Submitted:
18 July 2025
Posted:
21 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Microbial Basis for H2 Production in Deplete Oil Reservoirs
3. Depleted Oil Reservoir Conditions and Technological Challenges
4. Infrastructure Adaptation for H2 Production in DORs
5. Carbon Capture, Utilization, and Storage
6. Safety Aspects of H2Au Production
7. Economic Aspects of the Process of Obtaining H2 Using DORs as Bioreactors
8. Pilot Projects and Case Studies
8.1. CEMVITA - H2 Production from DORs - The “H2Au” Approach
8.2. Recôncavo Baiano Studies: Application Potential of Cemvita-Inspired H2Au Production
9. Relevance of H2 from DORs and Its Broader Impacts
10. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANP | Brazilian National Agency of Petroleum, Natural Gas and Biofuels |
| CO2 | Carbon dioxide |
| H2 | Molecular H2 |
| CH4 | Methane |
| VFAs | Volatile fatty acids |
| MECs | Microbial electrolysis cells |
| DORs | Depleted oil reservoirs |
| CCUS | Carbon capture, utilization, and storage |
| DF | Dark fermentation |
| assA | alkyl succinate synthase |
| bssA | benzyl succinate synthase |
| assR | alkyl succinate regulator |
| bssR | benzyl succinate regulator |
| hydA | hydrogenase synthase |
| fdh | putative formate dehydrogenase synthase |
| TRL | Technology readiness level |
| H2S | Hydrogen sulfide |
| SRB | Sulfate reducing bacteria |
| CAPEX | Capital expenditures |
| OPEX | Operational expenditures |
| PNH2 | Brazil’s National H2 Program |
References
- S. Meherkotay, D. Das, Biohydrogen as a renewable energy resource—Prospects and potentials, International Journal of Hydrogen Energy 33 (2008) 258–263. [CrossRef]
- M. Yue, H. Lambert, E. Pahon, R. Roche, S. Jemei, D. Hissel, Hydrogen energy systems: A critical review of technologies, applications, trends and challenges, Renewable and Sustainable Energy Reviews 146 (2021) 111180. [CrossRef]
- G. Piggot, C. Verkuijl, H. Van Asselt, M. Lazarus, Curbing fossil fuel supply to achieve climate goals, Climate Policy 20 (2020) 881–887. [CrossRef]
- Q. Hassan, A.Z. Sameen, H.M. Salman, M. Jaszczur, Large-scale green hydrogen production using alkaline water electrolysis based on seasonal solar radiation, Energy Harvesting and Systems 11 (2024) 20230011. [CrossRef]
- S. Gouws, J. Mackay, Production of green hydrogen through PEM water electrolysis, Pure and Applied Chemistry 96 (2024) 1383–1401. [CrossRef]
- J.M. Filgueira, A.O. Pereira Júnior, R.S. Barbosa De Araújo, N.F.D. Silva, Economic and Social Impacts of the Oil Industry on the Brazilian Onshore, Energies 13 (2020) 1922. [CrossRef]
- R. Jain, N.L. Panwar, S.K. Jain, T. Gupta, C. Agarwal, S.S. Meena, Bio-hydrogen production through dark fermentation: an overview, Biomass Conv. Bioref. (2022). [CrossRef]
- Y. Cao, H. Liu, W. Liu, J. Guo, M. Xian, Debottlenecking the biological hydrogen production pathway of dark fermentation: insight into the impact of strain improvement, Microb Cell Fact 21 (2022) 166. [CrossRef]
- M.M. Albuquerque, W.J. Martinez-Burgos, G. De Bona Sartor, A.B.P. Medeiros, J.C. De Carvalho, C.R. Soccol, Microbial Electrolysis Cells in Biohydrogen Production, in: C.R. Soccol, S.K. Brar, K. Permaul, K. Pakshirajan, J.C. De Carvalho (Eds.), Biohydrogen - Advances and Processes, Springer Nature Switzerland, Cham, 2024: pp. 429–453. [CrossRef]
- B. Logan, Pennsylvania StateUniversity, Novel Microbial Electrolysis Cell Design for Efficient Hydrogen Generation from Wastewaters, (n.d.).
- D. Vieira Barboza, M. Jasmim Meiriño, S.R. Da Silveira Barros, R.L. Fernandes Bella, Towards the Sustainable Decommissioning of Fixed Platforms by Aligning Ecosystem Services and Wind Generation: A Brazilian Case, IJEEP 13 (2023) 235–242. [CrossRef]
- B.M. Storey, R.H. Worden, D.D. McNamara, J. Wheeler, J. Parker, A. Kristen, Reactivation of Abandoned Oilfields for Cleaner Energy Generation: Three-Dimensional Modelling of Reservoir Heterogeneity and Geometry, Processes 12 (2024) 2883. [CrossRef]
- A. Meenakshisundaram, O.S. Tomomewo, L. Aimen, S.O. Bade, A comprehensive analysis of repurposing abandoned oil wells for different energy uses: Exploration, applications, and repurposing challenges, Cleaner Engineering and Technology 22 (2024) 100797. [CrossRef]
- D.S. Oliver, K. Fossum, T. Bhakta, I. Sandø, G. Nævdal, R.J. Lorentzen, 4D seismic history matching, Journal of Petroleum Science and Engineering 207 (2021) 109119. [CrossRef]
- W.-L. Kang, B.-B. Zhou, M. Issakhov, M. Gabdullin, Advances in enhanced oil recovery technologies for low permeability reservoirs, Petroleum Science 19 (2022) 1622–1640. [CrossRef]
- J. Magill, Oil, Gas Companies Deploy AI In The Fight To Reduce Carbon Emissions, Forbes (n.d.). https://www.forbes.com/sites/jimmagill/2021/03/28/oil-gas-companies-deploy-ai-in-the-fight-to-reduce-carbon-emissions/ (accessed May 30, 2023).
- Gold Hydrogen | The Gold Standard for the Energy Transition, (n.d.). https://goldhydrogen.com/ (accessed June 28, 2025).
- T. Jacobs, Firm Says Microbes Strike Hydrogen Gold in California Oil Field, Journal of Petroleum Technology (2025). https://jpt.spe.org/firm-says-microbes-strike-hydrogen-gold-in-california-oil-field (accessed July 13, 2025).
