Submitted:
06 August 2025
Posted:
07 August 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Lipidomic Analysis
2.3. Olink Analysis
2.4. Gene Ontology Analysis
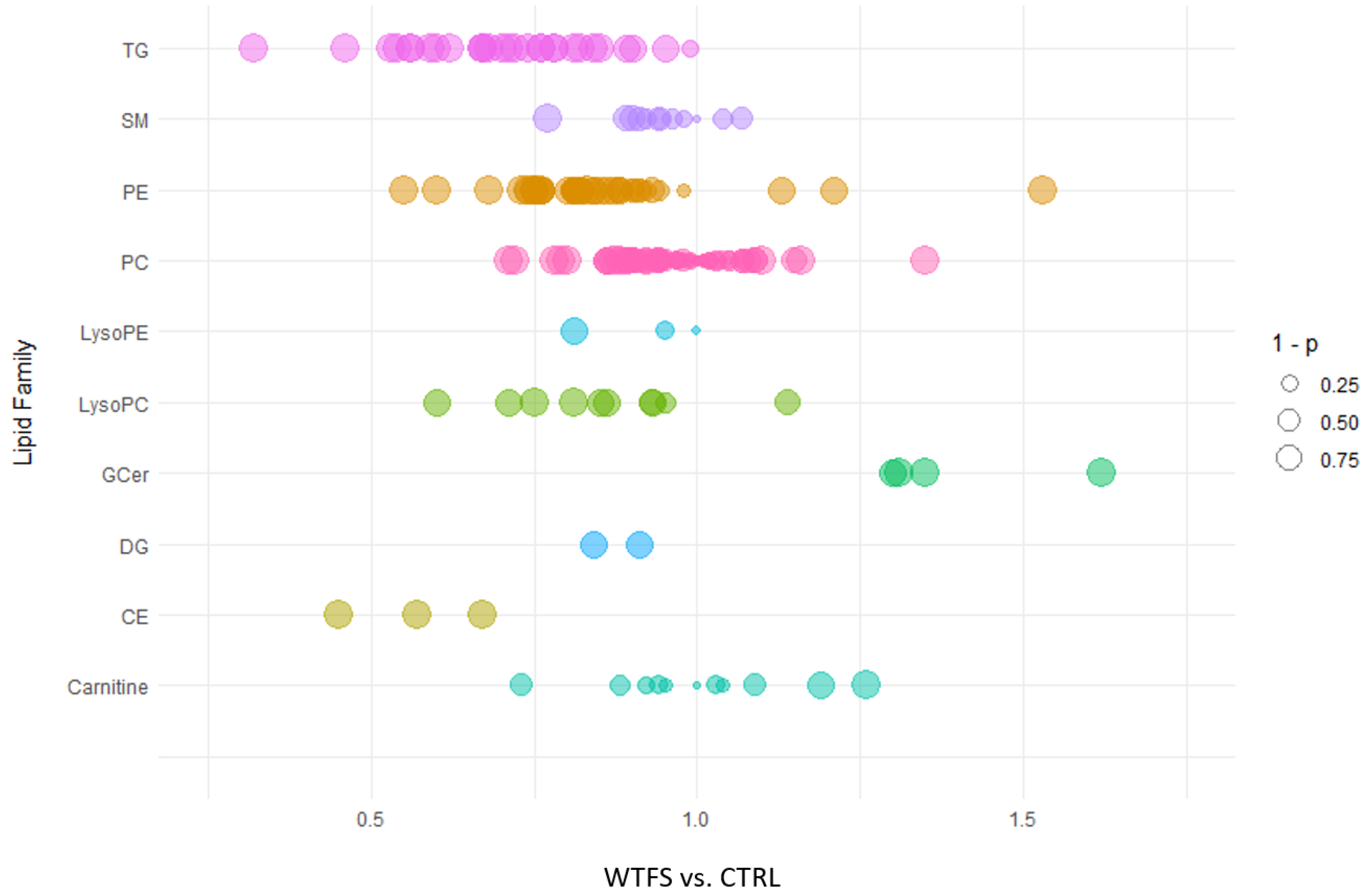
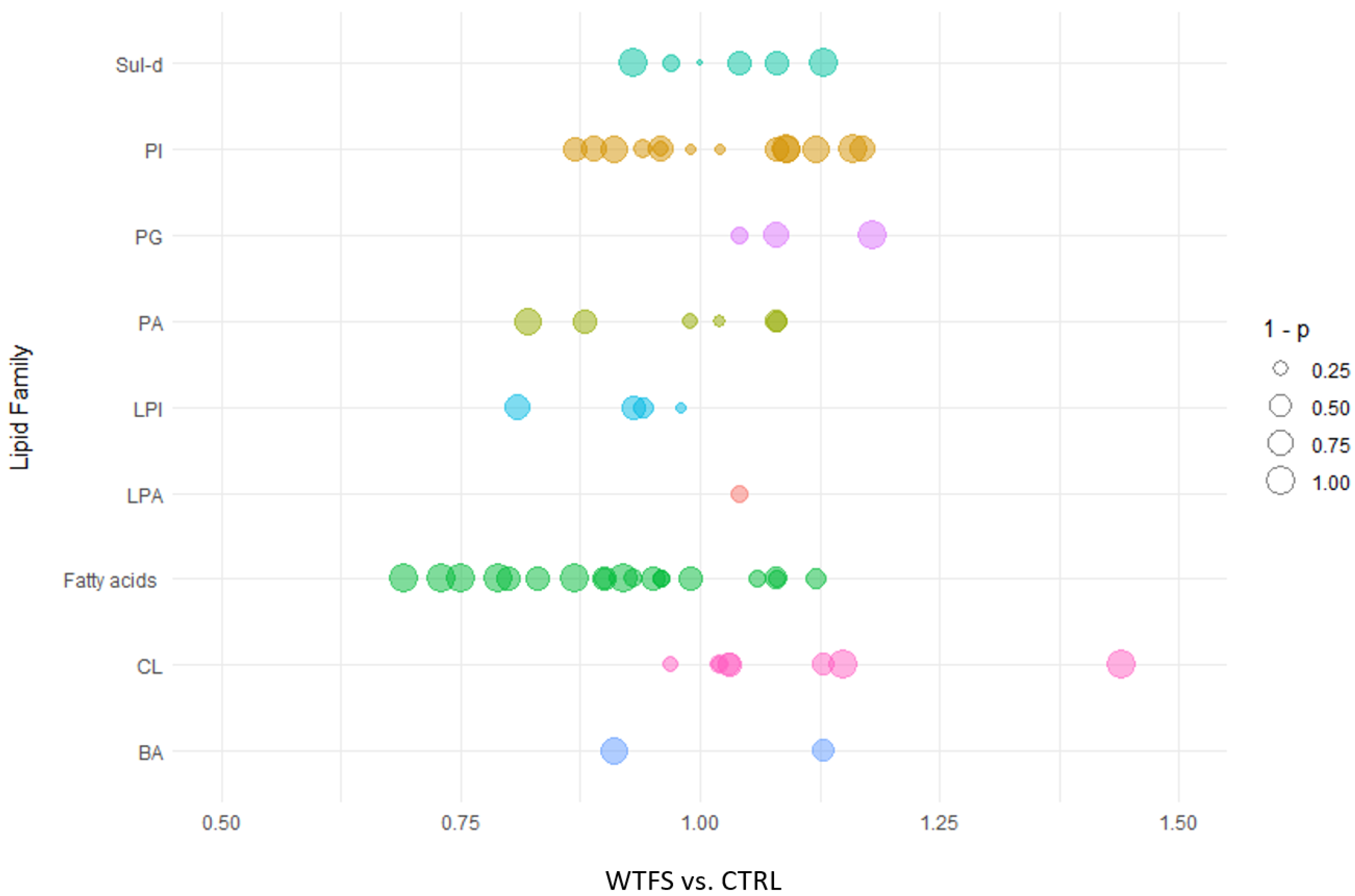
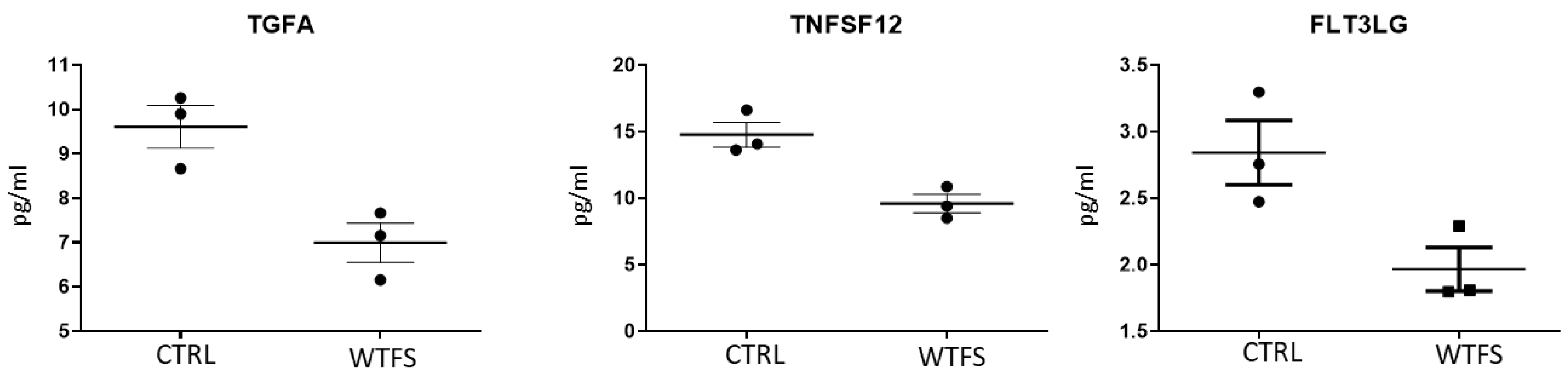
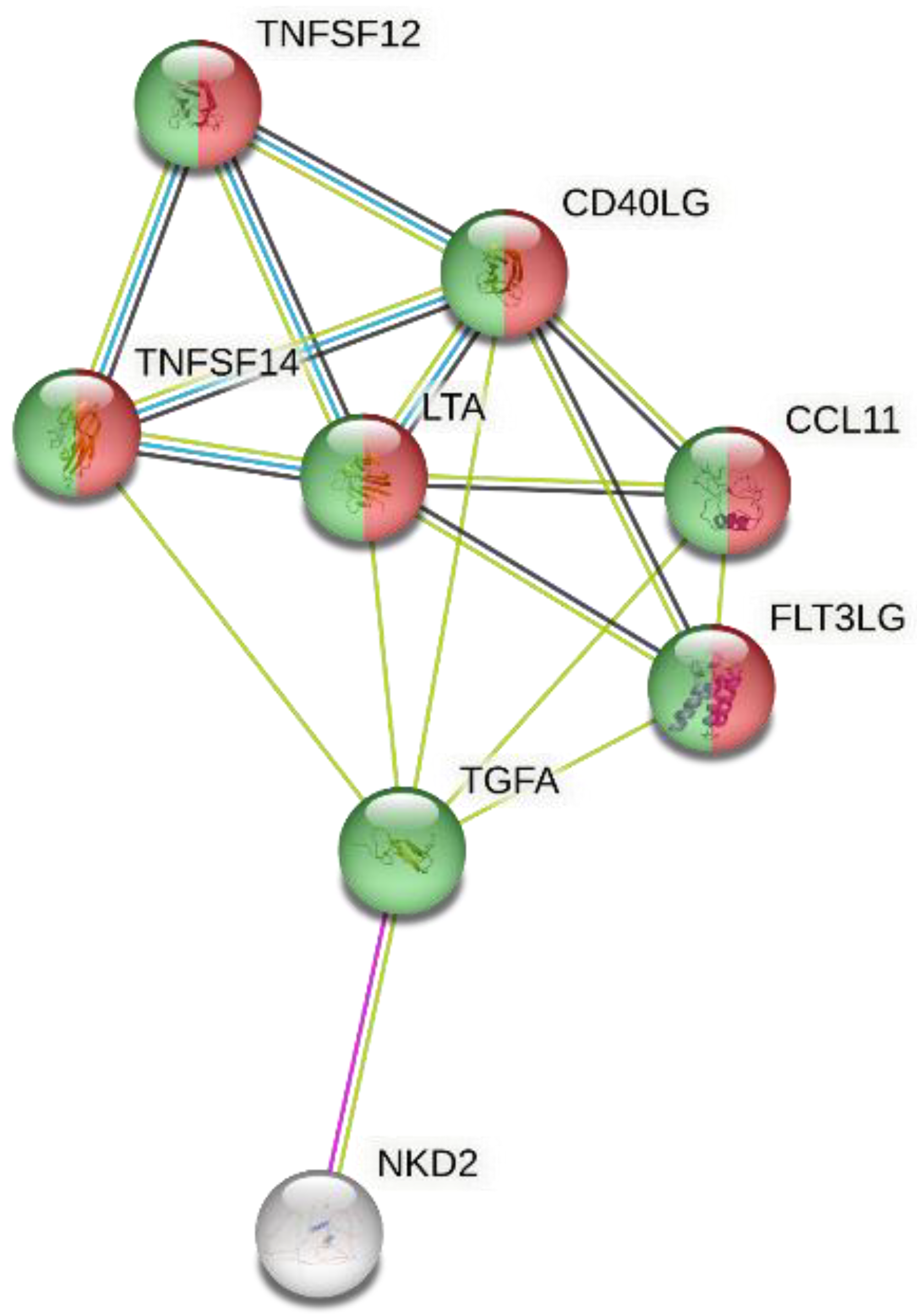
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CTRL | control |
| DMSO | dimethyl sulfoxide |
| NAFLD | non-alcoholic fatty liver disease |
| WTFS | whole tomato-based food supplement |
References
- Harrison, S.A.; Torgerson, S.; Hayashi, P.; Ward, J.; Schenker, S. Vitamin E and vitamin C treatment improves fibrosis in patients with nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2003, 98, 2485–2490. [Google Scholar] [CrossRef]
- Kugelmas, M.; Hill, D.B.; Vivian, B.; Marsano, L.; McClain, C.J. Cytokines and NASH: A pilot study of the effects of lifestyle modification and vitamin E. Hepatology 2003, 38, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Tessari, P.; Coracina, A.; Cosma, A.; Tiengo, A. Hepatic lipid metabolism and non-alcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Thorup, A.C.; Kristensen, H.L.; Kidmose, U.; Lambert, M.N.T.; Christensen, L.P.; Fretté, X.; Clausen, M.R.; Hansen, S.M.; Jeppesen, P.B. Strong and Bitter Vegetables from Traditional Cultivars and Cropping Methods Improve the Health Status of Type 2 Diabetics: A Randomized Control Trial. Nutrients 2021, 13, 1813. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, D.; Zhang, D.; Lv, Y.; Wei, Y.; Wu, W.; Zhou, F.; Tang, M.; Mao, T.; Li, M.; et al. Inhibitory effect of blueberry polyphenolic compounds on oleic acid-induced hepatic steatosis in vitro. J. Agric. Food Chem. 2011, 59, 12254–12263. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010, 52, 1836–1846. [Google Scholar] [CrossRef]
- Pouwels, S.; Sakran, N.; Graham, Y.; Leal, A.; Pintar, T.; Yang, W.; Kassir, R.; Singhal, R.; Mahawar, K. ; Ramnarain, D Non-alcoholic fatty liver disease (NAFLD): a review of pathophysiology, clinical management and effects of weight loss. BMC Endocr. Disord. 2022, 22, 63. [Google Scholar] [CrossRef]
- Wang, L.L.; Zhang, P.H.; Yan, H.H. Functional foods and dietary supplements in the management of non-alcoholic fatty liver disease: A systematic review and meta-analysis. Front. Nutr. 2023, 10, 1014010. [Google Scholar] [CrossRef]
- Subhash, K.; Bose, C.; Agrawal, B.K. Effect of short-term supplementation of tomatoes on antioxidant enzymes and lipid peroxidation in type-II diabetes. Indian J. Clin. Biochem. 2007, 22, 95–98. [Google Scholar] [CrossRef]
