1. Introduction
Chronic wounds, typically defined as wounds that fail to heal within six weeks, remain a major clinical and socioeconomic burden and markedly diminish patient quality of life [
1,
2]. Epidemiological studies estimate that chronic wounds affect approximately 1–2% of the population in developed countries, with elderly individuals and patients with diabetes or chronic venous insufficiency being particularly at risk [
3,
4]. The management of these wounds generates high healthcare costs and underscores the need for effective, personalized treatment strategies [
3,
4]. Among advanced treatment modalities, negative-pressure wound therapy (NPWT) and topical oxygen therapy (TOT
) are two of the most frequently used yet mechanistically distinct approaches.
NPWT applies sub-atmospheric pressure across a sealed foam interface, promoting granulation tissue formation, controlling exudate and reducing oedema [
5]. However, perfusion measurements indicate that the immediate increase in blood flow may be limited by the mechanical action of negative-pressure on blood vessels, with improved vascularization occurring gradually through angiogenesis [
6]. Conversely TOT delivers high-concentration oxygen directly to the wound bed, supporting fibroblast proliferation, collagen deposition and bacterial killing [
7]. TOT techniques vary, including continuous or cyclic oxygen delivery, often in a humidified environment, which optimizes metabolic and enzymatic processes essential for healing [
8]. Because oxygen tension is a limiting factor even under NPWT dressings, there is growing interest in combining both approaches to exploit their complementary mechanisms.
Evidence for this synergy is accumulating. Recent studies, including randomized controlled trials and meta-analyses, suggest that combining NPWT with adjunctive oxygen therapy significantly improves healing outcomes compared to monotherapy, particularly in complex or refractory wounds. A single-center randomized controlled trial on stage IV sacrococcygeal pressure ulcers reported that adding local oxygen to vacuum sealing drainage reduced median healing time by 30 days compared with NPWT alone [
9]. A 2024 systematic review and meta-analysis pooled ten studies and showed that adjunctive oxygen—whether topical or hyperbaric—combined with NPWT improved the odds of complete closure by 1.8-fold versus NPWT monotherapy [
10].
Equally important is the ability to stratify patients who are most likely to benefit. Readily available inflammatory ratios—neutrophil-to-lymphocyte (NLR), monocyte-to-lymphocyte (MLR) and platelet-to-lymphocyte (PLR)—have demonstrated prognostic value in diabetic and venous ulcer [
11,
12]. Serum total protein serves as a low-cost surrogate of nutritional reserve, deficiencies of which can delay collagen synthesis and epithelialisation [
13,
14]. Infra-red thermography offers a non-contact method of monitoring local perfusion, yet its predictive utility in chronic-wound trajectories is not firmly established [
15].
Against this background, the present pilot study prospectively compares combined TOT + NPWT with TOT alone in a clinically homogeneous cohort and explores whether baseline laboratory markers or early thermographic patterns can refine prognostic assessment. The overarching aim is to generate feasibility data that will inform personalized treatment algorithms for hard-to-heal wounds.
2. Materials and Methods
This prospective pilot study was conducted from January 1, 2024, to December 31, 2024. The study adopted a non-randomized, non-blinded exploratory design to assess the feasibility and preliminary effectiveness of combining topical oxygen therapy (TOT) with negative-pressure wound therapy (NPWT) in adults with chronic lower limb ulcers. The primary aim was to generate pilot data to inform future, more rigorous randomized controlled trials.
Adults (≥ 18 years) with a lower-limb ulcer of ≥ 6 weeks’ duration related to type 2 diabetes mellitus or chronic venous insufficiency were eligible. Additional inclusion criteria were wound surface area < 200 cm². Exclusion criteria comprised lack of consent, critical limb ischaemia, severe anaemia (haemoglobin < 8 g/dl), advanced renal failure (eGFR < 30 mL/min/1.73 m²), active malignancy, body-mass index < 18.5, immunosuppressive therapy or necrotising infection.
Participants were assigned 1:1 to topical oxygen therapy alone (TOT group) or TOT followed by negative-pressure wound therapy (TOT + NPWT group) using a computer-generated random list created with Excel
RAND; allocation was not concealed. TOT was delivered on study days 1–4 for 1.5 h/day at 10 l/min oxygen flow in accordance with 2022 Delphi-consensus guidelines [
16]. In the intervention arm, continuous NPWT at −125mmHg was applied from day 5 to day 7; the pressure setting and 48-hour dressing-change interval followed current practical recommendations for chronic wounds [
17]. Both groups received identical supportive care, including sharp debridement at every dressing change, irrigation with 0.05% sodium hypochlorite and multilayer compression for venous ulcers.
Wound morphology (surface area, granulation, necrotic tissue) was quantified using a validated three-dimensional imaging system (WoundMatrix™), while wound-bed and peri-wound temperatures were assessed by FLIR-One infrared thermography under standardized ambient conditions. Baseline laboratory tests included complete blood count, C-reactive protein and serum total protein; neutrophil-, monocyte- and platelet-to-lymphocyte ratios (NLR, MLR, PLR) were calculated accordingly.