- H. Wu, A. Li, H. Zhang, S. Li, C. Yang, H. Lv, Y. Yao, Microbial mechanisms for higher hydrogen production in anaerobic digestion at constant temperature versus gradient heating, Microbiome 12 (2024) 170. [CrossRef]
- K. Liang, Metabolic Pathways and Genetic Engineering of Anaerobic Bacteria for Biohydrogen Production, Be (2024). [CrossRef]
- M. Boll, ed., Anaerobic Utilization of Hydrocarbons, Oils, and Lipids, Springer International Publishing, Cham, 2020. [CrossRef]
- K. Laczi, Á. Erdeiné Kis, Á. Szilágyi, N. Bounedjoum, A. Bodor, G.E. Vincze, T. Kovács, G. Rákhely, K. Perei, New Frontiers of Anaerobic Hydrocarbon Biodegradation in the Multi-Omics Era, Front. Microbiol. 11 (2020) 590049. [CrossRef]
- H.R. Beller, A.M. Spormann, Analysis of the Novel Benzylsuccinate Synthase Reaction for Anaerobic Toluene Activation Based on Structural Studies of the Product, J Bacteriol 180 (1998) 5454–5457. [CrossRef]
- V. Grossi, C. Cravo-Laureau, A. Méou, D. Raphel, F. Garzino, A. Hirschler-Réa, Anaerobic 1-Alkene Metabolism by the Alkane- and Alkene-Degrading Sulfate Reducer Desulfatibacillum aliphaticivorans Strain CV2803T, Appl Environ Microbiol 73 (2007) 7882–7890. [CrossRef]
- H. Wilkes, W. Buckel, B.T. Golding, R. Rabus, Metabolism of Hydrocarbons in n-Alkane-Utilizing Anaerobic Bacteria, Microb Physiol 26 (2016) 138–151. [CrossRef]
- L. Cabrol, A. Marone, E. Tapia-Venegas, J.-P. Steyer, G. Ruiz-Filippi, E. Trably, Microbial ecology of fermentative hydrogen producing bioprocesses: useful insights for driving the ecosystem function, FEMS Microbiology Reviews 41 (2017) 158–181. [CrossRef]
- S. Gupta, A. Fernandes, A. Lopes, L. Grasa, J. Salafranca, Microbes and Parameters Influencing Dark Fermentation for Hydrogen Production, Applied Sciences 14 (2024) 10789. [CrossRef]
- F. Von Netzer, G. Pilloni, S. Kleindienst, M. Krüger, K. Knittel, F. Gründger, T. Lueders, Enhanced Gene Detection Assays for Fumarate-Adding Enzymes Allow Uncovering of Anaerobic Hydrocarbon Degraders in Terrestrial and Marine Systems, Appl Environ Microbiol 79 (2013) 543–552. [CrossRef]
- M.P. Díez, E. Villanueva-Galindo, I. Moreno-Andrade, E. Díaz, M.A. De La Rubia, A.F. Mohedano, M. Perez-Rangel, Enhanced hydrogen production from food waste via bioaugmentation with Clostridium and Lactobacillus, Biomass Conv. Bioref. (2025). [CrossRef]
- K.M. Muñoz-Páez, G. Buitrón, M. Vital-Jácome, Predicting metabolic pathways and microbial interactions in dark fermentation systems treating real cheese whey effluents, Bioresource Technology 413 (2024) 131536. [CrossRef]
- Y. Wang, X. Chen, K. Spengler, K. Terberger, M. Boehm, J. Appel, T. Barske, S. Timm, N. Battchikova, M. Hagemann, K. Gutekunst, Pyruvate:ferredoxin oxidoreductase and low abundant ferredoxins support aerobic photomixotrophic growth in cyanobacteria, eLife 11 (2022) e71339. [CrossRef]
- R. Biswas, T. Zheng, D.G. Olson, L.R. Lynd, A.M. Guss, Elimination of hydrogenase active site assembly blocks H2 production and increases ethanol yield in Clostridium thermocellum, Biotechnol Biofuels 8 (2015) 20. [CrossRef]
- L.C. De Souza, C.D. Herring, L.R. Lynd, Genetic investigation of hydrogenases in Thermoanaerobacterium thermosaccharolyticum suggests that redox balance via hydrogen cycling enables high ethanol yield, Appl Environ Microbiol 91 (2025) e01109-24. [CrossRef]
- S. Yadav, R. Haas, E.B. Boydas, M. Roemelt, T. Happe, U.-P. Apfel, S.T. Stripp, Oxygen sensitivity of [FeFe]-hydrogenase: a comparative study of active site mimics inside vs. outside the enzyme, Phys. Chem. Chem. Phys. 26 (2024) 19105–19116. [CrossRef]
- K. Schuchmann, N.P. Chowdhury, V. Müller, Complex Multimeric [FeFe] Hydrogenases: Biochemistry, Physiology and New Opportunities for the Hydrogen Economy, Front. Microbiol. 9 (2018) 2911. [CrossRef]
- H. Ogata, W. Lubitz, Y. Higuchi, Structure and function of [NiFe] hydrogenases, J Biochem 160 (2016) 251–258. [CrossRef]
- A.-R. Fleitas García, J.-S. Guez, C.G. Dussap, P. Fontanille, G. Christophe, Effect of redox potential on biohydrogen production during dark fermentation of food wastes in bioreactor, International Journal of Hydrogen Energy 141 (2025) 1199–1210. [CrossRef]
- X. Li, Y. Liu, X. Li, Z. Qin, Y. Su, S. Freguia, L. Feng, Y. Chen, Phenanthrene regulates metabolic pathways for hydrogen accumulation in sludge alkaline dark fermentation, Bioresource Technology 384 (2023) 129311. [CrossRef]
- X. Li, K. Sui, J. Zhang, X. Liu, Q. Xu, D. Wang, Q. Yang, Revealing the mechanisms of rhamnolipid enhanced hydrogen production from dark fermentation of waste activated sludge, Science of The Total Environment 806 (2022) 150347. [CrossRef]
- B. Zhao, S. Wang, Z. Dong, S. Cao, A. Yuan, H. Sha, N. Chen, Enhancing dark fermentative hydrogen production from wheat straw through synergistic effects of active electric fields and enzymatic hydrolysis pretreatment, Bioresource Technology 406 (2024) 130993. [CrossRef]
- S. Xue, D. Liang, J. Yan, Y. Xu, F. Wang, G. Lv, Geographic scale-based analysis of hydrogen production efficiency and mechanism in dark fermentation utilizing diverse inoculums, International Journal of Hydrogen Energy 89 (2024) 390–400. [CrossRef]
- Y. Fu, Y. Song, H. Chen, H. Chen, Y. Li, Q. Wu, Effects of biological carrier optimization on hydrogen production and biofilm formation in anaerobic circulating fluidized bed reactor via dark fermentation, International Journal of Hydrogen Energy 144 (2025) 85–95. [CrossRef]
- H. Sun, J. Shen, M. Hu, J. Zhang, Z. Cai, L. Zang, F. Zhang, D. Ji, Manganese ferrite nanoparticles enhanced biohydrogen production from mesophilic and thermophilic dark fermentation, Energy Reports 7 (2021) 6234–6245. [CrossRef]
- J.J. Rodríguez-Reyes, O. García-Depraect, S. Cantera, V. Mena-Navarro, E. León-Becerril, Assessment of the recovery of hydrogen production activity in dark fermentation reactors after a long period of shutdown, International Journal of Hydrogen Energy 144 (2025) 1134–1146. [CrossRef]
- G. Mohanakrishna, N.P. Sneha, S.M. Rafi, O. Sarkar, Dark fermentative hydrogen production: Potential of food waste as future energy needs, Science of The Total Environment 888 (2023) 163801. [CrossRef]
- A. Ouafa, D. Kerroum, P. Antonio, R. Lamis, B. Chaima, Z.-Z. Rania, B.-L. Mossaab, Coffee grounds and fruit and vegetable waste co-digestion in dark Fermentation: Evaluation of mixing ratio and hybrid pretreatments impact on bio-hydrogen production, Biomass and Bioenergy 199 (2025) 107896. [CrossRef]
- Y. Wei, Y. Jiao, H. Chen, Polydimethyldiallylammonium chloride inhibits dark fermentative hydrogen production from waste activated sludge, Bioresource Technology 393 (2024) 130003. [CrossRef]
- S. Feng, W. Guo, S. Zhang, G. Luo, H.T. Nguyen, N.C. Nguyen, D. Cheng, Y. Ye, H.H. Ngo, Optimization of hydraulic retention time in continuous orange peel crude enzyme - mediated dark fermentation for sustainable biohydrogen production from synthetic swine wastewater, Journal of Water Process Engineering 73 (2025) 107714. [CrossRef]