- Shafe, M.O.; Gumede, N.M.; Nyakudya, T.T.; Chivandi, E. Lycopene: A Potent Antioxidant with Multiple Health Benefits. J. Nutr. Metab. 2024, 6252426. [Google Scholar] [CrossRef]
- Wang, J.; Geng, T.; Zou, Q.; Yang, N.; Zhao, W.; Li, Y.; Tan, X.; Yuan, T.; Liu, X.; Zhigang. Liu. Lycopene prevents lipid accumulation in hepatocytes by stimulating PPARα and improving mitochondrial function. J. Funct. Foods 2020, 103857. [CrossRef]
- Mein, J.R.; Lian, F.; Wang, X.D. Biological activity of lycopene metabolites: implications for cancer prevention. Nutr Rev. 2008, 66, 667–683. [Google Scholar] [CrossRef]
- Sultan Alvi, S.; Ansari, I.A.; Khan, I.; Iqbal, J.; Khan, M.S. Potential role of lycopene in targeting proprotein convertase subtilisin/kexin type-9 to combat hypercholesterolemia. Free Radic. Biol. Med. 2017, 108, 394–403. [Google Scholar] [CrossRef]
- Schmitz, H. H.; Poor, C. L.; Wellman, R. B.; Erdman, J. W., Jr. Concentrations of selected carotenoids and vitamin A in human liver, kidney and lung tissue. J. Nutr. 1991, 121, 1613–1621. [Google Scholar] [CrossRef]
- Wang, X.D. Lycopene metabolism and its biological significance. Am. J. Clin. Nutr. 2012, 96, 1214S– 1222S. [CrossRef]
- Elvira-Torales, L.I.; García-Alonso, J.; Periago-Castón, M.J. Nutritional Importance of Carotenoids and Their Effect on Liver Health: A Review. Antioxidants (Basel). 2019, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- Makri, E.; Goulas, A.; Polyzos, S.A. Epidemiology, pathogenesis, diagnosis and emerging treatment of nonalcoholic fatty liver disease. Arch. Med. Res. 2021, 52, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Rühl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600685. [Google Scholar] [CrossRef] [PubMed]
- Marquez, C.S.; Reis Lima, M.J.; Oliveira, J:, Teixeira-Lemos, E. Tomato lycopene: functional proprieties and health benefits. Int. J. Agric. Eng. 2015, 9, 1089–1099. [CrossRef]
- Johnson, E.J.; Qin, J.; Krinsky, N.I.; Russell, R.M. Ingestion by men of a combined dose of beta-carotene and lycopene does not affect the absorption of beta-carotene but improves that of lycopene. J Nutr. 1997, 127, 1833–1837. [Google Scholar] [CrossRef]
- Tamanna, N.; Mahmood, N. Food processing and Maillard reaction products: effect on human health and nutrition. Int. J. Food Sci. 2015, 2015, 526762. [Google Scholar] [CrossRef]
