Submitted:
22 July 2025
Posted:
23 July 2025
You are already at the latest version
Abstract
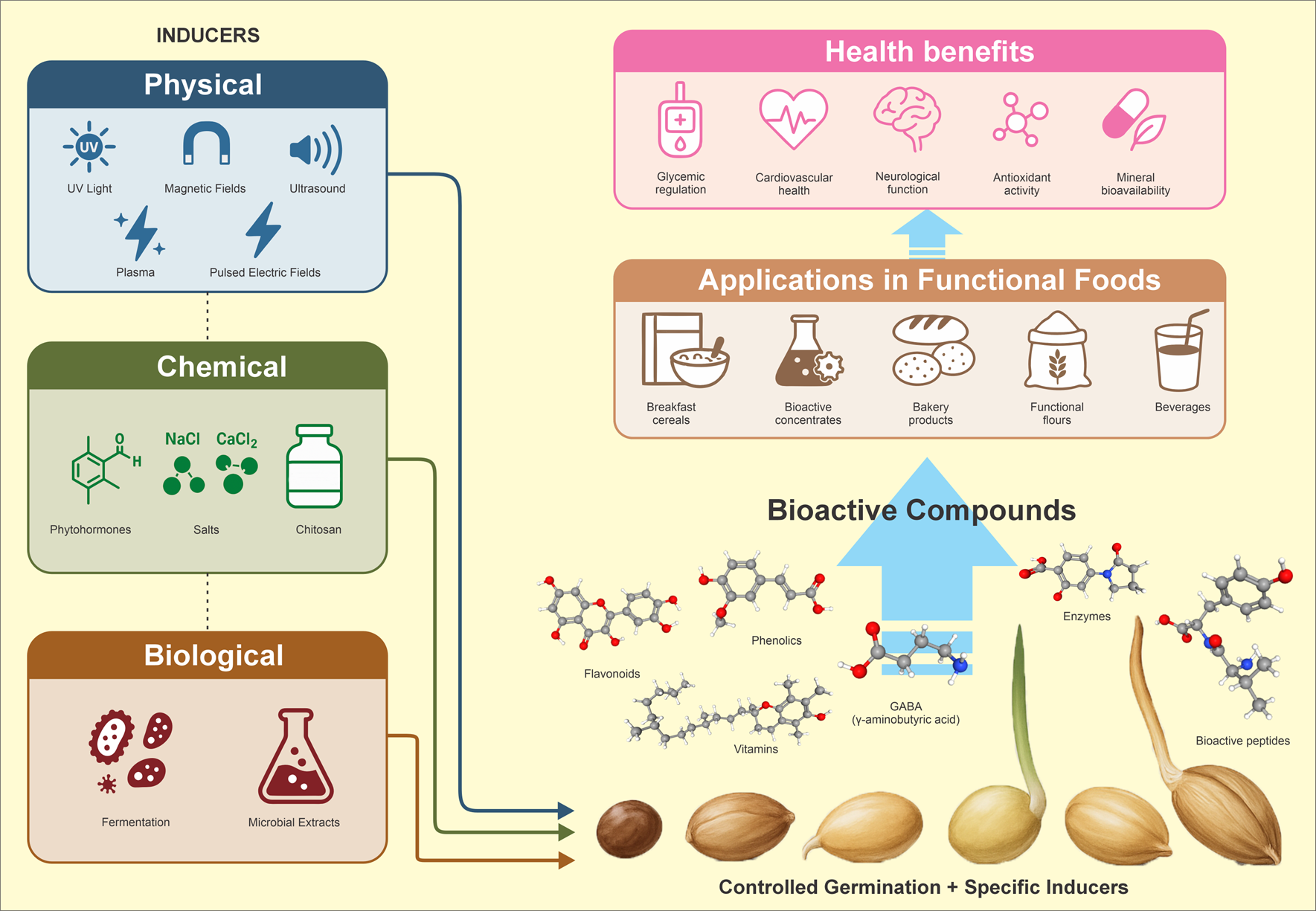
Keywords:
1. Introduction
2. Search Strategies and Brief Bibliometric Analysis
3. Fundamentals of Germination and Its Impact on Bioactive Compounds
3.1. Germination Process: Physiological and Biochemical Aspects
3.2. Main Bioactive Compounds in Germinated Seeds
3.1.1. Phenolic Compounds
3.1.2. GABA (γ-Aminobutyric Acid)
3.1.3. Bioactive Peptides
3.1.4. Melatonin and Indolic Compounds
3.1.5. Vitamins and Bioavailable Minerals
3.1.6. Antioxidant Enzymes
3.1.7. Diverse Phytochemicals
3.1.8. Dietary Fiber
4. Factors Influencing the Accumulation of Bioactive Compounds
4.1. Genetic Factors
4.2. Environmental Conditions During Growth
4.3. Germination Process Parameters
4.4. Other Processing Treatments
4.5. Abiotic Stress
5. Physical Inducers of Germination
5.1. Controlled Germination
5.2. Plasma Activated Water (PAW) Treatments
5.3. High Hydrostatic Pressure (HHP) Treatments
5.4. Pulsed Electric Fields (PEF)
5.5. High Voltage Electric Fields (HVEFs)
5.6. Magnetic Fields
5.7. High Pressure Carbon Dioxide (HPCD)
5.8. Microwave Irradiation
5.9. Light Intensity
5.10. Pulsed Light (PL)
5.11. Ultraviolet (UV) Radiation
5.12. Cold Atmospheric Plasma
5.13. Ultrasonication
6. Chemical Inducers of Germination
6.1. Plant-Derived Inducers
6.2. Minerals and Trace Elements
6.3. Plant Growth Regulators
6.4. Synthetic Chemical Inducers
6.5. Nanomaterials
7. Biological Inducers of Germination
7.1. Fermentation Concurrent with Germination
8. Combination of Inducers and Integrated Approaches
8.1. Synergies Between Physical and Biological Inducers
8.2. Synergies Between Physical and Chemical Inducers
8.3. Synergies Between Physical Inducers
9. Applications in the Food Industry and Technological Considerations
9.1. Functional Flours
9.2. Functional Bakery Products
9.3. Functional Breakfast Cereals and Snacks
9.4. Functional Beverages
9.5. Fermented Foods
9.6. Bioactive Concentrates
9.7. Functional Foods for Glycemic Control
9.8. Infant Foods
9.9. Foods with Improved Bioavailability
9.10. Functional Malted Products
10. Challenges and Technological Considerations
11. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Benincasa, P.; Falcinelli, B.; Lutts, S.; Stagnari, F.; Galieni, A. Sprouted Grains: A Comprehensive Review. Nutrients 2019, 11, 421. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, E.; Moroni, A.V.; Pagand, J.; Heirbaut, P.; Ritala, A.; Karlen, Y.; Lê, K.; Van Den Broeck, H.C.; Brouns, F.J.P.H.; De Brier, N.; et al. Impact of Cereal Seed Sprouting on Its Nutritional and Technological Properties: A Critical Review. Comp Rev Food Sci Food Safe 2019, 18, 305–328. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.; Hussain, M.B.; Waheed, M.; Ahmad, K.; Kassymov, S.; Shariati, M.A.; Akram, M.; Mishra, A.P.; Egbuna, C. Effect of Germination Processing on Bioactive Compounds of Cereals and Legumes. In Functional Foods and Nutraceuticals; Egbuna, C., Dable Tupas, G., Eds.; Springer International Publishing: Cham, 2020; ISBN 978-3-030-42318-6. [Google Scholar]
- Ding, J.; Hou, G.G.; Nemzer, B.V.; Xiong, S.; Dubat, A.; Feng, H. Effects of Controlled Germination on Selected Physicochemical and Functional Properties of Whole-Wheat Flour and Enhanced γ-Aminobutyric Acid Accumulation by Ultrasonication. Food Chemistry 2018, 243, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Johnson, J.; Chu, Y.F.; Feng, H. Enhancement of γ-Aminobutyric Acid, Avenanthramides, and Other Health-Promoting Metabolites in Germinating Oats (Avena Sativa L.) Treated with and without Power Ultrasound. Food Chemistry 2019, 283, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Yang, J.; Zhang, J.; Fang, W.; Yin, Y. Physiology and Metabolism Alterations in Flavonoid Accumulation During Buckwheat (Fagopyrum Esculentum Moench.) Sprouting. Plants 2024, 13, 3342. [Google Scholar] [CrossRef] [PubMed]
- Jan, R.; Saxena, D.C.; Singh, S. Effect of Germination on Nutritional, Functional, Pasting, and Microstructural Properties of Chenopodium (Chenopodium Album) Flour: Gluten Free Flour and Germination Effect on Various Properties. Journal of Food Processing and Preservation 2017, 41, e12959. [Google Scholar] [CrossRef]
- Lan, Y.; Wang, X.; Wang, L.; Zhang, W.; Song, Y.; Zhao, S.; Yang, X.; Liu, X. Change of Physiochemical Characteristics, Nutritional Quality, and Volatile Compounds of Chenopodium Quinoa Willd. during Germination. Food Chemistry 2024, 445, 138693. [Google Scholar] [CrossRef] [PubMed]
- Mawouma, S.; Cotârleț, M.; Lazăr, N.N.; Stănciuc, N.; Râpeanu, G. Effect of Combined Germination and Spontaneous Fermentation on the Bioactive, Mineral, and Microbial Profile of Red Sorghum and Pearl Millet Flours. AUDJG - Food Technology 2023, 47, 207–220. [Google Scholar] [CrossRef]
- Tyagi, A.; Chen, X.; Shabbir, U.; Chelliah, R.; Oh, D.H. Effect of Slightly Acidic Electrolyzed Water on Amino Acid and Phenolic Profiling of Germinated Brown Rice Sprouts and Their Antioxidant Potential. LWT 2022, 157, 113119. [Google Scholar] [CrossRef]
- AL-Ansi, W.; Mahdi, A.A.; Al-Maqtari, Q.A.; Mushtaq, B.S.; Ahmed, A.; Karrar, E.; Mohammed, J.K.; Fan, M.; Li, Y.; Qian, H.; et al. The Potential Improvements of Naked Barley Pretreatments on GABA, β-Glucan, and Antioxidant Properties. LWT 2020, 130, 109698. [Google Scholar] [CrossRef]
- Hsieh, C.-C.; Yu, S.-H.; Cheng, K.-W.; Liou, Y.-W.; Hsu, C.-C.; Hsieh, C.-W.; Kuo, C.-H.; Cheng, K.-C. Production and Analysis of Metabolites from Solid-State Fermentation of Chenopodium Formosanum (Djulis) Sprouts in a Bioreactor. Food Research International 2023, 168, 112707. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Hernández, A.A.; Rouzaud-Sández, O.; Frias, J.; Ayala-Zavala, F.; Astiazarán-García, H.; Salazar–López, N.J.; López-Saiz, C.M.; De La Reé-Rodríguez, S.C.; Sánchez, M.R. Antioxidant and Anti-Inflammatory Potential of a Food Produced from Irradiated (UV-A LED) Sorghum Sprouts Subjected to in Vitro Gastrointestinal Simulation. Journal of Functional Foods 2023, 110, 105857. [Google Scholar] [CrossRef]
- Yan, H.; Chen, H.; Liu, J.; Yao, T.; Xia, M.; Liao, Q.; Huang, L.; Li, W.; Song, Y.; Peng, L.; et al. Pyridoxal Phosphate Promotes the γ-Aminobutyric Acid Accumulation, Antioxidant and Anti-Hypertensive Activity of Germinated Tartary Buckwheat. Journal of Cereal Science 2024, 120, 104024. [Google Scholar] [CrossRef]
- Nsabimana, S.; Ismail, T.; Lazarte, C.E. Enhancing Iron and Zinc Bioavailability in Corn (Zea Mays) through Phytate Reduction: The Impact of Fermentation Alone and in Combination with Soaking and Germination. Front. Nutr. 2024, 11, 1478155. [Google Scholar] [CrossRef] [PubMed]
- Sim, U.; Sung, J.; Lee, H.; Heo, H.; Jeong, H.S.; Lee, J. Effect of Calcium Chloride and Sucrose on the Composition of Bioactive Compounds and Antioxidant Activities in Buckwheat Sprouts. Food Chemistry 2020, 312, 126075. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.; Martinez, E.; Carbonell, V.; Florez, M. Magnetic-Time Model for Triticale Seeds Germination. Romanian Journal of Physics 2019, 64, 10. [Google Scholar]
- He, W.; Wang, Y.; Luo, H.; Li, D.; Liu, C.; Song, J.; Zhang, Z.; Liu, C.; Niu, L. Effect of NaCl Stress and Supplemental CaCl2 on Carotenoid Accumulation in Germinated Yellow Corn Kernels. Food Chemistry 2020, 309, 125779. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Yeo, H.J.; Park, Y.E.; Chun, S.W.; Chung, Y.S.; Lee, S.Y.; Park, S.U. Influence of Chitosan, Salicylic Acid and Jasmonic Acid on Phenylpropanoid Accumulation in Germinated Buckwheat (Fagopyrum Esculentum Moench). Foods 2019, 8, 153. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ma, H.; Wang, S. Application of Ultrasound, Microwaves, and Magnetic Fields Techniques in the Germination of Cereals. Food Science and Technology Research 2019, 25, 489–497. [Google Scholar] [CrossRef]
- Chavarín-Martínez, C.D.; Reyes-Moreno, C.; Milán-Carrillo, J.; Perales-Sánchez, J.X.K.; Canizalez-Román, V.A.; Cuevas-Rodriguez, E.-O.; López-Valenzuela, J.A.; Gutiérrez-Dorado, R. Effect of Germination and UV-B Elicitation on Chemical Compositions, Antioxidant Activities, and Phytochemical Contents of Underutilised Mexican Blue Corn Seeds. IFRJ 2022, 29, 300–310. [Google Scholar] [CrossRef]
- Song, J.-S.; Lee, M.J.; Ra, J.E.; Lee, K.S.; Eom, S.; Ham, H.M.; Kim, H.Y.; Kim, S.B.; Lim, J. Growth and Bioactive Phytochemicals in Barley Sprouts Affected by Atmospheric Pressure Plasma during Seed Germination. J. Phys. D: Appl. Phys. 2020, 53, 314002. [Google Scholar] [CrossRef]
- Souid, A.; Bellani, L.; Tassi, E.L.; Ben Hamed, K.; Longo, V.; Giorgetti, L. Early Physiological, Cytological and Antioxidative Responses of the Edible Halophyte Chenopodium Quinoa Exposed to Salt Stress. Antioxidants 2023, 12, 1060. [Google Scholar] [CrossRef] [PubMed]
- Yodpitak, S.; Mahatheeranont, S.; Boonyawan, D.; Sookwong, P.; Roytrakul, S.; Norkaew, O. Cold Plasma Treatment to Improve Germination and Enhance the Bioactive Phytochemical Content of Germinated Brown Rice. Food Chemistry 2019, 289, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Z.; Manzoor, M.F.; Ahmad, N.; Zeng, X.; Din, Z.U.; Roobab, U.; Qayum, A.; Siddique, R.; Siddeeg, A.; Rahaman, A. Impact of Pulsed Electric Field Treatments on the Growth Parameters of Wheat Seeds and Nutritional Properties of Their Wheat Plantlets Juice. Food Science & Nutrition 2020, 8, 2490–2500. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Tao, Y.; Han, Y.; Ding, Y.; Jian, X.; Li, D. Preparation of Germinated Brown Rice with High γ-Aminobutyric Acid Content and Short Root by Magnetic Field Treatment. Journal of Cereal Science 2023, 112, 103720. [Google Scholar] [CrossRef]
