Submitted:
24 May 2025
Posted:
27 May 2025
You are already at the latest version
Abstract
Keywords:
1. INTRODUCTION
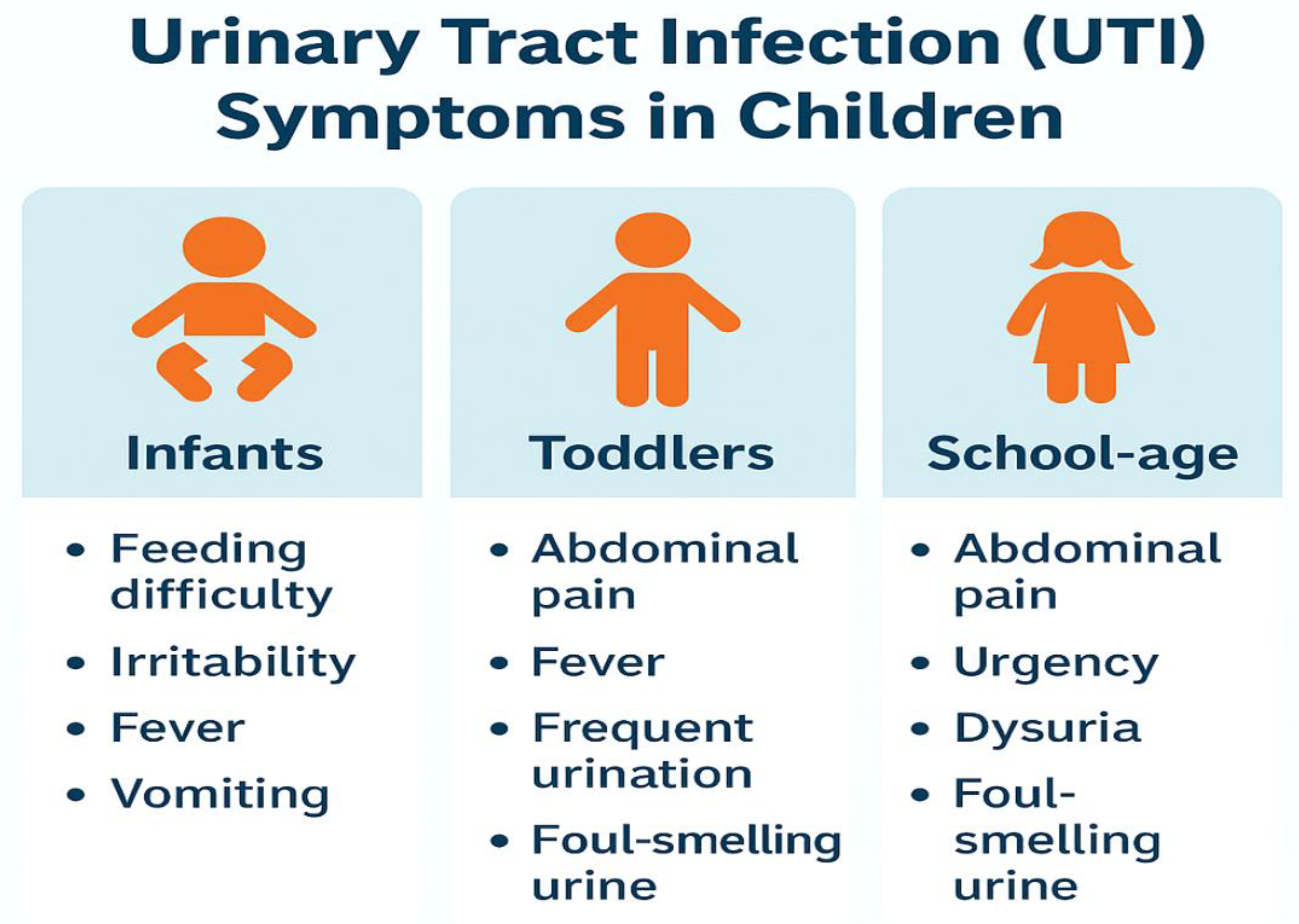
2. METHODOLOGY
2.1. Databases Searched
- PubMed/MEDLINE
- Scopus
- Web of Science
- Google Scholar
- World Health Organisation (WHO) Global Antimicrobial Resistance Surveillance System (GLASS)
- EMBASE
- African Journals Online (AJOL)
2.2. Search Terms and Strategy
2.3. Inclusion and Exclusion Criteria
- Studies published in peer-reviewed journals or validated surveillance sources.
- Articles focusing on pediatric populations (0–18 years).
- Reports addressing ESBL-producing organisms in urinary tract infections.
- Studies that discussed molecular biology, epidemiology, clinical impact, diagnostics, or treatment implications.
- Articles not in English.
- Studies not involving pediatric subjects.
- Case reports and opinion pieces without primary data.
- Studies focused solely on adult or animal models.
2.4. Selection and Review Process
3. GLOBAL EPIDEMIOLOGY OF ESBL-PRODUCING UROPATHOGENS
4. REGIONAL FOCUS: NIGERIA AND SUB-SAHARAN AFRICA
4.1. Prevalence and Clinical Trends in Nigeria
4.2. Molecular Profiles and Resistance Patterns
4.3. Contributing Factors to the ESBL Burden
- Inadequate diagnostic infrastructure, resulting in reliance on empirical therapy (39).
- Poor regulation of antibiotic sales, leading to widespread self-medication and overuse (40).
- Lack of pediatric-specific antimicrobial stewardship programs, particularly in primary and secondary care settings (41).
- Suboptimal infection control practices in both outpatient clinics and hospital wards (42).