- Rowles, J.L. 3r.; Erdman, J.W. Jr. Carotenoids and their role in cancer prevention. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2020, 1865, 158613. [Google Scholar]
- Linnewiel-Hermoni, K.; Khanin, M.; Danilenko, M.; Zango, G.; Amosi, Y.; Levy, J.; Sharoni, Y. The anti-cancer effects of carotenoids and other phytonutrients resides in their combined activity. Arch. Biochem. Biophys. 2015, 572, 28–35. [Google Scholar] [CrossRef]
- Landrier, J.F.; Breniere, T.; Sani, L.; Desmarchelier, C.; Mounien, L.; Borel, P. Effect of tomato, tomato-derived products and lycopene on metabolic inflammation: from epidemiological data to molecular mechanisms. Nutr. Res. Rev. 2023, 18, 1–17. [Google Scholar] [CrossRef]
- Mohri, S.; Takahashi, H.; Sakai, M.; Takahashi, S.; Waki, N.; Aizawa, K.; Suganuma, H.; Ara, T.; Matsumura, Y.; Shibata, D.; et al. Wide-range screening of anti-inflammatory compounds in tomato using LC-MS and elucidating the mechanism of their functions. PLoS One 2018, 13, e0191203. [Google Scholar] [CrossRef]
- Mazidi, M.; Katsiki, N.; George, E.S.; Banach, M. Tomato and lycopene consumption is inversely associated with total and cause-specific mortality: a population-based cohort study, on behalf of the International Lipid Expert Panel (ILEP). Br. J. Nutr. 2020, 124, 1303–10. [Google Scholar] [CrossRef] [PubMed]
- Fogliano, V.; Iacobelli, S.; Piantelli, M. Euro Patent 3 052 113 B1, Italian Health Ministry (registration n. 68843, 2018–2019). Available online: https://worldwide.espacenet.com/patent/search/family/049226079/publication/EP3052113A1?q=3052113 (accessed on July 8, 2025).
- Gholami, F.; Antonio, J.; Evans, C.; Cheraghi, K.; Rahmani, L.; Amirnezhad, F. Tomato powder is more effective than lycopene to alleviate exercise-induced lipid peroxidation in well-trained male athletes: randomized, double-blinded cross-over study. J. Int. Soc. Sports Nutr. 2021, 18, 17. [Google Scholar] [CrossRef] [PubMed]
- Natali, P.G.; Piantelli, M.; Minacori, M.; Eufemi, M.; Imberti, L. Improving whole tomato transformation for prostate health: benign prostate hypertrophy as an exploratory model. Int. J. Mol. Sci. 2023, 24, 5795. [Google Scholar] [CrossRef] [PubMed]
- Natali, P.G.; Piantelli, M.; Sottini, A.; Eufemi, M.; Banfi, C.; Imberti, L. A step forward in enhancing the health-promoting properties of whole tomato as a functional food to lower the impact of non-communicable diseases. Front. Nutr. 2025, 12, 1519905. [Google Scholar] [CrossRef]
