Submitted:
02 July 2025
Posted:
03 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. CO2 Reduction Technologies
- CO2 Capture and Storage (CCS)
- CO2 Capture and Utilization (CCU)
- CO2 Conversion into Value-Added Products
2.1. Mechanism of CO2 Reduction
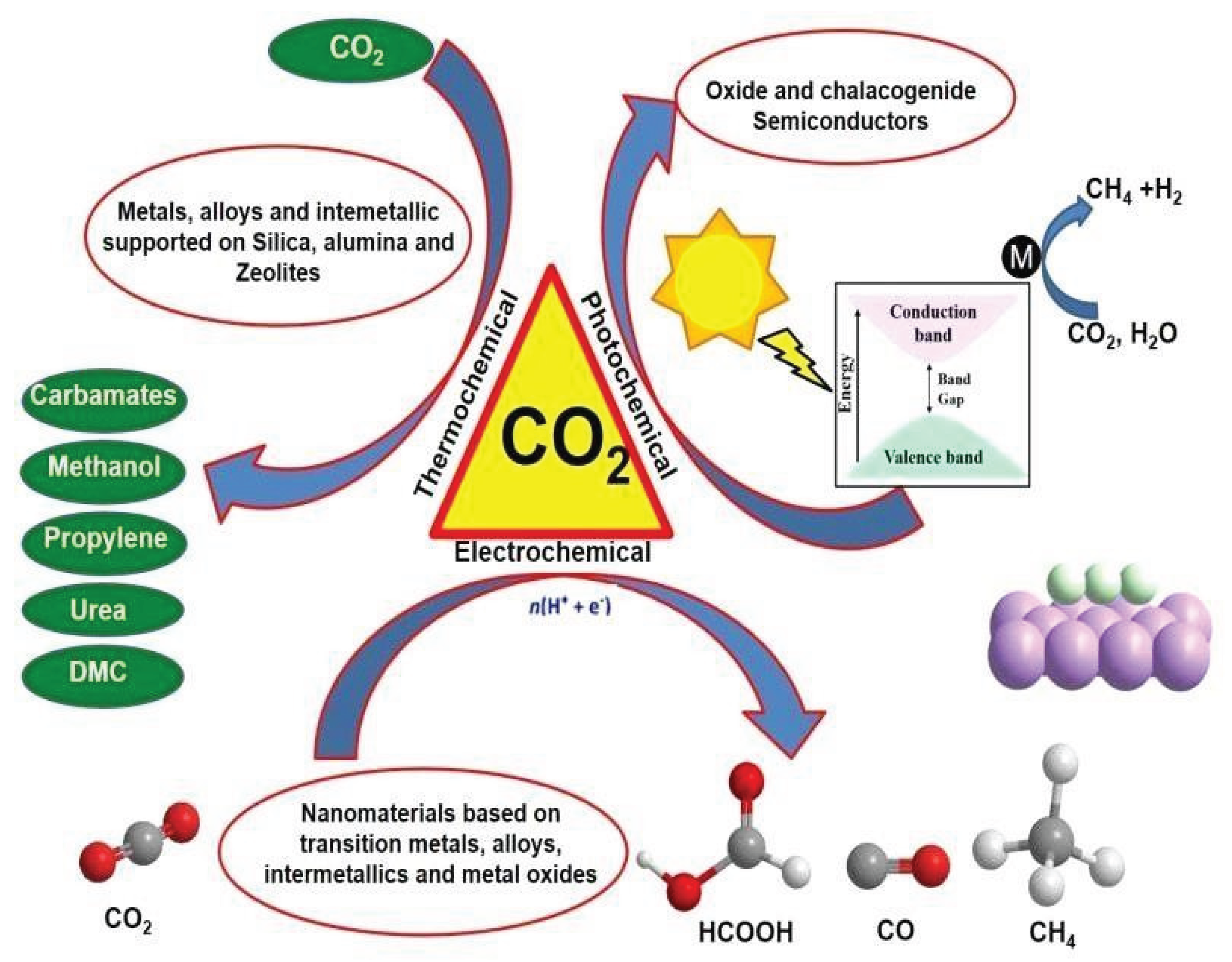
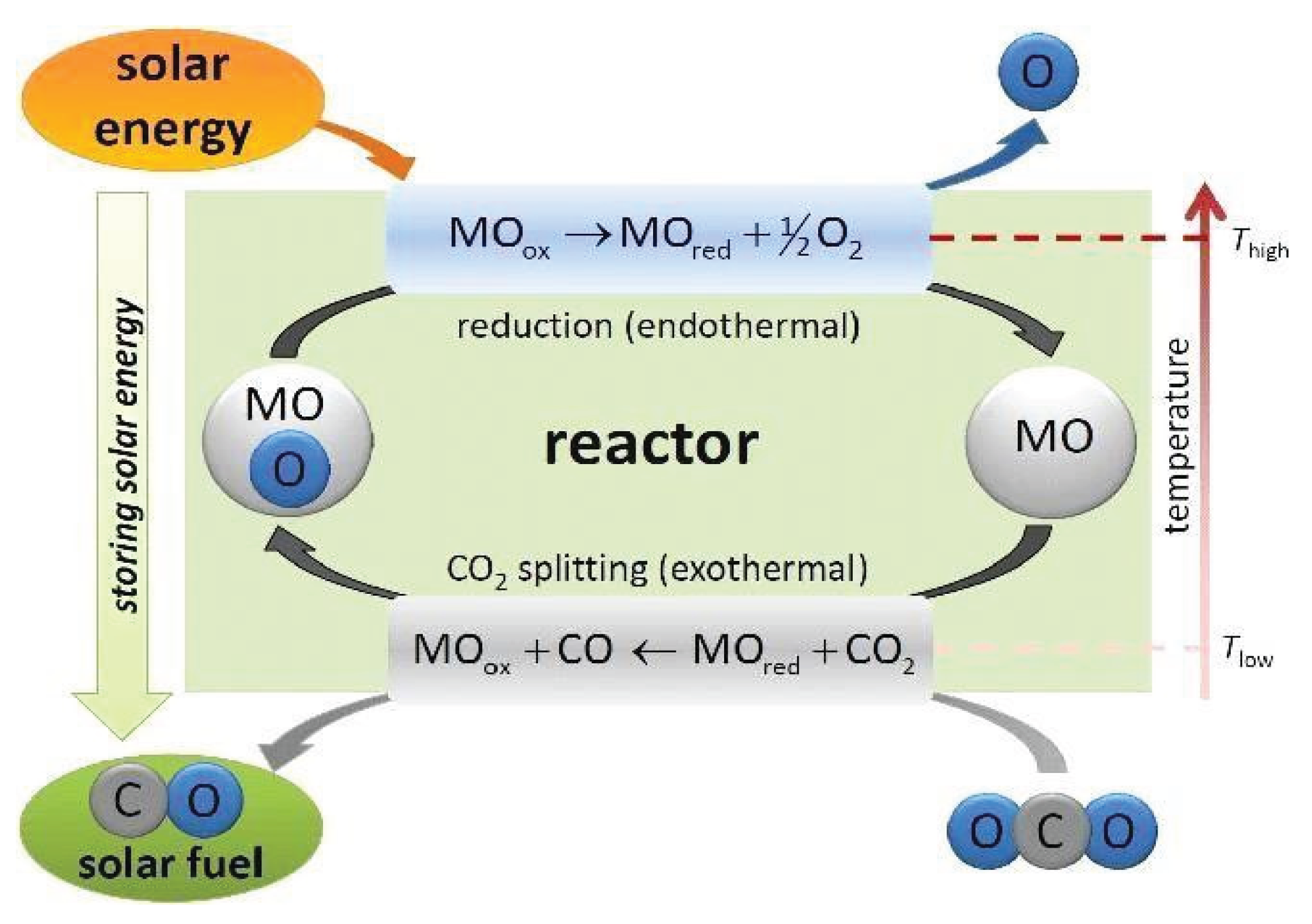
2.1.1. Thermochemical Processes
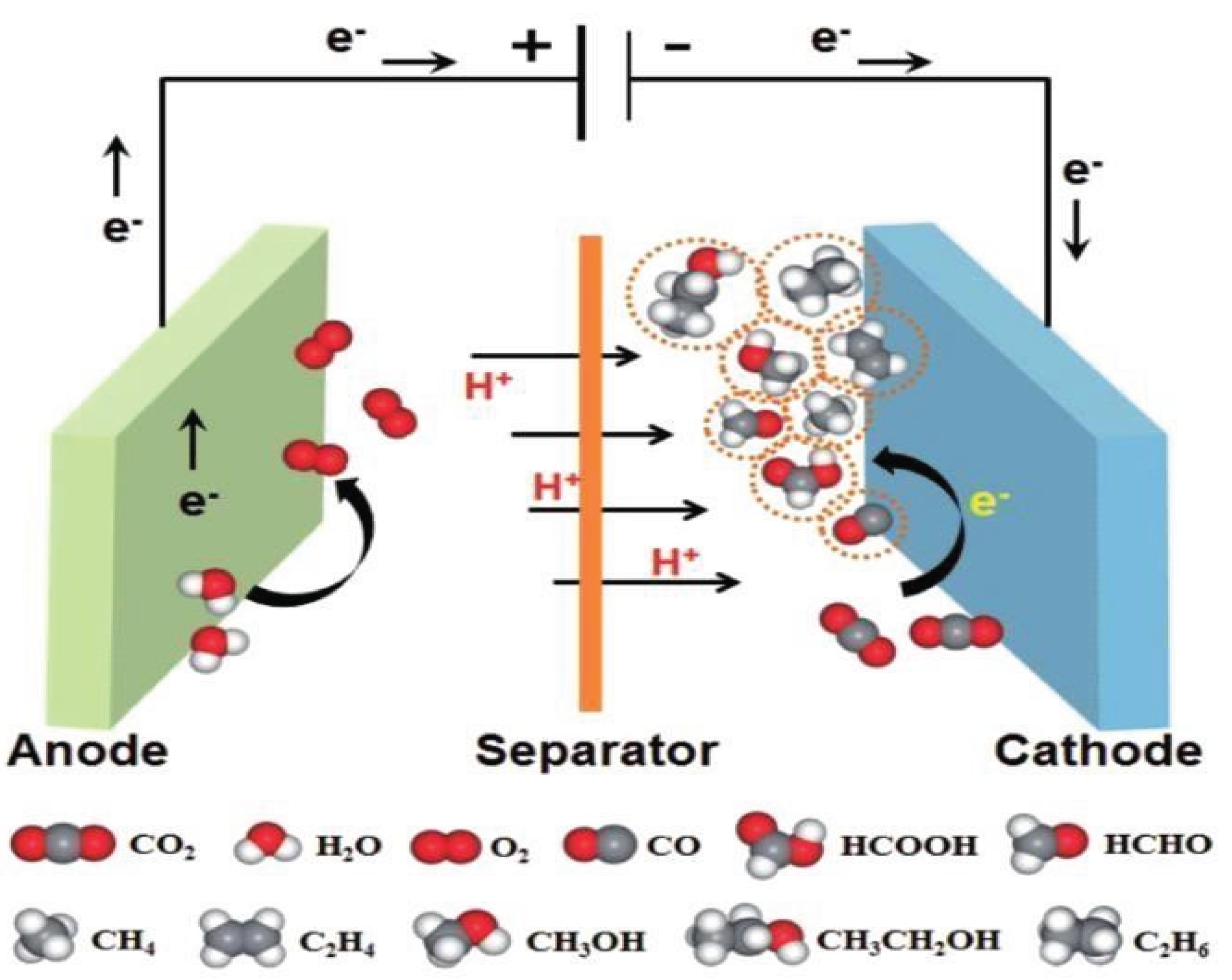
2.1.2. Electrochemical Reduction
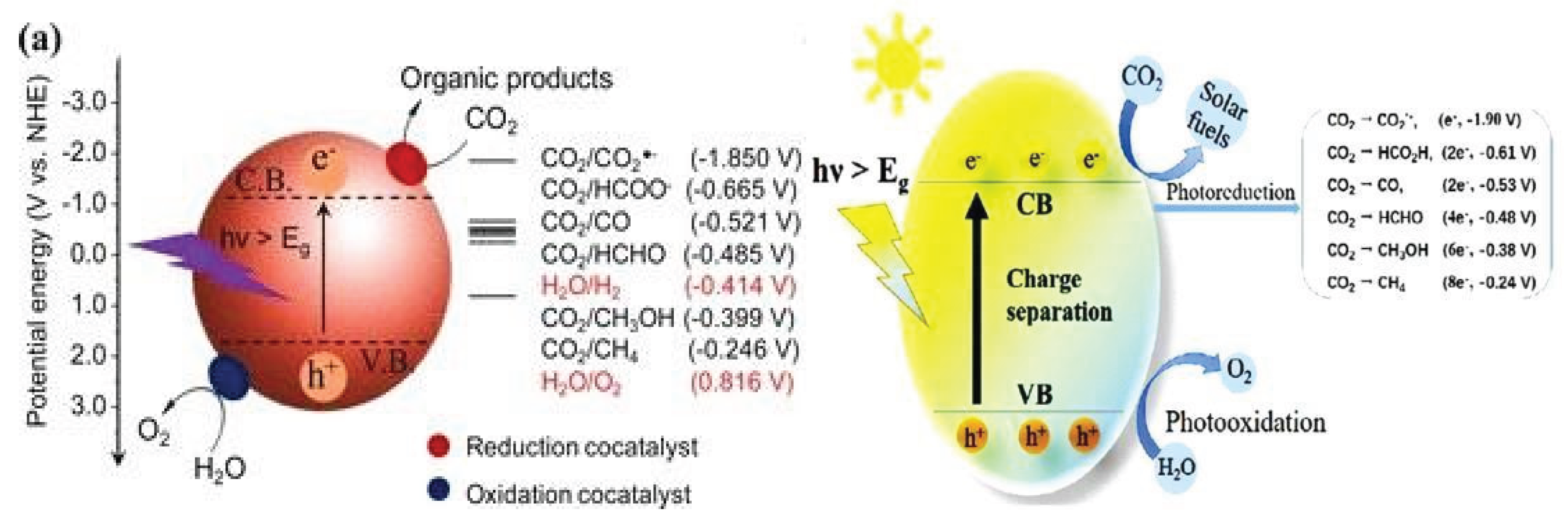
2.1.3. Photocatalytic Reduction
- Light adsorption (hv>Eg) by the photocatalysts: In this step photocatalysts adsorb photon light from sun light, promoting the generated electrons from valance band to conduction band and leaving behind holes in valance band so this step creation electron- hole pairs.
