Submitted:
12 July 2025
Posted:
15 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Sterilization Methods and Molecular Effects
2.1. Ionising Radiation
2.2. Electron Beam
2.3. Gamma Radiation
2.4. X-Ray
2.5. Ultraviolet Radiation
2.6. Ethylene Oxide
2.7. Autoclaving
2.8. Dry Heat
2.9. Ozone
2.10. Supercritical Carbon Dioxide
3. Sterilization of Polysaccharide-Based Materials
3.1. Electron Beam
3.2. Gamma
3.3. X-Ray
3.4. Ultra-Violet
3.5. Autoclave
3.6. Dry Heat
3.7. Ethylene Oxide
3.8. Ozone
3.9. SCCO2
3.10. Cold Plasma
4. Commercial Viability of Sterilized Biomaterials
4.1. Migratory Effects Caused by Sterilisation
4.2. Alteration of Physiochemical Properties
5. Commercial Advancements in Polysaccharides
6. Conclusion and Future Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ritchie H, Samborska V, Roser M. Plastic Pollution. Our World in Data 2023.
- Johnson R. Polymers in Chemistry: Versatile Materials with Diverse Applications. Journal of Chemistry 2023;12.
- Lu J. Polymer Materials in Daily Life: Classification, Applications, and Future Prospects. E3S Web of Conferences 2023;406. [CrossRef]
- Velazco-Medel MA, Camacho-Cruz LA, Bucio E. Modification of relevant polymeric materials for medical applications and devices. MEDICAL DEVICES & SENSORS 2020;3:e10073. [CrossRef]
- Rudin A, Choi P. Chapter 1 - Introductory Concepts and Definitions. In: Rudin A, Choi P, editors. The Elements of Polymer Science & Engineering (Third Edition), Boston: Academic Press; 2013, p. 1–62. [CrossRef]
- Speight JG. Chapter 14 - Monomers, polymers, and plastics. In: Speight JG, editor. Handbook of Industrial Hydrocarbon Processes (Second Edition), Boston: Gulf Professional Publishing; 2020, p. 597–649. [CrossRef]
- Smith O, Brisman A. Plastic Waste and the Environmental Crisis Industry. Crit Crim 2021;29:289–309. [CrossRef]
- Mohanan N, Montazer Z, Sharma PK, Levin DB. Microbial and Enzymatic Degradation of Synthetic Plastics. Front Microbiol 2020;11:580709. [CrossRef]
- Naderi Kalali E, Lotfian S, Entezar Shabestari M, Khayatzadeh S, Zhao C, Yazdani Nezhad H. A critical review of the current progress of plastic waste recycling technology in structural materials. Current Opinion in Green and Sustainable Chemistry 2023;40:100763. [CrossRef]
- OECD. Global plastic waste set to almost triple by 2060, says OECD. OECD 2022. https://www.oecd.org/en/about/news/press-releases/2022/06/global-plastic-waste-set-to-almost-triple-by-2060.html (accessed August 19, 2024).
- Ghasemlou M, Barrow CJ, Adhikari B. The future of bioplastics in food packaging: An industrial perspective. Food Packaging and Shelf Life 2024;43:101279. [CrossRef]
- Wellenreuther C, Wolf A, Zander N. Cost competitiveness of sustainable bioplastic feedstocks – A Monte Carlo analysis for polylactic acid. Cleaner Engineering and Technology 2022;6:100411. [CrossRef]
- Xiao L, Wang B, Yang G, Gauthier M. Poly(Lactic Acid)-Based Biomaterials: Synthesis, Modification and Applications. Biomedical Science, Engineering and Technology, 2012. [CrossRef]
- Azevedo HS, Reis RL. Enzymatic Degradation of Biodegradable Polymers and Strategies to Control Their Degradation Rate. Biodegradable Systems in Tissue Engineering and Regenerative Medicine n.d.