- P. Srivastava, E. García-Quismondo, J. Palma, C. González-Fernández, Coupling dark fermentation and microbial electrolysis cells for higher hydrogen yield: Technological competitiveness and challenges, International Journal of Hydrogen Energy 52 (2024) 223–239. [CrossRef]
- M. Hussien, H.O. Mohamed, D.A. Jadhav, A. Bahaa, S.-M. Jo, J.-H. Jang, J.-H. Kim, J.-Y. Kwon, E.T. Sayed, M.A. Abdelkareem, P. Castaño, K.-J. Chae, Synergistic integration of dark fermentation and microbial electrolysis cells for hydrogen production and sustainable swine manure treatment, International Journal of Hydrogen Energy 115 (2025) 299–309. [CrossRef]
- A. Jalil, Z. Yu, Impact of Substrates, Volatile Fatty Acids, and Microbial Communities on Biohydrogen Production: A Systematic Review and Meta-Analysis, Sustainability 16 (2024) 10755. [CrossRef]
- J. Zhang, B. Jiang, H. Zhang, S. Qian, T. Wei, Z. Zhang, L. Song, X. Yang, Fermentative Hydrogen Production from Lignocellulose by Mesophilic Clostridium populeti FZ10 Newly Isolated from Microcrystalline Cellulose-Acclimated Compost, Applied Sciences 12 (2022) 9562. [CrossRef]
- C.-H. Liu, C.-Y. Chang, C.-L. Cheng, D.-J. Lee, J.-S. Chang, Fermentative hydrogen production by Clostridium butyricum CGS5 using carbohydrate-rich microalgal biomass as feedstock, International Journal of Hydrogen Energy 37 (2012) 15458–15464. [CrossRef]
- M.J. Veshareh, M. Poulsen, H.M. Nick, K.L. Feilberg, A.A. Eftekhari, N. Dopffel, The light in the dark: In-situ biorefinement of crude oil to hydrogen using typical oil reservoir Thermotoga strains, International Journal of Hydrogen Energy 47 (2022) 5101–5110. [CrossRef]
- F.T. Jannat, K. Aftab, U. Kalsoom, M.A. Baig, A bibliometric analysis of the role of nanotechnology in dark fermentative biohydrogen production, Environ Sci Pollut Res 31 (2024) 24815–24835. [CrossRef]
- P. Kottenhahn, G. Philipps, S. Jennewein, Hexanol biosynthesis from syngas by Clostridium carboxidivorans P7 – product toxicity, temperature dependence and in situ extraction, Heliyon 7 (2021) e07732. [CrossRef]
- S. Maintinguer, B. Fernandes, I. Duarte, N. Saavedra, M. Adorno, M. Varesche, Fermentative hydrogen production by microbial consortium, International Journal of Hydrogen Energy 33 (2008) 4309–4317. [CrossRef]
- M. Černý, M. Vítězová, T. Vítěz, M. Bartoš, I. Kushkevych, Variation in the Distribution of Hydrogen Producers from the Clostridiales Order in Biogas Reactors Depending on Different Input Substrates, Energies 11 (2018) 3270. [CrossRef]
- H. Han, M. Cui, L. Wei, H. Yang, J. Shen, Enhancement effect of hematite nanoparticles on fermentative hydrogen production, Bioresource Technology 102 (2011) 7903–7909. [CrossRef]
- X.-J. Zheng, L.-F. Wei, Z.-H. Zhang, Q.-J. Jiang, Y.-J. Wei, B. Xie, M.-B. Wei, Research on photocatalytic H2 production from acetic acid solution by Pt/TiO2 nanoparticles under UV irradiation, International Journal of Hydrogen Energy 34 (2009) 9033–9041. [CrossRef]
- V. Beschkov, T. Parvanova-Mancheva, E. Vasileva, Experimental Study of Bio-Hydrogen Production by Clostridium beijerinckii from Different Substrates, Energies 16 (2023) 2747. [CrossRef]
- M. Martins, C. Mourato, I.A.C. Pereira, Desulfovibrio vulgaris Growth Coupled to Formate-Driven H2 Production, Environ. Sci. Technol. 49 (2015) 14655–14662. [CrossRef]
- N. Lackner, A. Hintersonnleitner, A.O. Wagner, P. Illmer, Hydrogenotrophic Methanogenesis and Autotrophic Growth of Methanosarcina thermophila, Archaea 2018 (2018) 1–7. [CrossRef]
- S. Saranya, L. Thamanna, V.P. Sreekutty, S. Dhayanithi, P. Chellapandi, Bioremediation of Oil and Natural Gas Industry Waste Using Methanogens: Current Status and Future Perspective to Biohythane Production, Arab J Sci Eng 50 (2025) 4457–4475. [CrossRef]
- M. Martins, I.A.C. Pereira, M. Pita, A.L. De Lacey, Biological Production of Hydrogen, in: J.J.G. Moura, I. Moura, L.B. Maia (Eds.), Enzymes for Solving Humankind’s Problems, Springer International Publishing, Cham, 2021: pp. 247–273. [CrossRef]
- G.M. Van Der Kraan, J. Bruining, B.P. Lomans, M.C.M. Van Loosdrecht, G. Muyzer, Microbial diversity of an oil-water processing site and its associated oil field: the possible role of microorganisms as information carriers from oil-associated environments, FEMS Microbiology Ecology 71 (2010) 428–443. [CrossRef]
- J. Xuan, L. He, W. Wen, Y. Feng, Hydrogenase and Nitrogenase: Key Catalysts in Biohydrogen Production, Molecules 28 (2023) 1392. [CrossRef]
- F.T. Mbow, A. Akbari, N. Dopffel, K. Schneider, S. Mukherjee, R.U. Meckenstock, Insights into the effects of anthropogenic activities on oil reservoir microbiome and metabolic potential, New Biotechnology 79 (2024) 30–38. [CrossRef]
- G.E. Thomas, J.L. Brant, P. Campo, D.R. Clark, F. Coulon, B.H. Gregson, T.J. McGenity, B.A. McKew, Effects of Dispersants and Biosurfactants on Crude-Oil Biodegradation and Bacterial Community Succession, Microorganisms 9 (2021) 1200. [CrossRef]
- C. Nikolova, T. Gutierrez, Biosurfactants and Their Applications in the Oil and Gas Industry: Current State of Knowledge and Future Perspectives, Front. Bioeng. Biotechnol. 9 (2021) 626639. [CrossRef]
- Y. Tian, S. Xue, Y. Ma, Comparative analysis of bacterial community and functional species in oil reservoirs with different in situ temperatures, Int Microbiol 23 (2020) 557–563. [CrossRef]
- M. Pannekens, L. Kroll, H. Müller, F.T. Mbow, R.U. Meckenstock, Oil reservoirs, an exceptional habitat for microorganisms, New Biotechnology 49 (2019) 1–9. [CrossRef]
- L. An, X. Liu, J. Wang, J. Xu, X. Chen, X. Liu, B. Hu, Y. Nie, X.-L. Wu, Global diversity and ecological functions of viruses inhabiting oil reservoirs, Nat Commun 15 (2024) 6789. [CrossRef]
- A. Sherry, N. Gray, C. Aitken, J. Dolfing, Microbial Oil Degradation Under Methanogenic Conditions, in: K.N. Timmis (Ed.), Handbook of Hydrocarbon and Lipid Microbiology, Springer Berlin Heidelberg, Berlin, Heidelberg, 2010: pp. 3905–3917. [CrossRef]
- J.J. Hamilton, M. Calixto Contreras, J.L. Reed, Thermodynamics and H2 Transfer in a Methanogenic, Syntrophic Community, PLoS Comput Biol 11 (2015) e1004364. [CrossRef]
- V.V. Kadnikov, N.V. Ravin, D.S. Sokolova, E.M. Semenova, S.K. Bidzhieva, A.V. Beletsky, A.P. Ershov, T.L. Babich, M.R. Khisametdinov, A.V. Mardanov, T.N. Nazina, Metagenomic and Culture-Based Analyses of Microbial Communities from Petroleum Reservoirs with High-Salinity Formation Water, and Their Biotechnological Potential, Biology 12 (2023) 1300. [CrossRef]
- I.C.C. Hsia, I.A. Shouki, J. Li, Feasibility of Rejuvenating Depleted Oil Fields with New Energy: Biohydrogen, in: Offshore Technology Conference Asia, OTC, Kuala Lumpur, Malaysia, 2024: p. D021S005R004. [CrossRef]
- H. Alkan, J.F. Bauer, O. Burachok, P. Kowollik, M. Olbricht, M. Amro, Hydrogen from Depleted/Depleting Hydrocarbon Reservoirs: A Reservoir Engineering Perspective, Applied Sciences 14 (2024) 6217. [CrossRef]
- M.M. Madirisha, B.D. Ikotun, Hydrogen storage in depleted geological hydrocarbon reservoirs: Enhancing wellbore integrity and sustainability with geopolymer cement – Review, Journal of Energy Storage 84 (2024) 110834. [CrossRef]
- T. Chaubey, B.S. Grover, P. Tessier, Staged Membrane Process for High Pressure Hydrogen Production, US2014170061A1, 2014.