- High patient-to-clinician ratios reduce the feasibility of individualised care (43).
4.4. Surveillance and Research Gaps
4.5. Regional Comparisons
5. MOLECULAR BIOLOGY OF ESBLS IN PEDIATRIC UTIS
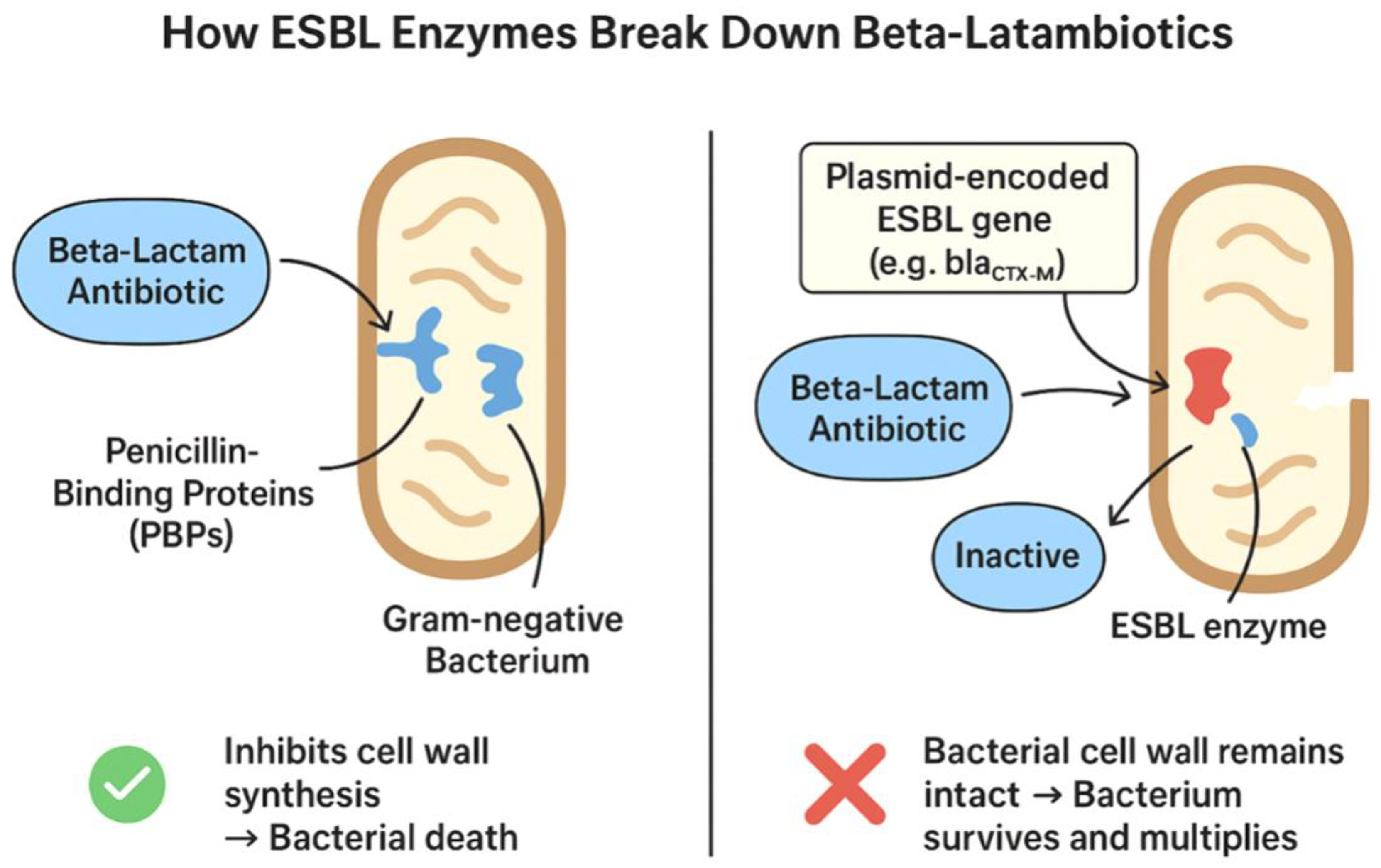
5.1. The TEM and SHV Families
5.2. The CTX-M Family
5.3. Genetic Context and Co-Resistance
5.4. Clonal Expansion and Horizontal Gene Transfer
5.5. Implications for Molecular Surveillance
6. CLINICAL IMPACT AND RISK FACTORS IN PEDIATRIC POPULATIONS
6.1. Disease Severity and Complications
6.2. Recurrent UTIs and Healthcare Burden
6.3. Risk Factors for ESBL Colonization and Infection
- Prior antibiotic exposure, particularly to cephalosporins and fluoroquinolones (67).
- Hospitalisation, catheterisation, and recent medical procedures (68).
- Congenital urinary anomalies (e.g., vesicoureteral reflux) (69).
- Poor hygiene and inadequate sanitation (70).
- Household or community-level transmission (71).
6.4. Vulnerable Subgroups
6.5. Psychological and Social Impact
7. DIAGNOSTIC APPROACHES: CURRENT PRACTICES AND MOLECULAR ADVANCES
7.1. Traditional Diagnostic Methods
7.2. Limitations of Phenotypic Testing
7.3. Molecular Diagnostics
7.4. Advanced Genomic Tools
7.5. Diagnostic Challenges in Pediatric Populations
7.6. The Role of Rapid Point-of-Care Testing (POCT)
7.7. Recommendations for Diagnostic Improvement
- Strengthening laboratory infrastructure and personnel training.
- Incorporating PCR-based diagnostics in tertiary hospitals.
