Submitted:
07 July 2025
Posted:
08 July 2025
You are already at the latest version
Abstract
Keywords:
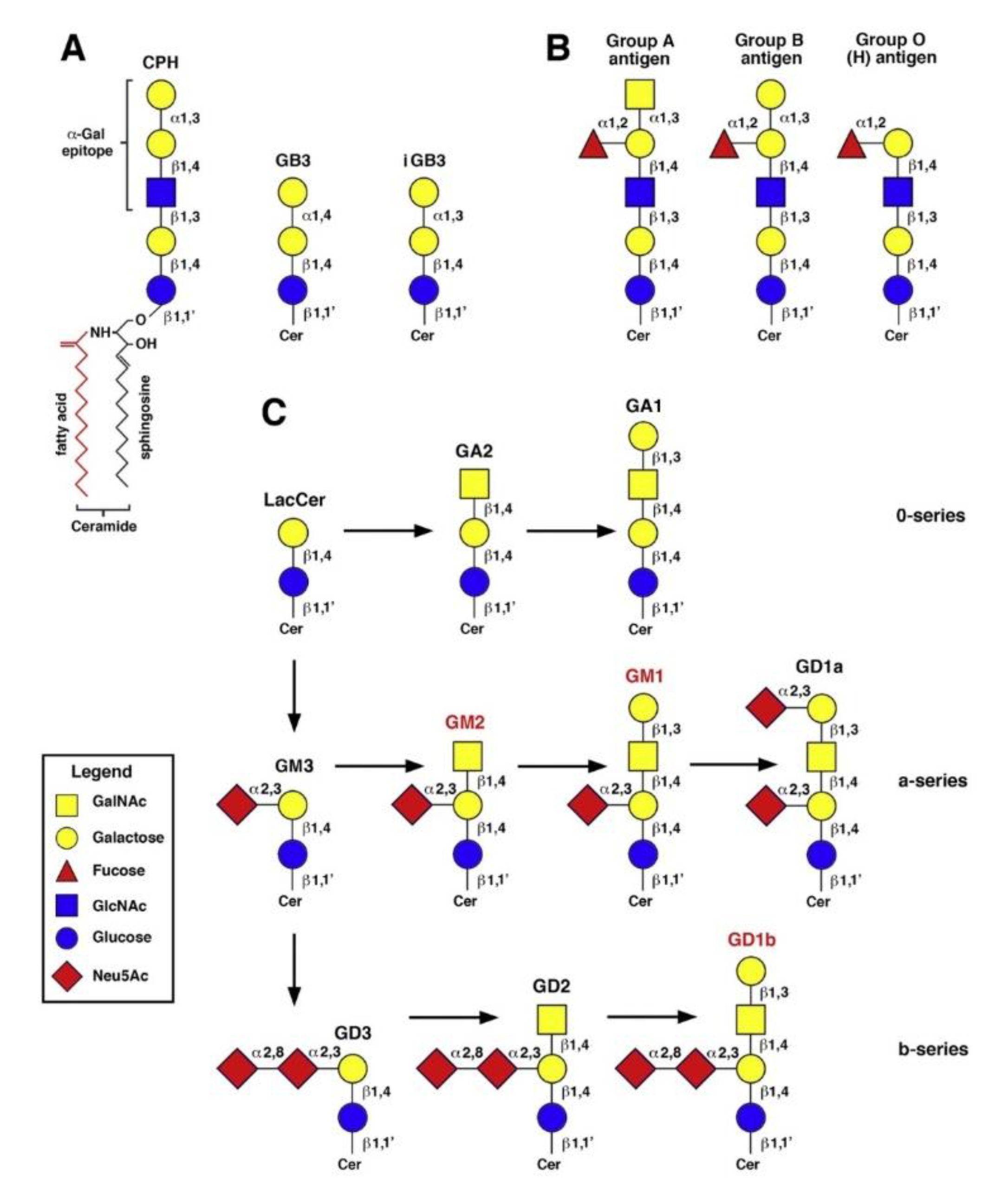
1. Introduction
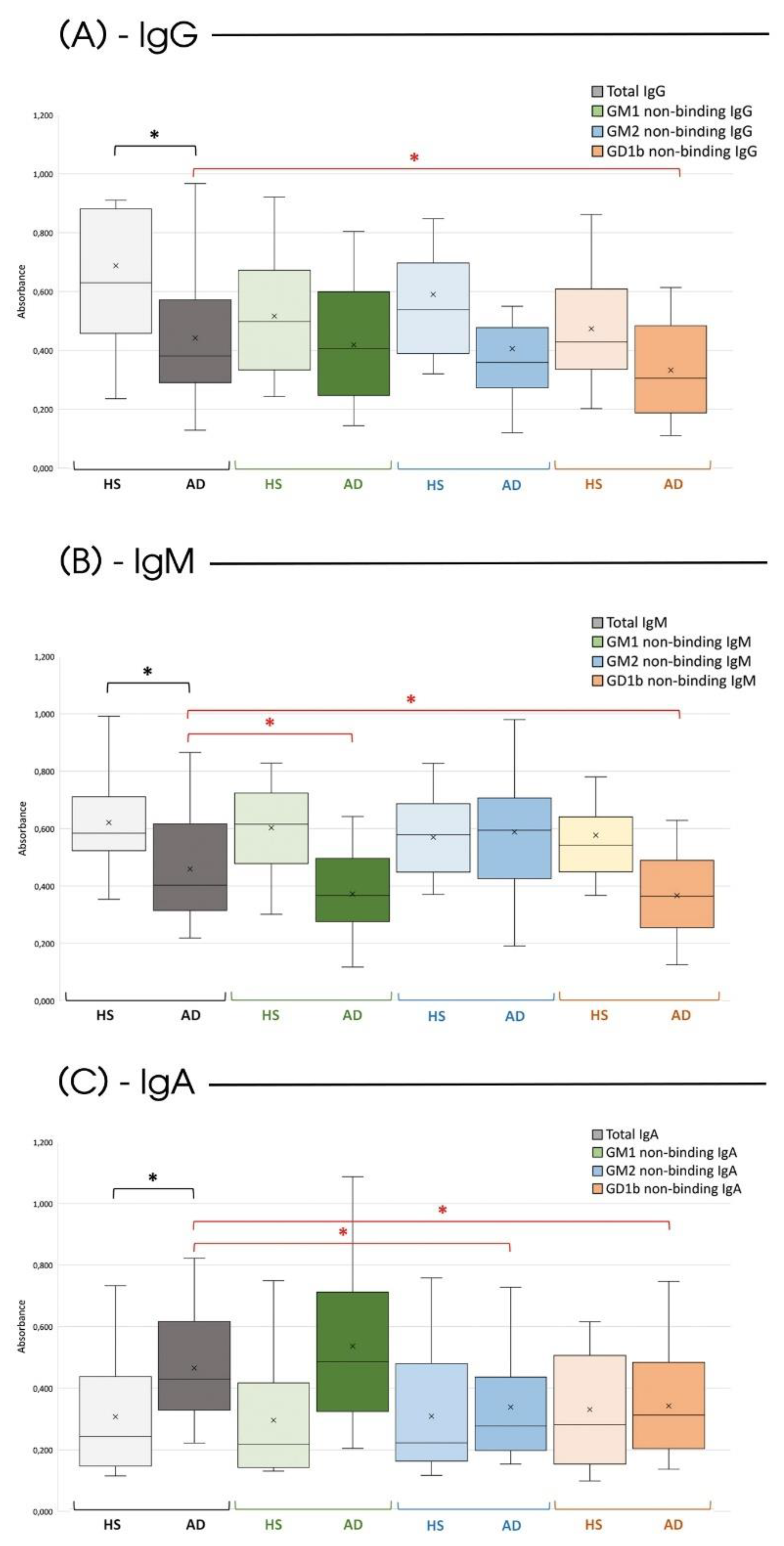
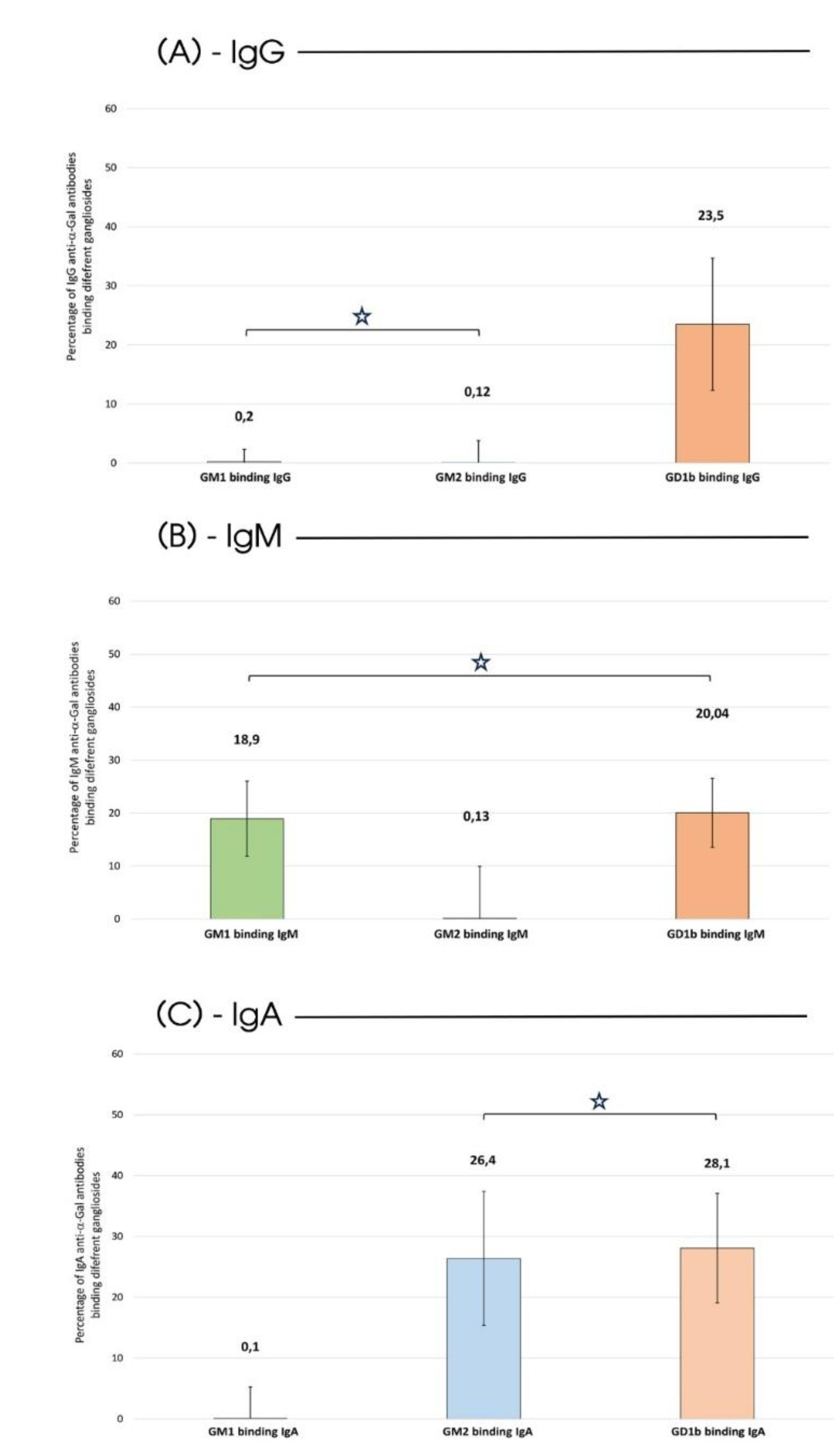
2. Results
3. Discussion
4. Materials and Methods
4.1. Patients and healthy controls
4.2. Blood collection and processing
4.3. Statistical analysis
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abs | Antibodies |
| AD | Alzheimer’s Disease |
| CNS | Central Nervous System |
| BBB | Blood-Brain Barrier |
| CSF | Cerebrospinal fluid |
| GBS | Guillan Barré Syndrome |
| GLS | Ganglioside |
| HS | Healthy Subjects |
| CerMA | Centre for Research and Training in Medicine of Aging |
| NIA-AA | National Institute of Aging and Alzheimer’s Association |
| MMSE | Mini-Mental State Examination |
| CDR | Clinical Dementia Rating |
| PBS | Phosphate-Buffered Saline |
| ELISA | Enzyme Linked Immunosorbent Assay |
| HSA | Human Serum Albumine |
| BMI | Body Mass Index |
| TIA | Transient Ischemic Attack |
References
- Hardy, J.; Selkoe, D.J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 2002, 297(5580), 353–356. [Google Scholar] [CrossRef] [PubMed]
- González-Sanmiguel, J.; Schuh, C.M.A.P.; Muñoz-Montesino, C.; Contreras-Kallens, P.; Aguayo, L.G.; Aguayo, S. Complex Interaction between Resident Microbiota and Misfolded Proteins: Role in Neuroinflammation and Neurodegeneration. Cells 2020, 9(11), 2476. [Google Scholar] [CrossRef]
- McGeer, PL.; McGeer, E.G. The amyloid cascade-inflammatory hypothesis of Alzheimer disease: implications for therapy. Acta Neuropathol 2013, 126(4), 479–497. [Google Scholar] [CrossRef]
