Submitted:
26 June 2025
Posted:
30 June 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Preparation of PTd and PTd Containing Vaccine Formulation
2.2. Assay of Neutralizing Epitopes in S1 and S2/3 Subunits
2.3. Immunization
2.4. Assay of Measuring PTx-Specific Immunoglobulins (Ig), Total IgG
2.5. Ethics Statement for Animal Experiments
3. Results
3.1. Establishment of the Neutralizing Epitopes Qualification ELISA Assay
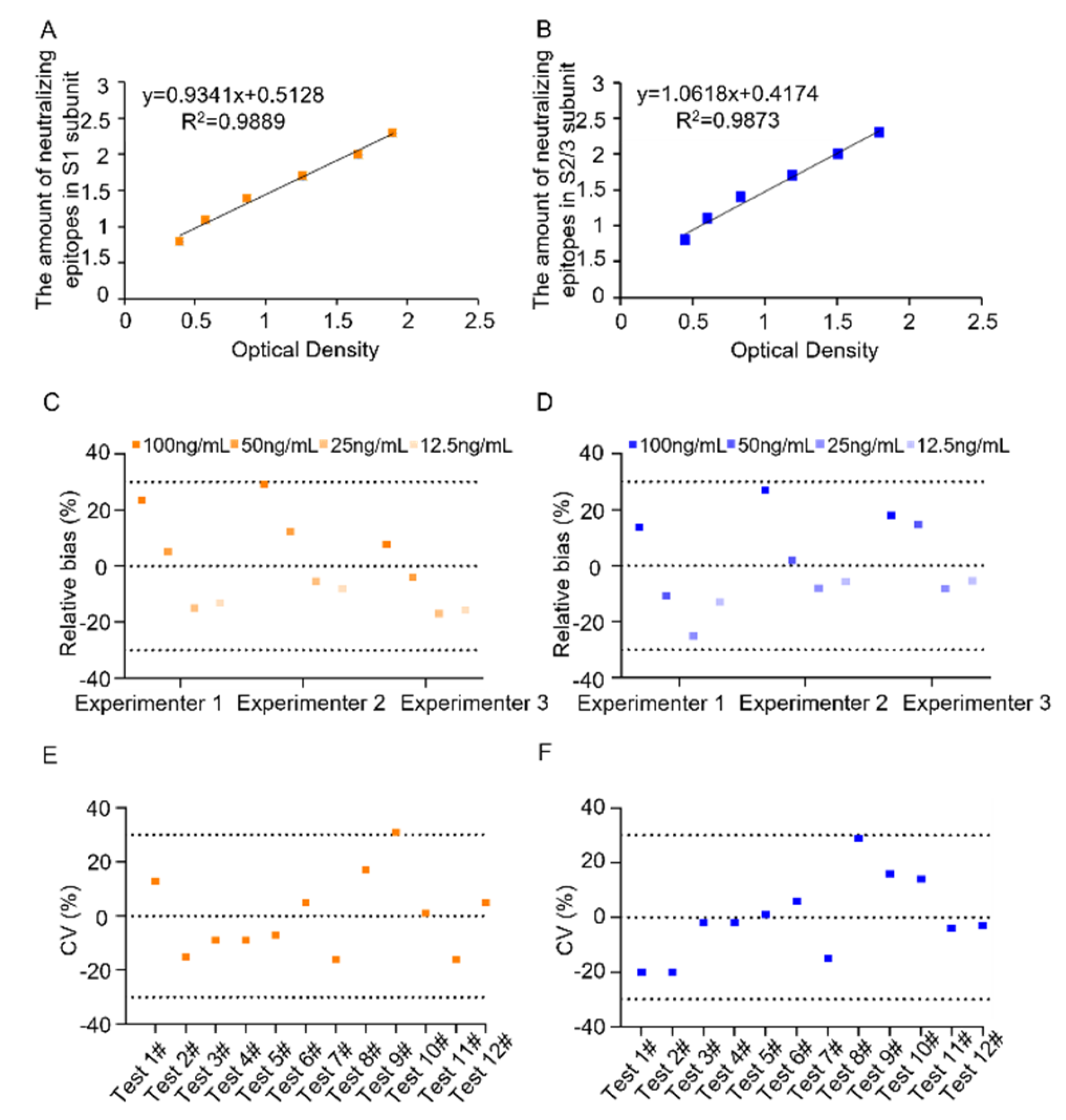
3.1.1. Working Range and Detection Limit
3.1.2. Specificity
3.1.3. Accuracy
3.1.4. Precision
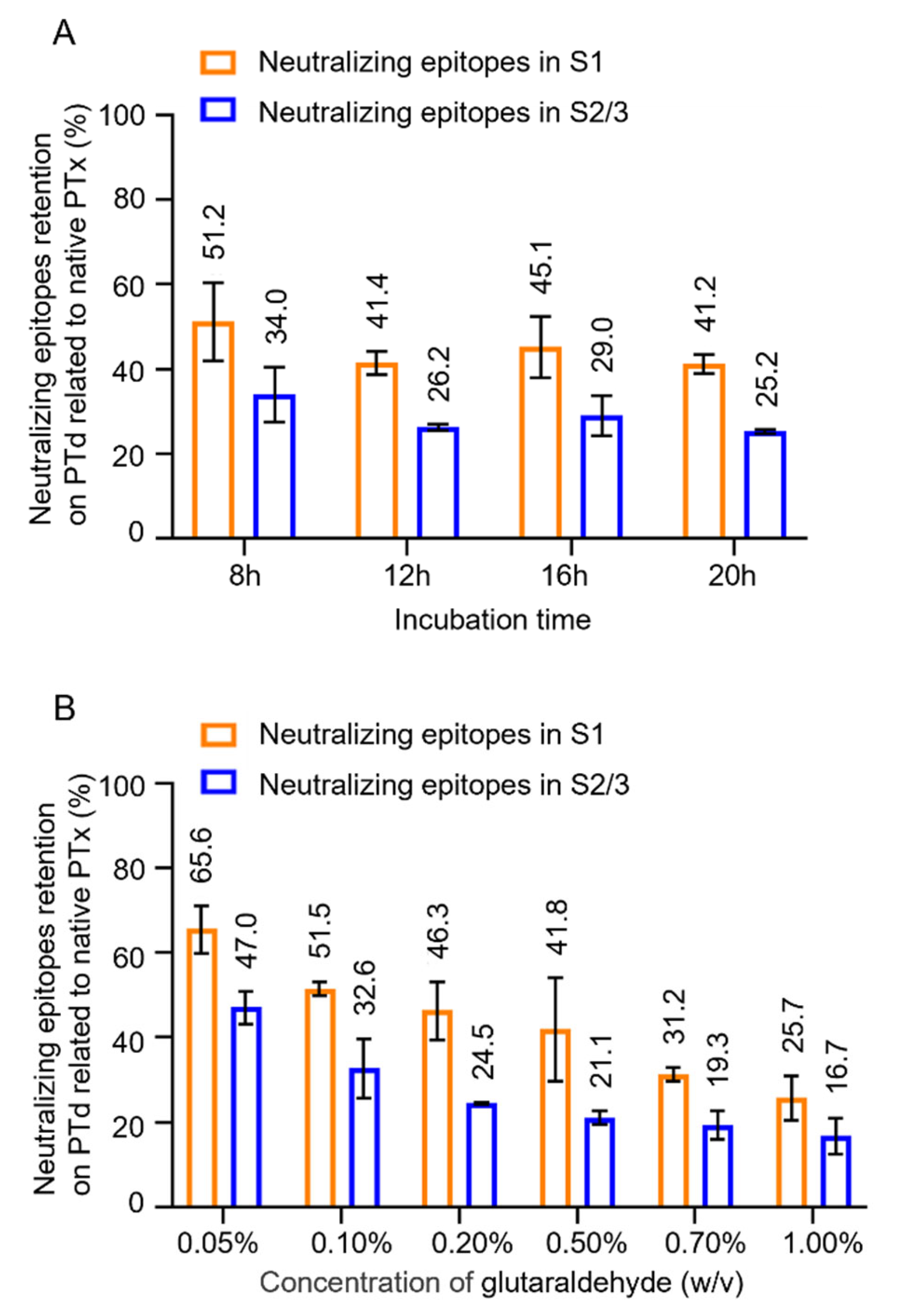
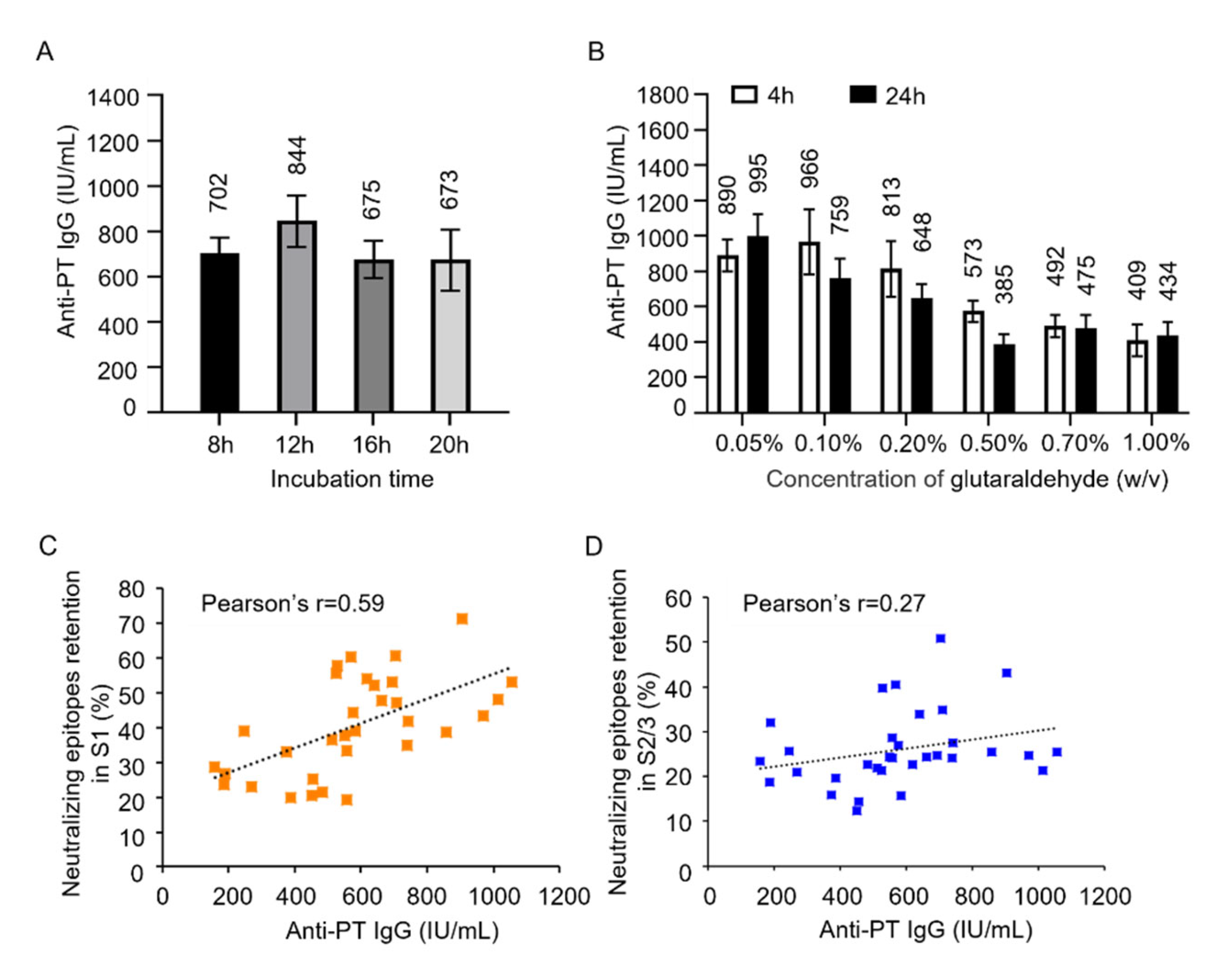