- Charge separation: this step prevents the recombination of generated electron- hole pairs, through catalyst modification such as Surface modifications or the introduction of co-catalysts can enhance this separation.
- Surface Reactions (Redox reaction): In this step reduction and oxidation both processes take place.
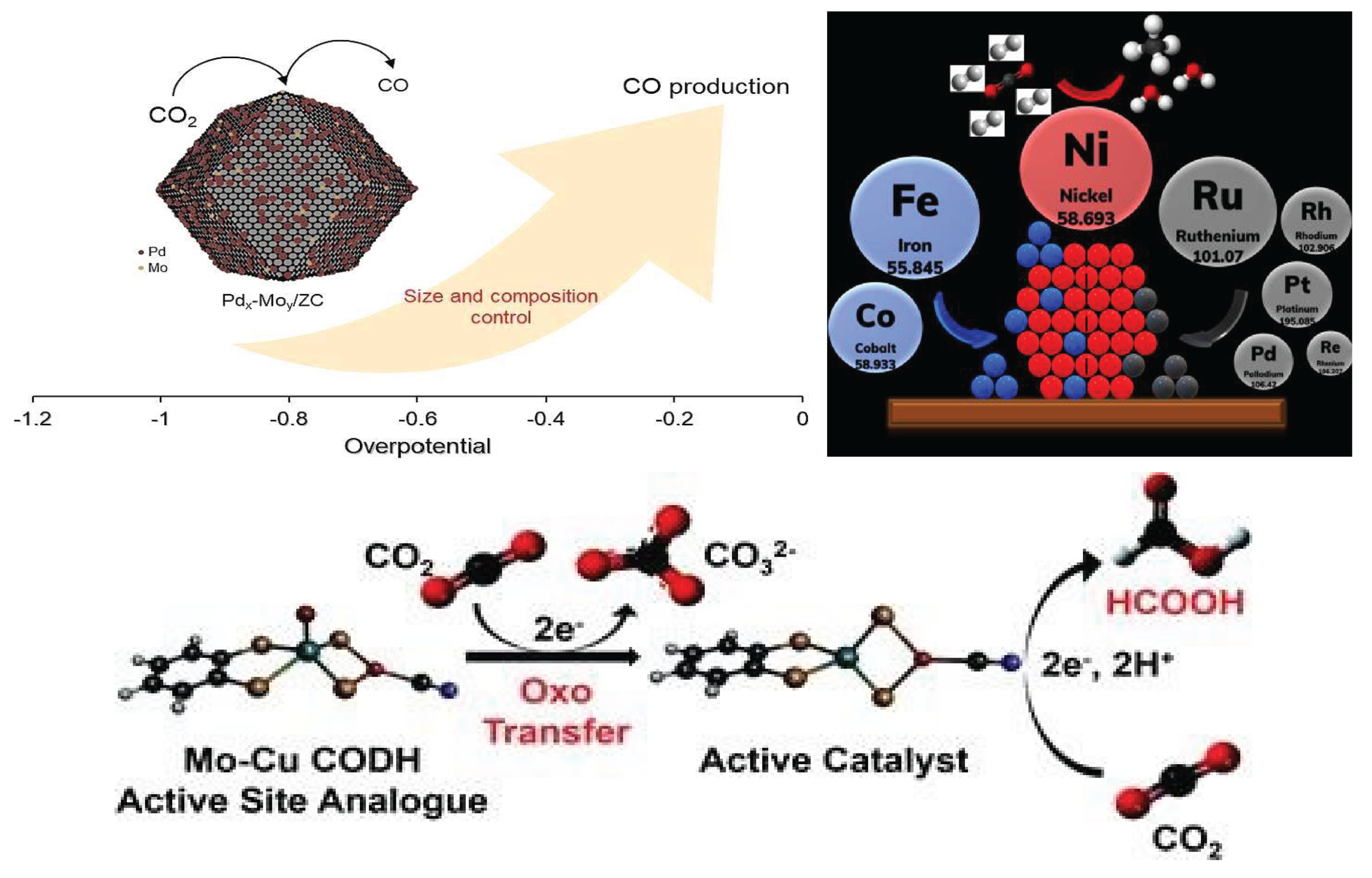
2.2. Role of Earth-Abundant Metals in CO2 Reduction
3. Earth – Abundant Metals as Catalysts
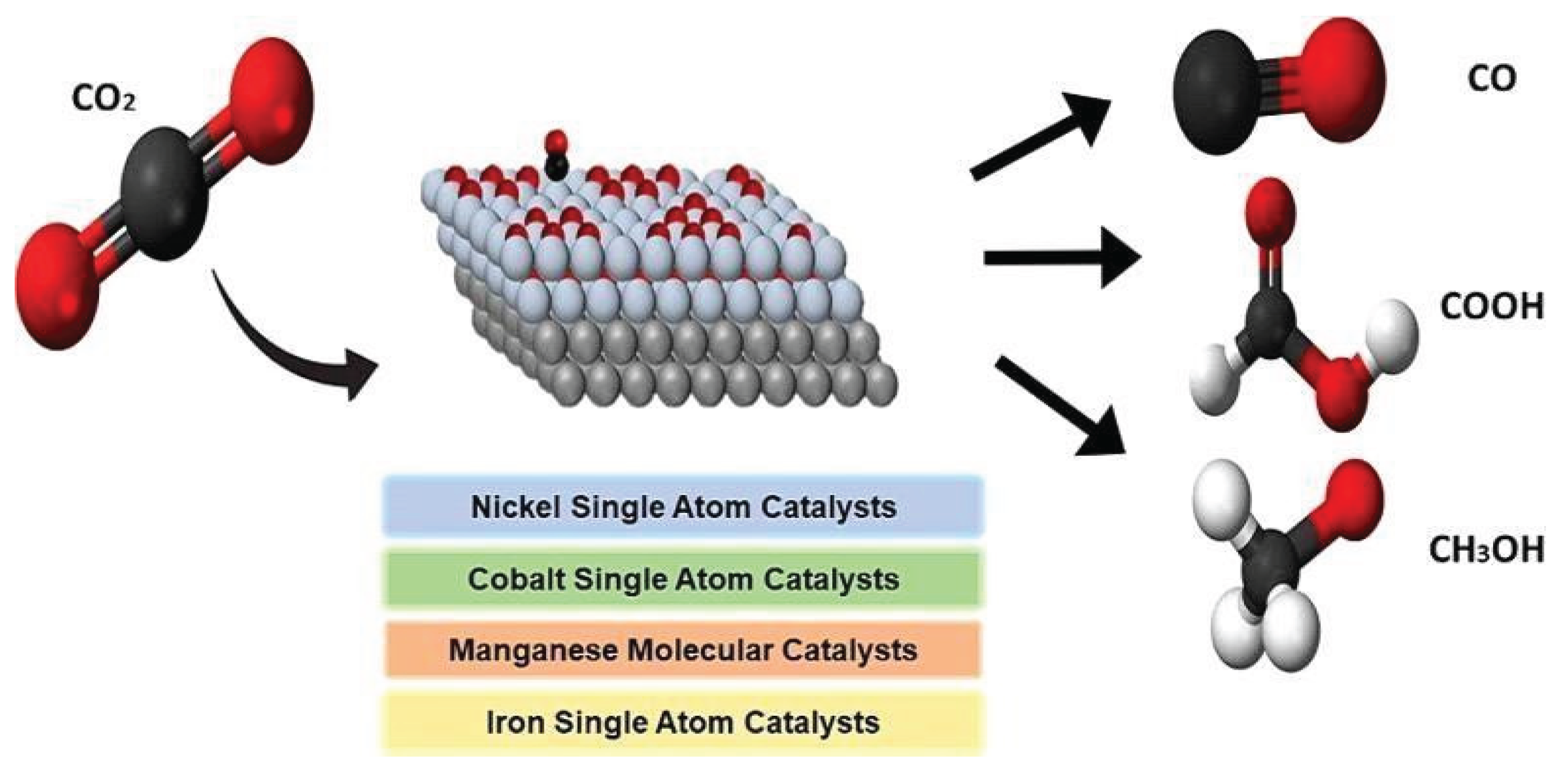
3.1. Molecular and Single Atom-Based Catalysts and Their Roles in CO2 Reduction
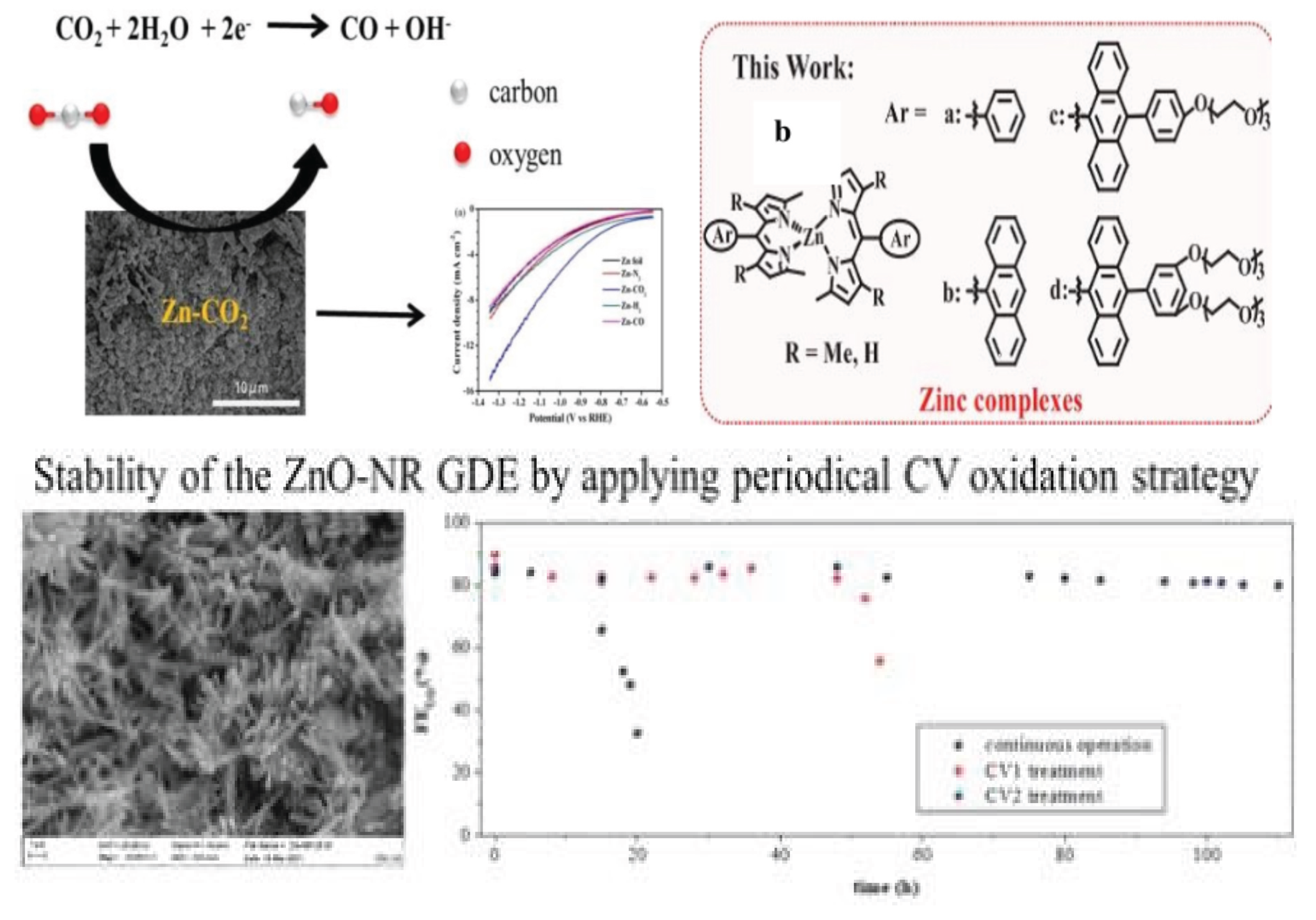
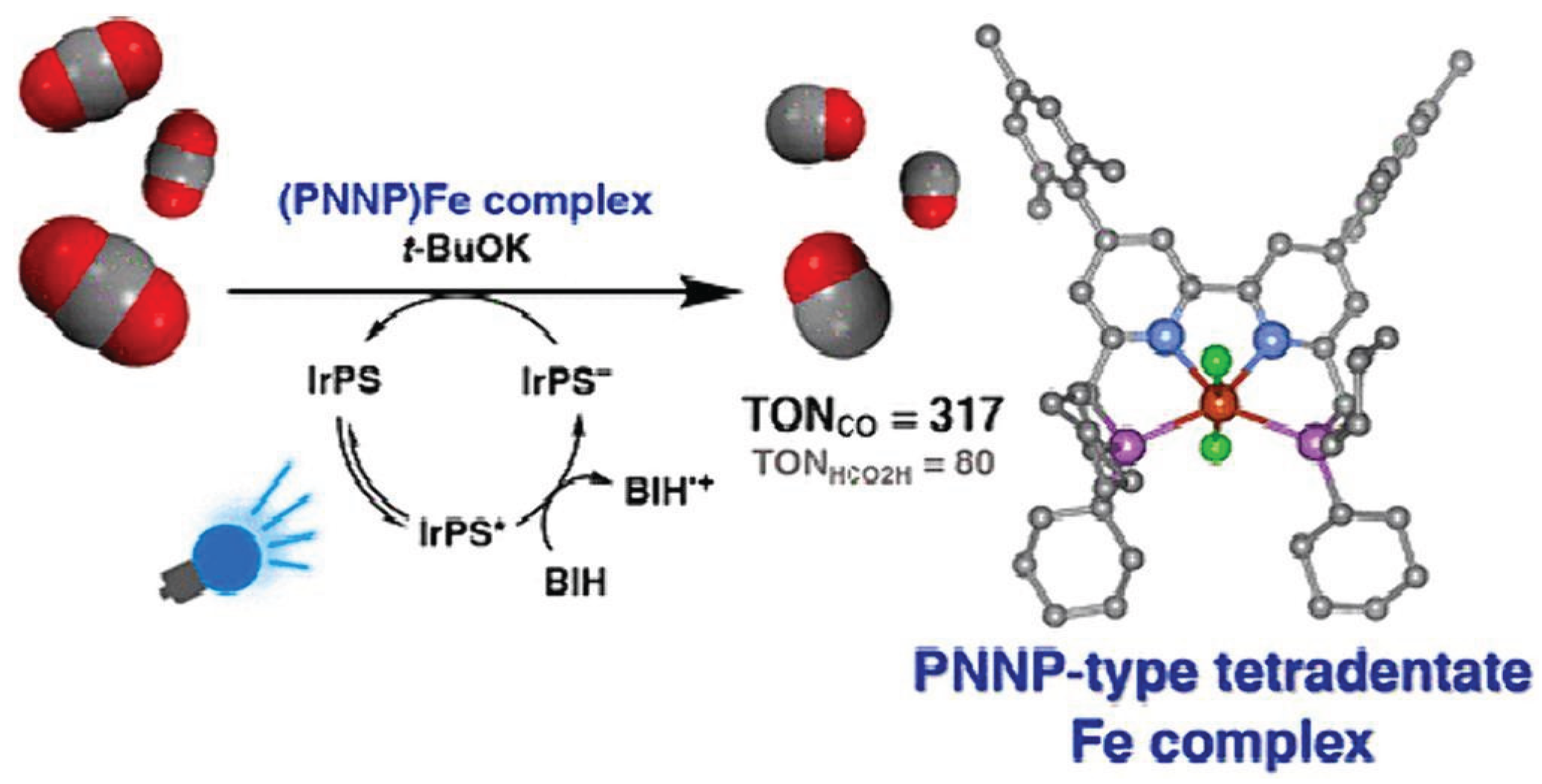
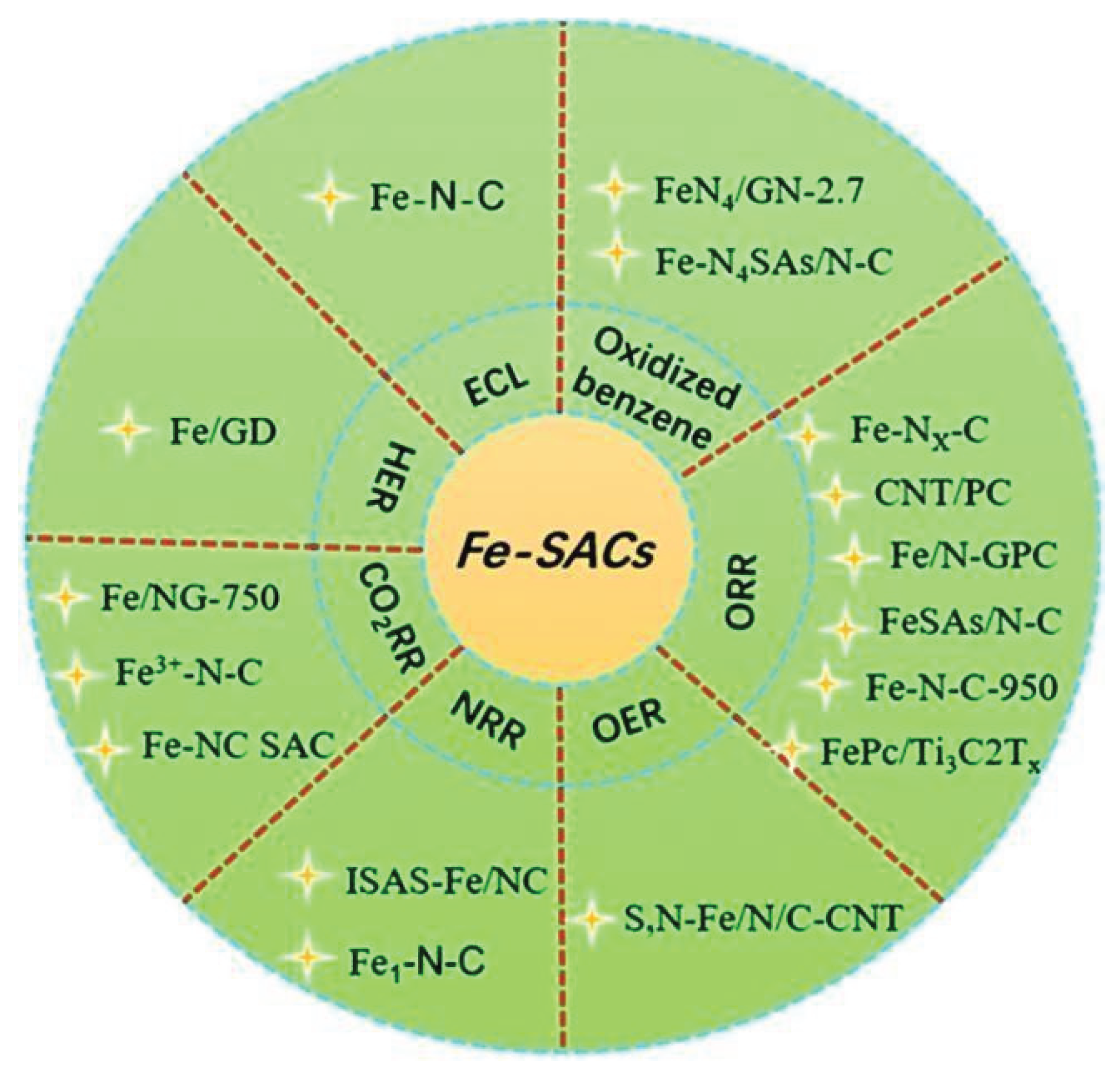
3.1.1. Iron Based Catalysts (Fe-SACs) for CO2 Reduction
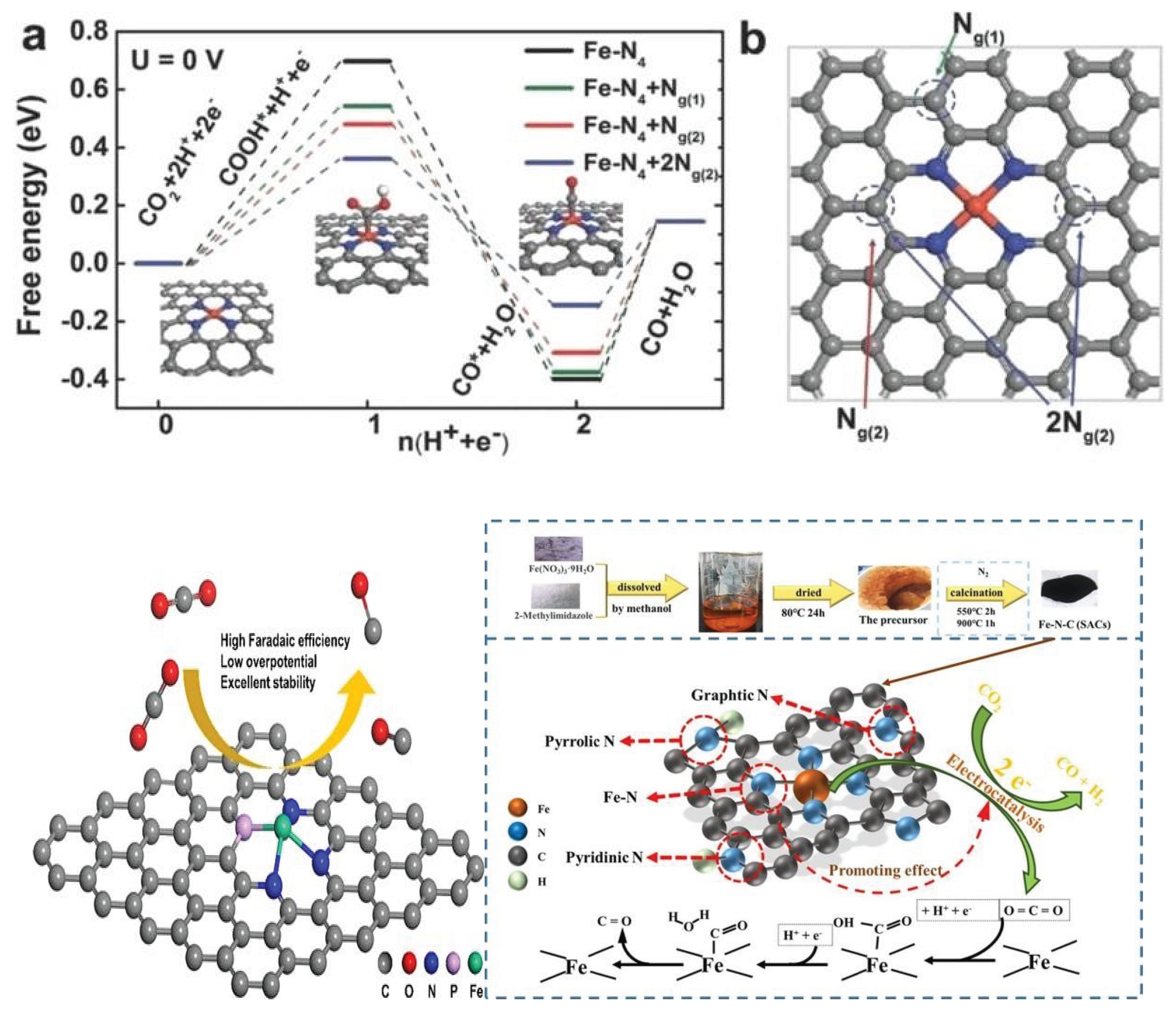
| Sr. No | Name of Metal Catalyst | Type of catalysts | Conversion Form | Conditions | Faradaic efficiency | References |
|---|---|---|---|---|---|---|
| 1. | Iron–porphyrin complex | Electrocatalyst | CO2 to CO | −0.97 V | 90% | [30] |
| 2. | Fe- Complex with Bipyridyl | Electrocatalytic reduction | CO2 to CO and HCOOH | -0.91 V | 86% | [31] |
| 3. | Fe-SACs | Electrocatalyst | CO2 to CO | -0.89 V | 92% | [32] |
| 4. | Fe/NG | Electrocatalytic reduction | CO2 to CO | -0.57V | 80% | [36] |
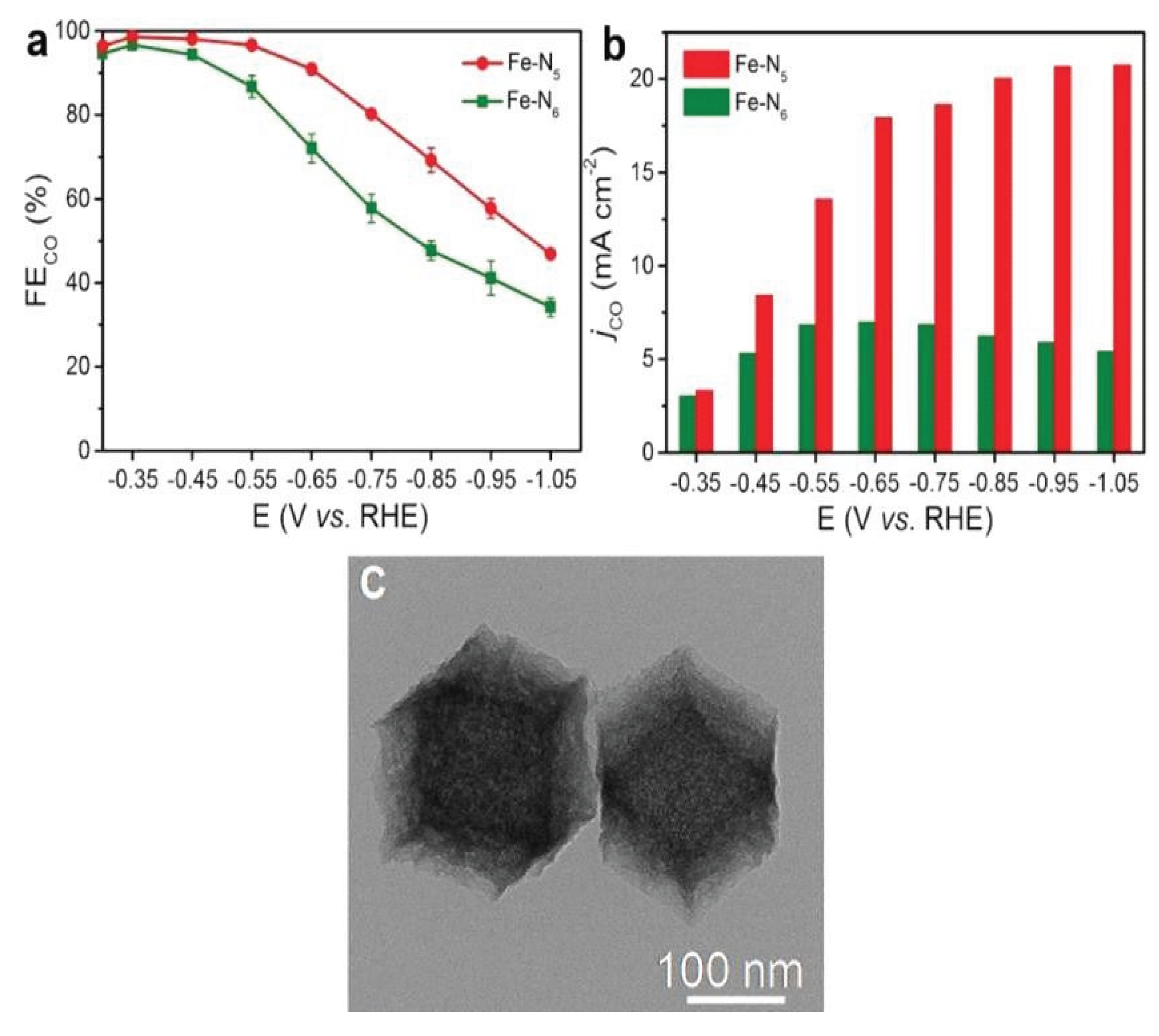
| 5. | Fe–N6 | Electroreduction | CO2 to CO | −0.35 to −0.65 V | >90% | [33] |
| 6. | Fe–N/P–C | Electrochemical reduction | CO2 to CO | 0.34 V | 98% | [35] |