- Aden, D.P.; Fogel, A.; Plotkin, S.; Damjanov, I.; Knowles, B.B. Controlled synthesis of HBsAg in a differentiated human liver carcinoma-derived cell line. Nature 1979, 282, 615–616. [Google Scholar] [CrossRef]
- Banfi, C.; Mussoni, L.; Risé, P.; Cattaneo, M.G.; Vicentini, L.; Battaini, F.; Galli, C.; Tremoli, E. Very low density lipoprotein-mediated signal transduction and plasminogen activator inhibitor type 1 in cultured HepG2 cells. Circ Res. 1999, 85, 208–217. [Google Scholar] [CrossRef]
- Rubini, E.; Minacori, M.; Paglia, G.; Macone, A.; Chichiarelli, S.; Altieri, F.; Eufemi, M. Tomato and olive bioactive compounds: A natural shield against the cellular effects induced by β-hexachlorocyclohexane-Activated Signaling Pathways. Molecules. 2021, 26, 7135. [Google Scholar] [CrossRef]
- Andus, T.; Bauer, J.; Gerok, W. Effects of cytokines on the liver. Hepatology 1991, 13, 364–375. [Google Scholar] [CrossRef]
- Kudlow, J.E.; Bjorge, J.D. TGF-alpha in normal physiology. Semin. Cancer Biol. 1990, 1, 293–302. [Google Scholar]
- Winkles, J.A. The TWEAK-Fn14 cytokine-receptor axis: discovery, biology and therapeutic targeting. Nat. Rev. Drug Discov. 2008, 7, 411–425. [Google Scholar] [CrossRef] [PubMed]
- Fausto, N. Liver regeneration and repair: Hepatocytes, progenitor cells, and stem cells. Hepatology 2004; 39, 1477-1487. [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Henry, L.; Paik, J.; Younossi, Z.M. Review article: the epidemiologic burden of non-alcoholic fatty liver disease across the world. Alimen. Pharmacol. Ther. 2022, 56, 942–956. [Google Scholar] [CrossRef]
- Goldner, D.; Lavine, J.E. Nonalcoholic Fatty Liver Disease in Children: Unique Considerations and Challenges. Gastroenterology 2020, 158, 1967–1983.e1. [Google Scholar] [CrossRef] [PubMed]
- Nobili. V.; Alisi, A.; Valenti, L.; Miele, L.; Feldstein, A.E.; Alkhouri, N. NAFLD in children: new genes, new diagnostic modalities and new drugs. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 517–530. [Google Scholar] [CrossRef]
- Haigh, L.; Kirk, C.; El Gendy, K.; Gallacher, J.; Errington, L.; Mathers, J.C. Anstee, Q.M. The effectiveness and acceptability of Mediterranean diet and calorie restriction in non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis. Clin. Nutr. 2022, 41, 1913–1931. [Google Scholar] [CrossRef]
- Hydes, T.J.; Ravi, S. , Loomba, R.; Gray, M.E. Evidence-based clinical advice for nutrition and dietary weight loss strategies for the management of NAFLD and NASH. Clin. Mol. Hepatol. 2020, 26, 383–400. [Google Scholar] [CrossRef]
