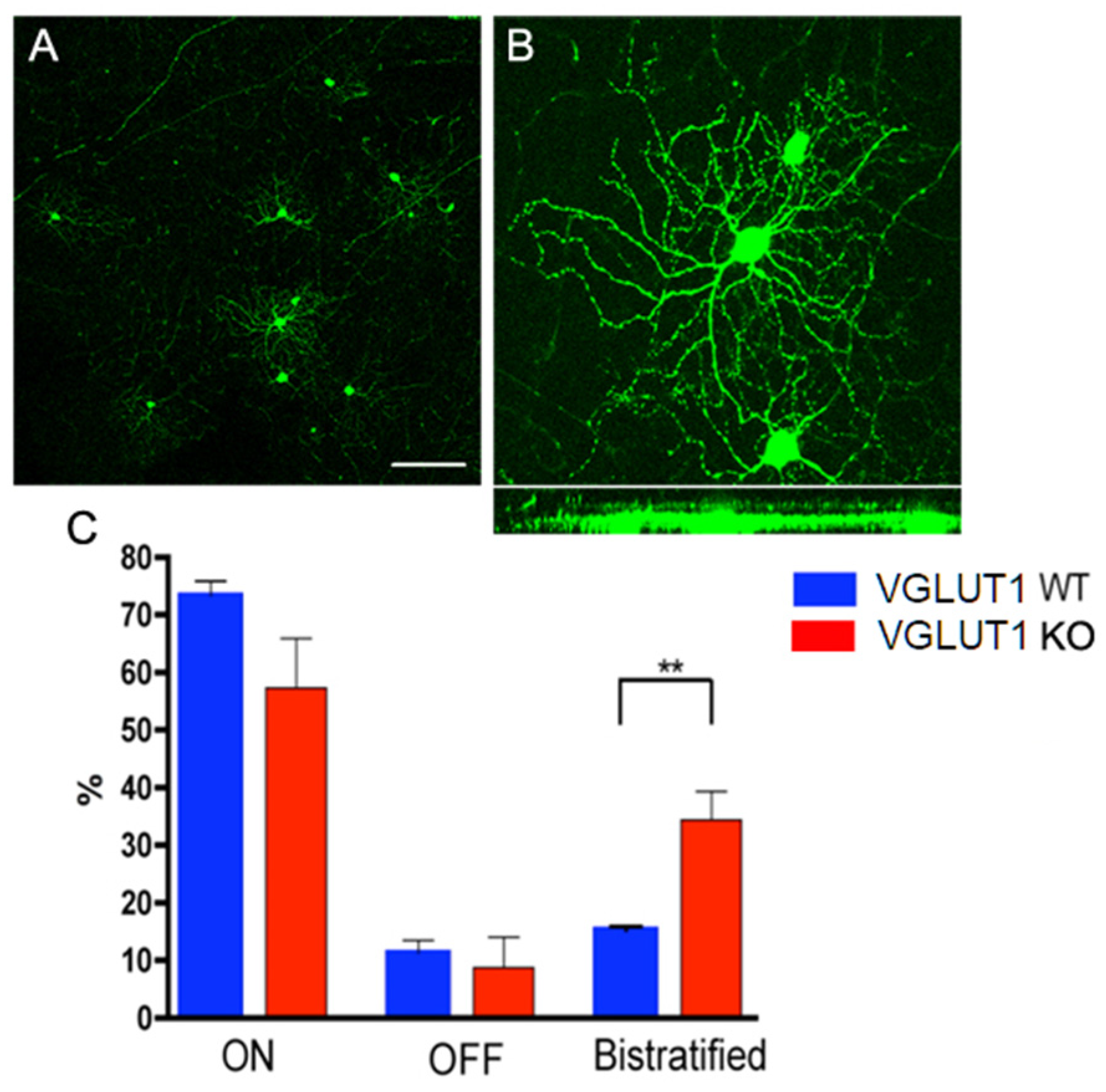
3.4. Postnatal Refinement of RGC Dendritic Arbors is Delayed in VGLUT1 Null Mice.
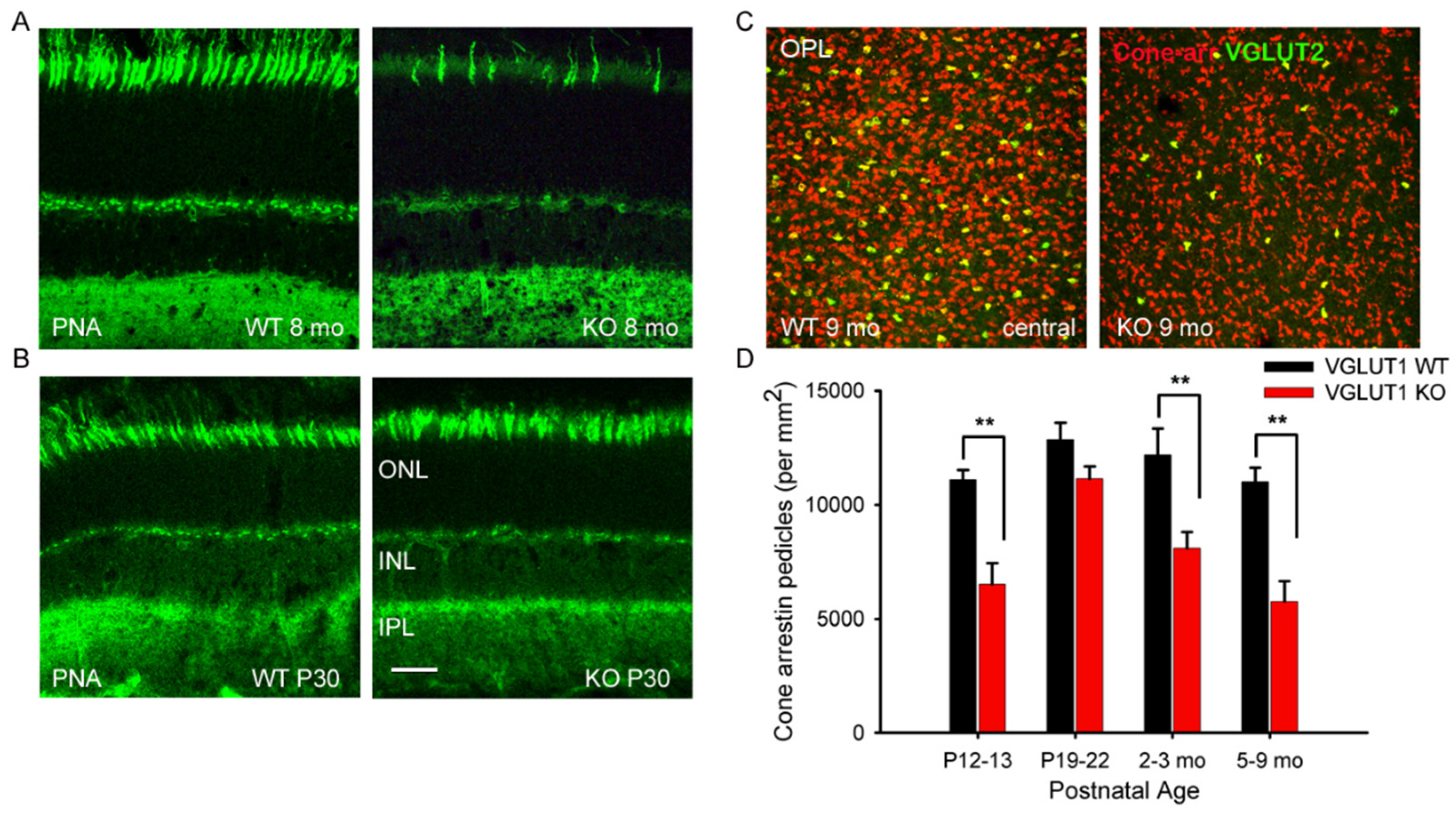
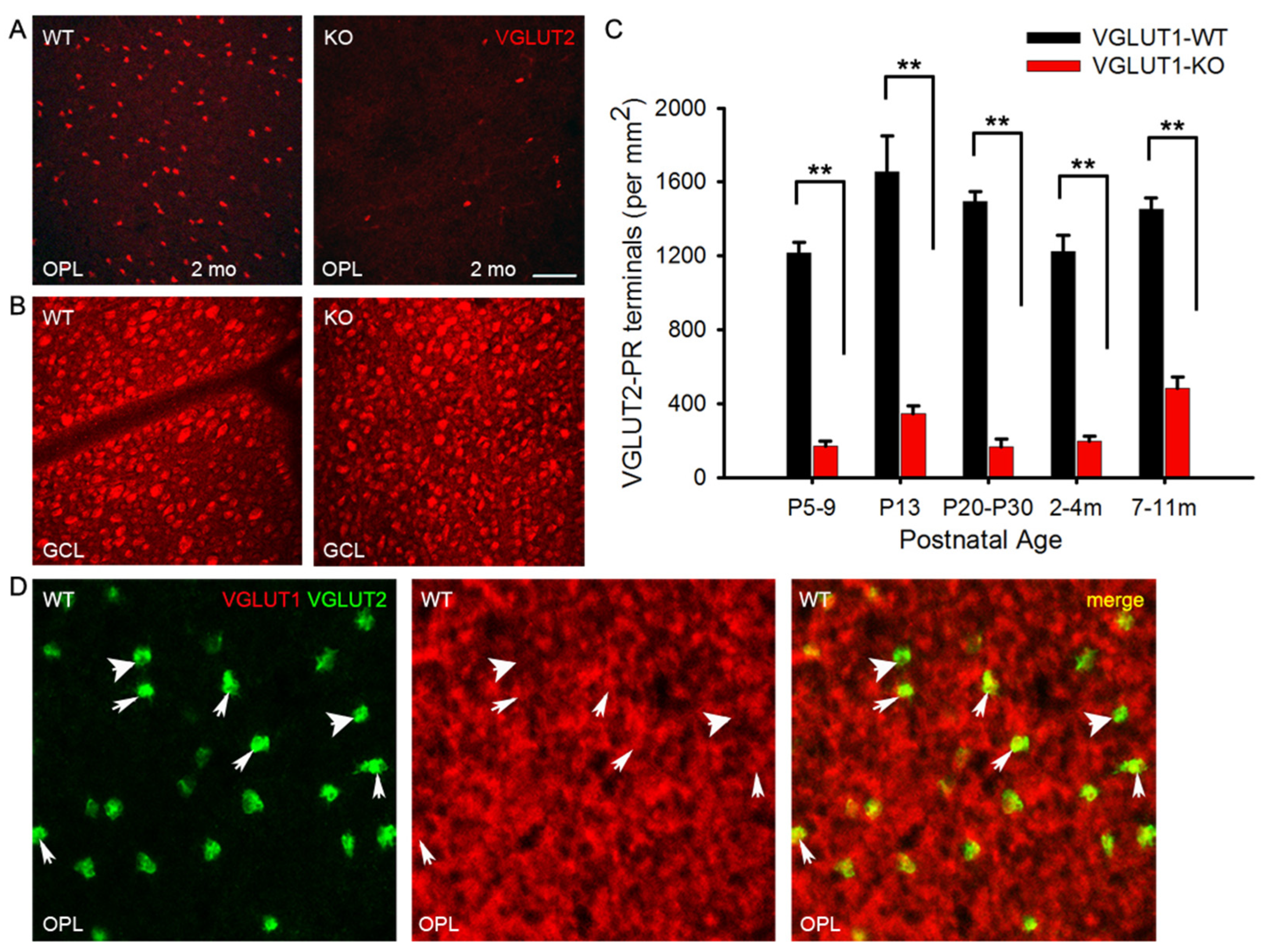
3.5. VGLUT2 Positive Cones are Fewer at all Ages in the VGLUT1 Null Retina
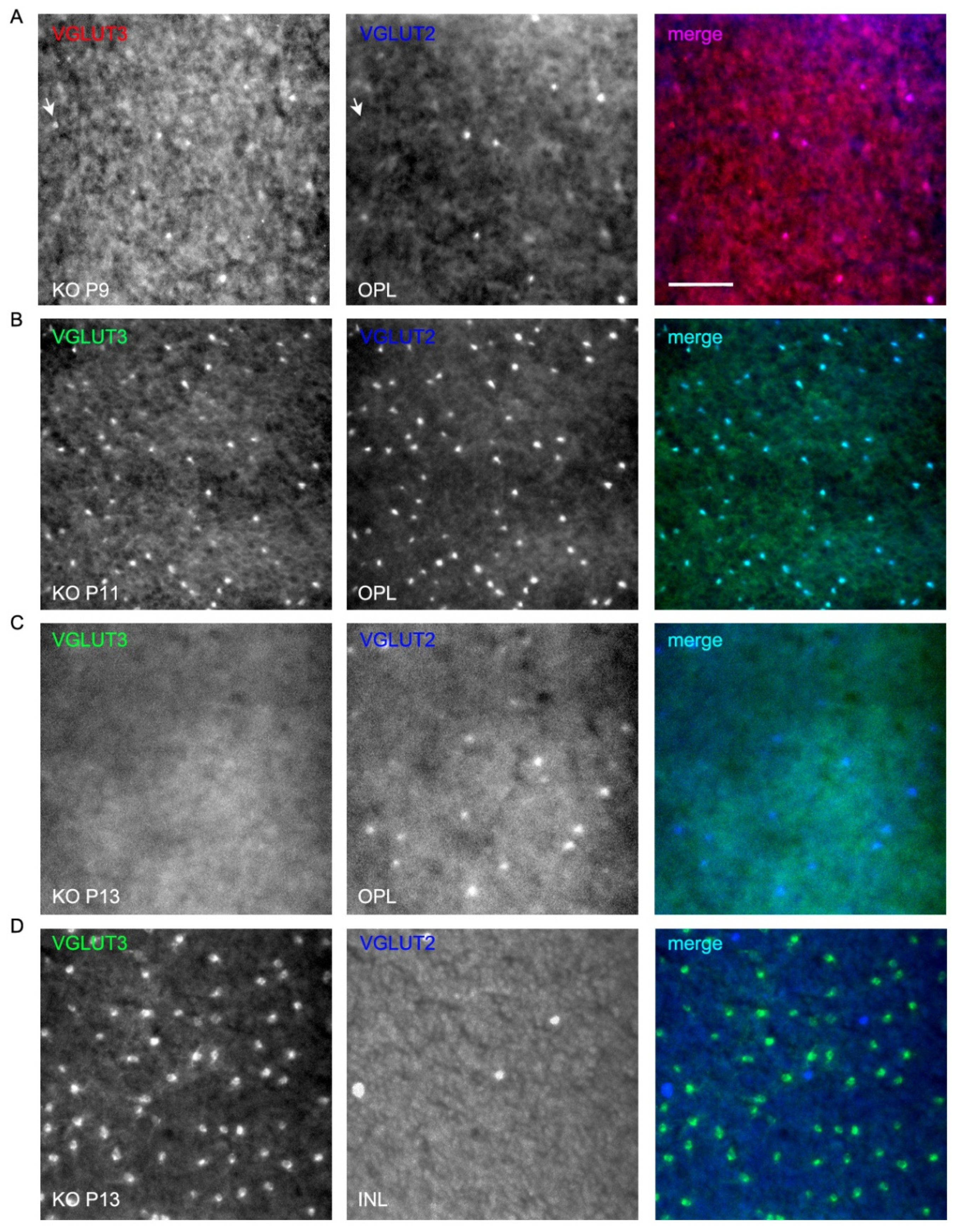
3.6. VGLUT2 and VGLUT3 Expression Change Dynamically During the First 2 Weeks of Postnatal Development
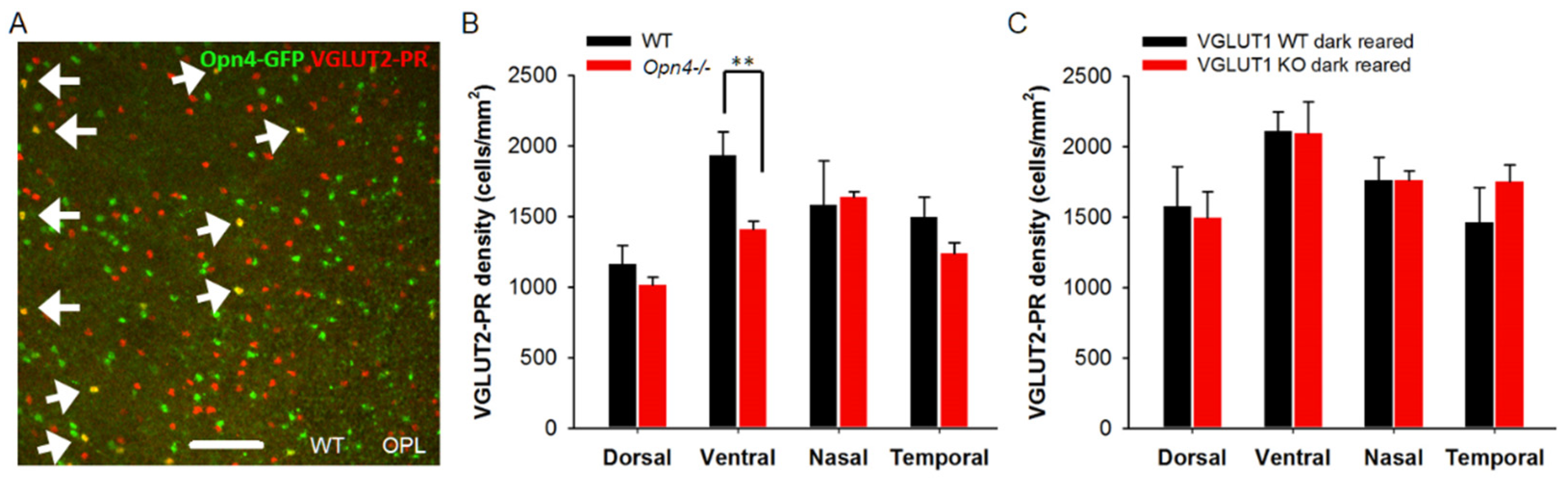
3.7. Light Activation of ipRGCs Suppresses VGLUT2 Expression in Cone Pedicles of VGLUT1 Null Retina
4. Discussion
4.1. Age Related Photoreceptor Degeneration and Increase in Histogenesis of Inner Retinal Glutamatergic Neurons in VGLUT1 Null Mice
4.2. Dynamic Expression of VGLUT2 in Some Cones in VGLUT1 Null Retina is Governed by Transient Expression of VGLUT3 in These Cones and Can Be Influenced by Melanopsin ipRGCs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Majumdar, S. Role of glutamate in the development of visual pathways. Front in Ophthanlmology 2023, 3, 1–5. [Google Scholar] [CrossRef]
- Ichinose, T.; Habib, S. ON and OFF Signaling Pathways in the Retina and the Visual System. Front Ophthalmol (Lausanne) 2022, 2, 1–14. [Google Scholar] [CrossRef]
- Martersteck, E.M.; Hirokawa, K.E.; Evarts, M.; Bernard, A.; Duan, X.; Li, Y.; Ng, L.; Oh, S.W.; Ouellette, B.; Royall, J.J.; Stoecklin, M.; Wang, Q.; Zeng, H.; Sanes, J.R.; Harris, J.A. Diverse Central Projection Patterns of Retinal Ganglion Cells. Cell Rep 2017, 2017 18, 2058–2072. [Google Scholar] [CrossRef]
- Central Projections of Retinal Ganglion Cells. Neuroscience, 2nd ed.; Purves, D., Augustine, G.J., Fitzpatrick, D., Katz, L.C., LaMantia, A.S., McNamara, J.O., Williams, S.M., Eds.; Sunderland (MA): Sinauer Associates, UK, 2001. [Google Scholar]