- D.J. Edlund, W. a Pledger, R.T. Studebaker, Hydrogen purification membranes, components and fuel processing systems containing the same, US6723156B2, 2004.
- D.J. Edlund, K. Hoshi, R.T. Studebaker, R.J. Studebaker, Hydrogen purification devices, US10717040B2, 2020.
- D.J. Edlund, Hydrogen generation assemblies and hydrogen purification devices, US9656215B2, 2017.
- Y. Li, Q. Zhang, Z. Zhang, Y. Jing, H. Zhang, J. Yue, D. Jiang, C. Lu, Y. Zhang, C. Wang, Biological hydrogen-alkane co-production fermentation system and method with negative carbon emission, CN114058479A, 2022.
- S.R. Gillick, M. Babaei, In-Situ Hydrogen Production from Natural Gas Wells with Subsurface Carbon Retention, SPE Journal 29 (2024) 2119–2129. [CrossRef]
- J.J. Abraham, A. Carvero, C. Teodoriu, M. AlMujalhem, M. Amani, Evaluating Potential Near Wellbore Integrity Issues in Cement and Casing Layers During Subsurface Hydrogen Storage and Production, in: GOTECH, SPE, Dubai City, UAE, 2025: p. D031S044R001. [CrossRef]
- » H2FIT Tubes: Paving the Way for Hydrogen Production, Storage, and Transport, (n.d.). https://www.centravis.com/en/news/h2fit-tubes-paving-the-way-for-hydrogen-production-storage-and-transport/ (accessed June 28, 2025).
- A. Naquash, A. Riaz, F. Yehia, Y.D. Chaniago, H. Lim, M. Lee, Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment, Gases 3 (2023) 92–105. [CrossRef]
- M. Yang, R. Hunger, S. Berrettoni, B. Sprecher, B. Wang, A review of hydrogen storage and transport technologies, Clean Energy 7 (2023) 190–216. [CrossRef]
- O. Massarweh, A.S. Abushaikha, CO2 sequestration in subsurface geological formations: A review of trapping mechanisms and monitoring techniques, Earth-Science Reviews 253 (2024) 104793. [CrossRef]
- H.S. Santos, H. Nguyen, F. Venâncio, D. Ramteke, R. Zevenhoven, P. Kinnunen, Mechanisms of Mg carbonates precipitation and implications for CO2 capture and utilization/storage, Inorg. Chem. Front. 10 (2023) 2507–2546. [CrossRef]
- W.H. Tay, K.K. Lau, A.M. Shariff, High frequency ultrasonic-assisted chemical absorption of CO 2 using monoethanolamine (MEA), Separation and Purification Technology 183 (2017) 136–144. [CrossRef]
- M. Sai Bhargava Reddy, D. Ponnamma, K.K. Sadasivuni, B. Kumar, A.M. Abdullah, Carbon dioxide adsorption based on porous materials, RSC Adv. 11 (2021) 12658–12681. [CrossRef]
- Y. Fan, W. Yu, A. Wu, W. Shu, Y. Zhang, Recent progress on CO2 separation membranes, RSC Adv. 14 (2024) 20714–20734. [CrossRef]
- M.G. De Morais, E.G. De Morais, J.H. Duarte, K.M. Deamici, B.G. Mitchell, J.A.V. Costa, Biological CO2 mitigation by microalgae: technological trends, future prospects and challenges, World J Microbiol Biotechnol 35 (2019) 78. [CrossRef]
- Case Studies of CO2 Storage in Depleted Oil and Gas Fields, IEAGHG (n.d.). https://ieaghg.org/publications/case-studies-of-co2-storage-in-depleted-oil-and-gas-fields/ (accessed June 24, 2025).
- Z.M. Shakor, N. Parsafard, E. Al-Shafei, Adsorption and separation modeling of CO2, hydrogen, and biogas: a mathematical review, J Mater Sci 60 (2025) 6850–6876. [CrossRef]
- A. Kamolov, Z. Turakulov, S. Rejabov, G. Díaz-Sainz, L. Gómez-Coma, A. Norkobilov, M. Fallanza, A. Irabien, Decarbonization of Power and Industrial Sectors: The Role of Membrane Processes, Membranes 13 (2023) 130. [CrossRef]
- M.M. Joachimski, J. Müller, T.M. Gallagher, G. Mathes, D.L. Chu, F. Mouraviev, V. Silantiev, Y.D. Sun, J.N. Tong, Five million years of high atmospheric CO2 in the aftermath of the Permian-Triassic mass extinction, Geology 50 (2022) 650–654. [CrossRef]
- R. Kumar, W.J. Chung, M.A. Khan, M. Son, Y.-K. Park, S.S. Lee, B.-H. Jeon, Breakthrough innovations in carbon dioxide mineralization for a sustainable future, Rev Environ Sci Biotechnol 23 (2024) 739–799. [CrossRef]
- N. Wang, D. Wang, A. Krook-Riekkola, X. Ji, MEA-based CO2 capture: a study focuses on MEA concentrations and process parameters, Front. Energy Res. 11 (2023) 1230743. [CrossRef]
- R. Prasad, S.K. Gupta, N. Shabnam, C.Y.B. Oliveira, A.K. Nema, F.A. Ansari, F. Bux, Role of Microalgae in Global CO2 Sequestration: Physiological Mechanism, Recent Development, Challenges, and Future Prospective, Sustainability 13 (2021) 13061. [CrossRef]
- S.V. Kiseleva, N.I. Chernova, M.S. Vlaskin, A.V. Grigorenko, E.A. Chunzhuk, S.Ya. Malaniy, E.A. Bakumenko, T.V. Rositskaya, Carbon Dioxide Absorption by Microalgae: Analysis of Technologies and Energy Costs, Therm. Eng. 71 (2024) 1038–1048. [CrossRef]
- Costs of CO2 sequestration, Thunder Said Energy (n.d.). https://thundersaidenergy.com/downloads/co2-disposal-in-geologic-formations-the-economics/ (accessed June 24, 2025).
- M. Mazzotti, J.C. Abanades, R. Allam, K.S. Lackner, F. Meunier, E. Rubin, J.C. Sanchez, K. Yogo, R. Zevenhoven, Mineral carbonation and industrial uses of carbon dioxide, in: IPCC Special Report on Carbon Dioxide Capture and Storage, Cambridge University Press, 2005. https://research.aalto.fi/en/publications/mineral-carbonation-and-industrial-uses-of-carbon-dioxide (accessed June 24, 2025).
- A. Raksajati, M.T. Ho, D.E. Wiley, Reducing the Cost of CO2 Capture from Flue Gases Using Aqueous Chemical Absorption, Ind. Eng. Chem. Res. 52 (2013) 16887–16901. [CrossRef]
- Cost of carbon capture by approach or technology, Statista (n.d.). https://www.statista.com/statistics/1304575/global-carbon-capture-cost-by-technology/ (accessed June 24, 2025).
- Is carbon capture too expensive? – Analysis, IEA (2021). https://www.iea.org/commentaries/is-carbon-capture-too-expensive (accessed June 24, 2025).