- Pul, R.; Dodel, R.; Stangel, M. Antibody-based therapy in Alzheimer's disease. Expert Opin Biol Ther 2011, 11(3), 343–357. [Google Scholar] [CrossRef] [PubMed]
- Macher, B.A.; Galili, U. The Galalpha1,3Galbeta1,4GlcNAc-R (alpha-Gal) epitope: a carbohydrate of unique evolution and clinical relevance. Biochim Biophys Acta 2008, 1780(2), 75–88. [Google Scholar] [CrossRef]
- Galili, U. The alpha-gal epitope and the anti-Gal antibody in xenotransplantation and in cancer immunotherapy. Immunol Cell Biol 2005, 83(6), 674–686. [Google Scholar] [CrossRef]
- Galili, U. Acceleration of wound healing by α-gal nanoparticles interacting with the natural anti-Gal antibody. J Immunol Res 2015, 2015, 589648. [Google Scholar] [CrossRef] [PubMed]
- Galili, U.; Anaraki, F.; Thall, A.; Hill-Black, C.; Radic, M. One percent of human circulating B lymphocytes are capable of producing the natural anti-Gal antibody. Blood 1993, 82(8), 2485–2493. [Google Scholar] [CrossRef]
- Jaison, P.L.; Kannan, V.M.; Geetha, M.; Appukuttan, P.S. Epitopes recognized by serum anti-α-galactoside antibody are present on brain glycoproteins in man. J Biosci 1993, 18, 187–193. [Google Scholar] [CrossRef]
- Bernth Jensen, J.M.; Skeldal, S.; Petersen, M.S.; Bjarne, K.M.; Steen, H.; Jens, C.J.; Uffe, B.S.S.; Steffen, T. The human natural anti-αGal antibody targets common pathogens by broad-spectrum polyreactivity. Immunology 2021, 162(4), 434–451. [Google Scholar] [CrossRef]
- Angiolillo, A.; Gandaglia, A.; Arcaro, A.; Carpi, A.; Gentile, F.; Naso, F.; Di Costanzo, A. Altered Blood Levels of Anti-Gal Antibodies in Alzheimer's Disease: A New Clue to Pathogenesis? Life (Basel) 2021, 11(6), 538. [Google Scholar] [CrossRef] [PubMed]