| 7. | Fe–N–C | Electrocatalytic reduction | CO2 to CO | −0.68 | 98% | [38] |
| 8. | FeBxCy | Electrocatalytic reduction | CO2 to CO | -0.45 V | 99.02% | [39] |
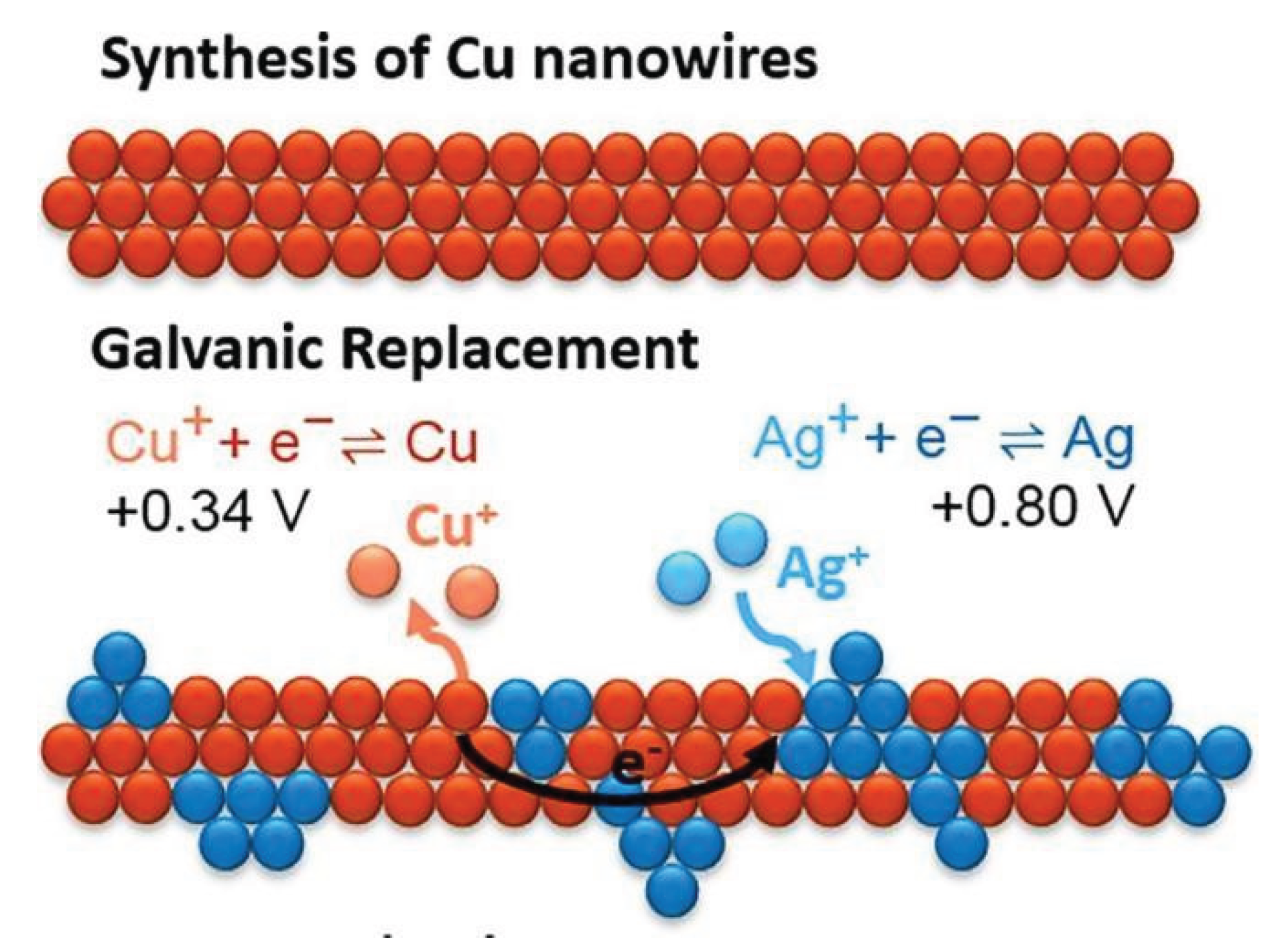
3.1.2. Cupper (Cu) Based Catalysts for CO2 Reduction
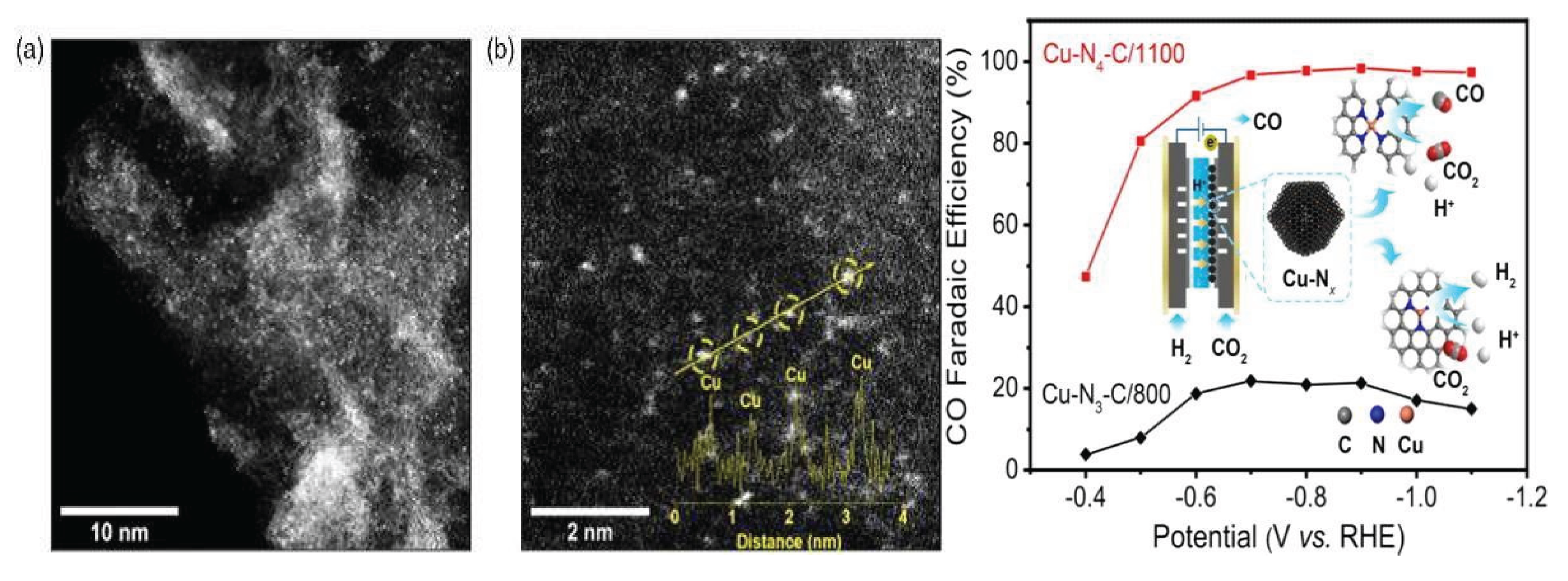
| S.R. No. | Name of Metal Catalyst | Type of catalysts | Conversion Form | Conditions | Faradaic efficiency | References |
|---|---|---|---|---|---|---|
| 1. | Cu-N-C | electroreduction | CO2 to CO | −0.67 V | 98% | [40] |
| 2. | Cu–N4– NG | electrocatalytic reduction | CO2 in CO | −1.0 V | 80.6% | [41] |
| 3. | Cu–N4 | electrocatalytic reduction | CO2 to CO | –0.9 V | 98% | [42] |
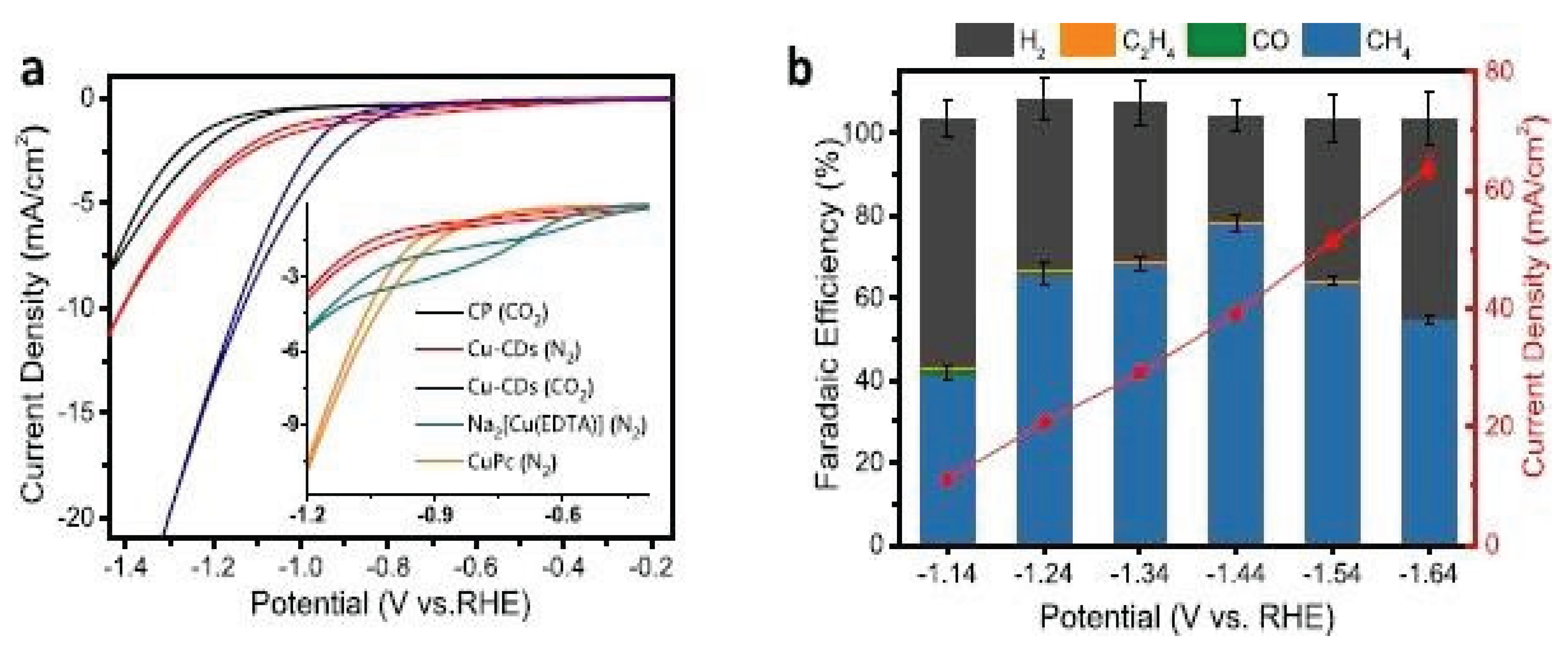
| 4. | Cu-CDs | electrochemical reduction | CO2 to CH4 | −1.64 V | 99% | [44] |
| 5. | Cu3N-derived Cu nanowires | electrocatalyst | CO2 to C2 | −1.0 V | 86% | [45] |
| 6. | Cu- Nx-C catalysts | electrocatalyst | CO2 reduction to CO | −1.1 V | 90% | [46] |
| 7. | CuN2O2 | electrochemical reduction | CO2 to C2 | −1.0 V | 86% | [48] |
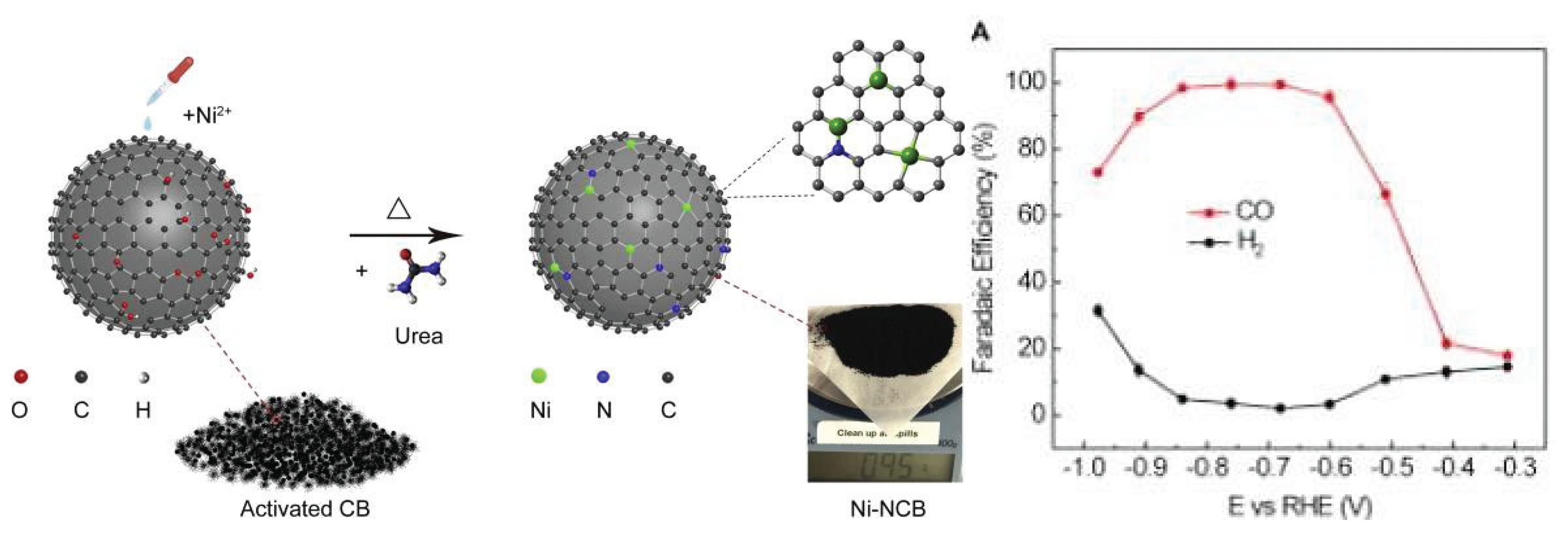
3.1.3. Nickel Based Single Atom Catalysts (Ni-SACs) for CO2 Reduction
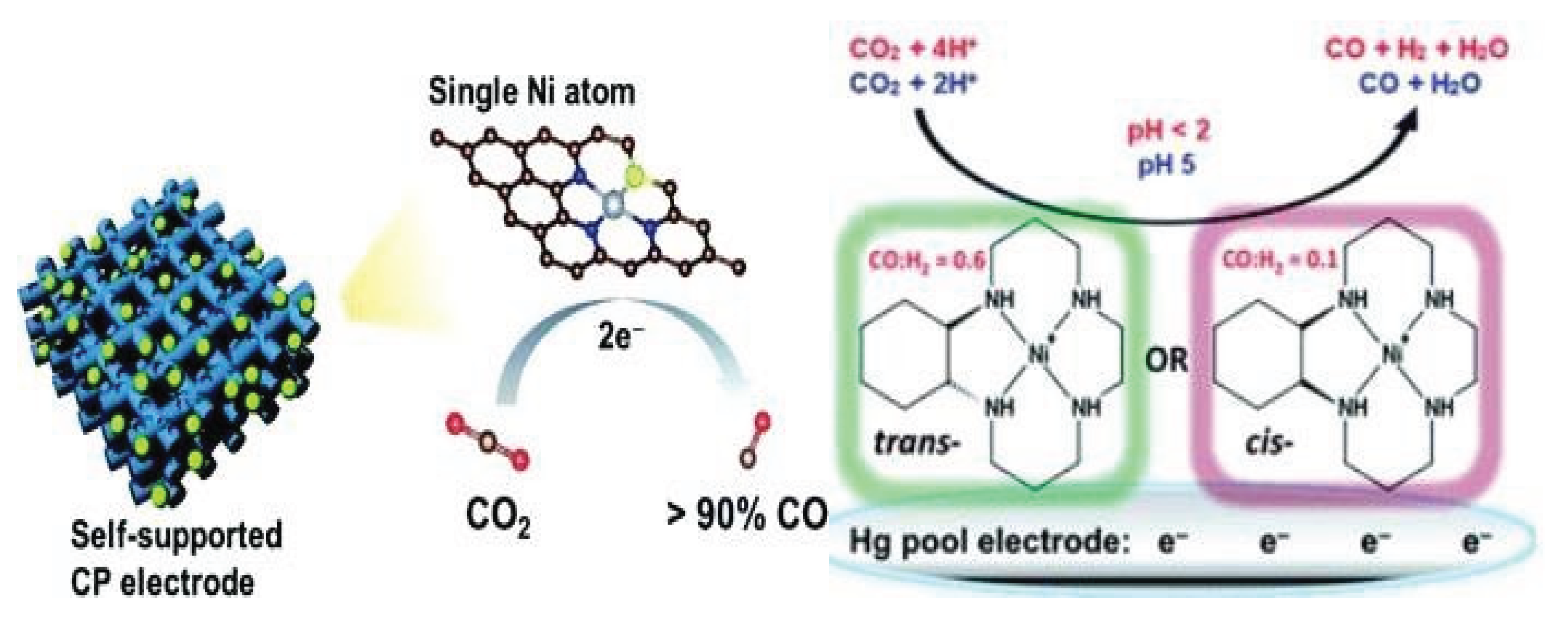
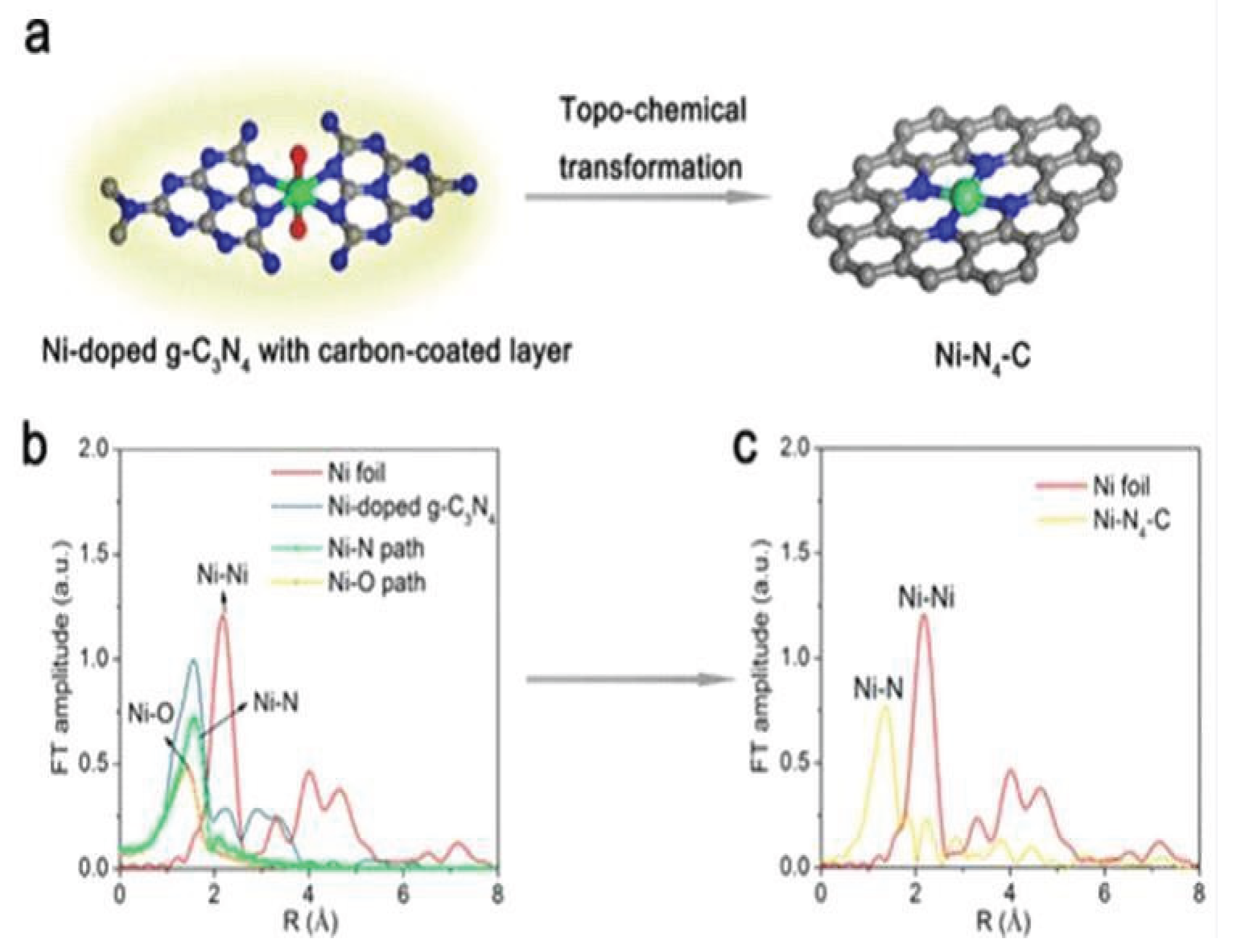
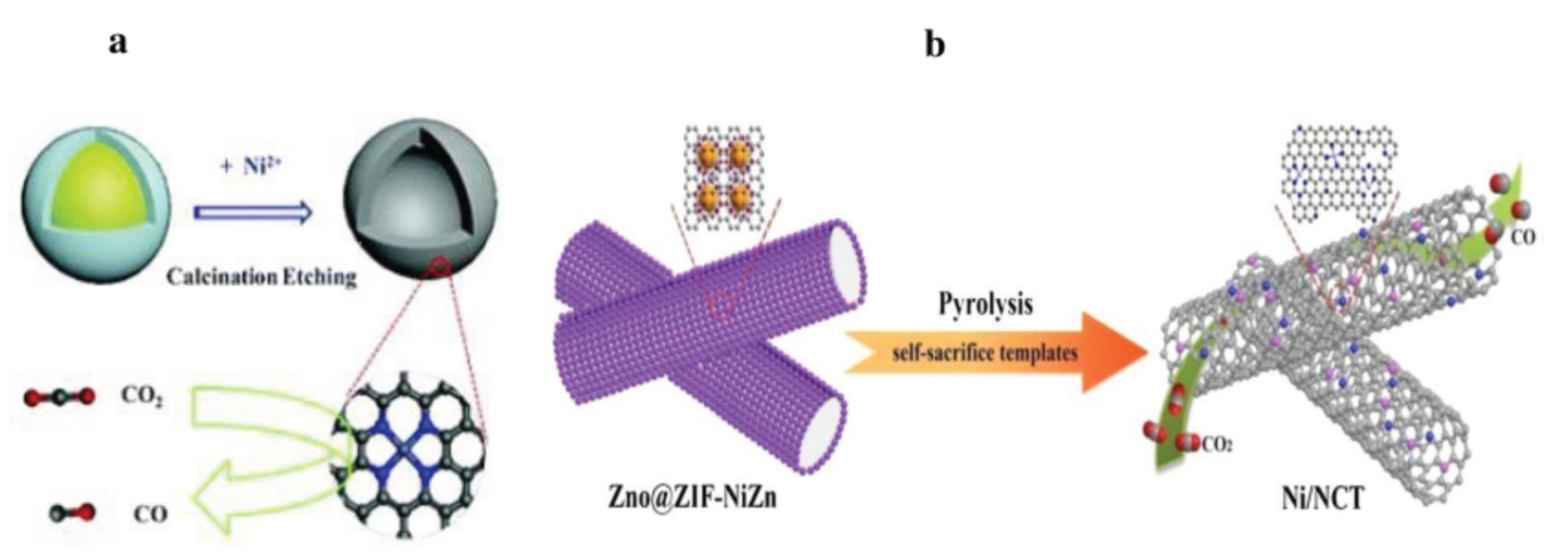
| Sr. No | Name of Metal Catalyst | Type of catalysts | Conversion Form | Conditions | Faradaic efficiency | References |
|---|---|---|---|---|---|---|
