Submitted:
07 August 2025
Posted:
08 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
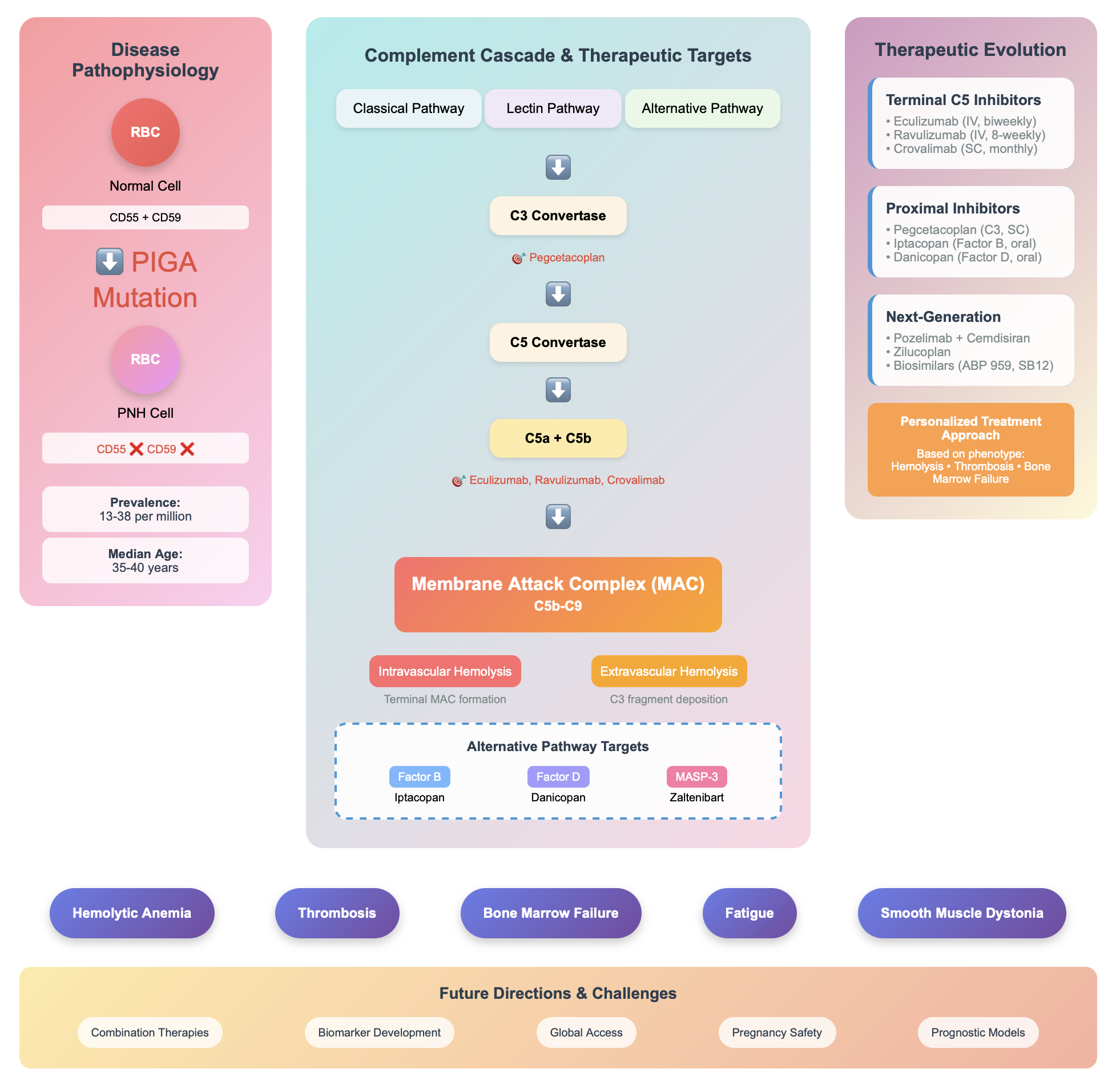
3. Disease Overview
4. Diagnosis
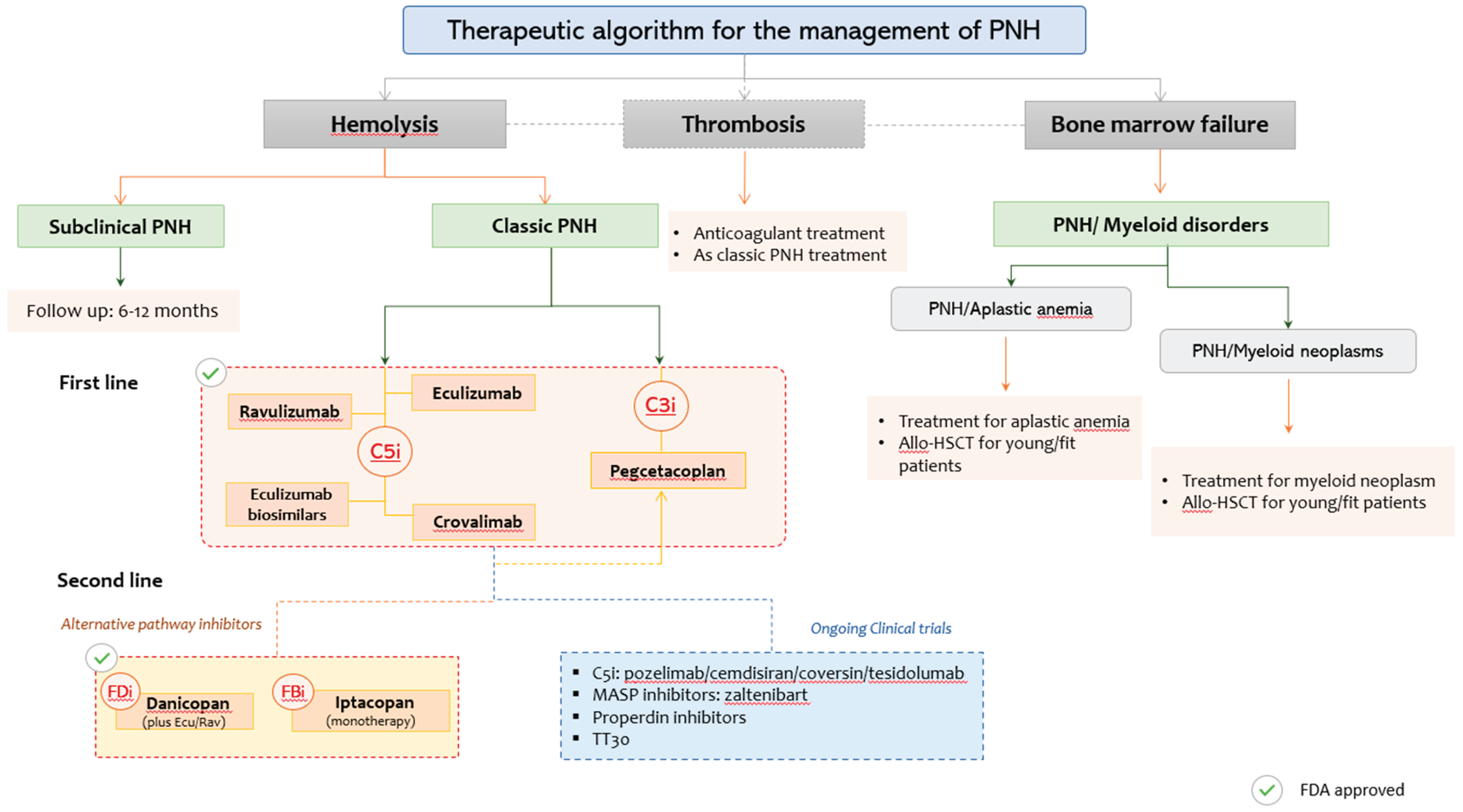
5. Evolvement in Complement Inhibition
5.1. Hemolysis-Predominant PNH
5.2. Thrombosis-Predominant PNH
5.3. PNH with Bone Marrow Failure Syndromes
5.4. Terminal Complement Inhibitors
5.4.1. Eculizumab
5.4.2. Ravulizumab
5.4.3. Crovalimab
5.5. Proximal Complement Inhibitors
5.5.1. Pegcetacoplan
5.5.2. Factor B Inhibitors
5.5.3. Factor D Inhibitors
6. Future Directions
6.1. Novel Therapeutic Approaches
6.2. Next Generation C5 Inhibitors
- Pozelimab (REGN3918): a fully human IgG4 monoclonal antibody targeting C5, offering potential advantages over first-generation agents. In the phase II trial (NCT03946748), pozelimab demonstrated rapid and sustained IVH control with a favorable safety profile in both treatment-naïve patients and those previously off C5 inhibitors. It is administered as a single IV loading dose (30 mg/kg) followed by weekly subcutaneous injections (800 mg). Importantly, it has shown efficacy in patients with C5 polymorphisms and is also under evaluation in combination regimens [58].
- Zilucoplan (RA101495): a small, synthetic macrocyclic peptide that binds C5 with high specificity, allowing daily subcutaneous self-administration. It inhibits MAC formation by preventing C5 cleavage. Two phase II trials (studies 201 and 203) assessed its efficacy in eculizumab-naïve and switch cohorts. While efficacy in naïve patients was encouraging, responses in patients previously treated with C5 inhibitors were comparatively less robust. Its favorable safety profile and pharmacokinetics support further investigation, possibly in combination with proximal inhibitors [59,60].
