Submitted:
27 July 2025
Posted:
28 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
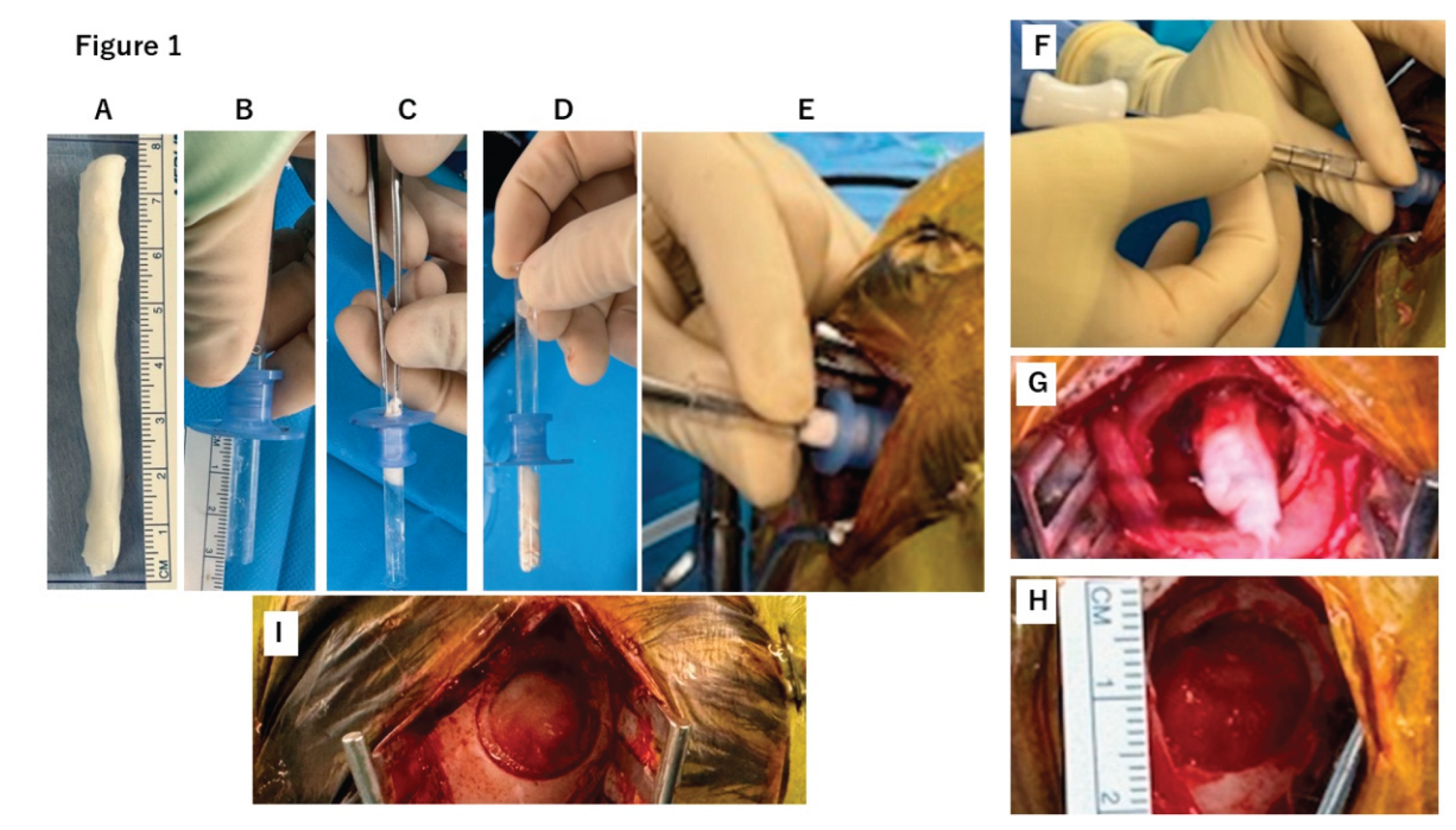
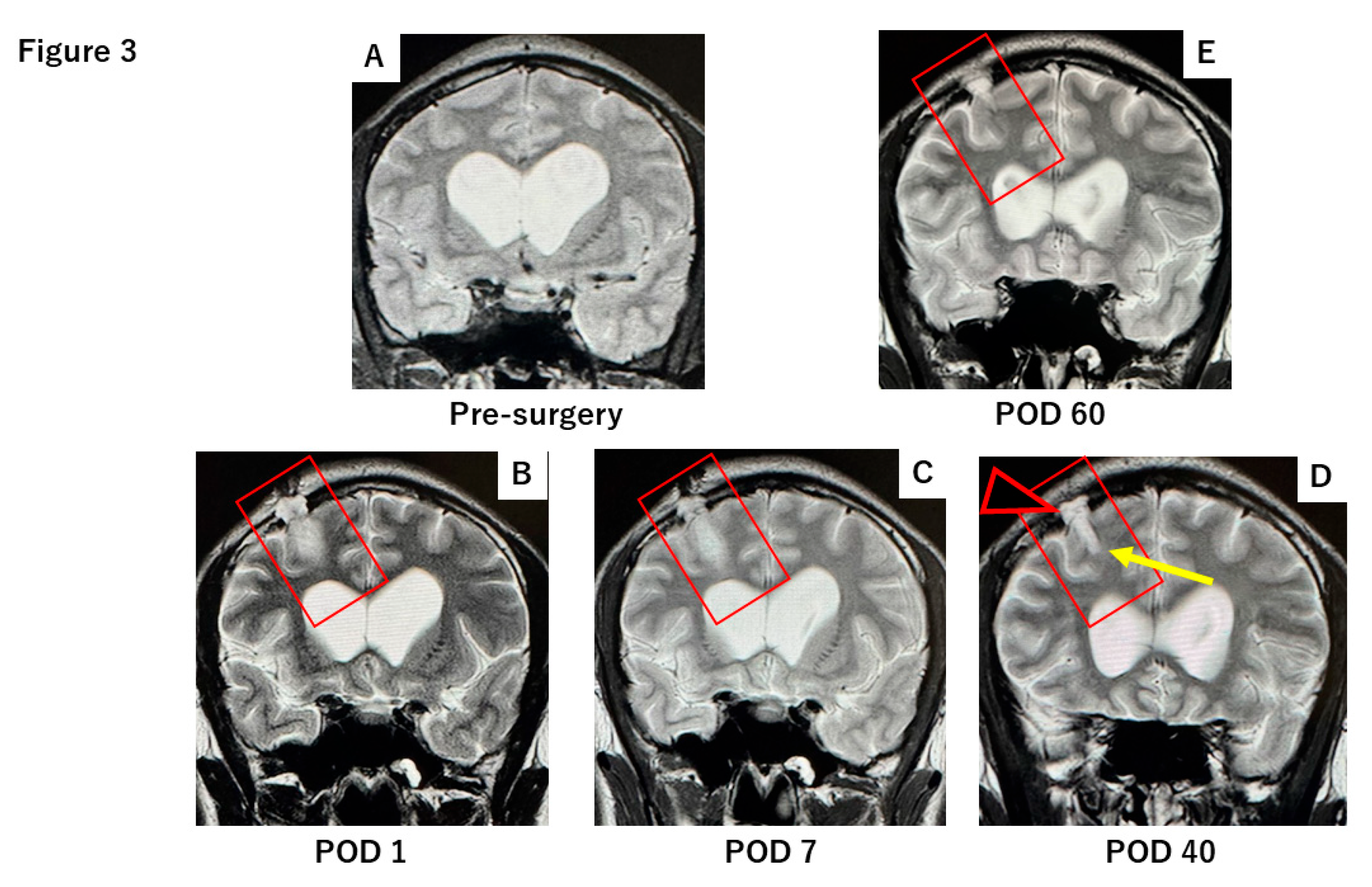
2.1. Case Report
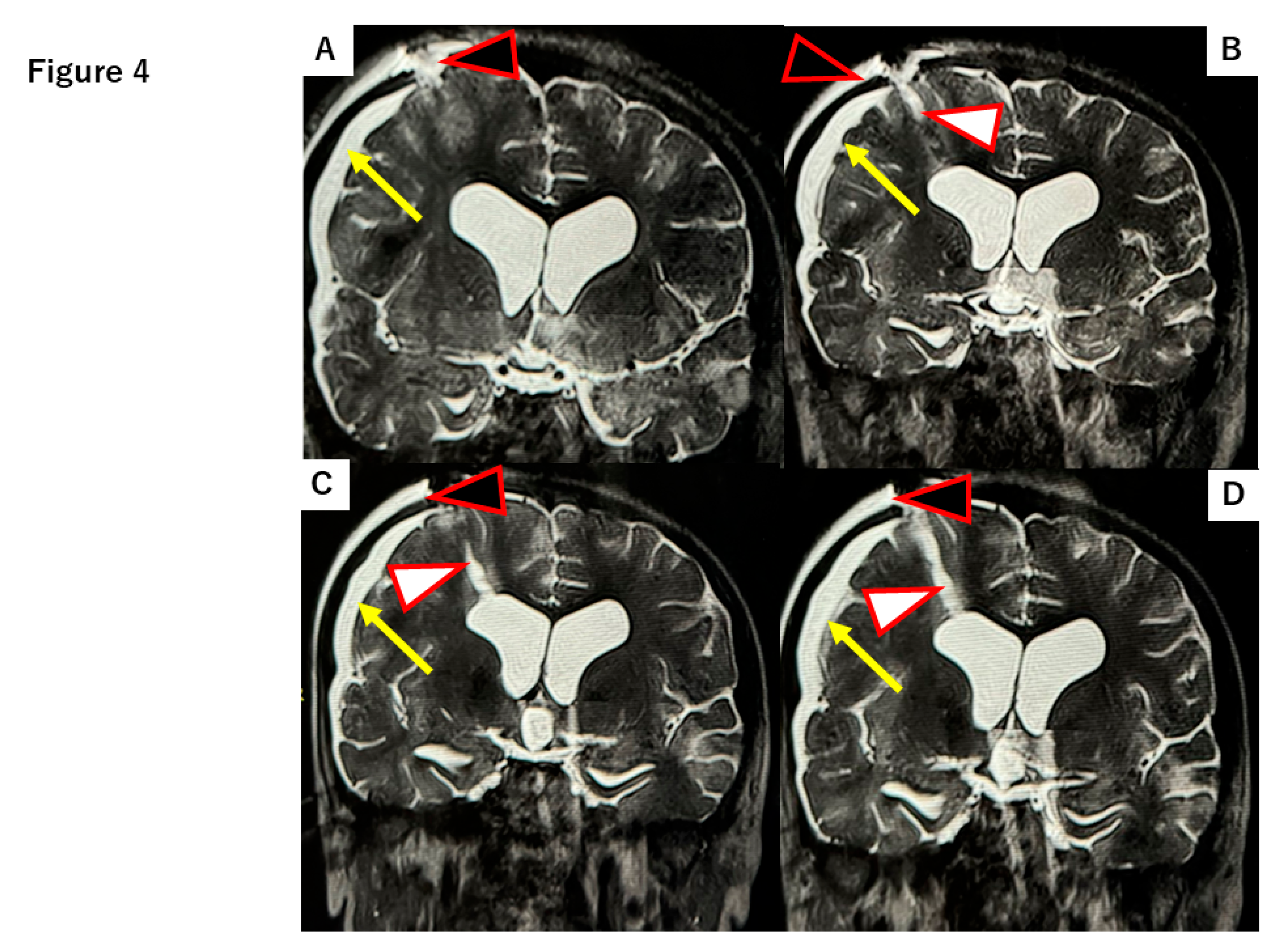
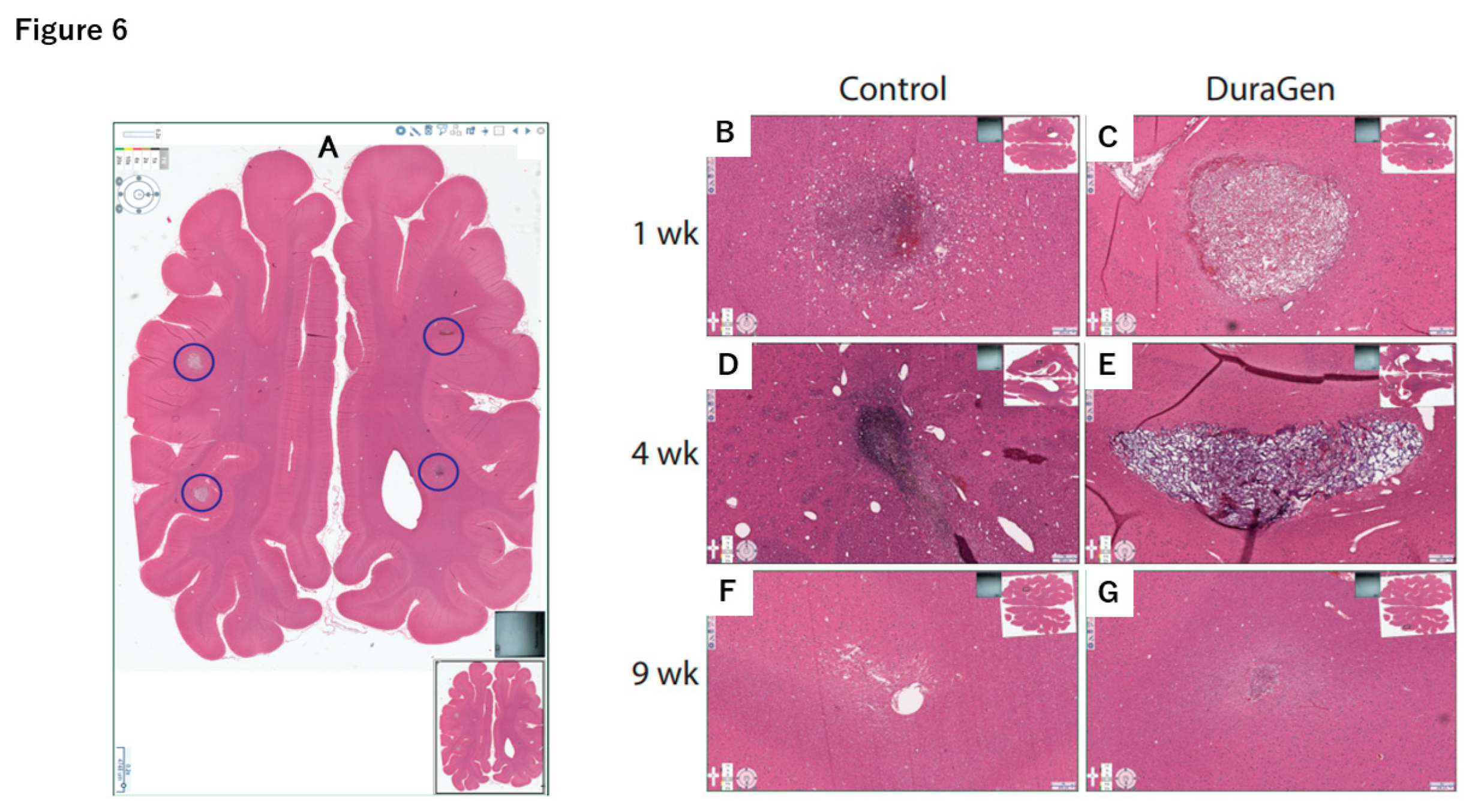
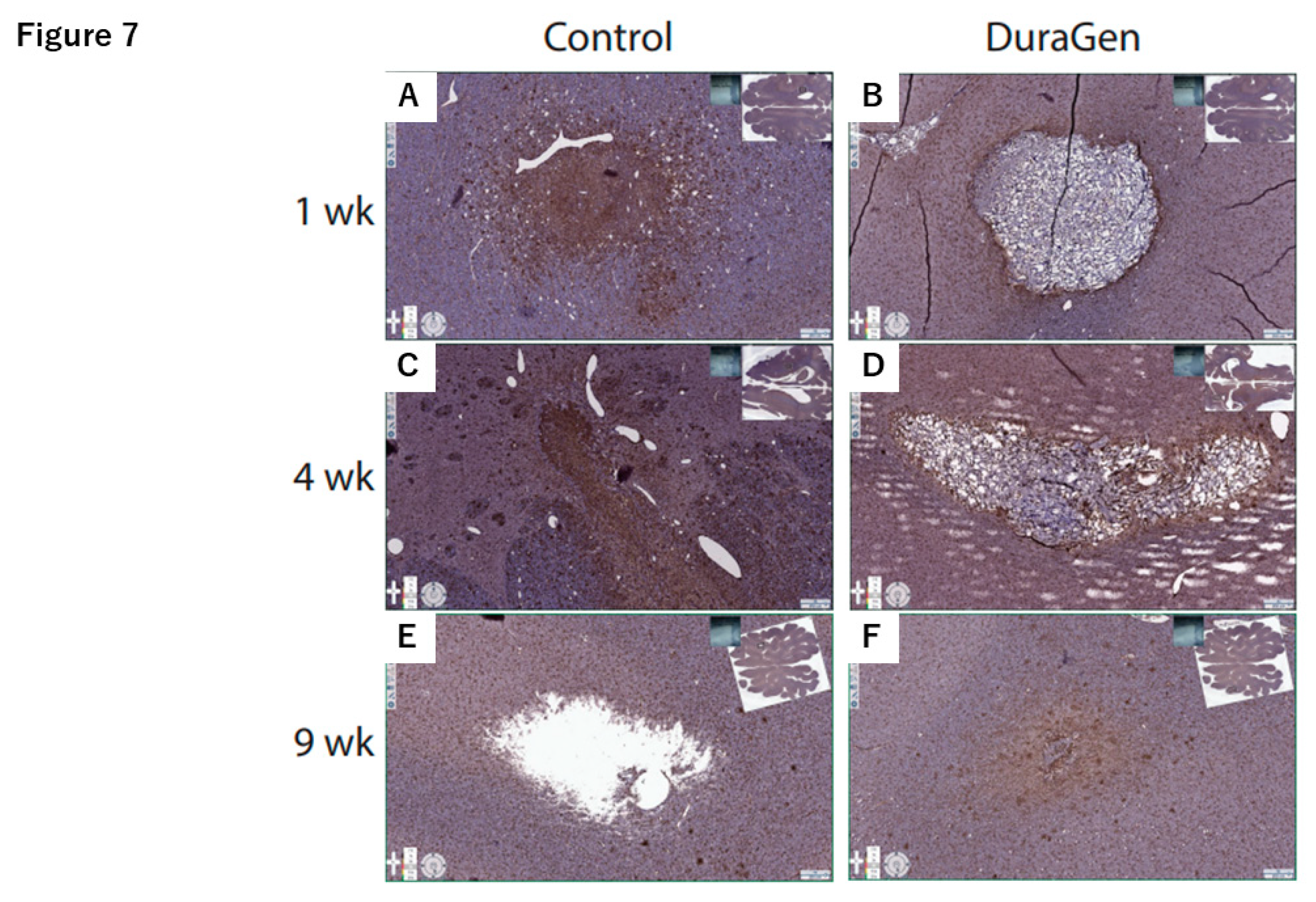
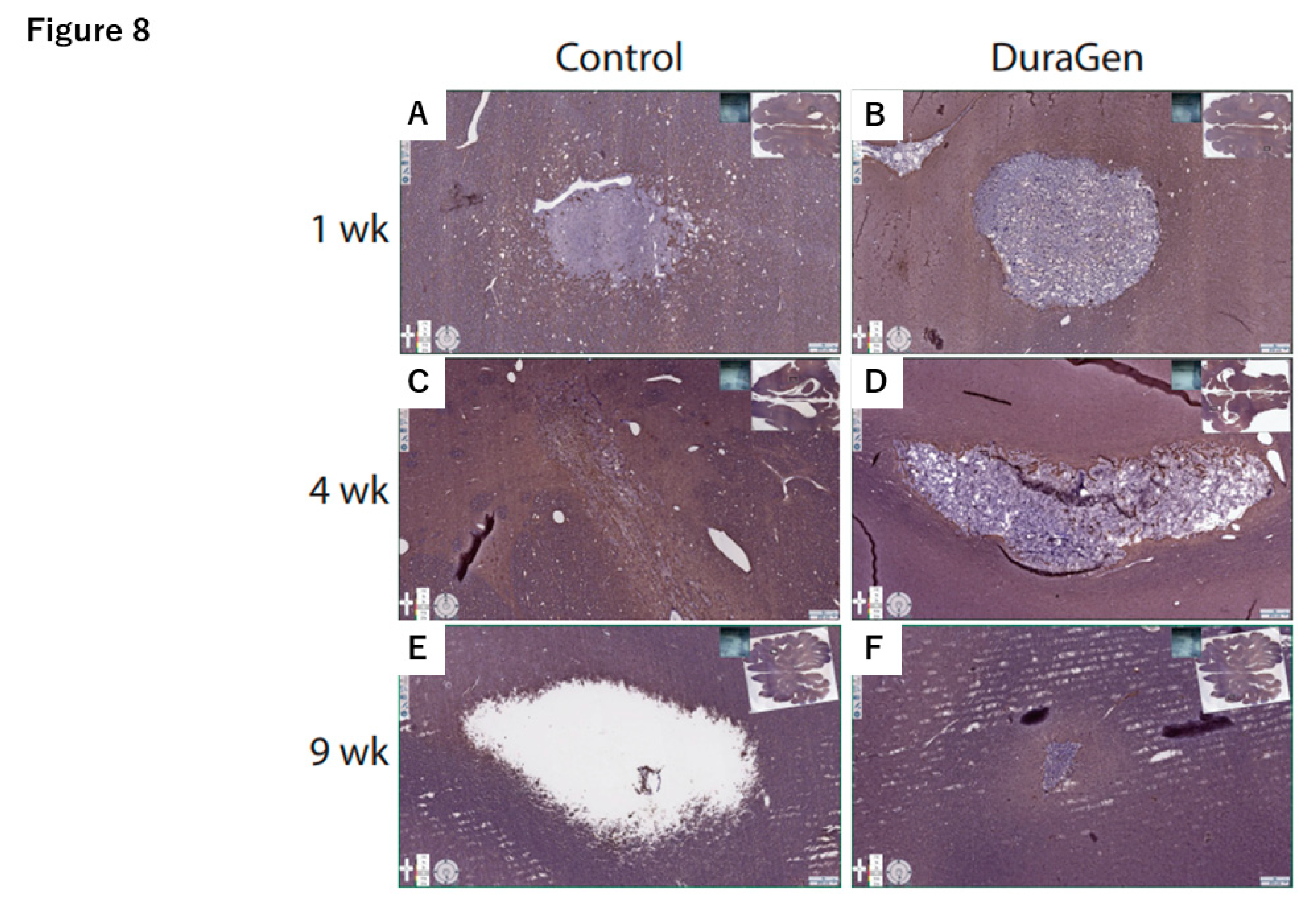
2.2. DuraGen Implant in Porcine Brain Parenchyma
3. Discussion
4. Materials and Methods
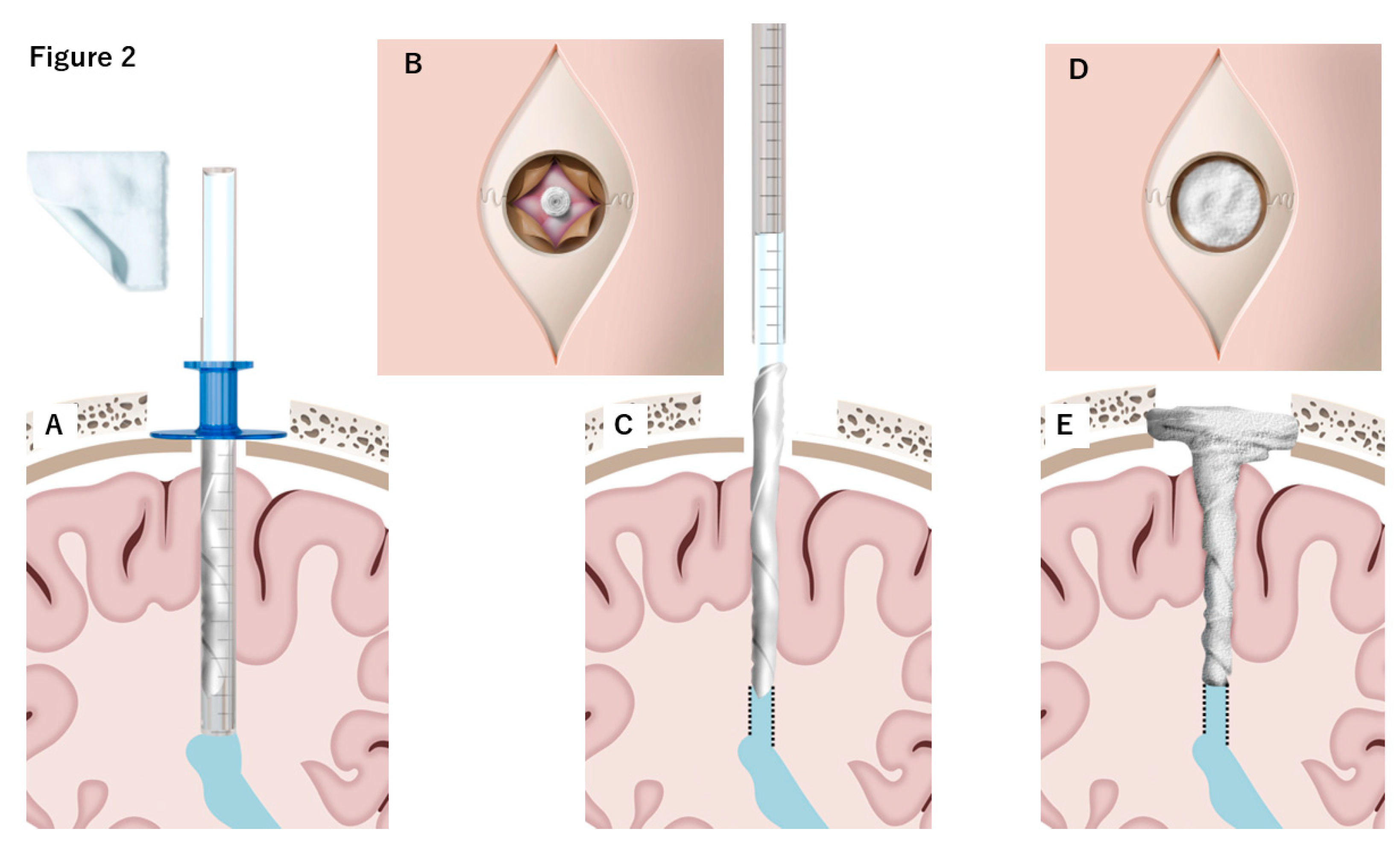
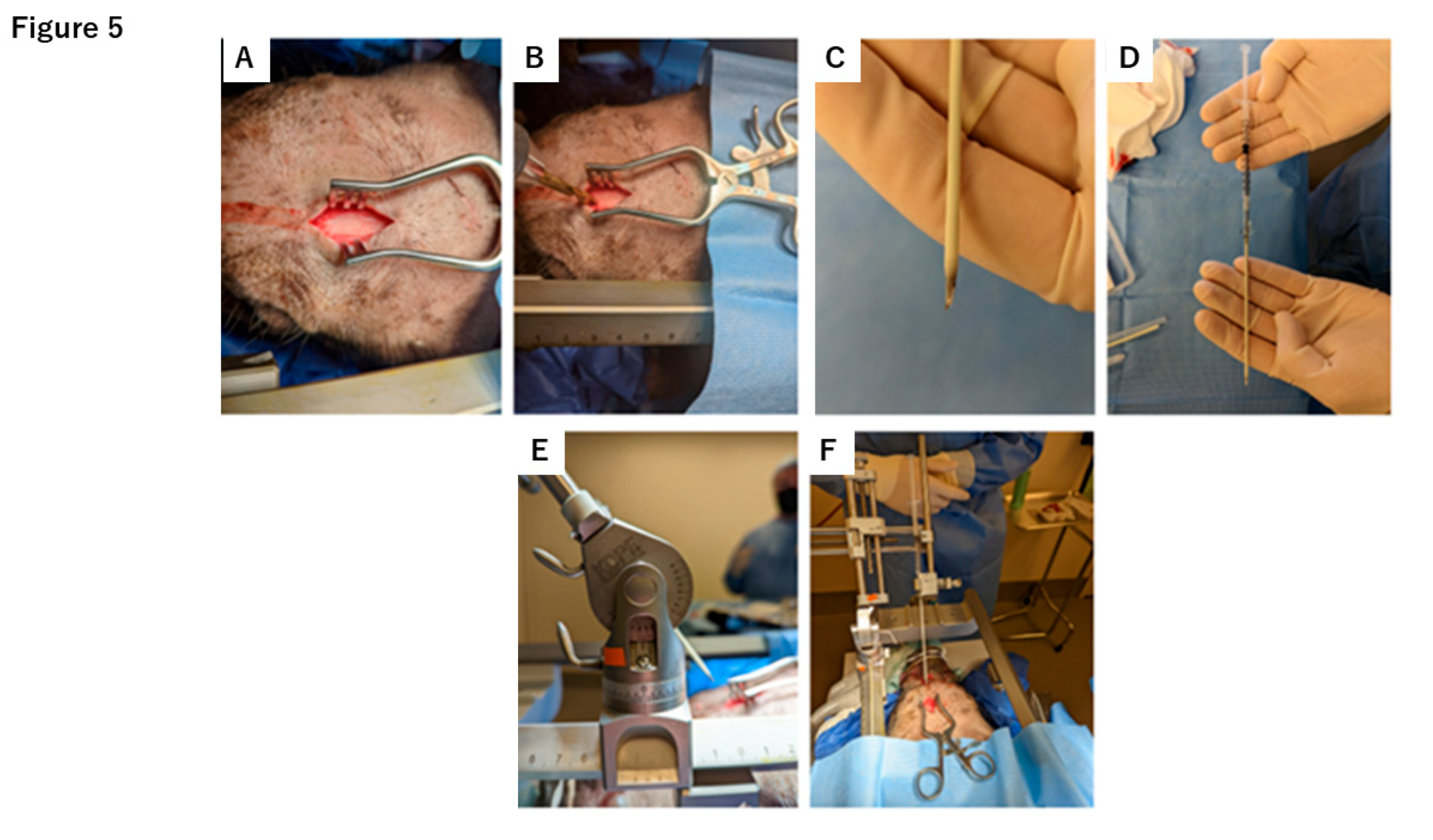
4.1. DuraGen Insertion Technique
4.2. DuraGen Implantation into Pig Brain Parenchyma
4.3. Histology
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishihara T, Teraoka A, Morita A, Ueki K, Kirino T. Transparent sheath for endoscopic surgery and its application in surgical evacuation of spontaneous intracerebral hematomas (technical note). J Neurosurg. 2000;92(6):1053–1055. [CrossRef]