| 1. | Ni-SACs | electrochemical reduction | CO2 to CO | 0.681 V | 99% | [49] |
| 2. | Ni-N-C | electrochemical reduction | CO2 to CO. | 0.6 V | 98% | [52] |
| 3. | Ni- SACs | electrochemical reduction | CO2 to CO | –0.681V | 99% | [53] |
| 4. | [Ni(cyclam)]2+ | electrocatalyst | CO2 to CO | 0.6 V | 99% | [54] |
| 5. | Ni-CTF | electrocatalytic reduction | CO2 to CO | −0.8 V | 90% | [55] |
| 6. | SA–Ni/N–CS | electrocatalytic reduction | CO2 to CO | −0.8 V | 95.1% | [58] |
| 7. | The Ni–N4–C | electrocatalytic reduction | CO2 to CO | −0.81 V | 99% | [59] |
3.1.4. Cobalt Based Single Atom Catalysts (Co-SACs) for CO2 Reduction
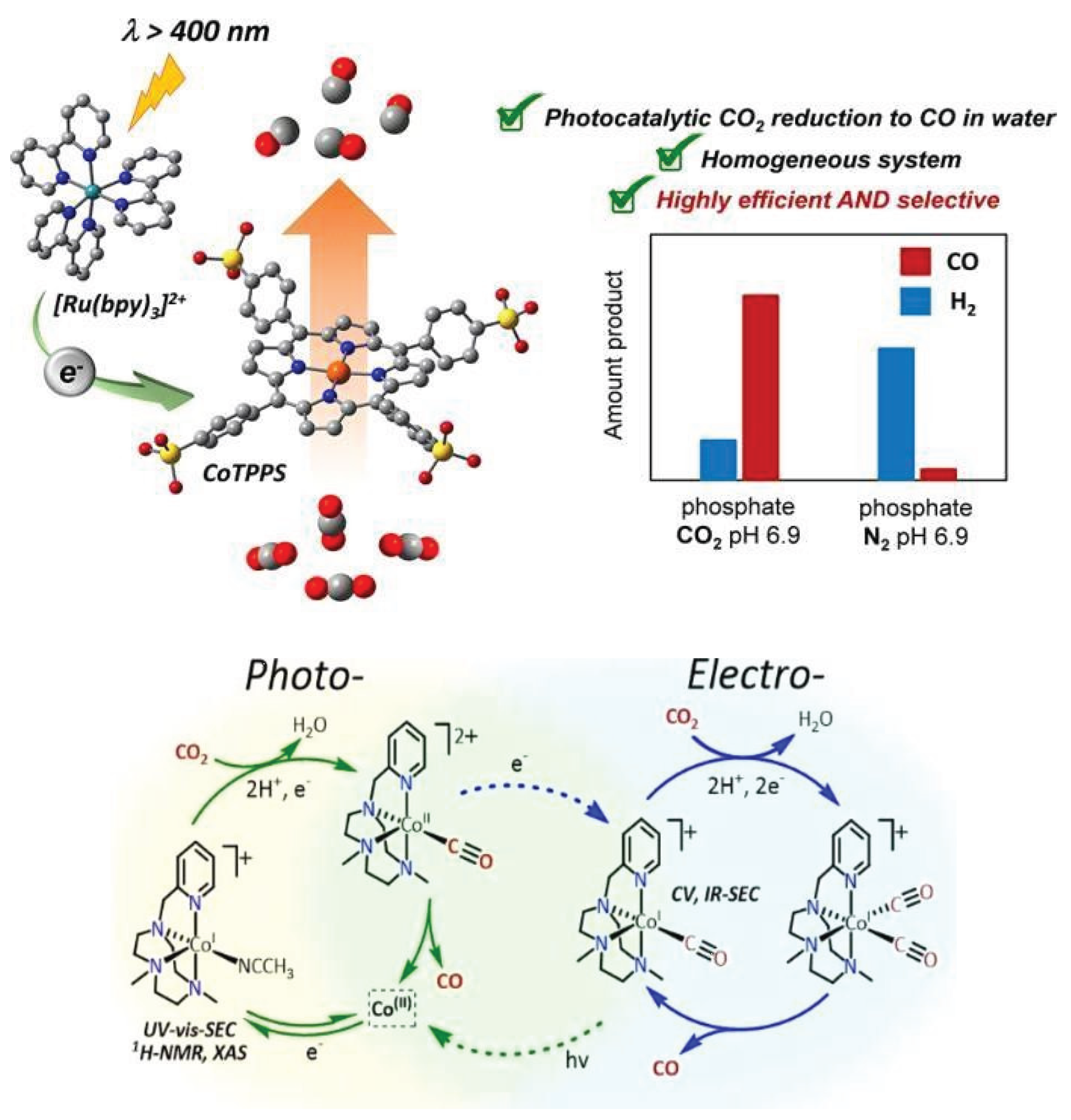

| Sr.No. | Name of Metal Catalyst | Type of catalysts | Conversion Form | Conditions | Faradaic efficiency | References |
|---|---|---|---|---|---|---|
| 1. | CoTPPS | photocatalytic reduction | CO2 to CO | −0.92 V | 90% | [65] |
| 2. | CO2RR | photocatalytic reduction | CO2 to CO | -0.94 V | > 95% | [68] |
| 3. | Co–N5 | photocatalytic reduction | CO2 to CO | −0.73 V | 99.2% | [70] |
3.1.5. Zinc Based Single Atom Catalysts (Zn-SACs) for CO2 Reduction
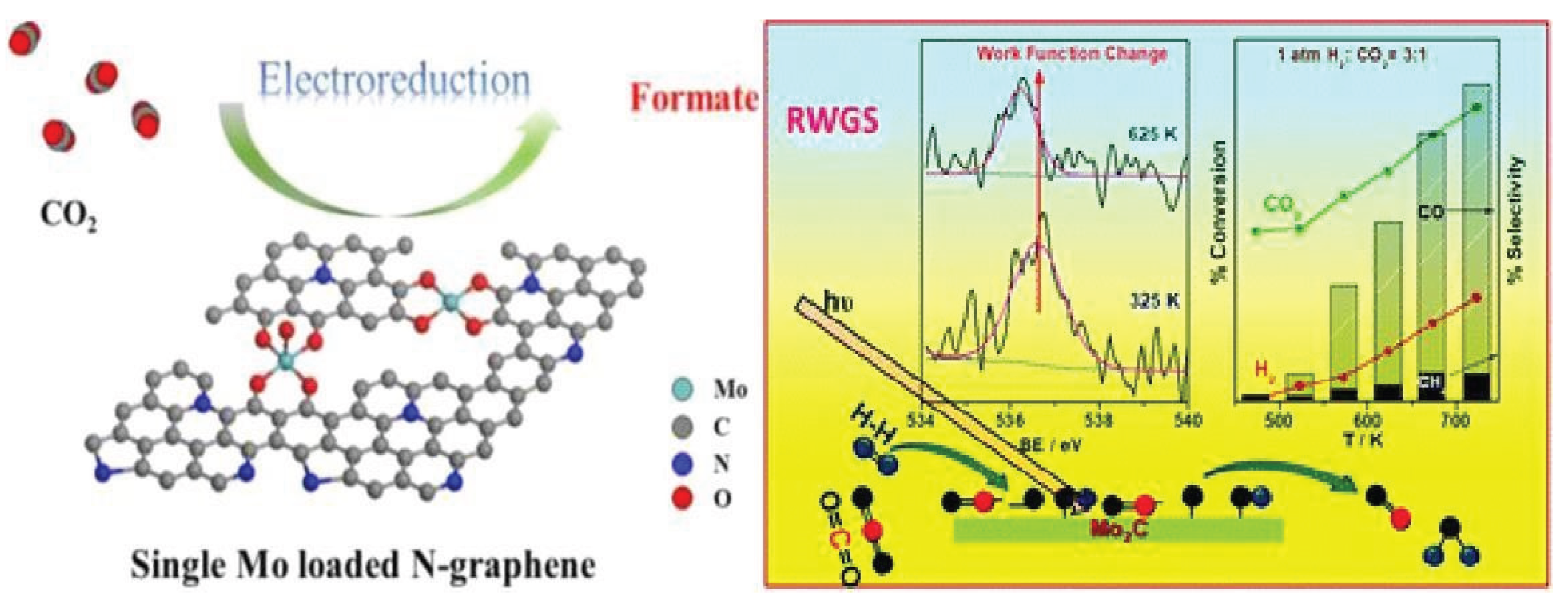
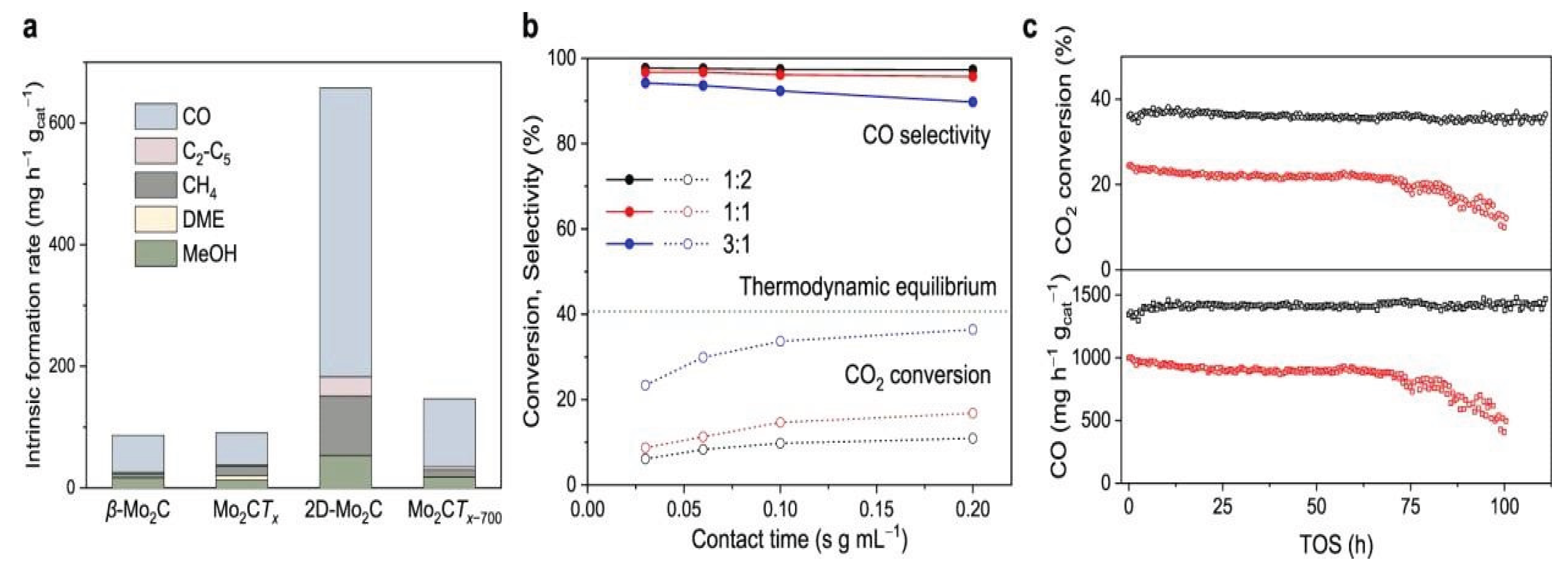
3.1.6. Molybdenum Based Single Atom Catalysts (Mo-SACs) for CO2 Reduction
3.1.7. Other Metal-Based Catalysts for CO2 Reduction
| Name of Metal Catalyst | Type of catalysts | Conversion Form | Conditions | Faradaic efficiency | References |
|---|---|---|---|---|---|