- Sherry, D.M.; Wang, M.M.; Bates, J.; Frishman, L.J. Expression of vesicular glutamate transporter 1 in the mouse retina reveals temporal ordering in development of rod vs. cone and ON vs. OFF circuits. J Comp Neurol 2003, 465, 480–498. [Google Scholar] [CrossRef] [PubMed]
- Fremeau, R.T. Jr.; Kam, K.; Qureshi, T.; Johnson, J.; Copenhagen, D.R.; Storm-Mathisen, J.; Chaudhry, F.A.; Nicoll, R.A.; Edwards, R.H. Vesicular glutamate transporters 1 and 2 target to functionally distinct synaptic release sites. Science 2004, 304, 1815–9. [Google Scholar] [CrossRef]
- Wässle, H.; Regus-Leidig, H.; Haverkamp, S. Expression of the vesicular glutamate transporter vGluT2 in a subset of cones of the mouse retina. J Comp Neurol 2006, 496, 544–55. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Sherry, D.M.; Liu, X.; Fremeau, R.T. Jr.; Seal, R.P.; Edwards, R.H.; Copenhagen, D.R. Vesicular glutamate transporter 3 expression identifies glutamatergic amacrine cells in the rodent retina. J Comp Neurol 2004, 477, 386–398. [Google Scholar] [CrossRef]
- Fremeau, R.T. Jr.; Voglmaier, S.; Seal, R.P.; Edwards, R.H. VGLUTs define subsets of excitatory neurons and suggest novel roles for glutamate. Trends Neurosci 2004, 27, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Wallén-Mackenzie, A.; Wootz, H.; Englund, H. Genetic inactivation of the vesicular glutamate transporter 2 (VGLUT2) in the mouse: what have we learnt about functional glutamatergic neurotransmission? Ups J Med Sci 2010, 115, 11–20. [Google Scholar] [CrossRef]
- Seal, R.P.; Akil, O.; Yi, E.; Weber, C.M.; Grant, L.; Yoo, J.; Clause, A.; Kandler, K.; Noebels, J.L.; Glowatzki, E.; Lustig, L.R.; Edwards, R.H. ; Sensorineural deafness and seizures in mice lacking vesicular glutamate transporter 3. Neuron 2008, 2008 57, 263–75. [Google Scholar] [CrossRef]