- Pezzino, S.; Sofia, M.; Mazzone, C.; Litrico, G.; Greco, L.P.; Gallo, L.; La Greca, G.; Latteri, S. Innovative treatments for obesity and NAFLD: A bibliometric study on antioxidants, herbs, phytochemicals, and natural compounds. Heliyon 2024, 10, e35498. [Google Scholar] [CrossRef]
- Christensen, K.; Lawler, T.; Mares, J. Dietary carotenoids and Non-Alcoholic Fatty Liver Disease among US adults, NHANES 2003⁻2014. Nutrients 2019, 11, 1101. [Google Scholar] [CrossRef]
- Stice, C.P.; Xia, H.; Wang, X.D. Tomato lycopene prevention of alcoholic fatty liver disease and hepatocellular carcinoma development. Chronic Dis. Transl. Med. 2018, 4, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Donghia, R.; Campanella, A.; Bonfiglio, C.; Cuccaro, F.; Tatoli, R.; Giannelli, G. Protective Role of Lycopene in Subjects with Liver Disease: NUTRIHEP Study. Nutrients 2024, 16, 562. [Google Scholar] [CrossRef] [PubMed]
- Paetau, I.; Khachik, F.; Brown, E.D.; Beecher, G.R.; Kramer, T.R.; Chittams, J.; Clevidence, B.A. Chronic ingestion of lycopene-rich tomato juice or lycopene supplements significantly increases plasma concentrations of lycopene and related tomato carotenoids in humans. Am. J. Clin. Nutr. 1998, 68, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Vuong, T.; Ruckle, J.; Synal, H.A.; Schulze-König, T.; Wertz, K.; Rümbeli, R.; Liberman, R.G.; Skipper, P.L.; Tannenbaum, S.R.; et al. Lycopene bioavailability and metabolism in humans: an accelerator mass spectrometry study. Am. J. Clin. Nutr. 2011, 93, 1263–1273. [Google Scholar] [CrossRef]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D; Bohn, T. ; Cámara, M.; Castenmiller, J.; De Henauw, S.; Jos, Á.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; et al. Safety of yellow tomato extract as a novel food pursuant to Regulation (EU) 2015/2283. EFSA J. 2025, 23, e9373. [Google Scholar] [CrossRef]
- Petyaev, I.M. Lycopene Deficiency in Ageing and Cardiovascular Disease. Oxidative Med. Cell. Longev. 2016, 2016, 321805. [Google Scholar] [CrossRef]
- Hirose, A.; Terauchi, M.; Tamura, M.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Tomato juice intake increases resting energy expenditure and improves hypertriglyceridemia in middle-aged women: an open-label, single-arm study. Nutr. J. 2015, 14, 34. [Google Scholar] [CrossRef]
- Nishimura, M.; Tominaga, N.; Ishikawa-Takano, Y.; Maeda-Yamamoto, M.; Nishihira, J. Effect of 12-Week Daily Intake of the High-Lycopene Tomato (Solanum Lycopersicum), A Variety Named “PR-7”, on Lipid Metabolism: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study. Nutrients 2019, 11, 1177. [Google Scholar] [CrossRef]