- Usuki, S.; O'Brien, D.; Rivner, M.H.; Yu, R.K. A new approach to ELISA-based anti-glycolipid antibody evaluation of highly adhesive serum samples. J Immunol Methods 2014, 408, 52–63. [Google Scholar] [CrossRef]
- Merrill, A.H. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem Rev 2011, 111(10), 6387–6422. [Google Scholar] [CrossRef]
- Simons, K.; Gerl, MJ. Revitalizing membrane rafts: new tools and insights. Nat Rev Mol Cell Biol 2010, 11(10), 688–699. [Google Scholar] [CrossRef] [PubMed]
- Mandal, P.K.; Pettegrew, J.W. Alzheimer's disease: NMR studies of asialo (GM1) and trisialo (GT1b) ganglioside interactions with Abeta(1-40) peptide in a membrane mimic environment. Neurochem Res 2004, 29(2), 447–453. [Google Scholar] [CrossRef]
- Sipione, S.; Monyror, J.; Galleguillos, D.; Steinberg, N.; Kadam, V. Gangliosides in the Brain: Physiology, Pathophysiology and Therapeutic Applications. Front Neurosci 2020, 14, 572965. [Google Scholar] [CrossRef] [PubMed]
- McGonigal, R.; Barrie, J.A.; Yao, D.; Black, L.E.; McLaughlin, M.; Willison, H.J. Neuronally expressed a-series gangliosides are sufficient to prevent the lethal age-dependent phenotype in GM3-only expressing mice. J Neurochem 2021, 158(2), 217–232. [Google Scholar] [CrossRef]
- Sarbu, M.; Ica, R.; Zamfir, A.D. Gangliosides as Biomarkers of Human Brain Diseases: Trends in Discovery and Characterization by High-Performance Mass Spectrometry. Int J Mol Sci 2022, 23(2), 693. [Google Scholar] [CrossRef]
- Di Scala, C.; Yahi, N.; Flores, A.; Boutemeur, S.; Kourdougli, N.; Chahinian, H.; Fantini, J. Broad neutralization of calcium-permeable amyloid pore channels with a chimeric Alzheimer/Parkinson peptide targeting brain gangliosides. Biochim Biophys Acta 2016, 1862(2), 213–222. [Google Scholar] [CrossRef]