- Johnson, J.; Fremeau, R.T. Jr.; Duncan, J.L.; Rentería, R.C.; Yang, H.; Hua, Z.; Liu, X.; LaVail, M.M.; Edwards, R.H.; Copenhagen, D.R. Vesicular glutamate transporter 1 is required for photoreceptor synaptic signaling but not for intrinsic visual functions. J Neurosci 2007, 2007 27, 7245–7255. [Google Scholar] [CrossRef]
- Johnson, J.; Tian, N.; Caywood, M.S.; Reimer, R,J. ; Edwards, R.H.; Copenhagen, D.R. Vesicular neurotransmitter transporter expression in developing postnatal rodent retina: GABA and glycine precede glutamate. J Neurosci 2003, 23, 518–29. [Google Scholar] [CrossRef]
- Boulland, J.L.; Qureshi, T.; Seal, R.P.; Rafiki, A.; Gundersen, V.; Bergersen, L.H.; Fremeau, R.T. Jr.; Edwards, R.H.; Storm-Mathisen, J.; Chaudhry, F.A. Expression of the vesicular glutamate transporters during development indicates the widespread corelease of multiple neurotransmitters. J Comp Neurol 2004, 480, 264–80. [Google Scholar] [CrossRef]
- Nakakubo, Y.; Abe, S.; Yoshida, T.; Takami, C.; Isa, M.; Wojcik, S.M.; Brose, N.; Takamori, S.; Hori, T. Vesicular Glutamate Transporter Expression Ensures High-Fidelity Synaptic Transmission at the Calyx of Held Synapses. Cell Rep 2020, 32, 108040. [Google Scholar] [CrossRef]
- Cepko, C. Intrinsically different retinal progenitor cells produce specific types of progeny. Nat Rev Neurosci 2014, 15, 615–27. [Google Scholar] [CrossRef]
- Duarte-Silva, A.T.; Ximenes, L.G.R.; Guimarães-Souza, M.; Domith, I.; Paes-de-Carvalho, R. Chemical signaling in the developing avian retina: Focus on cyclic AMP and AKT- dependent pathways. Front Cell Dev Biol 2022, 10, 1058925. [Google Scholar] [CrossRef] [PubMed]
- Delwig, A.; Majumdar, S.; Ahern, K.; LaVail, M.M.; Edwards, R.; Hnasko, T.S.; Copenhagen, D.R. Glutamatergic neurotransmission from melanopsin retinal ganglion cells is required for neonatal photoaversion but not adult pupillary light reflex. PLoS One 2013, 8, e83974. [Google Scholar] [CrossRef]