- Cemdisiran (ALN-CC5): an RNA interference (RNAi) therapeutic that silences C5 production at the mRNA level in hepatocytes, reducing circulating C5 protein. As monotherapy, it provides partial control of IVH. Ongoing trials are evaluating its combination with pozelimab and other agents for improved complement inhibition [56,61,62].
- Tesidolumab (LFG316): a human IgG1/λ monoclonal antibody binding to a distinct C5 epitope from eculizumab or ravulizumab. This may benefit patients with C5 variants or C5 inhibitor resistance. A phase II trial confirmed its efficacy in both wild-type and variant C5 populations, meeting the primary endpoint of LDH reduction [3,63].
6.3. Alternative Pathway Inhibitors
- Zaltenibart (OMS906): a fully humanized IgG4 antibody targeting MASP-3, the main activator of factor D in the alternative pathway. Zaltenibart may reduce infection risk while offering robust EVH and IVH control. It is under phase II and III investigation in both treatment-naïve and C5-experienced patients [64,65].
6.4. Eculizumab Biosimilars
6.5. Combination Therapies
7. Unmet Needs in PNH
- Incomplete control of EVH persists in a subset of patients treated with C5 inhibitors, resulting in continued anemia and transfusion dependency [6].
- The use of proximal complement inhibitors, although effective, is associated with a heightened risk of infections, emphasizing the need for enhanced safety profiles and appropriate prophylaxis [31].
- Access and affordability remain limiting factors in many regions, particularly in resource-constrained settings, where the high cost of complement inhibitors restricts equitable treatment access.
- Validated biomarkers for guiding treatment selection, predicting response, and monitoring disease activity are currently lacking, hindering the implementation of personalized therapy paradigms [72].
- PNH during pregnancy remains a uniquely challenging scenario. Among current therapies, eculizumab is the only extensively studied and routinely used option in this context [73].
- Additionally, the absence of a validated prognostic model to stratify patients according to risk of relapse or treatment failure represents a major gap. A robust prognostic tool could integrate multiple clinically relevant parameters including the size of the PNH clone, markers of hemolysis (e.g., LDH, hemoglobin, reticulocyte count), evidence of C3 fragment deposition, bone marrow status, history of thrombosis. Stratifying patients into low, intermediate, and high-risk categories could inform treatment escalation strategies, identify candidates for proximal inhibitors, and guide intensified monitoring. However, the low global prevalence of PNH poses a major barrier to developing and validating such algorithms through large, prospective datasets.
8. Discussion
9. Conclusions
Funding
Conflicts of Interest
References
- Brodsky, R.A. Paroxysmal nocturnal hemoglobinuria. Blood 2014, 124, 2804-2811. [CrossRef]
- Waheed, A.; Shammo, J.; Dingli, D. Paroxysmal nocturnal hemoglobinuria: Review of the patient experience and treatment landscape. Blood Rev 2024, 64, 101158. [CrossRef]
- Kulasekararaj, A.G.; Lazana, I. Paroxysmal nocturnal hemoglobinuria: Where are we going. Am J Hematol 2023, 98 Suppl 4, S33-s43. [CrossRef]
- Meyers, G.; Weitz, I.; Lamy, T.; Cahn, J.-Y.; Kroon, H.-A.; Severino, B.; Uranga, M.T.; Alonso, M.S.; Vela, J.A.G.; Hill, A. Disease-Related Symptoms Reported across a Broad Population of Patients with Paroxysmal Nocturnal Hemoglobinuria. Blood 2007, 110, 3683. [CrossRef]