- Nishihara T, Morita A, Teraoka A, Kirino T. Endoscopy-guided removal of spontaneous intracerebral hemorrhage: comparison with CT-guided stereotactic evacuation. Childs Nerv Syst. 2007;23(6):677–683. [CrossRef]
- Oi S, Abdullah SH. New transparent peel-away sheath with neuroendoscopic orientation markers (technical note). J Neurosurg. 2007;107(6):1244–1247. [CrossRef]
- Selesnick SH, Liu JC, Jen A, Newman J. The incidence of cerebrospinal fluid leak after vestibular schwannoma surgery. Otol Neurotol. 2004;25(3):387–393. [CrossRef]
- Graupman P, Nussbaum ES, Patel PD. Preventing cerebrospinal fluid leakage following endoscopy through a burr hole using a novel watertight closure (technical note). Br J Neurosurg. 2023;37(6):1915–1917. [CrossRef]
- Jung TY, Chong S, Kim IY, Lee JY, Phi JH, Kim SK. Prevention of complications in endoscopic third ventriculostomy. J Korean Neurosurg Soc. 2017;60(3):282–288. [CrossRef]
- Lo WB, Afshari FT, Rodrigues D, Kulkami AV. The “mushroom”: a simple and safe technique to avoid cerebrospinal fluid leak after endoscopic third ventriculostomy. Ann R Coll Surg Engl. 2020;102(4):312–319. [CrossRef]
- Oi S, Samii A, Samii M. Frameless free-hand maneuvering of a small-diameter rigid-rod neuroendoscope with a working channel used during high-resolution imaging (technical note). J Neurosurg Pediatr. 2005;102(1):113–118. [CrossRef]
- Integra LifeSciences Corporation. DuraGen® Collagen Matrix: Technical Information. Integra LifeSciences; 2019. Available at: https://www.cardion.cz/file/90/duragen-ultrapure-collagen.pdf.
