Submitted:
05 July 2025
Posted:
07 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Pregelatinized Cornstarch [9]
2.2.2. Preparation of Phosphate Pregelatinized Cornstarch [10]
2.2.3. Characterizations of Phosphate Pregelatinized Cornstarch
2.2.4. Paracetamol Characterizations
2.2.5. Tablet Formulation and Characterization
| Materials | F0 | F1 | F2 | F3 | F4 | F5 |
| Paracetamol (mg) | 500 | 500 | 500 | 500 | 500 | 500 |
| Corn starch (%) | 10 | 10 | 10 | 10 | 10 | 10 |
| Pregelatinized corn starch (%) | 7 | - | - | - | - | - |
| Phosphate pregelatinized corn starch (%) | - | 3 | 4 | 5 | 6 | 7 |
| Primogel (%) | 3 | 3 | 3 | 3 | 3 | 3 |
| Magnesium strearate (%) | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Talc (%) | 2 | 2 | 2 | 2 | 2 | 2 |
| Lactose (added to a final weight of 700 mg) | 700 | 700 | 700 | 700 | 700 | 700 |
3. Results and Discussion
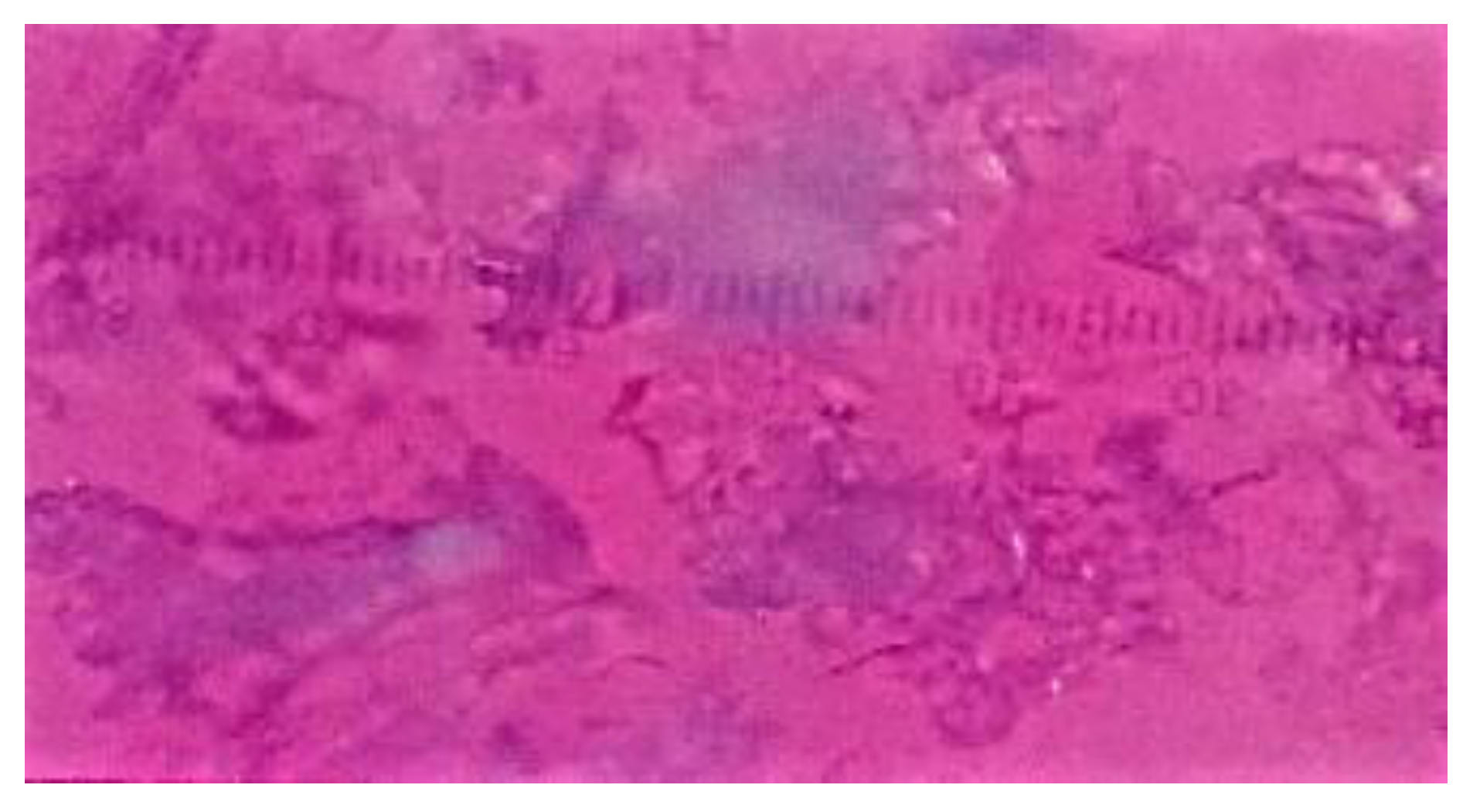
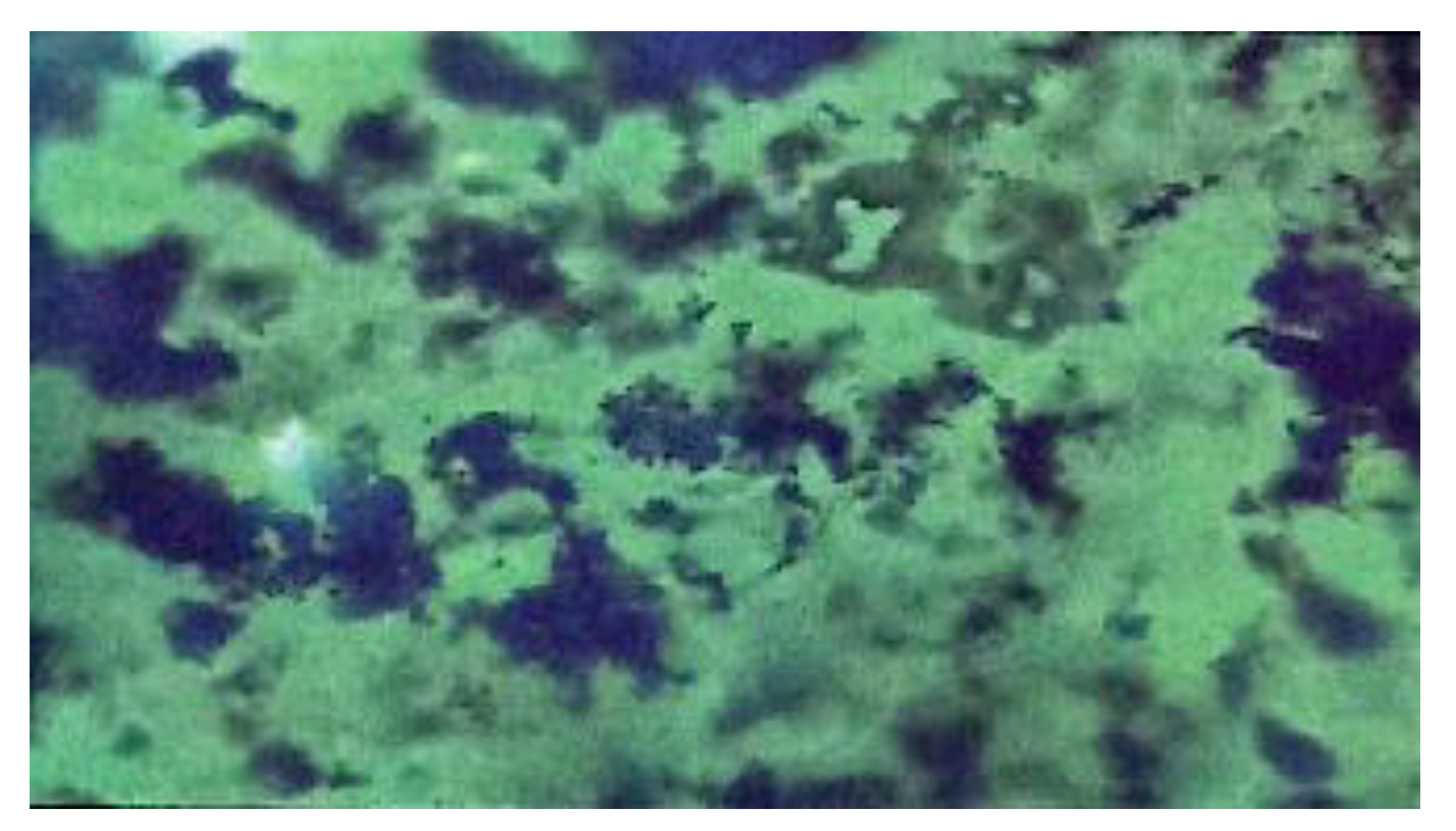
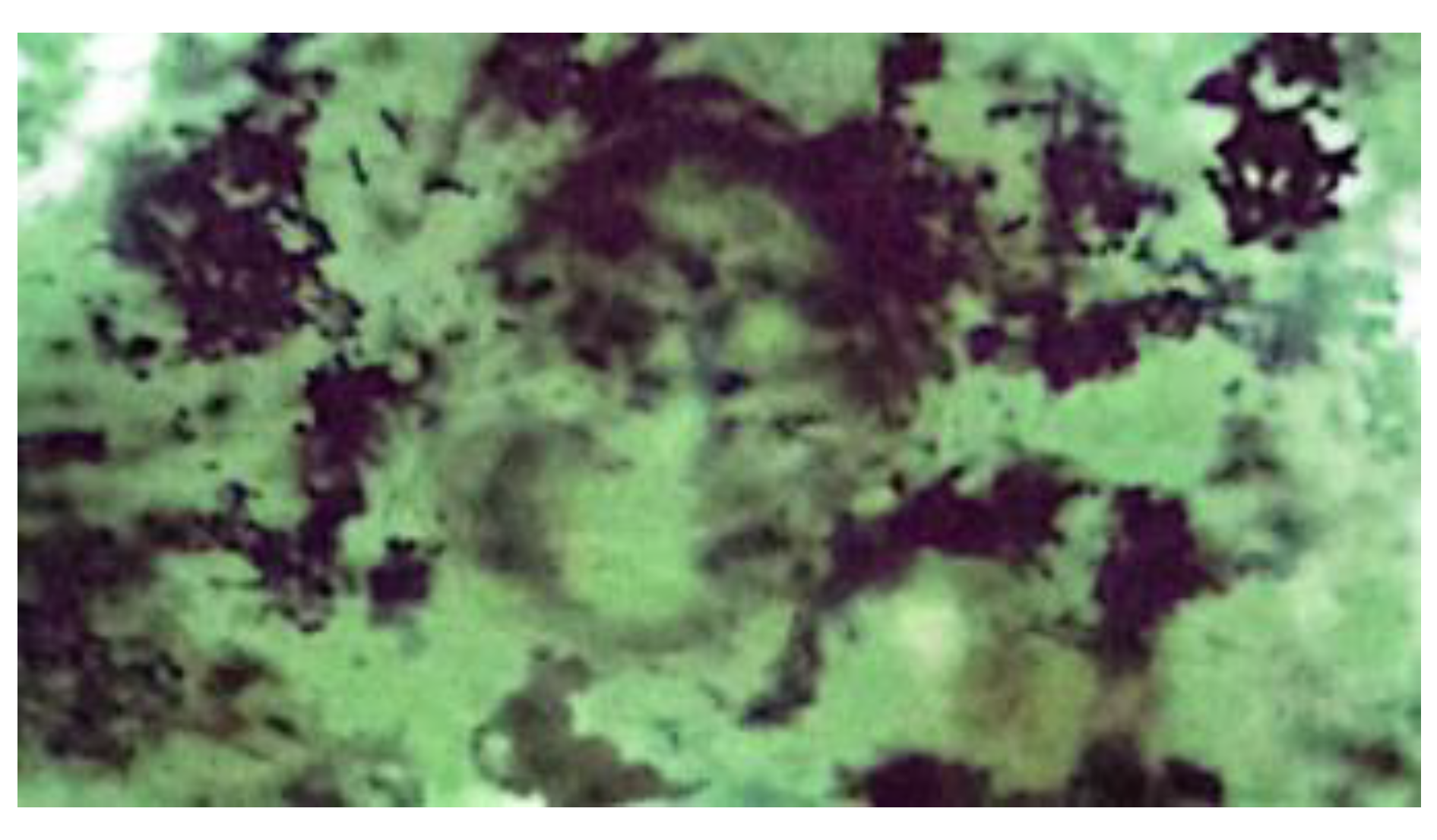
3.1. Characterization of Cornstarch, Pregelatinized Cornstarch, and Phosphate Pregelatinized Cornstarch
| Parameters | Corn starch |
Pregelatinized Corn starch |
Phosphate pregelatinized corn starch | ||
| 0.25%* | 0.30% | 0.35% | |||
| Viscosity (cP) | - | 72.9+0.45 | 37.7+0.75 | 45.5+0.30 | 34.3+0.55 |
| pH | 6.3+0.05 | 6.7+0.05 | 6.9+0.01 | 7.1+0.07 | 7.2+0.1 |
| Whiteness level (%) | 97.27 | 89.36 | 98.82 | 99.05 | 99.75 |
| Identification color | Purple | Dark blue | Dark blue | Dark blue | Dark blue |
| Loss on drying (%) | 10.56+0.06 | 5.59+0.30 | 6.19+0.05 | 6.16+0.06 | 6.19+0.01 |