- Schrezenmeier, H.; Röth, A.; Araten, D.J.; Kanakura, Y.; Larratt, L.; Shammo, J.M.; Wilson, A.; Shayan, G.; Maciejewski, J.P. Baseline clinical characteristics and disease burden in patients with paroxysmal nocturnal hemoglobinuria (PNH): updated analysis from the International PNH Registry. Ann Hematol 2020, 99, 1505-1514. [CrossRef]
- Oliver, M.; Patriquin, C.J. Paroxysmal Nocturnal Hemoglobinuria: Current Management, Unmet Needs, and Recommendations. J Blood Med 2023, 14, 613-628. [CrossRef]
- Du, Y.; Han, B. Advances in Hematopoietic Stem Cell Transplantation for Patients with Paroxysmal Nocturnal Hemoglobinuria. Transplant Cell Ther 2021, 27, 301-307. [CrossRef]
- Hillmen, P.; Young, N.S.; Schubert, J.; Brodsky, R.A.; Socie, G.; Muus, P.; Roth, A.; Szer, J.; Elebute, M.O.; Nakamura, R., et al. The complement inhibitor eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med 2006, 355, 1233-1243. [CrossRef]
- Lee, J.W.; Brodsky, R.A.; Nishimura, J.I.; Kulasekararaj, A.G. The role of the alternative pathway in paroxysmal nocturnal hemoglobinuria and emerging treatments. Expert Rev Clin Pharmacol 2022, 15, 851-861. [CrossRef]
- Versino, F.; Fattizzo, B. Complement inhibition in paroxysmal nocturnal hemoglobinuria: From biology to therapy. Int J Lab Hematol 2024, 46 Suppl 1, 43-54. [CrossRef]
- Ruiz-Argüelles, A.; Llorente, L. The role of complement regulatory proteins (CD55 and CD59) in the pathogenesis of autoimmune hemocytopenias. Autoimmun Rev 2007, 6, 155-161. [CrossRef]
- Geller, A.; Yan, J. The Role of Membrane Bound Complement Regulatory Proteins in Tumor Development and Cancer Immunotherapy. Front Immunol 2019, 10, 1074. [CrossRef]
- Schmidt, C.Q.; Hochsmann, B.; Schrezenmeier, H. The complement model disease paroxysmal nocturnal hemoglobinuria. Eur J Immunol 2024, 54, e2350817. [CrossRef]
- Dobó, J.; Kocsis, A.; Gál, P. Be on Target: Strategies of Targeting Alternative and Lectin Pathway Components in Complement-Mediated Diseases. Frontiers in Immunology 2018, Volume 9 - 2018. [CrossRef]
- Dobó, J.; Kocsis, A.; Farkas, B.; Demeter, F.; Cervenak, L.; Gál, P. The Lectin Pathway of the Complement System—Activation, Regulation, Disease Connections and Interplay with Other (Proteolytic) Systems. International Journal of Molecular Sciences 2024, 25, 1566.
- Risitano, A.M.; Frieri, C.; Urciuoli, E.; Marano, L. The complement alternative pathway in paroxysmal nocturnal hemoglobinuria: From a pathogenic mechanism to a therapeutic target. Immunol Rev 2023, 313, 262-278. [CrossRef]
- Richards, S.J.; Dickinson, A.J.; Newton, D.J.; Hillmen, P. Immunophenotypic assessment of PNH clones in major and minor cell lineages in the peripheral blood of patients with paroxysmal nocturnal hemoglobinuria. Cytometry B Clin Cytom 2022, 102, 487-497. [CrossRef]
- Borowitz, M.J.; Craig, F.E.; Digiuseppe, J.A.; Illingworth, A.J.; Rosse, W.; Sutherland, D.R.; Wittwer, C.T.; Richards, S.J. Guidelines for the diagnosis and monitoring of paroxysmal nocturnal hemoglobinuria and related disorders by flow cytometry. Cytometry B Clin Cytom 2010, 78, 211-230. [CrossRef]
- Babushok, D.V. When does a PNH clone have clinical significance? Hematology Am Soc Hematol Educ Program 2021, 2021, 143-152. [CrossRef]
- Cançado, R.D.; Araújo, A.D.S.; Sandes, A.F.; Arrais, C.; Lobo, C.L.C.; Figueiredo, M.S.; Gualandro, S.F.M.; Saad, S.T.O.; Costa, F.F. Consensus statement for diagnosis and treatment of paroxysmal nocturnal haemoglobinuria. Hematol Transfus Cell Ther 2021, 43, 341-348. [CrossRef]