| Mo-NG | electrocatalyst | CO2 into formate | -0.96 | 100% | [81] |
| Molybdenum carbide | electrocatalyst | CO2 to CO | -0.92 | 98% | [83] |
| Mn–C3N4/CNT | electrocatalyst | CO2 to CO | 0.44 V | 98% | [85] |
| Ag2–G | electrochemical reduction | CO2 to CO | −0.25 V | 93.4% | . [87] |
3.2. Alloy and Dua Atom Catalysts: Towards Better Selectivity
4. Challenges and Future Directions
Future Directions
Conclusions
Author Contributions
Acknowledgment
Conflicts of Interest
References
- Nunes, L.J.R. The Rising Threat of Atmospheric CO2: A Review on the Causes, Impacts, and Mitigation Strategies. Environments 2023, 10, 66. [Google Scholar] [CrossRef]
- Filonchyk, M.; Peterson, M.P.; Zhang, L.; Hurynovich, V.; He, Y. Greenhouse gases emissions and global climate change: Examining the influence of CO2, CH4, and N2O, Sci. Total Environ. 2024, 935, 173359. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Singh, S. Recent Advances in Earth-Abundant Metal Catalysts for CO2 Reduction, Chem. Sus. Chem. 2020, 13, 14–33. [Google Scholar]
- Ludwig, J.R.; Schindler, C.S. Catalyst: Sustainable Catalysis. Chem 2017, 2, 313–316. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, Y.; Hong, J. Electrochemical Reduction of Carbon Dioxide: Advances and Challenges. Chem. Rev. 2020, 119, 7439–7482. [Google Scholar]
- Gao, D.; Wang, C. Copper-Based Catalysts for CO2 Electroreduction: A Review. J. CO2 Util. 2021, 49, 101580. [Google Scholar]