3.2. The Effect Glutaraldehyde Treatment on Antigenic Properties of PTx
3.3. Correlation Between Neutralizing Epitope Content and Immunogenicity In Vivo
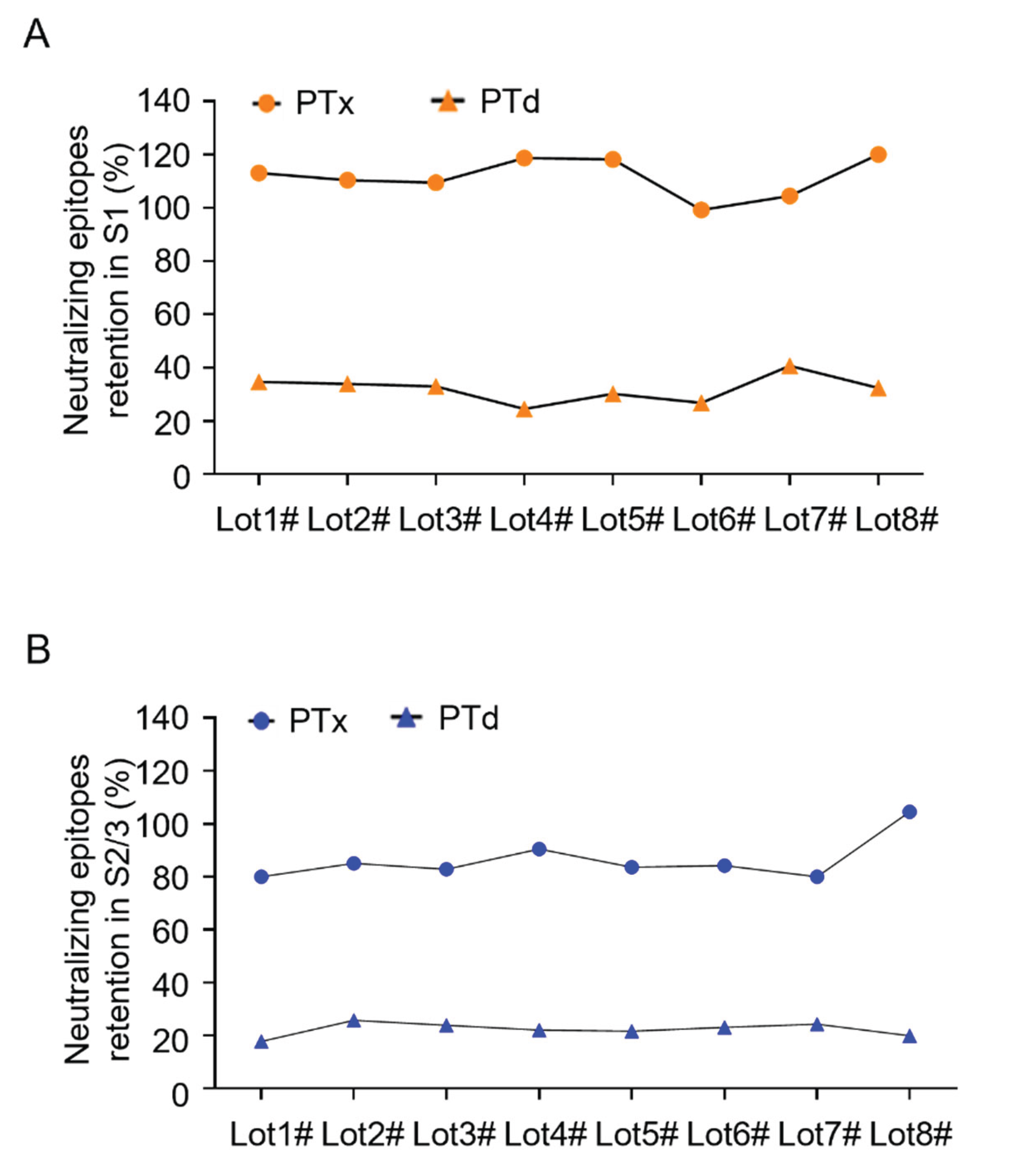
3.4. Monitoring Consistency of Neutralizing Epitopes in PTd Preparations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Esposito S, Stefanelli P, Fry NK, Fedele G, He QS, Paterson P, Tan T, Knuf M, Rodrigo C, Olivier CW, et al. Pertussis Prevention: Reasons for Resurgence, and Differences in the Current Acellular Pertussis Vaccines. Front Immunol. 2019;10: 1344. [CrossRef]
- Gregg KA, Merkel TJ. Pertussis Toxin: A Key Component in Pertussis Vaccines? Toxins.2019;11(10). [CrossRef]
