Submitted:
23 May 2025
Posted:
26 May 2025
You are already at the latest version
Abstract
Keywords:
Introduction
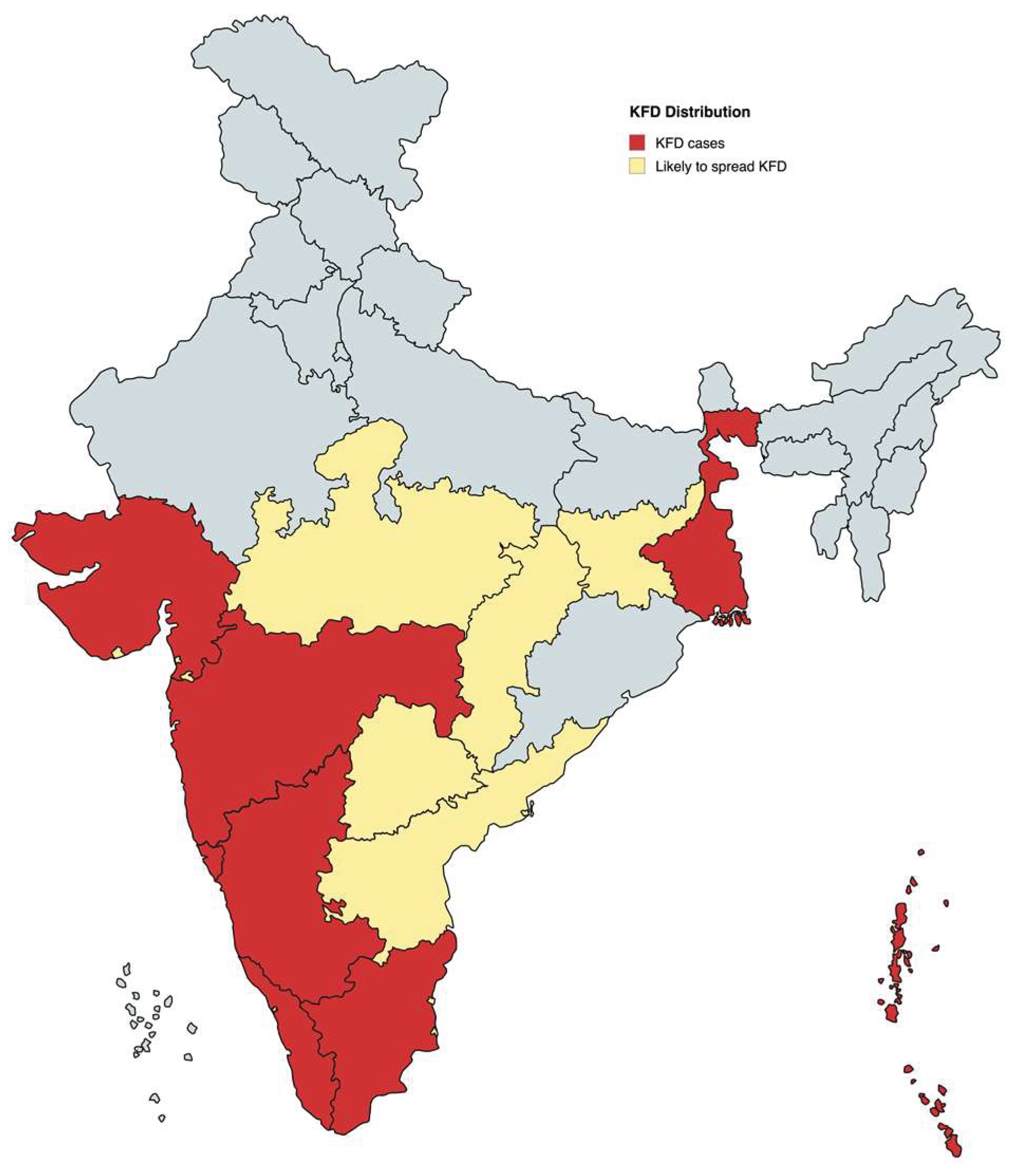
Epidemiology and Transmission of KFDV
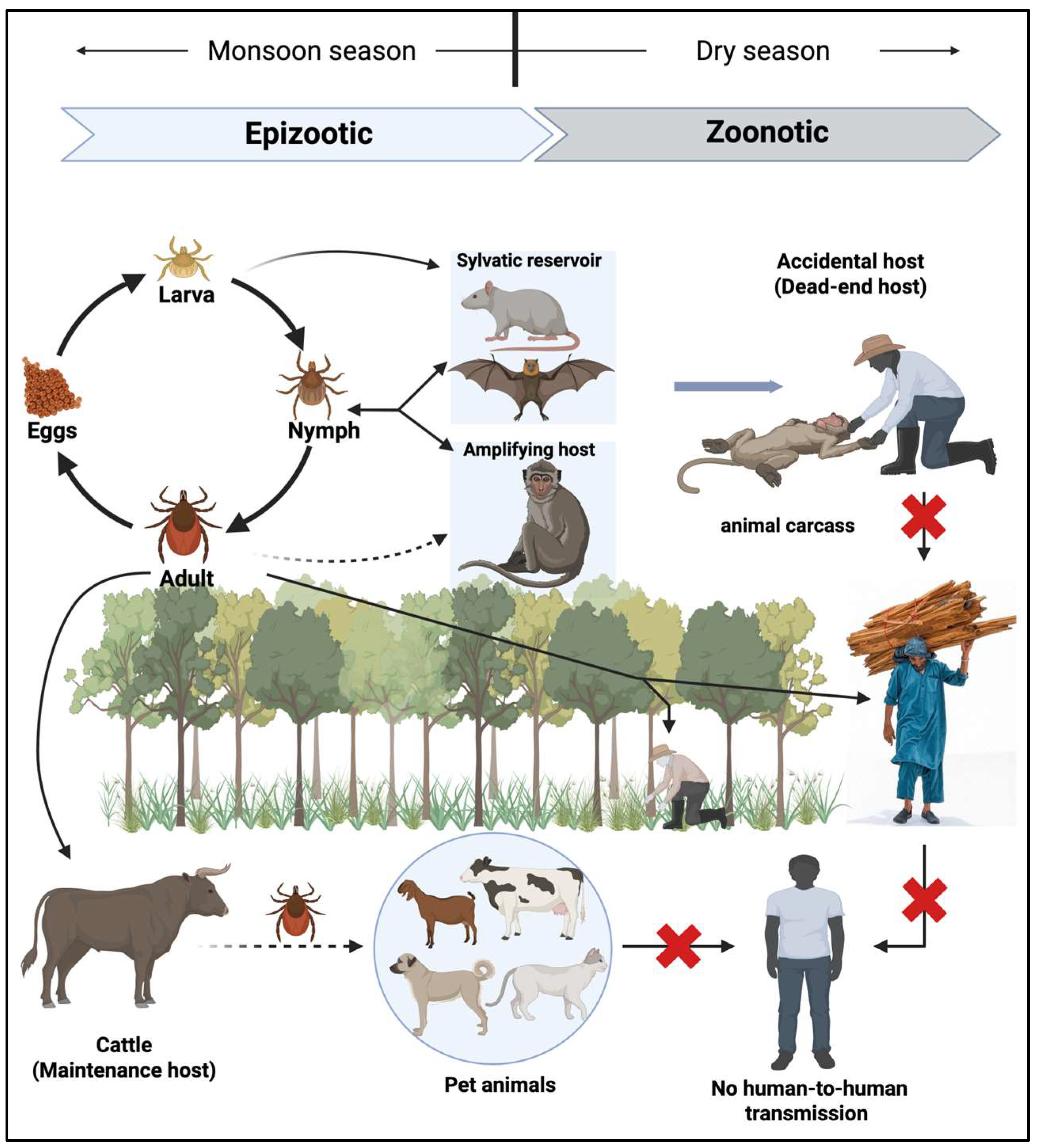
KFD virus and Pathogenesis
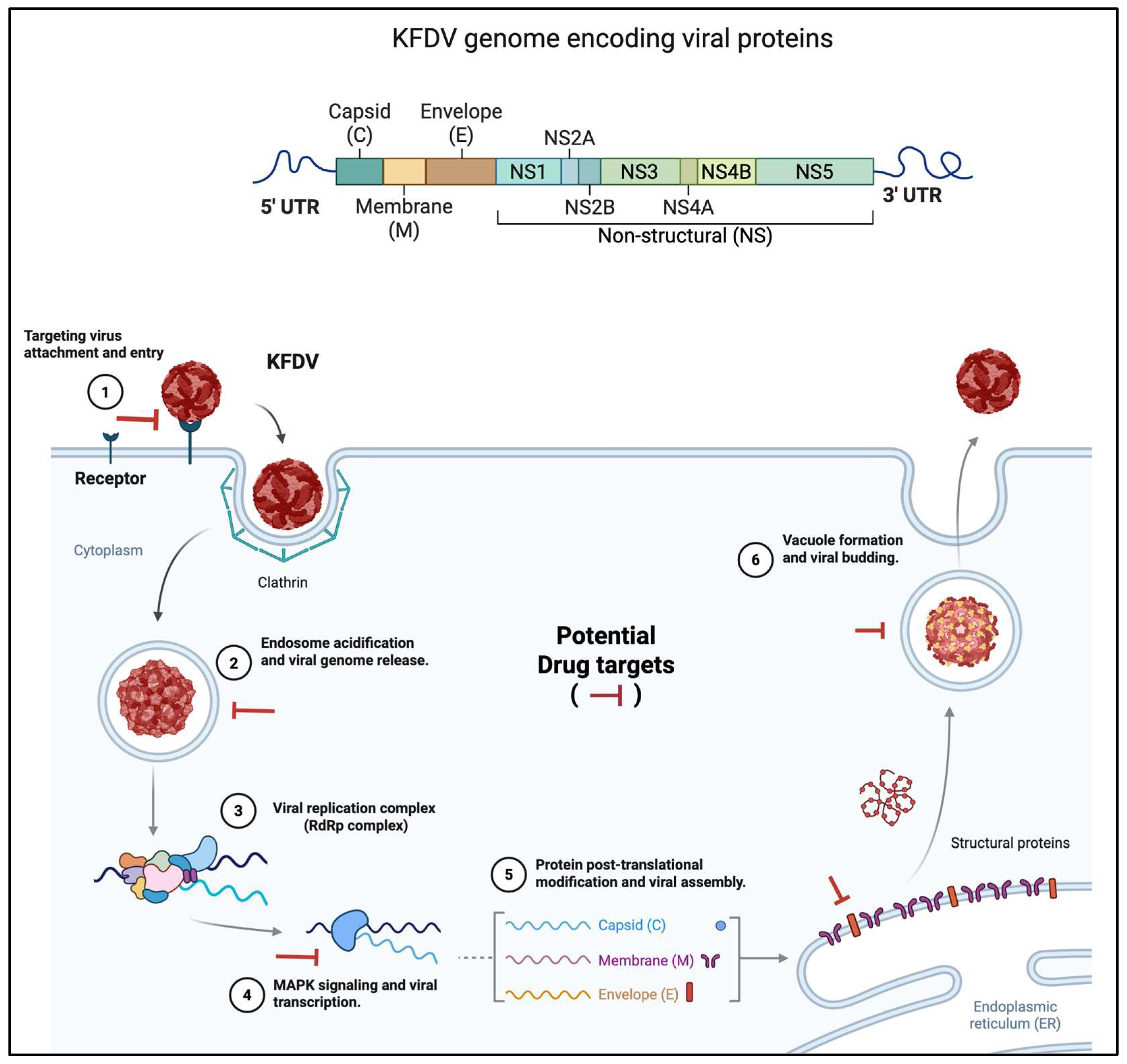
Therapeutic Options for KFDV
Current Status
Repurposed Drugs
| Drug Name | Mechanism of Action | Evidence of Activity | Current Status | Notes for KFDV Potential |
|---|---|---|---|---|
| NITD008 | Nucleoside analog; inhibits RNA synthesis | In vitro activity against KFDV, DENV, Zika [21,43,55] | Preclinical; discontinued due to toxicity | Demonstrated KFDV inhibition; safer analogs needed; AG129 model testing pending. |
| Favipiravir | RNA-dependent RNA polymerase inhibitor | In vitro/vivo activity against Ebola, DENV [56,57] | Clinically approved (e.g., influenza); not tested for KFDV | Broad-spectrum; potential for KFDV NS5 targeting; clinical trials required. |
| Sofosbuvir | NS5B polymerase inhibitor | In vitro activity against HCV, DENV [45,58] | Clinically approved (HCV); not tested for KFDV | Flavivirus cross-reactivity possible; in vivo KFDV studies needed. |
| Niclosamide | Induces autophagy; inhibits NS2B-NS3 protease | In vitro and in vivo activity[51]against DENV and SARS-CoV-2 [59] | FDA-approved (anthelmintic); not tested for KFDV | Promising for KFDV due to flavivirus similarity; AG129 model adaptation suggested. |