- Zhou, R.; Zhu, X.; Xie, T.; Li, W.; Xie, D.; Zhang, G.; Xiao, Y.; Zhang, L. Amadori compounds (N-(1-Deoxy-D-fructos-1-yl)-amino acid): The natural transition metal ions (Cu2+, Fe2+, Zn2+) chelators formed during food processing. LWT 2024, 191, 115600. [Google Scholar] [CrossRef]
- Pannellini, T.; Iezzi, M.; Liberatore, M.; Sabatini, F.; Iacobelli, S.; Rossi, C.; Alberti, S.; Di Ilio, C.; Vitaglione, P.; Fogliano, V.; et al. A dietary tomato supplement prevents prostate cancer in TRAMP mice. Cancer Prev. Res. 2010, 3, 1284–1291. [Google Scholar] [CrossRef]
- Arzumanian, V.A.; Kiseleva, O.I.; Poverennaya, E.V. The Curious Case of the HepG2 Cell Line: 40 Years of Expertise. Int. J. Mol. Sci. 2021, 22, 13135. [Google Scholar] [CrossRef]
- Park, S-J. ; Garcia Diaz, J.; Um, E.; Hahn, Y.S. Major roles of kupffer cells and macrophages in NAFLD development. Front. Endocrinol. 2023, 14, 1150118. [Google Scholar]
- Wiering, L.; Subramanian, P.; Hammerich. L. Hepatic Stellate Cells: Dictating Outcome in Nonalcoholic Fatty Liver Disease. Cell. Mol. Gastroenterol. Hepatol. 2023, 15, 1277–1292. [Google Scholar] [CrossRef]
- Moayedfard, Z.; Sani, F.; Alizadeh, A.; Bagheri Lankarani, K.; Zarei, M.; Azarpira, N. The role of the immune system in the pathogenesis of NAFLD and potential therapeutic impacts of mesenchymal stem cell-derived extracellular vesicles. Stem Cell Res. Ther. 2022, 13, 242. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Cohen, D.E. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J. Gastroenterol. 2013, 48, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.E.; Ramos-Roman, M.A.; Browning, J.D.; Parks, E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 2014, 146, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Alves-Bezerra, M.; Cohen, DE. Triglyceride Metabolism in the Liver. Compr. Physiol. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- Belloni, L.; Di Cocco, S.; Guerrieri, F.; Nunn, A.D.G.; Piconese, S.; Salerno, D.; Testoni, B.; Pulito, C.; Mori, F.; Pallocca, M.; et al. Targeting a phospho-STAT3-miRNAs pathway improves vesicular hepatic steatosis in an in vitro and in vivo model. Sci. Rep. 2018, 8, 13638. [Google Scholar] [CrossRef]
- Lee, J.H.; Wada, T.; Febbraio, M.; He, J.; Matsubara, T.; Lee, M.J.; Gonzalez, F.J.; Xie, W. A novel role for the dioxin receptor in fatty acid metabolism and hepatic steatosis. Gastroenterology 2010, 139, 653–663. [Google Scholar] [CrossRef]