- J. Meng, W. Liao, G. Zhang, Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China, Minerals 11 (2021) 274. [CrossRef]
- H. Zeng, X. Qu, D. Xu, Y. Luo, Porous Adsorption Materials for Carbon Dioxide Capture in Industrial Flue Gas, Front. Chem. 10 (2022) 939701. [CrossRef]
- E. Ruales, C. Gómez-Serrano, A. Morillas-España, C. González-López, M. Escolà Casas, V. Matamoros, M. Garfí, I. Ferrer, Resource recovery and contaminants of emerging concern mitigation by microalgae treating wastewater, Journal of Environmental Management 367 (2024) 121950. [CrossRef]
- Y.T. Yuen, P.N. Sharratt, B. Jie, Carbon dioxide mineralization process design and evaluation: concepts, case studies, and considerations, Environ Sci Pollut Res 23 (2016) 22309–22330. [CrossRef]
- M.H.J.-J. François, A. Grimstvedt, H.K. Knuutila, Iron Solubility Measurements in Aqueous MEA for CO2 Capture, Ind. Eng. Chem. Res. 64 (2025) 2318–2328. [CrossRef]
- F. Raganati, F. Miccio, P. Ammendola, Adsorption of Carbon Dioxide for Post-combustion Capture: A Review, Energy Fuels 35 (2021) 12845–12868. [CrossRef]
- C. Castel, R. Bounaceur, E. Favre, Membrane Processes for Direct Carbon Dioxide Capture From Air: Possibilities and Limitations, Front. Chem. Eng. 3 (2021) 668867. [CrossRef]
- IEA Greenhouse Gas R&D Programme, L. Stalker, K. Michael, C. Jenkins, K. Holland, L. Peeters, M. Myers, A. Ross, Reviewing the implications of unlikely but potential CO2 migration to the surface or shallow subsurface, IEAGHG, 2025. [CrossRef]
- M. Karimi, M. Shirzad, J.A.C. Silva, A.E. Rodrigues, Carbon dioxide separation and capture by adsorption: a review, Environ Chem Lett 21 (2023) 2041–2084. [CrossRef]
- S. Ray, C. Kuppam, S. Pandit, P. Kumar, Biogas Upgrading by Hydrogenotrophic Methanogens: An Overview, Waste Biomass Valor 14 (2023) 537–552. [CrossRef]
- S. Barbier, F. Huang, M. Andreani, R. Tao, J. Hao, A. Eleish, A. Prabhu, O. Minhas, K. Fontaine, P. Fox, I. Daniel, A Review of H2, CH4, and Hydrocarbon Formation in Experimental Serpentinization Using Network Analysis, Front. Earth Sci. 8 (2020) 209. [CrossRef]
- R. Kanaujiya, A.K. Metya, N. Choudhary, R. Kumar, T.K. Patra, Molecular dynamics insights into tetrahydrofuran-assisted formation of CH4, CO2, and H2 gas hydrates, Phys. Chem. Chem. Phys. (2025) 10.1039.D5CP01574J. [CrossRef]
- M. S, Carbonate and Sandstone Reservoirs in CO2 Sequestration: Assessing Porosity and Permeability for Enhanced Storage Potential, PPEJ 8 (2024) 1–12. [CrossRef]
- S. Ni, W. Lv, Z. Ji, K. Wang, CO2 Mineralized Sequestration and Assistance by Microorganisms in Reservoirs: Development and Outlook, Energies 16 (2023) 7571. [CrossRef]
- A. Askarova, A. Mukhametdinova, S. Markovic, G. Khayrullina, P. Afanasev, E. Popov, E. Mukhina, An Overview of Geological CO2 Sequestration in Oil and Gas Reservoirs, Energies 16 (2023) 2821. [CrossRef]
- Y. Chen, S. Chen, D. Li, X. Jiang, Density-Driven Convection for CO2 Solubility Trapping in Saline Aquifers: Modeling and Influencing Factors, Geotechnics 3 (2023) 70–103. [CrossRef]
- J.W. Johnson, J.J. Nitao, K.G. Knauss, Reactive transport modelling of CO2 storage in saline aquifers to elucidate fundamental processes, trapping mechanisms and sequestration partitioning, SP 233 (2004) 107–128. [CrossRef]
- Q. Chen, M. Ramdin, T.J.H. Vlugt, Solubilities of CO2, CH4, C2 H6, CO, H2, N2, N2 O, and H2 S in commercial physical solvents from Monte Carlo simulations, Molecular Simulation 49 (2023) 1341–1349. [CrossRef]
- D.V. Bhalani, B. Lim, Hydrogen Separation Membranes: A Material Perspective, Molecules 29 (2024) 4676. [CrossRef]
- J. Vilcáez, E. Chowdhury, Biogenic hydrogen production from oil hydrocarbons at geological carbon storage conditions, Energy Conversion and Management 325 (2025) 119438. [CrossRef]
- G.-C. Yang, L. Zhou, S.M. Mbadinga, J.-F. Liu, S.-Z. Yang, J.-D. Gu, B.-Z. Mu, Formate-Dependent Microbial Conversion of CO2 and the Dominant Pathways of Methanogenesis in Production Water of High-temperature Oil Reservoirs Amended with Bicarbonate, Front. Microbiol. 7 (2016). [CrossRef]
- R.L. Tyne, P.H. Barry, M. Lawson, D.J. Byrne, O. Warr, H. Xie, D.J. Hillegonds, M. Formolo, Z.M. Summers, B. Skinner, J.M. Eiler, C.J. Ballentine, Rapid microbial methanogenesis during CO2 storage in hydrocarbon reservoirs, Nature 600 (2021) 670–674. [CrossRef]
- Y.K. Leong, Y.-J. Huang, J.-S. Chang, Boosting CO2 capture and conversion to biohydrogen through enhanced microalgal biomass yield, International Journal of Hydrogen Energy 139 (2025) 998–1007. [CrossRef]
- M. Ashour, A.T. Mansour, Y.A. Alkhamis, M. Elshobary, Usage of Chlorella and diverse microalgae for CO2 capture - towards a bioenergy revolution, Front. Bioeng. Biotechnol. 12 (2024) 1387519. [CrossRef]
- A. Maghzian, A. Aslani, R. Zahedi, A comprehensive review on effective parameters on microalgae productivity and carbon capture rate, Journal of Environmental Management 355 (2024) 120539. [CrossRef]
- Y. Kikuchi, D. Kanai, K. Sugiyama, K. Fujii, Biogas Upgrading by Wild Alkaliphilic Microalgae and the Application Potential of Their Biomass in the Carbon Capture and Utilization Technology, Fermentation 10 (2024) 134. [CrossRef]
- G.E. King, D.E. King, Environmental Risk Arising From Well-Construction Failure—Differences Between Barrier and Well Failure, and Estimates of Failure Frequency Across Common Well Types, Locations, and Well Age, SPE Production & Operations 28 (2013) 323–344. [CrossRef]
- R.B. Jackson, The integrity of oil and gas wells, Proc. Natl. Acad. Sci. U.S.A. 111 (2014) 10902–10903. [CrossRef]
- J.D. Laumb, K.A. Glazewski, J.A. Hamling, A. Azenkeng, T.L. Watson, Wellbore corrosion and failure assessment for CO2 EOR and storage: Two case studies in the Weyburn field, International Journal of Greenhouse Gas Control 54 (2016) 479–489. [CrossRef]
- Pablo.G. Cirimello, J.L. Otegui, G. Carfi, W. Morris, Failure and integrity analysis of casings used for oil well drilling, Engineering Failure Analysis 75 (2017) 1–14. [CrossRef]
- N. Yousuf, O. Olayiwola, B. Guo, N. Liu, A comprehensive review on the loss of wellbore integrity due to cement failure and available remedial methods, Journal of Petroleum Science and Engineering 207 (2021) 109123. [CrossRef]
- M. Bai, Z. Zhang, X. Fu, A review on well integrity issues for CO 2 geological storage and enhanced gas recovery, Renewable and Sustainable Energy Reviews 59 (2016) 920–926. [CrossRef]
- D.A. Al-Shehri, Oil and Gas Wells: Enhanced Wellbore Casing Integrity Management through Corrosion Rate Prediction Using an Augmented Intelligent Approach, Sustainability 11 (2019) 818. [CrossRef]