- Johnson, M.A.; Kumar, M.; Thakur, S. Effect of Variation in Temperature and Light Duration on Morpho-Physiology and Phytochemical Content in Sprouts and Microgreens of Common Buckwheat (Fagopyrum Esculentum Moench). Plant Foods Hum Nutr 2024, 79, 875–885. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cheng, J.-H.; Sun, D.-W. Enhancement of Wheat Seed Germination, Seedling Growth and Nutritional Properties of Wheat Plantlet Juice by Plasma Activated Water. J Plant Growth Regul 2023, 42, 2006–2022. [Google Scholar] [CrossRef] [PubMed]
- Xia, Q.; Tao, H.; Li, Y.; Pan, D.; Cao, J.; Liu, L.; Zhou, X.; Barba, F.J. Characterizing Physicochemical, Nutritional and Quality Attributes of Wholegrain Oryza Sativa L. Subjected to High Intensity Ultrasound-Stimulated Pre-Germination. Food Control 2020, 108, 106827. [Google Scholar] [CrossRef]
- Lee, H.-H.; Yiu, E.; Zheng, A.-L.-T.; Bong, J.-C.-F.; Loh, S.-P.; Yiu, P.H. Optimisation of Phytate Degradation in Whole Grain Rice During Germination Processing Using Response Surface Methodology. BJRST 2023, 13, 132–141. [Google Scholar] [CrossRef]
- Yadav, A.; Singh, S. Effect of Exogenous Phytohormone Treatment on Antioxidant Activity, Enzyme Activity and Phenolic Content in Wheat Sprouts and Identification of Metabolites of Control and Treated Samples by UHPLC-MS Analysis. Food Research International 2023, 169, 112811. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Wang, L.L.; Chen, X.Y.; Yang, Y.; Wang, Z.; Xiong, F. Effects of Exogenous Gibberellic Acid and Abscisic Acid on Germination, Amylases, and Endosperm Structure of Germinating Wheat Seeds. Seed Sci. Technol. 2016, 44, 64–76. [Google Scholar] [CrossRef]
- Dębski, H.; Wiczkowski, W.; Szawara-Nowak, D.; Horbowicz, M. Elicitation with Sodium Silicate and Iron Chelate Affects the Contents of Phenolic Compounds and Minerals in Buckwheat Sprouts. Pol. J. Food Nutr. Sci. 2021, 21–28. [Google Scholar] [CrossRef]
- Sharma, R.; Sharma, S. Anti-Nutrient & Bioactive Profile, in Vitro Nutrient Digestibility, Techno-Functionality, Molecular and Structural Interactions of Foxtail Millet (Setaria Italica L.) as Influenced by Biological Processing Techniques. Food Chemistry 2022, 368, 130815. [Google Scholar] [CrossRef] [PubMed]
- Evrendilek, G.A.; Atmaca, B.; Bulut, N.; Uzuner, S. Development of Pulsed Electric Fields Treatment Unit to Treat Wheat Grains: Improvement of Seed Vigour and Stress Tolerance. Computers and Electronics in Agriculture 2021, 185, 106129. [Google Scholar] [CrossRef]
- Kanjevac, M.; Bojović, B.; Jakovljević, D. Improvement of Physiological Performance of Selected Cereals by Modulating Pregerminative Metabolic Activity in Seeds. CEREAL RESEARCH COMMUNICATIONS 2021, 50, 831–839. [Google Scholar] [CrossRef]
- Polachini, T.C.; Norwood, E.-A.; Le-Bail, P.; Le-Bail, A.; Cárcel, J.A. Pulsed Electric Field (PEF) Application on Wheat Malting Process: Effect on Hydration Kinetics, Germination and Amylase Expression. Innovative Food Science & Emerging Technologies 2023, 86, 103375. [Google Scholar] [CrossRef]
- Zhang, L.; Hao, N.; Li, W.; Zhang, B.; Shi, T.; Xie, M.; Yu, M. Effect of Ultrasonic Induction on the Main Physiological and Biochemical Indicators and γ–Aminobutyric Acid Content of Corn during Germination. Foods 2022, 11, 1358. [Google Scholar] [CrossRef]
- Bernate, I.; Kince, T.; Radenkovs, V.; Juhnevica-Radenkova, K.; Cinkmanis, I.; Bruveris, J.; Sabovics, M. Impact of Ozone Exposure on the Biochemical Composition of Wheat, Broccoli, Alfalfa, and Radish Seeds During Germination. Agronomy 2024, 14, 2571. [Google Scholar] [CrossRef]
- Feizollahi, E.; Jeganathan, B.; Reiz, B.; Vasanthan, T.; Roopesh, M.S. Reduction of Deoxynivalenol during Barley Steeping in Malting Using Plasma Activated Water and the Determination of Major Degradation Products. Journal of Food Engineering 2023, 352, 111525. [Google Scholar] [CrossRef]
- Lazukin, A.V.; Grabel’nykh, O.I.; Serdyukov, Yu.A.; Pobezhimova, T.P.; Nurminskii, V.N.; Korsukova, A.V.; Krivov, S.A. The Effect of Surface Barrier Discharge Plasma Products on the Germination of Cereals. Tech. Phys. Lett. 2019, 45, 16–19. [Google Scholar] [CrossRef]
- Shin, J.; Yang, J.; Yang, J.-Y. Germination of Tartary Buckwheat at Various Light Strengths to Enhance Flavonoid Content and Scale-up of the Process Using Smart-Farm Systems. Journal of Cereal Science 2023, 112, 103727. [Google Scholar] [CrossRef]
- Li, M.; Zhang, X.; Gao, Z.; Wu, M.; Ren, T.; Wu, C.; Wang, J.; Geng, Y.; Lv, W.; Zhou, Q.; et al. Metabolomic Insights into the Profile, Bioaccessibility, and Transepithelial Transport of Polyphenols from Germinated Quinoa during in Vitro Gastrointestinal Digestion/Caco-2 Cell Transport, and Their Prebiotic Effects during Colonic Fermentation. Food Research International 2024, 186, 114339. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, D.; Kumar, K.; Ahmed, N.; Pal Singh, T.; Thakur, P.; Hyder Rizvi, Q.-U.-E.; Yadav, A.N.; Dhaliwal, H.S. Effect of Processing Treatments on the Nutritional, Anti-Nutritional, and Bioactive Composition of Blue Corn (Zea Mays L.). Curr Res Nutr Food Sci 2022, 10, 171–182. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Nguyen, C.H. Effects of Processing Conditions on Change of Amino Acids, Reducing Sugar Andtotal Polyphenols of Caramelized Malt Produced from IR50404 Rice Variety. Food Res. 2024, 8, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Van De Velde, F.; Méndez-Galarraga, M.P.; Albarracín, M.; Garzón, A.G.; Aquino, M.; Cian, R.E.; Vinderola, G.; Drago, S.R. Exploring the Effects of Barley Germination on Chemical Composition, Phytic Acid, and Potential Malt Prebiotic Properties. Journal of Food Composition and Analysis 2025, 138, 107016. [Google Scholar] [CrossRef]
- Vingrys, K.; Mathai, M.; Ashton, J.F.; Stojanovska, L.; Vasiljevic, T.; McAinch, A.J.; Donkor, O.N. The Effect of Malting on Phenolic Compounds and Radical Scavenging Activity in Grains and Breakfast Cereals. Journal of Food Science 2022, 87, 4188–4202. [Google Scholar] [CrossRef] [PubMed]
- García-Santiesteban, A.E.; Ramírez-Corona, N.; López-Malo, A.; Palou, E.; Jiménez-Munguía, M.T. UVC Light Influence on the Sanitization of Alfalfa (Medicago Sativa), Wheat (Triticum Aestivum) and Chia (Salvia Hispanica) Seeds, Sprout Germination and Antioxidant Properties. Postharvest Biology and Technology 2024, 214, 112958. [Google Scholar] [CrossRef]
- Tene, S.T.; Ndinteh, D.T.; Dongmo, J.R.; Adebo, O.A.; Kewuyemi, Y.O.; Kengne Kamdem, M.H.; Obilana, A.O.; Klang, J.M.; Njobeh, P.B.; Womeni, H.M. Optimization Using Response Surface Methodology of Amylolytic Capacity of Corn Atp-Y and Coca-Sr Varieties: In Vitro Digestibility Capacity, Physico-Chemical and Functional Properties of Optimal Sample. Journal of Agriculture and Food Research 2022, 9, 100342. [Google Scholar] [CrossRef]
- Farooq, M.A.; Ma, W.; Shen, S.; Gu, A. Underlying Biochemical and Molecular Mechanisms for Seed Germination. IJMS 2022, 23, 8502. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, W.; Lu, H.; Shu, Q.; Zhang, Y.; Chen, Q. New Perspectives on Physiological, Biochemical and Bioactive Components during Germination of Edible Seeds: A Review. Trends in Food Science & Technology 2022, 123, 187–197. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, P.; Gu, Z.; Sun, M.; Yang, R. Effects of Germination on Physio-Biochemical Metabolism and Phenolic Acids of Soybean Seeds. Journal of Food Composition and Analysis 2022, 112, 104717. [Google Scholar] [CrossRef]
- Bhandari, U.; Gajurel, A.; Khadka, B.; Thapa, I.; Chand, I.; Bhatta, D.; Poudel, A.; Pandey, M.; Shrestha, S.; Shrestha, J. Morpho-Physiological and Biochemical Response of Rice to Drought Stress: A Review. Heliyon 2023, 9, e13744. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Xu, X.; Guo, L.; Yuzuak, S.; Lu, Y. Physiological and Biochemical Effects of Polystyrene Micro/Nano Plastics on Arabidopsis Thaliana. Journal of Hazardous Materials 2024, 469, 133861. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, X.; Gao, G.; Ali, I.; Wu, X.; Tang, M.; Chen, L.; Jiang, L.; Liang, T. Effects of Various Seed Priming on Morphological, Physiological, and Biochemical Traits of Rice under Chilling Stress. Front. Plant Sci. 2023, 14, 1146285. [Google Scholar] [CrossRef] [PubMed]
- Bellache, M.; Moltó, N.; Benfekih, L.A.; Torres-Pagan, N.; Mir, R.; Verdeguer, M.; Boscaiu, M.; Vicente, O. Physiological and Biochemical Responses to Water Stress and Salinity of the Invasive Moth Plant, Araujia Sericifera Brot., during Seed Germination and Vegetative Growth. Agronomy 2022, 12, 361. [Google Scholar] [CrossRef]
- Gao, Z.-W.; Mu, Y.-G.; Ding, J.-J.; Ding, K.-J.; Li, J.-T.; Li, X.-N.; He, L.-J.; Wang, Z.-J.; Mu, C.-S.; Alharbi, S.A.; et al. The Effects of Salinity Stress on Amorpha Fruticosa Linn. Seed Germination, Physiological and Biochemical Mechanisms. Not Bot Horti Agrobo 2024, 52, 13552. [Google Scholar] [CrossRef]
- Hafeez, A.; Rasheed, R.; Ashraf, M.A.; Qureshi, F.F.; Hussain, I.; Iqbal, M. Effect of Heavy Metals on Growth, Physiological and Biochemical Responses of Plants. In Plants and Their Interaction to Environmental Pollution; Elsevier, 2023; pp. 139–159 ISBN 978-0-323-99978-6.