- Developing cost-effective POCT for pediatric care.
- Encouraging public-private partnerships to scale diagnostic tools.
8. ANTIMICROBIAL RESISTANCE PATTERNS AND TREATMENT IMPLICATIONS
8.1. Resistance to Beta-Lactam Antibiotics
8.2. Cross-Resistance and Multidrug Resistance (MDR)
8.3. Retained Susceptibility and Alternative Options
8.4. Empirical Therapy Considerations
8.5. Treatment Challenges in LMICs
8.6. Role of Pharmacovigilance and Resistance Monitoring
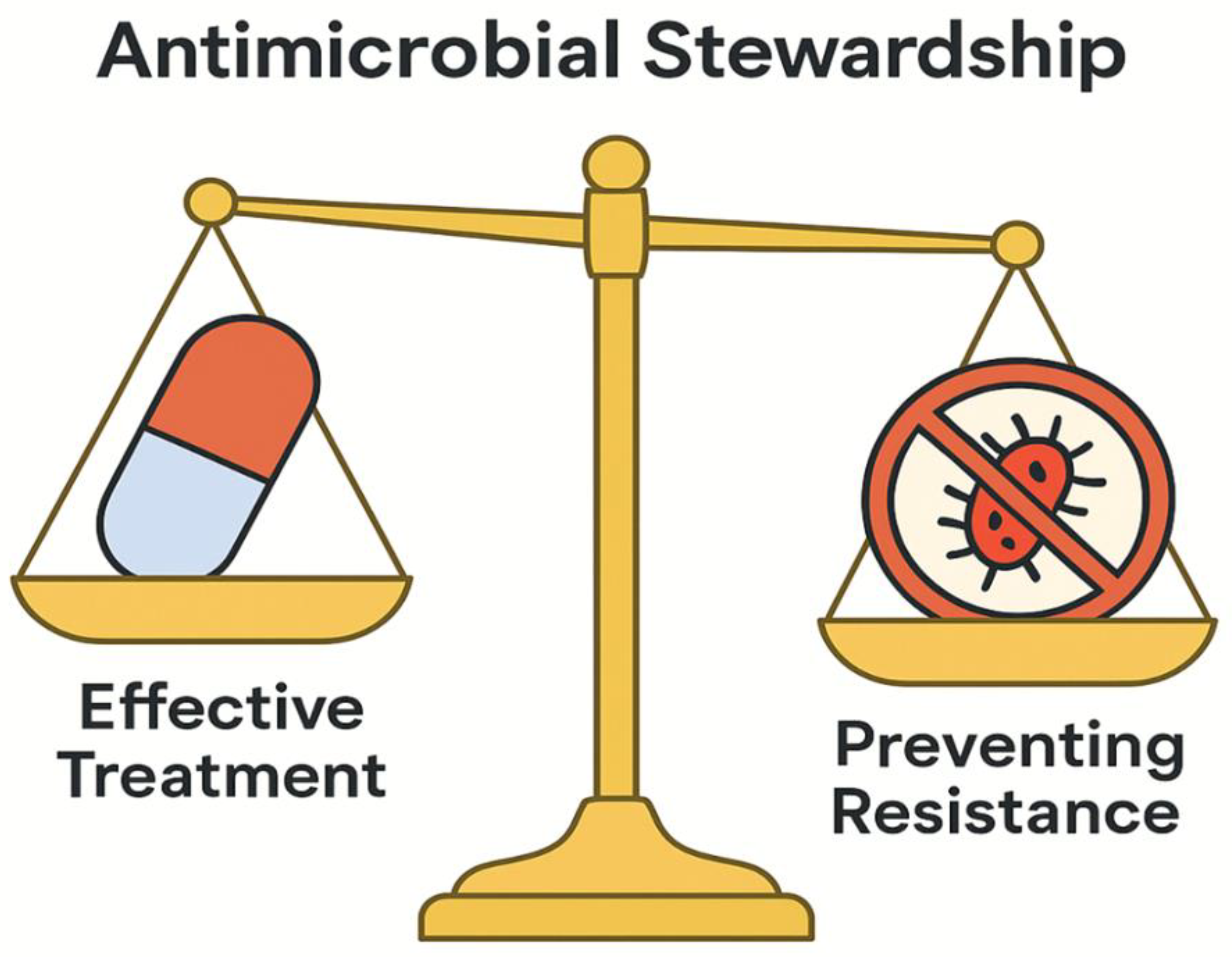
9. PUBLIC HEALTH AND STEWARDSHIP IMPLICATIONS
9.1. Strain on Health Systems
9.2. Implications for Infection Control and Hospital Policy
9.3. Antibiotic Stewardship in Pediatric Care
- Development and dissemination of pediatric-specific treatment guidelines.
- Regular audit and feedback mechanisms for antibiotic prescriptions.
- Engagement of paediatricians, pharmacists, and microbiologists in multidisciplinary stewardship teams.
- Training programs for junior clinicians on the principles of prudent antibiotic use.
9.4. Role of Pharmacists and Community Engagement
- When and why antibiotics are necessary (and when they are not).
- Risks of self-medication and incomplete courses.
- Recognition of early UTI symptoms and importance of clinical evaluation.
9.5. Policy and Regulatory Measures
- Expansion of diagnostic laboratories capable of detecting ESBL-producing strains.
- Investment in electronic medical records and surveillance systems.
- Inclusion of pediatric resistance data in national AMR reports.
