Submitted:
09 June 2025
Posted:
09 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Fabrication of Flexible Microelectrode Arrays (fMEAs)
Substrate Preparation
Zinc Oxide Nanorods Growth
Photolithography and Metal Deposition
Device Assembly and Connectivity
2.2. Devices Characterization
DMA Characterization
Thermogravimetric Analysis
SEM Characterization
EIS Characterization
2.3. System Testing
Biological Samples Preparation and Perfusion
Cell Culture Preparation for In Vitro Biocompatibility Testing
Recording Setup and Procedure
Signal Acquisition and Performance Evaluation
3. Results
3.1. Nanostructure Morphology and Characterization
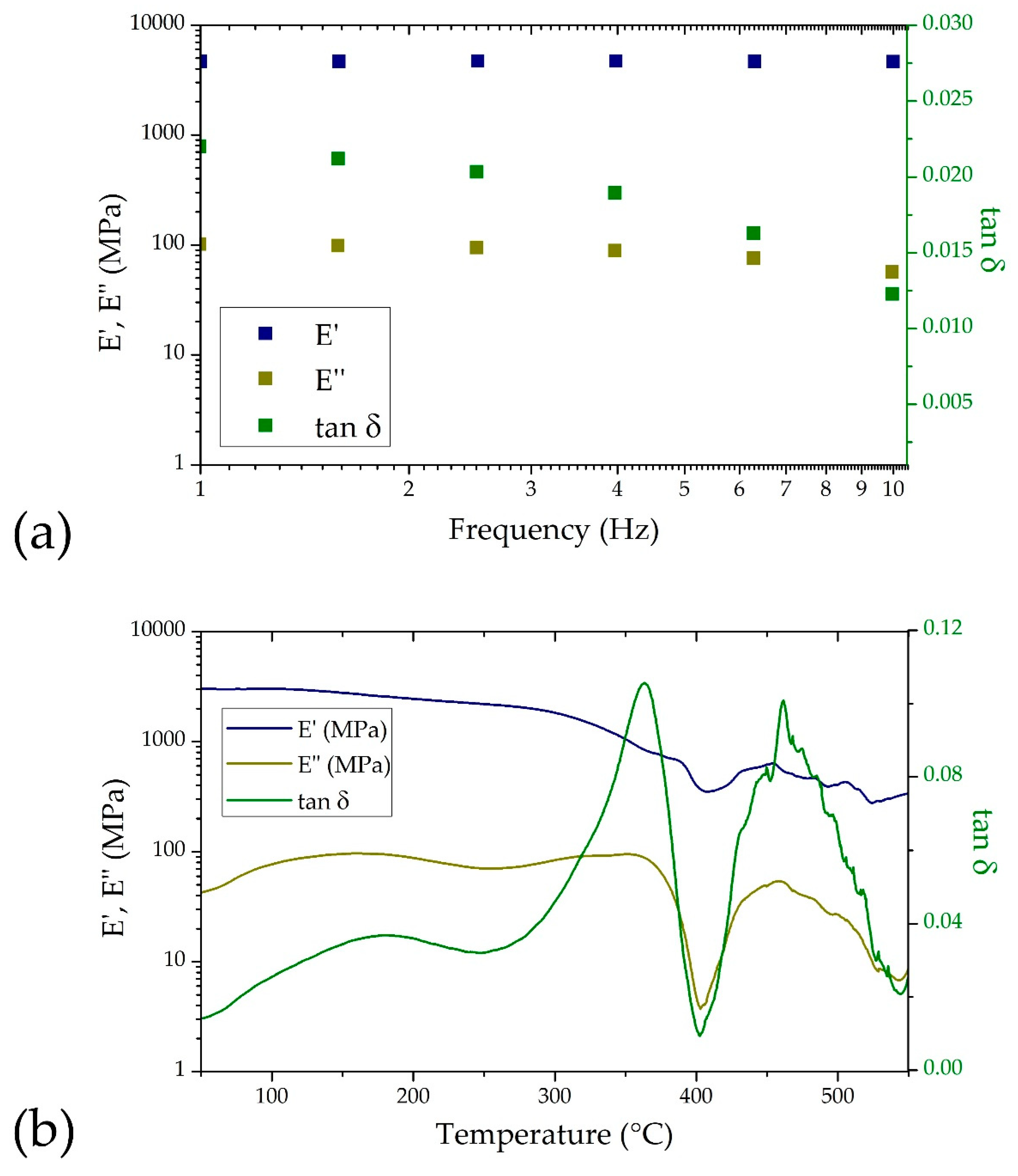
3.2. Dynamic Mechanical Analysis
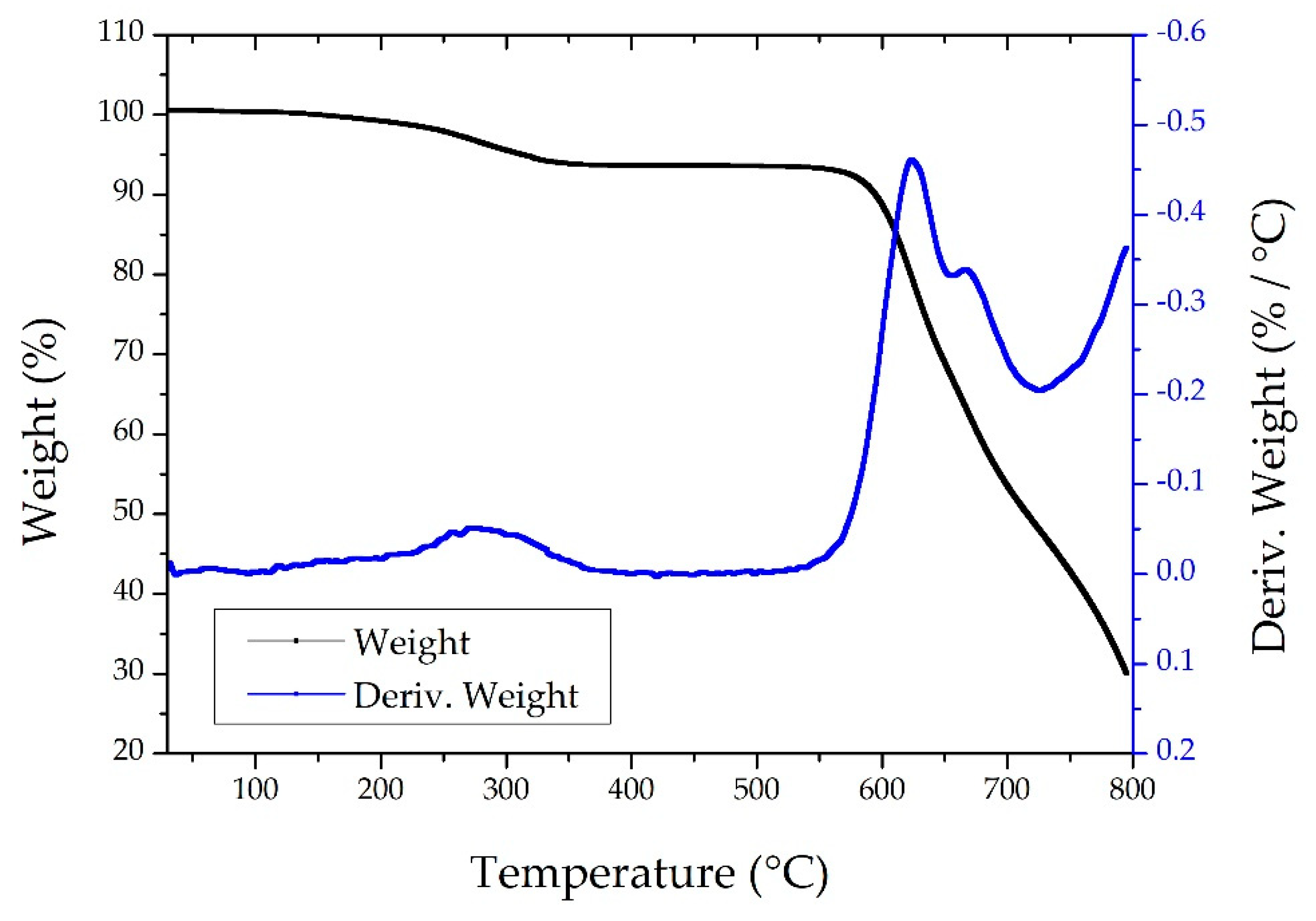
3.3. Thermal Characterization
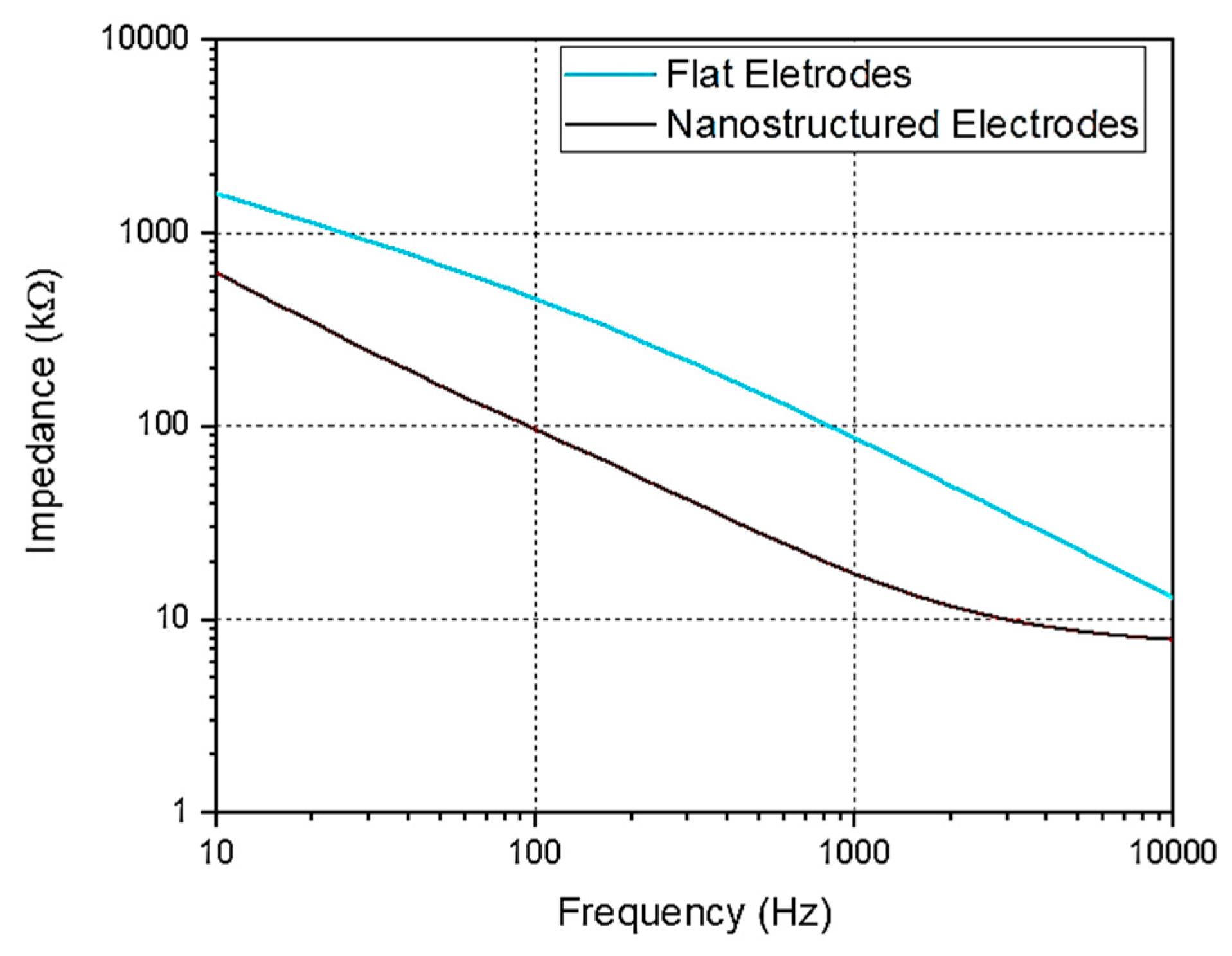
3.4. Electric Characterization
3.5. Device Design
3.6. Assessment of Nano-fMEA Biocompatibility in Neural Cells
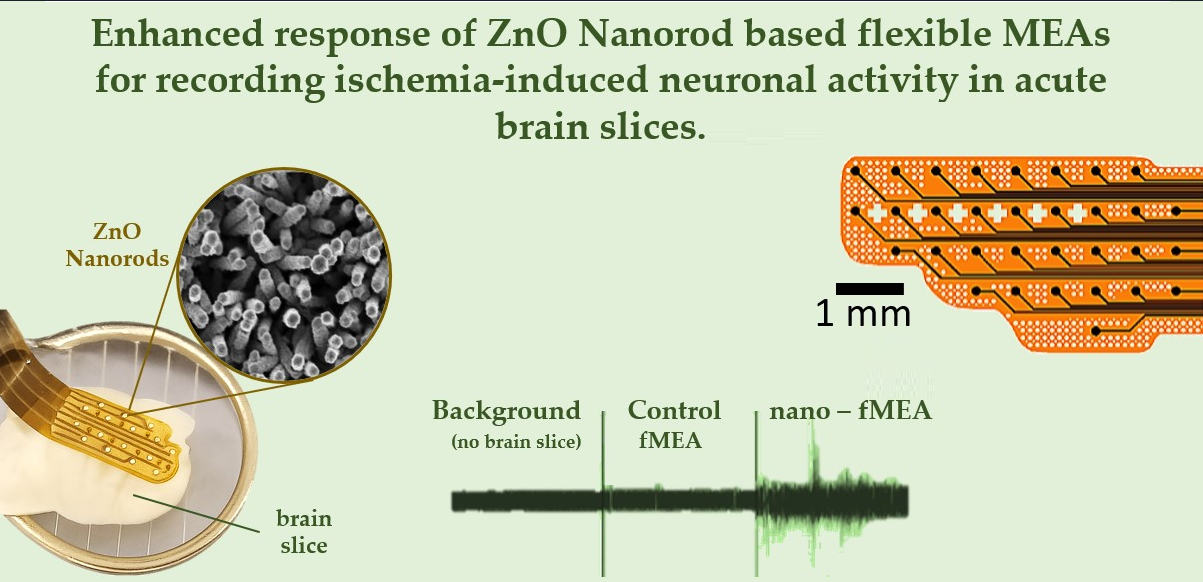
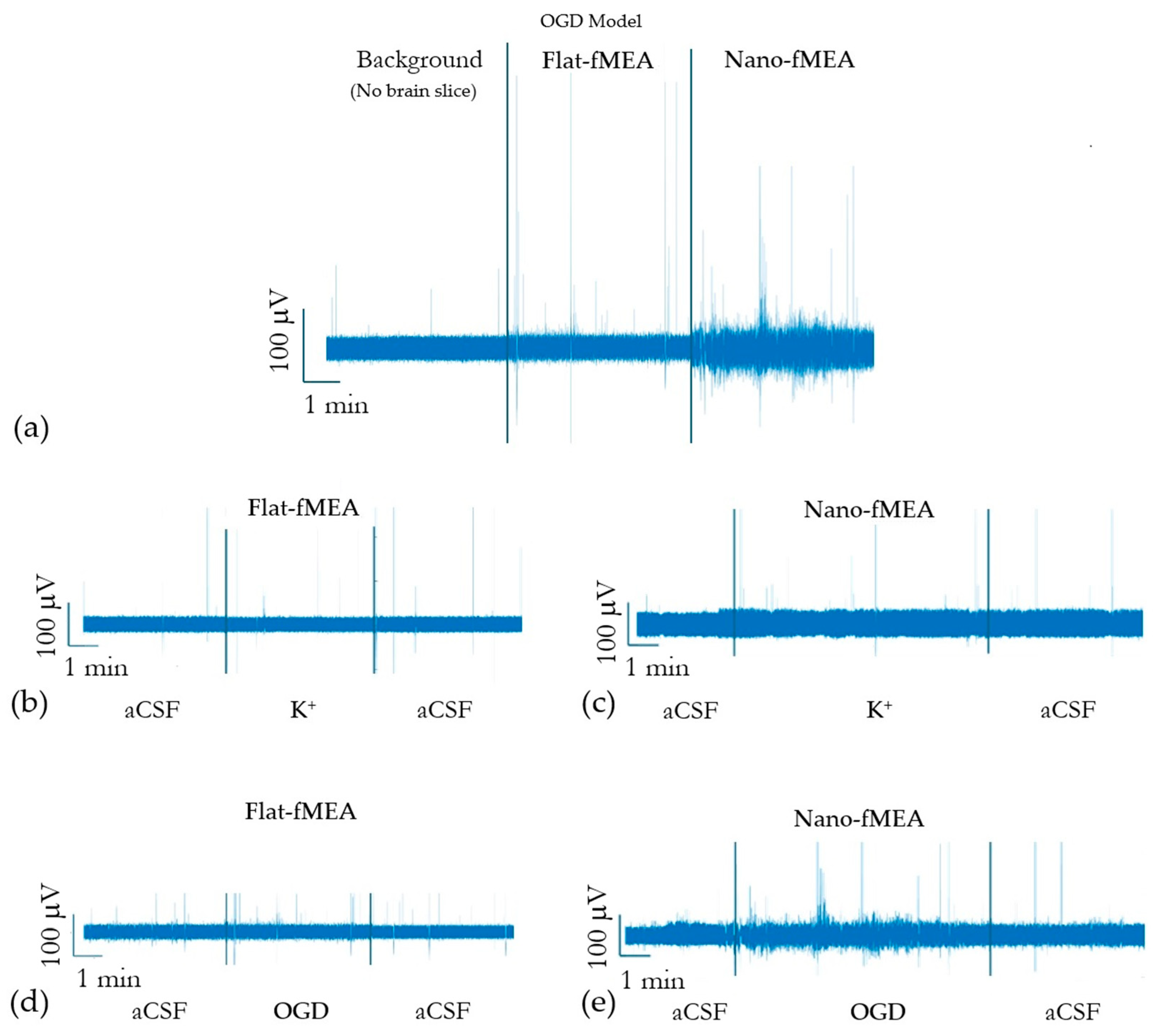
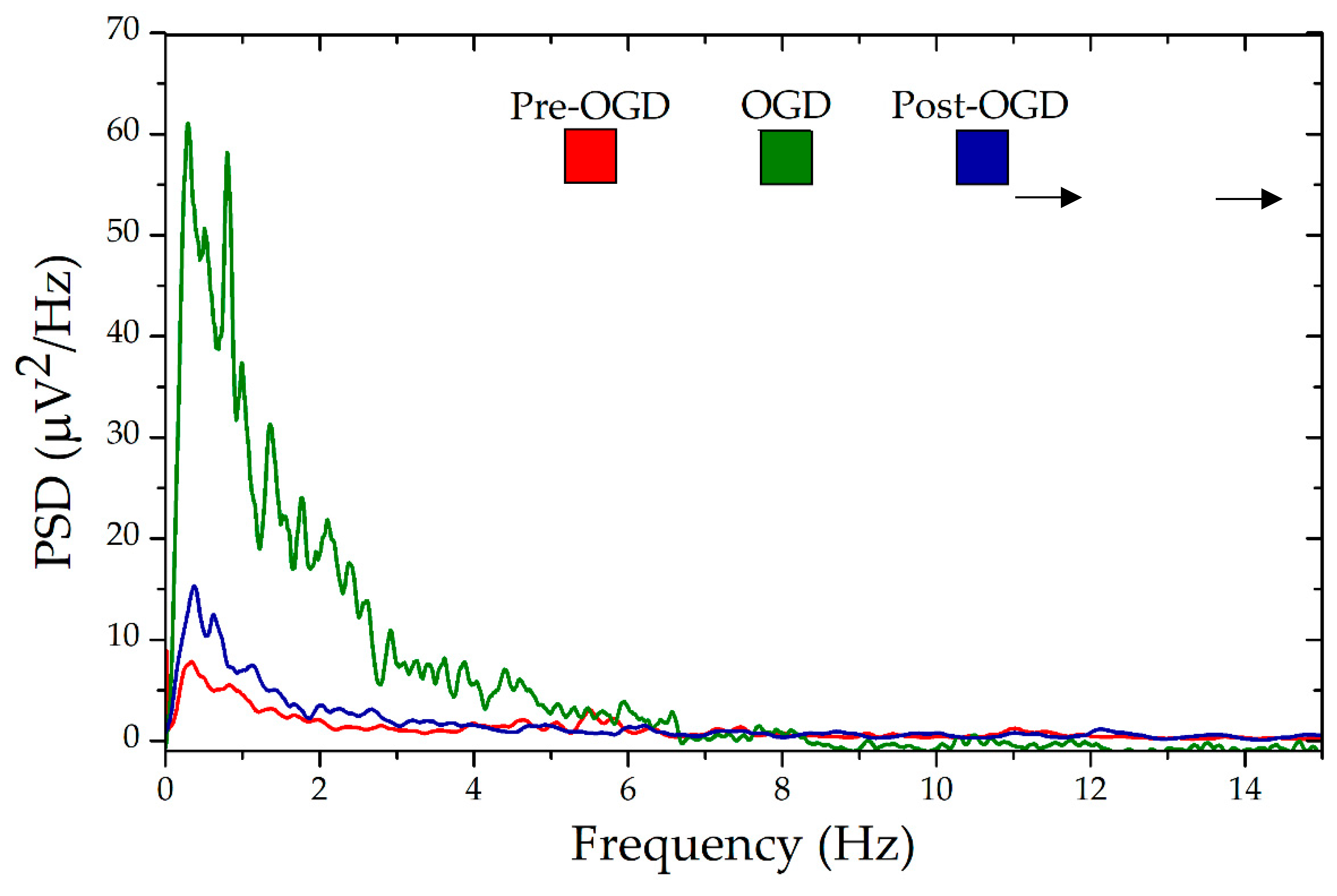
3.7. Signal Acquisition with Nano-fMEAs in Acute Brain Slices
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Abbreviations
| MEAs | Microelectrode arrays |
| fMEAs | flexible MEAs |
| nano-fMEAs | nanostructured flexible MEAs |
| ZnO NRs | Zinc Oxide Nanorods |
| SD | Spreading depolarization |
| OGD | Oxygen-glucose deprivation |