- Altıkardeş, E.; Güzel, N. Impact of Germination Pre-Treatments on Buckwheat and Quinoa: Mitigation of Anti-Nutrient Content and Enhancement of Antioxidant Properties. Food Chemistry: X 2024, 21, 101182. [Google Scholar] [CrossRef] [PubMed]
- Naumenko, N.; Fatkullin, R.; Popova, N.; Ruskina, A.; Kalinina, I.; Morozov, R.; Avdin, V.V.; Antonova, A.; Vasileva, E. Effect of a Combination of Ultrasonic Germination and Fermentation Processes on the Antioxidant Activity and γ-Aminobutyric Acid Content of Food Ingredients. Fermentation 2023, 9, 246. [Google Scholar] [CrossRef]
- Chu, C.; Yan, N.; Du, Y.; Liu, X.; Chu, M.; Shi, J.; Zhang, H.; Liu, Y.; Zhang, Z. iTRAQ-Based Proteomic Analysis Reveals the Accumulation of Bioactive Compounds in Chinese Wild Rice (Zizania Latifolia) during Germination. Food Chemistry 2019, 289, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Jan, R.; Riar, C.S.; Bansal, V. Analyzing the Effect of Germination on the Pasting, Rheological, Morphological and in- Vitro Antioxidant Characteristics of Kodo Millet Flour and Extracts. Food Chemistry 2021, 361, 130073. [Google Scholar] [CrossRef] [PubMed]
- Anwar, T.; Qureshi, H.; Akhtar, M.S.; Siddiqi, E.H.; Fatimah, H.; Zaman, W.; Alhammad, B.A.; Seleiman, M.F. Enhancing Corn Growth and Resilience to Environmental Stress with Biochar, Gibberellic Acid and Rhizobacteria. Front. Plant Sci. 2024, 15, 1396594. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Yang, Y.; Zhao, Y.; Liu, Z.; Li, C.; He, L.; Liu, L. Effect of Different Conditions on the Germination of Coix Seed and Its Characteristics Analysis. Food Chemistry: X 2024, 22, 101332. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Wang, Y.; Dai, Z.; Liu, C.; Xiao, Y.; Wei, Q.; Song, J.; Li, D. Effect of UV-B Radiation and a Supplement of CaCl2 on Carotenoid Biosynthesis in Germinated Corn Kernels. Food Chemistry 2019, 278, 509–514. [Google Scholar] [CrossRef] [PubMed]
- García-Mosqueda, C.; Cerón-García, A.; León-Galván, M.F.; Ozuna, C.; López-Malo, A.; Sosa-Morales, M.E. Changes in Phenolics and Flavonoids in Amaranth and Soybean Sprouts after UV-C Treatment. J Food Sci 2023, 88, 1280–1291. [Google Scholar] [CrossRef] [PubMed]
- Aly, B.E.; Mona, B.H.; Higazy, A.M. Green Synthesis of Silver Nanoparticles by Cyanobacterial Extracts: An Approach Guarantees Potential Bioactivity and Proper Cereal Seed Germination. Egyptian Pharmaceutical Journal 2023, 22, 613–631. [Google Scholar] [CrossRef]
- Rashid, M.T.; Liu, K.; Wei, D.-Z.; Jatoi, M.A.; Li, Q.; Sarpong, F. Drying Kinetics and Quality Dynamics of Ultrasound-Assisted Dried Selenium-Enriched Germinated Black Rice. Ultrasonics Sonochemistry 2023, 98, 106468. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, G.; Anwar, T.; Malik, M.; Rehman, H.U.; Danish, S.; Alahmadi, T.A.; Ansari, M.J. Evaluation of Potassium-Enriched Biochar and GA3 Effectiveness for Improving Wheat Growth under Drought Stress. BMC Plant Biol 2023, 23, 615. [Google Scholar] [CrossRef] [PubMed]
- Lalaleo, L.; Hidalgo, D.; Valle, M.; Calero-Cáceres, W.; Lamuela-Raventós, R.M.; Becerra-Martínez, E. Differentiating, Evaluating, and Classifying Three Quinoa Ecotypes by Washing, Cooking and Germination Treatments, Using 1H NMR-Based Metabolomic Approach. Food Chemistry 2020, 331, 127351. [Google Scholar] [CrossRef] [PubMed]
- Aparicio-García, N.; Martínez-Villaluenga, C.; Frias, J.; Peñas, E. Changes in Protein Profile, Bioactive Potential and Enzymatic Activities of Gluten-Free Flours Obtained from Hulled and Dehulled Oat Varieties as Affected by Germination Conditions. LWT 2020, 134, 109955. [Google Scholar] [CrossRef]
- Donkor, O.N.; Stojanovska, L.; Ginn, P.; Ashton, J.; Vasiljevic, T. Germinated Grains – Sources of Bioactive Compounds. Food Chemistry 2012, 135, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, H.; Liao, C.; Liu, X. Influences of UV-B Treatment and Cooking Methods on Bioactive Components in Germinated Highland Barley. LWT 2023, 186, 115194. [Google Scholar] [CrossRef]
- He, Y.; Song, S.; Li, C.; Zhang, X.; Liu, H. Effect of Germination on the Main Chemical Compounds and 5-Methyltetrahydrofolate Metabolism of Different Quinoa Varieties. Food Research International 2022, 159, 111601. [Google Scholar] [CrossRef] [PubMed]
- Kanjevac, M.; Bojović, B.; Ćirić, A.; Stanković, M.; Jakovljević, D. Seed Priming Improves Biochemical and Physiological Performance of Wheat Seedlings under Low-Temperature Conditions. Agriculture 2022, 13, 2. [Google Scholar] [CrossRef]
- Ahmed, Z.; Faisal Manzoor, M.; Hussain, A.; Hanif, M.; Zia-ud-Din; Zeng, X. -A. Study the Impact of Ultra-Sonication and Pulsed Electric Field on the Quality of Wheat Plantlet Juice through FTIR and SERS. Ultrasonics Sonochemistry 2021, 76, 105648. [Google Scholar] [CrossRef] [PubMed]
- Naseer, I.; Javed, S.; Shah, A.A.; Tariq, A.; Ahmad, A. Influence of Phyto-Mediated Zinc Oxide Nanoparticles on Growth of (Zea Mays L.). PAK. J. BOT. 2024, 56. [Google Scholar] [CrossRef] [PubMed]
- Masure, H.G.; Fierens, E.; Delcour, J.A. Current and Forward Looking Experimental Approaches in Gluten-Free Bread Making Research. Journal of Cereal Science 2016, 67, 92–111. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Szulc, P.; Szczepaniak, O.; Dziedziński, M.; Szymanowska, D.; Szymandera-Buszka, K.; Goryńska-Goldmann, E.; Gazdecki, M.; Telichowska, A.; Ligaj, M. Variability of Hordeum Vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity. Sustainability 2020, 12, 1938. [Google Scholar] [CrossRef]
- Popoola, O.O. Phenolic Compounds Composition and in Vitro Antioxidant Activity of Nigerian Amaranthus Viridis Seed as Affected by Autoclaving and Germination. Measurement: Food 2022, 6, 100028. [Google Scholar] [CrossRef]
- Ma, H.; Xu, X.; Wang, S.; Wang, J.; Wang, S. Effects of Microwave Irradiation of Fagopyrum Tataricum Seeds on the Physicochemical and Functional Attributes of Sprouts. LWT 2022, 165, 113738. [Google Scholar] [CrossRef]
- Dostalíková, L.; Hlásná Čepková, P.; Janovská, D.; Jágr, M.; Svoboda, P.; Dvořáček, V.; Viehmannová, I. The Impact of Germination and Thermal Treatments on Bioactive Compounds of Quinoa (Chenopodium Quinoa Willd.) Seeds. Eur Food Res Technol 2024, 250, 1457–1471. [Google Scholar] [CrossRef]
- Jena, K.; Vairakannu, P.; Singha, S. Microwave Drying of Sprouted Little Millet and Assessment of Their Kinetic and Physiochemical Properties. Journal of Cereal Science 2025, 122, 104122. [Google Scholar] [CrossRef]
- Paucar-Menacho, L.M.; Martínez-Villaluenga, C.; Dueñas, M.; Frias, J.; Peñas, E. Response Surface Optimisation of Germination Conditions to Improve the Accumulation of Bioactive Compounds and the Antioxidant Activity in Quinoa. International Journal of Food Science & Technology 2018, 53, 516–524. [Google Scholar] [CrossRef]
- Sneha, K.; Kumar, M.; Kaushik, D.; Kumar, A.; Bansal, V.; Oz, F.; Proestos, C. To Study the Germination Effect on Finger Millet, Pearl Millet and Buckwheat: It’s Impact on Phytochemical Properties and Their Prebiotic Effect. J Food Chem Nanotechnol 2023, 9. [Google Scholar] [CrossRef]
- Złotek, U.; Gawlik-Dziki, U.; Dziki, D.; Świeca, M.; Nowak, R.; Martinez, E. Influence of Drying Temperature on Phenolic Acids Composition and Antioxidant Activity of Sprouts and Leaves of White and Red Quinoa. Journal of Chemistry 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Paucar-Menacho, L.M.; Martínez-Villaluenga, C.; Dueñas, M.; Frias, J.; Peñas, E. Response Surface Optimisation of Germination Conditions to Improve the Accumulation of Bioactive Compounds and the Antioxidant Activity in Quinoa. Int J of Food Sci Tech 2018, 53, 516–524. [Google Scholar] [CrossRef]
- Dey, S.; Saxena, A.; Kumar, Y.; Maity, T.; Tarafdar, A. Synergistic Effects of Germination and Ultrasonication on Nutritional and Structural Characteristics of Kodo ( Paspalum Scrobiculatum ) and Little ( Panicum Sumatrense ) Millet. Journal of Food Quality 2024, 2024, 4951196. [Google Scholar] [CrossRef]
- Shakhov, I.V.; Kokorev, A.I.; Yuriev Institute of Plant Production, National Academy of Agrarian Sciences of Ukraine, 142 Heroiv Kharkova Ave. , Kharkiv 61060, Ukraine; Yastreb, T.O.; Yuriev Institute of Plant Production, National Academy of Agrarian Sciences of Ukraine, 142 Heroiv Kharkova Ave., Kharkiv 61060, Ukraine; Dmitriev, A.P.; Institute of Cell Biology and Genetic Engineering, National Academy of Sciences of Ukraine, 148 Akademika Zabolotnogo Str., Kyiv 03143, Ukraine; Kolupaev, Yu.E.; Yuriev Institute of Plant Production, National Academy of Agrarian Sciences of Ukraine, 142 Heroiv Kharkova Ave., Kharkiv 61060, Ukraine Increasing germination and antioxidant activity of aged wheat and triticale grains by priming with gamma-aminobutyric acid. Ukr. Bot. J. 2024, 81, 290–304. [Google Scholar] [CrossRef]
- Ge, X.; Saleh, A.S.M.; Jing, L.; Zhao, K.; Su, C.; Zhang, B.; Zhang, Q.; Li, W. Germination and Drying Induced Changes in the Composition and Content of Phenolic Compounds in Naked Barley. Journal of Food Composition and Analysis 2021, 95, 103594. [Google Scholar] [CrossRef]
- Xing, B.; Teng, C.; Sun, M.; Zhang, Q.; Zhou, B.; Cui, H.; Ren, G.; Yang, X.; Qin, P. Effect of Germination Treatment on the Structural and Physicochemical Properties of Quinoa Starch. Food Hydrocolloids 2021, 115, 106604. [Google Scholar] [CrossRef]
- Ramos-Pacheco, B.S.; Choque-Quispe, D.; Ligarda-Samanez, C.A.; Solano-Reynoso, A.M.; Palomino-Rincón, H.; Choque-Quispe, Y.; Peralta-Guevara, D.E.; Moscoso-Moscoso, E.; Aiquipa-Pillaca, Á.S. Effect of Germination on the Physicochemical Properties, Functional Groups, Content of Bioactive Compounds, and Antioxidant Capacity of Different Varieties of Quinoa (Chenopodium Quinoa Willd.) Grown in the High Andean Zone of Peru. Foods 2024, 13, 417. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Guan, X.; Gong, B.; Li, C. Chemical Components and Chain-Length Distributions Affecting Quinoa Starch Digestibility and Gel Viscoelasticity after Germination Treatment. Food Funct. 2021, 12, 4060–4071. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.; Sahu, J.K. Effect of Soaking and Germination on Grain Matrix and Glycaemic Potential: A Comparative Study on White Quinoa, Proso and Foxtail Millet Flours. Food Bioscience 2023, 56, 103105. [Google Scholar] [CrossRef]
- Al-Qabba, M.M.; El-Mowafy, M.A.; Althwab, S.A.; Alfheeaid, H.A.; Aljutaily, T.; Barakat, H. Phenolic Profile, Antioxidant Activity, and Ameliorating Efficacy of Chenopodium Quinoa Sprouts against CCl4-Induced Oxidative Stress in Rats. Nutrients 2020, 12, 2904. [Google Scholar] [CrossRef] [PubMed]
- Thakur, P.; Kumar, K.; Ahmed, N.; Chauhan, D.; Eain Hyder Rizvi, Q.U.; Jan, S.; Singh, T.P.; Dhaliwal, H.S. Effect of Soaking and Germination Treatments on Nutritional, Anti-Nutritional, and Bioactive Properties of Amaranth (Amaranthus Hypochondriacus L.), Quinoa (Chenopodium Quinoa L.), and Buckwheat (Fagopyrum Esculentum L.). Current Research in Food Science 2021, 4, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Gunathunga, C.; Senanayake, S.; Jayasinghe, M.A.; Brennan, C.S.; Truong, T.; Marapana, U.; Chandrapala, J. Germination Effects on Nutritional Quality: A Comprehensive Review of Selected Cereals and Pulses Changes. Journal of Food Composition and Analysis 2024, 128, 106024. [Google Scholar] [CrossRef]
- Jágr, M.; Hofinger-Horvath, A.; Ergang, P.; Čepková, P.H.; Schönlechner, R.; Pichler, E.C.; D́Amico, S.; Grausgruber, H.; Vagnerová, K.; Dvořáček, V. Comprehensive Study of the Effect of Oat Grain Germination on the Content of Avenanthramides. Food Chemistry 2024, 437, 137807. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, S.; Han, N.; Bian, H.; Song, D. Effects of Germinated and Ungerminated Grains on the Production of Non-Dairy Probiotic-Fermented Beverages. Qual. Assur. Saf. Crops Foods 2022, 14, 32–39. [Google Scholar] [CrossRef]
- Bassey, S.O.; Chinma, C.E.; Ezeocha, V.C.; Adedeji, O.E.; Jolayemi, O.S.; Alozie-Uwa, U.C.; Adie, I.E.; Ofem, S.I.; Adebo, J.A.; Adebo, O.A. Nutritional and Physicochemical Changes in Two Varieties of Fonio (Digitaria Exilis and Digitaria Iburua) during Germination. Heliyon 2023, 9, e17452. [Google Scholar] [CrossRef] [PubMed]