- Ariga, T.; McDonald, M.P.; Yu, R.K. Role of ganglioside metabolism in the pathogenesis of Alzheimer's disease--a review. J Lipid Res 2008, 49(6), 1157–1175. [Google Scholar] [CrossRef]
- Kracun, I.; Kalanj, S.; Talan-Hranilovic, J.; Cosovic, C. Cortical distribution of gangliosides in Alzheimer's disease. Neurochem Int 1992, 20(3), 433–438. [Google Scholar] [CrossRef] [PubMed]
- Pernber, Z.; Blennow, K.; Bogdanovic, N.; Månsson, J.E.; Blomqvist, M. Altered distribution of the gangliosides GM1 and GM2 in Alzheimer's disease. Dement Geriatr Cogn Disord 2012, 33(2-3), 174-188.
- Chapman, J.; Sela, B.A.; Wertman, E.; Michaelson, D.M. Antibodies to ganglioside GM1 in patients with Alzheimer's disease. Neurosci Lett 1988, 86(2), 235–240. [Google Scholar] [CrossRef]
- Hatzifilippou, E.; Koutsouraki, E.; Banaki, T.; Traka, M.; Costa, V.G.; Baloyannis, S.J. Antibodies against GM1 in demented patients. Am J Alzheimers Dis Other Demen 2008, 23(3), 274–279. [Google Scholar] [CrossRef]
- Lehmann, H.C.; Lopez, P.H.; Zhang, G.; Ngyuen, T.; Zhang, J.; Kieseier, B.C.; Mori, S.; Sheikh, K.A. Passive immunization with anti-ganglioside antibodies directly inhibits axon regeneration in an animal model. J Neurosci 2007, 27(1), 27–34. [Google Scholar] [CrossRef]
- Rodríguez, Y.; Rojas, M.; Pacheco, Y.; Acosta-Ampudia, Y.; Ramírez-Santana, C.; Monsalve, D.M.; Gershwin, E.C.; Anaya, J.M. Guillain-Barré syndrome, transverse myelitis and infectious diseases. Cell Mol Immunol 2018, 15(6), 547–562. [Google Scholar] [CrossRef] [PubMed]
- Yuki, N.; Ariga, T. Antibodies to fucogangliosides in neurological diseases. J Neurol Sci 1997, 150(1), 81–84. [Google Scholar] [CrossRef]