- Tao, L.; Zhang, S. Earth-Abundant Metal Catalysts for CO2 Reduction: Design and Mechanism. Adv. Mater. 2020, 32, 2002946. [Google Scholar]
- Wang, H.; Chen, Y. The Role of Iron and Nickel in Electrochemical CO2 Reduction, Nat. Rev. Chem. 2022, 6, 244–261. [Google Scholar]
- Kwon, Y.; Lee, J. Towards Sustainable CO2 Reduction: Innovations in Earth-Abundant Metal Catalysts. ACS Sustain Chem. & Eng. 2023, 11, 1072–1085. [Google Scholar]
- Zhang, H.; Huang, X. Review of Electrocatalytic CO2 Reduction with Copper-Based Catalysts. FrChem. 2022, 6, 185. [Google Scholar]
- Schwabe, T.; Kauffmann, K. Sustainable Pathways for CO2 Utilization: Catalytic Approaches Using Earth-Abundant Metals, Chem. Eur. J. 2022, 28, 202200045. [Google Scholar]
- Liu, X.; Zhu, Y. Mechanistic Insights into CO2 Electroreduction on Earth-Abundant Metal Catalyst, J. Am. Chem. Soc. 2021, 143, 9657–9670. [Google Scholar]
- Raghu, K.; Narayanan, R. Metallic Catalysts for CO2 Reduction: A Review of Earth-Abundant Metals. Catal. 2020, 10, 758. [Google Scholar]
- Rotundo, L.; Gobetto, R.; Nervi, C. Electrochemical CO2 reduction with earth-abundant metal catalysts,Curr. Opin. Green. Sustain. Chem. 2021, 31, 100509. [Google Scholar]
- Bui, M. Carbon capture technologies for the power sector. Nat. Energy. 2018, 3, 420–431. [Google Scholar]
- Zhou, P. Direct air capture: A technology review, Renew. Sustain. Energy Rev. 2021, 135, 110220. [Google Scholar]
- Global Status of CCS Institute, Retrieved from Global CCS Institute website 2020.