- Chavarín-Martínez, C.D.; Gutiérrez-Dorado, R.; Perales-Sánchez, J.X.K.; Cuevas-Rodríguez, E.O.; Milán-Carrillo, J.; Reyes-Moreno, C. Germination in Optimal Conditions as Effective Strategy to Improve Nutritional and Nutraceutical Value of Underutilized Mexican Blue Corn Seeds. Plant Foods Hum Nutr 2019, 74, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Al-Taher, F.; Nemzer, B. Effect of Germination on Fatty Acid Composition in Cereal Grains. Foods 2023, 12, 3306. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, P.M.; Sreerama, Y.N. Impact of Processing on the Phenolic Profiles of Small Millets: Evaluation of Their Antioxidant and Enzyme Inhibitory Properties Associated with Hyperglycemia. Food Chemistry 2015, 169, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, Á.L.; Rico, D.; Ronda, F.; Martín-Diana, A.B.; Caballero, P.A. Development of a Gluten-Free Whole Grain Flour by Combining Soaking and High Hydrostatic Pressure Treatments for Enhancing Functional, Nutritional and Bioactive Properties. Journal of Cereal Science 2022, 105, 103458. [Google Scholar] [CrossRef]
- Fang, Y.; Franke, C.; Manthei, A.; McMullen, L.; Temelli, F.; Gänzle, M.G. Effects of High-Pressure Carbon Dioxide on Microbial Quality and Germination of Cereal Grains and Beans. The Journal of Supercritical Fluids 2021, 175, 105272. [Google Scholar] [CrossRef]
- Karabín, M.; Jelínek, L.; Průšová, N.; Ovesná, J.; Stránská, M. Pulsed Electric Field Treatment Applied to Barley before Malting Reduces Fusarium Pathogens without Compromising the Quality of the Final Malt. LWT 2024, 206, 116575. [Google Scholar] [CrossRef]
- Yudaev, I.V.; Daus, Y.V.; Eviev, V.A.; Soumyanova, E.V.; Goldvarg, T.B. Pre-Sowing Stimulation of Cereal Seeds in High Voltage Electric Field. BIO Web Conf. 2024, 103, 00063. [Google Scholar] [CrossRef]
- Wang, S.; Wang, J.; Guo, Y. Microwave Irradiation Enhances the Germination Rate of Tartary Buckwheat and Content of Some Compounds in Its Sprouts. Pol. J. Food Nutr. Sci. 2018, 68, 195–205. [Google Scholar] [CrossRef]
- Kondratenko, E.P.; Soboleva, O.M.; Konstantinova, O.B. Synthesis of Phenolic Compounds in Barley Seedlings Under the Influence of the Microwave Electromagnetic Field of Ultrahigh Frequency. JAC 2023, 141–147. [Google Scholar] [CrossRef]
- Zhang, G.; Xu, J.; Wang, Y.; Hua, D.; Zhang, H.; He, Y.; Liu, Y.; Tang, A.; Liu, H.; Sun, J. A Multi-Omics Study Revealed the Effect of Pulsed Light Treatment on Germinated Brown Rice: Promotion of Sprouting Efficiency and Gamma-Aminobutyric Acid Enrichment. Food Bioscience 2024, 61, 104196. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, X.; Xu, L.; Xie, M.; Yu, M. Non-Targeted Metabolomics Analysis of γ–Aminobutyric Acid Enrichment in Germinated Corn Induced by Pulsed Light. Foods 2024, 13, 2675. [Google Scholar] [CrossRef]
- Wani, H.M.; Sharma, P.; Wani, I.A.; Kothari, S.L.; Wani, A.A. Influence of Γ-irradiation on Antioxidant, Thermal and Rheological Properties of Native and Irradiated Whole Grain Millet Flours. Int J of Food Sci Tech 2021, 56, 3752–3762. [Google Scholar] [CrossRef]
- Xue, J.; Hu, M.; Yang, J.; Fang, W.; Yin, Y. Optimization of Ultraviolet-B Treatment for Enrichment of Total Flavonoids in Buckwheat Sprouts Using Response Surface Methodology and Study on Its Metabolic Mechanism. Foods 2024, 13, 3928. [Google Scholar] [CrossRef] [PubMed]
- Amnuaysin, N.; Korakotchakorn, H.; Chittapun, S.; Poolyarat, N. Seed Germination and Seedling Growth of Rice in Response to Atmospheric Air Dielectric-Barrier Discharge Plasma. Songklanakarin Journal of Science & Technology 2018, 40, 819–823. [Google Scholar]
- Ivankov, A.; Naučienė, Z.; Degutytė-Fomins, L.; Žūkienė, R.; Januškaitienė, I.; Malakauskienė, A.; Jakštas, V.; Ivanauskas, L.; Romanovskaja, D.; Šlepetienė, A.; et al. Changes in Agricultural Performance of Common Buckwheat Induced by Seed Treatment with Cold Plasma and Electromagnetic Field. Applied Sciences 2021, 11, 4391. [Google Scholar] [CrossRef]
- Kriz, P.; Petr, B.; Zbynek, H.; Jaromir, K.; Pavel, O.; Petr, S.; Miroslav, D. Influence of Plasma Treatment in Open Air on Mycotoxin Content and Grain Nutriments. Plasma Med 2015, 5, 145–158. [Google Scholar] [CrossRef]
- Martinovs, A.; Martinovs, A.; Rēvalde, G.; Dombrovska, D.; Koļčs, G.; Tretjakova, R.; Zaicevs, E. Effect of Short-term Treatment of Some Cereal Grains with Atmospheric Pressure Ar–O2 and Ar–Air Plasma. Plasma Processes & Polymers 2024, 21, e2400093. [Google Scholar] [CrossRef]
- Peťková, M.; Švubová, R.; Kyzek, S.; Medvecká, V.; Slováková, Ľ.; Ševčovičová, A.; Gálová, E. The Effects of Cold Atmospheric Pressure Plasma on Germination Parameters, Enzyme Activities and Induction of DNA Damage in Barley. IJMS 2021, 22, 2833. [Google Scholar] [CrossRef] [PubMed]
- Szőke, C.; Nagy, Z.; Gierczik, K.; Székely, A.; Spitkól, T.; Zsuboril, Z.T.; Galiba, G.; Marton, C.L.; Kutasi, K. Effect of the Afterglows of Low Pressure Ar/N2 -O2 Surface-wave Microwave Discharges on Barley and Corn Seeds. Plasma Processes & Polymers 2018, 15, 1700138. [Google Scholar] [CrossRef]
- Zhang, G.; Xu, J.; Wang, Y.; Sun, X.; Huang, S.; Huang, L.; Liu, Y.; Liu, H.; Sun, J. Combined Transcriptome and Metabolome Analyses Reveal the Mechanisms of Ultrasonication Improvement of Brown Rice Germination. Ultrasonics Sonochemistry 2022, 91, 106239. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Pan, Y.; Yang, D.; Zheng, X.; Liu, Z.; Shang, J. Effect of NaCl Stress Germination on Microstructure and Physicochemical Properties of Wheat Starch. International Journal of Biological Macromolecules 2025, 297, 139924. [Google Scholar] [CrossRef] [PubMed]
- Islam, Md.A.; Shorna, Most. N.A.; Islam, S.; Biswas, S.; Biswas, J.; Islam, S.; Dutta, A.K.; Uddin, Md.S.; Zaman, S.; Akhtar-E-Ekram, Md.; et al. Hydrogen-Rich Water: A Key Player in Boosting Wheat Seedling Growth and Drought Resilience. Sci Rep 2023, 13, 22521. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yang, N.; Wu, F.; Jin, Z.; Xu, X. Effect of Acid Pretreatment on the Physicochemical and Antioxidant Properties of Germinated Adlay ( Coix Lachryma-jobi L.). J Food Process Preserv 2018, 42, e13511. [Google Scholar] [CrossRef]
- Sanchez, D.R.; Jespersen, B.M.; Rasmussen, L.H.; Andersen, M.L. Fungicidal Effect of Gaseous Ozone in Malting Barley: Implications for Fusarium Infections and Grain Germination. Journal of Cereal Science 2024, 118, 103973. [Google Scholar] [CrossRef]
- Kumar, R.; Dadhich, A.; Dhiman, M.; Sharma, L.; Sharma, M.M. Stimulatory Effect of ZnO Nanoparticles as a Nanofertilizer in Seed Priming of Pearl Millet (Pennisetum Glaucum) and Their Bioactivity Studies. South African Journal of Botany 2024, 165, 30–38. [Google Scholar] [CrossRef]
- Deore, A.; Athmaselvi, K.A.; Venkatachalapathy, N. Effect of Ultrasound and Microwave Pretreatment on Sprouting, GABA, Bioactive Compounds, and Other Physicochemical Properties of Sorghum. Grain & Oil Science and Technology 2023, 6, 91–99. [Google Scholar] [CrossRef]
| Group | Characteristics and Benefits | Bioactive Compounds * | Mechanism of Biological Action | Matrices Studied | Ref. |
| Phenolic compounds | Potent antioxidants that neutralize free radicals, reduce lipid peroxidation, prevent cellular oxidative damage, reduce chronic disease risk, and have anti-inflammatory effects. | PHEA: p-HBA, CHLA, ELLA, SALA, p-COU, GENT, FERA. FLVN: RUTI, QUER, KAEM, CATCH, EPIC. ANTH and other POLY including TAN. | Act as free radical scavengers, chelate pro-oxidant metals, modulate inflammatory pathways, inhibit oxidative enzymes, protect cell membranes. | Red sorghum, pearl millet, djulis (Chenopodium formosanum), naked barley (Hordeum vulgare L. var nudum), blue corn, foxtail millet, wheat (Triticum aestivum L.), barley, buckwheat, quinoa. | [9,11,12,34,44,59,60] |
| Neurotransmitters | Main inhibitory neurotransmitter in CNS. Related to hypotensive, antidepressant, and nervous system regulatory effects. Improves sleep, reduces anxiety, regulates blood pressure. | GABA and its precursors such as glutamic acid. | Functions as inhibitory neurotransmitter, modulates neuronal excitability, reduces neurotransmitter release, and exerts calming effect. | Soft wheat, barley, naked barley, djulis, rice (Oryza sativa L.), buckwheat, finger millet (Eleusine coracana (L.) Gaertn.), and sorghum. | [11,12,20,26,60,61,62] |
| Bioactive peptides | Possess antioxidant, antihypertensive, antimicrobial, immunomodulatory, and antithrombotic activity. Improve mineral bioavailability and may have hypocholesterolemic effects. | AABA, BIOP with antioxidant activity, antimicrobial peptides, oligopeptides and FAA, immunomodulatory peptides. | Inhibit key enzymes in physiological processes, interact with opioid receptors, neutralize free radicals, bind to minerals increasing bioavailability. | Rice, djulis, corn, buckwheat, wheat, quinoa. | [12,25,28,45,63,64,65] |
| Melatonin and indolic compounds | Melatonin is a potent antioxidant and circadian rhythm regulator. INDO have neuroprotective and anti-inflammatory activity. Improve sleep, protect neurons, modulate immune system. | MELA, TRYP, p-CQA, FERQ. | MELA neutralizes free radicals in lipophilic and hydrophilic environments. INDO act as neurotransmitter precursors, modulate immune pathways. | Amaranth (Amaranthus spp.) | [66] |
| Vitamins | Improves nutritional profile and biological value, helps combat micronutrient deficiencies, especially in vulnerable populations. | VITB: THIA, RIBO, NIAC. ASCA and VITA precursors. FOL. | Reduces antinutritional compounds, transforms inactive vitamin forms to active forms, increases solubility and stability. | Corn, sorghum, pearl millet, barley, blue corn, foxtail millet. | [9,11,12,15,34,44] |
| Antioxidant enzymes | Contribute to detoxification of reactive oxygen species, reducing oxidative stress. Help prevent chronic diseases related to oxidative damage. | SOD, CAT, POD, GPx, GR. | Dismutation of superoxide anion, degradation of hydrogen peroxide, reduction of hydroperoxides, maintenance of antioxidant cycle. | Wheat, buckwheat, quinoa, corn, millet, barley, rice. | [28,36,59,65,67,68,69] |
| Various phytochemicals | Possess antioxidant, anti-inflammatory, anticarcinogenic, and immune system modulating activity. Contribute to chronic disease prevention, benefit visual and cardiovascular health. | CAROT, CHL-a, CHL-b, TERP, TERD, SAPN, natural pigments, PHYS. | Neutralize free radicals, modulate inflammation pathways, protect cell membranes, photosensitize activity, induce apoptosis in tumor cells. | Corn, djulis, millet, rice, barley. | [12,25,28,36,65,69,70] |
| Dietary fiber | Contributes to gastrointestinal health, prebiotic effect, cholesterol reduction, glycemic control, satiety sensation. Prevents cardiovascular diseases, type 2 diabetes, certain cancers. | β-GLU, ARBX, OLIGS, SOLDF, INDF, REST. | Increases intestinal viscosity, ferments via microbiota producing short-chain fatty acids, binds bile acids, stimulates beneficial bacteria growth. | Barley, wheat, sorghum, millet. | [4,11,71,72] |
| Species Studied | Optimal Processing Parameters | Mechanism of Action | Bioactive Compounds Analyzed | Quantitative Results | Cost-effectiveness | Ref |
| Quinoa | 22°C, 80% RH, 144 h; 48 h dark then 16/8 h light/dark | Upregulated ADCS and pterine synthesis genes control folate biosynthesis pathways. | TPC, TFC, ANTH, 5-MTHF, β-CAR, LUT, ASCA, RIBO | 5-MTHF: +8567%, TPC: +397%, TFC: +325%, ANTH: +958%, LUT: +19787%, ASCA: +276%, RIBO: +728% | High | [74] |
| Quinoa | 25°C, 48 h, light treatment, 95% RH | Activation of hydrolytic enzymes modifying starch molecular structure. | TPC | TPC: +16.4-50% | Medium | [91] |
| Quinoa | 72-120 h at room temperature, 4 h soaking, 16/8 h light/dark | Germination promotes enzymatic activity, releases bioactive compounds, reduces antinutrients. | TPC, AOA, GACA, 4-HBA, CAFA, Q3OG, ISOQ, RUTI, SALA, QUER, NARG, KAEM, PINO, ISOR | RUT: +245%, KAEM: +760%, QUER: +8333%, PINO: +933%, ISOR: +1250%, AOA: variable increases | High | [82] |