9.6. International and Multisectoral Collaboration
10. FUTURE DIRECTION AND RECOMMENDATIONS
10.1. Revision of Empirical Treatment Protocols
10.2. Strengthening Antibiotic Stewardship Programs
10.3. Implementation of Routine Diagnostic Testing
10.4. Public Health Education Campaigns
10.5. Enhanced Surveillance and Research
11. CONCLUSIONS
References
- Shaikh, N., Morone, N. E., Bost, J. E. and Farrell, M. H.(2008). Prevalence of urinary tract infection in childhood: a meta-analysis. Pediatr Infect Dis J. 27(4):302–308.
- Masika, W. G., O’Meara, W. P., Holland, T. L. and Armstrong, J. (2017). Contribution of urinary tract infection to the burden of febrile illnesses in young children in rural Kenya. PLOS ONE 12(3): e0174199. [CrossRef]
- Coulthard, M. G., Lambert, H. J., Vernon, S. J., Hunter, E. W., Keir, M. J., & Matthews, J. N. (2014). Does prompt treatment of urinary tract infection in preschool children prevent renal scarring: mixed retrospective and prospective audits. Archives of disease in childhood, 99(4), 342–347. [CrossRef]
- Shaikh, N., Hoberman, A., Keren, R., Gotman, N., Docimo, S. G., Mathews, R., Bhatnagar, S., Ivanova, A., Mattoo, T. K., Moxey-Mims, M., Carpenter, M. A., Pohl, H. G., & Greenfield, S. (2016). Recurrent Urinary Tract Infections in Children With Bladder and Bowel Dysfunction. Pediatrics, 137(1), e20152982. [CrossRef]
- Ekpunobi, N. F., Mgbedo, U. G., Okoye, L. C. and Agu, K. C. (2024). Prevalence of ESBL genes in Klebsiella pneumoniae from individuals with community-acquired urinary tract infections in Lagos state, Nigeria. Journl of RNA and Genomics, 20 (2):1-6. [CrossRef]
- Ekpunobi, N., Adesanoye, S., Orababa, O., Adinnu, C., Okorie, C., Akinsuyi, O. (2024). Public health perspective of public abattoirs in Nigeria, challenges and solutions. GSC Biological and Pharmaceutical Sciences, 26. 115-127. [CrossRef]
- Okoye, L. C., Ugwu, M. C., Oli, A. N., Okoye, E. C. S., Ekpunobi, N. F., Okezie, U. M. and Mgbedo, U. G. (2024). Genotypic detection of metallo-Beta-Lactamases among multidrug resistant Klebsiella pneumoniae isolated from urine samples of UTI patients GSC Biological and Pharmaceutical Sciences, 29(03), 248-255. [CrossRef]
- Chukwunwejim, C.R., Ekpunobi, N.F., Ogunmola, T., Obidi, N., Ajasa, O. S. and Obidi, N. L. (2025). Molecular identification of multidrug-resistant bacteria from eggshell surfaces in Nigeria: A growing threat to public health. Magna Scientia Advanced Biology and Pharmacy, 2025, 15(01), 021-028. [CrossRef]
- Bryce, A., Hay, A. D., Lane, I. F., Thornton, H. V., Wootton, M., & Costelloe, C. (2016). Global prevalence of antibiotic resistance in paediatric urinary tract infections caused by Escherichia coli and association with routine use of antibiotics in primary care: systematic review and meta-analysis. BMJ (Clinical research ed.), 352, i939. [CrossRef]
- Obidi, N.O. and Ekpunobi, N. F. (2025). A narrative review exploring phage therapy as a sustainable alternative solution to combat antimicrobial resistance in Africa: Applications, challenges, and future directions. Afr. J. Clin. Exper. Microbiol. 26 (2): 106-113.
- Ekpunobi, N. F. and Agu, K.C. (2024). Emergence and Re-Emergence of Arboviruses: When Old Enemies Rise Again. Cohesive J Microbiol Infect Dis. 7(2). CJMI. 000658. [CrossRef]
- Chakupurakal, R., Ahmed, M., Sobithadevi, D. N., Chinnappan, S., & Reynolds, T. (2010). Urinary tract pathogens and resistance pattern. Journal of clinical pathology, 63(7), 652–654. [CrossRef]
- Murray, T.S. and Peaper, D.R. (2015) The Contribution of Extended-Spectrum β-Lactamases to Multidrug-Resistant Infections in Children. Current Opinion in Pediatrics, 27, 124-131. [CrossRef]
- Lukac, P. J., Bonomo, R. A., & Logan, L. K. (2015). Extended-spectrum β-lactamase-producing Enterobacteriaceae in children: old foe, emerging threat. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 60(9), 1389–1397. [CrossRef]
- Edlin, R. S., Shapiro, D. J., Hersh, A. L., & Copp, H. L. (2013). Antibiotic resistance patterns of outpatient pediatric urinary tract infections. The Journal of urology, 190(1), 222–227. [CrossRef]
- Parajuli, N. P., Maharjan, P., Parajuli, H., Joshi, G., Paudel, D., Sayami, S., & Khanal, P. R. (2017). High rates of multidrug resistance among uropathogenic Escherichia coli in children and analyses of ESBL producers from Nepal. Antimicrobial resistance and infection control, 6, 9. [CrossRef]
- Ferreira, I. C. D. S., Menezes, R. P., Jesus, T. A., Lopes, M. S. M., Araújo, L. B., Ferreira, D. M. L. M., & Röder, D. V. D. B. (2024). Unraveling the epidemiology of urinary tract infections in neonates: Perspective from a Brazilian NICU. American journal of infection control, 52(8), 925–933. [CrossRef]
- Reuland, E. A., Sonder, G. J., Stolte, I., Al Naiemi, N., Koek, A., Linde, G. B., van de Laar, T. J., Vandenbroucke-Grauls, C. M., & van Dam, A. P. (2016). Travel to Asia and traveller's diarrhoea with antibiotic treatment are independent risk factors for acquiring ciprofloxacin-resistant and extended spectrum β-lactamase-producing Enterobacteriaceae-a prospective cohort study. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases, 22(8), 731.e1–731.e7317. [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). (2020). Antimicrobial resistance in the EU/EEA (EARS-Net) – Annual epidemiological report for 2019. https://www.ecdc.europa.eu/en/publications-data/surveillance-antimicrobial-resistance-europe-2019.