Follow-up visits were conducted weekly for nine months. The primary endpoints were total treatment duration, defined as weeks from enrolment to complete epithelialization confirmed clinically, and the proportion of wounds achieving complete closure within the follow-up period. Secondary endpoints included early changes in wound dimensions and temperature as well as correlations between laboratory markers and healing metrics.
Descriptive statistics summarized the sample. The Wilcoxon rank-sum and Fisher’s exact test compared groups, while Spearman’s coefficient and simple linear regression assessed associations; no multivariable modeling or correction for multiple comparisons was performed given the exploratory, hypothesis-generating purpose.
The study received approval by the institutional Bioethics Committee (no. 1/2024). Written informed consent was obtained from all participants, in accordance with the Declaration of Helsinki and local regulatory requirements.
3. Results
A total of 18 patients were included in the analysis. Eight patients were men and ten were women. The intervention group included nine patients who received topical oxygen therapy combined with negative-pressure wound therapy (NPWT), and the control group included nine patients treated with topical oxygen therapy alone.
In 16 cases, the chronic wound was associated with venous insufficiency, while in two cases the etiology was diabetic. Baseline characteristics were comparable between groups (
Table 1).
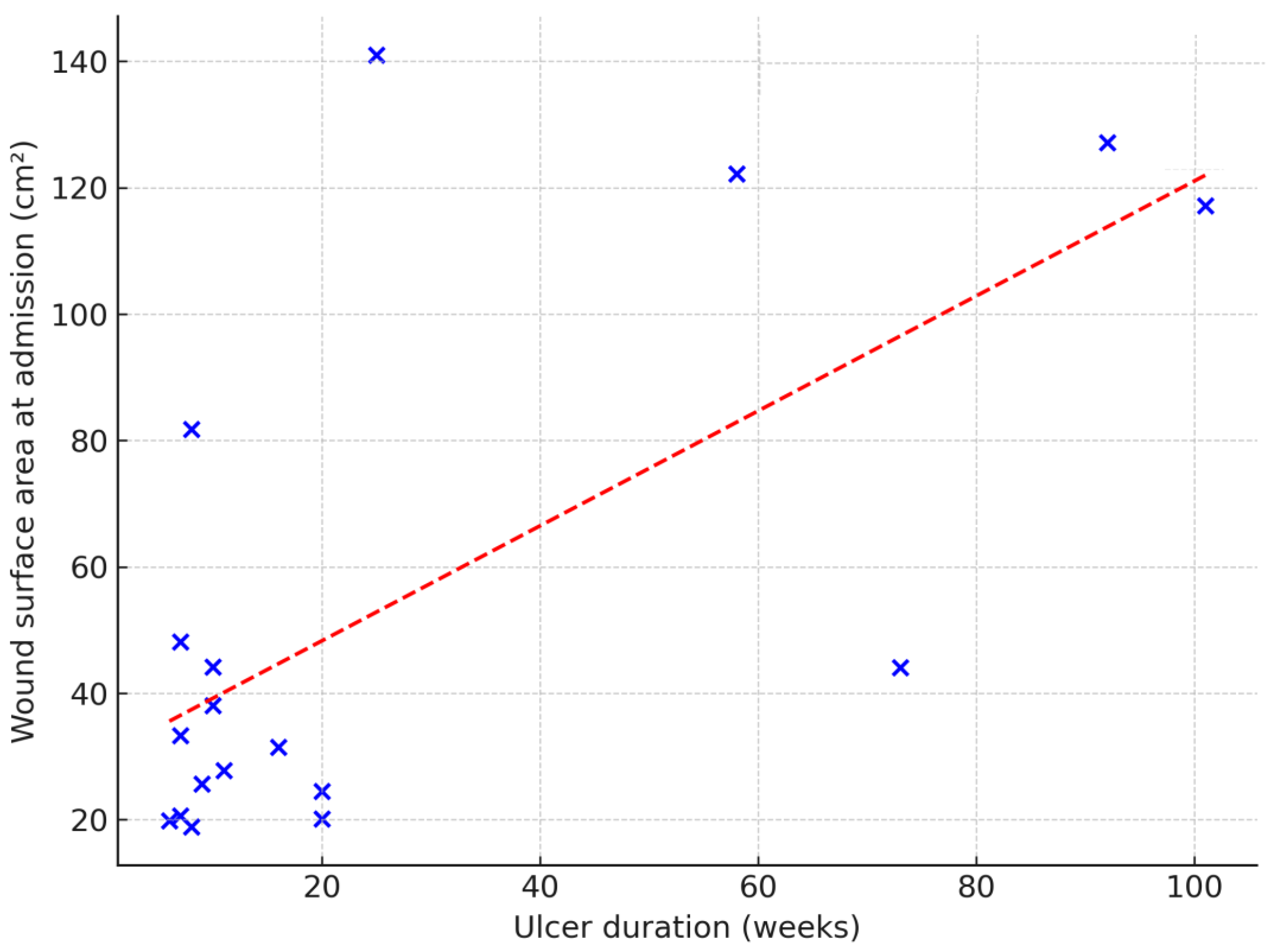
The influence of ulcer duration on treatment duration and outcome was analyzed, but no statistically significant association was found (odds ratio = -0.06,
p = 0.15). How-ever, ulcer duration was significantly associated with wound surface area at admission, with larger wound areas observed as ulcer duration increased (β = 0.91 cm²/week;
p = 0.003) (
Figure 1).