- Tombul, Z.; Bahaj, W.; Ozturk, M.; Patel, B.; Toprak, A.; Ibrahim, I.; Chen, W.; Fuda, F.; Ogbue, O.D.; Gurnari, C., et al. Ahemolytic PNH (white cell PNH): Clinical features and implications of a distinct phenotype of paroxysmal nocturnal haemoglobinuria. Br J Haematol 2024, 204, 2121-2124. [CrossRef]
- Rother, R.P.; Rollins, S.A.; Mojcik, C.F.; Brodsky, R.A.; Bell, L. Discovery and development of the complement inhibitor eculizumab for the treatment of paroxysmal nocturnal hemoglobinuria. Nat Biotechnol 2007, 25, 1256-1264. [CrossRef]
- Gavriilaki, E.; de Latour, R.P.; Risitano, A.M. Advancing therapeutic complement inhibition in hematologic diseases: PNH and beyond. Blood 2022, 139, 3571-3582. [CrossRef]
- Brodsky, R.A.; Young, N.S.; Antonioli, E.; Risitano, A.M.; Schrezenmeier, H.; Schubert, J.; Gaya, A.; Coyle, L.; de Castro, C.; Fu, C.L., et al. Multicenter phase 3 study of the complement inhibitor eculizumab for the treatment of patients with paroxysmal nocturnal hemoglobinuria. Blood 2008, 111, 1840-1847. [CrossRef]
- Schubert, J.; Hillmen, P.; Röth, A.; Young, N.S.; Elebute, M.O.; Szer, J.; Gianfaldoni, G.; Socié, G.; Browne, P.; Geller, R., et al. Eculizumab, a terminal complement inhibitor, improves anaemia in patients with paroxysmal nocturnal haemoglobinuria. Br J Haematol 2008, 142, 263-272. [CrossRef]
- Stern, R.M.; Connell, N.T. Ravulizumab: a novel C5 inhibitor for the treatment of paroxysmal nocturnal hemoglobinuria. Ther Adv Hematol 2019, 10, 2040620719874728. [CrossRef]
- Notaro, R.; Luzzatto, L. Breakthrough Hemolysis in PNH with Proximal or Terminal Complement Inhibition. N Engl J Med 2022, 387, 160-166. [CrossRef]
- Kelly, R.; Richards, S.; Hillmen, P.; Hill, A. The pathophysiology of paroxysmal nocturnal hemoglobinuria and treatment with eculizumab. Ther Clin Risk Manag 2009, 5, 911-921. [CrossRef]
- Risitano, A.M.; Marotta, S.; Ricci, P.; Marano, L.; Frieri, C.; Cacace, F.; Sica, M.; Kulasekararaj, A.; Calado, R.T.; Scheinberg, P., et al. Anti-complement Treatment for Paroxysmal Nocturnal Hemoglobinuria: Time for Proximal Complement Inhibition? A Position Paper From the SAAWP of the EBMT. Front Immunol 2019, 10, 1157. [CrossRef]
- Notaro, R.; Sica, M. C3-mediated extravascular hemolysis in PNH on eculizumab: Mechanism and clinical implications. Semin Hematol 2018, 55, 130-135. [CrossRef]
- Girmenia, C.; Barcellini, W.; Bianchi, P.; Di Bona, E.; Iori, A.P.; Notaro, R.; Sica, S.; Zanella, A.; De Vivo, A.; Barosi, G., et al. Management of infection in PNH patients treated with eculizumab or other complement inhibitors: Unmet clinical needs. Blood Rev 2023, 58, 101013. [CrossRef]
- Lee, S.E.; Lee, J.W. Safety of current treatments for paroxysmal nocturnal hemoglobinuria. Expert Opin Drug Saf 2021, 20, 171-179. [CrossRef]
- Kulasekararaj, A.G.; Hill, A.; Rottinghaus, S.T.; Langemeijer, S.; Wells, R.; Gonzalez-Fernandez, F.A.; Gaya, A.; Lee, J.W.; Gutierrez, E.O.; Piatek, C.I., et al. Ravulizumab (ALXN1210) vs eculizumab in C5-inhibitor-experienced adult patients with PNH: the 302 study. Blood 2019, 133, 540-549. [CrossRef]
- Brodsky, R.A.; Peffault de Latour, R.; Rottinghaus, S.T.; Röth, A.; Risitano, A.M.; Weitz, I.C.; Hillmen, P.; Maciejewski, J.P.; Szer, J.; Lee, J.W., et al. Characterization of breakthrough hemolysis events observed in the phase 3 randomized studies of ravulizumab versus eculizumab in adults with paroxysmal nocturnal hemoglobinuria. Haematologica 2021, 106, 230-237. [CrossRef]