- Galili, U.; Basbaum, C.B.; Shohet, S.B.; Buehler, J.; Macher, B.A. Identification of erythrocyte Gal alpha 1-3Gal glycosphingolipids with a mouse monoclonal antibody, Gal-13. J Biol Chem 1987, 262(10), 4683–4688. [Google Scholar] [CrossRef] [PubMed]
- Iweala, O.; Brennan, P.J.; Commins, S.P. Serum igE specific for alpha-gal sugar moiety can bind glycolipid. J Allergy Clin Immunol 2017, 139(2), AB88. [Google Scholar] [CrossRef]
- Yanagisawa, K.; Odaka, A.; Suzuki, N.; Ihara, Y. GM1 ganglioside-bound amyloid beta-protein (A beta): a possible form of preamyloid in Alzheimer's disease. Nat Med 1995, 1(10), 1062–1066. [Google Scholar] [CrossRef]
- Choo-Smith, L.P.; Garzon-Rodriguez, W.; Glabe, C.G.; Surewicz, W.K. Acceleration of amyloid fibril formation by specific binding of Abeta-(1-40) peptide to ganglioside-containing membrane vesicles. J Biol Chem 1997, 272(37), 22987–22990. [Google Scholar] [CrossRef]
- Matsuzaki, K.; Horikiri, C. Interactions of amyloid beta-peptide (1-40) with ganglioside-containing membranes. Biochemistry 1999, 38(13), 4137–4142. [Google Scholar] [CrossRef]
- Kakio, A.; Nishimoto, S.I.; Yanagisawa, K.; Kozutsumi, Y.; Matsuzaki, K. Cholesterol-dependent formation of GM1 ganglioside-bound amyloid beta-protein, an endogenous seed for Alzheimer amyloid. J Biol Chem 2001, 276(27), 24985–24990. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.P.; Suzuki, Y.; Bourne, N.T.; Asakura, T. Binding of amyloid beta-peptide to ganglioside micelles is dependent on histidine-13. Biochem J 2006, 397(3), 483–490. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K. Aβ-ganglioside interactions in the pathogenesis of Alzheimer's disease. Biochim Biophys Acta Biomembr 2020, 1862(8), 183233. [Google Scholar] [CrossRef]