- Gielen, D. The role of carbon capture and storage in the transition to a low-carbon energy system. Energy Econ. 2020, 87, 104559. [Google Scholar]
- Barton, J.R.; Ault, G.W. The role of energy storage in the future of the energy system, Renew. Sustain. Energy Rev. 2019, 113, 109270. [Google Scholar]
- Davidson, C.I. Sustainable bioenergy and carbon capture and storage. Nat. Clim. Change. 2018, 8, 963–971. [Google Scholar]
- Schembecker, G. Carbon Capture and Utilization: An Overview. J. CO2 Util. 2020, 41, 101203. [Google Scholar]
- Call, F.; Roeb, M.; Schmücker, M.; Bru, H. Thermogravimetric Analysis of Zirconia-Doped Ceria for Thermochemical Production of Solar Fuel. American J. Analyt. Chem. 2013, 4, 37–45. [Google Scholar] [CrossRef]
- Chen, Q.; Tsiakaras, P.; Shen, P. Electrochemical Reduction of Carbon Dioxide: Recent Advances on Au-Based Nano catalysts. Catal. 2022, 12, 1348. [Google Scholar] [CrossRef]
- Lingampalli, S.R.; Ayyub, M.M.; Rao, C.N. Recent Progress in the Photocatalytic Reduction of Carbon Dioxide. ACS Omega. 2017, 6, 1348. [Google Scholar] [CrossRef] [PubMed]
- Rotundo, L.; Gobetto, R.; Nervi, C. Electrochemical CO2 reduction with earth-abundant metal catalysts, Curr. Opin. Green Sustain. 2021, 31, 100509. [Google Scholar] [CrossRef]
- Fan, M.; Luo, Z.M.; Wang, J.W.; Aramburu-Trošelj, B.M.; Ouyang, G. Earth-abundant-metal complexes as photosensitizers in molecular systems for light-driven CO2 reduction, Coord. Chem. Rev. 2024, 500, 215529. [Google Scholar]
- Palade, E.A.; Gobetto, R.; Nervi, C. Molecular and single-atom catalysts based on earth-abundant transition metals for the electroreduction of CO2 to C1. Inorganica Chimica Acta 2024, 566, 122029. [Google Scholar] [CrossRef]
- Scarpa, D.; Sarno, M. Single-Atom Catalysts for the Electro-Reduction of CO2 to Syngas with a Tunable CO/H2 Ratio: A Review. Catal. 2022, 12, 275. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Chen, J.Y.; Siegbahn, P.E.M.; Liao, R.Z. Harnessing noninnocent porphyrin ligand to circumvent Fe-hydride formation in the selective Fe-catalyzed CO2 reduction in aqueous solution. ACS Catal. 2020, 10, 1–22. [Google Scholar] [CrossRef]
- Khakpour, R.; Laasonen, K.; Busch, M. Selectivity of CO2, carbonic acid and bicarbonate electroreduction over iron-porphyrin catalyst: a DFT study, Electrochim. Acta. 2023, 442, 141784. [Google Scholar] [CrossRef]
- Wakabayashi, T.; Kamada, K.; Sekizawa, K.; Sato, S.; Morikawa, T.; Jung, J. Photocatalytic CO2 reduction using an iron by two phosphines for improving catalyst durability. Organometallics 2022, 41, 1865–1871. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, Y.; Hua, Z.; Zhang, Z. Iron-based single-atom electrocatalysts: synthetic strategies and applications. RSC Adv. 2021, 11, 3079–3095. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, S.; Wu, J.; Liu, M.; Yazdi, S.; Ren, M.; Sha, J.; Zhong, J.; Nie, K.; Jalilov, A.S.; Li, Z.; Li, H.; Yakobson, B.I.; Wu, Q.; Ringe, E.; Xu, H.; Ajayan, P.M.; Tour, J.M. Electrochemical CO2 Reduction with Atomic Iron-Dispersed on Nitrogen-Doped Graphene, Adv. Energy Mater. 2018, 8, 1703487. [Google Scholar] [CrossRef]
- Chen, H.; Guo, X.; Kong, X.; Xing, Y.; Liu, Y.; Yu, B.; Li, Q.X.; Geng, Z.; Si, R.; Zeng, J. Tuning the coordination number of Fe single atoms for the efficient reduction of CO2. Green Chem. 2020, 22, 7529. [Google Scholar] [CrossRef]
- Chen, S.; Liu, T.; Olanrele, S.O.; Lian, Z.; Si, C.; Chen, Z.; Li, B. Boosting electrocatalytic activity for CO2 reduction on nitrogen-doped carbon catalysts by co-doping with phosphorus. J. Energy Chem. 2021, 54, 143–150. [Google Scholar] [CrossRef]
- Wu, S.; Lv, X.; Ping, D.; Zhang, G.; Wang, S.; Wang, H.; Yang, X.; Guo, D.; Fang, S. Highly exposed atomic Fe–N active sites within carbon nanorods towards electrocatalytic reduction of CO2 to CO. Electrochim. Acta. 2020, 340, 135930. [Google Scholar] [CrossRef]
- Jin, Z.; Jiao, D.; Dong, Y.; Liu, L.; Fan, J.; Gong, M.; Ma, X.; Wang, Y.; Zhang, W.; Zhang, L.; Yu, Z.G.; Voiry, D.; Zheng, W.; Cui, X. Boosting Electrocatalytic Carbon Dioxide Reduction via Self-Relaxation of Asymmetric Coordination in Fe-Based Single Atom Catalyst. Angew. Chem. 2023, 136, 1–14. [Google Scholar]
- Lyu, H.; Ma, C.; Zhao, J.; Shen, B.; Tang, J. A novel one-step calcination tailored single-atom iron and nitrogen co-doped carbon material catalyst for the selective reduction of CO2 to CO. Sep. Purifi. Technol. 2022, 303, 122221. [Google Scholar] [CrossRef]
- Ren, M.; Guo, X.; Huang, S. Coordination-tuned Fe single-atom catalyst for efficient CO2 electroreduction: The power of B atom, Chem. Eng. J. 2022, 433, 134270. [Google Scholar] [CrossRef]
- Chen, S.; Li, Y.; Bu, Z.; Yang, F.; Luo, J.; An, Q.; Zeng, Z.; Wang, J.; Deng, S. Boosting CO2-to-CO conversion on a robust single-atom copper decorated carbon catalyst by enhancing intermediate binding strength. J. Mater. Chem. A 2021, 9, 1705–1712. [Google Scholar] [CrossRef]
- Xu, C.; Zhi, X.; Vasileff, A.; Wang, D.; Jin, B.; Zheng, Y.; Qiao, S.Z. Highly Selective Two-Electron Electrocatalytic CO2 Reduction on Single-Atom Cu Catalysts. Small Struct. 2020, 2, 2000058. [Google Scholar] [CrossRef]
- Cheng, H.; Wu, X.; Li, X.; Nie, X.; Fan, S.; Feng, M.; Fan, Z.; Tan, M.; Che, Y.; He, G. Construction of atomically dispersed Cu-N4 sites via engineered coordination environment for high-efficient CO2 electroreduction. Chem. Eng. J. 2021, 407, 126842. [Google Scholar] [CrossRef]
- Chen, S.; Li, Y.; Bu, Z.; Yang, F.; Luo, J.; An, Q.; Zeng, Z.; Wang, J.; Deng, S. Boosting CO2-to-CO Conversion on a Robust Single-Atom Copper Decorated Carbon Catalyst by Enhancing Intermediate Binding Strength. J. Mater. Chem. A 2021, 9, 1705–1712. [Google Scholar] [CrossRef]
- Cai, Y.; Fu, J.; Zhou, Y.; Chang, Y.C.; Min, Q.; Zhu, J.J.; Lin, Y.; Zhu, W. Insights on forming N, O-coordinated Cu single-atom catalysts for electrochemical reduction CO2 to methane. Nat. Commun. 2021, 12, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Shen, S.; Peng, X.; Bao, H.; Liu, X.; Luo, J. Selective Electroreduction of CO2 to C2 Products over Cu3N-Derived Cu Nanowires. Chem. Electro. Chem. 2019, 6, 2393–2397. [Google Scholar] [CrossRef]
- Cheng, H.; Wu, X.; Li, X.; Nie, X.; Fan, S.; Feng, M.; Fan, Z.; Tan, M.; Che, Y.; He, G.G. Construction of atomically dispersed Cu-N4 sites via engineered coordination environment for high-efficient CO2 electroreduction. Chem. Eng. J. 2021, 407, 126842. [Google Scholar] [CrossRef]
- Fu, Y.C.J.; Zhou, Y.; Chang, Y.C.; Min, Q.; Zhu, J.; Lin, Y.; Zhu, W. Insights on forming N,O-coordinated Cu single-atom catalysts for electrochemical reduction CO2 to methane. Nat. Commun. 2021, 12, 586. [Google Scholar]
- Mi, Y.; Shen, S.; Peng, X.; Bao, H.; Liu, X. Selective Electroreduction of CO2 to C2 Products over Cu3N-Derived Cu Nanowires, Chem. Electro. Chem. 2019, 6, 2393–2397. [Google Scholar]
- Zheng, H.; Jiang, K.; Ta, N.; Hu, Y.; Zeng, J.; Liu, J.; Wang, H. Large-Scale and Highly Selective CO2 Electrocatalytic Reduction on Nickel Single-Atom Catalyst Tingting. Joule 2019, 3, 1–14. [Google Scholar] [CrossRef]
- Li, N.X.; Chen, Y.M.; Xu, Q.; Mu, W.H. Photocatalytic reduction of CO2 to CO using nickel(II)-bipyridine complexes with different substituent groups as catalysts. J. CO2 Util. 2023, 68, 102385. [Google Scholar] [CrossRef]
- Li, S.; Lu, X.; Frank, S.; Lock, N.; Roldan, A.; Hu, X.M.; Daasbjerg, K. Incorporation of nickel single atoms into carbon paper as self-standing electrocatalyst for CO2 reduction. J. Mater. Chem. A 2021, 9, 1583–1592. [Google Scholar] [CrossRef]
- Pellessier, J.; Gang, Y.; Li, Y. A Sustainable Synthesis of Nickel-Nitrogen-Carbon Catalysts for Efficient Electrochemical CO2 Reduction to CO. ES Mater. Manuf. 2021, 13, 66–75. [Google Scholar] [CrossRef]
- Zheng, T.; Jiang, K.; Ta, N.; Hu, Y.; Zeng, J.; Liu, J.; Wang, H. Large-Scale and Highly Selective CO2 Electrocatalytic Reduction on Nickel Single-Atom Catalyst. Joule. 2019, 3, 265–278. [Google Scholar] [CrossRef]
- Schneider, J.; Jia, H.; Kobiro, K.; Cabelli, D.E.; Muckermana, J.T.; Fujita, E. Nickel(ii) macrocycles: highly efficient electrocatalysts for the selective reduction of CO2 to CO. Energy Environ. Sci. 2012, 5, 9502–9510. [Google Scholar] [CrossRef]
- Su, P.; Iwase, K.; Harada, T.; Kamiya, K.; Nakanishi, S. Covalent triazine framework modified with coordinatively-unsaturated Co or Ni atoms for CO2 electrochemical reduction. Chem. Sci. 2018, 9, 3941–3947. [Google Scholar] [CrossRef]
- Yang, H.B.; Hung, S.F.; Liu, S.; Yuan, K.D.; Miao, S.; Zhang, L.P.; Huang, X.; Wang, H.Y.; Cai, W.Z.; Chen, R. Atomically dispersed Ni(i) as the active site for electrochemical CO2 reduction. Nat. Energy. 2018, 3, 140–147. [Google Scholar] [CrossRef]