| PCB | Printed circuit board |
| DMA | Dynamic mechanical analysis |
| TGA | Thermogravimetric analysis |
| SEM | Scanning electron microscopy |
| EIS | Electrochemical impedance spectroscopy |
| DAPI | 4′,6-diamidino-2-phenylindole |
| GFAP | Glial Fibrillary Acidic Protein |
References
- X. Tang, H. Shen, S. Zhao, N. Li, J. Liu, Flexible brain–computer interfaces, Nat. Electron. 6 (2023) 109–118. [CrossRef]
- T. Someya, Z. Bao, G.G. Malliaras, The rise of plastic bioelectronics, Nature 540 (2016) 379–385. [CrossRef]
- I.R. Minev, P. Musienko, A. Hirsch, Q. Barraud, N. Wenger, E.M. Moraud, J. Gandar, M. Capogrosso, T. Milekovic, L. Asboth, R.F. Torres, N. Vachicouras, Q. Liu, N. Pavlova, S. Duis, A. Larmagnac, J. Vörös, S. Micera, Z. Suo, G. Courtine, S.P. Lacour, Electronic dura mater for long-term multimodal neural interfaces, Science 347 (2015) 159–163. [CrossRef]
- S.P. Lacour, G. Courtine, J. Guck, Materials and technologies for soft implantable neuroprostheses, Nat. Rev. Mater. 1 (2016) 1–14. [CrossRef]
- Z. Fan, J.G. Lu, Zinc Oxide Nanostructures: Synthesis and Properties, J. Nanosci. Nanotechnol. 5 (2005) 1561–1573. [CrossRef]
- A. Rinaldi, M. Pea, A. Notargiacomo, E. Ferrone, S. Garroni, L. Pilloni, R. Araneo, A Simple Ball Milling and Thermal Oxidation Method for Synthesis of ZnO Nanowires Decorated with Cubic ZnO2 Nanoparticles, Nanomaterials 11 (2021) 475. [CrossRef]