- Peirano, G., & Pitout, J. D. (2019). Extended-spectrum β-lactamase-producing Enterobacteriaceae: update on molecular epidemiology and treatment options. Drugs, 79, 1529-1541.
- Laxminarayan, R., Duse, A., Wattal, C., Zaidi, A. K., Wertheim, H. F., Sumpradit, N., Vlieghe, E., Hara, G. L., Gould, I. M., Goossens, H., Greko, C., So, A. D., Bigdeli, M., Tomson, G., Woodhouse, W., Ombaka, E., Peralta, A. Q., Qamar, F. N., Mir, F., Kariuki, S., … Cars, O. (2013). Antibiotic resistance-the need for global solutions. The Lancet. Infectious diseases, 13(12), 1057–1098. [CrossRef]
- Aibinu I, Odugbemi T, Koenig W, Ghebremedhin B. Sequence Type ST131 and ST10 Complex (ST617) predominant among CTX-M-15-producing Escherichia coli isolates from Nigeria Clin Microbiol Infect. 2012;18:E49–51.
- Maina, D., Makau, P., Nyerere, A. and Revathi, G. (2013) Antimicrobial Resistance Patterns in Extended-Spectrum β-Lactamase Producing Escherichia coli and Klebsiella pneumoniae Isolates in a Private Tertiary Hospital, Kenya. Microbiology Discovery, 1, 5.
- Ouedraogo, A. S., Sanou, M., Kissou, A., Sanou, S., Solaré, H., Kaboré, F., Poda, A., Aberkane, S., Bouzinbi, N., Sano, I., Nacro, B., Sangaré, L., Carrière, C., Decré, D., Ouégraogo, R., Jean-Pierre, H., & Godreuil, S. (2016). High prevalence of extended-spectrum ß-lactamase producing enterobacteriaceae among clinical isolates in Burkina Faso. BMC infectious diseases, 16, 326. [CrossRef]
- Tamma, P. D., Avdic, E., Li, D. X., Dzintars, K., & Cosgrove, S. E. (2017). Association of Adverse Events With Antibiotic Use in Hospitalized Patients. JAMA internal medicine, 177(9), 1308–1315. [CrossRef]
- Pillonetto, M., Jordão, R. T. S., Andraus, G. S., Bergamo, R., Rocha, F. B., Onishi, M. C., de Almeida, B. M. M., Nogueira, K. D. S., Dal Lin, A., Dias, V. M. C. H., & de Abreu, A. L. (2021). The Experience of Implementing a National Antimicrobial Resistance Surveillance System in Brazil. Frontiers in public health, 8, 575536. [CrossRef]
- Pavez, M., Troncoso, C., Osses, I., Salazar, R., Illesca, V., Reydet, P., Rodríguez, C., Chahin, C., Concha, C., & Barrientos, L. (2019). High prevalence of CTX-M-1 group in ESBL-producing enterobacteriaceae infection in intensive care units in southern Chile. The Brazilian journal of infectious diseases : an official publication of the Brazilian Society of Infectious Diseases, 23(2), 102–110. [CrossRef]
- Woerther, P. L., Burdet, C., Chachaty, E., & Andremont, A. (2013). Trends in human fecal carriage of extended-spectrum β-lactamases in the community: toward the globalization of CTX-M. Clinical microbiology reviews, 26(4), 744–758. [CrossRef]
- Carattoli, A. (2013). Plasmids and the spread of resistance. International Journal of Medical Microbiology, 303(6-7), 298–304.
- Van Boeckel, T. P., Brower, C., Gilbert, M., Grenfell, B. T., Levin, S. A., Robinson, T. P., Teillant, A., & Laxminarayan, R. (2015). Global trends in antimicrobial use in food animals. Proceedings of the National Academy of Sciences, 112(18), 5649-5654.
- O'Neill, J. (2016). Tackling drug-resistant infections globally: Final report and recommendations. Review on Antimicrobial Resistance. https://amr-review.org.
- Ayukekbong, J.A., Ntemgwa, M. and Atabe, A.N. (2017) The Threat of Antimicrobial Resistance in Developing Countries: Causes and Control Strategies. Antimicrobial Resistance and Infection Control, 6, 47. [CrossRef]
- Ogbolu, D. O., Daini, O. A., Ogunledun, A., Alli, A. O., & Webber, M. A. (2011). High levels of multidrug resistance in clinical isolates of Gram-negative pathogens from Nigeria. International journal of antimicrobial agents, 37(1), 62–66. [CrossRef]
- Adekanmbi, A. O., Akinlabi, O. C., Usidamen, S., Olaposi, A. V., & Olaniyan, A. B. (2022). High burden of ESBL- producing Klebsiella spp., Proteus mirabilis, Enterobacter cloacae and Pseudomonas aeruginosa in diagnosed cases of urinary tract infection in a Nigerian Teaching Hospital. Acta microbiologica et immunologica Hungarica, 69(2), 127–134. [CrossRef]
- Gulumbe, B.H. and Faggo, A.A. (2019). Epidemiology of Multidrug-resistant Organisms in Africa. Mediterranean Journal of Infection Microbes and Antimicrobials, 8(1), 25-25.