- Kulasekararaj, A.; Brodsky, R.; Schrezenmeier, H.; Griffin, M.; Röth, A.; Piatek, C.; Ogawa, M.; Yu, J.; Patel, A.S.; Patel, Y., et al. Ravulizumab demonstrates long-term efficacy, safety and favorable patient survival in patients with paroxysmal nocturnal hemoglobinuria. Ann Hematol 2025, 104, 81-94. [CrossRef]
- Lee, J.W.; Sicre de Fontbrune, F.; Wong Lee Lee, L.; Pessoa, V.; Gualandro, S.; Fureder, W.; Ptushkin, V.; Rottinghaus, S.T.; Volles, L.; Shafner, L., et al. Ravulizumab (ALXN1210) vs eculizumab in adult patients with PNH naive to complement inhibitors: the 301 study. Blood 2019, 133, 530-539. [CrossRef]
- Dhillon, S. Crovalimab: First Approval. Drugs 2024, 84, 707-716. [CrossRef]
- Scheinberg, P.; Cle, D.V.; Kim, J.S.; Nur, E.; Yenerel, M.N.; Barcellini, W.; Bonito, D.; Giai, V.; Hus, M.; Lee, Y., et al. Phase 3 randomized COMMODORE 1 trial: Crovalimab versus eculizumab in complement inhibitor-experienced patients with paroxysmal nocturnal hemoglobinuria. Am J Hematol 2024, 99, 1757-1767. [CrossRef]
- Roth, A.; He, G.; Tong, H.; Lin, Z.; Wang, X.; Chai-Adisaksopha, C.; Lee, J.H.; Brodsky, A.; Hantaweepant, C.; Dumagay, T.E., et al. Phase 3 randomized COMMODORE 2 trial: Crovalimab versus eculizumab in patients with paroxysmal nocturnal hemoglobinuria naive to complement inhibition. Am J Hematol 2024, 99, 1768-1777. [CrossRef]
- Liu, H.; Xia, L.; Weng, J.; Zhang, F.; He, C.; Gao, S.; Jia, J.; Chang, A.C.; Lundberg, P.; Shi, J., et al. Efficacy and safety of the C5 inhibitor crovalimab in complement inhibitor-naive patients with PNH (COMMODORE 3): A multicenter, Phase 3, single-arm study. Am J Hematol 2023, 98, 1407-1414. [CrossRef]
- Gerber, G.F.; Brodsky, R.A. Pegcetacoplan for paroxysmal nocturnal hemoglobinuria. Blood 2022, 139, 3361-3365. [CrossRef]
- Heo, Y.A. Pegcetacoplan: A Review in Paroxysmal Nocturnal Haemoglobinuria. Drugs 2022, 82, 1727-1735. [CrossRef]
- Heo, Y.A. Correction to: Pegcetacoplan: A Review in Paroxysmal Nocturnal Haemoglobinuria. Drugs 2023, 83, 949. [CrossRef]
- Wong, R.S.M.; Navarro-Cabrera, J.R.; Comia, N.S.; Goh, Y.T.; Idrobo, H.; Kongkabpan, D.; Gomez-Almaguer, D.; Al-Adhami, M.; Ajayi, T.; Alvarenga, P., et al. Pegcetacoplan controls hemolysis in complement inhibitor-naive patients with paroxysmal nocturnal hemoglobinuria. Blood Adv 2023, 7, 2468-2478. [CrossRef]
- Hoy, S.M. Pegcetacoplan: First Approval. Drugs 2021, 81, 1423-1430. [CrossRef]
- Hillmen, P.; Szer, J.; Weitz, I.; Roth, A.; Hochsmann, B.; Panse, J.; Usuki, K.; Griffin, M.; Kiladjian, J.J.; de Castro, C., et al. Pegcetacoplan versus Eculizumab in Paroxysmal Nocturnal Hemoglobinuria. N Engl J Med 2021, 384, 1028-1037. [CrossRef]
- Horneff, R.; Czech, B.; Yeh, M.; Surova, E. Three Years On: The Role of Pegcetacoplan in Paroxysmal Nocturnal Hemoglobinuria (PNH) since Its Initial Approval. Int J Mol Sci 2024, 25. [CrossRef]
- Dighriri, I.M.; Al-Qahtani, R.M.; Almutairi, A.O.; Alhashbari, R.N.; Kanbeja, H.A.; AlOjaimi, S.A.; Aljuaid, M.A.; Albaradi, A.A.; Almanjumi, S.T.; Alqurashi, S.A., et al. Iptacopan Efficacy and Safety to Treat Paroxysmal Nocturnal Hemoglobinuria (PNH): A Systematic Review and Meta-Analysis. Cureus 2024, 16, e67830. [CrossRef]
- Risitano, A.M.; Kulasekararaj, A.; Roeth, A.; Scheinberg, P.; Ueda, Y.; de Castro, C.; Di Bona, E.; Griffin, M.; Langemeijer, S.M.; Schrezenmeier, H., et al. Factor B Inhibition with Oral Iptacopan Monotherapy Demonstrates Sustained Long-Term Efficacy and Safety in Anti-C5-Treated Patients (pts) with Paroxysmal Nocturnal Hemoglobinuria (PNH) and Persistent Anemia: Final 48-Week Results from the Multicenter, Phase III APPLY-PNH Trial. Blood 2023, 142, 571-571. [CrossRef]