- Ariga, T.; Kobayashi, K.; Hasegawa, A.; Kiso, M.; Ishida, H.; Miyatake, T. Characterization of high-affinity binding between gangliosides and amyloid beta-protein. Arch Biochem Biophys 2001, 388(2), 225–230. [Google Scholar] [CrossRef]
- Nishi, K.; Tanegashima, A.; Yamamoto, Y. ; Ushiyama, I:; Ikemoto, K.; Yamasaki, S; Nishimura, A.; Rand, S.; Brinkmann, B. Utilization of lectin-histochemistry in forensic neuropathology: lectin staining provides useful information for postmortem diagnosis in forensic neuropathology. Leg Med (Tokyo) 2003, 5(3), 117-131.
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Clifford, R.J.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement 2011, 7(3), 263–269. [Google Scholar] [CrossRef]
- Burk, C.M.; Beitia, R.; Lund, P.K.; Dellon, E.S. High rate of galactose-alpha-1,3-galactose sensitization in both eosinophilic esophagitis and patients undergoing upper endoscopy. Dis Esophagus 2016, 29(6), 558–562. [Google Scholar] [CrossRef]
- Safaie, P.; Ham, M.; Kuang, P.; Mehta, A.S; Wang, M.; Cheifetz, A.S.; Robson, S.; Lau, D.; Block, T.M.; Moss, A.C. Lectin-reactive anti-α-gal in patients with Crohn's disease: correlation with clinical phenotypes. Inflamm Bowel Dis 2013, 19(13), 2796–2800. [Google Scholar] [CrossRef] [PubMed]
- Chinuki, Y.; Morita, E. Alpha-Gal-containing biologics and anaphylaxis. Allergol Int 2019, 68(3), 296–300. [Google Scholar] [CrossRef]
- Naso, F.; Stefanelli, U.; Buratto, E.; Lazzari, G.; Perota, A. ; Galli, C; Gandaglia, A. Alpha Gal Inactivated Heart Valve Bioprostheses Exhibit an Anti-Calcification Propensity Similar to Knockout Tissues. Tissue Eng Part A 2017, 23(19-20), 1181-1195.
