Submitted:
01 September 2025
Posted:
03 September 2025
You are already at the latest version
Abstract
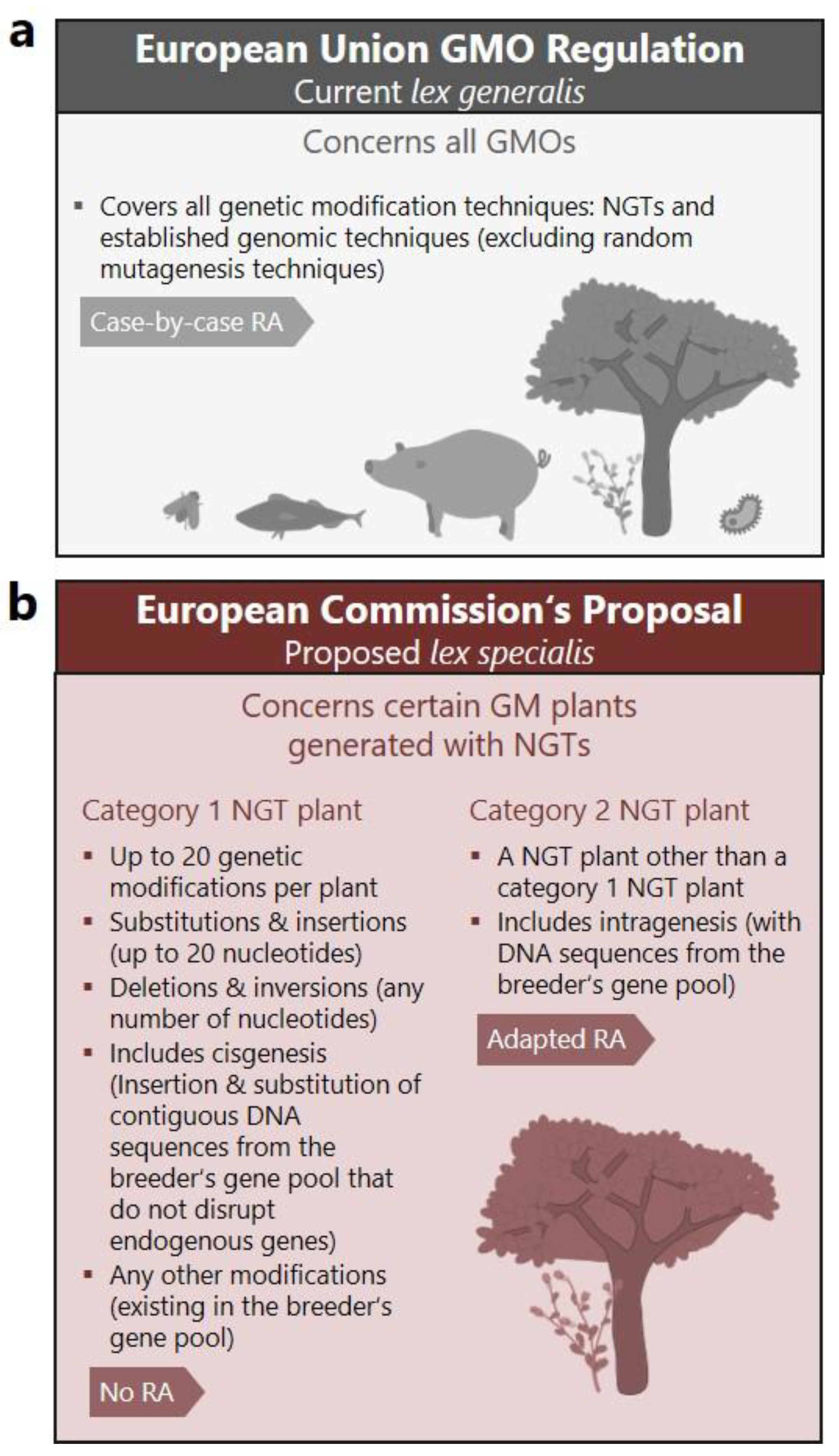
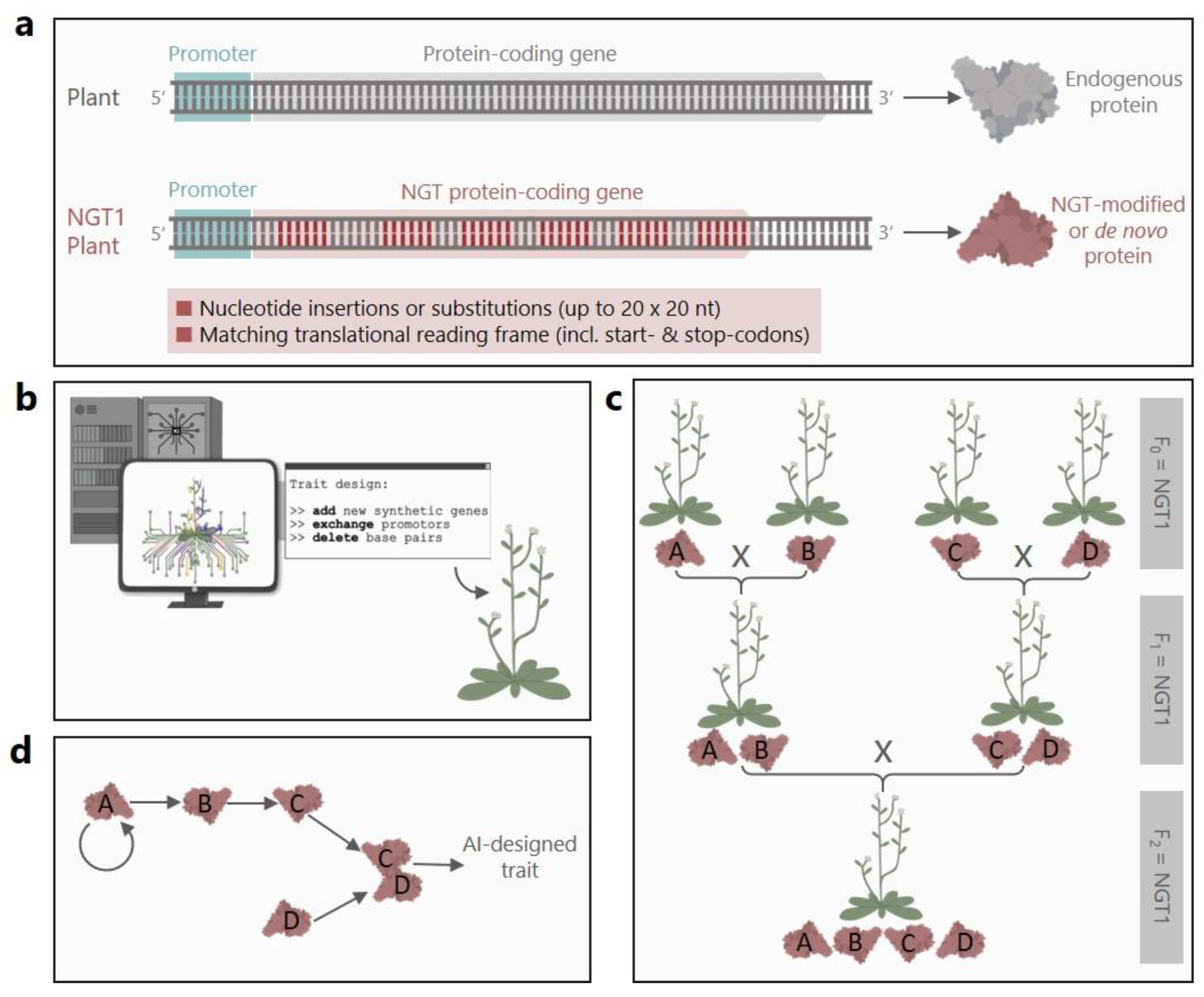
The European Commission has proposed to amend the EU GMO regulation, exempting certain genetically modified plants generated with new genomic techniques (NGTs) from risk assessment. In the suggested lex specialis so-called “category 1 NGT plants” would be treated as equivalent to conventionally bred plants, if they meet threshold-based criteria, which limit the number and size of induced genetic changes. Here, we critically analyze the scientific validity of these thresholds and show that the proposal oversimplifies genetic complexity – disregarding the biological context, mutational bias, and functional consequences. The proposal’s central claim of equivalence between NGT1 plants and conventionally bred plants is thus scientifically unfounded. Many conceivable genetic modifications produced with NGTs – including those created with CRISPR prime editing and AI-assisted design – could be highly complex and exceed the capabilities of conventional breeding. Nevertheless, the regulatory proposal treats all possible genetic changes as equally likely and overlooks the purpose and function of genetic edits. By eliminating case-by-case risk assessment, the proposal creates a regulatory gap that allows complex and novel traits to bypass scrutiny – undermining the EU’s legally binding precautionary principle. In contrast, a risk-based regulatory approach is needed to ensure safe and future-proof oversight of NGT plants.