- L. Wu, Z.-M. Hou, Z.-F. Luo, Y.-L. Fang, L.-C. Huang, X.-N. Wu, Q.-J. Chen, Q.-C. Wang, Impacts of microbial interactions on underground hydrogen storage in porous media: A comprehensive review of experimental, numerical, and field studies, Petroleum Science 21 (2024) 4067–4099. [CrossRef]
- K. Opoku Duartey, W. Ampomah, H. Rahnema, M. Mehana, Underground Hydrogen Storage: Transforming Subsurface Science into Sustainable Energy Solutions, Energies 18 (2025) 748. [CrossRef]
- A. Zappone, A.P. Rinaldi, M. Grab, Q.C. Wenning, C. Roques, C. Madonna, A.C. Obermann, S.M. Bernasconi, M.S. Brennwald, R. Kipfer, F. Soom, P. Cook, Y. Guglielmi, C. Nussbaum, D. Giardini, M. Mazzotti, S. Wiemer, Fault sealing and caprock integrity for CO2 storage: an in situ injection experiment, Solid Earth 12 (2021) 319–343. [CrossRef]
- IEA Greenhouse Gas R&D Programme, D. Bason, H. Nourollah, S. De Morton, M. Watson, G. Petho, I. Maffeis, M. Rieger, G. O’Brien, Geological Storage of CO2: Seal Integrity Review, IEAGHG, 2024. [CrossRef]
- N. Dopffel, B.A. An-Stepec, J.R. De Rezende, D.Z. Sousa, A. Koerdt, Editorial: Microbiology of underground hydrogen storage, Front. Energy Res. 11 (2023) 1242619. [CrossRef]
- Y. Sugai, Y. Owaki, K. Sasaki, Simulation Study on Reservoir Souring Induced by Injection of Reservoir Brine Containing Sulfate-Reducing Bacteria, Sustainability 12 (2020) 4603. [CrossRef]
- J. Hu, C. Li, Q. Zhang, Q. Guo, S. Zhao, W. Wang, D.-J. Lee, Y. Yang, Using chemical looping gasification with Fe2O3/Al2O3 oxygen carrier to produce syngas (H2+CO) from rice straw, International Journal of Hydrogen Energy 44 (2019) 3382–3386. [CrossRef]
- H.S. Hagar, J. Foroozesh, S. Kumar, D. Zivar, N. Banan, I. Dzulkarnain, Microbial H2S generation in hydrocarbon reservoirs: Analysis of mechanisms and recent remediation technologies, Journal of Natural Gas Science and Engineering 106 (2022) 104729. [CrossRef]
- M.J.L. McMahon, A.J. Daugulis, Gas phase H2S product recovery in a packed bed bioreactor with immobilized sulfate-reducing bacteria, Biotechnol Lett 30 (2008) 467–473. [CrossRef]
- Q. He, F. Kong, R. Sun, R. Li, J. Yang, Q. Min, Experimental study on dynamic response performance of hydrogen sensor in confined space under ceiling, Front. Energy Res. 12 (2024) 1456316. [CrossRef]
- H. Se, J. Jiang, C. Sun, K. Song, Hydrogen Leakage Detection in Confined Spaces with Drift Suppression Based on Subspace Alignment, in: H. Sun, W. Pei, Y. Dong, H. Yu, S. You (Eds.), Proceedings of the 10th Hydrogen Technology Convention, Volume 1, Springer Nature Singapore, Singapore, 2024: pp. 180–186. [CrossRef]
- J.J. Sheng, Techno-Economic Analysis of Hydrogen Generation in Hydrocarbon Reservoirs, SPE Journal 29 (2024) 5752–5760. [CrossRef]
- S. Sundeep, L. Sethuraman, D. Akindipe, L. Fingersh, Z. Wenrick, A. Munoz, Repurposing inactive oil and gas wells for energy storage: maximizing the potential via optimal drivetrain control, IET Conf. Proc. 2023 (2023) 486–493. [CrossRef]
- TresCantos, CAPEX y OPEX en proyectos de hidrógeno, ARIEMA (2023). https://www.ariema.com/capex-opex-proyectos-hidrogeno-ariema (accessed June 28, 2025).
- A. Ganguly, P. Sun, X. Liu, H.E. Delgado, L. Sun, A. Elgowainy, Techno-economic and life cycle analysis of bio-hydrogen production using bio-based waste streams through the integration of dark fermentation and microbial electrolysis, Green Chem. 27 (2025) 6213–6231. [CrossRef]
- G.M. Teke, B. Anye Cho, C.E. Bosman, Z. Mapholi, D. Zhang, R.W.M. Pott, Towards industrial biological hydrogen production: a review, World J Microbiol Biotechnol 40 (2024) 37. [CrossRef]
- N.Z.M. of F.A. and Trade, Japan: Strategic Hydrogen Roadmap - 30 October 2020, New Zealand Ministry of Foreign Affairs and Trade (2020). https://www.mfat.govt.nz/en/trade/mfat-market-reports/japan-strategic-hydrogen-roadmap-30-october-2020 (accessed June 28, 2025).
- Financial Incentives for Hydrogen and Fuel Cell Projects, Energy.Gov (n.d.). https://www.energy.gov/eere/fuelcells/financial-incentives-hydrogen-and-fuel-cell-projects (accessed June 28, 2025).
- R. Fujii-Rajani, S. Patnaik, What will happen to the Inflation Reduction Act under a Republican trifecta?, Brookings (2025). https://www.brookings.edu/articles/what-will-happen-to-the-inflation-reduction-act-under-a-republican-trifecta/ (accessed July 13, 2025).
- Cemvita Launches the Gold Hydrogen Program for Subsurface Biomanufacturing of Hydrogen | Gold Hydrogen, (n.d.). https://goldhydrogen.com/cemvita-launches-the-gold-hydrogen-program-for-subsurface-biomanufacturing-of-hydrogen/ (accessed June 28, 2025).
- V. Adomavicius, G. Simkoniene, Overview of innovations and recommendations for efficient operation of RES-based power plants, in: 2023. [CrossRef]
- K. Adeli, M. Nachtane, A. Faik, D. Saifaoui, A. Boulezhar, How Green Hydrogen and Ammonia Are Revolutionizing the Future of Energy Production: A Comprehensive Review of the Latest Developments and Future Prospects, Applied Sciences 13 (2023) 8711. [CrossRef]
- Cemvita’s Successful Field Test Demonstrates Gold HydrogenTM Production in Situ | Gold Hydrogen, (n.d.). https://goldhydrogen.com/cemvitas-successful-field-test-demonstrates-gold-hydrogen-production-in-situ/ (accessed June 28, 2025).
- J.D. Grinstein, Fighting Climate Change with Synthetic Biology: Cemvita Factory generates cost-effective, low-carbon solutions that net climate-positive results, Genetic Engineering & Biotechnology News 42 (2022) 12–13. [CrossRef]
- S. Ni, W. Lv, Z. Ji, K. Wang, Y. Mei, Y. Li, Progress of Crude Oil Gasification Technology Assisted by Microorganisms in Reservoirs, Microorganisms 12 (2024) 702. [CrossRef]
- I.M. Head, N.D. Gray, S.R. Larter, Life in the slow lane; biogeochemistry of biodegraded petroleum containing reservoirs and implications for energy recovery and carbon management, Front. Microbiol. 5 (2014). [CrossRef]
- S.F. Ahmed, N. Rafa, M. Mofijur, I.A. Badruddin, A. Inayat, M.S. Ali, O. Farrok, T.M. Yunus Khan, Biohydrogen Production From Biomass Sources: Metabolic Pathways and Economic Analysis, Front. Energy Res. 9 (2021) 753878. [CrossRef]
- P.D.S.J. of P. Technology, Biotech Firm To Create Gold Hydrogen by Injecting Microbes Into Depleted Oil Reservoirs, JPT (2022). https://jpt.spe.org/biotech-firm-to-create-gold-hydrogen-by-injecting-microbes-into-depleted-oil-reservoirs (accessed June 28, 2025).
- H2-View News: Start-up plans to generate low-cost hydrogen from old oil wells with microbes | H2 Energy Group, (2024). https://h2eg.com/h2-view-news-start-up-plans-to-generate-low-cost-hydrogen-from-old-oil-wells-with-microbes/ (accessed June 28, 2025).