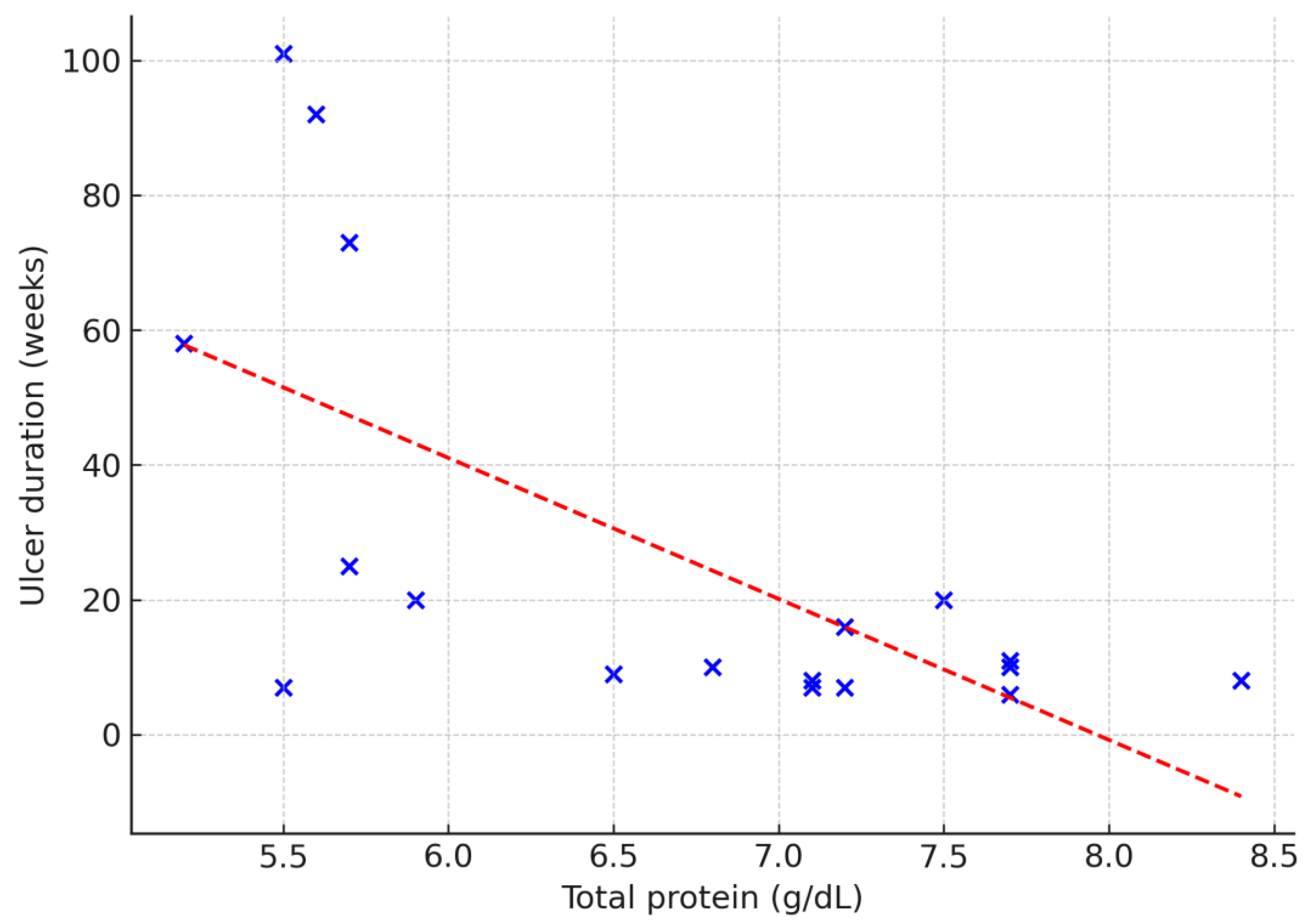
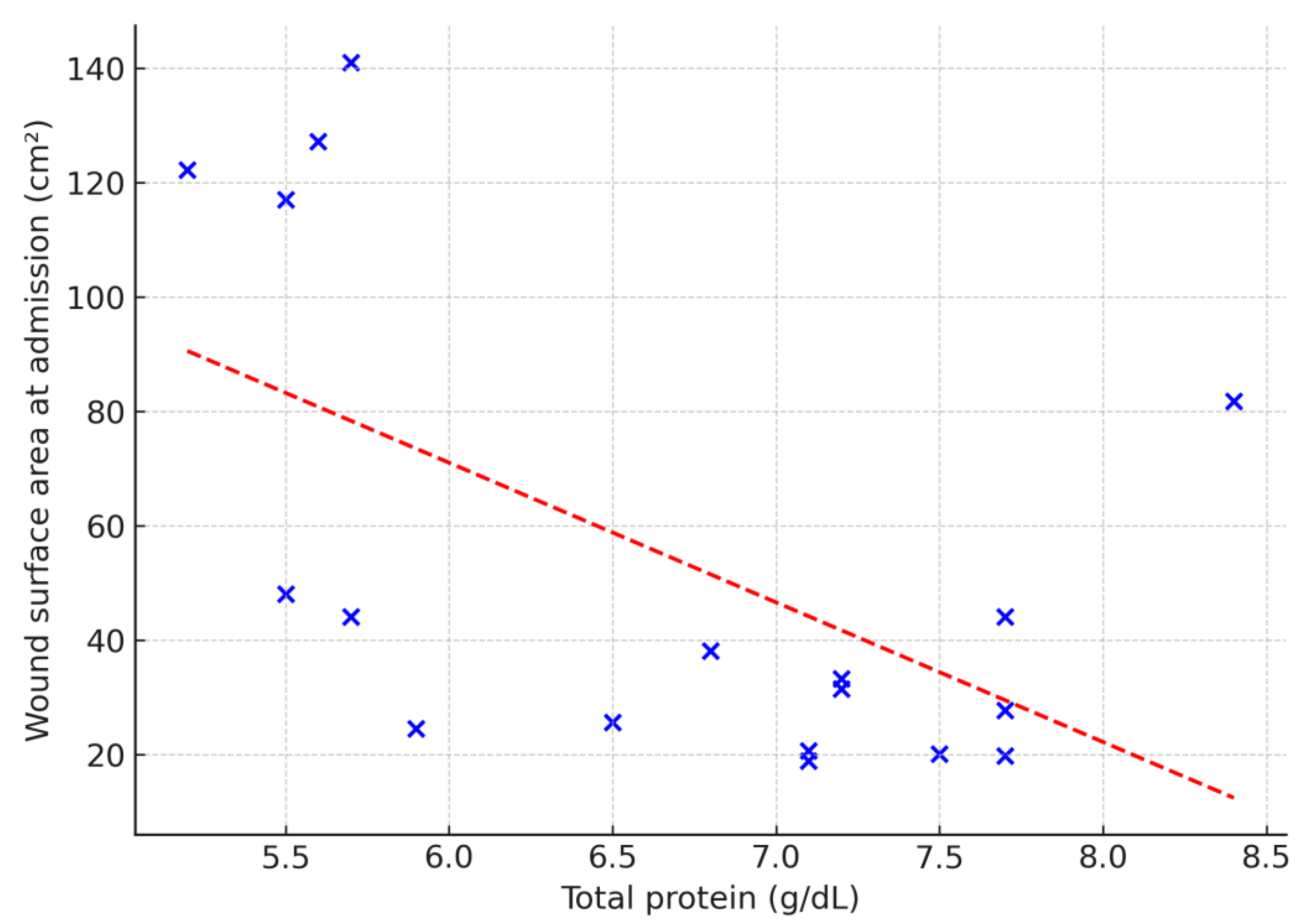
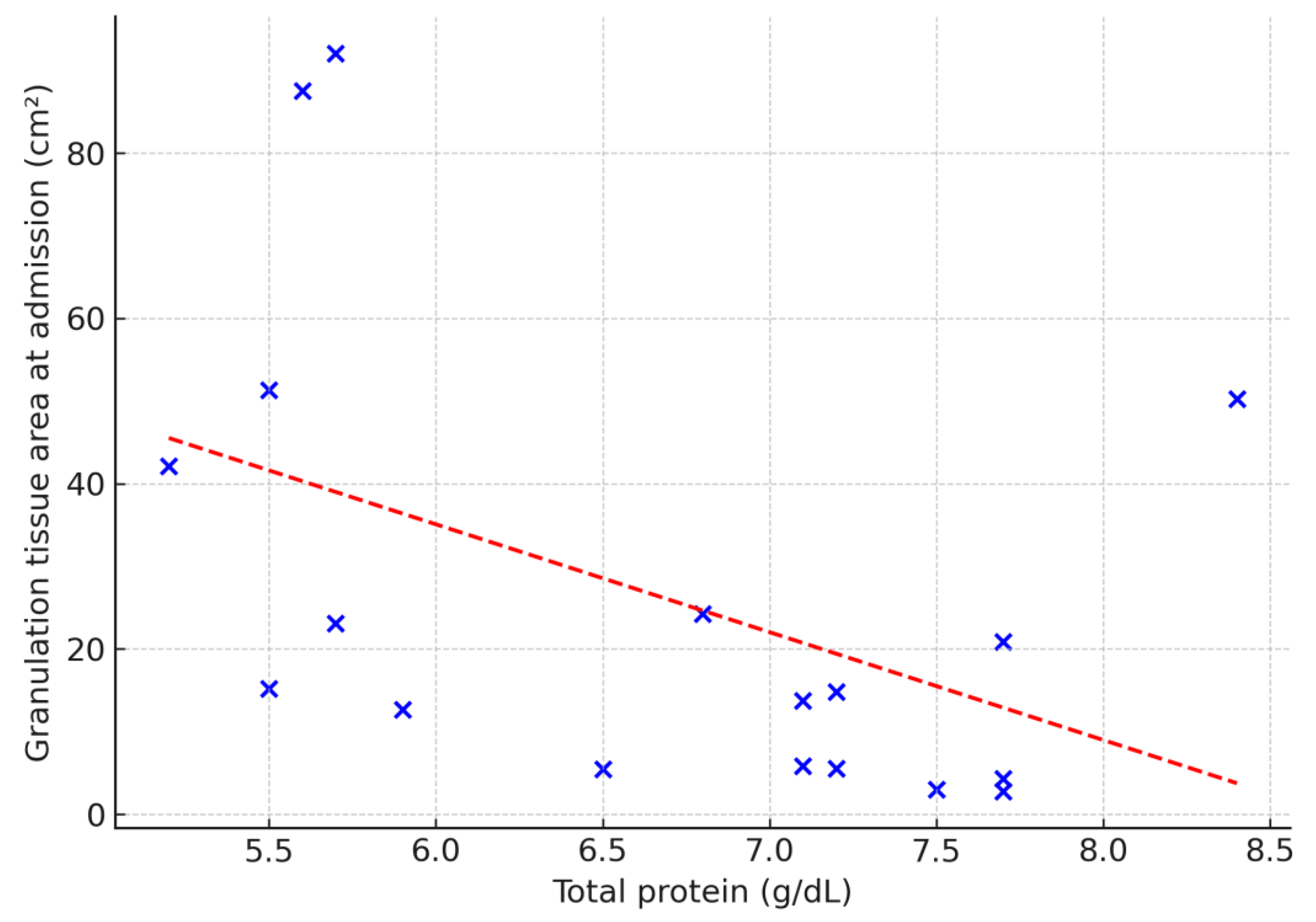
The predictive value of selected laboratory parameters in chronic wound treatment was also explored. Serum total protein correlated negatively with ulcer duration (ρ = –0.53;
p = 0.02), wound area (ρ = –0.50;
p = 0.03) and baseline granulation tissue (ρ = –0.53;
p = 0.02) (
Figure 2,
Figure 3 and