- Apriyanto A, Compart J, Fettke J. A review of starch, a unique biopolymer – Structure, metabolism and in planta modifications. Plant Science 2022;318:111223. [CrossRef]
- Agarwal S. Major factors affecting the characteristics of starch based biopolymer films. European Polymer Journal 2021;160:110788. [CrossRef]
- De Luca S, Milanese D, Gallichi-Nottiani D, Cavazza A, Sciancalepore C. Poly(lactic acid) and Its Blends for Packaging Application: A Review. Clean Technologies 2023;5:1304–43. [CrossRef]
- Bhaskar R, Zo SM, Narayanan KB, Purohit SD, Gupta MK, Han SS. Recent development of protein-based biopolymers in food packaging applications: A review. Polymer Testing 2023;124:108097. [CrossRef]
- Ibrahim NI, Shahar FS, Sultan MTH, Shah AUM, Safri SNA, Mat Yazik MH. Overview of Bioplastic Introduction and Its Applications in Product Packaging. Coatings 2021;11:1423. [CrossRef]
- Atanase L-I. Biopolymers for Enhanced Health Benefits. Int J Mol Sci 2023;24:16251. [CrossRef]
- Rosenboom J-G, Langer R, Traverso G. Bioplastics for a circular economy. Nat Rev Mater 2022;7:117–37. [CrossRef]
- Royer S-J, Greco F, Kogler M, Deheyn DD. Not so biodegradable: Polylactic acid and cellulose/plastic blend textiles lack fast biodegradation in marine waters. PLOS ONE 2023;18:e0284681. [CrossRef]
- Zhang L, Liao W, Huang Y, Wen Y, Chu Y, Zhao C. Global seaweed farming and processing in the past 20 years. Food Production, Processing and Nutrition 2022;4:23. [CrossRef]
- Thiviya P, Gamage A, Liyanapathiranage A, Makehelwala M, Dassanayake RS, Manamperi A, et al. Algal polysaccharides: Structure, preparation and applications in food packaging. Food Chemistry 2023;405:134903. [CrossRef]
- Sarangi MK, Rao MEB, Parcha V, Yi DK, Nanda SS. Chapter 22 - Marine polysaccharides for drug delivery in tissue engineering. In: Hasnain MS, Nayak AK, editors. Natural Polysaccharides in Drug Delivery and Biomedical Applications, Academic Press; 2019, p. 513–30. [CrossRef]
- Beaumont M, Tran R, Vera G, Niedrist D, Rousset A, Pierre R, et al. Hydrogel-Forming Algae Polysaccharides: From Seaweed to Biomedical Applications. Biomacromolecules n.d.;22:2021. [CrossRef]
- Mišurcová L, Škrovánková S, Samek D, Ambrožová J, Machů L. Chapter 3 - Health Benefits of Algal Polysaccharides in Human Nutrition. In: Henry J, editor. Advances in Food and Nutrition Research, vol. 66, Academic Press; 2012, p. 75–145. [CrossRef]
- Sionkowska A. Current research on the blends of natural and synthetic polymers as new biomaterials: Review. Progress in Polymer Science 2011;36:1254–76. [CrossRef]
- Ashfaq A, Clochard M-C, Coqueret X, Dispenza C, Driscoll MS, Ulański P, et al. Polymerization Reactions and Modifications of Polymers by Ionizing Radiation. Polymers (Basel) 2020;12:2877. [CrossRef]
- Malinowski M. Using X-ray Technology to Sterilize Medical Devices. AJBSR 2021;12:272–6. [CrossRef]
- L’annunziata MF. 1 - NUCLEAR RADIATION, ITS INTERACTION WITH MATTER AND RADIOISOTOPE DECAY. In: L’Annunziata MF, editor. Handbook of Radioactivity Analysis (Second Edition), San Diego: Academic Press; 2003, p. 1–121. [CrossRef]
- Tafti D, Maani CV. X-ray Production. StatPearls, Treasure Island (FL): StatPearls Publishing; 2024.
- Boardman EA. Deep ultraviolet (UVC) laser for sterilisation and fluorescence applications n.d.
- de Almeida MTG, de Almeida BG, Siqueira JPZ, Byzynski Soares G, Sigari Morais V, Mitsue Yasuoka FM, et al. Ultraviolet-C Light-emitting Device Against Microorganisms in Beauty Salons. Pathog Immun 2022;7:49–59. [CrossRef]
- Freeman DE. Chapter 10 - Sterilization and Antiseptics. In: Auer JA, Stick JA, editors. Equine Surgery (Third Edition), Saint Louis: W.B. Saunders; 2006, p. 112–23. [CrossRef]
- Lachenmeier DW. Chapter 24 - Antiseptic Drugs and Disinfectants. In: Ray SD, editor. Side Effects of Drugs Annual, vol. 36, Elsevier; 2014, p. 339–46. [CrossRef]
- Mendes GCC, Brandão TRS, Silva CLM. Ethylene oxide sterilization of medical devices: A review. American Journal of Infection Control 2007;35:574–81. [CrossRef]
- Pflug JJ, Holcomb R, Gomez M. Principles of thermal destruction of microorganisms. Disinfection, Sterilization and Preservation. 5th ed., Philadelphia: Lippincott Williams & Wilkins; 2001.
- Rogers WJ. 4 - The effects of sterilization on medical materials and welded devices. In: Zhou Y, Breyen MD, editors. Joining and Assembly of Medical Materials and Devices, Woodhead Publishing; 2013, p. 79–130. [CrossRef]
- Green H, Kidd J, Jackson LS. Novel and Emerging Cleaning and Sanitization Technologies. In: Smithers GW, editor. Encyclopedia of Food Safety (Second Edition), Oxford: Academic Press; 2024, p. 739–45. [CrossRef]
- Epelle EI, Macfarlane A, Cusack M, Burns A, Okolie JA, Vichare P, et al. Ozone Decontamination of Medical and Nonmedical Devices: An Assessment of Design and Implementation Considerations. Ind Eng Chem Res 2023;62:4191–209. [CrossRef]
- Jerrett M, Burnett RT, Pope CA, Ito K, Thurston G, Krewski D, et al. Long-Term Ozone Exposure and Mortality. New England Journal of Medicine 2009;360:1085–95. [CrossRef]
- Middleton J, Burks B, Wells T, Setters AM, Jasiuk I, Predecki P, et al. The effect of ozone on polymer degradation in Polymer Core Composite Conductors. Polymer Degradation and Stability 2013;98:436–45. [CrossRef]
- McHugh M, Krukonis V. Supercritical Fluid Extraction: Principles and Practice. Elsevier; 2013.