- Kimura, M.; Moteki, H.; Ogihara, M. Role of hepatocyte growth regulators in liver regeneration. Cells 2023, 12, 208. [Google Scholar] [CrossRef]
- Deng, Y.Q.; Zhao, H.; Ma, A.L.; Zhou, J.Y.; Xie, S.B.; Zhang, X.Q.; Zhang, D.Z.; Xie. Q.; Zhang, G.; Shang, J. et al. Selected cytokines serve as potential biomarkers for predicting liver inflammation and fibrosis in chronic hepatitis b patients with normal to mildly elevated aminotransferases. Medicine (Baltimore) 2015, 94, e2003. [Google Scholar] [CrossRef]
- Tirnitz-Parker, J.E.; Viebahn, C.S.; Jakubowski, A.; Klopcic, B.; Olynyk, J.; Yeoh, G.; Knight, B. Tumor necrosis factor–like weak inducer of apoptosis is a mitogen for liver progenitor cells. Hepatology 2010, 52, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Suppli, M.P.; Rigbolt, K.T.G.; Veidal, S.S.; Heebøll, S.; Eriksen, P.L.; Demant, M.; Bagger, J.I.; Nielsen, J.C.; Oró, D.; Thrane, S.W. Hepatic transcriptome signatures in patients with varying degrees of nonalcoholic fatty liver disease compared with healthy normal-weight individuals. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G462–G472. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Yang, Y.; Wu, S.; Li, W. TWEAK promotes inflammatory response in liver fibrosis. J. Biochem. Mol. Toxicol. 2023, 37, e23483. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, A.; Shepherd, E.L.; Amatucci, A.; Munir, M.; Reynolds, G.; Humphreys, E.; Resheq, Y.; Adams, D.H.; Hübscher, S.; Burkly, L.C. Interaction of TWEAK with Fn14 leads to the progression of fibrotic liver disease by directly modulating hepatic stellate cell proliferation. J. Pathol. 2016, 239, 109–121. [Google Scholar] [CrossRef]
- Ramachandran, P.; Dobie, R.; Wilson-Kanamori, J.R.; Dora, E.F.; Henderson, B.E.P.; Luu, N.T.; Portman, J.R.; Matchett, K.P.; Brice, M.; Marwick, J.A.; et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level. Nature 2019, 575, 512–518. [Google Scholar] [CrossRef]
- Xiao, M.H.; Ma, D.; Wu, S.; Huang, Z.; Liang, P.; Chen, H.; Zhong, Z.; Li, W.; Wang, F.; Tang, Y.; et al. Integrative single-cell and spatial transcriptomic analyses identify a pathogenic cholangiocyte niche and TNFRSF12A as therapeutic target for biliary atresia. Hepatology 2025, 81, 1146–1163. [Google Scholar] [CrossRef]
- Huang, T.; Zheng, D.; Song, Y.; Pan, H.; Qiu, G.; Xiang, Y.; Wang, Z.; Wang, F. Demonstration of the impact of COVID-19 on metabolic associated fatty liver disease by bioinformatics and system biology approach. Medicine (Baltimore) 2023, 102, e34570. [Google Scholar] [CrossRef]