- Eguchi S, Aihara Y, Hori T, Okada Y. Postoperative extra-axial cerebrospinal fluid collection—its pathophysiology and clinical management. Pediatr Neurosurg. 2011;47(2):125–132. [CrossRef]
- Peretta P, Ragazzi P, Galarza M, et al. Complications and pitfalls of neuroendoscopic surgery in children. J Neurosurg. 2006; 105:187-193. [CrossRef]
- Cohen, AR. Cohen AR. Endoscopic ventricular surgery. Pediatr Neurosurg. 1993;19(3):127–134. [CrossRef]
- Fries G, Perneczky A. Endoscope-assisted brain surgery: part 2 – analysis of 380 procedures. Neurosurgery. 1998;42(2):226–232. [CrossRef]
- Bouras T, Sgouros S. Complications of endoscopic third ventriculostomy: a review. J Neurosurg Pediatr. 2011;7(6):643–649. [CrossRef]
- Kulkarni AV, Riva-Cambrin J, Holubkov R, Browd SR, Cochrane DD, Drake JM. Endoscopic third ventriculostomy in children: prospective, multicenter results from the Hydrocephalus Clinical Research Network. J Neurosurg Pediatr. 2016;18(1):1–7. [CrossRef]
- Lin B, Yang H, Cui M,Yu J. Surgicel™ application in intracranial hemorrhage surgery contributed to giant-cell granuloma in a patient with hypertension: case report and review of the literature. World J Surg Oncol. 2014;12:101. [CrossRef]
- Lei T, Liu X, Cao J, Sun Y, Li G, Huang H. Intracranial foreign body granuloma caused by gelatin sponge: a case report and literature review. Int J Clin Exp Med. 2017;10(2):3996–4000. ISSN: 1940-5901/IJCEM0042191.
- Nagata Y, Takeuchi K, Sasaki H, Mizuno H, Tanahashi K. Modified shoelace dural closure with collagen matrix in extended transsphenoidal surgery. Neurol Med Chir (Tokyo). 2022;62(4):203–208. [CrossRef]