- Onanuga A, Eboh DD, Odetoyin B, Adamu OJ. (2020). Detection of ESBLs and NDM-1 genes among urinary Escherichia coli and Klebsiella pneumoniae fromhealthy students in Niger delta University, Amassoma, Bayelsa State, Nigeria.PAMJ - One Heal. 2:12.
- Bello, R., Ibrahim, Y.K., Olayinka, B.O., Jimoh, G.A., Afolabi-Balogun, N., Oni-Babatunde, A.O., Olabode, H.O., David, M.S., Aliyu, A., & Olufadi-Ahmed, H.Y. (2021). Molecular Characterization of Extended Spectrum Beta – Lactamase Producing Escherichia Coli Isolated from Pregnant Women with Urinary Tract Infections Attending Ante–Natal Clinics in Ilorin Metropolis. Nigerian Journal of Pharmaceutical Research.
- Auta, A., Hadi, M. A., Oga, E., Adewuyi, E. O., Abdu-Aguye, S. N., Adeloye, D., Strickland-Hodge, B., & Morgan, D. J. (2019). Global access to antibiotics without prescription in community pharmacies: A systematic review and meta-analysis. The Journal of infection, 78(1), 8–18. [CrossRef]
- Bastidas-Caldes, C., Hernández-Alomía, F., Almeida, M., Ormaza, M., Boada, J., Graham, J., Calvopiña, M., & Castillejo, P. (2024). Molecular identification and antimicrobial resistance patterns of enterobacterales in community urinary tract infections among indigenous women in Ecuador: addressing microbiological misidentification. BMC infectious diseases, 24(1), 1195. [CrossRef]
- Fadare, J. O., Ogunleye, O., Iliyasu, G., Adeoti, A., Schellack, N., Engler, D., Massele, A., & Godman, B. (2019). Status of antimicrobial stewardship programmes in Nigerian tertiary healthcare facilities: Findings and implications. Journal of global antimicrobial resistance, 17, 132–136. [CrossRef]
- Iregbu KC, Nwajiobi-Princewill PI, Medugu N, Umeokonkwo, C. D., Uwaezuoke, N. S., Peter, Y. J., Nwafia, I. N., Elikwu, C., 7Shettima, S. A., Suleiman, M. R., Awopeju, A. T. O., Udoh, U., Adedosu, N., Mohammed, A., Oshun, P., Ekuma, A., Manga, M. M., Osaigbovo, I. I., Ejembi, C. J., Akujobi, C. N., Samuel, S. O., Taiwo, S. S., and Oduyebo, O. O. (2021). Antimicrobial Stewardship Implementation in Nigerian Hospitals: Gaps and Challenges. African Journal of Clinical and Experimental Microbiology.22(1):60-66.https://www.ajol.info/index.php/ajcem/article/view/203077.
- Obiageli J.O., Uzoma I., Sanda U. I., Uzairue L. I., Emmanuel C. A. (2023). Systematic review of surveillance systems for AMR in Africa, Journal of Antimicrobial Chemotherapy, 78(1): 31–51, . [CrossRef]
- World Health Organization (WHO). (2021). Global antimicrobial resistance and use surveillance system (GLASS) report. https://www.who.int/publications/i/item/9789240027336.
- Olalekan, A., Onwugamba, F., Iwalokun, B., Mellmann, A., Becker, K., & Schaumburg, F. (2020). High proportion of carbapenemase-producing Escherichia coli and Klebsiella pneumoniae among extended-spectrum β-lactamase-producers in Nigerian hospitals. Journal of global antimicrobial resistance, 21, 8–12. [CrossRef]
- Sampah J, Owusu-Frimpong I, Aboagye FT, Owusu-Ofori A (2023) Prevalence of carbapenem-resistant and extended-spectrum beta-lactamase-producing Enterobacteriaceae in a teaching hospital in Ghana. PLoS ONE 18(10): e0274156.