- Verweij, J.; Chaney, S.Y.; Bredl, D.; Vemaraju, S.; König, G.M.; Kostenis, E.; Lang, R.A.; Copenhagen, D.R. Light responses of melanopsin-expressing ganglion cells in the foetal mammalian retina. bioRxiv 2019, 675702. [Google Scholar] [CrossRef]
- Blankenship, A.G.; Ford, K.J.; Johnson, J.; Seal, R.P.; Edwards, R.H.; Copenhagen, D.R.; Feller, M.B. Synaptic and extrasynaptic factors governing glutamatergic retinal waves. Neuron 2009, 62, 230–41. [Google Scholar] [CrossRef]
- Biswas, S.; Shahriar, S.; Bachay, G.; Arvanitis, P.; Jamoul, D.; Brunken, W.J.; Agalliu, D. Glutamatergic neuronal activity regulates angiogenesis and blood-retinal barrier maturation via Norrin/β-catenin signaling. Neuron 2024, 112, 1978–1996. [Google Scholar] [CrossRef] [PubMed]
- Tomomura, M.; Rice, D.S.; Morgan, J.I.; Yuzaki, M. Purification of Purkinje cells by fluorescence-activated cell sorting from transgenic mice that express green fluorescent protein. Eur J Neurosci 2001, 14, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Wong, H.; Walsh, J.H.; Brecha, N.C. Expression of the somatostatin subtype 2A receptor in the rabbit retina. J Comp Neurol 1998, 393, 93–101. [Google Scholar] [CrossRef]
- Haverkamp, S.; Wassle, H. Immunocytochemical analysis of the mouse retina. J Comp Neurol 2000, 424, 1–23. [Google Scholar] [CrossRef]
- Buckley, K.; Kelly, R.B. Identification of a transmembrane glycoprotein specific for secretory vesicles of neural and endocrine cells. J Cell Biol 1985, 100, 1284–1294. [Google Scholar] [CrossRef]
- Montana, V.; Ni, Y.; Sunjara, V.; Hua, X.; Parpura, V. Vesicular glutamate transporter- dependent glutamate release from astrocytes. J Neurosci 2004, 24, 2633–2642. [Google Scholar] [CrossRef] [PubMed]