- Inoue T, Shitara S, Shima A, Goto Y, Fukushima T. Double collagen matrix grafting for dural closure in microvascular decompression: an alternative to autologous fascial graft. Acta Neurochir (Wien). 2021;163(10):2395–2401. [CrossRef]
- Koyama J, Akutsu N, Kawamura A. Duraplasty using a combination of a pedicled dural flap and collagen matrix in posterior fossa decompression for pediatric Chiari malformation type I with syrinx. Acta Neurochir (Wien). 2024;166(1):70. [CrossRef]
- Narotam PK, Reddy K, Fewer D, Qiao F, Nathoo N. Collagen matrix duraplasty for cranial and spinal surgery: a clinical and imaging study. J Neurosurg. 2007;106(1):45–51. [CrossRef]
- Narotam PK, Qiao F, Nathoo N. Collagen matrix duraplasty for posterior fossa surgery: evaluation of surgical technique in 52 adult patients. J Neurosurg. 2009;111(2):380–386. [CrossRef]
- Yannas IV, Lee E, Orgill DP, Skrabut EM, Murphy GF. Synthesis and characterization of a model extracellular matrix that induces partial regeneration of adult mammalian skin. Proc Natl Acad Sci U S A. 1989;86(3):933–937. [CrossRef]
- Narotam PK, Van Dellen JR, Bhoola K, Raidoo D. Experimental evaluation of collagen sponge as a dural graft. Br J Neurosurg. 1993;7(6):635–641. [CrossRef]
- Narotam PK, Van Dellen JR, Bhoola KD. A clinicopathological study of collagen sponge as a dural graft in neurosurgery. J Neurosurg. 1995;82(3):406–412. [CrossRef]
- Shaffrey CI, Spotnitz WD, Shaffrey ME. Neurosurgical applications of fibrin glue: augmentation of dural closure in 134 patients. Neurosurgery. 1990;26(2):207–210.