| Flow rate (g/s) | 0.51+0.04 | 0.85+0.04 | 1.04+0.03 | 1.25+0.10 | 1.41+0.11 |
| Repose angle (o) | 37.75+0.45 | 24.94+0.42 | 23.37+0.59 | 21.70+0.10 | 20.89+0.72 |
| Compressibility (%) | 20.32+0.32 | 15.41+0.55 | 13.93+0.24 | 12.93+0.27 | 11.33+1.02 |
3.2. Characterization of Paracetamol
| Parameters | Results |
| Flow rate (g/s) | - |
| Repose angle (o) | - |
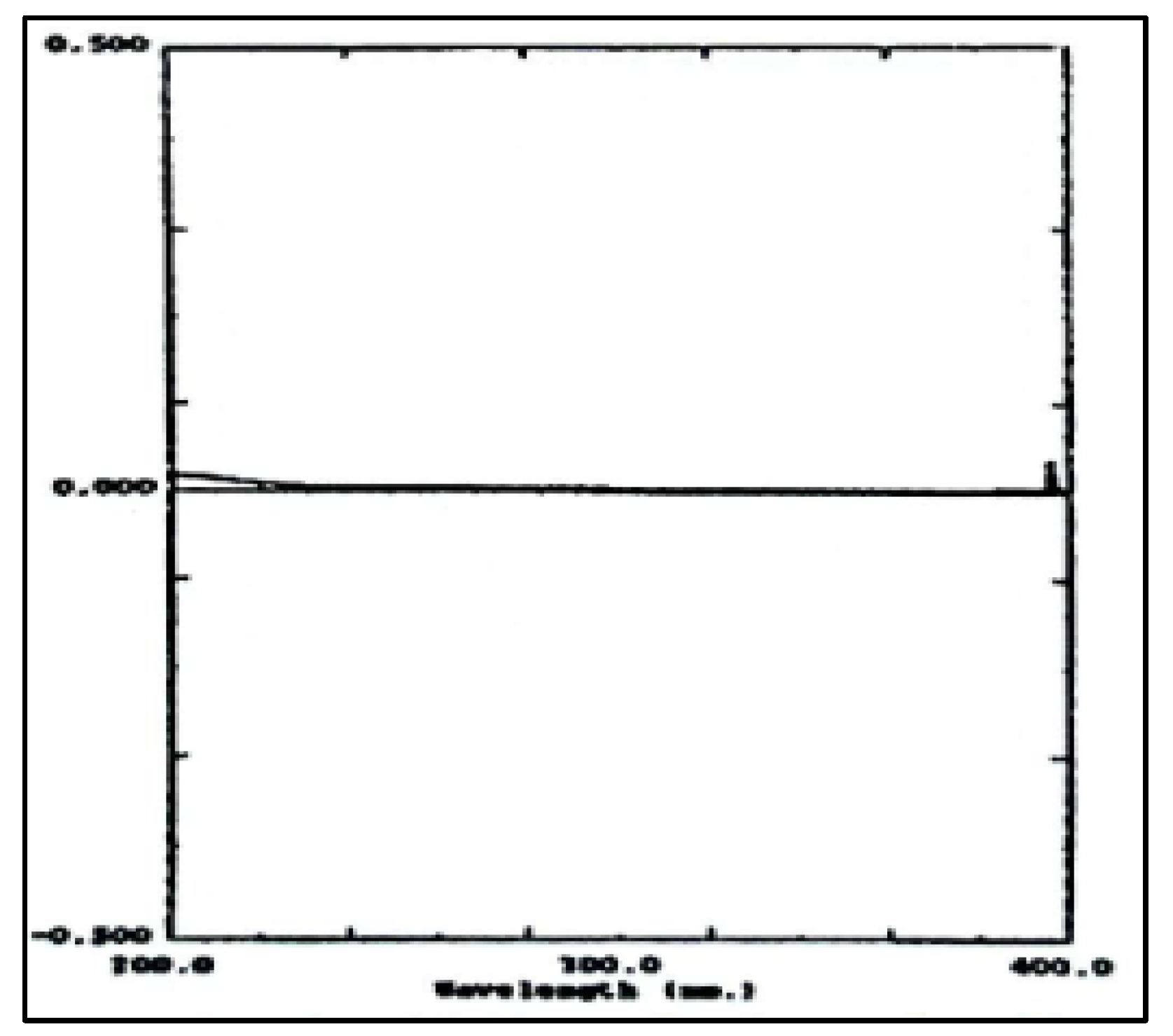
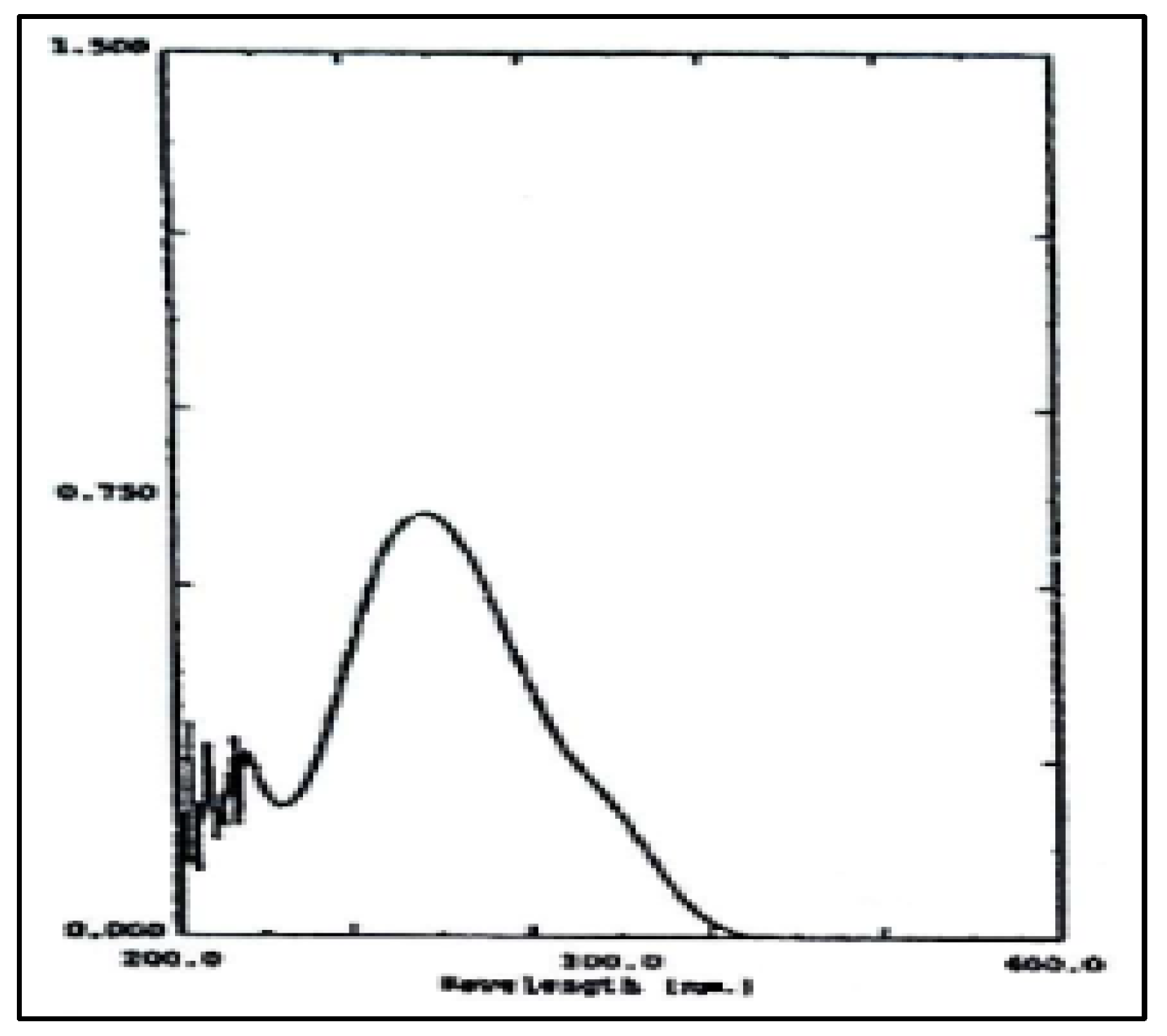
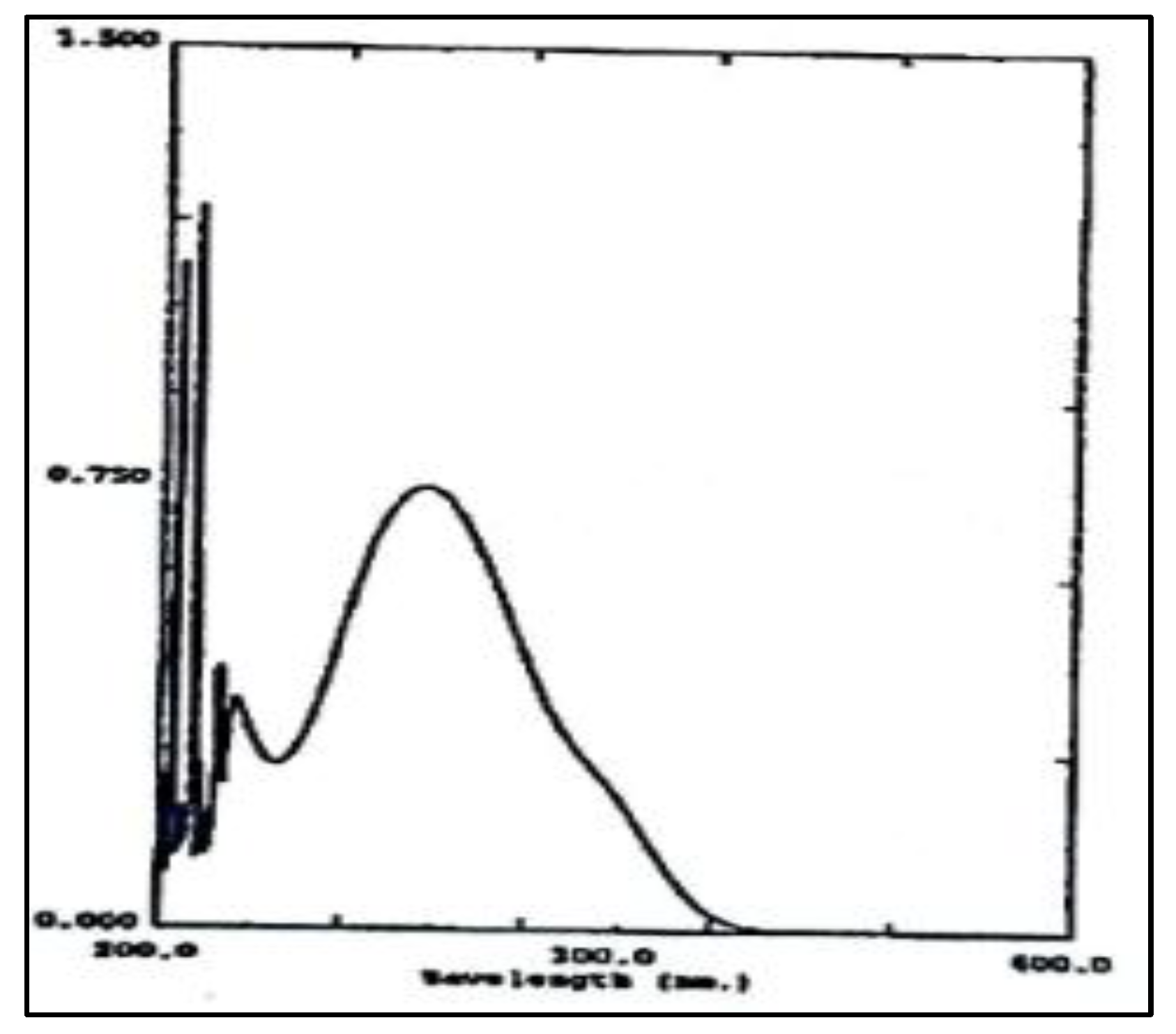
| Absorbance peak (nm) | NaOH 0.1N at 257.20 nm Phosphate buffer at pH 5.8 at 243.60 nm |
| Organoleptic | White powder, with no odor and bitter taste |
| Compressibility (%) | 23.59+0.42 |
| Solubility | Soluble in boil water, NaOH 1N, and ethanol |
3.3. Characterization of Tablet Mass Before Compression
| Parameters | F0 | F1 | F2 | F3 | F4 | F5 | |
| LOD (%) | 3.52+0.02 | 3.56+0.02 | 3.55+0.02 | 3.61+0.01 | 3.54+0.01 | 3.52+0.01 | |
| F. rate (g/s) | 28.74+0.30 | 22.73+0.72 | 24.21+0.08 | 26.18+0.93 | 25.32+0.32 | 25.84+0.73 | |
| R. angle (o) | 22.56+0.24 | 25.54+1.51 | 24.49+0.36 | 23.99+0.29 | 23.68+0.13 | 23.15+0.28 | |
| Compress. (%) | 8.70+0.30 | 15.68+0.08 | 14.34+0.34 | 12.70+0.30 | 11.60+0.40 | 10.70+0.30 | |
| Con. hom. (%) | 99.47+0.21 | 101.98+0.52 | 99.18+0.08 | 99.05+0.039 | 101.92+0.08 | 101.14+0.17 | |
3.4. Particle Size Distribution (%)
| Mesh# | F0 | F1 | F2 | F3 | F4 | F5 | |
| 14 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 16 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 20 | 0.97+0.03 | 0.95+0.01 | 0.97+0.01 | 0.85+0.04 | 1.58+0.02 | 0.90+0.02 | |