- Jiang, K.; Siahrostami, S.; Akey, A.J.; Li, Y. Transition-Metal Single Atoms in a Graphene Shell as Active Centers for Highly Efficient Artificial Photosynthesis. Chem. 2019, 3, 950–965. [Google Scholar] [CrossRef]
- Yuan, C.Z.; Zhan, L.Y.; Liu, S.; Chen, F.; Lin, H.; Wu, X.L.; Chen, J. Semi-sacrificial template synthesis of single-atom Ni sites supported on hollow carbon nanospheres for efficient and stable electrochemical CO2 reduction. Inorg. Chem. Front. 2020, 7, 1719–1725. [Google Scholar] [CrossRef]
- Li, X.; Bi, W.; Chen, M.; Sun, Y.; Ju, H.; Yan, W.; Zhu, J.; Wu, X.; Chu, W.; Wu, C. Exclusive Ni−N4 Sites Realize Near-Unity CO Selectivity for Electrochemical CO2 Reduction. J. Am. Chem. Soc. 2017, 139, 14889–14892. [Google Scholar] [CrossRef]
- Hou, Y.; Liang, Y.L.; Shi, P.C.; Huang, Y.B.; Cao, R. Atomically dispersed Ni species on N-doped carbon nanotubes for electroreduction of CO2 with nearly 100% CO selectivity. Appl. Catal. B Environ. 2020, 271, 11892. [Google Scholar] [CrossRef]
- Zhu, W.; Fu, J.; Liu, J.; Chen, Y.; Li, X.; Huang, K.; Cai, Y.; He, Y.; Zhou, Y.; Su, D. Tuning single atom-nanoparticle ratios of Ni-based catalysts for synthesis gas production from CO2. Appl. Catal. B Environ. 2020, 264, 118502. [Google Scholar] [CrossRef]
- Suraj Gupta, Rohan Fernandes, Rupali Patel, Matjaž Spreitzer, Nainesh Patel, A review of cobalt-based catalysts for sustainable energy and environmental applications. Appl. Catal. 2023, 661, 119254. [CrossRef]
- Usman, M.; Humayun, M.; Garba, M.D.; Ullah, L.; Zeb, Z.; Helal, A.; Suliman, M.H.; Alfaifi, B.Y.; Iqbal, N.; Abdinejad, M.; Tahir, A.A.; Ullah, H. Electrochemical Reduction of CO2: A Review of Cobalt Based Catalysts for Carbon Dioxide Conversion to Fuels. Nanomater. 2021, 11, 2029. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Cometto, C.; Ishitani, O.; Robert, M. Electrons, Photons, Protons and Earth-Abundant Metal Complexes for Molecular Catalysis of CO2 Reduction. ACS Catal. 2017, 7, 70–88. [Google Scholar] [CrossRef]
- Call, A.; Cibian, M.; Takashi, K.Y.; Yamauchi, N.K.; Sakai, K. Highly Efficient and Selective Photocatalytic CO2 Reduction to CO in Water by a Cobalt Porphyrin Molecular Catalyst. ACS Catal. 2019, 9, 4867–4874. [Google Scholar] [CrossRef]
- Wang, M.; Torbensen, K.; Salvatore, D.; Ren, S.; Joulié, D.; Dumoulin, F.; Mendoza, D.; Lassalle-Kaiser, B.; Işci, U.; Berlinguette, C.P.; Robert, M. CO2 electrochemical catalytic reduction with a highly active cobalt phthalocyanine. Nat. Commun. 2019, 10, 3602. [Google Scholar] [CrossRef]
- Hou, P.; Song, W.; Wang, X.; Hu, Z.; Kang, P. Well-Defined Single-Atom Cobalt Catalyst for Electrocatalytic Flue Gas CO2 Reduction. Small. 2020, 16, 2001896. [Google Scholar] [CrossRef]
- Fernández, S.; Franco, F.; Casadevall, C.; Martin-Diaconescu, V.; Luis, J.M.; Lloret-Fillol, J. A Unified Electro- and Photocatalytic CO2 to CO Reduction Mechanism with Aminopyridine Cobalt Complexes. J. Am. Chem. Soc. 2020, 142, 120–133. [Google Scholar] [CrossRef]
- Wang, L.; Chen, W.; Zhang, D.; Du, Y.; Qiao, S.; Wu, J.; Yin, Z. Surface strategies for catalytic CO2 reduction: from two-dimensional materials to nanoclusters to single atoms. Chem. Soc. Rev. 2019, 48, 5310–5349. [Google Scholar] [CrossRef]
- Pan, Y.; Lin, R.; Chen, Y.; Liu, S.; Zhu, W.; Cao, X.; Chen, W.; Wu, K.; Cheong, W.C.; Wang, Y. Design of Single-Atom Co–N5 Catalytic Site: A Robust Electrocatalyst for CO2 Reduction with Nearly 100% CO Selectivity and Remarkable Stability. J. Am. Chem. Soc. 2018, 140, 4218–4221. [Google Scholar] [CrossRef]
- Amal, A.; Wang, L.; Chen, W.; Zhang, D.; Du, Y.; Qiao, S.; Wu, J.; Yin, Z. Surface strategies for catalytic CO2 reduction: from two-dimensional materials to nanoclusters to single atoms. Chem. Soc. Rev. 2019, 48, 5310–5349. [Google Scholar]
- Song, X.; Zhang, H.; Yang, Y.; Zhang, B.; Zuo, M.; Cao, X.; Sun, J.; Lin, C.; Li, X.; Jiang, Z. Bifunctional Nitrogen and Cobalt Codoped Hollow Carbon for Electrochemical Syngas Production. Adv. Sci. 2018, 5, 1800177. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Jagvaral, Y. Electrochemical reduction of CO2 on graphene supported transition metals—Towards single atom catalysts. Phys. Chem. Chem. Phys. 2017, 19, 11436–11446. [Google Scholar] [CrossRef] [PubMed]
- Stamatelos, I.; Dinh, C.T.; Lehnert, W.; Shviro, M. Zn-Based Catalysts for Selective and Stable Electrochemical CO2 Reduction at High Current Densities. ACS Appl. Energy Mater. 2022, 6, 1–22. [Google Scholar] [CrossRef]
- Yang, F.; Song, P.; Liu, X.Z.; Mei, B.B.; Xing, W.; Jiang, Z.; Gu, L.; Xu, W.L. Highly efficient CO2 electroreduction on ZnN4-based single-atom catalyst. Angew. Chem. Int. Ed. 2018, 57, 12303–12307. [Google Scholar] [CrossRef]
- Stamatelos, I.; Scheepers, F.; Pasel, J.; Dinh, C.T.; Stolten, D. Ternary Zn-Ce-Ag catalysts for selective and stable electrochemical CO2 reduction at large-scale. Appl. Catal. B: Environ. Energy. 2024, 353, 124062. [Google Scholar] [CrossRef]
- Yang, F.; Song, P.; Liu, X.; Mei, B.; Xing, W.; Jiang, Z.; Gu, L.; Xu, W. Highly Efficient CO2 Electroreduction on ZnN4 -based Single-Atom Catalyst, Angew. Chem. Int. Ed. Eng. 2018, 57, 12303–12307. [Google Scholar] [CrossRef]
- Guo, M.; Li, X.; Huang, Y.; Li, L.; Li, J.; Lu, Y.; Zhang, L. CO2-Induced Fibrous Zn Catalyst Promotes Electrochemical Reduction of CO2 to CO. Catalysts 2021, 11, 477. [Google Scholar] [CrossRef]
- Guo, M.; Li, X.; Li, X.; Huang, Y.; Li, J.; Lu, Y.; Xu, Y.; Zhang, L. CO2-Induced Fibrous Zn Catalyst Promotes Electrochemical Reduction of CO2 to CO. Catalysts 2021, 11, 477. [Google Scholar] [CrossRef]
- Huang, P.; Cheng, M.; Zhang, H.; Zuo, M.; Xiao, C.; Xie, Y. Single Mo atom realized enhanced CO2 electro-reduction into formate on N-doped graphene. Nano Energy 2019, 61, 428–434. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, Z.; Kountoupi, E.; Tsoukalou, A.; Abdala, P.M.; Florian, P. Alexey Fedorov & Christoph R. Müller, Two-dimensional molybdenum carbide 2D-Mo2C as a superior catalyst for CO2 hydrogenation. Nat. Commun. 2021, 12, 5510. [Google Scholar]
- Reddy, K.P.; Dama, S.; Mhamane, N.B.; Ghosalya, M.K.; Raja, T.; Satyanarayan, C.V.; Gopinath, C.S. Molybdenum carbide catalyst for the reduction of CO2 to CO: surface science aspects by NAPPES and catalysis studies. Dalton Trans. 2019, 48, 12199–12209. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, Z.; Kountoupi, E.; Tsoukalou, A.; Abdala, P.M.; Florian, P.; Fedorov, A.; Müller, C.R. Two-dimensional molybdenum carbide 2D-Mo2C as a superior catalyst for CO2 hydrogenation. Nat. Commun. 2021, 12, 5510. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Gao, H.; Zheng, L.; Chen, Z.; Zeng, S.; Jiang, C.; Dong, H.; Liu, L.; Zhang, S.; Zhang, X. A Mn-N3 single-atom catalyst embedded in graphitic carbon nitride for efficient CO2 electroreduction. Nature Commun. 2020, 11, 4341. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Deng, W.; Justiniano, C.; Li, Y. Identification of champion transition metals centers in metal and nitrogen-codoped carbon catalysts for CO2 reduction. Appl. Catal. B Environ. 2018, 226, 463–472. [Google Scholar] [CrossRef]
- Li, Y.; Chen, C.; Cao, R.; Pan, Z.; He, H.; Zhou, K. Dual-atom Ag2/graphene catalyst for efficient electroreduction of CO2 to CO. Appl. Catal. B Environ. 2020, 268, 118747. [Google Scholar] [CrossRef]
- He, Q.; Lee, J.H.; Liu, D.; Liu, Y.; Lin, Z.; Xie, Z.; Hwang, S.; Kattel, S.; Song, L.; Chen, J.G. Accelerating CO2 Electroreduction to CO Over Pd Single-Atom Catalyst. Adv. Funct. Mater. 2020, 30, 2000407. [Google Scholar] [CrossRef]
- Podyacheva, O.Y.; Bulushev, D.A.; Suboch, A.N.; Svintsitskiy, D.A.; Lisitsyn, A.S.; Modin, E. Highly Stable Single-Atom Catalyst with Ionic Pd Active Sites Supported on N-Doped Carbon Nanotubes for Formic Acid Decomposition. Chem. sustain. Energy Environ. 2018, 11, 3724–3727. [Google Scholar] [CrossRef]
- Wijaya, D.T.; Lee, C.W. Pd–Mo bimetallic catalysts forelectrochemical reduction of carbon dioxideto carbon monoxide. J. Electroanalyt. Chem. 2024, 953, 118007. [Google Scholar] [CrossRef]
- Ahmed, M.; Tanya, T.; Subal, K.D.; Marc, F.; Mougel, V. A bioinspired molybdenum–copper molecular catalyst for CO2 electroreduction. Chem. Sci. 2020, 11, 5503. [Google Scholar]
- Wang, L.; Peng, H.; Lamaison, S.; Abild-Pederser, F.; Jaramillo, T.F.; Hahn, C. Bimetallic effects on Zn-Cu electrocatalysts enhance activity and selectivity for the conversion of CO2 to CO. Chem. Ctalyt. 2021, 1, 663–680. [Google Scholar] [CrossRef]
- Tsiotsias, A.I.; Charisiou, N.D.; Yentekakis, I.V.; Goula, M.A. Bimetallic Ni-Based Catalysts for CO2 Methanation: A Review. Nanomaterials 2021, 11, 28. [Google Scholar] [CrossRef]
- Hannah, L.A.; Symes, M.D. Recent progress in CO2 reduction using bimetallic electrodes containing copper. Electrochem. Commun. 2022, 135, 107212. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).