| Monoclonal Antibodies | Neutralizes E protein; blocks viral entry | Effective in DENV passive immunization [54] | Preclinical/early clinical for DENV; none for KFDV | KFDV-specific antibodies needed; risk of cross-reactivity with other flaviviruses. |
Challenges to develop therapeutics for KFDV
Vaccines
| Vaccine Name | Platform/Technology | Evidence of Efficacy | Current Status | Notes for KFDV Potential |
|---|---|---|---|---|
| Formalin-Inactivated Vaccine | Inactivated whole virus (mouse brain-derived) | 62.4% efficacy (2 doses), 82.9% (with boosters) [14,62] | In use since 1960s; suspended in 2022 | Partial protection; waning immunity; production challenges; booster dependency. |
| VSV-Based Vaccine | Recombinant vesicular stomatitis virus (VSV) expressing KFDV E protein | 100% protection in BALB/c mice [16]; reduced viral load in macaques [15]; cross-protects against AHFV | Preclinical (mice, macaques) | Promising efficacy and safety; Phase I/II trials needed; scalable production is potential. |
| Multi-Epitope Subunit Vaccine | Recombinant subunit (in-silico designed E protein epitopes) | Strong B/T-cell responses predicted in silico; binds TLR-2 [18] | Preclinical (in silico) | Cost-effective; in vivo validation pending; potential AHFV cross-protection. |