- Zhang J, Davis TA, Matthews MA, Drews MJ, LaBerge M, An YH. Sterilization using high-pressure carbon dioxide. The Journal of Supercritical Fluids 2006;38:354–72. [CrossRef]
- Shyam R, Palaniappan A. Effect of sterilization techniques on biomaterial inks’ properties and 3D bioprinting parameters. Bioprinting 2023;33:e00294. [CrossRef]
- Rubi RVC, Quitain AT, Agutaya JKCN, Doma BT, Soriano AN, Auresenia J, et al. Synergy of in-situ formation of carbonic acid and supercritical CO2-expanded liquids: Application to extraction of andrographolide from Andrographis paniculata. The Journal of Supercritical Fluids 2019;152:104546. [CrossRef]
- Zhou X, Li Y, editors. Chapter 1 - Basic Biology of Oral Microbes. Atlas of Oral Microbiology, Oxford: Academic Press; 2015, p. 1–14. [CrossRef]
- Dillow AK, Dehghani F, Hrkach JS, Foster NR, Langer R. Bacterial inactivation by using near- and supercritical carbon dioxide. Proceedings of the National Academy of Sciences 1999;96:10344–8. [CrossRef]
- Spilimbergo S, Elvassore N, Bertucco A. Microbial inactivation by high-pressure. The Journal of Supercritical Fluids 2002;22:55–63. [CrossRef]
- Liang W, Zhao W, Liu X, Zheng J, Sun Z, Ge X, et al. Understanding how electron beam irradiation doses and frequencies modify the multiscale structure, physicochemical properties, and in vitro digestibility of potato starch. Food Research International 2022;162:111947. [CrossRef]
- Zhou X, Ye X, He J, Wang R, Jin Z. Effects of electron beam irradiation on the properties of waxy maize starch and its films. International Journal of Biological Macromolecules 2020;151:239–46. [CrossRef]
- Cleland M, Galloway R, Genin F, Lindholm M. The use of dose and charge distributions in electron beam processing. Radiation Physics and Chemistry 2002;63:729–33. [CrossRef]
- Driscoll M, Stipanovic A, Winter W, Cheng K, Manning M, Spiese J, et al. Electron beam irradiation of cellulose. Radiation Physics and Chemistry 2009;78:539–42. [CrossRef]
- Clinical Applicable Carboxymethyl Chitosan with Gel-Forming and Stabilizing Properties Based on Terminal Sterilization Methods of Electron Beam Irradiation | ACS Omega n.d. https://pubs.acs.org/doi/full/10.1021/acsomega.4c01299 (accessed April 15, 2025).
- Gryczka U. LONG-TERM RADIATION-INDUCED EFFECTS ON SOLID STATE CHITOSAN. | Progress on Chemistry & Application of Chitin & its Derivatives | EBSCOhost 2021;26:80. [CrossRef]
- Iuliano A, Fabiszewska A, Kozik K, Rzepna M, Ostrowska J, Dębowski M, et al. Effect of Electron-Beam Radiation and Other Sterilization Techniques on Structural, Mechanical and Microbiological Properties of Thermoplastic Starch Blend. J Polym Environ 2021;29:1489–504. [CrossRef]
- Farno M, Lamarche C, Tenailleau C, Cavalié S, Duployer B, Cussac D, et al. Low-energy electron beam sterilization of solid alginate and chitosan, and their polyelectrolyte complexes. Carbohydrate Polymers 2021;261:117578. [CrossRef]
- Chung K-H, Othman Z, Lee J-S. Gamma irradiation of corn starches with different amylose-to-amylopectin ratio. J Food Sci Technol 2015;52:6218–29. [CrossRef]
- Ibrahim SM. Characterization, mechanical, and thermal properties of gamma irradiated starch films reinforced with mineral clay. Journal of Applied Polymer Science 2011;119:685–92. [CrossRef]
- Atrous H, Benbettaieb N, Chouaibi M, Attia H, Ghorbel D. Changes in wheat and potato starches induced by gamma irradiation: A comparative macro and microscopic study. International Journal of Food Properties 2017;20:1532–46. [CrossRef]
- Abad LV, Aranilla CT, Relleve LS, Dela Rosa AM. Emerging applications of radiation-modified carrageenans. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2014;336:167–72. [CrossRef]
- Choi J, Gu Lee S, Jong Han S, Cho M, Cheon Lee P. Effect of gamma irradiation on the structure of fucoidan. Radiation Physics and Chemistry 2014;100:54–8. [CrossRef]
- Choi J, Kim H-J. Preparation of low molecular weight fucoidan by gamma-irradiation and its anticancer activity. Carbohydrate Polymers 2013;97:358–62. [CrossRef]
- Lee D, Choi W-S, Byun M, Park H, Yu Y-M, Lee C. Effect of γ-Irradiation on Degradation of Alginate. Journal of Agricultural and Food Chemistry 2003;51:4819–23. [CrossRef]
- Lim L-Y, Khor E, Koo O. γ Irradiation of chitosan. Journal of Biomedical Materials Research 1998;43:282–90. [CrossRef]
- Li B, Li J, Xia J, Kennedy JF, Yie X, Liu TG. Effect of gamma irradiation on the condensed state structure and mechanical properties of konjac glucomannan/chitosan blend films. Carbohydrate Polymers 2011;83:44–51. [CrossRef]