- M. Carofiglio, S. Barui, V. Cauda, M. Laurenti, Doped Zinc Oxide Nanoparticles: Synthesis, Characterization and Potential Use in Nanomedicine, Appl. Sci. 10 (2020) 5194. [CrossRef]
- M. Maddah, C.P. Unsworth, G.J. Gouws, N.O.V. Plank, Synthesis of encapsulated ZnO nanowires provide low impedance alternatives for microelectrodes, PLOS ONE 17 (2022) e0270164. [CrossRef]
- F. Maita, L. Maiolo, I. Lucarini, I. Del Rio De Vicente, E. Palmieri, E. Fiorentini, V. Mussi, Low cost and label free Raman sensors based on Ag-coated ZnO nanorods for monitoring astronaut’s health, in: 2023 IEEE 10th Int. Workshop Metrol. Aerosp. MetroAeroSpace, 2023: pp. 363–367. [CrossRef]
- D. Sudha, E.R. Kumar, S. Shanjitha, A.M. Munshi, G.A.A. Al-Hazmi, N.M. El-Metwaly, S.J. Kirubavathy, Structural, optical, morphological and electrochemical properties of ZnO and graphene oxide blended ZnO nanocomposites, Ceram. Int. 49 (2023) 7284–7288. [CrossRef]
- J.I. Del Río De Vicente, I. Lucarini, F. Maita, D. Salvò, V. Marchetti, M. Anderova, J. Gómez, L. Maiolo, Development of ZnO NRs-rGO Low-Impedance Electrodes for Astrocyte Cell Signal Recording, in: 2023 IEEE Sens., 2023: pp. 1–4. [CrossRef]
- M. Wang, G. Mi, D. Shi, N. Bassous, D. Hickey, T.J. Webster, Nanotechnology and Nanomaterials for Improving Neural Interfaces, Adv. Funct. Mater. 28 (2018) 1700905. [CrossRef]