- Wilson, J.M.; Erickson, L.; Levin, M.; Ailsworth, S.M.; Commins, S.P.; Platts-Mills, T.A.E. Tick bites, IgE to galactose-alpha-1,3-galactose and urticarial or anaphylactic reactions to mammalian meat: The alpha-gal syndrome. Allergy 2024, 9(6), 1440–1454. [Google Scholar] [CrossRef]
- Dhana, K.; Beck, T.; Desai, P.; Wilson, R.S.; Evans, D.A.; Rajan, K.B. Prevalence of Alzheimer's disease dementia in the 50 US states and 3142 counties: A population estimate using the 2020 bridged-race postcensal from the National Center for Health Statistics. Alzheimer's Dement 2023, 19(10), 4388-4395.
- Bianchi, J.; Walters, A.; Fitch, Z.W.; Turek, J.W. Alpha-gal syndrome: Implications for cardiovascular disease. Glob. cardiol. Sci 2019, 3. [Google Scholar] [CrossRef]
- Qi, C.; Liu, F.; Zhang, W.; Han, Y.; Zhang, N.; Liu, Q.; Li, H. Alzheimer's disease alters the transcriptomic profile of natural killer cells at single-cell resolution. Front Immunol 2022, 13, 1004885. [Google Scholar] [CrossRef] [PubMed]
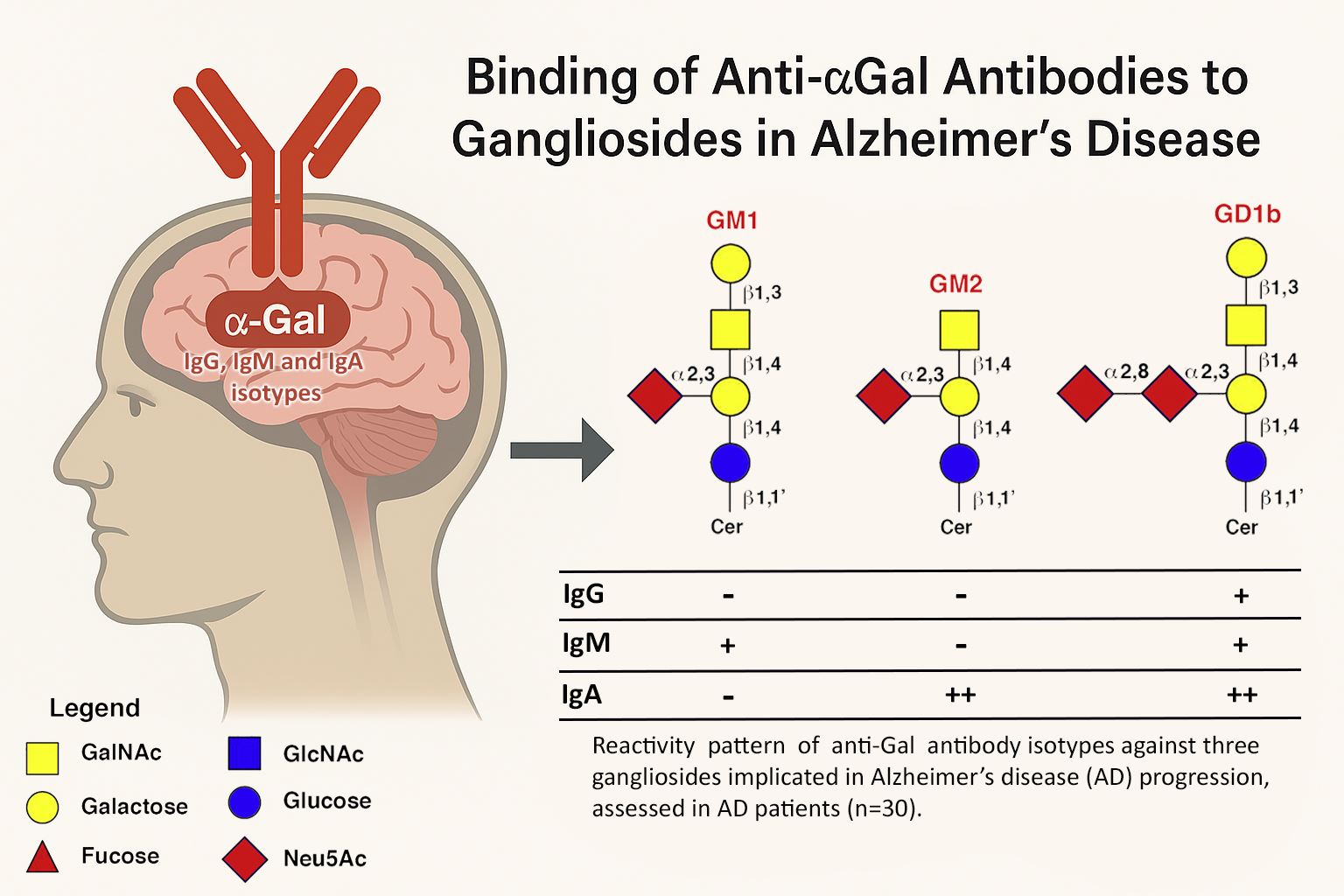
| GM1 | GM2 | GD1b | ||
|---|---|---|---|---|
|
HS |
IgG | - | - | - |
| IgM | - | - | - | |
| IgA | - | - | - | |
|
AD |
IgG | - | - | + |
| IM | + | - | + | |
| IgA | - | + + | + + |
| Characteristics | AD (N. 30) |
HS (N. 30) |
F (1,59)/X2 † |
P |
|---|---|---|---|---|