| 30 | 59.29+0.31 | 37.07+0.28 | 36.36+0.46 | 27.90+0.40 | 65.05+0.32 | 22.51+0.31 | |
| 40 | 26.31+0.50 | 36.81+1.01 | 51.01+0.38 | 55.29+0.30 | 14.36+0.16 | 56.83+0.10 | |
| 50 | 10.97+0.69 | 20.90+0.09 | 6.11+2.03 | 12.17+0.44 | 15.04+0.35 | 17.12+0.71 | |
| Pan | 2.46+0.08 | 4.26+0.61 | 3.85+0.23 | 3.78+0.29 | 3.58+0.17 | 2.72+0.17 | |
3.5. Granule Friability (%)
| Mesh# | F0 | F1 | F2 | F3 | F4 | F5 | |
| 14 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 16 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 20 | 0.88+0.01 | 0.28+0.02 | 0.49+0.62 | 0.65+0.04 | 1.33+0.10 | 0.80+0.89 | |
| 30 | 58.48+0.44 | 12.38+1.07 | 21.80+0.80 | 27.90+0.40 | 65.05+0.32 | 22.51+0.31 | |
| 40 | 25.85+0.53 | 68.851+0.48 | 64.96+0.86 | 54.05+0.05 | 16.59+0.35 | 56.44+0.30 | |
| 50 | 12.56+0.90 | 18.58+0.34 | 7.67+0.24 | 15.13+0.50 | 14.68+0.40 | 17.99+0.76 | |
| Pan | 2.36+0.02 | 5.42+0.55 | 5.07+0.25 | 4.29+0.28 | 3.23+0.18 | 2.55+0.24 | |
3.6. Physical Data of Paracetamol Tablet Formulations
| Test type | F0 | F1 | F2 | F3 | F4 | F5 | |
| Organoleptic | White, no odor, Uhamka logo | White, no odor, Uhamka logo | White, no odor, Uhamka logo | White, no odor, Uhamka logo | White, no odor, Uhamka logo | White, no odor, Uhamka logo | |
| Diameter (cm) | 1.32+0.00 | 1.32+0.00 | 1.32+0.00 | 1.32+0.00 | 1.32+0.00 | 1.32+0.00 | |
| Thicknss (cm) | 0.50+0.01 | 0.50+0.00 | 0.50+0.00 | 0.50+0.00 | 0.50+0.00 | 0.50+0.00 | |
| Hardness (kg) | 5.57+0.08 | 5.05+0.03 | 5.28+0.04 | 5.60+0.06 | 5.64+0.02 | 5.80+0.06 | |