| Quinoa | 25°C, 36-72 h, 90-95% RH, darkness | Enzymatic activation, increased cell permeability, degradation of cell wall components. | GABA, TFC, TPC, FERA, VANA, p-HBA, t-HCA, CAFA, SALA, RUTI, KAEM, DIOS, ISOQ | GABA: +117%, FPHE: +32%, FLVN: +81%, BPHE: +22%, BFLVN: +127%, FERA: +104%, SALA: +597%, KAEM: +1331%, AOA: +1722% | High | [8] |
| Quinoa | 25°C, 72 h, 95% RH, followed by 40°C drying | Enzymatic activation mobilizing nutrient reserves and reducing antinutrients. | TPC, TFC | TPC: +43.3-74.4%, TFC: +95.4%, AOA: +99.6-100.8% | High | [92] |
| Quinoa | 25°C, 8 h soaking (1:10), germination to 1 cm sprouts | Enzymatic action modifies polyphenol state, promotes hydrolysis of ester bonds. | TPC, FERA, Q3OG, HYGE, 4-HBA, RUTI, MALT, QUER, GACA | TPC: +21.91%, FERA: +289.11%, Q3OG: +55.11%, QUER: +75.21%, GACA: +79.85% | High | [43] |
| Quinoa | 20°C, 96 h darkness, watering each 12 h | Activation of endogenous amylases preferentially degrading amylose molecules. | TPC | TPC: +13.7-50% | Medium | [93] |
| White quinoa | 28°C, 48 h, periodic water spraying | Amylolytic and debranching enzyme activation, cell wall degradation. | TPC, TFC | TPC: +15-36%, TFC: +4.7-25.4% | High | [94] |
| Red and yellow quinoa | 17±1°C, 90% RH, darkness, 6 days | Autolytic enzyme activation releasing free phenolic compounds. | TPC, AOA, CAROT, SAPN, ANTH, TFC, FLVL, GACA, PROTA, p-HBA, CATCH, SCOP, FERA, SINA, p-COU, RUTI, APG7G, ROSA, CINA, QUER, APG, KAEM, CHRY | TPC red: +178.9%, TPC yellow: +130.4%, AOA red: +69.8%, AOA yellow: +75.3%, FERA red: +114.6%, FERA yellow: +47.2%, TFC red: +47.2%, TFC yellow: +165.4% | High | [95] |
| Red and white quinoa | 20°C, 4 days darkness, drying at 30°C | Endogenous esterases release phenolic compounds or induce de novo synthesis. | TPC, TFC, p-HBA, VANA, p-COU, SALA, FERA | TPC: +105%, VANA: +9242%, FERA: +367%, AOA: +50% | Medium | [86] |
| Quinoa | 20°C, 42 h | Enzymatic hydrolysis, release of phenolic compounds, increased GABA synthesis. | TPC, TFC, FERA, p-COU, QUER, KAEM, GABA | TPC: +84.2%, TFC: +44.1%, GABA: +445.7%, AOA: +30% | High | [84] |
| Quinoa | 4°C 24 h stratification, 10°C 72 h, 16/8 h photoperiod, >10,000 lx | Activation of metabolic pathways increasing bioactive compound synthesis. | TRIG, BET, CHOL, SAPN | SAPN: -60% to +80% depending on ecotype | Medium | [70] |
| Species Studied | Optimal Parameters | Mechanism of Action | Bioactive Compounds | Quantitative Results | Cost-Effectiveness | Ref |
| Buckwheat | 25°C, 72 h | Activation of flavonoid-metabolizing enzymes enhancing flavonoid biosynthesis pathway. | FLVN, TPC, ANTZ (CAT, POD, SOD, APX) | FLVN: +49.3-53.2%, TPC: +30%, AOA: significant increase | High | [6] |
| Amaranth, quinoa, buckwheat | 25°C, 72 h, soaking 16 h | Activation of endogenous enzymes promoting phenolic compound biosynthesis and reducing phytic acid. | TPC, AOA | TPC amaranth: +126.62%, TPC buckwheat: +125.32%, TPC quinoa: +71.56%, AOA buckwheat: +178.38%, AOA amaranth: +87.47%, AOA quinoa: +34.88% | High | [96] |
| Amaranth | 35.86°C, 22 h | Degradative enzyme activation, especially phytase and proteases reducing antinutrients. | TPC, TFC, AOA | AOA: +43.8%, TPC: +4.5%, TFC: +3.2%, OLEA: +8.2%, LINA: +18.6% | Medium | [44] |
| Amaranth | 28±2°C, 72 h darkness, 30 min 0.2% formaldehyde pretreatment | Hydrolytic enzyme activation, de novo synthesis of secondary metabolites. | TPC, TFC, HYBZ, HYCA | TPC: +52.7%, TFC: +33.0%, FERA: +28.0%, p-HBA: +19.4%, AOA: +54.3% | High | [80] |
| Rice, Corn | 30-35°C, 192 h, 12 h initial soaking, watering every 12 h | Enzymatic activation promoting transformation and biosynthesis of phenolic compounds. | TPC, TFC, AOA | TPC corn: +600%, TPC rice: +10%, TFC corn: +230%, TFC rice: +74%, AOA corn: -86%, AOA FRAP corn: +348%, AOA FRAP rice: +27% | High | [97] |
| Chinese wild rice | 30°C darkness, 120 h | Activation of key proteins in metabolic pathways for phenolic synthesis and GABA production. | TPC, FERA, p-COU, CATCH, EPIC, GABA | TPC: +96.6%, FERA: +75%, CATCH: +67.3%, GABA: +729%, AOA: +50% | Medium | [61] |
| Oat | 20°C, 48-72 h, >85% RH, 24 h soaking | Activation of enzymes for avenanthramide synthesis during germination. | AVEN (AVN 2c, AVN 2p, AVN 2f, AVN 1a, AVN 2a, AVN 2ad) | Total AVEN: +29,300%, AVN 2c: +1,993-2,130%, AVN 2p: +900%, AVN 2f: +1,100%, AVN-hexosides: +2,100% | Medium | [98] |
| Hulled oat, dehulled oat | 16°C 216 h (Barra), 18°C 156 h (Meeri) | Hydrolytic enzyme activation and de novo synthesis of phenolic compounds. | FPHE, β-GLU, AOA | FPHE Barra: +63.7%, FPHE Meeri: +165.8%, β-GLU Barra: -46.8%, β-GLU Meeri: -55.9%, AOA Barra: +172.2%, AOA Meeri: +369.5% | High | [71] |
| Barley | 22°C, 240 h, ad libitum irrigation, 13.48-19.98 cm height | Accumulation of phenolic compounds and flavonols with antioxidant properties. | POLY, PHEA, FLVL (CATCH, EPIC, QUER, RUTI, KAEM) | POLY: +49.8%, PHEA: +41.0%, AOA DPPH: +175.0%, AOA ABTS: +127.3%, AChE inhibition: +610.0% | Medium | [79] |
| Barley | 25°C/72 h for phenolics, 20°C/35 h for prebiotics | Hydrolytic enzyme activation modifying cell walls and releasing phenolic compounds. | TPC, GACA, PROTA, HYBA, VANA, CAFA, p-COU, SINA, FERA | TPC: +61.8%, FERA: +89.1%, p-COU: +127.3%, GACA: +91.8% | High | [46] |
| Barley, Tibetan barley, rice | 30°C, 96 h barley/Tibetan barley, 48 h rice, soaking 8 h at 30°C, drying 55±5°C | Starch hydrolysis by amylases, increased nitrogen content, release of antioxidant compounds. | AOA, TPC | TPC: +23-41%, AOA: +36-64% | Medium | [99] |
| Naked barley | 25°C, 36 h, infrared drying (600 W/m², 20°C) | Activation of secondary metabolic pathways associated with phenolic metabolism. | FLVN, PHEA | VITX: +386%, RUTI: +379%, HESP: +775%, FERA: +766% | Medium | [90] |
| Chenopodium album | 25°C, 48 h, drying at 45°C for 12 h | Enzymatic degradation of starch and structural modification of cell wall. | TPC | TPC V1: +73.7%, TPC V2: +134.4%, AOA V1: +26.5%, AOA V2: +29.1% | Medium | [7] |
| White fonio (Digitaria exilis), brown fonio (Digitaria iburua) | 28°C, 72 h, 92% RH, darkness, 7 h soaking | Hydrolytic enzyme activation modifying endosperm and releasing bioactive compounds. | TPC, AOA | TPC brown: +297.28%, TPC white: +279.27%, AOA DPPH brown: +78.24%, AOA DPPH white: +78.42%, AOA ORAC brown: +18.97%, AOA ORAC white: +20.10% | High | [100] |
| Blue corn | 26.9°C, 207.7 h | Enzymatic degradation of major components and activation of phenolic compound biosynthesis. | TPC, ANTH | TPC: +79%, ANTH: +9.9%, AOA ABTS: +192%, AOA ORAC: +160%, AOA DPPH: +148% | Medium | [101] |
| Millet, amaranth, quinoa, other cereals | 19-23°C, 72 h, 92% RH, darkness, overnight soaking | Lipase enzyme activation and enzymatic desaturases converting saturated fatty acids. | Polyunsaturated fatty acids | Polyunsaturated fatty acids millet: +1.6%, amaranth: +5.9%, buckwheat: +11.7%, LINA millet: +0.5%, amaranth: +4.9%, Omega-3 millet: +67.2%, amaranth: +181.0%, buckwheat: +40.7% | Medium | [102] |
| Barnyard millet (Echinochloa frumentacea Link), foxtail millet, proso millet (Panicum miliaceum L.) | 25±2°C, 48 h, 16 h soaking (1:3 w/v) | Activation of glucosidases releasing aglycones from glycosides and biosynthesis of phenolic compounds. | TPC, TFC, PHEA, FLVN | TPC proso: +220.3%, TFC foxtail: +80.0%, FERA proso: +67.1%, AOA foxtail: +41.8%, α-glucosidase inhibition barnyard: +59.1% | High | [103] |
| Kodo millet | Soaking: 4 h at 23-24 °C; Germination: 48 h at 25 °C with 80-90% RH to maximize phenolic content and antioxidant activity. | Hydrolytic enzymes activated during germination degrade the protein-starch matrix, releasing bioactive compounds like phenols and GABA, while glutamate decarboxylase enzyme converts glutamate to GABA under stress conditions. | Total phenolic content (TPC), total antioxidant capacity (TAC), GABA. | TPC: kodo millet +30%. TPC: little millet (Panicum sumatrense Roth ex Roem. & Schult.) +20%. TFC: kodo millet +50%. TFC: little millet +70%. RSA: kodo millet 88.46%. RSA: little millet 89.06%. | ||
| Kodo millet | 25°C, 48 h, 80-90% RH, 4 h soaking at 23-24°C | Hydrolytic enzymes degrading protein-starch matrix, releasing bioactive compounds. | TPC, AOA, GABA | TPC: +52.2%, AOA: +48.3%, GABA: +410.6%, AOA DPPH: +13.4%, AOA H₂O₂: +69.7% | High | [62] |
| Kodo millet and little millet | 40°C, 72 h, 80-90% RH, drying at 45°C to 10% moisture | Cell wall degrading enzymes and activation of phenylalanine ammonia-lyase. | TPC, TFC, AOA | TPC kodo: +30%, TPC little: +20%, TFC kodo: +50%, TFC little: +70%, AOA kodo: 88.46%, AOA little: 89.06% | Medium | [88] |
| Little millet | 30°C, 72 h, 90% RH, microwave drying 1050 W at 50°C for 720 s | Rapid volumetric heating releases bound phenolic compounds and degrading cellular constituents. | TPC, TFC, p-COU, DHFA, SYRA, ACA, EPGC, p-COU hexoside | TPC: +23.2%, TFC: +6.5%, AOA: +35.45% | High | [83] |
| Pearl millet, finger millet, buckwheat | 22°C (buckwheat), 30°C (millets), 72 h | Altered enzyme activity and metabolic rates affecting bioactive compound levels. | TPC, TAN, AOA | TPC finger: -52.0%, TPC pearl: -42.0%, TPC buckwheat: +55.3%, AOA buckwheat 22°C: +89.5%, AOA buckwheat 30°C: +97.9%, TAN finger: -82.5%, TAN pearl: +373.1%, TAN buckwheat: -33.4% | Medium | [85] |
| Coix | 29°C, 24 h germination, 36°C 10 h soaking | Activation of glutamate decarboxylase enzyme and degradation of macromolecules. | GABA, SOLP, FAA | GABA: +683%, SOLP: +31.9%, FAA: +41.3% | Medium | [64] |
| Seven grains: various cereals and buckwheat | 16.5°C, 98% RH, 120 h darkness, intermittent watering, aeration | Enzymatic activation degrading proteins, carbohydrates and phytic acid. | TPC, PHEA, FLVN (QUER, RUTI, TRIF), GABA, ARBX, inositol phosphates | GABA rye: +700%, ARBX wheat: +33%, α-amylase inhibition barley: +650%, α-glucosidase inhibition sorghum: +25%, AOA rye: +51% | Medium | [72] |
| Wheat and triticale | 24°C germination, 1 mM GABA 3 h soaking | GABA reduces oxidative stress, increases antioxidant activity through catalase activation. | TPC, ANTH | TPC wheat: +29%, ANTH triticale: +92%, Germination: +18-21% | Medium | [89] |
| Wheat, barley, sorghum | 20°C, 96 h | Hydrolytic enzyme activation and Maillard reaction producing endogenous antioxidants. | TPC, AOA, PHEA, FLVN | TPC wheat: +181%, TPC breakfast wheat: +181%, TPC barley: +69%, TPC breakfast barley: +72%, TPC sorghum: +102%, AOA wheat: +107%, AOA barley: +42%, AOA breakfast barley: +158%, AOA sorghum: +16% | Medium | [47] |
| Type of Inductor | Species Studied | Optimal Parameters | Mechanism of Action | Bioactive Compounds | Quantitative Results | Ref. |
| Plasma-activated water (PAW) | Wheat | PAW-3 treatment, 15 mm distance, Ar-O₂ gas (98% Ar, 2% O₂), 40 L/min, 600 W | Reactive nitrogen species act as a nitrogen source and signaling molecules for metabolism. | TPC, CHL-a, CHL-b, CAROT, SOLP, ASCA, GABA, FAA | TPC: +10.46%, CHL-a: +89.46%, CHL-b: +112.46%, CAROT: +91.58%, SOLP: +19.48%, GABA: +32.56%, FAA: +28.23%, SOD: +47.12% | [28] |
| Plasma-activated water (PAW) with atmospheric plasma jet (APPJ) | Barley | Treatment C: 30 min APPJ, 4.5 h soaking, 19 h air rest, 15°C, 76% RH | PAW contains reactive oxygen/nitrogen species that degrade DON through oxidation. | α-AMY, β-AMY, β-GLU | β-AMY: +18.8%, germination: improved acrospire growth | [40] |
| High hydrostatic pressure (HHP) and soaking | Buckwheat | Soaking 40°C 4 h, 600 MPa 30 min single cycle | Pressure modifies protein-starch structure, increases cell permeability, improves extraction. | TPC, AOA | TPC: +16%, AOA: +2.5% | [104] |
| Constant High Voltage Electric Field (CHVEF), Alternating High Voltage Electric Field (AHVEF), Pulsed High Voltage Electric Field (PHVEF) | Winter triticale | AHVEF (3 kV·cm-1, 60s) for germination energy and uniformity | Redistribution of electric charges altering physicochemical processes and intensifies biological processes. | Not analyzed | Root length: +28.7%, grains per spike: +31.0%, grain yield: +57.8%, germination uniformity: +4% | [107] |
| Pulsed electric field (PEF) | Wheat | PEF 6 kV·cm-1, 50 pulses prior to imbibition | PEF increases cell membrane permeability through electroporation, facilitating water absorption. | TPC, CHL, CAROT, SOLP, FAA | TPC: +18.56%, CHL: +373%, CAROT: +34%, SOLP: +12.08%, AOA: +5.78% | [25] |