- De Kerf M, Mondelaers W, Lahorte P, Vervaet C, Remon JP. Characterisation and disintegration properties of irradiated starch. International Journal of Pharmaceutics 2001;221:69–76. [CrossRef]
- Bajer D, Kaczmarek H, Bajer K. The structure and properties of different types of starch exposed to UV radiation: A comparative study. Carbohydrate Polymers 2013;98:477–82. [CrossRef]
- Kurdziel M, Łabanowska M, Pietrzyk S, Pająk P, Królikowska K, Szwengiel A. The effect of UV-B irradiation on structural and functional properties of corn and potato starches and their components. Carbohydrate Polymers 2022;289:119439. [CrossRef]
- Nowak E, Wisła-Świder A, Leszczyńska T, Koronowicz A. Physicochemical and Molecular Properties of Spelt and Wheat Starches Illuminated with UV Light. Applied Sciences 2023;13:2360. [CrossRef]
- Sedayu BB, Cran MJ, Bigger SW. Effects of surface photocrosslinking on the properties of semi-refined carrageenan film. Food Hydrocolloids 2021;111:106196. [CrossRef]
- Prasetyaningrum A, Widayat W, Jos B, Dharmawan Y, Ratnawati R. UV Irradiation and Ozone Treatment of κ-Carrageenan: Kinetics and Products Characteristics. Bulletin of Chemical Reaction Engineering & Catalysis 2020;15:319–30. [CrossRef]
- Wang L, Jayawardena TU, Hyun J, Wang K, Fu X, Xu J, et al. Antioxidant and anti-photoaging effects of a fucoidan isolated from Turbinaria ornata. International Journal of Biological Macromolecules 2023;225:1021–7. [CrossRef]
- Habibi M, Golmakani M-T, Eskandari MH, Hosseini SMH. Potential prebiotic and antibacterial activities of fucoidan from Laminaria japonica. International Journal of Biological Macromolecules 2024;268:131776. [CrossRef]
- Meynaud S, Huet G, Brulé D, Gardrat C, Poinssot B, Coma V. Impact of UV Irradiation on the Chitosan Bioactivity for Biopesticide Applications. Molecules 2023;28:4954. [CrossRef]
- Sionkowska A, Kaczmarek B, Gnatowska M, Kowalonek J. The influence of UV-irradiation on chitosan modified by the tannic acid addition. Journal of Photochemistry and Photobiology B: Biology 2015;148:333–9. [CrossRef]
- Chansoria P, Narayanan LK, Wood M, Alvarado C, Lin A, Shirwaiker RA. Effects of Autoclaving, EtOH, and UV Sterilization on the Chemical, Mechanical, Printability, and Biocompatibility Characteristics of Alginate. ACS Biomater Sci Eng 2020;6:5191–201. [CrossRef]
- (PDF) A Preliminary Investigation of UV Crosslinked Alginate- based Hydrogel for Cardiac-tissue Mimicking Material Potential. ResearchGate n.d. [CrossRef]
- Hofmann S, Stok KS, Kohler T, Meinel AJ, Müller R. Effect of sterilization on structural and material properties of 3-D silk fibroin scaffolds. Acta Biomaterialia 2014;10:308–17. [CrossRef]
- Hashimoto T, Nakamura Y, Tamada Y, Kurosu H, Kameda T. The influence of thermal treatments on the secondary structure of silk fibroin scaffolds and their interaction with fibroblasts. PeerJ Materials Science 2020;2:e8. [CrossRef]
- Zheng M, Xiao Y, Yang S, Liu H, Liu M, Yaqoob S, et al. Effects of heat–moisture, autoclaving, and microwave treatments on physicochemical properties of proso millet starch. Food Science & Nutrition 2020;8:735–43. [CrossRef]
- Zhu Y, Liu L, Sun Z, Ji Y, Wang D, Mei L, et al. Fucoidan as a marine-origin prebiotic modulates the growth and antibacterial ability of Lactobacillus rhamnosus. International Journal of Biological Macromolecules 2021;180:599–607. [CrossRef]
- Stoppel WL, White JC, Horava SD, Henry AC, Roberts SC, Bhatia SR. Terminal sterilization of alginate hydrogels: Efficacy and impact on mechanical properties. J Biomed Mater Res B Appl Biomater 2014;102:877–84. [CrossRef]
- Ofori-Kwakye K, Martin GP. Viscoelastic characterisation of calcium alginate gels intended for wound healing. Journal of Science and Technology (Ghana) 2005;25:46–52. [CrossRef]
- Juan AS, Montembault A, Gillet D, Say JP, Rouif S, Bouet T, et al. Degradation of chitosan-based materials after different sterilization treatments. IOP Conf Ser: Mater Sci Eng 2012;31:012007. [CrossRef]
- Gossla E, Tonndorf R, Bernhardt A, Kirsten M, Hund R-D, Aibibu D, et al. Electrostatic flocking of chitosan fibres leads to highly porous, elastic and fully biodegradable anisotropic scaffolds. Acta Biomaterialia 2016;44:267–76. [CrossRef]
- Mohd Adzahan N, Dzulkifly MH, Abdul Rahman R. Effect of heat treatment on the physico-chemical properties of starch from different botanical sources. International Food Research Journal 2010;17:127–35.