| Females/males (N.) | 19/11 | 15/15 | 1.086 | 0.297 |
| Age (mean ± SD, y) (range, y) |
83.77 ± 5.89 (70-96) |
80.83 ± 6.04 (70-93) |
3.631 | 0.062 |
| Education level (mean + SD, y) | 9.10 ± 5.27 | 11.77 ± 4.19 | 4.708 | 0.034 |
| BMI (mean + SD, kg/m2) | 24.37 ± 4.56 | 26.37±3.70 | 3.475 | 0.067 |
| MMSE (score) | 17.88 ± 6.85 | 30.07±1.31 | 91.632 | <0.001 |
| Blood group (N; %) | ||||
| 0 | 13; 43.3% | 14; 46.6% | 0.067 | 0.795 |
| A | 13; 43.3% | 8; 26.6% | 1.832 | 0.176 |
| B | 3; 10% | 6; 20% | 1.176 | 0.278 |
| AB | 1; 3.3% | 2; 6.6% | 0.351 | 0.554 |
| Medical History (N; %) | ||||
| Smoke* | 3; 10% | 3; 10% | 0.000 | 1.000 |
| Dyslipidemia | 11; 36.6% | 12; 40% | 0.071 | 0.791 |
| Diabetes | 8; 26.6% | 6; 20% | 0.373 | 0.542 |
| Hypertension | 17; 56.6% | 19; 63.3 % | 0.278 | 0.598 |
| Myocardial infarction | 3; 10% | 3; 10% | 0.000 | 1.000 |
| TIA/Stroke | 3; 10% | 1; 3.33% | 1.071 | 0.301 |
| Drugs (N; %) | ||||
| Antihypertensive | 17; 56.6% | 18; 60% | 0.069 | 0.793 |
| Lipid-lowering | 10; 30% | 11; 36.6% | 0.073 | 0.787 |
| Hypoglycemic | 8; 26.6% | 6; 20% | 0.373 | 0.542 |
| Antiacid | 12; 40% | 11; 36.6% | 0.071 | 0.791 |
| Antiplatelet | 13; 43.3% | 12; 40% | 0.069 | 0.793 |
| Anti-inflammatory | 3; 10% | 3; 10% | 0.000 | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