- B.L. Rodilla, A. Arché-Núñez, S. Ruiz-Gómez, A. Domínguez-Bajo, C. Fernández-González, C. Guillén-Colomer, A. González-Mayorga, N. Rodríguez-Díez, J. Camarero, R. Miranda, E. López-Dolado, P. Ocón, M.C. Serrano, L. Pérez, M.T. González, Flexible metallic core–shell nanostructured electrodes for neural interfacing, Sci. Rep. 14 (2024) 3729. [CrossRef]
- E. Saracino, L. Maiolo, D. Polese, M. Semprini, A.I. Borrachero-Conejo, J. Gasparetto, S. Murtagh, M. Sola, L. Tomasi, F. Valle, L. Pazzini, F. Formaggio, M. Chiappalone, S. Hussain, M. Caprini, M. Muccini, L. Ambrosio, G. Fortunato, R. Zamboni, A. Convertino, V. Benfenati, A Glial-Silicon Nanowire Electrode Junction Enabling Differentiation and Noninvasive Recording of Slow Oscillations from Primary Astrocytes, Adv. Biosyst. 4 (2020) 1900264. [CrossRef]
- J. Kriska, Z. Hermanova, T. Knotek, J. Tureckova, M. Anderova, On the Common Journey of Neural Cells through Ischemic Brain Injury and Alzheimer’s Disease, Int. J. Mol. Sci. 22 (2021) 9689. [CrossRef]
- D. Belov Kirdajova, J. Kriska, J. Tureckova, M. Anderova, Ischemia-Triggered Glutamate Excitotoxicity From the Perspective of Glial Cells, Front. Cell. Neurosci. 14 (2020) 51. [CrossRef]
- Y. Du, W. Wang, A.D. Lutton, C.M. Kiyoshi, B. Ma, A.T. Taylor, J.W. Olesik, D.M. McTigue, C.C. Askwith, M. Zhou, Dissipation of transmembrane potassium gradient is the main cause of cerebral ischemia-induced depolarization in astrocytes and neurons, Exp. Neurol. 303 (2018) 1–11. [CrossRef]