- Stendel R, Danne M, Fiss I, et al. Efficacy and safety of a collagen matrix for cranial and spinal dural reconstruction using different fixation techniques. J Neurosurg. 2008;109(2):215–221. [CrossRef]
- Tamura R, Kuranari Y, Orikasa H, Katayama M. Meningioma cell invasion into DuraGen-derived dura mater: a case report. Medicines (Basel). 2022;9(4):30. [CrossRef]
- Danish SF, Samdani A, Hanna A,Storm P, Sutton L. Experience with acellular human dura and bovine collagen matrix for duraplasty after posterior fossa decompression for Chiari malformations. J Neurosurg. 2006;104(1):16–20. [CrossRef]
- Neulen A, Gutenberg A, Takács I, Weber G, Wegmann J, Schulz-Schaeffer W. Evaluation of efficacy and biocompatibility of a novel semisynthetic collagen matrix as a dural onlay graft in a large animal model. Acta Neurochir (Wien). 2011;153(11):2241–2250. [CrossRef]
- Ellingsworth LR, DeLustro F, Brennan JE, Sawamura S, Mcpherson J. The human immune response to reconstituted bovine collagen. J Immunol. 1986;136(3):877–882.
- Jinnin, M. Jinnin M. [An experimental study on the prevention of postlaminectomy scar formation: efficacy of interpositional collagen-sponge]. Nippon Seikeigeka Gakkai Zasshi. 1995;69(2):198–208. (Japanese).
- Calikoglu C, Cakir M, Tuzun Y. Histopathological investigation of the effectiveness of collagen matrix in the repair of experimental spinal dura mater defects. Eurasian J Med. 2019;51(2):133–138. [CrossRef]
- Narotam PK, José S, Nathoo N, Taylon C, Vora Y. Collagen matrix (DuraGen) in dural repair: analysis of a modified technique. Spine (Phila Pa 1976). 2004;29(24):2861–2867; discussion 2868–2869. [CrossRef]
- Haq I, Cruz-Almeida Y, Siqueira EB, Norenberg M, Green BA, Levi AD. Postoperative fibrosis after surgical treatment of the porcine spinal cord: a comparison of dural substitutes. J Neurosurg Spine. 2005;2(1):50–5. [CrossRef]
- Khorasani L, Kapur RP, Lee C, Avellino M. Histological analysis of DuraGen in a human subject: case report. Clin Neuropathol. 2008;27(5):361–364. [CrossRef]
- Esposito F, Grimod G, Cavallo LM, Lanterna L, Biroli F, Cappabianca P. Collagen-only biomatrix as dural substitute: results at 5-year follow-up. Clin Neurol Neurosurg. 2013;115(9):1735–1737. [CrossRef]
- Sade B, Oya S, Lee JH. Non-watertight dural reconstruction in meningioma surgery: results in 439 consecutive patients and a review of the literature. J Neurosurg. 2011;114(3):714–718. [CrossRef]
- Zerris VA, James KS, Roberts JB, Bell E, Heilman B. Repair of the dura mater with processed collagen devices. J Biomed Mater Res B Appl Biomater. 2007;83(2):580–588. [CrossRef]
- Aksekili MAE, Ateş O, Yüksel K, et al. The comparison of histopathological results of repairing dural defects in rats using different collagen-based grafts. Electron J Biol. 2017;13(2):188–194. ISSN 1860-3122.
- Petrov D, Katiyar KS, Struzyna LA, Harris JP, Cullen DK. Extracellular matrix-derived tissues for neurological applications. In: Mooradian DL, ed. Extracellular Matrix–Derived Implants in Clinical Medicine. Woodhead Publishing: Sawston, Cambridge, UK; 2016:83–118. [CrossRef]
- Rabinowitz L, Monnerie H, Shashidhara S, Le Roux P. Growth of rat cortical neurons on DuraGen, a collagen-based dural graft matrix. Neurol Res. 2005;27(8):887–894. [CrossRef]
- Finch L, Harris S, Solomou G, Sen J, Tzerakis N, Emes RD. Safe nanoengineering and incorporation of transplant populations in a neurosurgical-grade biomaterial (DuraGen Plus™) for protected cell therapy applications. J Control Release. 2020;321:553-563. [CrossRef]
- Kameo K, Kaku Y, Ohmori Y, Takemoto Y, Uekawa K, Mukasa A. Artificial dural regeneration matrix as a substitute for autologous tissue in indirect bypass in Moyamoya disease: Investigation of a rat model of chronic cerebral hypoperfusion. Neurosurg Rev. 2025;48(1):48. [CrossRef]
- Maeda T, Suzuki K, Nisson PL, et al. Histopathological analysis of Duragen collagen matrix over time in humans. Sci Rep. 2025;15(1):11119. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