Figure 4). No other laboratory parameters demonstrated statistically significant correlations with ulcer duration, wound area, or baseline granulation tissue (all
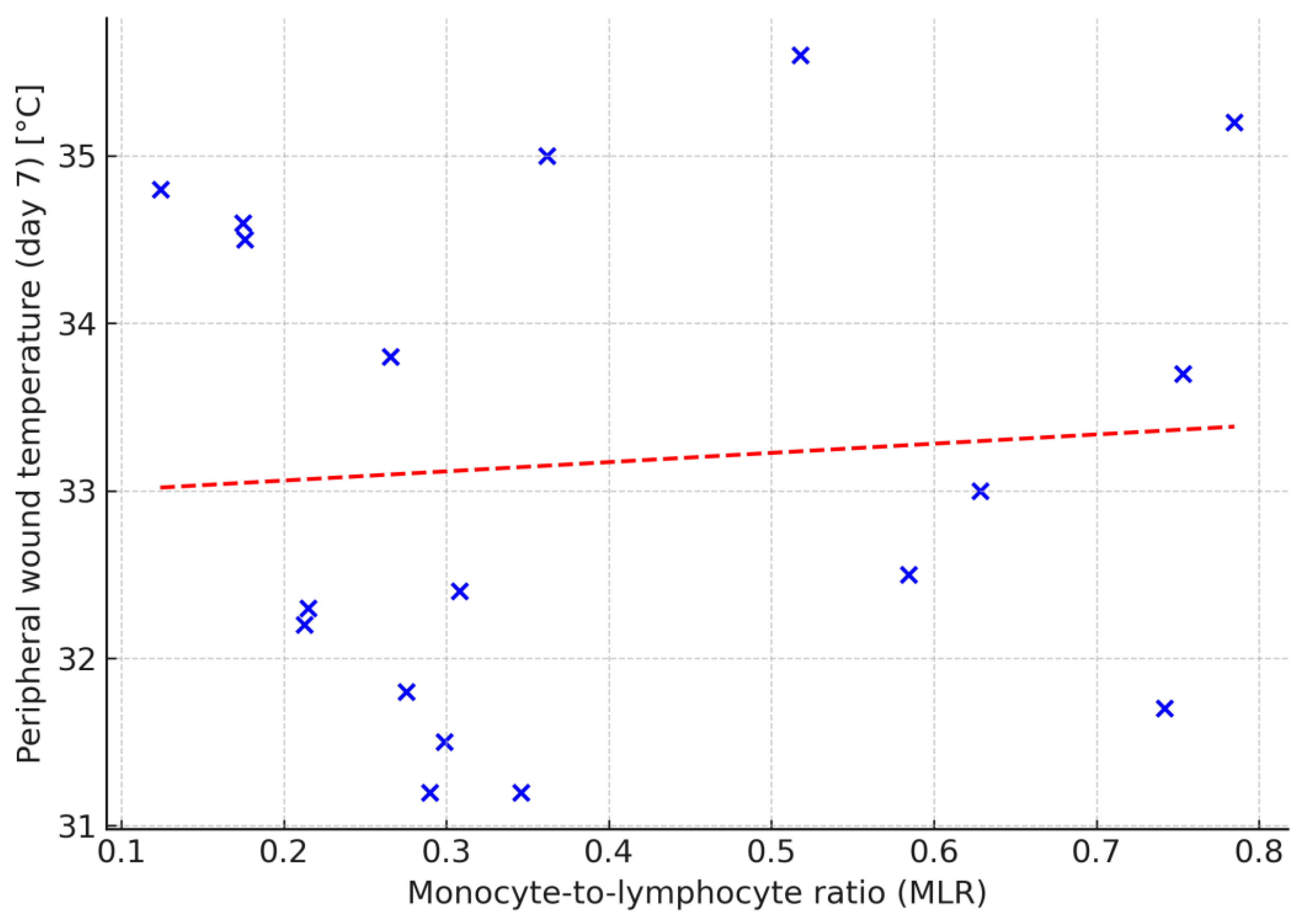
p > 0.05). Laboratory parameters were also correlated with temperatures measured at the wound bed and at the wound margin using thermography. Only one statistically significant correlation was found: between MLR and peripheral wound temperature on day 7 in the control group (ρ = 0.74,