| Friability (%) | 0.34+0.05 | 1.61+0.03 | 1.31+0.07 | 0.97+0.02 | 0.75+0.06 | 0.50+0.03 | |
| Disint. time (s) | 79+0.01 | 72+0.03 | 76+0.01 | 63+0.03 | 74+0.04 | 82+0.01 | |
| W. vari.(mg) | 710.26+2.53 | 710.85+4.90 | 710.56+4.22 | 709.49+2.68 | 711.44+2.79 | 711.60+2.33 | |
| W. dev. (%) | 0.30+0.27 | 0.58+0.41 | 0.52+0.31 | 0.29+0.25 | 0.32+0.25 | 0.28+0.18 | |
3.7. Chemical Data of Paracetamol Tablet Formulations
| Test type | F0 | F1 | F2 | F3 | F4 | F5 | |
| Par. contn (%) | 99.85+0.14 | 102.92+0.06 | 98.84+0.28 | 99.27+0.17 | 101.71+0.19 | 101.86+0.08 | |
| C. hom. (%) | 99.88+0.12 | 102.91+0.13 | 98.84+0.04 | 99.27+0.10 | 101.75+0.11 | 101.80+0.03 | |
3.8. Dissolution Data of Paracetamol Tablet Formulations
| Time | Minute-3 | Minute-5 | Minute-7 | Minute-10 | Minute-20 | Minute-30 | Minute-45 |
|
Disso-lusion (%) |
F0 | ||||||
| 35.56+1.80 | 50.40+2.17 | 64.85+2.08 | 74.80+1.95 | 84.63+2.12 | 97.55+4,65 | 101.87+0.9 | |
| F1 | |||||||
| 99.13+0.84 | 100.80+0.69 | 101.34+0.78 | 101.68+0.82 | 102.06+0.79 | 102.41+0.83 | 102.79+0.9 | |
| F2 | |||||||
| 90.95+2.95 | 95.00+2.35 | 98.25+1.79 | 100.99+0.70 | 100.99+0.81 | 100.43+1.12 | 100.07+1.3 | |
| F3 | |||||||
| 55.90+6.14 | 73.32+4.99 | 86.48+4.00 | 94.02+2.19 | 101.43+0.83 | 100.89+0.95 | 100.58+1.2 | |
| F4 | |||||||
| 49.57+1.37 | 66.06+1.63 | 73.57+1.99 | 81.76+1.85 | 97.29+2.44 | 103.20+0.59 | 102.41+1.0 | |
| F5 | |||||||
| 43.10+2.74 | 57.98+5.83 | 69.34+2.83 | 77.29+4.88 | 86.95+2.74 | 98.90+2.47 | 103.99+0.6 | |
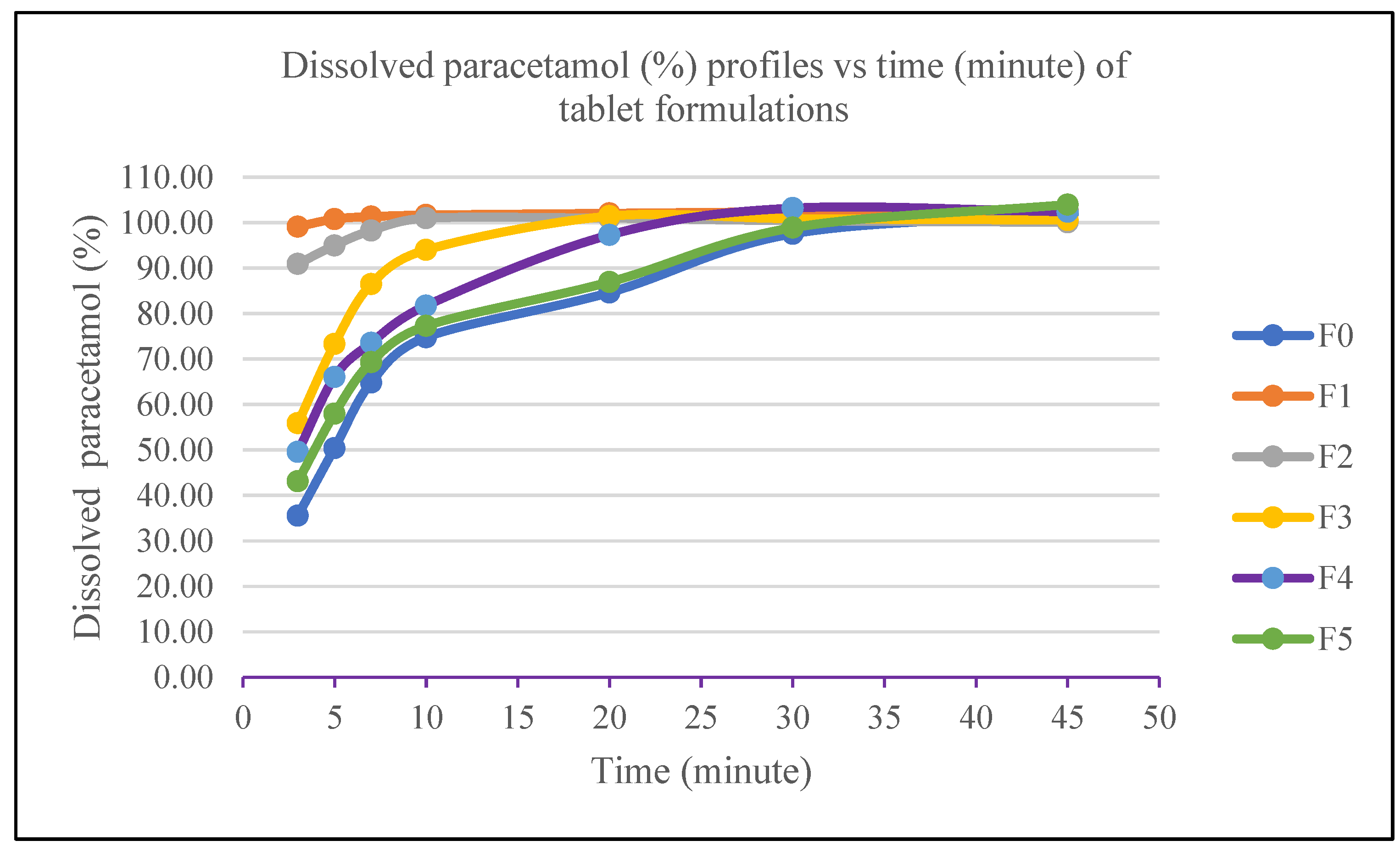
3.9. Dissolusion Efficiency (%)