- tom Dieck, S.; Altrock, W,D. ; Kessels, M.M.; Qualmann, B.; Regus, H.; Brauner, D.; Fejtová, A.; Bracko, O.; Gundelfinger, E.D.; Brandstätter, J.H. Molecular dissection of the photoreceptor ribbon synapse: physical interaction of Bassoon and RIBEYE is essential for the assembly of the ribbon complex. J Cell Biol 2005, 168, 825–836. [Google Scholar]
- Wässle, H.; Puller, C.; Müller, F.; Haverkamp, S. Cone contacts, mosaics, and territories of bipolar cells in the mouse retina. J Neurosci 2009, 29, 106–117. [Google Scholar] [CrossRef]
- Sulaiman, P.; Fina, M.; Feddersen, R.; Vardi, N. Ret-PCP2 colocalizes with protein kinase C in a subset of primate ON cone bipolar cells. J Comp Neurol 2010, 518, 1098–1112. [Google Scholar] [CrossRef]
- Specht, D.; Wu, S.B.; Turner, P.; Dearden, P.; Koentgen, F.; Wolfrum, U.; Maw, M.; Brandstätter, J.H.; tom Dieck, S. Effects of presynaptic mutations on a postsynaptic Cacna1s calcium channel colocalized with mGluR6 at mouse photoreceptor ribbon synapses. Invest Ophthalmol Vis Sci 2009, 50, 505–515. [Google Scholar] [CrossRef]
- Barone, I.; Novelli, E.; Strettoi, E. Long-term preservation of cone photoreceptors and visual acuity in rd10 mutant mice exposed to continuous environmental enrichment. Mol Vis 2014, 20, 1545–56. [Google Scholar]
- Shiromani, P.J.; Floyd, C.; Velázquez-Moctezuma, J. Pontine cholinergic neurons simultaneously innervate two thalamic targets. Brain Res 1990, 532, 317–322. [Google Scholar] [CrossRef]
- Berson, D.M.; Castrucci, A.M.; Provencio, I. Morphology and mosaics of melanopsin- expressing retinal ganglion cell types in mice. J Comp Neurol 2010, 518, 2405–2422. [Google Scholar] [CrossRef]
- Johnson, J.; Wu, V.; Donovan, M.; Majumdar, S.; Rentería, R.C.; Porco, T.; Van Gelder, R.N.; Copenhagen, D.R. Melanopsin-dependent light avoidance in neonatal mice. Proc Natl Acad Sci USA 2010, 107, 17374–17378. [Google Scholar] [CrossRef] [PubMed]