- A. Menyhárt, D. Zölei-Szénási, T. Puskás, P. Makra, O. M. Tóth, B. Szepes, R. Tóth, O. Ivánkovits-Kiss, T. Obrenovitch, F. Bari, E. Farkas, Spreading depolarization remarkably exacerbates ischemia-induced tissue acidosis in the young and aged rat brain, Sci. Rep. 7 (2017). [CrossRef]
- X.-Y. Shen, Z.-K. Gao, Y. Han, M. Yuan, Y.-S. Guo, X. Bi, Activation and Role of Astrocytes in Ischemic Stroke, Front. Cell. Neurosci. 15 (2021) 755955. [CrossRef]
- L. Maiolo, S. Mirabella, F. Maita, A. Alberti, A. Minotti, V. Strano, A. Pecora, Y. Shacham-Diamand, G. Fortunato, Flexible pH sensors based on polysilicon thin film transistors and ZnO nanowalls, Appl. Phys. Lett. 105 (2014) 093501. [CrossRef]
- Luca Maiolo, Vincenzo Guarino, Emanuela Saracino, Annalisa Convertino, Manuela Melucci, Michele Muccini, Luigi Ambrosio, Roberto Zamboni, Valentina Benfenati, Glial Interfaces: Advanced Materials and Devices to Uncover the Role of Astroglial Cells in Brain Function and Dysfunction, Adv. Healthcare Mater., 2021, 10 (1), 2001268. https://advanced.onlinelibrary.wiley.com/doi/full/10.1002/adhm.202001268.
- E. Castagnola, L. Maiolo, E. Maggiolini, A. Minotti, M. Marrani, F. Maita, A. Pecora, G.N. Angotzi, A. Ansaldo, M. Boffini, L. Fadiga, G. Fortunato, D. Ricci, PEDOT-CNT-Coated Low-Impedance, Ultra-Flexible, and Brain-Conformable Micro-ECoG Arrays, IEEE Trans. Neural Syst. Rehabil. Eng. Publ. IEEE Eng. Med. Biol. Soc. 23 (2015) 342–350. [CrossRef]