- Syed, Y.Y. Iptacopan: First Approval. Drugs 2024, 84, 599-606. [CrossRef]
- Peffault de Latour, R.; Roth, A.; Kulasekararaj, A.G.; Han, B.; Scheinberg, P.; Maciejewski, J.P.; Ueda, Y.; de Castro, C.M.; Di Bona, E.; Fu, R., et al. Oral Iptacopan Monotherapy in Paroxysmal Nocturnal Hemoglobinuria. N Engl J Med 2024, 390, 994-1008. [CrossRef]
- Gavriilaki, E.; Papakonstantinou, A.; Agrios, K.A. Novel Insights into Factor D Inhibition. Int J Mol Sci 2022, 23. [CrossRef]
- Yuan, X.; Gavriilaki, E.; Thanassi, J.A.; Yang, G.; Baines, A.C.; Podos, S.D.; Huang, Y.; Huang, M.; Brodsky, R.A. Small-molecule factor D inhibitors selectively block the alternative pathway of complement in paroxysmal nocturnal hemoglobinuria and atypical hemolytic uremic syndrome. Haematologica 2017, 102, 466-475. [CrossRef]
- Risitano, A.M.; Kulasekararaj, A.G.; Lee, J.W.; Maciejewski, J.P.; Notaro, R.; Brodsky, R.; Huang, M.; Geffner, M.; Browett, P. Danicopan: an oral complement factor D inhibitor for paroxysmal nocturnal hemoglobinuria. Haematologica 2021, 106, 3188-3197. [CrossRef]
- Lee, J.W.; Griffin, M.; Kim, J.S.; Lee Lee, L.W.; Piatek, C.; Nishimura, J.I.; Carrillo Infante, C.; Jain, D.; Liu, P.; Filippov, G., et al. Addition of danicopan to ravulizumab or eculizumab in patients with paroxysmal nocturnal haemoglobinuria and clinically significant extravascular haemolysis (ALPHA): a double-blind, randomised, phase 3 trial. Lancet Haematol 2023, 10, e955-e965. [CrossRef]
- Panse, J.P.; Höchsmann, B.; Schubert, J. Paroxysmal Nocturnal Hemoglobinuria, Pathophysiology, Diagnostics, and Treatment. Transfus Med Hemother 2024, 51, 310-320. [CrossRef]
- Risitano, A.M.; Peffault de Latour, R. How we(‘ll) treat paroxysmal nocturnal haemoglobinuria: diving into the future. Br J Haematol 2022, 196, 288-303. [CrossRef]
- Jang, J.-H.; Weyne, J.; Chaudhari, U.; Harari, O.; Miller, J.; Dain, B.; Meagher, K.A.; Rodgers, M.L.; Perlee, L.; Morton, L., et al. Pozelimab, a Human Monoclonal Antibody Against Complement Factor C5, Provided Inhibition of Intravascular Hemolysis in Patients with Paroxysmal Nocturnal Hemoglobinuria. Blood 2021, 138, 1128. [CrossRef]
- Tang, G.Q.; Tang, Y.; Dhamnaskar, K.; Hoarty, M.D.; Vyasamneni, R.; Vadysirisack, D.D.; Ma, Z.; Zhu, N.; Wang, J.G.; Bu, C., et al. Zilucoplan, a macrocyclic peptide inhibitor of human complement component 5, uses a dual mode of action to prevent terminal complement pathway activation. Front Immunol 2023, 14, 1213920. [CrossRef]
- Tang, G.Q.; Tang, Y.; Dhamnaskar, K.; Hoarty, M.D.; Vyasamneni, R.; Vadysirisack, D.D.; Ma, Z.; Zhu, N.; Wang, J.G.; Bu, C., et al. Corrigendum: Zilucoplan, a macrocyclic peptide inhibitor of human complement component 5, uses a dual mode of action to prevent terminal complement pathway activation. Front Immunol 2023, 14, 1282155. [CrossRef]
- Badri, P.; Jiang, X.; Borodovsky, A.; Najafian, N.; Kim, J.; Clausen, V.A.; Goel, V.; Habtemariam, B.; Robbie, G.J. Pharmacokinetic and Pharmacodynamic Properties of Cemdisiran, an RNAi Therapeutic Targeting Complement Component 5, in Healthy Subjects and Patients with Paroxysmal Nocturnal Hemoglobinuria. Clin Pharmacokinet 2021, 60, 365-378. [CrossRef]
- Brodsky, R.A. Paroxysmal nocturnal hemoglobinuria without GPI-anchor deficiency. J Clin Invest 2019, 129, 5074-5076. [CrossRef]
- Nishimura, J.I.; Ando, K.; Masuko, M.; Noji, H.; Ito, Y.; Mayer, J.; Griskevicius, L.; Bucher, C.; Mullershausen, F.; Gergely, P., et al. Tesidolumab (LFG316) for treatment of C5-variant patients with paroxysmal nocturnal hemoglobinuria. Haematologica 2022, 107, 1483-1488. [CrossRef]