- Burger, C.A.; Jiang, D.; Mackin, R.D.; Samuel, M.A. Development and maintenance of vision’s first synapse. Dev Biol 2021, 476, 218–239. [Google Scholar] [CrossRef] [PubMed]
- Jackson, G.R.; Owsley, C.; Curcio, C.A. Photoreceptor degeneration and dysfunction in aging and age-related maculopathy. Ageing Res Rev 2002, 1, 381–96. [Google Scholar] [CrossRef]
- Jeon, C.J.; Strettoi, E.; Masland, R.H. The major cell populations of the mouse retina. J Neurosci 1998, 18, 8936–46. [Google Scholar] [CrossRef] [PubMed]
- Jin, N.; Zhang, Z.; Keung, J.; Youn, S.B.; Ishibashi, M.; Tian, L.M.; Marshak, D.W.; Solessio, E.; Umino, Y.; Fahrenfort, I.; Kiyama, T.; Mao, C.A.; You, Y.; Wei, H.; Wu, J.; Postma, F.; Paul, D.L.; Massey, S.C.; Ribelayga, C.P. Molecular and functional architecture of the mouse photoreceptor network. Sci Adv 2020, 6, eaba7232. [Google Scholar] [CrossRef]
- Lee, S.; Chen, L.; Chen, M.; Ye, M.; Seal, R.P.; Zhou, Z.J. An unconventional glutamatergic circuit in the retina formed by vGluT3 amacrine cells. Neuron 2014, 84, 708–15. [Google Scholar] [CrossRef]
- Prigge, C.L.; Yeh, P.T.; Liou, N.F.; Lee, C.C.; You, S.F.; Liu, L.L.; McNeill, D.S.; Chew, K.S.; Hattar, S.; Chen, S.K.; Zhang, D.Q. M1 ipRGCs Influence Visual Function through Retrograde Signaling in the Retina. J Neurosci 2016, 36, 7184–97. [Google Scholar] [CrossRef]
- Maloney, R.; Quattrochi, L.; Yoon, J.; Souza, R.; Berson, D. Efficacy and specificity of melanopsin reporters for retinal ganglion cells. J Comp Neurol 2024, 532, e25591. [Google Scholar] [CrossRef]
- Hooks, B.M.; Chen, C. Critical periods in the visual system: changing views for a model of experience-dependent plasticity. Neuron 2007, 56, 312–26. [Google Scholar] [CrossRef]