Keywords:
Background
Beyond Thresholds: Why Genetic Context and Function Matters
NGTs: From Targeted Errors to AI-Driven Design
Risks: Within the Scope of a Simplified Regulation
Summary and Outlook
Supplementary Materials
Author Contributions
Funding
Ethics Approval and Consent to Participate
Consent for Publication
Availability of Data and Material
Acknowledgments
Conflicts of Interest
List of Abbreviations
| AI | Artificial intelligence |
| ANSES | Agence Nationale de Sécurité Sanitaire |
| Cas | CRISPR-associated system |
| COM | European Commission |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| DSB | Double-stranded breaks |
| EFSA | European Food Safety Authority |
| RA | Risk assessment |
| GM | Genetically modified |
| GMO | Genetically modified organism |
| miRNA | Micro RNA |
| NGT | New genomic technique |
| NGT1 | Category 1 NGT |
| NGT2 | Category 2 NGT |
| nt | Nucleotide |
| SDN1 | Site-directed nucleases 1 |
References
- European Commission. Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on plants obtained by certain new genomic techniques and their food and feed, and amending Regulation (EU) 2017/625: COM(2023) 411 final; 2023.
- AGENCE NATIONALE DE SÉCURITÉ SANITAIRE (ANSES) de l’alimentation, de l’environnement et du travail. OPINION of the French Agency for Food, Environmental and Occupational Health & Safety on the scientific analysis of Annex I of the European Commission’s Proposal for a Regulation of 5 July 2023 on new genomic techniques (NGTs): Review of the proposed equivalence criteria for defining category 1 NGT plants; 2023.
- European Commission. ANNEXES to the Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on plants obtained by certain new genomic techniques and their food and feed, and amending Regulation (EU) 2017/625: COM(2023) 411 final ANNEXES 1 to 3; 2023.
- European Commission. Regulation on new genomic techniques (NGT)—Technical paper on the rationale for the equivalence criteria in Annex I: 2023/0226(COD), 14204/23; 2023.
- Ossowski S, Schneeberger K, Lucas-Lledó JI, Warthmann N, Clark RM, Shaw RG, et al. The rate and molecular spectrum of spontaneous mutations in Arabidopsis thaliana. Science. 2010;327:92–4. [CrossRef]
- Weng M-L, Becker C, Hildebrandt J, Neumann M, Rutter MT, Shaw RG, et al. Fine-grained analysis of spontaneous mutation spectrum and frequency in Arabidopsis thaliana. Genetics. 2019;211:703–14. [CrossRef]
- Quiroz D, Lensink M, Kliebenstein DJ, Monroe JG. Causes of mutation rate variability in plant genomes. Annu Rev Plant Biol. 2023;74:751–75. [CrossRef]
- Szurman-Zubrzycka M, Kurowska M, Till BJ, Szarejko I. Is it the end of TILLING era in plant science? Front Plant Sci. 2023;14:1160695. [CrossRef]
- Otto SP, Payseur BA. Crossover interference: shedding light on the evolution of recombination. Annual Review of Genetics. 2019;53:19–44. [CrossRef]
- Epstein R, Sajai N, Zelkowski M, Zhou A, Robbins KR, Pawlowski WP. Exploring impact of recombination landscapes on breeding outcomes. Proc Natl Acad Sci U S A. 2023;120:e2205785119. [CrossRef]
- Chen Y-Y, Schreiber M, Bayer MM, Dawson IK, Hedley PE, Lei L, et al. The evolutionary patterns of barley pericentromeric chromosome regions, as shaped by linkage disequilibrium and domestication. Plant J. 2022;111:1580–94. [CrossRef]
- Des Danguy Déserts A, Bouchet S, Sourdille P, Servin B. Evolution of recombination landscapes in diverging populations of bread wheat. Genome Biol Evol 2021. [CrossRef]