- E.G. Barbagiovanni, V. Strano, G. Franzò, I. Crupi, S. Mirabella, Photoluminescence transient study of surface defects in ZnO nanorods grown by chemical bath deposition, Appl. Phys. Lett. 106 (2015) 093108. [CrossRef]
- V. Strano, R.G. Urso, M. Scuderi, K.O. Iwu, F. Simone, E. Ciliberto, C. Spinella, S. Mirabella, Double Role of HMTA in ZnO Nanorods Grown by Chemical Bath Deposition, J. Phys. Chem. C 118 (2014) 28189–28195. [CrossRef]
- Gyu-Chul Yi, Chunrui Wang and Won Il Park, ZnO nanorods: synthesis, characterization and applications, Semicond. Sci. Technol. 20 (2005) S22–S34. [CrossRef]
- L. Pazzini, D. Polese, J.F. Weinert, L. Maiolo, F. Maita, M. Marrani, A. Pecora, M.V. Sanchez-Vives, G. Fortunato, An ultra-compact integrated system for brain activity recording and stimulation validated over cortical slow oscillations in vivo and in vitro, Sci. Rep. 8 (2018) 16717. [CrossRef]
- Corish, P., & Tyler-Smith, C. (1999). Attenuation of green fluorescent protein half-life in mammalian cells. Protein engineering, 12(12), 1035–1040. https://doi-org.d360prx.biomed.cas.cz/10.1093/protein/12.12.1035.
- P.M.L. Janssen, B.J. Biesiadecki, M.T. Ziolo, J.P. Davis, The Need for Speed; Mice, Men, and Myocardial Kinetic Reserve, Circ. Res. 119 (2016) 418–421. [CrossRef]