- Microbes that eat oil and excrete cheap, clean hydrogen | GlobalSpec, (n.d.). https://insights.globalspec.com/article/19402/microbes-that-eat-oil-and-excrete-cheap-clean-hydrogen (accessed June 28, 2025).
- P.K. Kevin, L.B.D.S. Marcio, K. Mojtaba, A.G. Renata, A.H. Roger, C.T. Aaron, R.B. Zachary, K. Tahereh, L.A.L. Luiza, D.F.M. Barbara, W.R. Christian, Method. GB2613039A, 2023.
- Z.R. Broussard, K.P. Kincaid, M. Karimi, T. Karimi, S.M.L.B. Da, R.A. Gonçalv, R.A. Harris, A.C. Trevino, L.L.A. Lahme, B.D.F. Magalhães, C.W. Rimbau, Method for Microbiological Production of Hydrogen, US2024392324A1, 2024.
- R.B. Zachary, T. Tj, S. Michael, J.K. Edward, S.R. Raja, A.G. Renata, C.T. Aaron, W. Addien, D.F.M. Barbara, T. Edris, Process and plant, GB2629486A, 2024.
- Z.R. Broussard, T. Tidwell, M. Samuel, E.J. Koch, R.S. Ramanathan, R.A. Gonçalves, A. Trevino, A. Wray, B.D.F. Magalhães, Process and Plant, WO2024206528A2, 2024.
- T. Karimi, M. Karimi, Methods and Systems for Producing Organic Compounds in a Subterranean Environment, US2022282604A1, 2022.
- L. Shen, Q. Zhao, X. Wu, X. Li, Q. Li, Y. Wang, Interspecies electron transfer in syntrophic methanogenic consortia: From cultures to bioreactors, Renewable and Sustainable Energy Reviews 54 (2016) 1358–1367. [CrossRef]
- B. Hu, S. Chen, Pretreatment of methanogenic granules for immobilized hydrogen fermentation, International Journal of Hydrogen Energy 32 (2007) 3266–3273. [CrossRef]
- J.I. Ghignone, G.D. Andrade, General Geology and Major Oil Fields of Recôncavo Basin, Brazil1, in: Geology of Giant Petroleum Fields, American Association of Petroleum Geologists, 1970. [CrossRef]
- Universidade Federal da Bahia: Isolamento e identificação molecular de bactérias redutoras de sulfato de amostras de água produzida em campo de petróleo, (n.d.). https://repositorio.ufba.br/handle/ri/18451 (accessed March 18, 2025).
- S. Myhr, B.L.P. Lillebø, E. Sunde, J. Beeder, T. Torsvik, Inhibition of microbial H2S production in an oil reservoir model column by nitrate injection., Applied Microbiology and Biotechnology 58 (2002) 400–8. [CrossRef]
- S. Deng, B. Wang, S. Su, S. Sun, Y. She, F. Zhang, Dynamics of Microbial Community and Removal of Hydrogen Sulfide (H2 S) Using a Bio-Inhibitor and Its Application under the Oil Reservoir Condition, Energy Fuels 36 (2022) 14128–14135. [CrossRef]
- S. Sarma, D. Ortega, N.P. Minton, V.K. Dubey, V.S. Moholkar, Homologous overexpression of hydrogenase and glycerol dehydrogenase in Clostridium pasteurianum to enhance hydrogen production from crude glycerol, Bioresource Technology 284 (2019) 168–177. [CrossRef]
- G. Meshulam-Simon, S. Behrens, A.D. Choo, A.M. Spormann, Hydrogen Metabolism in Shewanella oneidensis MR-1, Appl Environ Microbiol 73 (2007) 1153–1165. [CrossRef]
- Programa Nacional de Hidrogênio - PNH2 — Ministério de Minas e Energia, (n.d.). https://www.gov.br/mme/pt-br/programa-nacional-do-hidrogenio-1 (accessed July 1, 2025).
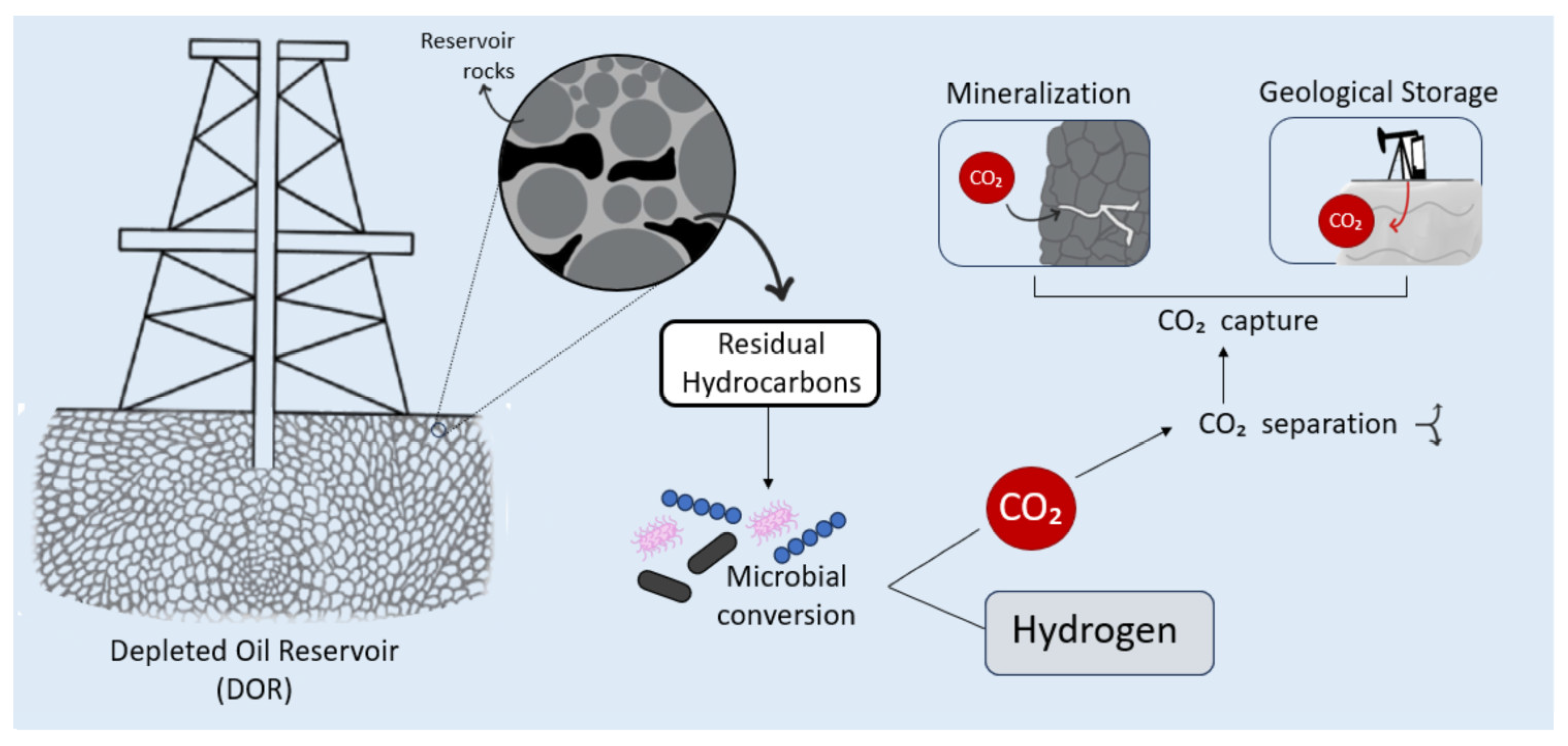
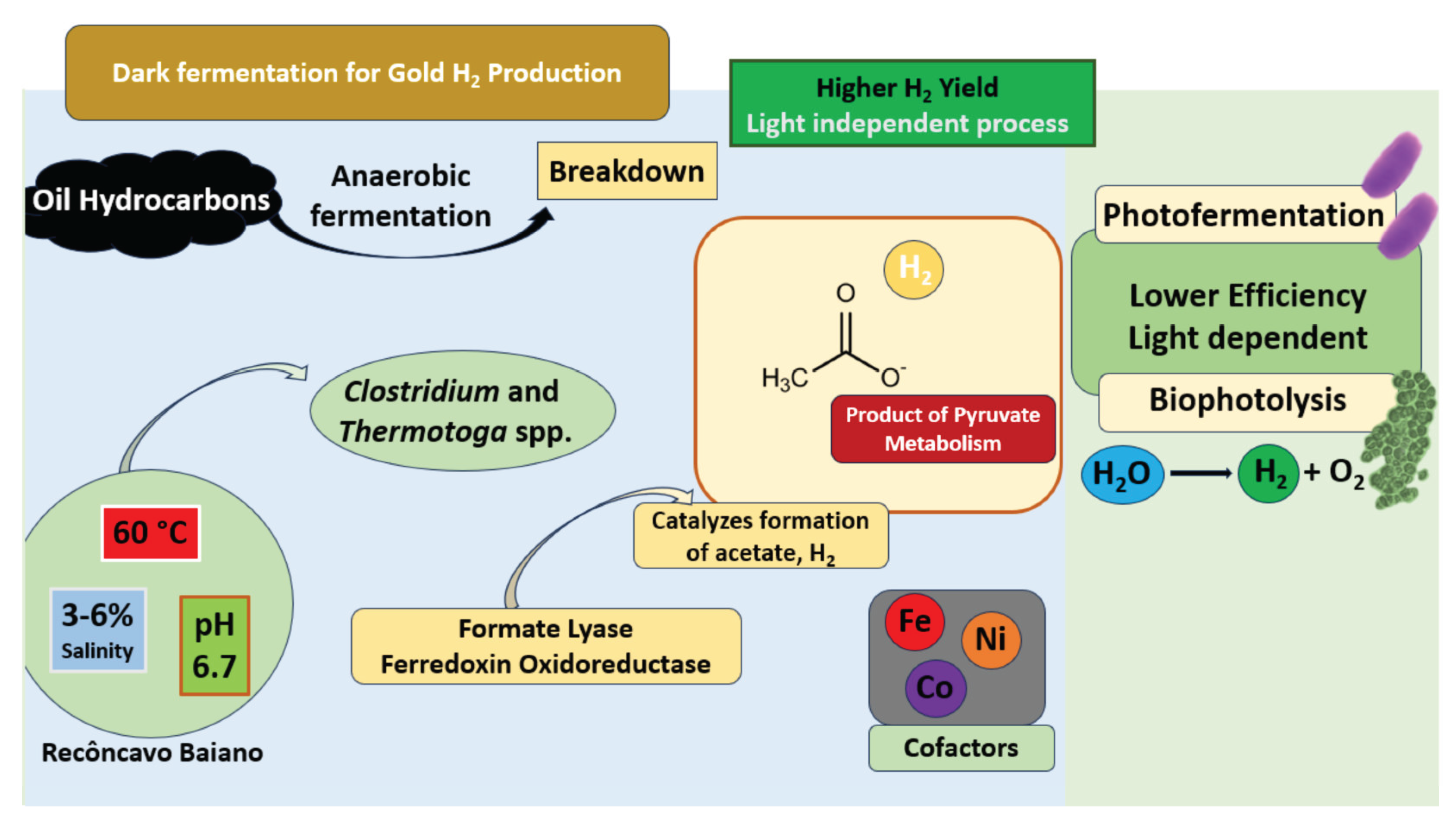
| Infrastructure Component | Existing in Oil & Gas Fields | Required Modifications for H2 Production | New Installations for H2 Bioproduction |
| Wells and Boreholes | Vertical and directional production/injection wells | Casing reinforcement; corrosion-resistant linings; zonal isolation with packers/plugs [85] | New wells if reservoir geometry or access is insufficient |
| Well Cement and Casings | Standard steel casing and Portland cement | Replacement or coating with corrosion-resistant alloys; acid-tolerant cement materials [86] | — |
| Subsurface Infrastructure | Reservoir access through perforated casings | Isolation of fermentation zones; installation of retrievable plugs [85] | Bioreactor adaptation: enhanced sealing for anaerobic containment |