- Tian, N.; Copenhagen, D.R. Visual stimulation is required for refinement of ON and OFF pathways in postnatal retina. Neuron 2003, 39, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Grishanin, R.N.; Tolwani, R.J.; Rentería, R.C.; Xu, B.; Reichardt, L.F.; Copenhagen, D.R. Brain-derived neurotrophic factor and TrkB modulate visual experience- dependent refinement of neuronal pathways in retina. J Neurosci 2007, 27, 7256–67. [Google Scholar] [CrossRef] [PubMed]
- Péquignot, M.; Provost, A.; Sallé, S.; Taupin, P.; Sainton, K.M.; Marchant, D.; Martinou, J.C.; Ameisen, J.C.; Jais, J.P.; Abitbol, M. Major role of BAX in apoptosis during retinal development and in establishment of a functional postnatal retina. Dev Dynam 2003, 228, 231–238. [Google Scholar] [CrossRef]
- O’Driscoll, C.; Donovan, M.; Cotter, T.G. Analysis of apoptotic and survival mediators in the early post-natal and mature retina. Exp Eye Res 2006, 83, 1482–92. [Google Scholar] [CrossRef] [PubMed]
- Tiriac, A,; Smith, B. E,; Feller, M.B. Light Prior to Eye Opening Promotes Retinal Waves and Eye-Specific Segregation. Neuron 2018, 100, 1059–1065. [Google Scholar] [CrossRef]
- Renna, J.M.; Chellappa, D.K.; Ross, C.L.; Stabio, M.E.; Berson, D.M. Melanopsin ganglion cells extend dendrites into the outer retina during early postnatal development. Dev Neurobiol 2015, 75, 935–46. [Google Scholar] [CrossRef]
- Hu, J.; Shi, Y.; Zhang, J.; Huang, X.; Wang, Q.; Zhao, H.; Shen, J.; Chen, Z.; Song, W.; Zheng, P.; Zhan, S.; Sun, Y.; Cai, P.; An, K.; Ouyang, C.; Zhao, B.; Zhou, Q.; Xu, L.; Xiong, W.; Zhang, Z.; Meng, J.; Chen, J.; Ma, Y.; Zhao, H.; Zhang, M.; Qu, K.; Hu, J.; Luo, M.; Xu, F.; Chen, X.; Xiong, Y.; Bao, J.; Xue, T. Melanopsin retinal ganglion cells mediate light-promoted brain development. Cell 2022, 185, 3124–3137. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, S.P.; Upton, B.A.; Eldred, K.C.; Glass, I.; Nayak, G.; Grover, K.; Ahmed, A.; Nguyen, M.T.; Hu, Y.C.; Gamlin, P.; Lang, R.A. Developmental control of rod number via a light- dependent retrograde pathway from intrinsically photosensitive retinal ganglion cells. Dev Cell 2024, 59, 2897–2911. [Google Scholar] [CrossRef]