- Liu K, Hao Y, Chen Y, Gao Q. Effects of dry heat treatment on the structure and physicochemical properties of waxy potato starch. International Journal of Biological Macromolecules 2019;132:1044–50. [CrossRef]
- Eha K, Pehk T, Heinmaa I, Kaleda A, Laos K. Impact of short-term heat treatment on the structure and functional properties of commercial furcellaran compared to commercial carrageenans. Heliyon 2021;7. [CrossRef]
- Ouyang Q-Q, Hu Z, Li S-D, Quan W-Y, Wen L-L, Yang Z-M, et al. Thermal degradation of agar: Mechanism and toxicity of products. Food Chemistry 2018;264:277–83. [CrossRef]
- Mao B, Bentaleb A, Louerat F, Divoux T, Snabre P. Heat-induced aging of agar solutions: Impact on the structural and mechanical properties of agar gels. Food Hydrocolloids 2017;64:59–69. [CrossRef]
- Soazo M, Báez G, Barboza A, Busti PA, Rubiolo A, Verdini R, et al. Heat treatment of calcium alginate films obtained by ultrasonic atomizing: Physicochemical characterization. Food Hydrocolloids 2015;51:193–9. [CrossRef]
- Lim LY, Khor E, Ling CE. Effects of dry heat and saturated steam on the physical properties of chitosan. J Biomed Mater Res 1999;48:111–6. [CrossRef]
- Marreco PR, Moreira P da L, Genari SC, Moraes ÂM. Effects of different sterilization methods on the morphology, mechanical properties, and cytotoxicity of chitosan membranes used as wound dressings. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2004;71B:268–77. [CrossRef]
- Rediguieri CF, de Jesus Andreoli Pinto T, Bou-Chacra NA, Galante R, de Araújo GLB, do Nascimento Pedrosa T, et al. Ozone Gas as a Benign Sterilization Treatment for PLGA Nanofiber Scaffolds. Tissue Eng Part C Methods 2016;22:338–47. [CrossRef]
- Tyubaeva P, Zykova A, Podmasteriev V, Olkhov A, Popov A, Iordanskii A. The Investigation of the Structure and Properties of Ozone-Sterilized Nonwoven Biopolymer Materials for Medical Applications. Polymers 2021;13:1268. [CrossRef]
- Bento CSA, Alarico S, Empadinhas N, de Sousa HC, Braga MEM. Sequential scCO2 drying and sterilisation of alginate-gelatine aerogels for biomedical applications. The Journal of Supercritical Fluids 2022;184:105570. [CrossRef]
- Donegan M, Milosavljević V, Dowling DP. Activation of PET Using an RF Atmospheric Plasma System. Plasma Chem Plasma Process 2013;33:941–57. [CrossRef]
- Pankaj SK, Bueno-Ferrer C, Misra NN, Milosavljević V, O’Donnell CP, Bourke P, et al. Applications of cold plasma technology in food packaging. Trends in Food Science & Technology 2014;35:5–17. [CrossRef]
- Sobhan A, Sher M, Muthukumarappan K, Zhou R, Wei L. Cold plasma treatment for E. coli inactivation and characterization for fresh food safety. Journal of Agriculture and Food Research 2024;18:101403. [CrossRef]
- Pan Y, Zhang Y, Cheng J-H, Sun D-W. Inactivation of Listeria Monocytogenes at various growth temperatures by ultrasound pretreatment and cold plasma. LWT 2020;118:108635. [CrossRef]
- Liew KJ, Zhang X, Cai X, Ren D, Chen J, Chang Z, et al. The Biological Responses of Staphylococcus aureus to Cold Plasma Treatment. Processes 2023;11:1188. [CrossRef]
- Misra `N.N, Yadav B, Roopesh MS, Jo C. Cold Plasma for Effective Fungal and Mycotoxin Control in Foods: Mechanisms, Inactivation Effects, and Applications. Comprehensive Reviews in Food Science and Food Safety 2019;18. [CrossRef]
- Guo Z, Gou Q, Yang L, Yu Q, Han L. Dielectric barrier discharge plasma: A green method to change structure of potato starch and improve physicochemical properties of potato starch films. Food Chemistry 2022;370:130992. [CrossRef]
- Sifuentes-Nieves I, Mendez-Montealvo G, Flores-Silva PC, Nieto-Pérez M, Neira-Velazquez G, Rodriguez-Fernandez O, et al. Dielectric barrier discharge and radio-frequency plasma effect on structural properties of starches with different amylose content. Innovative Food Science & Emerging Technologies 2021;68:102630. [CrossRef]
- Aronson JK, editor. Ethylene oxide. Meyler’s Side Effects of Drugs (Sixteenth Edition), Oxford: Elsevier; 2016, p. 198–202. [CrossRef]
- Rederstorff E, Fatimi A, Sinquin C, Ratiskol J, Merceron C, Vinatier C, et al. Sterilization of Exopolysaccharides Produced by Deep-Sea Bacteria: Impact on Their Stability and Degradation. Marine Drugs 2011;9:224–41. [CrossRef]
- Mäkelä NM. Cereal β-glucan in aqueous solutions: Oxidation and structure formation. Academic Dissertation. University of Helsinki, 2017.