| mRNA Vaccine | mRNA encoding KFDV antigens | Effective for Zika, SARS-CoV-2 [64,65,66] | None of the study done for KFDV | Rapid development potential; adaptable to strains; requires KFDV-specific design. |
| Live-Attenuated Vaccine | Attenuated KFDV strain | Successful for yellow fever [67] 2017, DENV [68] | Not developed for KFDV | Could induce robust immunity; safety concerns need addressing; preclinical testing needed. |
Diagnostics
| Diagnostic methods | Technique | Detection Target | Sensitivity/Specificity | Current Status | Notes on Limitations and Potential Improvements |
|---|---|---|---|---|---|
| RT-PCR (Standard) | Reverse transcription PCR | Viral RNA (envelope gene) | ~95% / ~98% [69] | Routine in labs | Limited to viremic phase (days 3–8); requires infrastructure; dry-down version improves field use. |
| Dry-Down RT-PCR | Lyophilized RT-PCR | Viral RNA (envelope gene) | ~95% / ~98% [22] | Emerging (field testing) | Reduces turnaround to 4–6 hours; needs validation in remote settings; scalable production needed. |
| ELISA (IgM/IgG) | Enzyme-linked immunosorbent assay | IgM/IgG antibodies | ~92% / ~94% (IgM) [70] | Routine in labs | Cross-reactivity with flaviviruses; delayed detection (days 5–14); enhance with recombinant antigens. |