| Pulsed electric field (PEF) | Barley | 10 min pre-soaking in phosphate buffer, PEF 3.8 kV·cm-1, 100 pulses, 20 μs width | PEF causes electroporation, affecting ion transport and generating oxidative stress. | α-AMY, β-GLUC | α-AMY: -4%, β-GLUC: +12%, diastatic power: +2% with optimized treatment; α-AMY: -73%, β-GLUC: -87%, diastatic power: -45% with non-optimized treatment | [106] |
| Pulsed electric field (PEF) | Wheat | PEF 3 kV·cm-1, 200 pulses (19.8 kJ/kg) before first hydration cycle, or 100 pulses (9.9 kJ/kg) after first cycle | Temporary cell membrane electroporation facilitates water absorption and enzyme activation. | α-AMY, β-AMY | α-AMY: +104%, β-AMY: +25%, water absorption: +25%, water retention: +15% | [37] |
| Pulsed electric field (PEF) | Wheat | 161.8 Hz, 6.1 J, 19.5 s | PEF creates membrane pores, increasing permeability for nutrient and water absorption. | Not analyzed | Germination rate: +10%, normal seedlings: +28% | [35] |
| Static magnetic field | Triticale | 3.72 mT, 6 h, 10°C | Acceleration of the germination process by modifying time parameters. | Not analyzed | Maximum germination: +9%, time to 50% germination: -12.4%, time to 75% germination: -16% | [17] |
| Static magnetic field | Brown rice | 10 mT, 60 min, 25°C, followed by 24 h germination at 30°C in 5-10 mM GABA | Increased cell membrane permeability, reduced GABA-aminotransferase activity. | GABA | GABA: +207.6%, GABA-T activity: -16.14%, root length maintained <3 mm | [26] |
| High-pressure carbon dioxide (HPCD) | Barley | 57 bar, 25-35°C | Dissolved CO₂ penetrates seeds, modifies cellular pH affects key enzymes. | Not analyzed | Oat germination: -13.8% to -100%, barley germination: -100% | [105] |
| Gamma irradiation | Various millet varieties | 2.5 kGy, 12% moisture | Breakdown of macromolecules through the hydrolysis of chemical bonds from free radicals. | TPC, AOA | TPC: +24.5%, AOA DPPH: +55.6%, reducing power: +120.8% | [108] |
| Microwave irradiation | Tartary buckwheat | 300 W, 50 s, 25°C, 85% RH, 5 days darkness | Microwave increases the activity of key enzymes in the flavonoid biosynthesis pathway. | FLVN, PAL, CHI, FLS | FLVN: +31.78%, PAL: +6.50%, CHI: +8.64%, FLS: +14.55%, AOA: significant increase | [81] |
| Microwave | Bitter buckwheat | 600 W, 10 s prior to 7 days germination, 25±2°C, 85% RH | Microwaves activate key enzymes regulating gene expression of antioxidant enzymes. | FLVN, ANTZ (CAT, SOD), PHEA | FLVN: +377%, AOA: +264%, CAT: +300%, SOD: +58% | [108] |
| Ultra-high frequency microwave (UHF EMF) | Spring barley | 0.42 kW, 11 s, 7 days at 18°C, 60% RH | Microwaves induce ROS production, triggering protective responses including phenolic synthesis. | PHEC: CAFA, FERA, VANA, GACA, p-COU, SYRA, SALA | CAFA in leaves: +95.2%, FERA in leaves: +50.7%, VANA in leaves: +329.3%, SYRA in endosperm: +1871%, TPC in leaves: +167.6% | [109] |
| Light (different intensities) | Bitter buckwheat | 20°C, 99% RH, 6,000 lux for rutin/flavonoids, 600g buckwheat/plate, 5° inclination | Light stimulates flavonoid and phenolic compound biosynthesis as a protection mechanism. | RUTI, MYR, QUER, KAEM, TPC, TFC | RUTI:+34%, MYR highest at 6,000 lux, QUER highest at 6,000 lux, KAEM: +12%, TPC: +47%, TFC: +64%, AOA DPPH: +361%, AOA ABTS: +250% | [42] |
| Pulsed light (PL) | Corn | 6 h soaking, 400 pulses (0.50 J/cm²), 30°C, 90% RH, 48-72 h | Pulsed light improves membrane permeability, increases GAD activity, reduces GABA-T activity. | GABA, GLUAS, FAA, GAD, GABA-T | GABA: +27.20%, GABA vs non-germinated: +801%, GLUAS: +11.79%, FAA: +239.65%, GAD: significant increase, GABA-T: reduction | [111] |
| Pulsed light (PLT) | Brown rice | 300 pulses at 400 J, 28°C, 95% RH, 36 h | PLT promotes phenylalanine biosynthesis, carbohydrate and energy metabolism pathways. | GABA | GABA: >30% increase in all varieties, OsbZIP56 gene: +20% | [110] |
| Temperature and photoperiod (light) | Common buckwheat | 16°C, 20/4 h light/dark photoperiod, 288 h | Moderate abiotic stress activates defense mechanisms increasing secondary metabolite synthesis. | CHL, CAROT, ANTH, TPC, TFC, AOA | TPC: +76.6%, TFC: +20%, CAROT: +18.19%, CAROT with extended photoperiod: +21.34%, CHL: +35.40%, AOA: +15% | [27] |
| Type of Inductor | Species Studied | Optimal Parameters | Mechanism of Action | Bioactive Compounds | Quantitative Results | Ref |
| UV-A LED Light | Sorghum) | 35°C, 98% RH, 28 h germination, 36 h sprouting, 11.9 h UV-A at 5.1 μW/cm² | UV-A activates antioxidant defense systems and stimulates phenolic biosynthesis. | TPC, GACA, PROTA, p-COU, FERA, SINA, CATCH | TPC: +143.57%, FPHE: +210.45%, AOA: +168.86% | [13] |
| UV-B light (280-311 nm) | Mexican blue corn | 26.9°C, 80-90% RH, 207.7 h, UV-B 37.0 h after 96 h | Activation of PAL and TAL enzymes converting amino acids into secondary metabolites. | TPC, ANTH, GABA | TPC: +587.2%, FPHE: +1148%, BPHE: +469%, ANTH: +29.9%, GABA: +199.9%, AOA ABTS: +133.9%, AOA DPPH: +173.4% | [21] |
| UV-B light (wavelengths between 280 and 315 nanometers) | Highland barley | Germination: 72 h at 25°C. UV-B radiation: 0.2 W m⁻² for 6 h/day for flavonoids; 0.2 W m⁻² for 6 h/day for polyphenols; 0.2 W m⁻² for 12 h/day for riboflavin; 0.2 W m⁻² for 3 h/day for GABA. | UV-B radiation activates defense mechanisms that stimulate the synthesis of secondary metabolites such as flavonoids and polyphenols, increases the activity of enzymes such as glutamate decarboxylase (GAD) for GABA synthesis, and accelerates the redox process for riboflavin synthesis. | TFC, TPC, RIBO, GABA, β-GLU. | TPC:: +49.4%. GABA: +40.21% | [73] |
| UV-B light (UV-B) | Buckwheat or common buckwheat | 28.7°C, 3.0 days, UV-B 30.0 μmol·m⁻²·s⁻¹ for 7.6 h/day | UV-B increases activity and gene expression of phenylpropanoid pathway enzymes. | FLVN, TPC, AOA | FLVN: +97%, TPC: +54%, AOA DPPH: +54%, AOA ABTS: +66%, AOA FRAP: +54% | [113] |
| UV-C light (200-280 nm) | Amaranth | 3 cm distance, 15 min exposure | Oxidative stress induces defense mechanisms, stimulating phenolic compound biosynthesis. | TPC, TFC, CAFQ, TRYP, p-CQA, FERQ | TPC: +196%, p-CQA: +17.7%, TRYP: +12.4% | [66] |
| UV-C light (254 nm) | Wheat .and Chia | UV-C 120 min chia (35.7×10⁴ J m⁻²), 180 min wheat (141.7×10⁴ J m⁻²), germination 25°C | UV-C produces photooxidation affecting biological molecules and activating protective enzymes. | TPC, AOA | AOA chia: +317%, AOA wheat: +78%, TPC: no significant effect | [48] |
| Plasma (low pressure plasma and sliding arc plasma) | Barley, Wheat, Triticale | Low pressure plasma, 5 min | Not specifically reported | Not reported | Low pressure plasma: +18%, atmospheric plasma: -58% | [116] |
| Cold Atmospheric Pressure Plasma (CAPP) | Barley | 10-20 s for ambient air/nitrogen, 10-30 s for oxygen, 24±2°C | RONS act as signal molecules modulating germination pathways and activating enzymes. | ANTZ | SOD: +40%, G-POX: +132%, germination acceleration: +56%, root growth: +20.6%, sprout weight: +19% | [118] |
| Dielectric Barrier Discharge (DBD) air atmospheric plasma | Rice | DBD plasma for 60 s | Surface modification increases hydrophilicity and water absorption capacity. | CHL-a, CHL-b, CAROT | Germination: +9.0%, vigor index: +18.0%, germination speed: +7.3%, CHL-b: +10.3%, CAROT: +7.6% | [114] |
| Surface Barrier Discharge (SBD) plasma | Various winter and spring cereals | SBD plasma for 60 min (winter wheat), 24°C | SBD plasma products generate ROS inducing moderate oxidative stress. | Not analyzed | Shoot length wheat: +31%, root length wheat: +15% (30 min), +33% (60 min), +21% at 6 days | [41] |
| Atmospheric pressure plasma (SDBD) | Barley | 6 min SDBD plasma, 51.7 W, 8 kVp-p, 14.4 kHz, 15°C, 16/8 h light/dark | ROS/RNS modify physico-chemical properties, acidification and supply of nitrate ions. | TPC, SAPO, GABA, POLI | TPC: +9%, SAPO: +50%, GABA: +40%, POLI: +90% | [22] |
| Cold plasma (CP) and electromagnetic field (EMF) | Common buckwheat | CP7 for ‘VB Nojai’, CP5 for ‘VB Nojai’, EMF15 for ‘VB Vokiai’ | Changes in EPR signal, phytohormone balance, ROS production during germination. | Not analyzed | In vitro germination: 100% in all groups, germination time: -7%, field emergence: -13% to -20% | [115] |
| Cold plasma (DBD) | Brown rice | Plasma at 135 W, 75 s, argon flow 22 mL/min, germination 25-28°C, 1-1.5 days | Plasma causes “surface etching” facilitating water absorption and activating pathways. | TPC, TOCO, γ-ORY, ANTH, PHYS | TPC: +86%, TOCO: +290%, γ-ORY: +80%, ANTH: +38%, PHYS: +40.6%, TERP: +80.5% | [24] |
| Cold plasma (microwave discharges) | Barley, corn | Barley: Ar-20%O₂ 180 s at 4 mbar; corn: Ar-20%O₂ 240 s + N₂-2%O₂ 120 s at 8 mbar | ROS/RNS act on seed surfaces; atomic oxygen species are main contributors. | Not analyzed | No significant effect on germination, slight positive effect on root/shoot length | [119] |
| Atmospheric cold plasma (CAP): DBD and APPJ | Various cereals | APPJ Ar+O₂ 11 days for barley, APPJ Ar+air 10 days for wheat | Surface etching creating micropores, chemical modification by RONS, hydrophilic changes. | Not analyzed | Root dry mass rye: +15.6%, barley: +16.2%, wheat: +14.3%, germination rate barley: +21.4%, oats: +28.8%, wheat: +33.3% | [117] |
| Ultrasound | Oat | Soaking 4 h at 23-24°C, ultrasound 5 min at 25 kHz (16 W/L), germination 72-96 h at 24±2°C, 95±3% RH | Ultrasound alters grain microstructure through acoustic cavitation, creating micropores. | GABA, AVEN, TPC, AOA | GABA: +256.9%, AVEN 2c: +3403.2%, AVEN 2p: +2024.6%, AVEN 2f: +1267.6%, TPC: +11.24%, AOA: +72.45% | [5] |
| Ultrasound | Corn | 45 kHz, 30°C, 30 min, germination at 30°C, 90% RH for 60 h | Cavitation generates mechanical force and thermal effects, disrupting biological structures. | GABA | GABA: +30.55% | [38] |
| Ultrasound (40 kHz, 30 min) | Brown rice | 40 kHz, 30 min, germination 36 h at 28±1°C | Ultrasound creates pores in seed coat improving water absorption and oxygenation. | GABA, FAA, BIOP | Germination: +28%, metabolomic profile changes | [120] |
| High intensity ultrasound (HIU) | Brown rice | HIU: 28 kHz, 17.83 W/cm², 5 min, germination at 37°C for 32 h | HIU acts as abiotic elicitor generating cellular stress and activating secondary metabolites. | GABA, FAA, ANTZ, PROL, MINB | GABA: +56.92%, amino acid index: +137.5%, AOA FRAP: +43.9%, iron bioaccessibility: +147.1% | [29] |
| Ultrasound and controlled germination | Red spring wheat, white soft wheat | 28±2°C, 95±3% RH, 6 h soaking, 72 h germination, 30 min ultrasound at 25 kHz | Ultrasound causes acoustic stress increasing GAD activity and GABA production. | GABA, SOLDF, INDF, TDF | GABA: +339%, GABA with ultrasound: +30.7%, SOLDF in SW: -18.4%, glucose in HW: +471% | [4] |
| Type of Inducer | Species Studied | Optimal Processing Parameters | Mechanism of Action | Bioactive Compounds Analyzed | Quantitative Results | Ref |
| Vegetable salts (ashes) | Corn | Atp-Y: 25.12 h at 25.54°C, 0.52% salt, 144.37 h germination, 37.65 h maturation. Coca-sr: 1.608 h at 36.63°C, 1.11% salt, 144.37 h germination, 27.07 h maturation | Enzymatic activation, antinutrient-protein complex destruction, metallic cofactor release. | TPC, TFC, CONP, HYDT, SAPN | TPC Coca-sr: +72.4%, TFC Coca-sr: +126.4%, AOA DPPH Coca-sr: +89.9%, AOA FRAP Coca-sr: +193.6% | [49] |
| Chitosan, jasmonic acid (JA), salicylic acid (SA) | Common buckwheat | Chitosan 0.1%, JA 150 μM, 72 h, 25°C | Inducers activate key enzymes in phenylpropanoid pathway, increasing phenolic biosynthesis. | GACA, CATCH, CHLA, CAFA, EPIC, RUTI | Chitosan: TPC: +23%, GACA: +51%, CATCH: +72%, CHLA: +69%, EPIC: +122%; JA: TPC: +147%, RUTI: +138%, CAFA: +48%, EPIC: +695% | [19] |
| Slightly acidic electrolyzed water (SAEW) | Brown rice | SAEW pH 5.5-6.0, redox potential 940-968 mV, available chlorine 10 ppm, 35±1°C, 85% RH, 48 h darkness | Moderate stress activates enzymes, particularly GAD, initiating signal transduction cascade. | GABA, ASCA, FERA, p-COU, QUER, TPC, TFC, FAA | TPC: +743%, TFC: +578%, GABA: +299%, FERA: +2751%, p-COU: +1339%, ASCA: detected only in SAEW, QUER (SAEW): -69%, QUER (water): +154%, AOA DPPH: +839%, AOA ABTS: +792%, AOA FRAP: +934% | [10] |