- Wang L, Cardenas RB, Watson C. An isotope dilution ultra high performance liquid chromatography-tandem mass spectrometry method for the simultaneous determination of sugars and humectants in tobacco products. Journal of Chromatography A 2017;1514:95–102. [CrossRef]
- Abad LV, Kudo H, Saiki S, Nagasawa N, Tamada M, Katsumura Y, et al. Radiation degradation studies of carrageenans. Carbohydrate Polymers 2009;78:100–6. [CrossRef]
- Mzoughi Z, Chakroun I, Hamida SB, Rihouey C, Mansour HB, Le Cerf D, et al. Ozone treatment of polysaccharides from Arthrocnemum indicum: Physico-chemical characterization and antiproliferative activity. International Journal of Biological Macromolecules 2017;105:1315–23. [CrossRef]
- Uyarcan M, Güngör SC. Improving functional properties of starch-based films by ultraviolet (UV-C) technology: Characterization and application on minced meat packaging. International Journal of Biological Macromolecules 2024;282:137085. [CrossRef]
- Ribeiro N, Soares GC, Santos-Rosales V, Concheiro A, Alvarez-Lorenzo C, García-González CA, et al. A new era for sterilization based on supercritical CO2 technology. J Biomed Mater Res 2020;108:399–428. [CrossRef]
- Gryczka U, Dondi D, Chmielewski AG, Migdal W, Buttafava A, Faucitano A. The mechanism of chitosan degradation by gamma and e-beam irradiation. Radiation Physics and Chemistry 2009;78:543–8. [CrossRef]
- Ohara T. Using X-ray Technology to Sterilize Medical Devices. AJBSR 2021;12:272.
- Bajer D, Kaczmarek H, Bajer K. The structure and properties of different types of starch exposed to UV radiation: A comparative study. Carbohydrate Polymers 2013;98:477–82. [CrossRef]
- Zheng M, Xiao Y, Yang S, Liu H, Liu M, Yaqoob S, et al. Effects of heat–moisture, autoclaving, and microwave treatments on physicochemical properties of proso millet starch. Food Science & Nutrition 2020;8:735–43. [CrossRef]
- Liu K, Hao Y, Chen Y, Gao Q. Effects of dry heat treatment on the structure and physicochemical properties of waxy potato starch. International Journal of Biological Macromolecules 2019;132:1044–50. [CrossRef]
- Marreco PR, Moreira P da L, Genari SC, Moraes ÂM. Effects of different sterilization methods on the morphology, mechanical properties, and cytotoxicity of chitosan membranes used as wound dressings. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2004;71B:268–77. [CrossRef]
- Tyubaeva P, Zykova A, Podmasteriev V, Olkhov A, Popov A, Iordanskii A. The Investigation of the Structure and Properties of Ozone-Sterilized Nonwoven Biopolymer Materials for Medical Applications. Polymers 2021;13:1268. [CrossRef]
- Bento CSA, Alarico S, Empadinhas N, de Sousa HC, Braga MEM. Sequential scCO2 drying and sterilisation of alginate-gelatine aerogels for biomedical applications. The Journal of Supercritical Fluids 2022;184:105570. [CrossRef]
- Lee Y, Cho J, Sohn J, Kim C. Health Effects of Microplastic Exposures: Current Issues and Perspectives in South Korea. Yonsei Med J 2023;64:301. [CrossRef]
- Yan J, Zhang Z, Lai B, Wang C, Wu H. Recent advances in marine-derived protein/polysaccharide hydrogels: Classification, fabrication, characterization, mechanism and food applications. Trends in Food Science & Technology 2024;151:104637. [CrossRef]
- Jeong G-J, Khan F, Kim D-K, Cho K-J, Tabassum N, Choudhury A, et al. Marine polysaccharides for antibiofilm application: A focus on biomedical fields. International Journal of Biological Macromolecules 2024;283:137786. [CrossRef]