- Bennett EP, Petersen BL, Johansen IE, Niu Y, Yang Z, Chamberlain CA, et al. INDEL detection, the ‘Achilles heel’ of precise genome editing: a survey of methods for accurate profiling of gene editing induced indels. Nucleic Acids Res. 2020;48:11958–81. [CrossRef]
- Cardi T, Murovec J, Bakhsh A, Boniecka J, Bruegmann T, Bull SE, et al. CRISPR/Cas-mediated plant genome editing: outstanding challenges a decade after implementation. Trends Plant Sci. 2023;28:1144–65. [CrossRef]
- Bhuyan SJ, Kumar M, Ramrao Devde P, Rai AC, Mishra AK, Singh PK, Siddique KHM. Progress in gene editing tools, implications and success in plants: a review. Front Genome Ed. 2023;5:1272678. [CrossRef]
- Abdelrahman M, Wei Z, Rohila JS, Zhao K. Multiplex genome-editing technologies for revolutionizing plant biology and crop Improvement. Front Plant Sci. 2021;12:721203. [CrossRef]
- Nei M, Rooney AP. Concerted and birth-and-death evolution of multigene families. Annual Review of Genetics. 2005:121–52. [CrossRef]
- Sánchez-León S, Gil-Humanes J, Ozuna CV, Giménez MJ, Sousa C, Voytas DF, Barro F. Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnol J. 2018;16:902–10. [CrossRef]
- Li J, Ding J, Zhu J, Xu R, Gu D, Liu X, et al. Prime editing-mediated precise knockin of protein tag sequences in the rice genome. Plant Commun. 2023;4:100572. [CrossRef]
- Yasmeen E, Wang J, Riaz M, Zhang L, Zuo K. Designing artificial synthetic promoters for accurate, smart, and versatile gene expression in plants. Plant Commun. 2023;4:100558. [CrossRef]
- Bravo-Vázquez LA, Méndez-García A, Chamu-García V, Rodríguez AL, Bandyopadhyay A, Paul S. The applications of CRISPR/Cas-mediated microRNA and lncRNA editing in plant biology: shaping the future of plant non-coding RNA research. Planta. 2023;259:32. [CrossRef]
- Bohle F, Schneider R, Mundorf J, Zühl L, Simon S, Engelhard M. Where does the EU-path on new genomic techniques lead us? Front. Genome Ed. 2024. [CrossRef]
- Winnifrith A, Outeiral C, Hie BL. Generative artificial intelligence for de novo protein design. Curr Opin Struct Biol. 2024;86:102794. [CrossRef]
- Zhang D, Xu F, Wang F, Le L, Pu L. Synthetic biology and artificial intelligence in crop improvement. Plant Commun. 2024:101220. [CrossRef]
- Hayes T, Rao R, Akin H, Sofroniew NJ, Oktay D, Lin Z, et al. Simulating 500 million years of evolution with a language model. Science. 2025:eads0018. [CrossRef]
- Huang P-S, Boyken SE, Baker D. The coming of age of de novo protein design. Nature. 2016;537:320–7. [CrossRef]
- EFSA Panel on Genetically Modified Organisms. Scientific opinion on the ANSES analysis of Annex I of the EC proposal COM (2023) 411 (EFSA-Q-2024-00178). EFSA Journal 2024. [CrossRef]
- Eckerstorfer MF, Grabowski M, Lener M, Engelhard M, Simon S, Dolezel M, et al. Biosafety of genome editing applications in plant breeding: Considerations for a focused case-specific risk assessment in the EU. BioTech 2021. [CrossRef]
- European Court of Justice. Judgement of the Court (Grand Chamber) in Case C-528/16. 2018, July 25.
- European Parliament and Council of the European Union. Directive 2001/18/EC on the deliberate release into the environment of genetically modified organisms and repealing Council Directive 90/220/EEC; 2001.
- Parisi C, Rodríguez-Cerezo E. Current and future market applications of new genomic techniques. Publications Office of the European Union 2001. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