- Vonk, W. I. M., Rainbolt, T. K., Dolan, P. T., Webb, A. E., Brunet, A., & Frydman, J. (2020). Differentiation Drives Widespread Rewiring of the Neural Stem Cell Chaperone Network. Molecular cell, 78(2), 329–345.e9. [CrossRef]
- C Benincasa, J., Madias, M. I., Kandell, R. M., Delgado-Garcia, L. M., Engler, A. J., Kwon, E. J., & Porcionatto, M. A. (2024). Mechanobiological Modulation of In Vitro Astrocyte Reactivity Using Variable Gel Stiffness. ACS biomaterials science & engineering, 10(7), 4279–4296. [CrossRef]
- Lucarini, F. Maita, G. Conte, E. Saracino, F. Formaggio, E.Palmieri, R. Fabbri, A. Konstantoulaki, C. Lazzarini, M. Caprini, V. Benfenati, L. Maiolo and A.Convertino, Silicon Nanowire Mats Enable Advanced Bioelectrical Recordings in Primary DRG Cell Cultures, Adv. Healthcare Mater. 2025, 2500379. [CrossRef]
- Joshi, I., & Andrew, R. D. (2001). Imaging anoxic depolarization during ischemia-like conditions in the mouse hemi-brain slice. Journal of neurophysiology, 85(1), 414–424. [CrossRef]
- C.P. Taylor, M.L. Weber, C.L. Gaughan, E.J. Lehning, R.M. LoPachin, Oxygen/glucose deprivation in hippocampal slices: altered intraneuronal elemental composition predicts structural and functional damage, J. Neurosci. Off. J. Soc. Neurosci. 19 (1999) 619–629. [CrossRef]
- Reappraisal of anoxic spreading depolarization as a terminal event during oxygen–glucose deprivation in brain slices in vitro Scientific Reports, (n.d.). https://www.nature.com/articles/s41598-020-75975-w (accessed on 17 April 2025).