- Dobo, J.; Pal, G.; Cervenak, L.; Gal, P. The emerging roles of mannose-binding lectin-associated serine proteases (MASPs) in the lectin pathway of complement and beyond. Immunol Rev 2016, 274, 98-111. [CrossRef]
- Li, Y.; Yabuki, M.; Cummings, W.J. Alternative Pathway Masp-3 Inhibitor OMS906 Effectively and Potently Inhibits Complement-Mediated Hemolysis in Preclinical Models Mechanistically Similar to Paroxysmal Nocturnal Hemoglobinuria. Blood 2023, 142, 4082-4082. [CrossRef]
- Chen, J.Y.; Cortes, C.; Ferreira, V.P. Properdin: A multifaceted molecule involved in inflammation and diseases. Mol Immunol 2018, 102, 58-72. [CrossRef]
- Chen, J.Y.; Galwankar, N.S.; Emch, H.N.; Menon, S.S.; Cortes, C.; Thurman, J.M.; Merrill, S.A.; Brodsky, R.A.; Ferreira, V.P. Properdin Is a Key Player in Lysis of Red Blood Cells and Complement Activation on Endothelial Cells in Hemolytic Anemias Caused by Complement Dysregulation. Front Immunol 2020, 11, 1460. [CrossRef]
- Kulasekararaj, A.; Brodsky, R.; Kulagin, A.; Jang, J.H. Biosimilars in rare diseases: a focus on paroxysmal nocturnal hemoglobinuria. Haematologica 2023, 108, 1232-1243. [CrossRef]
- Kulasekararaj, A.; Lanza, F.; Arvanitakis, A.; Langemeijer, S.; Chonat, S.; Tombak, A.; Hanes, V.; Cao, J.; Miller, M.J.; Colbert, A., et al. Comparative clinical efficacy and safety of biosimilar ABP 959 and eculizumab reference product in patients with paroxysmal nocturnal hemoglobinuria. Am J Hematol 2024, 99, 2108-2117. [CrossRef]
- Jang, J.H.; Gomez, R.D.; Bumbea, H.; Nogaieva, L.; Wong, L.L.L.; Lim, S.M.; Kim, Y.; Park, J. A phase III, randomised, double-blind, multi-national clinical trial comparing SB12 (proposed eculizumab biosimilar) and reference eculizumab in patients with paroxysmal nocturnal haemoglobinuria. EJHaem 2023, 4, 26-36. [CrossRef]
- Patriquin, C.; Jang, J.-H.; Aurand, L.; Taneja, D.; Magyar, A.; Dain, B.; Meagher, K.; Perlee, L.; Souttou, A.; Griffin, M. Efficacy and Safety of Pozelimab Plus Cemdisiran Vs Ravulizumab in Patients with Paroxysmal Nocturnal Hemoglobinuria Who Are Naïve to Complement Inhibition. Blood 2024, 144, 306. [CrossRef]
- Kulasekararaj, A.G.; Kuter, D.J.; Griffin, M.; Weitz, I.C.; Röth, A. Biomarkers and laboratory assessments for monitoring the treatment of patients with paroxysmal nocturnal hemoglobinuria: Differences between terminal and proximal complement inhibition. Blood Rev 2023, 59, 101041. [CrossRef]
- Kelly, R.J.; Höchsmann, B.; Szer, J.; Kulasekararaj, A.; de Guibert, S.; Röth, A.; Weitz, I.C.; Armstrong, E.; Risitano, A.M.; Patriquin, C.J., et al. Eculizumab in Pregnant Patients with Paroxysmal Nocturnal Hemoglobinuria. N Engl J Med 2015, 373, 1032-1039. [CrossRef]
| Agent | Target | Route | Dosing Interval | Key Benefits | Limitations |
|---|---|---|---|---|---|
| Eculizumab | C5 | IV | Biweekly | Reduces IVH, improves survival | BTH, IV only |
| Ravulizumab | C5 | IV | Every 8 weeks | Longer half-life, fewer BTH events | Cost, infection risk |
| Crovalimab | C5 (recycling Ab) |
SC | Monthly | SC, convenient, robust IVH control | Limited long-term data |
| Pegcetacoplan | C3 | SC | Twice weekly | Controls both IVH and EVH | Injection site reactions, infection risk |
| Iptacopan | Factor B | Oral | BID | Improves Hb, oral route, monotherapy | Mild infections, long-term safety under review |
| Danicopan | Factor D | Oral | TID | Add-on therapy, effective in EVH | Short half-life, TID dosing |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