| Parameters | Dissolusion efficiency (%) | ||||||
| F0 | F1 | F2 | F3 | F4 | F5 | ||
| Sample size | 18 | 18 | 18 | 18 | 18 | 18 | |
| Average | 78.79 | 95.88 | 95.01 | 90.53 | 86.09 | 80.89 | |
| SD | 0.67 | 0.19 | 0.47 | 0.79 | 0.70 | 1.26 | |
| RSD | 0.85 | 0.20 | 0.49 | 0.87 | 0.81 | 1.55 | |
3.10. Discussion
3.10.1. Preparation of Phosphate Pregelatinized Cornstarch
3.10.2. Characterization of Phosphate Pregelatinized Cornstarch
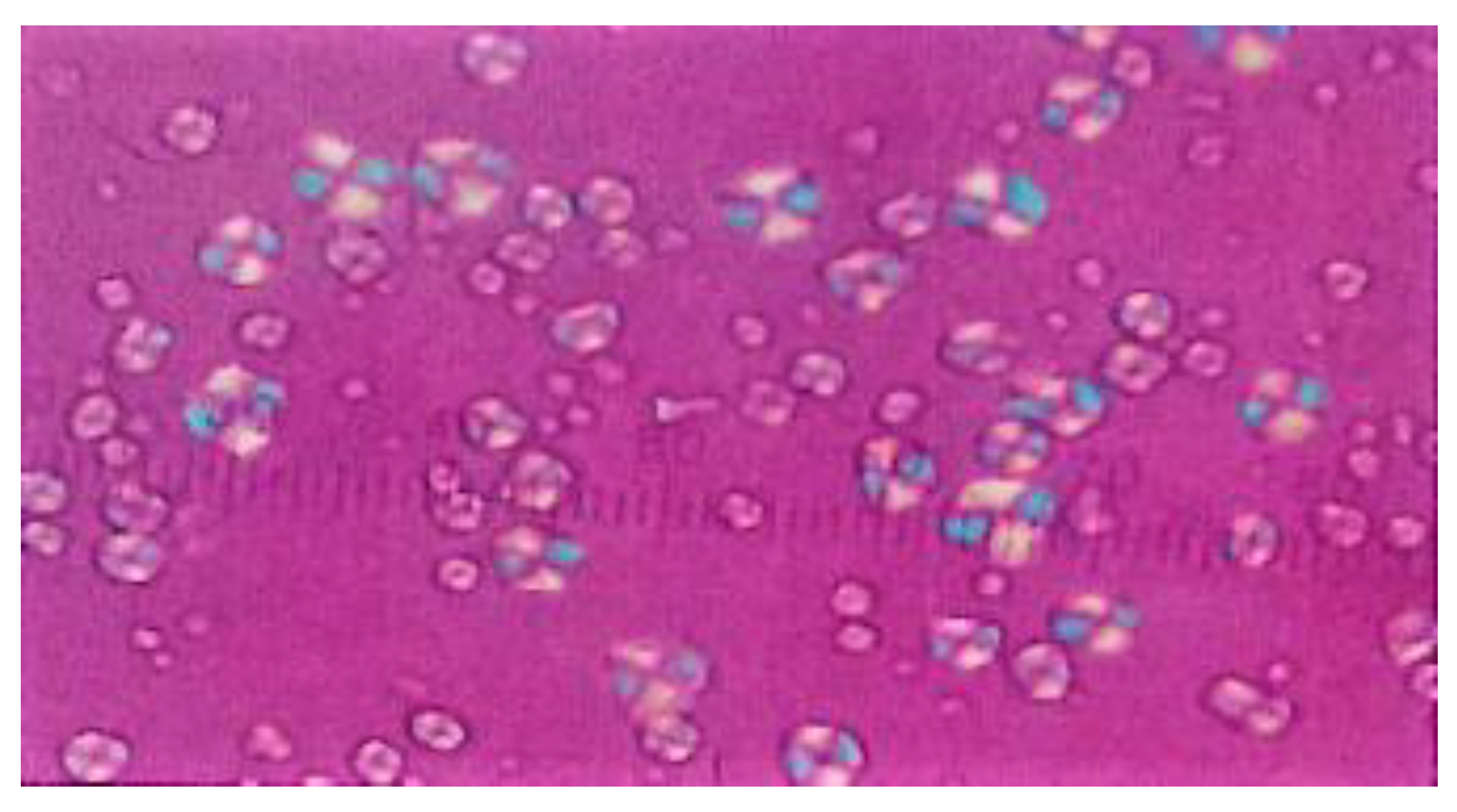
3.10.3. Examination of Paracetamol Raw Materials
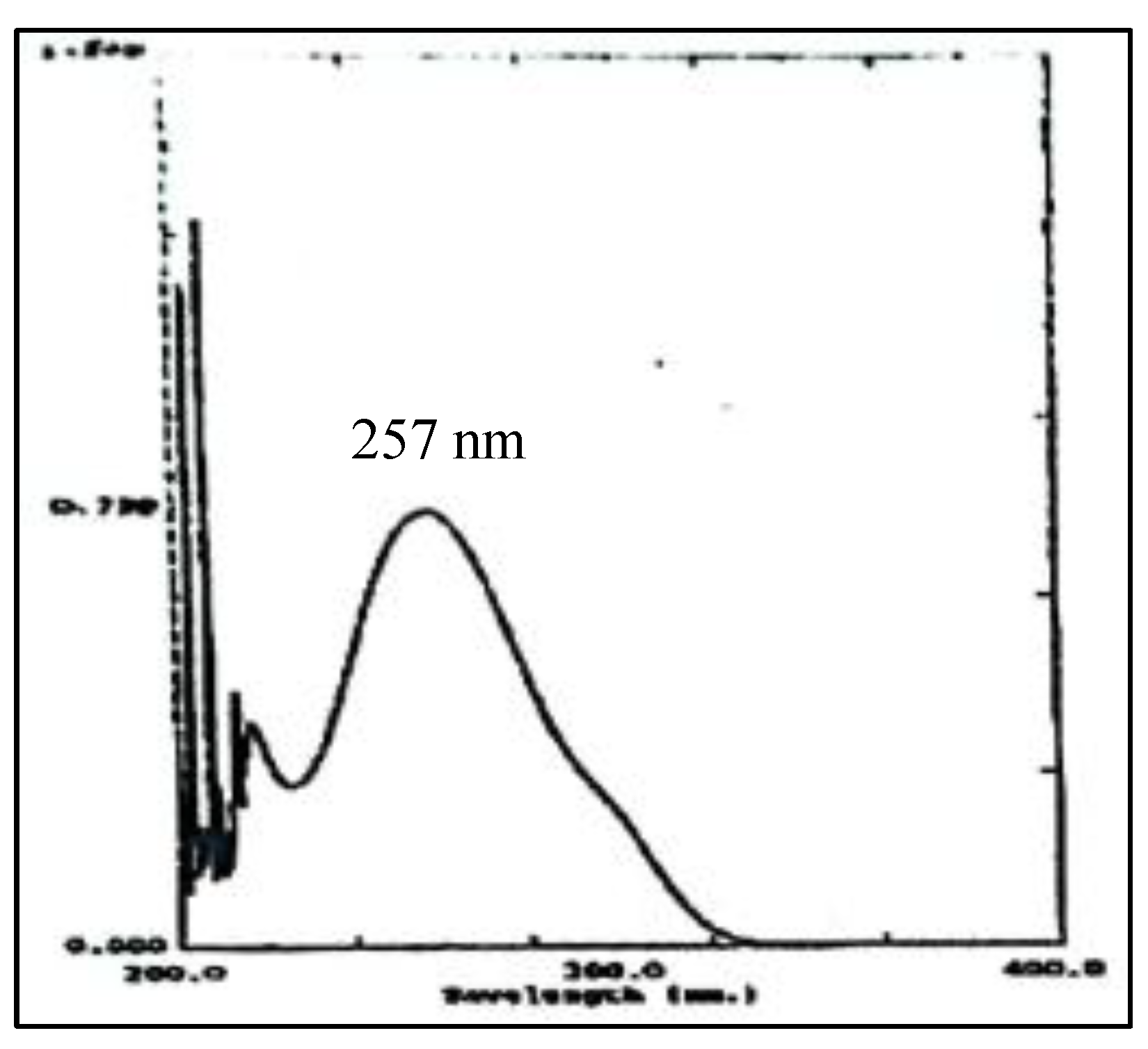
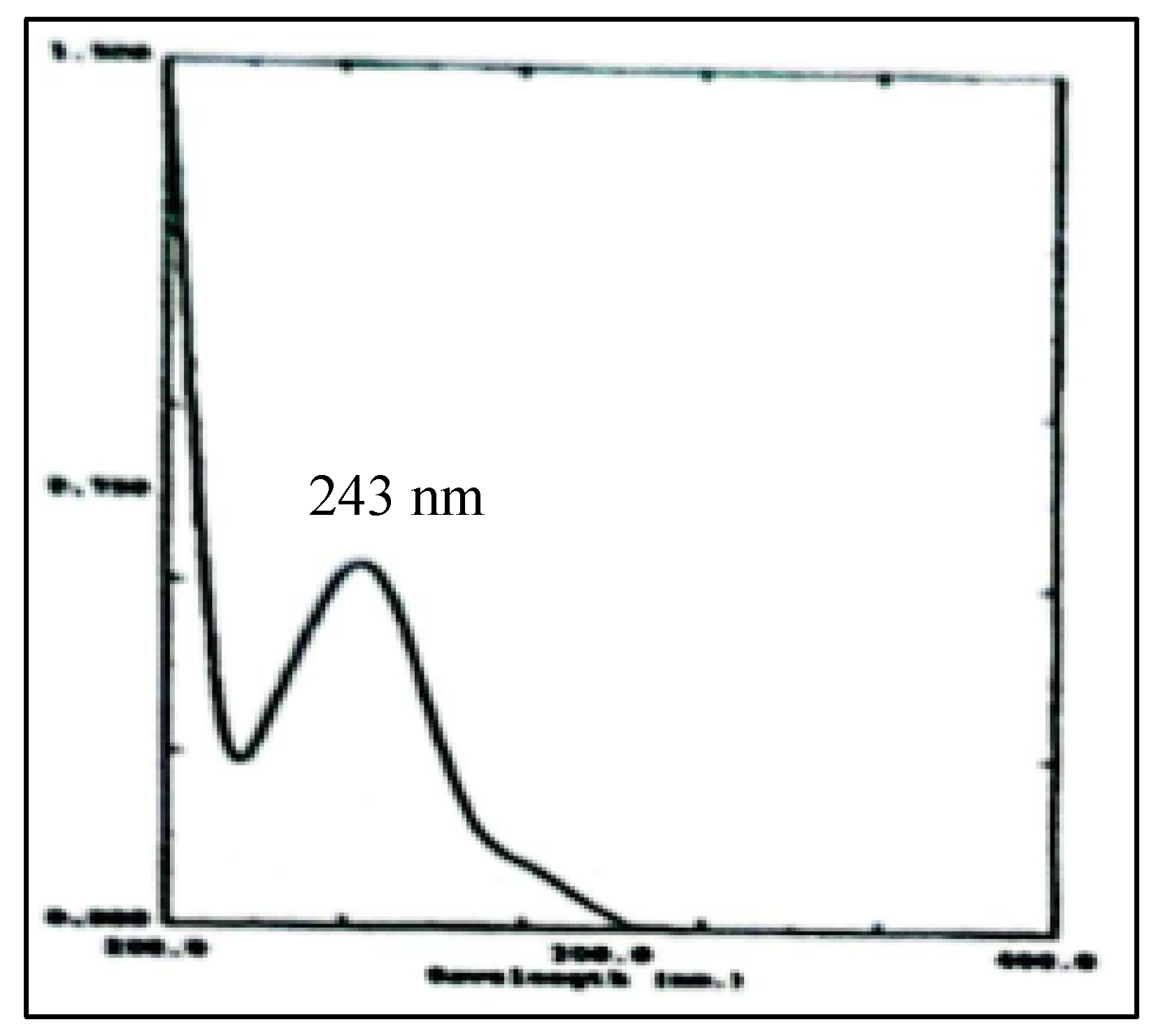

3.10.4. Tablet Preparations
3.10.5. Granule Evaluation
3.10.6. Tablet Evaluation
3.10.7. Statistical Analysis
4. Conclusion
Disclaimer (Artificial Intelligence)
Acknowledgements
Competing Interests
References
- M. Arief. (2020, October 25). Kemenperin: Impor bahan baku obat terus berkurang hingga 2024 [Internet]. Available: https://ekonomi.bisnis.com/read/20201025/257/1309563/kemenperin-impor-bahan-baku-obat-terus berkurang-hingga-2024.
- M. S. Arshad, et al., “A review of emerging technologies enabling improved solid oral dosage form manufacturing and processing.” Advanced Drug Delivery Reviews, vol. 178, pp. 113840, 2021. PMID: 34147533. [CrossRef]
- T. N. S. Sulaiman, Wahyono, A. N. Bestari, and F. N. Aziza, “Preparation and characterization of pregelatinized Sago starch (PSS) from native Sago starch (NSS) (Metroxylon sp.) and its evaluation as tablet disintegrant and filler-binder on direct compression tablet.” Indonesian Journal of Pharmacy, vol. 33, no. 2, pp. 251–260, 2022. [CrossRef]