p = 0.02;
Figure 5). No significant correlations were observed in the intervention group. After one week of therapy, no statistically significant differences were found between the groups in wound area, granulation tissue area, or necrotic tissue area (
p > 0.05). Similarly, no significant differences were noted in wound bed temperature or peripheral temperature (
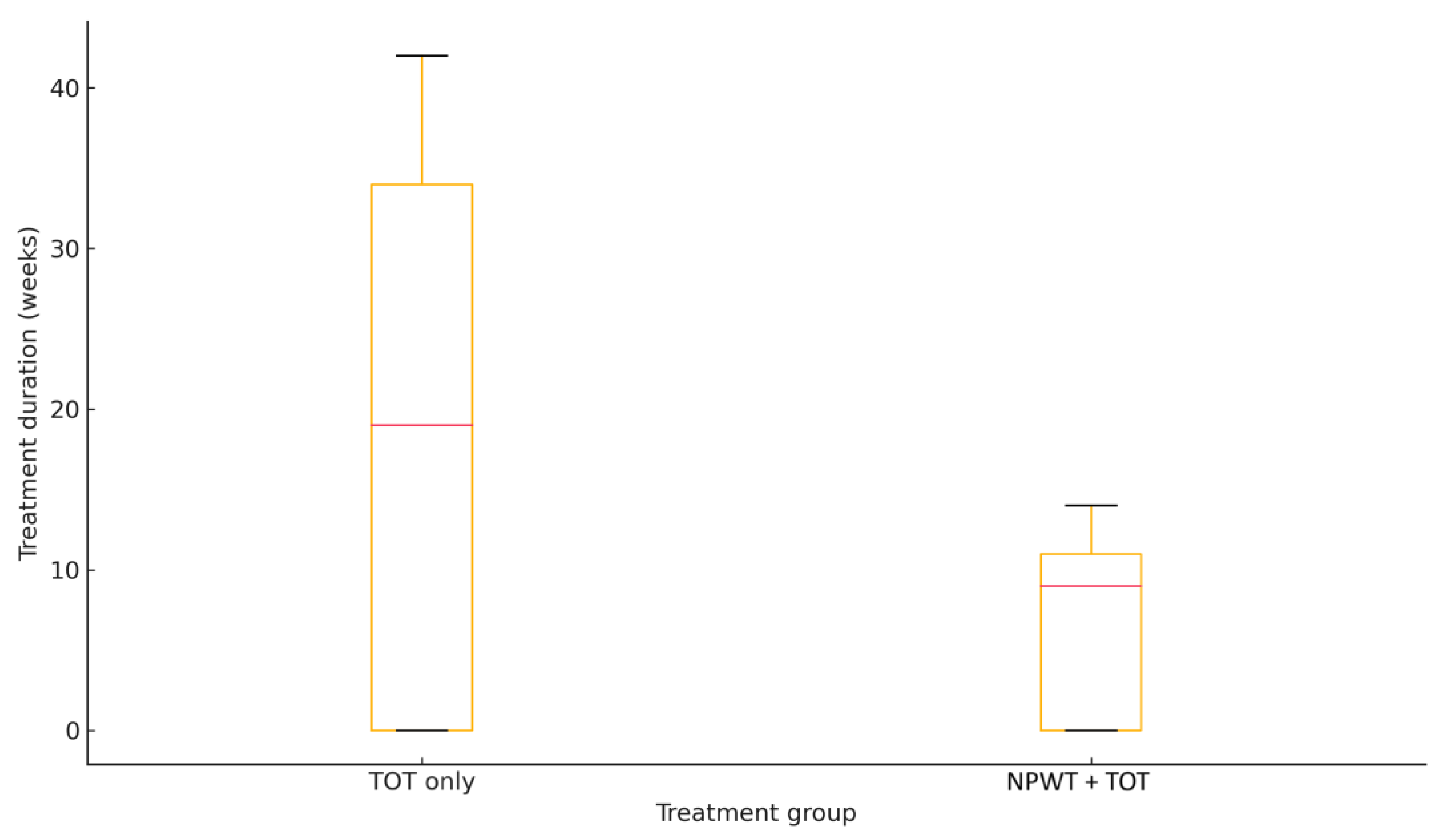
p > 0.05). The primary endpoint—total treatment duration—remained markedly shorter in the intervention arm, with a median of 10 weeks versus 34 weeks in the TOT-only group (
p = 0.008;
Figure 6). Complete closure occurred in 67% of combined therapy patients and 33% of controls, a difference that did not reach statistical significance (
p = 0.18) (
Table 2). No adverse events or complications related to the treatment methods were reported during the study period.