| Next-Generation Sequencing (NGS) | High-throughput sequencing | Whole viral genome | Variable (research-grade [71] | Research tool | Costly and complex; not routine; potential for AI integration to track strains. |
| Point-of-Care (POC) Devices | Lateral flow or RT-PCR-based | KFDV Antigens or RNA | Under validation (~90% est.;[72] | Prototypes (development) | Limited validation; needs thermostable, affordable design for rural deployment. |
Conclusion
Discussion and Future Directions
Research Gaps
Future Directions
Global Implications and Integrated Strategies
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Work Th, Trapido H, Murthy Dp, Rao Rl, Bhatt Pn, Kulkarni Kg. Kyasanur forest disease. III. A preliminary report on the nature of the infection and clinical manifestations in human beings. Indian J Med Sci. 1957;11(8):619-645.
- Work Th. Russian spring-summer virus in India: Kyasanur Forest disease. Prog Med Virol. 1958;1:248-279.
- Trapido H, Rajagopalan Pk, Work Th, Varma Mg. Kyasanur Forest disease. VIII. Isolation of Kyasanur Forest disease virus from naturally infected ticks of the genus Haemaphysalis. Indian J Med Res. 1959;47(2):133-138.
- Ocular manifestations of Kyasanur forest disease (a clinical study). Indian J Ophthalmol. 1983;31(5):700-702.