- Chegu Krishnamurthi M, Tiwari S, Veera Bramhachari P, Swarnalatha GV. Exploitation of Marine-Derived Multifunctional Biomaterials in Biomedical Engineering and Drug Delivery. In: Veera Bramhachari P, Berde CV, editors. Marine Bioactive Molecules for Biomedical and Pharmacotherapeutic Applications, Singapore: Springer Nature Singapore; 2023, p. 231–50. [CrossRef]
- Liu J, Zhang Z, Deng Y, Chen G. Effect of extraction method on the structure and bioactivity of polysaccharides from activated sludge. Water Research 2024;253:121196. [CrossRef]
- Sacramento MMA, Borges J, Correia FJS, Calado R, Rodrigues JMM, Patrício SG, et al. Green approaches for extraction, chemical modification and processing of marine polysaccharides for biomedical applications. Front Bioeng Biotechnol 2022;10:1041102. [CrossRef]
- Premarathna AD, Ahmed TAE, Kulshreshtha G, Humayun S, Shormeh Darko CN, Rjabovs V, et al. Polysaccharides from red seaweeds: Effect of extraction methods on physicochemical characteristics and antioxidant activities. Food Hydrocolloids 2024;147:109307. [CrossRef]
- Akbal A, Şahin S, Güroy B. Optimization of ultrasonic-assisted extraction of polysaccharides from Ulva rigida and evaluation of their antioxidant activity. Algal Research 2024;77:103356. [CrossRef]
- Abolore RS, Jaiswal S, Jaiswal AK. Green and sustainable pretreatment methods for cellulose extraction from lignocellulosic biomass and its applications: A review. Carbohydrate Polymer Technologies and Applications 2024;7:100396. [CrossRef]
- Riseh RS, Vazvani MG, Hassanisaadi M, Thakur VK. Agricultural wastes: A practical and potential source for the isolation and preparation of cellulose and application in agriculture and different industries. Industrial Crops and Products 2024;208:117904. [CrossRef]
- Dorantes-Fuertes M-G, López-Méndez MC, Martínez-Castellanos G, Meléndez-Armenta RÁ, Jiménez-Martínez H-E. Starch Extraction Methods in Tubers and Roots: A Systematic Review. Agronomy 2024;14:865. [CrossRef]
- Sheridan E, Filonenko S, Volikov A, Sirviö JA, Antonietti M. A systematic study on the processes of lignin extraction and nanodispersion to control properties and functionality. Green Chem 2024;26:2967–84. [CrossRef]
- Wibowo CH, Ariawan D, Surojo E, Sunardi S. Microcrystalline Cellulose as Composite Reinforcement: Assessment and Future Prospects. MSF 2024;1122:65–80. [CrossRef]
- Gorgun E, Ali A, Islam MdS. Biocomposites of Poly(Lactic Acid) and Microcrystalline Cellulose: Influence of the Coupling Agent on Thermomechanical and Absorption Characteristics. ACS Omega 2024;9:11523–33. [CrossRef]
- Salehi Abar E, Vandghanooni S, Torab A, Jaymand M, Eskandani M. A comprehensive review on nanocomposite biomaterials based on gelatin for bone tissue engineering. International Journal of Biological Macromolecules 2024;254:127556. [CrossRef]
- He H, Wu Z, Xiao S, Yin P. Characterization of corn starch/chitosan composite films incorporated with amino-, carboxyl-, and hydroxyl-terminated hyperbranched dendrimers: A comparative study. International Journal of Biological Macromolecules 2024;283:137573. [CrossRef]
- Shi B, Hao Z, Du Y, Jia M, Xie S. Mechanical and Barrier Properties of Chitosan-based Composite Film as Food Packaging: A Review. BioResources 2024;19:4001–14.