4. Discussion
The present pilot study demonstrates that adjunctive negative-pressure wound therapy (NPWT) administered after a short course of topical oxygen therapy (TOT) is associated with a clinically relevant reduction in overall healing time compared with TOT alone. Although the higher proportion of complete closures in our intervention arm did not achieve statistical significance, the numerical trend is consistent with findings from a recent randomized trial in advanced pressure ulcers, in which adjunctive topical oxygen shortened time-to-closure by approximately one month, as well as with a 2024 meta-analysis indicating nearly a two-fold increase in the likelihood of closure when oxygen delivery is combined with NPWT [
9,
10].
A significant association was also observed between ulcer duration and wound surface area, reinforcing the importance of early intervention in chronic wound care. These findings are consistent with prior studies indicating that prolonged inflammation in chronic wounds leads to progressive tissue damage and delayed healing [
14,
15].
Our data also highlight the potential prognostic value of serum total protein levels. The observed negative correlations between total protein and wound severity indicators (ulcer duration, wound area, granulation tissue size) suggest that nutritional status may play an important role in wound healing, aligning with previous evidence linking protein-energy malnutrition to delayed epithelialization and higher infection risk [
13]. Given its low cost and widespread availability, total protein could serve as a pragmatic triage marker prompting early dietetic referral and targeted supplementation.
Inflammatory markers—including NLR, MLR, PLR and CRP—have been proposed as indicators of systemic inflammation and tissue-repair capacity in chronic wounds, although most published work has focused on their ability to stratify amputation risk in diabetic-foot syndrome rather than to predict healing kinetics per se [
11,
12,
18]. In our study, only one statistically significant correlation was found: a strong positive relationship between MLR and peripheral wound temperature in the TOT-only group. This may reflect an interplay between systemic immune activation and local perfusion or inflammation. The absence of such correlations in the intervention group may suggest that NPWT modulates local tissue response, though the small sample size limits definitive conclusions.
Thermographic assessment of wound healing offers a promising non-invasive method to monitor local physiological changes [
15,
19]. Early wound-morphometry and thermography (day 7) did not differ between groups, indicating that macroscopic parameters measured within the first treatment week are insufficient to predict the longer-term divergence in healing trajectories. Animal data suggest that oxygen supplementation exerts its principal influence during the proliferative phase (post-day 7), when collagen synthesis and neovascularization are maximal [
20].
Strengths of the study include prospective data acquisition, objective three-dimensional wound measurement, standardized thermography, uniform treatment protocols and a nine-month follow-up schedule with minimal attrition. The study has several important limitations. First, the small sample size precluded multivariable analysis and resulted in wide confidence intervals around effect estimates. Second, the non-randomized and unblinded design may have introduced selection and performance bias, although baseline characteristics were balanced across groups. Third, the cohort comprised only venous and diabetic ulcers, limiting generalizability to arterial or pressure ulcers. Fourth, bacteriological assessment of wound biofilm was not performed, preventing evaluation of microbial dynamics in response to the interventions. Fifth, thermographic monitoring was restricted to three early time-points, which may have limited detection of perfusion changes occurring later in the healing trajectory.
Our observations strengthen the mechanistic rationale for pairing topical oxygen with negative-pressure wound therapy and highlight serum total protein as a readily obtainable marker worthy of further investigation. Ongoing multicenter work will determine whether systematically addressing nutritional deficits and integrating molecular read-outs can further enhance the effectiveness of combined advanced therapies in chronic-wound care.
5. Conclusions
The observed association between ulcer duration and wound size underscores the importance of early intervention in chronic wound management. Our laboratory and thermographic findings point to the potential value of selected biomarkers in predicting wound healing outcomes; however, these results should be interpreted with caution given the exploratory nature of the study.
Combining topical oxygen therapy with negative-pressure wound therapy was feasible, safe and associated with a clinically meaningful reduction in healing time compared with topical oxygen alone. Given the small sample and non-randomized design of this pilot, the results should be viewed as hypothesis-generating. Larger, rigorously controlled studies are required to validate these findings and to determine whether biomarker-guided, personalized protocols can further optimize chronic-wound outcomes.
Author Contributions
Conceptualization, B.M. and S.G.; methodology, B.M.; software, M.F.; validation, B.M., M.F. and R.K.; formal analysis, B.M.; investigation, B.M.; resources, R.K.; data curation, M.F.; writing—original draft preparation, B.M.; writing—review and editing, M.F. and S.G.; visualization, R.K.; supervision, S.G.; project administration, B.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Approved by the Bioethics Committee of The Jan Kochanowski University Medical College (approval no. 1/2024).
Informed Consent Statement
Obtained from all participants.