| Hydrogen-rich water (HRW) | Wheat | HRW 4 h, 23±2°C, 50% RH | HRW acts as molecular signaler activating antioxidant enzymes and increasing Dreb1 gene expression. | Not analyzed | Germination: +21.1%, vigor index: +84.7%, chlorophyll: +76.8% | [122] |
| 3% sucrose and 7.5 mM calcium chloride | Common buckwheat | 3% sucrose + 7.5 mM CaCl₂, sprayed every 6 h, 8 days, 25°C, 60% RH, darkness | Sucrose induces defense response; calcium acts as secondary messenger in signal transduction. | TPC, TFC, GABA, ORI, ISOV, VITX, RUTI, ASCA, β-CAR, TOCO | TPC: +64%, TFC: +58%, GABA: +59%, ORI: +43%, ISOV: +30%, VITX: +36%, ISOV: +38%, RUTI: +34%, ASCA: +10%, α-TOCO: +31%, AOA DPPH: +51% | [16] |
| Acidic medium | Brown rice | pH 2.7, 25°C, 12 h | Acidic conditions facilitate phytate complex degradation by increasing solubility. | PHYA, PHYT | PHYA: -46.2%, PHYT: +402%, calcium bioaccessibility: +32.9%, zinc bioaccessibility: +44.4% | [30] |
| Gaseous ozone (O₃) | Spring malting barley | 50 ppm, 1-6 h, 20°C, 98% RH, 19.8% grain moisture | Not specifically reported | Not analyzed | Germination energy: 96.0% vs 99.3% in control, significant reductions after 1 h | [124] |
| Gaseous ozone (O₃) | Wheat | Ozone: 3 h at 50 ppm, flow 1 L min⁻¹; Soaking: 24 h at 22±2°C (1:2); Germination: 72 h at 22±2°C, 80% RH, darkness | Ozone induces oxidative stress triggering defense responses with moderate exposure. | TPC, AOA | TPC grains: +1.5% (3 h), -56.2% (4 h), -54.8% (5 h); TPC germinated: -15.5% (3 h), -19.5% (4 h), -18.9% (5 h); AOA grains: +2.4% (3 h), -13.0% (4 h), -10.0% (5 h) | [39] |
| Citric and lactic acid | Adlay | 1% citric acid, 12 h at 25°C, germination 60 h at 25°C, 95% RH, darkness | Citric acid stimulates phenolic biosynthetic pathway by increasing PAL activity. | TPC, TFC | TPC citric acid: +18.3%, TPC lactic acid: -14.9%, TFC citric acid: +17.0%, TFC lactic acid: -9.0%, AOA citric acid: +39.1%, AOA lactic acid: -16.8% | [123] |
| Sodium Chloride (NaCl) | Wheat | 48 h at 17°C, 80% RH, darkness, 60 mM NaCl solution | NaCl alters ionic strength, stabilizing enzymatic structures and increasing activity. | TPC | TPC: +242.3% | [121] |
| Sodium Chloride (NaCl) and CaCl₂ supplement | Yellow corn | 16 h water soaking, 8 h 300 mM NaCl + 5 mM CaCl₂, 72 h germination at 24±1°C in darkness | NaCl induces oxidative stress activating antioxidant systems; CaCl₂ regulates gene expression. | CAROT: LUT, ZEAX, α-CRY, β-CRY, α-CAR, β-CAR | LUT NaCl+CaCl₂: +21.50%, LUT CaCl₂: +36.55%, ZEAX NaCl+CaCl₂: +30.18%, α-CRY NaCl+CaCl₂: +23.33%, AOA ORAC NaCl: +127.80% | [18] |
| Sodium chloride (NaCl) | Quinoa | 300 mM NaCl, 24°C, 240 h (10 days) | Salt stress induces oxidative stress activating plant’s antioxidant system. | TPC, TFC, FLVL, ANTH, AOA, ANTZ | TPC sprouts: +152%, TFC sprouts: +243%, FLVL sprouts: +92%, ANTH sprouts: +238%, AOA DPPH sprouts: +3700% | [23] |
| Sodium silicate and iron chelate (Fe-EDTA) | Common buckwheat | 7 days, 4 mM sodium silicate (SIL) or SIL + 0.5 mM Fe-EDTA (SIL-Fe), 15 min immersion twice daily | Modification of phenolic profile and alteration of micronutrient absorption. | FLVN, PHEA | FLVN SIL-Fe: -20.6%, PHEA SIL: +11.2%, EPIC SIL-Fe: -24.2%, CAFA esters SIL: +80.8%, Fe SIL-Fe: +335%, Si SIL-Fe: +204% | [33] |
| Zinc oxide nanoparticles (ZnO NPs) | Pearl millet | 150 ppm ZnO NPs, 6 h imbibition, 28°C | Activation of pre-germinative metabolism increases enzymatic activity and mobilization. | Not specifically analyzed | Germination: +20%, vigor index: +51% | [125] |
| Zinc oxide nanoparticles (ZnONPs) | Corn | 700-1000 mg/L ZnONPs for germination, 800 mg/L for carotenoids | ZnONPs participate in chlorophyll biosynthesis and protect SH groups. | CHL, TPC, CAROT, PROL | CHL: +170.8%, TPC: +3.4%, CAROT: +221.5%, PROL: +66.8% | [77] |
| Gibberellic acid (GA₃) and abscisic acid (ABA) | Wheat | GA₃ 150 μM, 12 h, 25°C, 16 h photoperiod | GA₃ promotes α-amylase mRNA synthesis and hydrolytic enzymes in aleurone layer. | α-AMY | α-AMY Yangmai 13: +13.5 mg g⁻¹·min⁻¹, α-AMY Yannong 19: +12.5 mg·g⁻¹·min⁻¹ | [32] |
| Phytohormones (IAA, SA, GA) | Wheat | IAA: 0.01 mg/mL, GA: 0.01 mg/mL, SA: 0.001 mg/mL or combination, 72-120 h, room temperature, darkness | Phytohormones act as elicitors generating oxidative stress inducing PAL activity. | TPC, TFC, FERA, CINA, p-COU, SYRA, CAFA, VANA, QUER, KAEM, LUTL, APG, CATCH, TRIC, GABA | TPC IAA+GA+SA: +128%, TFC IAA+GA+SA: +182%, FERA: +949.3%, NARG: +438.3%, TRIC: +76%, GABA: -64%, AOA DPPH: +106%, AOA FRAP: +108% | [31] |
| Vitamin B6 (Pyridoxal phosphate, PLP) | Bitter buckwheat | 2 days at 22°C, 75% RH, darkness, then 2.5 mM PLP at 30°C for 24 h | GAD activation, increased glutamate substrate, positive regulation of gene expression. | GABA, TFC, TPC | GABA: +867%, GLUAS: +175%, FLVN: +11%, TPC: +33%, AOA DPPH: +23%, AOA ABTS: +31%, ACE inhibition: +135% | [14] |
| Type of Inducer | Species Studied | Optimal Parameters | Mechanism of Action | Bioactive Compounds Analyzed | Quantitative Results | Refer |
| Cyanobacteria extracts and biologically synthesized silver nanoparticles (AgNPs) | Barley and wheat | AgNPs synthesized with 1.0 mM AgNO₃ at 30°C in light, smallest particles (7.3-28 nm) | Not clearly specified for germination process, possible protective and antimicrobial properties. | Not analyzed | GRI barley cv. Giza 123: +3.7%, GVC barley cv. Giza 123: +1.1%, MGT barley cv. Giza 123: -7.1% | [67] |
| Fermentation, soaking and controlled germination | Corn | Soaking: 24 h at 18°C (1:3 w/v), germination: 80 h at 18°C, fermentation: 24 h at 30°C with L. plantarum 299v | Activation of endogenous phytases during germination and soaking; microbial phytase production. | PHYA, MINB (Fe, Zn) | PHYA FLp-SG: -85.6%, PHYA FYLc: -68.7%, PHYA FLp: -65.3%, PHYA FSp: -51.8%, PHYA germination: -31.9%, PHYA soaking: -12.6%, phytate molar ratio FLp-SG: -81%, phytate molar ratio FLp-SG: -85% | [15] |
| Spontaneous fermentation and germination | Red sorghum and pearl millet | Germination: 48 h at 30°C with intermittent water spraying, drying at 50°C for 12 h, fermentation: 48 h at 30°C | Enzymatic and microbial degradation of antinutritional compounds improving mineral bioavailability. | TPC, TFC, TDC, PHYA, AOA | Red sorghum (G+F): TPC: -69%, TFC: -54%, TDC: -89.2%, PHYA: -90.1%, AOA DPPH: -30%, AOA ABTS: +3%; Pearl millet (G+F): TPC: -26.3%, TFC: -56.9%, TDC: -86.1%, PHYA: -85.1%, AOA DPPH: -69.1%, AOA ABTS: +11% | [9] |
| Fermentation with Rhizopus oligosporus (SSF) in two systems: traditional plate fermentation (PF) and bioreactor fermentation (BF) | djulis | Germination: 4 days at room temperature (42.6 ± 9.5 mm sprouts), fermentation: bioreactor at 35°C, 0.4 vvm aeration, 5 rpm rotation, 4 days | Hydrolytic enzymes decompose polysaccharides and proteins; microbial enzymes degrade complex molecules. | TPC, FLVL, ISOF, CHL, ANTH, GABA, FAA, QUER, RUTI, TRIF | AOA: +101%, FAA: +172%, FPHE: +23%, CAROT: +37%, CHL-a: +13%, CHL-b: +133%, ANTH: +134% | [12] |
| Fermentation and controlled germination | Naked barley | Germination: 30°C, 48 h, 80-85% RH; germination-fermentation: 48 h germination + 24 h fermentation at 35°C | Activation of GAD converting glutamate to GABA; degradative enzyme activation; PAL activation. | GABA, β-GLU, TPC, AOA, VOLA | GABA germination: +116.63%, GABA germination-fermentation: +87.53%, GABA soaking: +78.51%, TPC germination-fermentation: +68.39%, TPC germination: +26.21%, AOA DPPH germination-fermentation: +267.46%, AOA DPPH germination: +146%, AOA ABTS germination-fermentation: +36.1%, β-GLU germination: -9.68%, β-GLU germination-fermentation: -5.51% | [11] |
| Natural fermentation, soaking and roasting | Blue corn | Soaking 16 h, germinate at 25°C for 72 h with periodic watering | Hydrolytic enzyme activation degrades antinutritional factors, increasing mineral availability. | TPC, ANTH, AOA | TPC germination: +36.02%, TPC fermentation: +14.88%, TPC roasting: -20.38%, ANTH germination: -3.11%, ANTH fermentation: -3.23%, ANTH roasting: -5.07%, AOA germination: +81.07%, AOA fermentation: +39.28%, AOA roasting: -6.53% | [44] |
| Fermentation and controlled germination | Foxtail millet | Soaking: 12 h at 25°C (1:15), germination: 48 h at 25°C in darkness, fermentation: 20 h at 38°C with lactic acid bacteria to pH 3.0 | Enzymatic activation during germination and microbial enzyme production during fermentation. | TPC, TFC | TPC combined: +98.2%, TFC combined: +16.6%, AOA DPPH combined: +81.5%, AOA FRAP combined: +33.5%, reducing power combined: +184.5% | [34] |
| Combination of Inductors | Reported Synergies | Species Studied | Bioactive Compounds Analyzed | Quantitative Results | Reported Limitations | Ref. |
| Ultrasound and fermentation with complex starter culture | Synergy between ultrasound treatment and fermentation with complex starter culture | Spring soft wheat and spring barley | GABA, TFC, AOA | GABA wheat Zauralochka: +370%, GABA wheat Erythrosperium: +210%, GABA barley Chelyabinets: +220%, TFC wheat Zauralochka: +35%, TFC wheat Erythrosperium: +45%, TFC barley Chelyabinets: +68%, AOA wheat Zauralochka: +31%, AOA wheat Erythrosperium: +38%, AOA barley Chelyabinets: +51%. | Germination and fermentation reduce starch content | [60] |
| Controlled germination, gibberellic acid (GA3), indole-3-acetic acid (IAA), KNO₃, MgSO₄, H₂O₂, ascorbic acid (AA) and H₂O | GA3 and KNO3 showed synergistic effects for wheat and oats, while GA3 and IAA were more effective for barley | Wheat, barley and oats | CHL (a+b), CHL-a, CHL-b, CAROT | Wheat with MgSO₄: CHL: +35%, wheat with H₂O₂: CHL: +15%, barley with H₂O₂: CHL: +150%, oats with IAA: CHL: +77%, CAROT in wheat with KNO₃: +63%, wheat germination with GA₃: +12% | Variation in response depending on cereal type | [36] |
| Pretreatments (soaking, ultrasound and alkaline thermo-hydrolysis) + controlled germination | Ultrasound pretreatment combined with germination shows synergistic effects | Buckwheat and quinoa | TPC, FLVN (RUTI, QUER, KAEM, CHRY, HESP, CATCH, EPIC), PHEA | Buckwheat + ultrasound/germination: TPC: +34%, AOA: +20%, FLVN: +201%; Quinoa + ultrasound/germination: TPC: +8%, AOA: +64%, FLVN: +43%; Buckwheat + thermo-alkaline: TAN: -83%; Quinoa + ultrasound: PHYA: -85.5% | Alkaline treatment decreases TPC | [59] |
| UV-B radiation and CaCl₂ supplement | Combined treatment shows synergistic effects for carotenoid enhancement | Yellow corn | CAROT: LUT, ZEAX, α-CRY, β-CRY, α-CAR, β-CAR | LUT: +77.38%, ZEAX: +121.07%, α-CRY: +75.19%, β-CRY: +65.52%, α-CAR: +79.17%, β-CAR: +86.49%, SOD: +14.12%, POD: +2.66% | UV-B radiation inhibits growth | [65] |
| Ultrasound and selenium (selenium enrichment with sodium selenite) | Synergy between ultrasound and low drying temperature (50°C) | Black rice | PHEA (GACA, CHLA, ELLA, FERA, HYBA, ISOFA, p-COU, PROTA, SINA, VANA), FLVN (KAEM, QUER, RUTI), ANTH | GACA: +271%, PROTA: +268%, CYA3GL: +732%, total phenolics: +146% with 10 min ultrasound, 50°C | Limited ultrasound time | [68] |
| Potassium-enriched biochar (KBC) and gibberellic acid (GA₃) | GA₃+KBC synergy for increasing germination, chlorophyll content and reducing antioxidant enzymes under osmotic stress | Wheat | CHL-a, CHL-b, ANTZ | CHL-a: +34.35%, CHL-b: +20.96%, Total chlorophyll: +29.12%, electrolyte leakage: -11.02% with GA₃+KBC under low stress | More field-level research needed | [69] |
| Hormopriming (GA₃, IAA), halopriming (KNO₃, MgSO₄), osmopriming (H₂O₂, ascorbic acid), hydropriming (distilled water) | No combinations of inductors reported in this study | Wheat | TPC, TFC, PHEA, FLVN, ANTZ | TPC KNO₃: +16.7%, TFC H₂O₂: +5.9%, CATCH H₂O: +60.3%, CHLA GA₃: +1008.8%, HYBA GA₃: +1183.3%, SINA H₂O: +113.3%, RUTI H₂O: +158.3%, NARG GA₃: +214.5%, QUER GA₃: +282.4%, AOA KNO₃: +20.8% | Study limited to laboratory conditions | [75] |
| Controlled germination and thermal treatment (tempering and roasting) | Synergy between germination and thermal treatment for phenolic compounds and antioxidants | Rice | TPC, AOA | TPC green malt roasted (150°C, 45 min): +113.8%, TPC tempered malt roasted (125°C, 90 min): +83.1%, amino acids (50°C, 60 min): +80%, AOA tempering (60-90 min): +46.21% | High temperatures (>175°C) reduce bioactives | [45] |