- B. M. J. Martens, W. J. J. Gerrits, and E. M. A. M. Bruininx, et al., “Amylopectin structure and crystallinity explains variation in digestion kinetics of starches across botanic sources in an in vitro pig model.” Journal of Animal Science and Biotechnology, vol. 9, no. 91, pp. 1-13, 2018. [CrossRef]
- M. El Farkhani, et al., “A review of the chemical modification and applications of starch.” Bio Web Conference, vol. 109, pp. 01020, 2024. [CrossRef]
- J. Compart, A. Singh, J. Fettke, and A. Apriyanto, “Customizing starch properties: A review of starch modifications and their applications.” Polymers (Basel), vol. 15, no. 16, pp. 3491, 2023. PMID: 37631548; PMCID: PMC10459083. [CrossRef]
- E. Sophia. Yuk Mengenal Parasetamol [Internet]. Jakarta: Ditjen Keslan; 2023 [cited 2025 January 5].
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Pharmaceutical Drugs. Lyon (FR): International Agency for Research on Cancer; 1990. (IARC Monographs on the Evaluation of the Carcinogenic Risks to Humans, No. 50.) Paracetamol (Acetaminophen) Available from: https://www.ncbi.nlm.nih.gov/books/NBK526213/.
- R. Klingler, and K-G Busch. “Pregelatinized starches and processes for their production.” US Patent 7045003B2. 2023-06-17.
- M. Wu, et al., “Preparation and structural properties of starch phosphate modified by alkaline phosphatase.” Carbohydrate Polymer, vol. 276, pp. 118803, 2021. [CrossRef]
- R. Berdan, “The microscopic structure of starch grains food microscopy.” Motic America. 2022; March 15. https://moticmicroscopes.com/blogs/articles/food-microscopy-starch-grains#.