- A.P. Antunes, A.J. Schiefecker, R. Beer, B. Pfausler, F. Sohm, M. Fischer, A. Dietmann, P. Lackner, W.O. Hackl, J.-P. Ndayisaba, C. Thomé, E. Schmutzhard, R. Helbok, Higher brain extracellular potassium is associated with brain metabolic distress and poor outcome after aneurysmal subarachnoid hemorrhage, Crit. Care Lond. Engl. 18 (2014) R119. [CrossRef]
- Hansen A. J. (1978). The extracellular potassium concentration in brain cortex following ischemia in hypo- and hyperglycemic rats. Acta physiologica Scandinavica, 102(3), 324–329. [CrossRef]
- Somjen G., G. (1979). Extracellular potassium in the mammalian central nervous system. Annual review of physiology, 41, 159–177. https://doi-org.d360prx.biomed.cas.cz/10.1146/annurev.ph.41.030179.
- Blaeser AS, Connors BW, Nurmikko AV. Spontaneous dynamics of neural networks in deep layers of prefrontal cortex. J Neurophysiol. 2017;117(4):1581-1594. [CrossRef]
- Schmidt SL, Chew EY, Bennett DV, Hammad MA, Fröhlich F. Differential effects of cholinergic and noradrenergic neuromodulation on spontaneous cortical network dynamics. Neuropharmacology. 2013;72:259-273. [CrossRef]
- Walch, E., Murphy, T. R., Cuvelier, N., Aldoghmi, M., Morozova, C., Donohue, J., Young, G., Samant, A., Garcia, S., Alvarez, C., Bilas, A., Davila, D., Binder, D. K., & Fiacco, T. A. (2020). Astrocyte-Selective Volume Increase in Elevated Extracellular Potassium Conditions Is Mediated by the Na+/K+ ATPase and Occurs Independently of Aquaporin 4. ASN neuro, 12, 1759091420967152. [CrossRef]
- Ding, F., Sun, Q., Long, C., Rasmussen, R. N., Peng, S., Xu, Q., Kang, N., Song, W., Weikop, P., Goldman, S. A., & Nedergaard, M. (2024). Dysregulation of extracellular potassium distinguishes healthy ageing from neurodegeneration. Brain : a journal of neurology, 147(5), 1726–1739. [CrossRef]
- Xie, M., Wang, W., Kimelberg, H. K., & Zhou, M. (2008). Oxygen and glucose deprivation-induced changes in astrocyte membrane potential and their underlying mechanisms in acute rat hippocampal slices. Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism, 28(3), 456–467. [CrossRef]
- Mestre ALG, Inácio PMC, Elamine Y, et al. Extracellular Electrophysiological Measurements of Cooperative Signals in Astrocytes Populations. Front Neural Circuits. 2017;11:80. Published 2017 Oct 23. [CrossRef]
- Chiang CC, Durand DM. Subthreshold Oscillating Waves in Neural Tissue Propagate by Volume Conduction and Generate Interference. Brain Sci. 2022;13(1):74. Published 2022 Dec 30. [CrossRef]




| Compounds | aCSF (mM) | Isolation solution (mM) |
Hyperkalemic (mM) | OGD (mM) |
|---|---|---|---|---|
| NaCl | 122 | - | 115 | 122 |
| NMDG | - | 110 | - | - |
| KCl | 3 | 2.5 | 10 | 3 |
| NaHCO3 | 28 | 24.5 | 28 | 28 |
| Na2HPO4 | 1.25 | 1.25 | 1.25 | 1.25 |
| Glucose | 10 | 20 | 10 | - |
| CaCl2 | 1.5 | 0.5 | 1.5 | 1.5 |
| MgCl2 | 1.3 | 7 | 1.3 | 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