- Wadia RS. Neurological involvement in Kyasanur Forest Disease. Neurol India. 1975;23(3):115-120.
- Muraleedharan M. Kyasanur Forest Disease (KFD): Rare Disease of Zoonotic Origin. J Nepal Health Res Counc. 2016;14(34):214-218.
- Chakraborty S, Andrade FCD, Ghosh S, Uelmen J, Ruiz MO. Historical Expansion of Kyasanur Forest Disease in India From 1957 to 2017: A Retrospective Analysis. Geohealth. 2019;3(2):44-55. Published 2019 Feb 5. [CrossRef]
- N, Srilekha et al. “Kyasanur Forest Disease: A Comprehensive Review.” Cureus vol. 16,7 e65228. 23 Jul. 2024. [CrossRef]
- Amogh Verma, Ayush Anand, Ajeet Singh, Abhinav Khare, Ahmad Neyazi, Sarvesh Rustagi, Neelima Kukreti, Abhay M Gaidhane, Quazi Syed Zahiruddin, Prakasini Satapathy, Kyasanur Forest Disease: Clinical manifestations and molecular dynamics in a zoonotic landscape, Clinical Infection in Practice, Volume 21, 2024, 100352, ISSN 2590-1702, .
- Ajesh K, Nagaraja BK, Sreejith K. Kyasanur forest disease virus breaking the endemic barrier: An investigation into ecological effects on disease emergence and future outlook. Zoonoses Public Health. 2017;64(7):e73-e80. [CrossRef]
- Purse BV, et al. Climate change and vector-borne diseases: A review of the evidence. Philos Trans R Soc Lond B Biol Sci 2020; 375:20180362.
- Pattnaik S, Agrawal R, Murmu J, Kanungo S, Pati S. Does the rise in cases of Kyasanur forest disease call for the implementation of One Health in India?. IJID Reg. 2023;7:18-21. Published 2023 Feb 12. [CrossRef]
- Dandawate CN, Desai GB, Achar TR, Banerjee K. Field evaluation of formalin inactivated Kyasanur forest disease virus tissue culture vaccine in three districts of Karnataka state. Indian J Med Res. 1994;99:152-158.
- Kasabi GS, Murhekar MV, Sandhya VK, et al. Coverage and effectiveness of Kyasanur forest disease (KFD) vaccine in Karnataka, South India, 2005-10. PLoS Negl Trop Dis. 2013;7(1):e2025. [CrossRef]
- Bhatia B, Tang-Huau TL, Feldmann F, et al. Single-dose VSV-based vaccine protects against Kyasanur Forest disease in nonhuman primates. Sci Adv. 2023;9(36):eadj1428. [CrossRef]
- Bhatia B, Meade-White K, Haddock E, Feldmann F, Marzi A, Feldmann H. A live-attenuated viral vector vaccine protects mice against lethal challenge with Kyasanur Forest disease virus. NPJ Vaccines. 2021;6(1):152. Published 2021 Dec 14. [CrossRef]
- Das M, Kumar V, Madhukalya R, et al. Purification and characterization of kyasanur forest disease virus EDIII domain of major envelope glycoprotein. J Virol Methods. 2025;333:115089. [CrossRef]
- Arumugam S, Varamballi P. In-silico design of envelope based multi-epitope vaccine candidate against Kyasanur forest disease virus. Sci Rep. 2021;11(1):17118. Published 2021 Aug 24. [CrossRef]
- Kasibhatla SM, Rajan L, Shete A, et al. Construction of an immunoinformatics-based multi-epitope vaccine candidate targeting Kyasanur forest disease virus. PeerJ. 2025;13:e18982. Published 2025 Mar 21. [CrossRef]
- Pillai, S. B., Jeyachandran, A., Pariyapurath, N. K., Jagadibabu, S., Rajaiah, P., Channappa, S. K., Pachamuthu, R. G., Premkumar, A. A., & Jagannathan, S. (2025). Advancements in Vaccine Development : A comprehensive design of a Multi-Epitopic Immunodominant Peptide Vaccine Targeting Kyasanur Forest Disease via Reverse Vaccinology. bioRxiv (Cold Spring Harbor Laboratory). [CrossRef]
- Lo MK, Shi PY, Chen YL, Flint M, Spiropoulou CF. In vitro antiviral activity of adenosine analog NITD008 against tick-borne flaviviruses. Antiviral Res. 2016;130:46-49. [CrossRef]
- Yadav P, Sharma S, Dash PK, Dhankher S, V K S, Kiran SK. Dry- down probe free qPCR for detection of KFD in resource limited settings. PLoS One. 2023;18(5):e0284559. Published 2023 May 10. [CrossRef]
- Hasan S, et al. Advances in multiplex diagnostics for arboviruses. Emerg Infect Dis 2025; 31:in press.