Data Availability Statement
The data are not publicly available due to ethical and privacy restrictions imposed by the institutional review board. An anonymized dataset may be made available by the corresponding author upon reasonable request and pending ethics approval.
Acknowledgments
Thanks to the nursing staff for assistance in follow-up.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript
TOT – Topical Oxygen Therapy
NPWT – Negative Pressure Wound Therapy
NLR – Neutrophil-to-Lymphocyte Ratio
MLR – Monocyte-to-Lymphocyte Ratio
PLR – Platelet-to-Lymphocyte Ratio
CRP – C-Reactive Protein
References
- Graves N, Phillips CJ, Harding K. A narrative review of the epidemiology and economics of chronic wounds. Br J Dermatol. 2022;187(2):141-148. [CrossRef]
- Olsson M, Järbrink K, Divakar U, Bajpai R, Upton Z, Schmidtchen A, et al. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019;27(1):114-125. [CrossRef]
- Díaz-Herrera MÁ, González-Durán M, Rodríguez-Martínez FJ, Tujillo-Flores G, Tuset-Mateu N, Verdú-Soriano J, et al. The financial burden of chronic wounds in primary care: A real-world data analysis on cost and prevalence. Int J Nurs Stud Adv. 2025;8:100313. [CrossRef]
- Kapp S, Santamaria N. The financial and quality-of-life cost to patients living with a chronic wound in the community. Int Wound J. 2017;14(6):1108. [CrossRef]
- Zens Y, Barth M, Bucher HC, Dreck K, Felsch M, Groß W, et al. Negative pressure wound therapy in patients with wounds healing by secondary intention: a systematic review and meta-analysis of randomized controlled trials. Syst Rev. 2020;9(1). [CrossRef]
- Normandin S, Safran T, Winocour S, Chu CK, Vorstenbosch J, Murphy AM, et al. Negative Pressure Wound Therapy: Mechanism of Action and Clinical Applications. Semin Plast Surg. 2021;35(3):164. [CrossRef]
- Cates NK, Kim PJ. Topical Oxygen Therapy for Wound Healing: A Critical Evaluation. Surg Technol Int. 2022;40. [CrossRef]
- Frykberg RG. Topical Wound Oxygen Therapy in the Treatment of Chronic Diabetic Foot Ulcers. Medicina (B Aires). 2021;57(9):917. [CrossRef]
- Zhao Y, Zhang M, Zhang C, Yan H. Effect of local oxygen therapy combined with vacuum sealing drainage on the healing of stage IV sacrococcygeal pressure ulcers. Int Wound J. 2023;20(10):4253-4261. [CrossRef]
- Yang L, Kong J, Xing Y, Pan L, Li C, Wu Z, et al. Adjunctive hyperbaric oxygen therapy and negative pressure wound therapy for hard-to-heal wounds: a systematic review and meta-analysis. J Wound Care. 2024;33(12):950-957. [CrossRef]
- Vatankhah N, Jahangiri Y, Landry GJ, McLafferty RB, Alkayed NJ, Moneta GL, et al. Predictive value of neutrophil-to-lymphocyte ratio in diabetic wound healing. J Vasc Surg. 2016;65(2):478-483. [CrossRef]
- Eren MA, Güneş AE, Kırhan İ, Sabuncu T. The role of the platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio in the prediction of length and cost of hospital stay in patients with infected diabetic foot ulcers: A retrospective comparative study. Acta Orthop Traumatol Turc. 2020;54(2):127-131. [CrossRef]
- Ju M, Kim Y, Seo KW. Role of nutrition in wound healing and nutritional recommendations for promotion of wound healing: a narrative review. Annals of Clinical Nutrition and Metabolism. 2023;15(3):67-71. [CrossRef]
- Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366(9498):1736-1743. [CrossRef]
- Iruela Sánchez M, García-Sierra R, Medrano-Jiménez R, Bonachela-Mompart D, Maella-Rius N, Soria-Martín E, et al. Use of Infrared Thermometry to Observe Temperature Variation Associated with the Healing Process in Wounds and Ulcers: TIHUAP Cohort Study Protocol. Healthcare (Basel). 2023;11(12). [CrossRef]
- Serena TE, Andersen C, Cole W, Garoufalis M, Frykberg R, Simman R. Guidelines for the use of topical oxygen therapy in the treatment of hard-to-heal wounds based on a Delphi consensus. J Wound Care. 2022;31(Sup3):S20-S24. [CrossRef]
- Gupta S, Gabriel A, Lantis J, Téot L. Clinical recommendations and practical guide for negative pressure wound therapy with instillation. Int Wound J. 2015;13(2):159. [CrossRef]
- Yürük Atasoy P, Gürbüz E, Alkan S. Prognostic Significance of NLR, LMR, PLR, and CRP-Albumin Ratio in Lower Extremity Cellulitis: A Hospitalization and Recurrence Analysis. Int J Low Extrem Wounds. Published online 2024. [CrossRef]
- Lu G, HS, CY, et al. The role of inflammatory markers in chronic wound healing. J Inflamm Res. 2020;13:51-59.
- Yip WL. Influence of oxygen on wound healing. Int Wound J. 2014;12(6):620. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).