- Voronov, I.; Manolson, M.F. Editorial: Flt3 ligand-friend or foe? J. Leukoc. Biol. 2016, 99, 401–403. [Google Scholar] [CrossRef]
- Rahman, A.H. , Aloman, C. Dendritic cells and liver fibrosis. Biochim. Biophys. Acta. 2013, 1832, 998–1004. [Google Scholar] [CrossRef]
- Lovisa, S. Epithelial-to Mesenchymal Transition in Fibrosis: Concepts and Targeting Strategies. Front. Pharmacol. 2021, 12, 737570. [Google Scholar] [CrossRef]
- Daniello, V.; De Leo, V.; Lasalvia, M.; Hossain, M.N.; Carbone, A.; Catucci, L.; Zefferino, R.; Ingrosso, C.; Conese, M.; Di Gioia, S. Solanum lycopersicum (Tomato)-derived nanovesicles accelerate wound healing by eliciting the migration of keratinocytes and fibroblasts. Int. J. Mol. Sci. 2024, 25, 2452. [Google Scholar] [CrossRef] [PubMed]
- McEneny, J.; Henry, S.-L.; Woodside, J.; Moir, S.; Rudd, A.; Vaughan, N.; Thies. F. Lycopene-rich diets modulate HDL functionality and associated inflammatory markers without aecting lipoprotein size and distribution in moderately overweight, disease-free, middle-aged adults: A randomized controlled trial. Front. Nutr. 2022, 9, 954593. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Sanchez, J.I.; Saldarriaga, O.A.; Solis, L.M.; Tweardy, D.J.; Maru, D.M.; Stevenson, H. L.; Beretta, L. Spatial molecular and cellular determinants of STAT3 activation in liver fibrosis progression in non-alcoholic fatty liver disease. JHEP Rep. 2022, 5, 100628. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, N.; Plaz Torres, M.C.; Petta, S.; Valenti, L.; Giannini, E.G.; Aghemo, A. Is there an ‘ideal’ diet for patients with NAFLD? Eur. J. Clin. Invest. 2022, 52, e13659. [Google Scholar] [CrossRef]
- Kiani, A.K.; Medori, M.C.; Bonetti, G.; Aquilanti, B.; Velluti, V.; Matera, G.; Iaconelli, A.; Stuppia, L.; Connelly, S.T.; Herbst, K.L.; et al. Modern vision of the Mediterranean diet. J. Prev. Med. Hyg. 2022, 63, E36–E43. [Google Scholar]
- Caparello, G.; Galluccio, A.; Giordano, C.; Lofaro, D.; Barone, I.; Morelli, C.; Sisci, D.; Catalano, S.; Andò, S.; Bonofiglio, D. Adherence to the Mediterranean diet pattern among university staff: a cross-sectional web-based epidemiological study in Southern Italy. Int. J. Food Sci. Nutr. 2020, 71, 581–592. [Google Scholar] [CrossRef]
- Leh, H.E.; Lee, L.K. Lycopene: a potent antioxidant for the amelioration of type II diabetes mellitus. Molecules 2022, 27, 2335. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