| Method | Mechanism | Effects on Polysaccharides | Potential Byproducts | Reference |
|---|---|---|---|---|
| Ethylene Oxide (EtO) | Alkylation, gaseous diffusion | Absorption into hydrophilic matrix, slow desorption | Residual EtO; cytotoxicity; potential DNA alkylation | [108] |
| Autoclaving | Moist heat, hydrolysis | Glycosidic bond cleavage; structural degradation | Release of simple sugars, Maillard reaction forms mutagens | [109] |
| Dry Heat | High-temperature thermal degradation | Dehydration of plasticizers like glycerol | Acrolein / thermal degradation products | [110] |
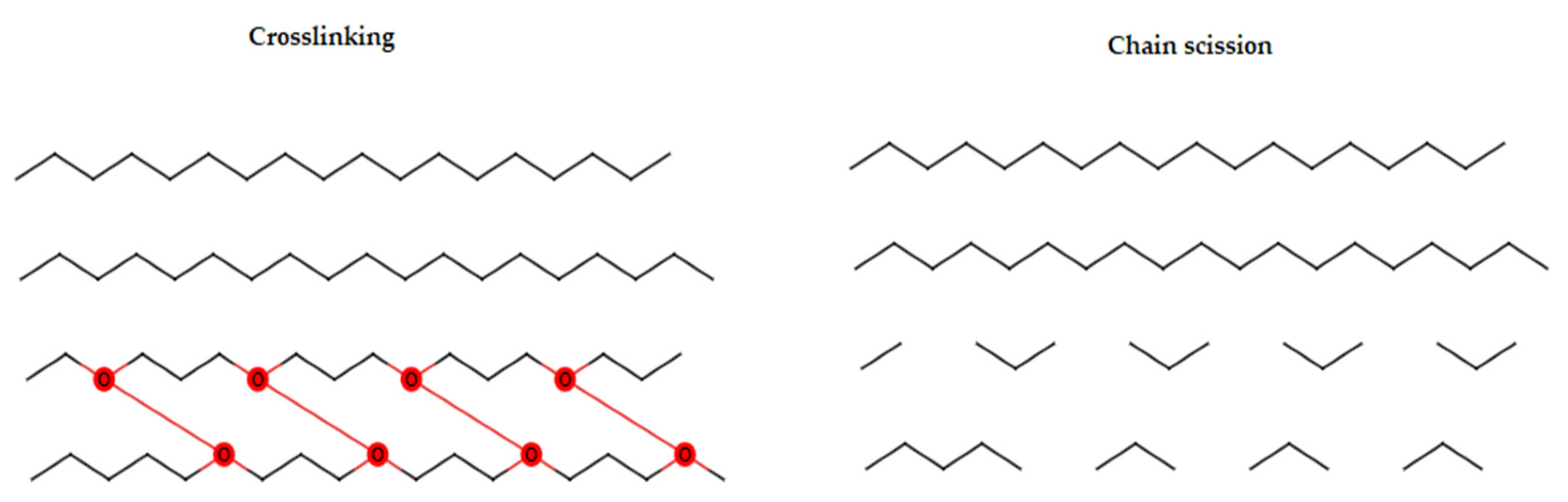
| Gamma / E-Beam Radiation | Radiolysis via free radicals | Oxidative chain scission; sugar ring opening | Formaldehyde (from formyl radicals), acetic acid | [111] |
| Ozone Sterilization | Strong oxidizer surface oxidation | Oxidation of sugar residues and additives | Aldehydes, carboxylic acids degradation of structural groups | [112] |
| UV-C Radiation | Surface photolysis, oxidation | Limited penetration surface oxidation | Surface-level carbonyls, aldehydes (low concentration) | [113] |
| Supercritical CO2 | Penetrating, inert under typical conditions | Physically absorbed, minimal chemical reactivity | Minimal unless additives are used (H2O2/peracetic acid) | [114] |
| Sterilization Method | Mechanical | Morphological | Thermal | Molecular | Reference |
|---|---|---|---|---|---|
| E-Beam | Increased elastic modulus at moderate-high doses; reduced flexibility | Chain scission reduces crystallinity; crosslinking increases rigidity | Potential increase in solubility and density | Increased modifiability; oxidative degradation; WVP increases | [115] |
| Gamma | Sometimes improves mechanical performance without sacrificing elongation | Backbone cleavage; enhanced gelling behavior in some polymers | Effects vary by dose and polymer | Reduced chain scission at sterilization doses crosslinking observed | [115] |
| X-Ray | Undocumented but likely less severe than gamma due to shorter exposure | Effects inferred to be similar to gamma radiation | Not well characterized | Expected ionizing radiation effects more research required | [116] |
| UV | Mechanical improvement with photo-crosslinking agents | Surface degradation in sensitive materials | Not widely observed due to surface-limited effect | Limited degradation in UV-resistant polymers like amylopectin | [117] |
| Autoclaving | Generally negative; reduced mechanical integrity | Complete loss of crystallinity in starches; hydrolytic breakdown | Thermally induced degradation in hydrophilic polymers | Glycosidic bond cleavage; sugar leeching; Maillard reactions | [118] |
| Dry Heat | Not well documented; likely poor due to thermal sensitivity | Loss of crystallinity varies without humidity | Distinct from autoclave; less hydrolysis | Thermal dehydration of plasticizers, acrolein formation possible | [119] |
| EtO | Slight increase in mechanical strength; increased brittleness | Unclear due to limited data | Minimal thermal impact | Trace residues require long aeration slow desorption | [120] |
| Ozone | Mechanical improvement in some fibers like PHB | Reduction in crystallinity; minimal impact on PLGA | Alters thermal behaviour in degraded fibers | Chain scission; aldehyde and carboxylic acid formation | [121] |
| ScCO2 | Maintains mechanical performance | Minimal structural degradation | Operates at low temperature; thermally gentle | Negligible chemical changes unless oxidative additives are used | [122] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