| Injection Systems | Water/gas/polymer injection systems | Modified for microbial inoculum and nutrient solution delivery | Modular microbial and nutrient injection skids |
| Production Tubing | Hydrocarbon production tubing | Material upgrade to resist H2S, CO2, and acids [87] | - |
| Surface Separation Units | Oil/gas/water separators | Adapted to separate and handle H2-rich biogas [88] | H2-specific gas-liquid separators |
| Gas Processing Units | Natural gas dehydration, compression, and sweetening | Integration of H2-compatible compressors and piping | H2 purification systems (pressure swing adsorption, membrane units) |
| Monitoring Equipment | Pressure, temperature, flow rate sensors | Additional sensors for pH, redox, microbial activity, gas composition (H2, CO2, H2S) | Real-time bioprocess monitoring modules |
| Control Systems | Present for process automation and remote monitoring | Integration with new sensors and fermentation specific controls | AI-enabled microbial fermentation control algorithms |
| Storage Tanks | Crude oil, water, and gas storage facilities | H2 compatible materials for gas storage (e.g., high-alloy steel) [89] | H2-specific pressurized storage tanks or absorption beds |
| Pipelines | Steel pipelines for oil/gas transport | Retrofitting or replacement with H2 compatible materials | New dedicated H2 pipelines (short range/local) |
| Waste Management | Produced water treatment, gas flaring | Treatment of microbial byproducts and acidic effluents | Biosludge and fermentation waste handling units |
| Laboratory Facilities | On-site labs for chemical analysis | Capability expansion for microbial, gas, and fermentation monitoring | Mobile genetic and microbial culture labs |
| Parameter | Geological (in situ) | Mineralization (in situ) | Chemical Absorption | Adsorption | Membrane Separation | Microalgae-Based Sequestration |
| Mechanism | CO2 is trapped in porous rocks via structural, capillary, solubility, or residual trapping [90] | CO2 reacts with minerals (Ca, Mg silicates) to form stable carbonates [91] | CO2 chemically reacts with monoethanolamine [92] | CO2 adheres to solid porous materials under pressure [93] | Selective diffusion of CO2 through polymer/inorganic membranes [94] | CO2 fixed by photosynthesis into algal biomass [95] |
| Maturity/TRL | High (especially for DORs) [96] |
Moderate to high | High | Moderate to high [97] | Moderate [98] | Moderate (higher for closed photobioreactors) |
| CO2 Permanence | Very High (millions of years) [99] | Very High (solid carbonates are stable) [100] | Low to Moderate [101] | Low to Moderate [97] | Low to Moderate | Moderate [102] |
| Energy Requirement | Low | Moderate | High | Moderate | Low to Moderate | Moderate [103] |
| Cost (USD/ton CO2) | ~$10–20 [104] |
~$50–100 [105] | ~$62–80 [106] | ~$15–130 [107] | ~$30–80 [107] | ~$30–200 [102] |
| Scalability | High [108] | Moderate | High | Moderate | High | Moderate |
| Co-benefits | Enhanced oil/H2 recovery | Heavy metal stabilization [109] | None | May support pressure swing adsorption for H2 purification [110] | Energy-efficient for H2 purification | Biomass for fuels, wastewater treatment |
| Key Limitations | Risk of leakage, site-specific [111] | Slow natural kinetics [112] | Solvent degradation [113] | Sorbent regeneration [114] | Fouling/selectivity [115] | Growth depends on conditions |
| Environmental Impact | Low (if monitored) [116] | Low to moderate [116] | Chemical waste | Sorbent disposal [117] | Low | Low; improves air/water quality [111] |
| Application in H2 | Explored in microbial H2 | Emerging for microbial mineralization | Used in gas purification | Used in downstream cleanup | Growing interest | Promising for integrated systems |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