- Bush, K., & Bradford, P. A. (2020). Epidemiology of β-Lactamase-Producing Pathogens. Clinical microbiology reviews, 33(2), e00047-19. [CrossRef]
- Paterson, D. L., & Bonomo, R. A. (2005). Extended-spectrum beta-lactamases: a clinical update. Clinical microbiology reviews, 18(4), 657–686. [CrossRef]
- Cantón, R., González-Alba, J. M., & Galán, J. C. (2012). CTX-M Enzymes: Origin and Diffusion. Frontiers in microbiology, 3, 110. [CrossRef]
- Bevan, E. R., Jones, A. M., & Hawkey, P. M. (2017). Global epidemiology of CTX-M β-lactamases: temporal and geographical shifts in genotype. The Journal of antimicrobial chemotherapy, 72(8), 2145–2155. [CrossRef]
- Bonnet R. (2004). Growing group of extended-spectrum beta-lactamases: the CTX-M enzymes. Antimicrobial agents and chemotherapy, 48(1), 1–14. [CrossRef]
- Bradford P. A. (2001). Extended-spectrum beta-lactamases in the 21st century: characterization, epidemiology, and detection of this important resistance threat. Clinical microbiology reviews, 14(4), 933–951. [CrossRef]
- Jacoby, G. A., & Munoz-Price, L. S. (2005). The new beta-lactamases. The New England journal of medicine, 352(4), 380–391. [CrossRef]
- D'Andrea, M. M., Arena, F., Pallecchi, L., & Rossolini, G. M. (2013). CTX-M-type β-lactamases: a successful story of antibiotic resistance. International journal of medical microbiology : IJMM, 303(6-7), 305–317. [CrossRef]
- Rodríguez-Baño, J., Gutiérrez-Gutiérrez, B., Machuca, I., & Pascual, A. (2018). Treatment of Infections Caused by Extended-Spectrum-Beta-Lactamase-, AmpC-, and Carbapenemase-Producing Enterobacteriaceae. Clinical microbiology reviews, 31(2), e00079-17. [CrossRef]
- Coque, T. M., Novais, A., Carattoli, A., Poirel, L., Pitout, J., Peixe, L., Baquero, F., Cantón, R., & Nordmann, P. (2008). Dissemination of clonally related Escherichia coli strains expressing extended-spectrum beta-lactamase CTX-M-15. Emerging infectious diseases, 14(2), 195–200. [CrossRef]
- Mathers, A. J., Peirano, G., & Pitout, J. D. (2015). The role of epidemic resistance plasmids and international high-risk clones in the spread of multidrug-resistant Enterobacteriaceae. Clinical microbiology reviews, 28(3), 565–591. [CrossRef]
- Duicu, C., Cozea, I., Delean, D., Aldea, A. A., & Aldea, C. (2021). Antibiotic resistance patterns of urinary tract pathogens in children from Central Romania. Experimental and therapeutic medicine, 22(1), 748. [CrossRef]
- Nicolas-Chanoine, M. H., Bertrand, X., & Madec, J. Y. (2014). Escherichia coli ST131, an intriguing clonal group. Clinical microbiology reviews, 27(3), 543–574. [CrossRef]
- Tchesnokova, V., Radey, M., Chattopadhyay, S., Larson, L., Weaver, J. L., Kisiela, D., & Sokurenko, E. V. (2019). Pandemic fluoroquinolone resistant Escherichia coli clone ST1193 emerged via simultaneous homologous recombinations in 11 gene loci. Proceedings of the National Academy of Sciences of the United States of America, 116(29), 14740–14748. [CrossRef]
- Stoesser, N., Sheppard, A. E., Pankhurst, L., De Maio, N., Moore, C. E., Sebra, R., Turner, P., Anson, L. W., Kasarskis, A., Batty, E. M., Kos, V., Wilson, D. J., Phetsouvanh, R., Wyllie, D., Sokurenko, E., Manges, A. R., Johnson, T. J., Price, L. B., Peto, T. E., Johnson, J. R., … Modernizing Medical Microbiology Informatics Group (MMMIG) (2016). Evolutionary History of the Global Emergence of the Escherichia coli Epidemic Clone ST131. mBio, 7(2), e02162. [CrossRef]
- Enyi, E. O., Ekpunobi, N. F. (2022). Secondary metabolites from endophytic fungi of Moringa oleifera: antimicrobial and antioxidant properties. Journal of Microbiology and Experimentation 10(5):150‒154. [CrossRef]
- Ekpunobi, N. F. and Adeleye, I. A. (2020). Phenotypic characterization of biofilm formation and efflux pump activity in multidrug-resistant Staphylococcus species isolated from asymptomatic students. Journal of Microbiology and Experimentation 8(6): 223-229. [CrossRef]
- Logan, L. K., Braykov, N. P., Weinstein, R. A., Laxminarayan, R., & CDC Epicenters Prevention Program (2014). Extended-Spectrum β-Lactamase-Producing and Third-Generation Cephalosporin-Resistant Enterobacteriaceae in Children: Trends in the United States, 1999-2011. Journal of the Pediatric Infectious Diseases Society, 3(4), 320–328. [CrossRef]
- Hughes, K., Cannings-John, R., Jones, H., Lugg-Widger, F., Lau, T. M. M., Paranjothy, S., Francis, N., Hay, A. D., Butler, C. C., Angel, L., Van der Voort, J., & Hood, K. (2024). Long-term consequences of urinary tract infection in childhood: an electronic population-based cohort study in Welsh primary and secondary care. The British journal of general practice : the journal of the Royal College of General Practitioners, 74(743), e371–e378. [CrossRef]
- Afsharpaiman S, Bairaghdar F, Torkaman M, Kavehmanesh Z, Amirsalari S, Moradi M, Safavimirmahalleh MJ. Bacterial Pathogens and Resistance Patterns in Children With Community-Acquired Urinary Tract Infection: A Cross Sectional Study. J Compr Ped. 2012;3(1):16- 20. [CrossRef]
- Habib S. (2012). Highlights for management of a child with a urinary tract infection. International journal of pediatrics, 2012, 943653. [CrossRef]
- Drekonja DM, Filice GA, Greer N, Olson A, MacDonald R, Rutks I, Wilt TJ. 2015. Antimicrobial stewardship in outpatient settings: a systematic review. Infect Control Hosp Epidemiol 36:142–152.
- Cardoso T, Almeida M, Friedman ND, Aragao I, Costa-Pereira A, Sarmento AE, et al. (2014) Classification of healthcare-associated infection: a systematic review 10 years after the first proposal. BMC Med. 12:40.