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapters, <1174> Powder flow. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <912> Viscosity-Rotational methods. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <731> Loss on drying. [CrossRef]
- UV calibration and whiteness FAQs. Datacolor. https://www.datacolor.com/wp-content/ uploads/2022/03/UV-Calibration-and-Whiteness-FAQs.pdf.
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <791> pH. [CrossRef]
- M. Zuhdi, and A.N. Khairi, “Analysis of organoleptic properties and consumer acceptance of frozen noodle products.” Journal of Halal Science and Research, vol. 3, no. 1, pp. 15-19, 2022. [CrossRef]
- USP 2024 (United State Pharmacopeia 47-NF 42): Acetaminophen. https://doi.org/10.31003/USPNF_M150_05_01.
- N.P. Ashok, and G.J. Khan, “A simple UV-Vis spectrophotometric assay study on different brand of paracetamol.” WJPLS, vol. 8, no. 9, pp, 186-193, 2022. https://www.wjpls.org/home/article_abstract/2751.
- A. Sucipto, F. Iskandar, Hanifah, W. Rahmawati, and S.A. Fiqrotinnisa, “The application of Amylum manihot as disintegrant in the formulation of paracetamol tablets by wet granulation method.” Journal of Scince and Technology Research for Pharmacy, vol. 1, no. 2, pp. 26-30, 2022. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <786> Particle Size Distribution Estimation by Analytical Sieving. [CrossRef]
- S. Ahmad, “Thickness test for tablets” [Internet]. Pharmainfo; 2022 [cited 2025 January 10]. https://www.pharmainform.com/2022/07/thickness-test-for-tablets.html.