- Pramanik P, et al. Climate-driven changes in tick-borne disease risk. J Med Entomol 2021; 58:1234–1242.
- Pattnaik P. Kyasanur forest disease: an epidemiological view in India [published correction appears in Rev Med Virol. 2008 May-Jun;18(3):211]. Rev Med Virol. 2006;16(3):151-165. [CrossRef]
- Munivenkatappa, Ashok et al. “Clinical & epidemiological significance of Kyasanur forest disease.” The Indian journal of medical research vol. 148,2 (2018): 145-150. [CrossRef]
- Rajaiah P. Kyasanur Forest Disease in India: innovative options for intervention. Hum Vaccin Immunother. 2019;15(10):2243-2248. [CrossRef]
- Balasubramanian R, Yadav PD, Sahina S, Nadh VA. The species distribution of ticks & the prevalence of Kyasanur forest disease virus in questing nymphal ticks from Western Ghats of Kerala, South India. Indian J Med Res. 2021;154(5):743-749. [CrossRef]
- Vedachalam SK, Rajput BL, Choudhary S, et al. Kyasanur Forest Disease: An Epidemiological Investigation and Case-Control Study in Shivamogga, Karnataka, India-2022. Int J Public Health. 2024;69:1606715. Published 2024 Oct 18. [CrossRef]
- Vedachalam, S.K., Rajput, B.L., Choudhary, S. et al. Descriptive epidemiology of Kyasanur forest disease in Thirthahalli taluk, Shivamogga, Karnataka, 2018–2022. Discov Public Health 22, 130 (2025). [CrossRef]
- Chakraborty S, Sander W, Allan BF, Andrade FCD. Sociodemographic factors associated with Kyasanur forest disease in India - a retrospective study. IJID Reg. 2024;10:219-227. Published 2024 Feb 15. [CrossRef]
- Bhat HR, Naik SV. Transmission of Kyasanur forest disease virus by Haemaphysalis wellingtoni Nuttall and Warburton, 1907 (Acarina : Ixodidae). Indian J Med Res. 1978;67(5):697-703.
- Zhang C, Li Y, Samad A, et al. Kyasanur Forest disease virus NS3 helicase: Insights into structure, activity, and inhibitors. Int J Biol Macromol. 2024;254(Pt 3):127856. [CrossRef]
- Cook BW, Cutts TA, Court DA, Theriault S. The generation of a reverse genetics system for Kyasanur Forest Disease Virus and the ability to antagonize the induction of the antiviral state in vitro. Virus Res. 2012;163(2):431-438. [CrossRef]
- Cook BW, Ranadheera C, Nikiforuk AM, et al. Limited Effects of Type I Interferons on Kyasanur Forest Disease Virus in Cell Culture. PLoS Negl Trop Dis. 2016;10(8):e0004871. Published 2016 Aug 1. [CrossRef]
- Sirmarova J, Salat J, Palus M, et al. Kyasanur Forest disease virus infection activates human vascular endothelial cells and monocyte-derived dendritic cells. Emerg Microbes Infect. 2018;7(1):175. Published 2018 Nov 7. [CrossRef]
- Sawatsky B, McAuley AJ, Holbrook MR, Bente DA. Comparative pathogenesis of Alkhumra hemorrhagic fever and Kyasanur forest disease viruses in a mouse model. PLoS Negl Trop Dis. 2014;8(6):e2934. Published 2014 Jun 12. [CrossRef]
- Patil, D.R., Yadav, P.D., Shete, A. et al. Study of Kyasanur forest disease viremia, antibody kinetics, and virus infection in target organs of Macaca radiata. Sci Rep 10, 12561 (2020). [CrossRef]
- Robertson SJ, Mitzel DN, Taylor RT, Best SM, Bloom ME. Tick-borne flaviviruses: dissecting host immune responses and virus countermeasures. Immunol Res. 2009;43(1-3):172-186. [CrossRef]
- Avirutnan P, Hauhart RE, Somnuke P, Blom AM, Diamond MS, Atkinson JP. Binding of flavivirus nonstructural protein NS1 to C4b binding protein modulates complement activation. J Immunol. 2011;187(1):424-433. [CrossRef]