- Znoiko, S.L.; Rohrer, B.; Lu, K.; Lohr, H. R.; Crouch, R.K.; Ma, J.X. Downregulation of cone-specific gene expression and degeneration of cone photoreceptors in the Rpe65-/- mouse at early ages. Invest Ophthalmol Vis Sci 2005, 46, 1473–1479. [Google Scholar] [CrossRef]
- Berry, C.T.; Sceniak, M.P.; Zhou, L.; Sabo, S.L. Developmental up-regulation of vesicular glutamate transporter-1 promotes neocortical presynaptic terminal development. PLoS One 2012, 7, e50911. [Google Scholar] [CrossRef] [PubMed]
| Antigen | Antibody dilution | Source Animal, Company |
|---|---|---|
| PKCα | 1:500 [23] | Mouse, Santa Cruz Biotechnology, USA |
| PKCα | 1:10000 [24] | Rabbit, Sigma-Aldrich, USA |
| SV-2 | 1:5000 [25] | Mouse, Developmental Studies Hybridoma Bank, USA |
| Calbindin | 1:2000 [24] | Rabbit, Millipore, USA |
| VGLUT1 | 1:1000 [5] | Rabbit, Synaptic Systems, Germany |
| VGLUT2 | 1:2000 [7] | Guinea pig, Millipore, USA |
| VGLUT2 | 1:500 [7] | Rabbit, Millipore, USA |
| VGLUT3 | 1:2000 [26] | Guinea pig, Millipore, USA |
| VGLUT3 | 1:1000 [8] | Rabbit, Millipore, USA |
| CtBP2 | 1:5000 [27] | Mouse, BD Transduction, USA |
| PKARIIβ | 1:1000 [28] | Rabbit, BD Transduction, USA |
| mGLUR6 | 1:500 [29] | Guinea Pig, Neuromics, USA |
| GluK1 (GluR5) | 1:100 [30] | Goat, Santa Cruz Biotechnologies, USA |
| Cone arrestin | 1:1000 [31] | Rabbit, Millipore, USA |
| TH | 1:400 [24] | Rabbit, Millipore, USA |
| ChAT | 1:400 [32] | Goat, Millipore, USA |
| Melanopsin | 1:2000 [33] | Rabbit, Millipore, USA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