- Biggelaar VD, A. H. J. , Poolman AHJ, Jan T. Predicting future trends in the burden of pertussis in the 21st century: implications for infant pertussis and the success of maternal immunization. Expert Review of Vaccines. 2016;15(1): 69-80. [CrossRef]
- Habeeb A, Hiramoto R. Reaction of proteins with glutaraldehyde. Arch Biochem Biophys.1968; 126(1): 16-26. [CrossRef]
- Richards F, Knowles JR. Glutaraldehyde as a protein cross-linking reagent. JMol Bid. 1968; 37: 231–233.
- Goldsmith JA, Nguyen AW, Wilen RE, Wijagkanalan W, McLellan JS, Maynard JA. Structural Basis for Antibody Neutralization of Pertussis Toxin. bioRxiv. 2024. [CrossRef]
- Hokyun Oh, Kim BG, Nam KT, Hong SH, Ahn DH, Choi GS, Kim H, Hong JT, Ahn BY. Characterization of the carbohydrate binding and ADP-ribosyltransferase activi-ties of chemically detoxified pertussis toxins. Vaccine. 2013; 31(29): 2988-2993. [CrossRef]
- Yuen C, Asokanathan C, Cook S. Effect of different detoxification procedures on the residual pertussis toxin activities in vaccines. Vaccine. 2016; 34(18): 2129-2134. [CrossRef]
- Knuutila A, Dalby T, Barkoff AM, Jorgensen CS, Fuursted K, Mertsola J, Markey K, He QS. Differences in epitope-specific antibodies to pertussis toxin after infection and acellular vaccinations. Clinical & Translational Immunology. 2020; 9: e1161. [CrossRef]
- Seubert A, D’Oro U, Scarselli M, Pizza M. Genetically detoxified pertussis toxin (PT-9K/129G): implications for immunization and vaccines. Expert Rev Vaccines. 2014; 13(10): 1191-1204. [CrossRef]
- Ibsen, P. The effect of formaldehyde, hydrogen peroxide and genetic detoxification of pertussis toxin on epitope recognition by murine monoclonal antibodies. Vaccine. 1996; 14(5): 359-368. [CrossRef]
- Sato H, Sato Y. Relationship between structure and biological and protective activities of pertussis toxin. Dev Biol Stand. 1991; 73: 121-132.