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter <1217> Tablet breaking force. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter <1216> Tablet friability. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <701> Disintegration. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <905> Uniformity of dosage units. [CrossRef]
- G. Marisa, J. Kapala, T. Mafuru, R. Matinde, E. Kimaro, and E. Kaale, “Quality evaluation of locally manufactured paracetamol tablets in East Africa.” BioMed Research International, pp. 9437835, 2024 Sep 14. PMID: 39310289; PMCID: PMC11416170. [CrossRef]
- USP 2024 (United State Pharmacopeia 47 - NF 42): General chapter, <711> Dissolution. [CrossRef]
- Y. Hong, and X. Liu, “Pre-gelatinized Modification of Starch.” In: Z. Sui, and X. Kong, (eds), “Physical Modifications of Starch.” Springer: Singapore, 2018. https://link.springer.com/chapter/10.1007/978-981-13-0725-6_4.
- N.G. Marotta, and H. Bell, “Method of preparing pregelatinized starch compositions.” US3464857A (Patent) 1967.
- D. Anggraini, A. Lukman, and H.Y. Teruna, “Physicochemical properties of phosphate pregelatinized Musa balbisiana starch as pharmaceutical excipient.” Pharmacology and Clinical Pharmacy Research, vol. 1, no. 3, pp. 90-96, 2016. [CrossRef]
- Depkes RI, “Farmakope Indonesia, edisi 4.” Jakarta: Depkes RI; 1995.
- N. Fadjria, A. Arfiandia, N.D. Auliyah, “Analysis of amylose content in sweet corn starch (Zea Mays L. Var. Saccharata) by UV-Vis spectrophotometry.” Journal of Pharmaceutical and Sciences, vol. 7, no. 2, pp. 152-158, 2024. [CrossRef]
- E. Bertoft, “Understanding starch structure: Recent progress.” Agronomy, vol. 7, no. 3, pp. 56, 2017. [CrossRef]
- E.I. Yousif, M.G.E. Gadallah, and A.M. Sorour, “Physico-chemical and rheological properties of modified corn starches and its effect on noodle quality.” Annals of Agricultural Sciences, vol. 57, no. 1, pp. 19-27, 2012. [CrossRef]
- C. Ophardt, “Starch and iodine” [internet]. California: LibreTexts. https://chem.libretexts.org/Bookshelves/ Biological_Chemistry/Supplemental_Modules.
- N. Aini, and P. Hariyadi, “Utilization of modified white corn starch in producing marshmallow cream.” IJFAC, vol. 3, no. 2, pp. 40-46, 2018. [CrossRef]
- P.M. Lestari, A. Widayanti, and H. Afifah, “The effect of pregelatinized taro starch (Colocasia esculenta (L.) Schott) on temperature as filler on thiamine hidrochloride tablet. Open Access Macedonian Journal of Medical Sciences, vol. 7, no. 22, pp. 3827-3832, 2019. PMID: 32127985; PMCID: PMC7048343. [CrossRef]
- E.G.C. Clarke, and A.C. Moffat, eds. “Clarke's Isolation and Identification of Drugs in Pharmaceuticals, Body Fluids, and Post-mortem Material, 2nd ed. 2.” Michigan: Pharmaceutical Press; 1986.
- B. Setyono, and F.A. Purnawiranita, “Analysis of flow characteristics and paracetamol tablet hardness using 2D double mixer of design drum type with rotation and mixing time variations.” Journal of Mechanical Engineering, Science, and Innovation, vol. 1, no. 2, pp. 38-48, 2021. [CrossRef]
- B. Hartesi, Sriwidodo, M. Abdassah, and A.Y. Chaerunisa, “Starch as pharmaceutical excipient.” International Journal of Pharmaceutical Sciences Review and Research, vol. 41, no. 2, 59-64, 2016. https://globalresearchonline.net/journalcontents/v41-2/14.
- P. Hiremath, K. Nuguru, and V. Agrahari, “Chapter 8 - Material Attributes and Their Impact on Wet Granulation Process Performance,” Editor(s): A.S. Narang, and S.I.F. Badawy, “Handbook of Pharmaceutical Wet Granulation.” Academic Press, pp. 263-315, 2019, ISBN 9780128104606. [CrossRef]
- P. Thapa, A.R. Lee, D.H. Choi, and S.H. Jeong, “Effects of moisture content and compression pressure of various deforming granules on the physical properties of tablets.” Powder Technology, vo. 310, pp. 92-102, 2017. [CrossRef]
- A. Patel, C. Jin, B. Handzo, and R. Kalyanaraman, “Measurement of moisture content in pharmaceutical tablets by handheld near-infrared spectrometer: Adopting quality by design approach to analytical method lifecycle management.” Journal of Pharmaceutical and Biomedical Analysis, vol 229, pp. 115381, 2023. [CrossRef]
- D.S. Shah, K.K. Moravkar, D.K. Jha, V. Lonkar, P.D. Amin, S.S. Chalikwar, “A concise summary of powder processing methodologies for flow enhancement.” Heliyon, vol. 9, no. 6, pp. e16498, 2023 May 24. PMID: 37292344; PMCID: PMC10245010. [CrossRef]
- Utsumi R. An attrition test with a sieve shaker for evaluating granule strength. Powder Technology, vol. 122, no. 2, pp. 199-204, 2001. [CrossRef]
- A.L. Skelbæk-Pedersen, T.K. Vilhelmsen, J. Rantanen, and P. Kleinebudde, “The relevance of granule fragmentation on reduced tabletability of granules from ductile or brittle materials produced by roll compaction/dry granulation.” International Journal of Pharmaceutics, vol. 592, pp. 120035, 2021. [CrossRef]
- H. Leuenberger, “The compressibility and compactibility of powder systems.” International Journal of Pharmaceutics, vol. 12, no. 1, pp. 41-55, 1982. [CrossRef]
- O. Macho, et al., “Systematic study of paracetamol powder mixtures and granules tabletability: Key role of rheological properties and dynamic image analysis.” International Journal of Pharmaceutics, vol. 608, pp. 121110, 2021. [CrossRef]
- Azimuddin, M.F. Roslan, and R.T. Widodo, “Formulation and in vitro evaluations of paracetamol orally disintegrating tablets.” Journal of Food and Pharmaceutical Sciences, vol. 11, no. 1, pp. 780-787, 2023. [CrossRef]
- R. Kamour, I. El-Sharaa, and A. Eswayah, “Physical and chemical evaluation of different brands of paracetamol tablet.” AlQalam Journal of Medical and Applied Sciences, vol. 7, no. 2, pp. 257-260, 2024. [CrossRef]
- Y.L. Achille, et al., “Quality control of paracetamol generic tablets marketed in Benin and search of its two impurities p-aminophenol and p-nitrophenol by HPLC-UV/Visible.” American Journal of Analytical Chemistry, vol. 13, no. 11, pp. 449-460, 2022. [CrossRef]
- British Pharmacopoeia Commission. “British Pharmacopoeia. 2nd ed.” London: The Stationery Office, 2007.
- C.H.R. Serra, K.H. Chang, T.M. Dezani, V. Porta, and S. Storpirtis, “Dissolution efficiency and bioequivalence study using urine data from healthy volunteers: a comparison between two tablet formulations of cephalexin.” Brazilian Journal of Pharmaceutical Sciences, vol. 51, no. 2, pp. 383-392, 2015. [CrossRef]
- Jubril, J. Muazu, and G.T. Mohammed, “Effects of phosphate modified and pregelatinized sweet potato starches on disintegrant property of paracetamol tablet formulations.” Journal of Applied Pharmaceutical Science, vol. 2, no. 2, pp. 32-36, 2012. https://www.researchgate.net/publication/265219787.
- IBM SPSS Statistics, “Analyze data with ease, forecast trends accurately, and drive key outcomes using IBM SPSS Statistics.” Available from: https://www.ibm.com/products/spss-statistics (accessed April 1, 2025).

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