- Sencan, A., Carvas, F., Hekimoglu, I. C., Caf, N., Sencan, A., Chow, J., & Nguyen, H. T. (2014). Urinary tract infection and vesicoureteral reflux in children with mild antenatal hydronephrosis. Journal of pediatric urology, 10(6), 1008–1013. [CrossRef]
- Laxminarayan, R., Duse, A., Wattal, C., Zaidi, A. K., Wertheim, H. F., Sumpradit, N., Vlieghe, E., Hara, G. L., Gould, I. M., Goossens, H., Greko, C., So, A. D., Bigdeli, M., Tomson, G., Woodhouse, W., Ombaka, E., Peralta, A. Q., Qamar, F. N., Mir, F., Kariuki, S., … Cars, O. (2013). Antibiotic resistance-the need for global solutions. The Lancet. Infectious diseases, 13(12), 1057–1098. [CrossRef]
- Woerther, P. L., Burdet, C., Chachaty, E., & Andremont, A. (2013). Trends in human fecal carriage of extended-spectrum β-lactamases in the community: toward the globalization of CTX-M. Clinical microbiology reviews, 26(4), 744–758. [CrossRef]
- Magiorakos, A. P., Srinivasan, A., Carey, R. B., Carmeli, Y., Falagas, M. E., Giske, C. G., Harbarth, S., Hindler, J. F., Kahlmeter, G., Olsson-Liljequist, B., Paterson, D. L., Rice, L. B., Stelling, J., Struelens, M. J., Vatopoulos, A., Weber, J. T., & Monnet, D. L. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases, 18(3), 268–281. [CrossRef]
- Subcommittee on Urinary Tract Infection, Steering Committee on Quality Improvement and Management, Roberts, K. B. (2011). Urinary tract infection: clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics, 128(3), 595–610.
- CLSI. (2023). Performance Standards for Antimicrobial Susceptibility Testing. Clinical and Laboratory Standards Institute.
- Pitout, J. D., & Laupland, K. B. (2008). Extended-spectrum beta-lactamase-producing Enterobacteriaceae: an emerging public-health concern. The Lancet. Infectious diseases, 8(3), 159–166. [CrossRef]
- Giske, C. G., Sundsfjord, A. S., Kahlmeter, G., Woodford, N., Nordmann, P., Paterson, D. L., Cantón, R., & Walsh, T. R. (2009). Redefining extended-spectrum beta-lactamases: balancing science and clinical need. The Journal of antimicrobial chemotherapy, 63(1), 1–4. [CrossRef]
- Notomi, T., Okayama, H., Masubuchi, H., Yonekawa, T., Watanabe, K., Amino, N., & Hase, T. (2000). Loop-mediated isothermal amplification of DNA. Nucleic acids research, 28(12), E63. [CrossRef]
- Chen, H., Liu, K., Li, Z., & Wang, P. (2019). Point of care testing for infectious diseases. Clinica chimica acta; international journal of clinical chemistry, 493, 138–147. [CrossRef]
- Sunmonu, A. and Ekpunobi, N. (2023). Larvicidal potential of silver nanoparticles synthesized from Ocimum gratissimum leaf extracts against anopheles' mosquito. GSC Biological and Pharmaceutical Sciences. 25. 41-48. [CrossRef]
- Gajdács, M., Burián, K., & Terhes, G. (2019). Resistance Levels and Epidemiology of Non-Fermenting Gram-Negative Bacteria in Urinary Tract Infections of Inpatients and Outpatients (RENFUTI): A 10-Year Epidemiological Snapshot. Antibiotics (Basel, Switzerland), 8(3), 143. [CrossRef]
- Livermore, D. M., & Woodford, N. (2006). The beta-lactamase threat in Enterobacteriaceae, Pseudomonas and Acinetobacter. Trends in microbiology, 14(9), 413–420. [CrossRef]
- Codjoe, F. S., & Donkor, E. S. (2017). Carbapenem Resistance: A Review. Medical sciences (Basel, Switzerland), 6(1), 1. [CrossRef]
- Ekpunobi, N., Markjonathan, I., Olanrewaju, O. and Olanihun, D. (2020). Idiosyncrasies of COVID- 19;A Review. Iranian Journal of Medical Microbiology 14(3): 290-296. [CrossRef]
- Gandra, S., Alvarez-Uria, G., Turner, P., Joshi, J., Limmathurotsakul, D., & van Doorn, H. R. (2020). Antimicrobial Resistance Surveillance in Low- and Middle-Income Countries: Progress and Challenges in Eight South Asian and Southeast Asian Countries. Clinical microbiology reviews, 33(3), e00048-19. [CrossRef]
- Alqahtani, A., Almubayedh, T., Alkhamis, I. H., AlMubayedh, A. A., Shuiel, H. K., Aldakail, M. (2025). Knowledge, attitudes, and practices of Saudi parents regarding antibiotic use for children: A cross-sectional study. International Journal of Pediatrics and Adolescent Medicine 12(1): 20-29.
- National Institute for Health and Care Excellence (NICE). (2018). Urinary tract infection in under 16s: Diagnosis and management. https://www.nice.org.uk/guidance/cg54.
- Nordmann, P., Naas, T., & Poirel, L. (2011). Global spread of Carbapenemase-producing Enterobacteriaceae. Emerging infectious diseases, 17(10), 1791–1798. [CrossRef]
- Hersh, A. L., Jackson, M. A., Hicks, L. A., & American Academy of Pediatrics Committee on Infectious Diseases (2013). Principles of judicious antibiotic prescribing for upper respiratory tract infections in pediatrics. Pediatrics, 132(6), 1146–1154. [CrossRef]
- Ventola C. L. (2015). The antibiotic resistance crisis: part 1: causes and threats. P & T : a peer-reviewed journal for formulary management, 40(4), 277–283.
- Ogunleye, O. O., Fadare, J. O., Yinka-Ogunleye, A. F., Anand Paramadhas, B. D., & Godman, B. (2019). Determinants of antibiotic prescribing among doctors in a Nigerian urban tertiary hospital. Hospital practice (1995), 47(1), 53–58. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