- Sutherland J, Maynard JA. Characterization of a key neutralizing epitope on pertussis toxin recognized by monoclonal antibody 1B7. Biochemistry. 2009; 48(50): 11982-11993. [CrossRef]
- Wagner E, Wang X, Bui A, Maynard JA. Synergistic Neutralization of Pertussis Toxin by a Bispecific Antibody In Vitro and In Vivo. Clin Vaccine Immunol. 2016; 23(11): 851-862. [CrossRef]
- Sato H, Sato Y, Ohishi I. Comparison of Pertussis Toxin (PT)-Neutralizing Activities and Mouse-Protective Activities of Anti-PT Mouse Monoclonal Antibodies. Infection and immunity. 1991, p. 3832-3835.
- Sato H, Sato Y. Protective Activities in Mice of Monoclonal Antibodies against Pertussis Toxin. Infection and immunity. 1990, p. 3369-3374.
- Tan L, Fahim RE, Jackson G, Phillips K, Wah P, Alkema D, Zobrist G, Herbert A, Boux L, Chong P. A novel process for preparing an acellular pertussis vaccine composed of non-pyrogenic toxoids of pertussis toxin and filamentous hemagglutinin. Mol Immunol. 1991 ; 28(3) : 251-255. doi : 10.1016/0161-5890(91)90070-z.
- Wu CY; Wang X, Zhou Y, Zhu XS, Ma Y, Wei WM, Zhang YT. Development and Implementation of a Single Radial Diffusion Technique for Quality Control of Acellular Pertussis Vaccines. Vaccines. 2025; 13, 116. [CrossRef]
- Winsnes R, Sesardic D, Daas A, Terao E, Behr-Gross M. Collaborative study on a Guinea pig serological method for the assay of acellu-lar pertussis vaccines. Pharmeur Bio Sci Notes. 2009 ; 1109(1) : 27-40. [PubMed]
- Zhang YT, Guo YC, Dong Y, Liu YW, Zhao YX, Yu SZ, Li SH, Wu CY, Yang BF, Li WL, et al. Safety and immunogenicity of a combined DTacP-sIPV-Hib vaccine in animal models. Hum Vaccin Immunother. 2005; 18(7): 2160158. [CrossRef]
- Schild GC, Wood JM, Newman RW. A single radial immunodiffusion technique for the assay of influenza hemagglutinin antigen: Proposals for an assay method for the haemagglutinin content of influenza vaccines. Bull WHO. 1975; 52, 223–231. [PubMed]
- Aydin S, Emre E, Ugur K, Aydin MA, Sahin B, Cinar V, Akbulut T. An overview of ELISA: a review and update on best laboratory practices for quantifying peptides and proteins in biological fluids. J Int Med Res. 2025; 53(2). [CrossRef]
- Waritani T, Chang J, McKinney B, Terato K. An ELISA protocol to improve the accuracy and reliability of serological antibody assays. MethodsX. 2017; 4:153-165. [CrossRef]
- Alhajj M, Zubair M, Farhana A. Enzyme Linked Immunosorbent Assay. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2025; Available from: https://www.ncbi.nlm.nih.gov/books/NBK555922/.
- Arciniega J, Shahin RD, Burnette WN, Bartley TD, Burns D. Contribution of the B oligomer to the protective activity of genetically attenuated pertussis toxin. Infect Immun. 1991 ; 59(10). doi : 10.1128/iai.59.10.3407-3410.1991.
- Sutherland J, Chang C, Yoder SM, Rock MT, Maynard JA. Antibodies Recognizing Protective Pertussis Toxin Epitopes Are Preferentially Elicited by Natural Infection versus Acellular Immunization. Clinical and Vaccine Immunology. 2011; 18(6): 954-962. [CrossRef]
| Detecting Abs | Samples | Concentration (ng/mL) | OD value | Cut-off value |
| 1B7 | PTx | 6.25 | 0.372 | 0.347 |
| S24 | 5000 | 0.193 | ||
| Assay buffer | N/A | 0.165 | N/A | |
| 11E6 | PTx | 6.25 | 0.353 | 0.306 |
| S24 | 6.25 | 0.358 | ||
| Assay buffer | N/A | 0.146 | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