- Chen Y, Maguire T, Hileman RE, et al. Dengue virus infectivity depends on envelope protein binding to target cell heparan sulfate. Nat Med. 1997;3(8):866-871. [CrossRef]
- Kroschewski H, Allison SL, Heinz FX, Mandl CW. Role of heparan sulfate for attachment and entry of tick-borne encephalitis virus. Virology. 2003;308(1):92-100. [CrossRef]
- Yin Z., Chen Y.-L., Schul W., Wang Q.-Y., Gu F., Duraiswamy J., Kondreddi R.R., Niyomrattanakit P., Lakshminarayana S.B., Goh A. An adenosine nucleoside inhibitor of dengue virus. Proc. Natl. Acad. Sci. USA. 2009;106:20435–20439. [CrossRef]
- Bixler SL, Bocan TM, Wells J, et al. Efficacy of favipiravir (T-705) in nonhuman primates infected with Ebola virus or Marburg virus. Antiviral Res. 2018;151:97-104. [CrossRef]
- Keating G. M. (2014). Sofosbuvir: a review of its use in patients with chronic hepatitis C. Drugs 74, 1127–1146. [CrossRef]
- Marlin, R., Desjardins, D., Contreras, V. et al. Antiviral efficacy of favipiravir against Zika and SARS-CoV-2 viruses in non-human primates. Nat Commun 13, 5108 (2022). [CrossRef]
- Morrey J, Taro B, Siddharthan V, et al. Efficacy of orally administered T-705 pyrazine analog on lethal West Nile virus infection in rodents. Antiviral Res 2008; 80: 377–379.
- Julander JG, Shafer K, Smee DF, et al. Activity of T-705 in a hamster model of yellow fever virus infection in comparison with that of a chemically related compound, T-1106. Antimicrob Agents Chemother 2009; 53: 202–209.
- Ferreira ACReis PA, de Freitas CSSacramento CQVillas Bôas Hoelz L, Bastos MM, Mattos MRocha NGomes de Azevedo Quintanilha I, da Silva Gouveia Pedrosa C, Rocha Quintino Souza L, Correia Loiola E, Trindade P, Rangel Vieira YBarbosa-Lima G, de Castro Faria Neto HC, Boechat N, Rehen SKBrüning K, Bozza FABozza PT, Souza TML 2019. Beyond Members of the Flaviviridae Family, Sofosbuvir Also Inhibits Chikungunya Virus Replication. Antimicrob Agents Chemother 63:10.1128/aac.01389-18.
- Sacramento, C., de Melo, G., de Freitas, C. et al. The clinically approved antiviral drug sofosbuvir inhibits Zika virus replication. Sci Rep 7, 40920 (2017). [CrossRef]
- Kao JC, HuangFu WC, Tsai TT, et al. The antiparasitic drug niclosamide inhibits dengue virus infection by interfering with endosomal acidification independent of mTOR. PLoS Negl Trop Dis. 2018;12(8):e0006715. Published 2018 Aug 20. [CrossRef]
- Gassen NC, Papies J, Bajaj T, et al. SARS-CoV-2-mediated dysregulation of metabolism and autophagy uncovers host-targeting antivirals. Nat Commun. 2021;12(1):3818. Published 2021 Jun 21. [CrossRef]
- Li, Z., Brecher, M., Deng, YQ. et al. Existing drugs as broad-spectrum and potent inhibitors for Zika virus by targeting NS2B-NS3 interaction. Cell Res 27, 1046–1064 (2017). [CrossRef]
- Gunale B, Farinola N, Kamat CD, et al. An observer-blind, randomised, placebo-controlled, phase 1, single ascending dose study of dengue monoclonal antibody in healthy adults in Australia. Lancet Infect Dis. 2024;24(6):639-649. [CrossRef]
- Deng YQ, Zhang NN, Li CF, et al. Adenosine Analog NITD008 Is a Potent Inhibitor of Zika Virus. Open Forum Infect Dis. 2016;3(4):ofw175. Published 2016 Aug 30. [CrossRef]
- Guedj J, Piorkowski G, Jacquot F, et al. Antiviral efficacy of favipiravir against Ebola virus: A translational study in cynomolgus macaques. PLoS Med. 2018;15(3):e1002535. Published 2018 Mar 27. [CrossRef]