| Ultrasound (US) and pulsed electric field (PEF) | Synergistic effect between US and PEF, resulting in greater bioactive increase | Wheat | TPC, TFC, CHL, AOA | TPC US+PEF: +8.59%, TFC US+PEF: +14.06%, CHL US+PEF: +12.06%, AOA DPPH US+PEF: +8.58%, AOA ORAC US+PEF: +2.34% | Long-term stability unknown | [76] |
| Ultrasound (US) and microwave (MW) | No specific synergies reported between inductors | Sorghum | GABA, TPC | GABA ultrasound (15 min): +182%, GABA microwave (10% power): +117%, TPC ultrasound (20 min): +18.8%, AOA ultrasound (10 min): 84.53% vs 60.61% in control | Prolonged ultrasound times reduce effectiveness | [126] |
| Application | Recommended Inductors | Ideal Matrices | Benefits and Characteristics | Development Status | Technological Challenges | Market Opportunities | Ref |
| Foods with ACE inhibitory activity | Specific inductors (PLP, chitosan) | Barley, buckwheat, rice | High GABA content; ACE inhibitory peptides; phenolic compounds (rutin, catechin) with antihypertensive activity; reduction of factors that promote hypertension | Research | Clinical validation; effective concentrations in final product; stability during processing; optimization of treatment conditions | Functional foods for blood pressure control; preventive products for cardiovascular health; foods targeted at hypertensive population | [10,11,14,19,38] |
| Foods with improved mineral bioaccessibility | Germination combined with lactic acid fermentation | Sorghum, millet, corn | Significant reduction of phytates, tannins, oxalates and other antinutrients; increase in bioavailability of iron, zinc and calcium; moderate increase in phenolic compounds | Research/ Pilot | Prolonged processing time; optimization by cereal type; balance between antinutrient reduction and bioactive preservation | Naturally fortified food products; foods for populations with micronutrient deficiencies, especially women and children in Africa | [9,15,30,62] |
| Sports foods | Ultrasound + low-intensity microwave | Oats, rye, corn | High protein content with branched-chain amino acids; bioactive peptides with regenerative properties; GABA for recovery; antioxidants to reduce post-exercise oxidative stress; gradually absorbed carbohydrates; bioavailable minerals | Pilot | Standardization; organoleptic profile; formulation for different consumption times; stability | Muscle recovery; sports performance; pre/post-training products; natural alternatives to synthetic supplements |
[44,126] |
| Infant foods | Low-temperature germination (28°C) + ultrasound | Quinoa, amaranth, millet | Improved essential amino acid profile; increase in B vitamins; reduction of antinutritional factors; increased DHA and essential fatty acids; bioavailable phenolic compounds; higher protein digestibility | Research | Microbiological safety; digestibility; allergenicity; sensory acceptability; stability during processing | Nutritious porridges and snacks for cognitive development; foods for early childhood; premium organic products | [5,15] |
| Foods for glycemic control | Cold plasma (CAPP and DBD) + germination | Rice, barley, wheat, buckwheat | Modification of starch structure for reduced digestibility; increase in phenolic compounds with α-amylase and α-glucosidase inhibitory properties; increase in soluble fiber; high GABA content; bioactive compounds with hypoglycemic activity | Research | Specialized plasma equipment; parameter optimization according to cereal; in vivo validation; variable effect depending on variety | Foods for prevention and management of diabetes; low glycemic index products; foods for weight control | [22,29,93,118] |
| Foods for brain health | Hydrogen-rich water (HRW) + controlled germination | Wheat, rice, buckwheat | Increase in GABA, improvement in enzymatic antioxidant content (SOD, CAT), increase in neuroprotective phenolic compounds, higher bioactive protein content, stimulation of metabolic pathways related to neuroprotection | Research | Stability of dissolved hydrogen; specific equipment; optimization of treatment conditions; partially understood mechanisms of action | Foods for cognitive improvement; products for prevention of age-related mental deterioration; functional foods for students and professionals | [26,28,120,122] |
| Functional beverages | Germination + lactic fermentation | Oats, barley, quinoa | High content of β-glucans with hypocholesterolemic properties; avenanthramides with anti-inflammatory activity; phenolic compounds and flavonoids with antioxidant capacity; bioactive peptides with ACE inhibitory activity | Commercial (limited) | Stability during shelf life; processing that preserves bioactives; standardization of content; limited solubility | Products for cardiovascular health; premium plant-based beverage market; athletes and fitness | [9,11,15,44] |
| GABA-rich functional beverages | Plasma-activated water (PAW) + controlled germination | Barley, rice, wheat | High GABA content; increase in total free amino acids; increase in antioxidant enzymes; higher chlorophyll content; release of phenolic compounds with antioxidant activity; improved sensory profile (reduction of undesirable volatile compounds) | Research | Limited shelf life of PAW; industrial scalability; precise control of reactive species; need for specialized equipment | Beverages for stress reduction; products to improve sleep quality; functional foods for hypertension; fermented beverages with probiotic properties | [28,38,40,110] |
| Sprouts enriched with specific phytonutrients | Moderate abiotic stress (salinity, CaCl₂, sucrose) | Buckwheat, rice, corn | Specific increase of target compounds: carotenoids, GABA, flavonoids (rutin, quercetin, kaempferol), specific phenolic acids (ferulic, p-coumaric); metabolic adaptation that increases antioxidant defense systems | Research | Balance between stress to induce bioactive compounds and acceptable yield; optimization by species; knowledge transfer to commercial scale | Functional foods directed at specific conditions; ingredients with specific health claims; components for specialized supplements | [16,18,23,69] |
| Functional sprouts for direct consumption | Pulsed electric field (PEF 3-6 kV·cm-1) + cold plasma | Wheat, buckwheat, quinoa | Increased content of chlorophylls, GABA, flavonoids, carotenoids, antioxidant enzymes, significantly elevated antiradical capacity (DPPH, ABTS) | Research/ Pilot | Specialized equipment; precise parameter control; energy cost; variable yield depending on species | Ready-to-eat superfoods; functional living foods; components for smoothies and juices; nutritious vegetable snacks | [28,29,38,42,76] |
| Enriched breakfast cereals | Germination (malting) + controlled drying | Wheat, oats, barley, quinoa | Increase in total polyphenols, flavonoids, improved antioxidant activity (DPPH, ABTS, FRAP), increased B vitamins, Maillard reaction compounds with antioxidant properties, improved amino acid profile | Commercial | Balance between thermal processing and preservation of bioactive compounds; development of attractive sensory profiles; standardization of processes | Value-added cereals; products for premium segments; foods with “whole grain plus” claims; alternatives to conventional cereals | [45,47,82,90] |
| Bioactive concentrates | Specific inductors depending on target compound (MgSO₄, KNO₃, H₂O₂) | Rice, buckwheat, barley, sorghum | Standardized GABA concentrates for antihypertensive applications; extracts rich in avenanthramides for anti-inflammatory applications; concentrates of specific flavonoids (rutin, quercetin) with high bioavailability; purified bioactive peptides | Laboratory/Pilot | Purification; standardization; stability; scaling up of extraction processes; production cost | Ingredients for nutraceuticals; food supplements; medical foods; specialized food ingredients; techno-functional ingredients | [14,19,22,61] |
| Germinated seeds with optimized phytochemical profile | Static magnetic fields (1-10 mT) | Rice, buckwheat, wheat, barley | Significant increase in polyphenols, flavonoids, rutin, GABA, alteration of secondary metabolism without negatively affecting germination, modification of enzymatic activity in key biosynthetic pathways | Research | Equipment for magnetic field generation; optimization of intensity and exposure time; mechanisms of action not fully elucidated | Premium sprouts with specific functional properties; ingredients for food supplements; components for designer foods | [17,26,115] |
| Germinated seeds with improved sensory profile | Pretreatment with organic acids (citric, lactic) | Adlay, rice, millet | Moderate increase in total phenols, reduction of undesirable volatile compounds, improved amino acid profile, improved antioxidant activity, optimized texture | Research | pH optimization; balance between sensory profile and bioactive compounds; variability between cereals; microbiological control | Sprouts with better sensory acceptance; products for introduction to conventional consumer market; sensorially attractive functional foods | [8,30,121,123] |
| Functional flours | Moderate thermal treatments + salicylic acid | Wheat, rice, amaranth | Increase in alkylresorcinols with anticancer activity; total phenolic compounds; increase in GABA; improved soluble dietary fiber; reduction of antinutrients; higher mineral bioavailability | Pilot/Commercial | Maintaining technological properties; treatment homogeneity; quality control during processing; balance between bioactive compound content and functionality | Functional bakery; premium gluten-free market; foods with specialized nutritional value; products for diabetics | [2,124,126] |
| Germinated flours with improved antioxidant activity | UV-C light (200-280 nm) + controlled germination | Wheat, amaranth, millet | Increased total phenolic compounds; increased total flavonoids; improved antioxidant activity (DPPH); reduction of antinutritional factors; improved essential amino acid profile; structural modification of starch for better functionality | Research | Control of exposure time and distance; optimization of conditions according to cereal; balance between bioactive activity and yield | Ingredients for bakery with functional properties; premium flours for conscious consumers; foods with natural antioxidant claims | [44,48,62,66,80] |
| Naturally biofortified ingredients | Nanoparticles (ZnO, Fe) + germination | Corn, rice, millet | Significant increase in carotenoids, phenolic compounds, higher bioavailability of essential minerals, improved nutritional profile (amino acids, vitamins), increased stress resistance | Research | Controlled synthesis of nanoparticles; regulation and consumer acceptance; dose optimization; long-term safety evaluation | Naturally biofortified foods; products to combat nutritional deficiencies in vulnerable populations; alternatives to chemical fortification | [18,33,77,125] |
| Functional malts | Chemical elicitors (gibberellic acid, salicylic acid) | Barley, wheat, sorghum | Increase in enzymatic activity (α-amylase, β-glucanase) for better functionality; higher content of phenolic compounds and flavonoids; reduction of antinutrients during malting; optimized biochemical profile for specific applications | Pilot | Precise control of hormonal treatments; specific optimization according to variety; balance between enzymatic activity and bioactive compounds | Functional beers; malt extracts as ingredients; specialty malts for craft breweries; ingredients for bakery and pastry | [31,36,37,75,106] |
| Functional microgreens | Controlled germination with saline stress (NaCl 100-300 mM) + UV-B | Buckwheat, quinoa, barley | High flavonoid content, superior antioxidant capacity, increased GABA content, unique phytochemical profile with high levels of rutin, catechins and phenolic acids. Significant concentrations of anthocyanins in colored varieties. | Research/ Pilot | Precise control of stress conditions; commercial scalability; reduced shelf life; consistent quality control; batch-to-batch variability | High value-added foods; gourmet market; health-conscious consumers; ingredients for premium culinary applications | [13,21,23,27] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