- Franco EJ, Pires de Mello CP, Brown AN. Antiviral Evaluation of UV-4B and Interferon-Alpha Combination Regimens against Dengue Virus. Viruses. 2021;13(5):771. Published 2021 Apr 27. [CrossRef]
- Xu HT, Colby-Germinario SP, Hassounah SA, et al. Evaluation of Sofosbuvir (β-D-2'-deoxy-2'-α-fluoro-2'-β-C-methyluridine) as an inhibitor of Dengue virus replication<sup/>. Sci Rep. 2017;7(1):6345. Published 2017 Jul 24. [CrossRef]
- Weiss A, Touret F, Baronti C, et al. Niclosamide shows strong antiviral activity in a human airway model of SARS-CoV-2 infection and a conserved potency against the Alpha (B.1.1.7), Beta (B.1.351) and Delta variant (B.1.617.2). PLoS One. 2021;16(12):e0260958. Published 2021 Dec 2. [CrossRef]
- Yeo KL, Chen YL, Xu HY, et al. Synergistic suppression of dengue virus replication using a combination of nucleoside.
- Tien SM, Chang PC, Lai YC, et al. Therapeutic efficacy of humanized monoclonal antibodies targeting dengue virus nonstructural protein 1 in the mouse model. PLoS Pathog. 2022;18(4):e1010469. Published 2022 Apr 29. [CrossRef]
- Devadiga S, McElroy AK, Prabhu SG, Arunkumar G. Dynamics of human B and T cell adaptive immune responses to Kyasanur Forest disease virus infection. Sci Rep. 2020;10(1):15306. Published 2020 Sep 17. [CrossRef]
- Chakraborty S, Sander WE, Allan BF, Andrade FCD. Retrospective Study of Kyasanur Forest Disease and Deaths among Nonhuman Primates, India, 1957-2020. Emerg Infect Dis. 2021;27(7):1969-1973. [CrossRef]
- Gote V, Bolla PK, Kommineni N, et al. A Comprehensive Review of mRNA Vaccines. Int J Mol Sci. 2023;24(3):2700. Published 2023 Jan 31. [CrossRef]
- Chaudhary, N., Weissman, D. & Whitehead, K.A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov 20, 817–838 (2021). [CrossRef]
- Pardi, N., Hogan, M., Porter, F. et al. mRNA vaccines — a new era in vaccinology. Nat Rev Drug Discov 17, 261–279 (2018). [CrossRef]
- Barrett ADT. Yellow fever live attenuated vaccine: A very successful live attenuated vaccine but still we have problems controlling the disease. Vaccine. 2017;35(44):5951-5955. [CrossRef]
- Kallás EG, Cintra MAT, Moreira JA, et al. Live, Attenuated, Tetravalent Butantan-Dengue Vaccine in Children and Adults. N Engl J Med. 2024;390(5):397-408. [CrossRef]
- N S, Hewson R, Afrough B, Bewley K, Arunkumar G. Development of a quantitative real-time RT-PCR assay that differentiates between Kyasanur Forest disease virus and Alkhurma hemorrhagic fever virus. Ticks Tick Borne Dis. 2020;11(3):101381. [CrossRef]
- Rajak A, Kumar JS, Dhankher S, et al. Development and application of a recombinant Envelope Domain III protein based indirect human IgM ELISA for Kyasanur forest disease virus. Acta Trop. 2022;235:106623. [CrossRef]
- Sharma S, Yadav P, Dash PK, Dhankher S. Molecular epidemiology of Kyasanur forest disease employing ONT-NGS a field forward sequencing. J Clin Virol. 2025;177:105783. [CrossRef]
- Majumdar T, Shete A, Yadav P, et al. Point of care real-time polymerase chain reaction-based diagnostic for Kyasanur forest disease. Int J Infect Dis. 2021;108:226-230. [CrossRef]
- Suder E, Furuyama W, Feldmann H, Marzi A, de Wit E. The vesicular stomatitis virus-based Ebola virus vaccine: From concept to clinical trials. Hum Vaccin Immunother. 2018;14(9):2107-2113. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




